Anaerobic Co-Digestion of Bioplastics and Food Waste under Mesophilic and Thermophilic Conditions: Synergistic Effect and Biodegradation
Abstract
1. Introduction
2. Materials and Methods
2.1. Substrate and Inoculum Preparation
2.2. Biochemical Methane Potential (BMP) Assay
2.3. Liquid and Solid Characteristics
2.4. Calculations
3. Results and Discussion
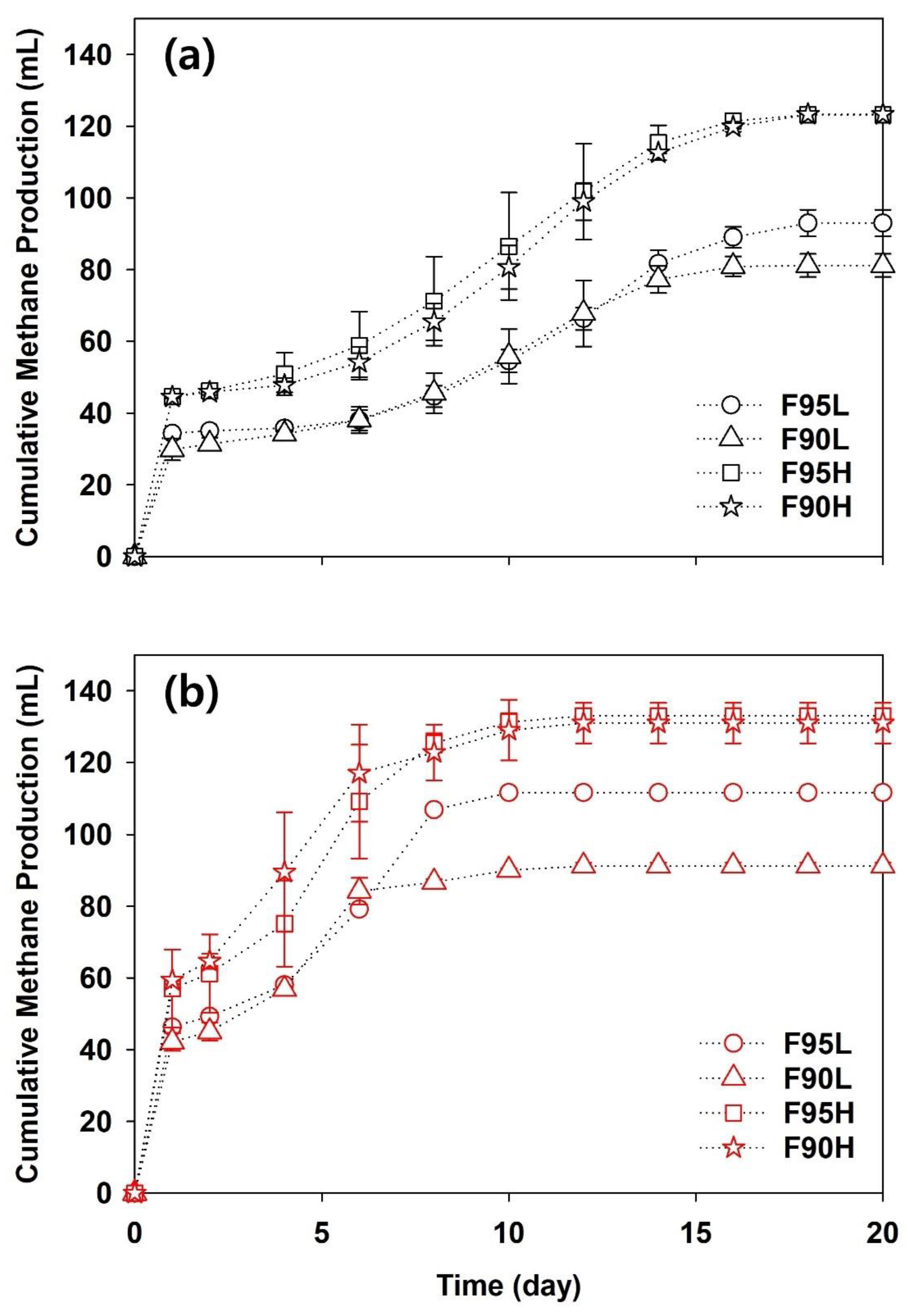
3.1. Mono-Digestion of Food Waste, PLA, and PHA
3.2. Co-Digestion and Its Synergistic Effect
3.3. Biodegradation of Bioplastics
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cucina, M.; De Nisi, P.; Trombino, L.; Tambone, F.; Adani, F. Degradation of bioplastics in organic waste by mesophilic anaerobic digestion, composting and soil incubation. Waste Manag. 2021, 134, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Statista. Available online: https://www.statista.com/statistics/282732/global-production-of-plastics-since-1950/ (accessed on 10 November 2022).
- Bellasi, A.; Binda, G.; Pozzi, A.; Galafassi, S.; Volta, P.; Bettinetti, R. Microplastic contamination in freshwater environments: A review, focusing on interactions with sediments and benthic organisms. Environments 2020, 7, 30. [Google Scholar] [CrossRef]
- Unmar, G.; Mohee, R. Assessing the effect of biodegradable and degradable plastics on the composting of green wastes and compost quality. Bioresour. Technol. 2008, 99, 6738–6744. [Google Scholar] [CrossRef]
- Abraham, A.; Park, H.; Choi, O.; Sang, B.I. Anaerobic co-digestion of bioplastics as a sustainable mode of waste management with improved energy production—A review. Bioresour. Technol. 2021, 322, 124537. [Google Scholar] [CrossRef]
- Kakadellis, S.; Lee, P.H.; Harris, Z.M. Two Birds with One Stone: Bioplastics and Food Waste Anaerobic Co-Digestion. Environments 2022, 9, 9. [Google Scholar] [CrossRef]
- Brodhagen, M.; Goldberger, J.R.; Hayes, D.G.; Inglis, D.A.; Marsh, T.L.; Miles, C. Policy considerations for limiting unintended residual plastic in agricultural soils. Environ. Sci. Policy 2017, 69, 81–84. [Google Scholar] [CrossRef]
- Narodoslawsky, M.; Shazad, K.; Kollmann, R.; Schnitzer, H. LCA of PHA production–Identifying the ecological potential of bio-plastic. Chem. Biochem. Eng. Q. 2015, 29, 299–305. [Google Scholar] [CrossRef]
- Siracusa, V.; Rocculi, P.; Romani, S.; Dalla Rosa, M. Biodegradable polymers for food packaging: A review. Trends Food Sci. Technol. 2008, 19, 634–643. [Google Scholar] [CrossRef]
- Sudesh, K.; Abe, H.; Doi, Y. Synthesis, structure and properties of polyhydroxyalkanoates: Biological polyesters. Prog. Polym. Sci. 2000, 25, 1503–1555. [Google Scholar] [CrossRef]
- Yagi, H.; Ninomiya, F.; Funabashi, M.; Kunioka, M. Mesophilic anaerobic biodegradation test and analysis of eubacteria and archaea involved in anaerobic biodegradation of four specified biodegradable polyesters. Polym. Degrad. Stab. 2014, 110, 278–283. [Google Scholar] [CrossRef]
- Meeks, D.; Hottle, T.; Bilec, M.M.; Landis, A.E. Compostable biopolymer use in the real world: Stakeholder interviews to better understand the motivations and realities of use and disposal in the US. Resour. Conserv. Recycl. 2015, 105, 134–142. [Google Scholar] [CrossRef]
- Solano, G.; Rojas-Gätjens, D.; Rojas-Jimenez, K.; Chavarría, M.; Romero, R.M. Biodegradation of plastics at home composting conditions. Environ. Chall. 2022, 7, 100500. [Google Scholar] [CrossRef]
- Benn, N.; Zitomer, D. Pretreatment and anaerobic co-digestion of selected PHB and PLA bioplastics. Front. Environ. Sci. 2018, 5, 93. [Google Scholar] [CrossRef]
- Stieb, M.; Schink, B. A new 3-hydroxybutyrate fermenting anaerobe, Ilyobacter polytropus, gen. nov. sp. nov., possessing various fermentation pathways. Arch. Microbiol. 1984, 140, 139–146. [Google Scholar] [CrossRef]
- Yoshie, N.; Oike, Y.; Kasuya, K.I.; Doi, Y.; Inoue, Y. Change of surface structure of poly (3-hydroxybutyrate) film upon enzymatic hydrolysis by PHB depolymerase. Biomacromolecules 2002, 3, 1320–1326. [Google Scholar] [CrossRef] [PubMed]
- Shang, L.; Fei, Q.; Zhang, Y.H.; Wang, X.Z.; Fan, D.D.; Chang, H.N. Thermal properties and biodegradability studies of poly (3-hydroxybutyrate-co-3-hydroxyvalerate). J. Polym. Environ. 2012, 20, 23–28. [Google Scholar] [CrossRef]
- Venkiteshwaran, K.; Bocher, B.; Maki, J.; Zitomer, D. Relating anaerobic digestion microbial community and process function: Supplementary issue: Water microbiology. Microbiol. Insights 2015, 8, 37–44. [Google Scholar] [CrossRef]
- Hobbs, S.R.; Parameswaran, P.; Astmann, B.; Devkota, J.P.; Landis, A.E. Anaerobic codigestion of food waste and polylactic acid: Effect of pretreatment on methane yield and solid reduction. Adv. Mater. Sci. Eng. 2019, 2019, 6. [Google Scholar] [CrossRef]
- Cazaudehore, G.; Guyoneaud, R.; Vasmara, C.; Greuet, P.; Gastaldi, E.; Marchetti, R.; Leonardi, F.; Turon, R.; Monlau, F. Impact of mechanical and thermo-chemical pretreatments to enhance anaerobic digestion of poly (lactic acid). Chemosphere 2022, 297, 133986. [Google Scholar] [CrossRef]
- García-Depraect, O.; Lebrero, R.; Rodriguez-Vega, S.; Bordel, S.; Santos-Beneit, F.; Martínez-Mendoza, L.J.; Börner, R.A.; Börner, T.; Munoz, R. Biodegradation of bioplastics under aerobic and anaerobic aqueous conditions: Kinetics, carbon fate and particle size effect. Bioresour. Technol. 2022, 344, 126265. [Google Scholar] [CrossRef]
- Stroot, P.G.; McMahon, K.D.; Mackie, R.I.; Raskin, L. Anaerobic codigestion of municipal solid waste and biosolids under various mixing conditions—I. Digester performance. Water Res. 2001, 35, 1804–1816. [Google Scholar] [CrossRef]
- Cho, H.S.; Moon, H.S.; Kim, M.; Nam, K.; Kim, J.Y. Biodegradability and biodegradation rate of poly (caprolactone)-starch blend and poly (butylene succinate) biodegradable polymer under aerobic and anaerobic environment. Waste Manag. 2011, 31, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Yagi, H.; Ninomiya, F.; Funabashi, M.; Kunioka, M. Anaerobic biodegradation tests of poly (lactic acid) under mesophilic and thermophilic conditions using a new evaluation system for methane fermentation in anaerobic sludge. Int. J. mol. Sci. 2009, 10, 3824–3835. [Google Scholar] [CrossRef]
- Zhang, W.; Heaven, S.; Banks, C.J. Degradation of some EN13432 compliant plastics in simulated mesophilic anaerobic digestion of food waste. Polym. Degrad. Stab. 2018, 147, 76–88. [Google Scholar] [CrossRef]
- Cruz, F.B.D.L.; Barlaz, M.A. Estimation of waste component-specific landfill decay rates using laboratory-scale decomposition data. Environ. Sci. Technol. 2010, 44, 4722–4728. [Google Scholar] [CrossRef] [PubMed]
- Garaffa, C.; Yepsen, R. Managing compostable bags at anaerobic digestion plants. BioCycle 2012, 53, 37–40. [Google Scholar]
- Appels, L.; Baeyens, J.; Degrève, J.; Dewil, R. Principles and potential of the anaerobic digestion of waste-activated sludge. Prog. Energy Combus. Sci. 2008, 34, 755–781. [Google Scholar] [CrossRef]
- Bernat, K.; Kulikowska, D.; Wojnowska-Baryła, I.; Zaborowska, M.; Pasieczna-Patkowska, S. Thermophilic and mesophilic biogas production from PLA-based materials: Possibilities and limitations. Waste Manag. 2021, 119, 295–305. [Google Scholar] [CrossRef]
- Im, S.; Yun, Y.M.; Song, Y.C.; Kim, D.H. Enhanced anaerobic digestion of glycerol by promoting DIET reaction. Biochem. Eng. J. 2019, 142, 18–26. [Google Scholar] [CrossRef]
- American Public Health Association—APHA, AWWA, WEF. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; APHA: Washington, DC, USA, 2012. [Google Scholar]
- Xiaohui, L.; Changmin, L.; Jae, Y.K. Thermal hydrolysis pre-treatment combined with anaerobic digestion for energy recovery from organic wastes. J. Mater. Cycles Waste Manag. 2020, 22, 1370–1381. [Google Scholar] [CrossRef]
- Rehm, H.J.; Reed, G.; Klein, J. Biotechnology: Environmental Processes II. Soil Decontamination; Wiley-VCH: Weinheim, Germany, 2000; Volume 11b. [Google Scholar]
- Shin, S.R.; Lee, M.K.; Im, S.; Kim, D.H. Effect of seaweed addition on enhanced anaerobic digestion of food waste and sewage sludge. Environ. Eng. Res. 2019, 24, 449–455. [Google Scholar] [CrossRef]
- Mata-Alvarez, J.; Dosta, J.; Romero-Güiza, M.S.; Fonoll, X.; Peces, M.; Astals, S. A critical review on anaerobic co-digestion achievements between 2010 and 2013. Renew. Sustain. Energy Rev. 2014, 36, 412–427. [Google Scholar] [CrossRef]
- Hegde, S.; Diaz, C.A.; Dell, E.M.; Trabold, T.A.; Lewis, C.L. Investigation of process parameters on the anaerobic digestion of a poly(hydroxyalkonate) film. Eur. Polym. J. 2021, 148, 110349. [Google Scholar] [CrossRef]
- Massardier-Nageotte, V.; Pestre, C.; Cruard-Pradet, T.; Bayard, R. Aerobic and anaerobic biodegradability of polymer films and physico-chemical characterization. Polym. Degrad. Stab. 2006, 91, 620–627. [Google Scholar] [CrossRef]
- Ryan, C.A.; Billington, S.L.; Criddle, C.S. Assessment of models for anaerobic biodegradation of a model bioplastic: Poly (hydroxybutyrate-co-hydroxyvalerate). Bioresour. Technol. 2017, 227, 205–213. [Google Scholar] [CrossRef] [PubMed]


| Units | TS a | VS | COD | |
|---|---|---|---|---|
| Food waste | g/kg | 85.7 | 81.9 | 114.5 |
| PLA | kg/kg | 0.99 | 0.98 | 1.3 |
| PHA | kg/kg | 0.99 | 0.98 | 1.5 |
| Mixing Condition | P a (mL) | Rm b (mL/d) | λ c (d) | r2 | |
|---|---|---|---|---|---|
| Mesophilic AD (37 °C) | Food waste d | 126.0 | 6.9 | 6.59 | 0.99 |
| PLA | 5.8 | 1.2 | 3.59 | 0.99 | |
| PHA | 76.9 | 9.5 | 2.42 | 0.99 | |
| F95L d | 89.4 | 5.2 | 6.11 | 0.99 | |
| F90L d | 78.6 | 4.1 | 3.46 | 0.99 | |
| F95H d | 123.1 | 6.4 | 1.34 | 0.99 | |
| F90H d | 123.2 | 6.4 | 2.02 | 0.99 | |
| Thermophilic AD (55 °C) | Food waste | 141.0 | 17.2 | <0.1 | 0.97 |
| PLA | 12.8 | 2.4 | 2.26 | 0.99 | |
| PHA | 86.0 | 10.4 | 0.42 | 0.99 | |
| F95L | 114.1 | 13.6 | <0.1 | 0.97 | |
| F90L | 91.4 | 15.2 | <0.1 | 0.97 | |
| F95H | 134.4 | 18.4 | <0.1 | 0.97 | |
| F90H | 130.4 | 23.0 | <0.1 | 0.98 | |
| Experimental Conditions | Theoretical CH4 Production Yield (mL/g COD) | Actual CH4 Production Yield from Co-Digestion (mL/g COD) | Synergistic Effect (%) | |
|---|---|---|---|---|
| Mesophilic AD (37 °C) | Food waste | - | 252.0 | - |
| PLA | - | 11.6 | - | |
| PHA | - | 153.8 | - | |
| F95L | 164.9 | 178.9 | 8.5 | |
| F90L | 124.2 | 157.2 | 26.6 | |
| F95H | 213.1 | 246.2 | 15.5 | |
| F90H | 196.3 | 246.4 | 25.5 | |
| Thermophilic AD (55 °C) | Food waste | - | 281.9 | - |
| PLA | - | 25.6 | - | |
| PHA | - | 172.0 | - | |
| F95L | 189.1 | 228.3 | 20.7 | |
| F90L | 145.6 | 182.7 | 25.5 | |
| F95H | 238.4 | 268.7 | 12.7 | |
| F90H | 219.6 | 260.7 | 18.7 | |
| Inoculum (g VS/L) | Food Waste (g VS/L) | PHA (g VS/L) | PLA (g VS/L) | Total (g VS/L) | |
|---|---|---|---|---|---|
| Blank | 4.00 | - | - | - | 4.00 |
| Food waste | 4.00 | 1.43 | - | - | 5.43 |
| PLA | 4.00 | - | - | 1.51 | 5.51 |
| PHA | 4.00 | - | 1.31 | - | 5.31 |
| FW95L | 4.00 | 0.90 | - | 0.54 | 5.44 |
| FW90L | 4.00 | 0.63 | - | 0.76 | 5.39 |
| FW95H | 4.00 | 0.85 | 0.51 | - | 5.36 |
| FW90H | 4.00 | 0.58 | 0.70 | - | 5.28 |
| Experimental Conditions | Inoculum (g VS/L) | Food Waste (g VS/L) | PLA d (g VS/L) | PHA d (g VS/L) | Total a (g VS/L) | |
|---|---|---|---|---|---|---|
| Mesophilic AD (37 °C) | Blank | 3.10 | - | - | - | 3.10 |
| Food waste | 3.10 | 0.30 b | - | - | 3.41 | |
| PLA | 3.10 | - | 1.42 | - | 4.52 | |
| PHA | 3.10 | - | - | 0.67 | 3.77 | |
| F95L | 3.10 | 0.19 c | 0.41 | - | 3.71 | |
| F90L | 3.10 | 0.13 c | 0.52 | - | 3.75 | |
| F95H | 3.10 | 0.18 c | - | 0.20 | 3.48 | |
| F90H | 3.10 | 0.12 c | - | 0.26 | 3.48 | |
| Thermophilic AD (55 °C) | Blank | 2.93 | - | - | - | 2.93 |
| Food waste | 2.93 | 0.17 b | - | - | 3.10 | |
| PLA | 2.93 | - | 1.30 | - | 4.23 | |
| PHA | 2.93 | - | - | 0.62 | 3.55 | |
| F95L | 2.93 | 0.11 c | 0.30 | - | 3.33 | |
| F90L | 2.93 | 0.07 c | 0.46 | - | 3.46 | |
| F95H | 2.93 | 0.10 c | - | 0.10 | 3.13 | |
| F90H | 2.93 | 0.07 c | - | 0.16 | 3.15 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, J.-H.; Kang, S.-W.; Kim, W.-J.; Kim, D.-H.; Im, S.-W. Anaerobic Co-Digestion of Bioplastics and Food Waste under Mesophilic and Thermophilic Conditions: Synergistic Effect and Biodegradation. Fermentation 2022, 8, 638. https://doi.org/10.3390/fermentation8110638
Kang J-H, Kang S-W, Kim W-J, Kim D-H, Im S-W. Anaerobic Co-Digestion of Bioplastics and Food Waste under Mesophilic and Thermophilic Conditions: Synergistic Effect and Biodegradation. Fermentation. 2022; 8(11):638. https://doi.org/10.3390/fermentation8110638
Chicago/Turabian StyleKang, Jeong-Hee, Sung-Won Kang, Weon-Jae Kim, Dong-Hoon Kim, and Seong-Won Im. 2022. "Anaerobic Co-Digestion of Bioplastics and Food Waste under Mesophilic and Thermophilic Conditions: Synergistic Effect and Biodegradation" Fermentation 8, no. 11: 638. https://doi.org/10.3390/fermentation8110638
APA StyleKang, J.-H., Kang, S.-W., Kim, W.-J., Kim, D.-H., & Im, S.-W. (2022). Anaerobic Co-Digestion of Bioplastics and Food Waste under Mesophilic and Thermophilic Conditions: Synergistic Effect and Biodegradation. Fermentation, 8(11), 638. https://doi.org/10.3390/fermentation8110638








