Lactic Acid for Green Chemical Industry: Recent Advances in and Future Prospects for Production Technology, Recovery, and Applications
Abstract
1. Introduction
2. Lactic Acid Production Technologies
2.1. Chemical Synthesis
2.2. Biological Synthesis
2.2.1. Lactic Acid-Producing Microorganisms
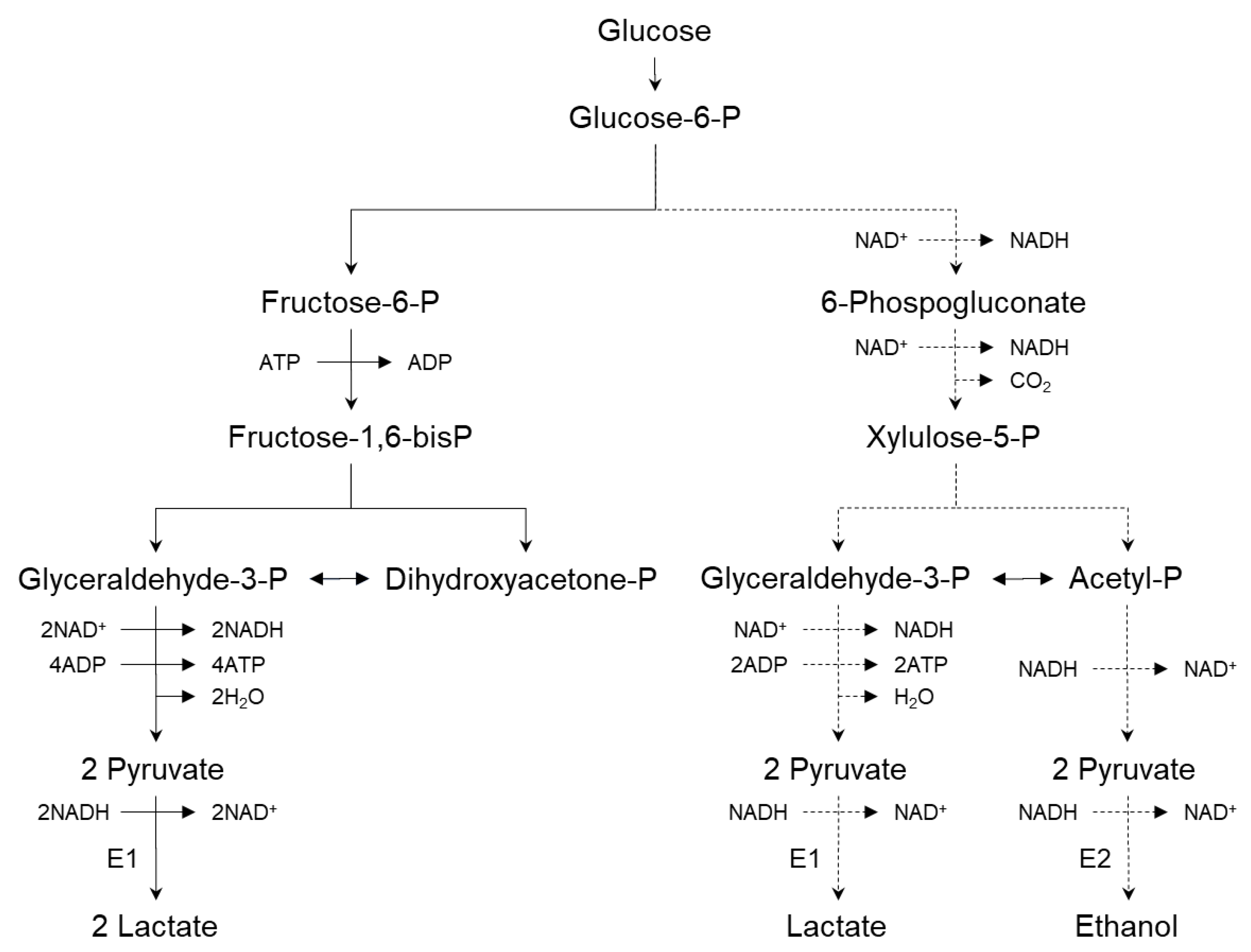
2.2.2. Biochemistry of Lactic Acid Production
2.2.3. Factors Affecting Lactic Acid Production
2.2.4. Substrates for Lactic Acid Production
Glucose
Starch
Lignocellulose
Whey
Food Waste
Glycerol
Algae
2.2.5. Neutralizing Agents Used in Lactic Acid Fermentation
2.2.6. Lactic Acid Fermentation Technologies
2.2.7. Genetic Engineering in Lactic Acid Production
3. Recovery Processes of Lactic Acid
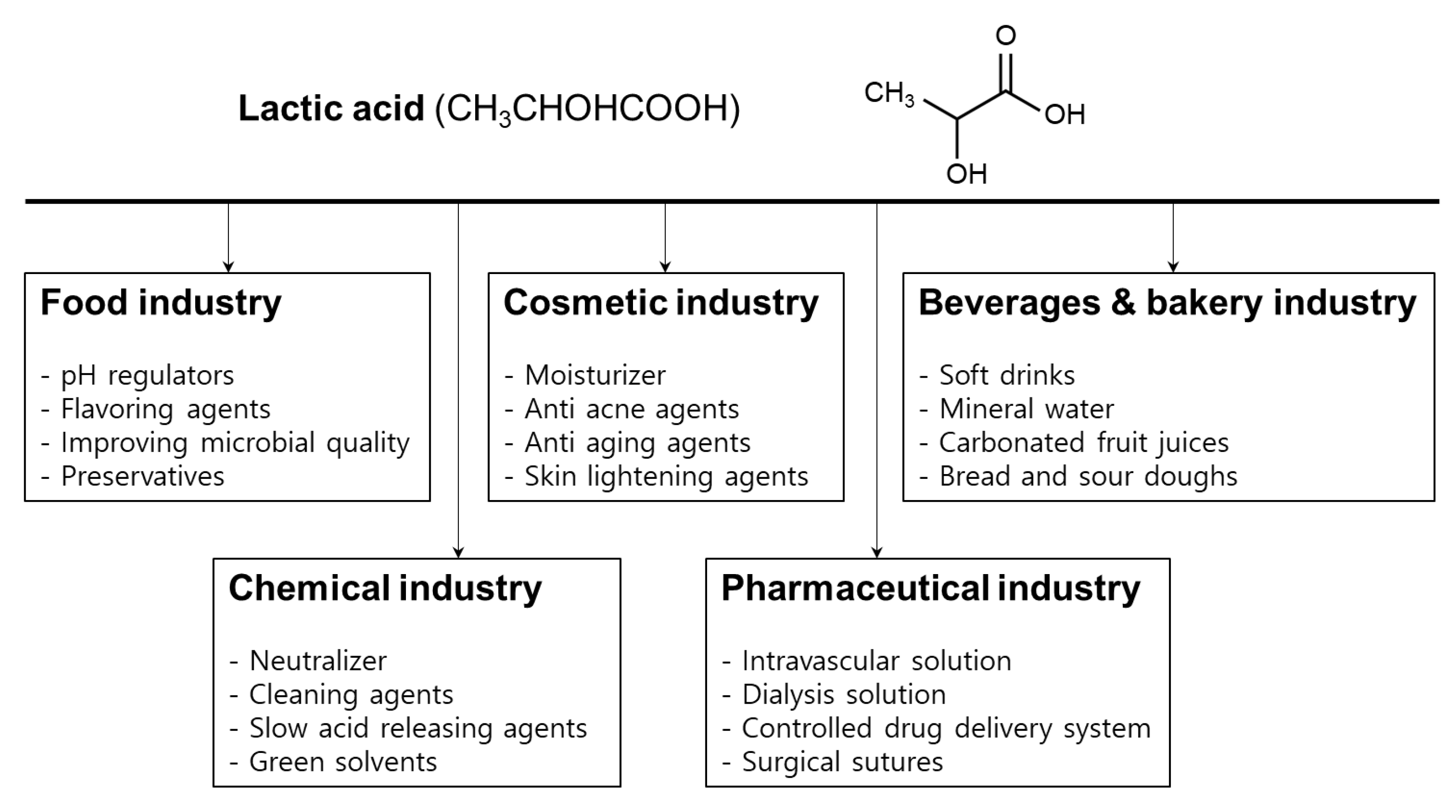
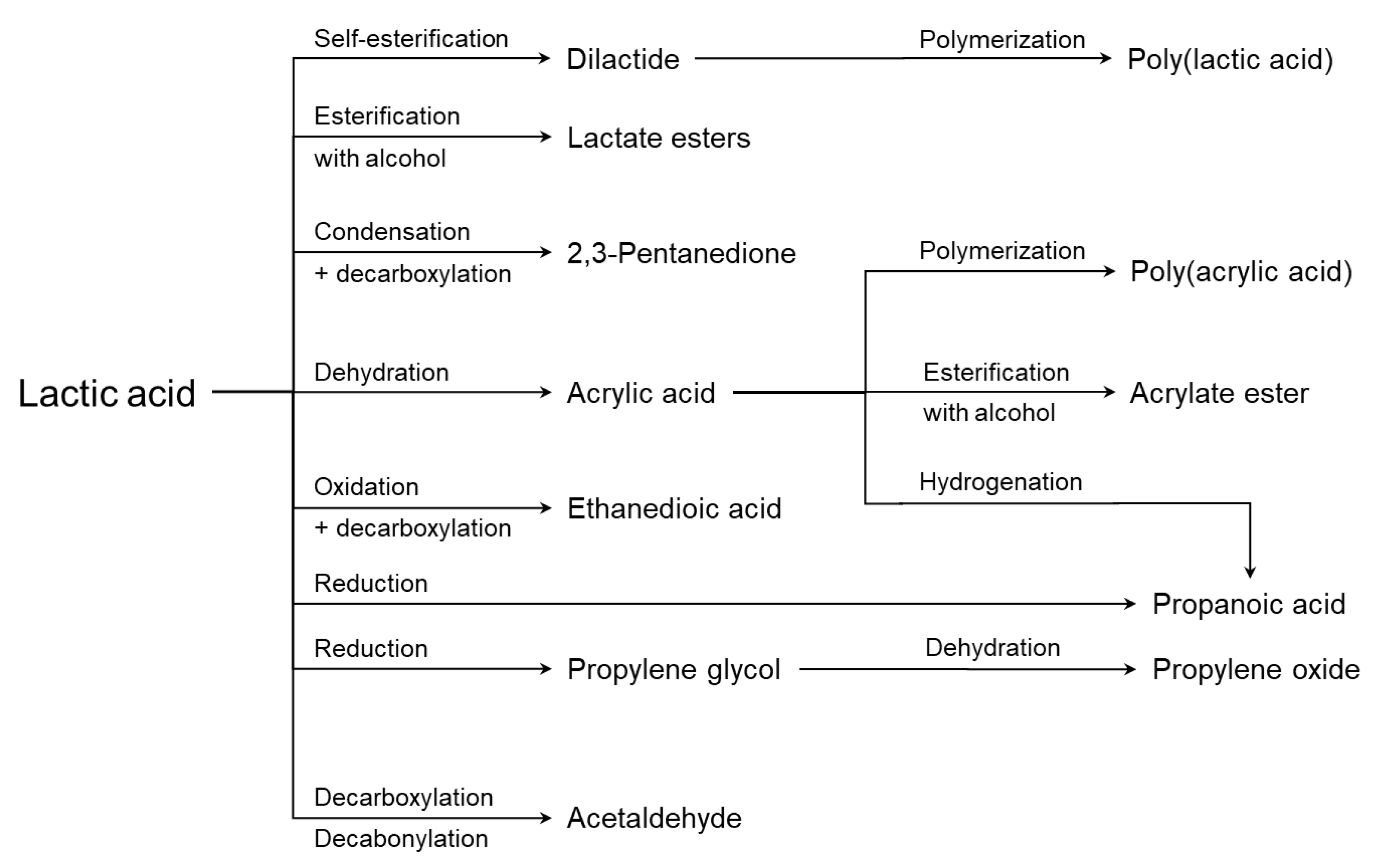
4. Applications of Lactic Acid
5. Future Prospects
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Philp, J.C.; Ritchie, R.J.; Allan, J.E.M. Biobased chemicals: The convergence of green chemistry with industrial biotechnology. Trends Biotechnol. 2013, 31, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Rahman, M.A.; Tashiro, Y.; Sonomoto, K. Recent advances in lactic acid production by microbial fermentation processes. Biotechnol. Adv. 2013, 31, 877–902. [Google Scholar] [CrossRef] [PubMed]
- Precedence Research. Lactic Acid Market–Global Industry Analysis, Size, Share, Growth, Trends, Regional Outlook, and Forecast 2022–2030. Available online: https://www.precedenceresearch.com/lactic-acid-market (accessed on 28 October 2022).
- Budhavaram, N.K.; Fan, Z. Production of lactic acid from paper sludge using acid-tolerant, thermophilic Bacillus coagulans strains. Bioresour. Technol. 2009, 100, 5966–5972. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.M.; Kim, J.S.; Hwang, H.J.; Park, M.S.; Choi, G.J.; Choi, Y.H.; Kim, J.C. Production of l-lactic acid from a green microalga, Hydrodictyon reticulum, by Lactobacillus paracasei LA104 isolated from the traditional Korean food, makgeolli. Bioresour. Technol. 2012, 110, 552–559. [Google Scholar] [CrossRef]
- Watanabe, M.; Makino, M.; Kaku, N.; Koyama, M.; Nakamura, K.; Sasano, K. Fermentative l(+)-lactic acid production from non-sterilized rice washing drainage containing rice bran by a newly isolated lactic acid bacteria without any additions of nutrients. J. Biosci. Bioeng. 2013, 115, 449–452. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Hassan, S.E.; Azab, M.S.; Mahin, A.A.; Gaber, M.A. High improvement in lactic acid productivity by new alkaliphilic bacterium using repeated batch fermentation integrated with increased substrate concentration. BioMed Res. Int. 2019, 2019, 7212870. [Google Scholar] [CrossRef]
- Benninga, H.A. A History of Lactic Acid Making; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1990; pp. 1–21. [Google Scholar]
- John, R.P.; Nampoothiri, K.M. Co-culturing of Lactobacillus paracasei subsp. paracasei with a Lactobacillus delbrueckii subsp. delbrueckii mutant to make high cell density for increased lactate productivity from cassava bagasse hydrolysate. Curr. Microbiol. 2011, 62, 790–794. [Google Scholar] [CrossRef]
- Corma, A.; Iborra, S.; Velty, A. Chemical routes for the transformation of biomass into chemicals. Chem. Rev. 2007, 107, 2411–2502. [Google Scholar] [CrossRef]
- Dodds, D.R.; Gross, R.A. Chemicals from biomass. Science 2007, 318, 1250–1251. [Google Scholar] [CrossRef]
- Holm, M.S.; Saravanamurugan, S.; Taarning, E. Conversion of sugars to lactic acid derivatives using heterogeneous zeotype catalysts. Science 2010, 328, 602–605. [Google Scholar] [CrossRef]
- de Clippel, F.; Dusselier, M.; Rompaey, R.V.; Vanelderen, P.; Dijkmans, J.; Makshina, E.; Giebeler, L.; Oswald, S.; Baron, G.V.; Denayer, J.F.M.; et al. Fast and selective sugar conversion to alkyl lactate and lactic acid with bifunctional carbon-silica catalysts. J. Am. Chem. Soc. 2012, 134, 10089–10101. [Google Scholar] [CrossRef] [PubMed]
- Datta, R.; Henry, M. Lactic acid: Recent advances in products, processes and technologies—A review. J. Chem. Technol. Biotechnol. 2006, 81, 1119–1129. [Google Scholar] [CrossRef]
- Litchfield, J.H. Lactic acid, microbially produced. In Encyclopedia of Microbiology, 3rd ed.; SchaechterMosel, O., Ed.; Academic Press: Oxford, UK, 2009; pp. 362–372. [Google Scholar]
- Lunelli, B.H.; Andrade, R.R.; Atala, D.I.P.; Filho, F.; Filho, M.R. Production of lactic acid from sucrose: Strain selection, fermentation and kinetic modelling. Appl. Biochem. Biotechnol. 2010, 16191, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, G.; Yu, X.; Chen, H.; Sun, Y.; Chen, G. Pretreatment of corn stover by solid acid for d-lactic acid fermentation. Bioresour. Technol. 2017, 239, 490–495. [Google Scholar] [CrossRef]
- Balakrishnan, R.; Tadi, S.R.R.; Rajaram, S.K.; Mohan, N.; Sivaprakasam, S. Batch and fed-batch fermentation of optically pure d(-) lactic acid from Kodo millet (Paspalum scrobiculatum) bran residue hydrolysate: Growth and inhibition kinetic modeling. Prep. Biochem. Biotechnol. 2020, 50, 265–278. [Google Scholar] [CrossRef] [PubMed]
- de la Torre, I.; Acedos, M.G.; Ladero, M.; Santos, V.E. On the use of resting L. delbrueckii spp. delbrueckii cells for d-lactic acid production from orange peel wastes hydrolysates. Biochem. Eng. J. 2019, 145, 162–169. [Google Scholar]
- de la Torre, I.; Ladero, M.; Santos, V.E. d-Lactic acid production from orange waste enzymatic hydrolysates with L. delbrueckii cells in growing and resting state. Ind. Crops Prod. 2020, 146, 112176. [Google Scholar] [CrossRef]
- Liu, P.; Zheng, Z.; Xu, Q.; Qian, Z.; Liu, J.; Ouyang, J. Valorization of dairy waste for enhanced d-lactic acid production at low cost. Process Biochem. 2018, 71, 18–22. [Google Scholar] [CrossRef]
- Kwan, T.H.; Hu, Y.; Lin, C.Z.K. Valorisation of food waste via fungal hydrolysis and lactic acid fermentation with Lactobacillus casei Shirota. Bioresour. Technol. 2016, 217, 129–136. [Google Scholar] [CrossRef]
- Oonkhanonda, B.; Jonglertjunya, W.; Srimaruta, N.; Bunpacharta, P.; Tantinukula, S.; Nasongkla, N.; Sakdaronnarong, C. Lactic acid production from sugarcane bagasse by an integrated system of lignocellulose fractionation, saccharification, fermentation, and ex-situ nanofiltration. J. Environ. Chem. Eng. 2017, 5, 2533–2541. [Google Scholar] [CrossRef]
- Bernardo, M.P.; Coelho, L.F.; Sass, D.C.; Contiero, J. l-(+)-Lactic acid production by Lactobacillus rhamnosus B103 from dairy industry waste. Braz. J. Microbiol. 2016, 47, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Marquesa, S.; Matosad, C.T.; Gírioa, F.M.; Roseiroa, J.C.; Santos, J.A.L. Lactic acid production from recycled paper sludge: Process intensification by running fed-batch into a membrane-recycle bioreactor. Biochem. Eng. J. 2017, 120, 63–72. [Google Scholar] [CrossRef]
- Bahry, H.; Rawa, A.; Pons, A.; Taha, S.; Vial, C. Optimization of lactic acid production using immobilized Lactobacillus rhamnosus and carob pod waste from the Lebanese food industry. J. Biotechnol. 2019, 306, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Lech, M. Optimisation of protein-free waste whey supplementation used for the industrial microbiological production of lactic acid. Biochem. Eng. J. 2020, 157, 107531. [Google Scholar] [CrossRef]
- Okano, K.; Hama, S.; Kihara, M.; Noda, H.; Tanaka, T.; Kondo, A. Production of optically pure d-lactic acid from brown rice using metabolically engineered Lactobacillus plantarum. Appl. Bicrobiol. Biotechnol. 2017, 101, 1869–1875. [Google Scholar] [CrossRef]
- Chen, P.T.; Hong, Z.S.; Cheng, C.L.; Ng, I.S.; Lo, Y.C.; Nagarajan, D.; Chang, J.S. Exploring fermentation strategies for enhanced lactic acid production with polyvinyl alcohol-immobilized Lactobacillus plantarum 23 using microalgae as feedstock. Bioresour. Technol. 2020, 308, 123266. [Google Scholar] [CrossRef]
- Mladenovića, D.; Pejin, J.; Kocić-Tanackov, S.; Radovanović, Ž.; Djukić-Vuković, A.; Mojović, L. Lactic acid production on molasses enriched potato stillage by Lactobacillus paracasei immobilized onto agro-industrial waste supports. Ind. Crops Prod. 2018, 124, 142–148. [Google Scholar] [CrossRef]
- Cubas-Cano, E.; González-Fernández, C.; Ballesteros, I.; Tomás-Pejó, E. Efficient utilization of hydrolysates from steam-exploded gardening residues for lactic acid production by optimization of enzyme addition and pH control. Waste Manag. 2020, 107, 235–243. [Google Scholar] [CrossRef]
- Kim, Y.S.; Jang, J.Y.; Park, S.J.; Um, B.H. Dilute sulfuric acid fractionation of Korean food waste for ethanol and lactic acid production by yeast. Waste Manag. 2018, 74, 231–240. [Google Scholar] [CrossRef]
- Trakarnpaiboon, S.; Srisuk, N.; Piyachomkwan, K.; Yang, S.T.; Kitpreechavanich, V. l-Lactic acid production from liquefied cassava starch by thermotolerant Rhizopus microsporus: Characterization and optimization. Process Biochem. 2017, 63, 26–34. [Google Scholar] [CrossRef]
- Ma, X.; Gao, M.; Yin, Z.; Zhu, W.; Liu, S.; Wang, Q. Lactic acid and animal feeds production from Sophora flavescens residues by Rhizopus oryzae fermentation. Process Biochem. 2020, 92, 401–408. [Google Scholar] [CrossRef]
- Abedi, E.; Hashemi, S.M.B. Lactic acid production–producing microorganisms and substrates sources–state of art. Heliyon 2020, 6, 10. [Google Scholar] [CrossRef] [PubMed]
- Kyla-Nikkila, K.; Hujanen, M.; Leisola, M.; Palva, A. Metabolic engineering of Lactobacillus helveticus CNRZ32 for production of pure l-(+)-lactic acid. Appl. Environ. Microbiol. 2020, 66, 3835–3841. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Dominguez, J.M.; Cao, N.; Du, J.; Tsao, G.T. Optimization of l-lactic acid production from glucose by Rhizopus oryzae ATCC 52311. Appl. Biochem. Biotechnol. 1999, 78, 401–407. [Google Scholar] [CrossRef]
- Kosakai, Y.; Park, Y.S.; Okabe, M. Enhancement of l(+)-lactic acid production using mycelial flocs of Rhizopus oryzae. Biotechnol. Bioeng. 1999, 55, 461–470. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Jina, B.; Kelly, J.M. Production of lactic acid from renewable materials by Rhizopus fungi. Biochem. Eng. J. 2007, 35, 251–263. [Google Scholar] [CrossRef]
- Nigatu, A. Evaluation of numerical analyses of RAPD and API 50CH patterns to differentiate Lactobacillus plantarum, Lact. fermentum, Lact. rhamnosus, Lact. sake, Lact. parabuchneri, Lact. gallinarum, Lact. casei, Weissella minor and related taxa isolated from kocho and tef. J. Appl. Microbiol. 2000, 89, 969–978. [Google Scholar]
- Sanders, M.E.; Klaenhammer, T.R. Invited review: The scientific basis of Lactobacillus acidophilus NCFM functionality as a probiotic. J. Dairy Sci. 2001, 84, 319–331. [Google Scholar] [CrossRef]
- Yun, J.S.; Wee, Y.J.; Ryu, H.W. Production of optically pure l(+)-lactic acid from various carbohydrates by batch fermentation of Enterococcus faecalis RKY1. Enzyme Microb. Technol. 2003, 33, 416–442. [Google Scholar] [CrossRef]
- Joshi, D.; Singhvi, M.; Khire, J.; Gokhale, D. Strain improvement of Lactobacillus lactis for d-lactic acid production. Biotechnol. Lett. 2010, 32, 517–520. [Google Scholar] [CrossRef]
- Bustos, G.; Moldes, A.B.; Cruz, J.M.; Dominguez, J.M. Production of lactic acid from vine-trimming wastes and viticulture lees using a simultaneous saccharification fermentation. J. Sci. Food Agri. 2005, 472, 466–472. [Google Scholar] [CrossRef]
- Tyree, R.W.; Clausen, E.C.; Gaddy, J.L. The fermentative characteristics of Lactobacillus xylosus on glucose and xylose. Biotechnol. Lett. 1990, 12, 51–56. [Google Scholar] [CrossRef]
- Reddy, L.V.; Kim, Y.M.; Yun, J.S.; Ryu, H.W.; Wee, Y.J. l-Lactic acid production by combined utilization of agricultural bioresources as renewable and economical substrates through batch and repeated-batch fermentation of Enterococcus faecalis RKY1. Bioresour. Technol. 2016, 209, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Iyer, P.V.; Thomas, S.; Lee, Y.Y. High-yield fermentation of pentoses into lactic acid. Appl. Biochem. Biotechnol. 2000, 84–86, 665–677. [Google Scholar] [CrossRef]
- Ou, M.S.; Ingram, L.O.; Shanmugam, K.T. l(+)-Lactic acid production from non-food carbohydrates by thermotolerant Bacillus coagulans. J. Ind. Microbiol. Biotechnol. 2011, 38, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sun, L.; Wei, D.; Wang, R. Reducing byproduct formation in l-lactic acid fermentation by Rhizopus oryzae. J. Ind. Microbiol. Biotechnol. 2005, 32, 38–40. [Google Scholar] [CrossRef]
- Cui, F.; Li, Y.; Wan, C. Lactic acid production from corn stover using mixed cultures of Lactobacillus rhamnosus and Lactobacillus brevis. Bioresour. Technol. 2011, 102, 1831–1836. [Google Scholar] [CrossRef]
- Hofvendahl, K.; Hahn-Hägerdal, B. l-Lactic acid production from whole wheat flour hydrolysate using strains of Lactobacilli and Lactococci. Enzyme Microb. Technol. 1997, 20, 301–307. [Google Scholar] [CrossRef]
- Yoshida, S.; Okano, K.; Tanaka, T.; Ogino, C.; Kondo, A. Homo-d-lactic acid production from mixed sugars using xylose assimilating operon-integrated Lactobacillus plantarum. Appl. Microbiol. Biotechnol. 2001, 92, 67–76. [Google Scholar] [CrossRef]
- Korbekandi, H.; Abedi, D.; Jalali, M.; Fazeli, M.; Heidari, M. Optimization of Lactobacillus casei growth and lactic acid production in batch culture. J. Biotechnol. 2007, 131, S182–S183. [Google Scholar] [CrossRef]
- Nout, M.J.R.; Motarjemi, Y. Assessment of fermentation as a household technology for improving food safety: A joint FAO/WHO workshop. Food Control 1997, 8, 221–226. [Google Scholar] [CrossRef]
- Burgos-Rubio, C.N.; Okos, M.R.; Wankat, P.C. Kinetic studies of the conversion of different substrates to lactic acid using Lactobacillus bulgaricus. Biotechnol. Prog. 2000, 16, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, L.M.D.; Xavier, A.N.R.B.; Almanda, J.S.; Corronodo, M.J.T. Concomitant substrate and product inhibition kinetics in lactic acid production. Enzyme Microb. Technol. 1991, 13, 314–319. [Google Scholar] [CrossRef]
- Büyükkileci, A.O.; Harsa, S. Batch production of l-(+) lactic acid from whey by Lactobacillus casei NRRL B-441. J. Chem. Technol. Biotechnol. 2004, 79, 1036–1040. [Google Scholar] [CrossRef]
- Schepers, A.; Thibault, J.; Lacroix, C. Lactobacillus helveticus growth and lactic acid production during pH-controlled batch cultures in whey permeate/yeast extract medium. Part I. Multiple factor kinetic analysis. Enzyme Microb. Technol. 2002, 30, 176–186. [Google Scholar] [CrossRef]
- Ishizaki, A.; Ohta, T.; Kobayashi, G. Computer simulation for l-lactate batch process employing Lactococcus lactis IO-1. J. Biotechnol. 1992, 24, 85–107. [Google Scholar] [CrossRef]
- Bai, D.M.; Wei, Q.; Yan, Z.H.; Zhao, X.M.; Li, X.G.; Xu, S.M. Fed-batch fermentation of Lactobacillus lactis for hyper-production of l-lactic acid. Biotechnol. Lett. 2003, 25, 1833–1835. [Google Scholar] [CrossRef]
- Roukas, T.; Kotzekidou, P. Production of lactic acid from deproteinized whey by coimmobilized Lactobacillus casei and Lactococcus lactis cell. Enzyme Microb. Technol. 1991, 13, 33–38. [Google Scholar] [CrossRef]
- Juturu, V.; Wu, J.C. Microbial production of lactic acid: The latest development. Crit. Rev. Biotechnol. 2016, 36, 967–977. [Google Scholar] [CrossRef]
- Nancib, N.; Nancib, A.; Boudjelal, A.; Benslimane, C.; Blanchard, F.; Boudrant, J. The effect of supplementation by different nitrogen sources on the production of lactic acid from date juice by Lactobacillus casei subsp. rhamnosus. Bioresour. Technol. 2001, 78, 149–153. [Google Scholar] [CrossRef]
- Talukder, M.M.R.; Probir, D.; Wu, J.C. Microalgae (Nannochloropsis salina) biomass to lactic acid and lipid. Biochem. Eng. J. 2012, 68, 109–113. [Google Scholar] [CrossRef]
- Gómez-Gómez, J.A.; Giraldo-Estrada, C.; Habeych, D.; Baena, S. Evaluation of biological production of lactic acid in a synthetic medium and in Aloe Vera (L.) Burm. f. processing by-products. Univ. Sci. 2015, 20, 369–385. [Google Scholar] [CrossRef]
- Chasoy, G.R.; Chairez, I.; Durán-Páramo, E. Carbon/nitrogen ratio and initial pH effects on the optimization of lactic acid production by Lactobacillus casei susp. casei NRRL-441. Wulfenia J. 2020, 27, 37–59. [Google Scholar]
- Munanga, D.J.; Loiseau, G.; Grabulos, J.; Mestres, C. Modeling lactic fermentation of Gowé using Lactobacillus starter culture. Microorganisms 2016, 4, 44. [Google Scholar] [CrossRef]
- Hetényi, K.; Németh, Á.; Sevella, B. Role of pH-regulation in lactic acid fermentation: Second steps in a process improvement. Chem. Eng. Process 2011, 50, 293–299. [Google Scholar] [CrossRef]
- Peeva, L.; Peev, G. A new method for pH stabilization of the lactoacidic fermentation. Enzyme Microb. Technol. 1997, 21, 176–181. [Google Scholar] [CrossRef]
- John, R.P.; Nampoothiri, K.M.; Pandey, A. Solid-state fermentation for l-lactic acid production from agro wastes using Lactobacillus delbrueckii. Process Biochem. 2006, 41, 759–763. [Google Scholar] [CrossRef]
- Atangoa, M.S.; Ghalya, A.E. Effect of temperature on lactic acid production from cheese whey using Lactobacillus helveticus under batch conditions. Biomass Bioenergy 1999, 16, 61–78. [Google Scholar]
- Papadimitriou, K.; Alegría, Á.; Bron, P.A.; de Angelis, M.; Gobbetti, M.; Kleerebezem, M.; Lemos, J.A.; Linares, D.M.; Ross, P.; Stanton, C.; et al. Stress physiology of lactic acid bacteria. Microbiol. Mol. Biol. Rev. 2016, 80, 837–890. [Google Scholar] [CrossRef]
- Roy, D.; LeDuy, A.; Goulet, J. Kinetic of growth and lactic acid production from whey permeate by Lactobacillus helveticus. Can. J. Chem. Eng. 1987, 68, 597–603. [Google Scholar] [CrossRef]
- Peleg, M. A model of microbial growth and decay in a closed habitat based on combined Fermi’s and the logistic equations. J. Sci. Food Agric. 1996, 71, 225–230. [Google Scholar] [CrossRef]
- Qin, H.; Gong, S.S.; Ge, X.Y.; Zhang, W.G. The effect of temperature on l-lactic acid production and metabolite distribution of Lactobacillus casei. Prep. Biochem. Biotechnol. 2012, 42, 564–573. [Google Scholar] [CrossRef] [PubMed]
- Nagarjun, P.A. Parametric optimization of lactic acid production and its scale up using free and immobilized cells of Lactobacillus amylovorus NRRL B- 4542. Int. J. Pure App. Biosci. 2015, 3, 159–168. [Google Scholar] [CrossRef]
- Chiarini, L.; Mara, L.; Tabacchioni, S. Influence of growth supplements on lactic acid production in whey ultrafiltrate by Lactobacillus helveticus. Appl. Microbiol. Biotechnol. 1992, 36, 461–464. [Google Scholar] [CrossRef]
- Gandhi, D.N.; Patel, R.S.; Wadhwa, B.K.; Bansal, N.; Kaur, M.; Kumar, G. Effect of agro-based by-products on production of lactic acid in whey permeate medium. J. Food Sci. Technol. 2000, 37, 292–295. [Google Scholar]
- Kumar, S.; Jha, Y.K.; Chauhan, G.S. Process optimization for lactic acid production from whey using Lactobacillus strains. J. Food Sci. Technol. 2001, 38, 59–61. [Google Scholar]
- Cubas-Cano, E.; González-Fernández, C.; Ballesteros, M.; Tomás-Pejó, E. Biotechnological advances in lactic acid production by lactic acid bacteria: Lignocellulose as novel substrate. Biofuels Bioprod. Biorefining 2018, 12, 290–303. [Google Scholar] [CrossRef]
- Wiselogel, A.; Tyson, J.; Johnsson, D. Biomass feedstock resources and composition. In Handbook on Bioethanol: Production and Utilization; Wyman, C.E., Ed.; Taylor & Francis: Washington, DC, USA, 1996; pp. 105–118. [Google Scholar]
- Wang, Y.; Meng, H.; Cai, D.; Wang, B.; Qin, P.; Wang, Z.; Tan, T. Improvement of l-lactic acid productivity from sweet sorghum juice by repeated batch fermentation coupled with membrane separation. Bioresour. Technol. 2016, 211, 291–297. [Google Scholar] [CrossRef]
- Vijayakumar, J.; Aravindan, R.; Viruthagiric, T. Recent trends in the production, purification and application of lactic acid. Chem. Biochem. Eng. Q. 2008, 22, 245–264. [Google Scholar]
- Komesu, A.; de Oliveira, J.A.R.; Martins, L.H.d.S.; Maciel, M.R.W.; Filho, R.M. Lactic acid production to purification: A review. BioResources 2017, 12, 4364–4383. [Google Scholar] [CrossRef]
- Massoud, M.I.; El-Razek, A.M.A. Suitability of Sorghum bicolor L. stalks and grains for bioproduction of ethanol. Ann. Agric. Sci. 2011, 56, 83–87. [Google Scholar] [CrossRef][Green Version]
- Naveena, B.J.; Altaf, M.; Bhadrayya, K.; Madhavendra, S.S.; Reddy, G. Direct fermentation of starch to l(+) lactic acid in SSF by Lactobacillus amylophilus GV6 using wheat bran as support and substrate medium optimization using RSM. Process Biochem. 2005, 40, 681–690. [Google Scholar] [CrossRef]
- Morlon-Guyot, J.; Guyot, J.P.; Pot, B.; de Haut, I.J.; Raimbault, M. Lactobacillus rnanihotivorans sp. nov., a new starch-hydrolysing lactic acid bacterium isolated during cassava sour starch fermentation. Int. J. Syst. Bacteriol. 1998, 48, 1101–1109. [Google Scholar] [CrossRef] [PubMed]
- Olympia, M.; Fukuda, H.; Ono, H.; Kaneko, Y.; Takano, M. Characterization of starch-hydrolyzing lactic acid bacteria from a fermented fish and rice food, “burong isda”, and its amylolytic enzyme. J. Ferment. Bioeng. 1995, 80, 124–130. [Google Scholar] [CrossRef]
- Sanni, A.; Morlon-Guyot, J.; Guyot, J.P. New efficient amylase-producing strains of Lactobacillus plantarum and L. fermentum isolated from different Nigerian traditional fermented foods. Int. J. Food Microbiol. 2002, 72, 53–62. [Google Scholar] [CrossRef]
- Rodrıguez-Sanoja, R.; Ruiz, B.; Guyot, J.P.; Sanchez, S. Starch-binding domain affects catalysis in two Lactobacillus α-amylases. Appl. Environ. Microbiol. 2005, 71, 297–302. [Google Scholar] [CrossRef]
- Nakamura, L.K. Lactobacillus amylovorus, a new starch-hydrolyzing species from cattle waste corn fermentation. Int. J. Syst. Bacteriol. 1981, 31, 56–63. [Google Scholar] [CrossRef]
- Chatterjee, M.; Chakrabarty, S.L.; Chattopadhyay, B.D.; Mandal, R.K. Production of lactic acid by direct fermentation of starchy wastes by an amylase-producing Lactobacillus. Biotechnol. Lett. 1997, 19, 873–874. [Google Scholar] [CrossRef]
- Giraud, E.; Champailler, A.; Raimbault, M. Degradation of raw starch by a wild amylolytic strain of Lactobacillus plantarum. Appl. Environ. Microbiol. 1994, 60, 4319–4323. [Google Scholar] [CrossRef]
- Vishnu, C.; Seenayya, G.; Reddy, G. Direct fermentation of various pure and crude starchy substrates to l(+) lactic acid using Lactobacillus amylophilus GV6. World J. Microbiol. Biotechnol. 2002, 18, 429–433. [Google Scholar] [CrossRef]
- Naveena, B.J.; Altaf, M.; Bhadrayya, K.; Reddy, G. Production of l(+) lactic acid by Lactobacillus amylophilus GV6 in semi-solid state fermentation using wheat bran. Food Technol. Biotechnol. 2004, 42, 147–152. [Google Scholar]
- Reddy, G.; Altaf, M.; Naveena, B.J.; Venkateshwar, M.; Vijay Kumar, E. Amylolytic bacterial lactic acid fermentation–a review. Biotechnol. Adv. 2008, 26, 22–34. [Google Scholar] [CrossRef]
- Kristoficova, L.; Rosenberg, M.; Vlnova, A.; Sajbidor, J.; Cetrik, M. Selection of Rhizopus strains for l(+) lactic acid and gammalinolenic acid production. Folia Microbiol. 1991, 36, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.; Yin, P.; Ma, Y.; Zhao, L. Production of lactic acid and fungal biomass by Rhizopus fungi from food processing waste streams. J. Ind. Microbiol. Biotechnol. 2005, 32, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.P.; Jin, B.; Lant, P.; Zhou, J. Biotechnological production of lactic acid integrated with potato wastewater treatment by Rhizopus arrhizus. J. Chem. Technol. Biotechnol. 2003, 78, 899–906. [Google Scholar] [CrossRef]
- Yu, R.C.; Hang, Y.D. Kinetics of direct fermentation of agricultural commodities to l(+) lactic acid by Rhizopus oryzae. Biotechnol. Lett. 1989, 11, 597–600. [Google Scholar] [CrossRef]
- Tay, A.; Yang, S.T. Production of l(+)-lactic acid from glucose and starch by immobilized cells of Rhizopus oryzae in a rotating fibrous bed bioreactor. Biotechnol. Bioeng. 2002, 80, 1–12. [Google Scholar] [CrossRef]
- Oda, Y.; Saito, K.; Yamauchi, H.; Mori, M. Lactic acid fermentation of potato pulp by the fungus Rhizopus oryzae. Curr. Microbiol. 2002, 45, 1–4. [Google Scholar] [CrossRef]
- Yin, P.M.; Yahiro, K.; Ishigaki, T.; Park, Y.; Okabe, M. l(+)-Lactic acid production by repeated batch culture of Rhizopus oryzae in air-lift bioreactor. J. Ferment. Bioeng. 1998, 85, 96–100. [Google Scholar] [CrossRef]
- Miura, S.; Arimura, T.; Hoshino, M.; Kojima, M.; Dwiarti, L.; Okabe, M. Optimizaiton and scale-up of l-lactic acid fermentation by mutant strain Rhizopus sp. MK-96-1196 in airlift bioreactors. J. Biosci. Bioeng. 2003, 96, 65–69. [Google Scholar] [CrossRef]
- Miura, S.; Dwiarti, L.; Arimura, T.; Hoshino, M.; Tiejun, L.; Okabe, M. Enhanced production of l-lactic acid by ammonia-tolerant mutant strain Rhizopus sp. MK-96-1196. J. Biosci. Bioeng. 2004, 97, 19–23. [Google Scholar] [CrossRef]
- Kumar, A.K.; Sharma, S. Recent updates on different methods of pretreatment of lignocellulosic feedstocks: A review. Bioresour. Bioprocess. 2017, 4, 7. [Google Scholar] [CrossRef] [PubMed]
- Alvira, P.; Tomas-Pejo, E.; Ballesteros, M.; Negro, M.J. Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: A review. Bioresour. Technol. 2010, 10, 4851–4861. [Google Scholar] [CrossRef] [PubMed]
- Wyman, C.E.; Dale, B.E.; Elander, R.T.; Holtzapple, M.; Ladisch, M.R.; Lee, Y.Y. Coordinated development of leading biomass pretreatment technologies. Bioresour. Technol. 2005, 96, 1959–1966. [Google Scholar] [CrossRef] [PubMed]
- Mussatto, S.I.; Roberto, I.C. Alternatives for detoxification of diluted-acid lignocellulosic hydrolyzates for use in fermentative processes: A review. Bioresour. Technol. 2004, 93, 1–10. [Google Scholar] [CrossRef]
- Sakdaronnarong, C.; Srimarut, N.; Lucknakhul, N.; Na-songkla, N.; Jonglertjunya, W. Two-step acid and alkaline ethanolysis/alkaline peroxide fractionation of sugarcane bagasse and rice straw for production of polylactic acid precursor. Biochem. Eng. J. 2014, 85, 49–62. [Google Scholar] [CrossRef]
- Zhao, X.; Cheng, K.; Liu, D. Organosolv pretreatment of lignocellulosic biomass for enzymatic hydrolysis. Appl. Microbiol. Biotechnol. 2009, 82, 815–827. [Google Scholar] [CrossRef]
- Zhao, X.-b.; Wang, L.; Liu, D.-h. Effect of several factors on peracetic acid pretreatment of sugarcane bagasse for enzymatic hydrolysis. J. Chem. Technol. Biotechnol. 2007, 82, 1115–1121. [Google Scholar] [CrossRef]
- Sakdaronnarong, C.; Saengsawang, A.; Siriyutta, A.; Jonglertjunya, W.; Nasongkla, N.; Laosiripojana, N. An integrated system for fractionation and hydrolysis of sugarcane bagasse using heterogeneous catalysts in aqueous biphasic system. Chem. Eng. J. 2016, 285, 144–156. [Google Scholar] [CrossRef]
- Meethit, P.; Rattanaprasit, P.; Sakdaronnarong, C. Candida shehatae and Saccharomyces cerevisiae work synergistically to improve ethanol fermentation from sugarcane bagasse and rice straw hydrolysate in an immobilized cell bioreactor. Eng. Life Sci. 2016, 16, 706–719. [Google Scholar] [CrossRef]
- Xu, G.C.; Ding, J.C.; Han, R.Z.; Dong, J.J.; Ni, Y. Enhancing cellulose accessibility of corn stover by deep eutectic solvent pretreatment for butanol fermentation. Bioresour. Technol. 2016, 203, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Konda, N.M.; Shi, J.; Singh, S.; Blanch, H.W.; Simmons, B.A.; Klein-Marcuschamer, D. Understanding cost drivers and economic potential of two variants of ionic liquid pretreatment for cellulosic biofuel production. Biotechnol. Biofuels 2014, 7, 86. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Konda, N.V.S.N.M.; Parthasarathi, R.; Dutta, T.; Valiev, M.; Xu, F.; Simmons, B.A.; Singh, S. One-pot integrated biofuel production using low-cost biocompatible protic ionic liquids. Green Chem. 2017, 19, 3152–3163. [Google Scholar] [CrossRef]
- Chen, H.Y.; Xue, D.S.; Feng, X.Y.; Yao, S.J. Screening and production of ligninolytic enzyme by a marine-derived fungal Pestalotiopsis sp. J63. Appl. Biochem. Biotechnol. 2011, 165, 1754–1769. [Google Scholar] [CrossRef]
- Sindhu, R.; Binod, P.; Pandey, A. Biological pretreatment of lignocellulosic biomass—An overview. Bioresour. Technol. 2016, 199, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Lynd, L.R.; Wyman, C.E.; Gerngross, T.U. Biocommodity engineering. Biotechnol. Prog. 1999, 15, 777–793. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, M.; Hoshina, M.; Tanabe, S.; Higuchi, Y.; Sakai, K.; Ohtsubo, S.; Hoshino, K.; Tanaka, T. Production of l-lactic acid by simultaneous saccharification and fermentation using unsterilized defatted rice bran as a carbon source and nutrient components. Food Sci. Technol. Res. 2005, 11, 400–406. [Google Scholar] [CrossRef][Green Version]
- Tu, M.; Saddler, J.N. Potential enzyme cost reduction with the addition of surfactant during the hydrolysis of pretreated softwood. Appl. Biochem. Biotechnol. 2010, 161, 274–287. [Google Scholar] [CrossRef]
- Lynd, L.R.; Weimer, P.J.; van Zyl, W.H.; Pretorius, I.S. Microbial cellulose utilization: Fundamentals and biotechnology. Microbiol. Mol. Biol. Rev. 2002, 66, 506–577. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, S.; Singh, O.V. Bioconversion of lignocellulosic biomass: Biochemical and molecular perspectives. J. Ind. Microbiol. Biotechnol. 2008, 35, 377–391. [Google Scholar] [CrossRef]
- Carvalheiro, F.; Silva-Fernandes, T.; Duarte, L.C.; Gírio, F.M. Wheat straw autohydrolysis: Process optimization and products characterization. Appl. Biochem. Biotechnol. 2009, 153, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Keshwani, D.R.; Cheng, J.J. Switchgrass for bioethanol and other value-added applications: A review. Bioresour. Technol. 2009, 100, 1515–1523. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Zhong, L.; Gao, J.; Sun, N.; Wang, Y.; Sun, G.; Qu, Y.; Zhong, Y. Production of highly efficient cellulase mixtures by genetically exploiting the potentials of Trichoderma reesei endogenous cellulases for hydrolysis of corncob residues. Microb. Cell Fact. 2017, 16, 207. [Google Scholar] [CrossRef]
- Panesar, P.S.; Kennedy, J.F.; Gandhi, D.N.; Bunko, K. Bioutilisation of whey for lactic acid production. Food Chem. 2007, 105, 1–14. [Google Scholar] [CrossRef]
- Roukas, T.; Kotzekidou, P. Lactic acid production from deproteinized whey by mixed cultures of free and coimmobilized Lactobacillus casei and Lactococcus lactis cells using fedbatch culture. Enzyme Microb. Technol. 1998, 22, 199–204. [Google Scholar] [CrossRef]
- Göksungur, Y.; Gündüz, M.; Harsa, Ş. Optimization of lactic acid production from whey by L. casei NRRL B-441 immobilized in chitosan stabilized Ca-alginate beads. J. Chem. Technol. Biotechnol. 2005, 80, 1282–1290. [Google Scholar] [CrossRef]
- Taleghani, H.G.; Najafpour, G.D.; Ghoreyshi, A.A. A study on the effect of parameters on lactic acid production from whey. Pol. J. Chem. Technol. 2016, 18, 58–63. [Google Scholar] [CrossRef]
- Panesar, P.S.; Kennedy, J.F. Biotechnological approaches for the value addition of whey. Crit. Rev. Biotechnol. 2012, 32, 327–348. [Google Scholar] [CrossRef]
- Alonso, S.; Herrero, M.; Rendueles, M.; Díaz, M. Residual yoghurt whey for lactic acid production. Biomass Bioenerg. 2010, 34, 931–938. [Google Scholar] [CrossRef]
- Saadoun, J.H.; Bertani, G.; Levante, A.; Vezzosi, F.; Ricci, A.; Bernini, V.; Lazzi, C. Fermentation of agri-food waste: A promising route for the production of aroma compounds. Foods 2021, 10, 707. [Google Scholar] [CrossRef]
- Zhang, B.; He, P.; Ye, N.; Shao, L. Enhanced isomer purity of lactic acid from the non-sterile fermentation of kitchen wastes. Bioresour. Technol. 2008, 99, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Pleissner, D.; Lam, W.C.; Sun, Z.; Lin, C.S.K. Food waste as nutrient source in heterotrophic microalgae cultivation. Bioresour. Technol. 2013, 137, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Kitpreechavanich, V.; Hayami, A.; Talek, A.; Chin, C.F.S.; Tashiro, Y.; Sakai, K. Simultaneous production of l-lactic acid with high optical activity and a soil amendment with food waste that demonstrates plant growth promoting activity. J. Biosci. Bioeng. 2016, 122, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, Y.; Inokuchi, S.; Poudel, P.; Okugawa, Y.; Miyamoto, H.; Miayamoto, H.; Sakai, K. Novel pH control strategy for efficient production of optically active l-lactic acid from kitchen refuse using a mixed culture system. Bioresour. Technol. 2016, 216, 52–59. [Google Scholar] [CrossRef]
- Neu, D.; Pleissner, K.; Mehlmann, R.; Schneider, G.I.; Puerta-quintero, J.V. Fermentative utilization of coffee mucilage using Bacillus coagulans and investigation of down-stream processing of fermentation broth for optically pure l(+)-lactic acid production. Bioresour. Technol. 2016, 211, 398–405. [Google Scholar] [CrossRef]
- Posada, J.A.; Rincón, L.E.; Cardona, C.A. Design and analysis of biorefineries based on raw glycerol: Addressing the glycerol problem. Bioresour. Technol. 2012, 111, 282–293. [Google Scholar] [CrossRef]
- Biebl, H. Fermentation of glycerol by Clostridium pasteurianum—batch and continuous culture studies. J. Ind. Microbiol. Biotechnol. 2001, 27, 18–26. [Google Scholar] [CrossRef]
- Cheng, K.K.; Zhang, J.A.; Liu, D.H.; Sun, Y.; Yang, M.D.; Xu, J.M. Production of 1,3-propanediol by Klebsiella pneumoniae from glycerol broth. Biotechnol. Lett. 2006, 28, 1817–1821. [Google Scholar] [CrossRef]
- El-Ziney, M.G.; Arneborg, N.; Uyttendaele, M.; Debevere, J.; Jakobsen, M. Characterization of growth and metabolite production of Lactobacillus reuteri during glucose/glycerol cofermentation in batch and continuous cultures. Biotechnol. Lett. 1998, 20, 913–916. [Google Scholar] [CrossRef]
- Durnin, G.; Clomburg, J.; Yeates, Z.; Alvarez, P.J.J.; Zygourakis, K.; Campbell, P.; Gonzalez, R. Understanding and harnessing the microaerobic metabolism of glycerol in Escherichia coli. Biotechnol. Bioeng. 2009, 103, 148–161. [Google Scholar] [CrossRef]
- Hong, A.A.C.; Tanino, K.K.; Peng, F.; Zhou, S.; Sun, Y.; Liu, C.M.; Liu, D.H. Strain isolation and optimization of process parameters for bioconversion of glycerol to lactic acid. J. Chem. Technol. Biotechnol. 2009, 84, 1576–1581. [Google Scholar] [CrossRef]
- Mazumdar, S.; Clomburg, J.M.; Gonzalez, R. Escherichia coli strains engineered for homofermentative production of d-lactic acid from glycerol. Appl. Environ. Microbiol. 2010, 76, 4327–4336. [Google Scholar] [CrossRef] [PubMed]
- Posada, J.A.; Cardona, C.A.; Gonzalez, R. Analysis of the production process of optically pure d-lactic acid from raw glycerol using engineered Escherichia coli strains. Appl. Biochem. Biotechnol. 2012, 166, 680–699. [Google Scholar] [CrossRef] [PubMed]
- Tian, K.; Chen, X.; Shen, W.; Prior, B.A.; Shi, G.; Singh, S.; Wang, Z. High-efficiency conversion of glycerol to d-lactic acid with metabolically engineered Escherichia coli. Afr. J. Biotechnol. 2012, 11, 4860–4867. [Google Scholar]
- Mazumdar, S.; Blankschien, M.D.; Clomburg, J.M.; Gonzalez, R. Efficient synthesis of l-lactic acid from glycerol by metabolically engineered Escherichia coli. Microb. Cell Fact. 2013, 12, 7. [Google Scholar] [CrossRef]
- Niccolai, A.; Shannon, E.; Abu-Ghannam, N.; Biondi, N.; Rodolfi, L.; Tredici, M.R. Lactic acid fermentation of Arthrospira platensis (spirulina) biomass for probiotic-based products. J. Appl. Phycol. 2019, 31, 1077–1083. [Google Scholar] [CrossRef]
- Hwang, H.J.; Kim, S.M.; Chang, J.H.; Lee, S.B. Lactic acid production from seaweed hydrolysate of Enteromorpha prolifera (Chlorophyta). J. Appl. Phycol. 2012, 24, 935–940. [Google Scholar] [CrossRef]
- Yen, H.W.; Che, T.J.; Pan, W.C.; Wu, H.J. Effects of neutralizing agents on lactic acid production by Rhizopus oryzae using sweet potato starch. World J. Microbiol. Biotechnol. 2010, 26, 437–441. [Google Scholar] [CrossRef]
- Pal, P.; Sikder, J.; Roy, S.; Giorno, L. Process intensification in lactic acid production: A review of membrane based processes. Chem. Eng. Proc. 2009, 48, 1549–1559. [Google Scholar] [CrossRef]
- Othman, M.; Ariff, A.B.; Rios-Solis, L.; Halim, M. Extractive fermentation of lactic acid in lactic acid bacteria cultivation: A review. Front. Microbiol. 2017, 8, 2285. [Google Scholar] [CrossRef]
- Moon, S.K.; Wee, Y.J.; Choi, G.W. A novel lactic acid bacterium for the production of high purity l-lactic acid, Lactobacillus paracasei subsp. paracasei CHB2121. J. Biosci. Bioeng. 2012, 114, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Taherzadeh, M.J.; Karimi, K. Enzymatic-based hydrolysis processes for ethanol from lignocellulosic materials: A review. BioResoures 2007, 2, 707–738. [Google Scholar]
- Alfani, F.; Cantarella, L.; Gallifuoco, A.; Cantarella, M. Membrane reactors for the investigation of product inhibition of enzyme activity. J. Membr. Sci. 1990, 52, 339–350. [Google Scholar] [CrossRef]
- Hong, J.; Michael, R.L.; Chen-shung, G.; Phillip, C.W.; Tsao, G.T. Combined product and substrate inhibition equation for cellobiase. Biotechnol. Bioeng. 1981, 23, 2779–2788. [Google Scholar] [CrossRef]
- Kühl, P.W. Excess-substrate inhibition in enzymology and high-dose inhibition in pharmacology: A re-interpretation. Biochem. J. 1994, 298, 171–180. [Google Scholar] [CrossRef]
- Olofsson, K.; Bertilsson, M.; Lidén, G. A short review on SSF-an interesting process option for ethanol production from lignocellulosic feedstocks. Biotechnol. Biofuels 2008, 1, 7. [Google Scholar] [CrossRef]
- Nakano, S.; Ugwu, C.U.; Tokiwa, Y. Efficient production of d-(–)-lactic acid from broken rice by Lactobacillus delbrueckii using Ca(OH)2 as a neutralizing agent. Bioresour. Technol. 2012, 104, 791–794. [Google Scholar] [CrossRef]
- Marques, S.; Santos, J.A.L.; Girio, F.M.; Roseiro, J.C. Lactic acid production from recycled paper sludge by simultaneous saccharification and fermentation. Biochem. Eng. J. 2008, 41, 210–216. [Google Scholar] [CrossRef]
- Wu, Z.; Lee, Y.Y. Nonisothermal simultaneous saccharification and fermentation for direct conversion of lignocellulosic biomass to ethanol. Appl. Biochem. Biotechnol. 1998, 70–72, 479–492. [Google Scholar] [CrossRef]
- Teixeira, L.C.; Linden, J.C.; Schroeder, H.A. Optimizing peracetic acid pretreatment conditions for improved simultaneous saccharification and co-fermentation (SSCF) of sugar cane bagasse to ethanol fuel. Renew. Energ. 1999, 16, 1070–1073. [Google Scholar] [CrossRef]
- Hamelinck, C.N.; van Hooijdonk, G.; Faaij, A.P.C. Ethanol from lignocellulose biomass: Techno-economic performance in short-, middle-, and long-term. Biomass Bioenergy 2005, 28, 384–410. [Google Scholar] [CrossRef]
- Tomás-Pejó, E.; Oliva, J.M.; Ballesteros, M.; Olsson, L. Comparison of SHF and SSF processes from steam-exploded wheat straw for ethanol production by xylose-fermenting and robust glucose-fermenting Saccharomyces cerevisiae strains. Biotechnol. Bioeng. 2008, 100, 1122–1131. [Google Scholar] [CrossRef] [PubMed]
- Alfani, F.; Gallifuoco, A.; Saporosi, A.; Spera, A.; Cantarella, M. Comparison of SHF and SSF processes for the bioconversion of steam-exploded wheat straw. J. Ind. Microbiol. Biotechnol. 2000, 25, 184–192. [Google Scholar] [CrossRef]
- Öhgren, K.; Bengtsson, O.; Gorwa-Grauslund, M.F.; Galbe, M.; Hahn-Hägerdal, B.; Zacchi, G. Simultaneous saccharification and co-fermentation of glucose and xylose in steam-pretreated corn stover at high fiber content with Saccharomyces cerevisiae TMB3400. J. Biotechnol. 2006, 126, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.M.; Lee, Y.Y.; Elander, R.T. Conversion of aqueous ammonia-treated corn stover to lactic acid by simultaneous saccharification and cofermentation. Appl. Biochem. Biotechnol. 2007, 136–140, 721–738. [Google Scholar]
- Qiu, Z.; Gao, Q.; Bao, J. Engineering Pediococcus acidilactici with xylose assimilation pathway for high titer cellulosic l-lactic acid fermentation. Bioresour. Technol. 2018, 249, 9–15. [Google Scholar] [CrossRef]
- Nagamori, E.; Fujita, H.; Shimizu, K.; Tokuhiro, K.; Ishida, N.; Takahashi, H. Fed-batch system for cultivating genetically engineered yeast that produces lactic acid via the fermentative promoter. J. Biosci. Bioeng. 2013, 115, 193–195. [Google Scholar] [CrossRef]
- Mladenović, D.; Djukić-Vuković, A.; Stanković, M.; Milašinović-Šeremešić, M.; Radosavljević, M.; Pejin, J.; Mojović, L. Bioprocessing of agro-industrial residues into lactic acid and probiotic enriched livestock feed. J. Sci. Food Agric. 2019, 99, 5293–5302. [Google Scholar] [CrossRef]
- Choi, H.Y.; Ryu, H.K.; Park, K.M.; Lee, E.G.; Lee, H.; Kim, S.W.; Choi, E.S. Direct lactic acid fermentation of Jerusalem artichoke tuber extract using Lactobacillus paracasei without acidic or enzymatic inulin hydrolysis. Bioresour. Technol. 2012, 114, 745–747. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, X.; Qi, B.; Luo, J.; Shen, F.; Su, Y.; Khan, R.; Wan, Y. Improving lactic acid productivity from wheat straw hydrolysates by membrane integrated repeated batch fermentation under non-sterilized conditions. Bioresour. Technol. 2014, 163, 160–166. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Tashiro, Y.; Zendo, T.; Sakai, K.; Sonomoto, K. Highly efficient l-lactic acid production from xylose in cell recycle continuous fermentation using Enterococcus mundtii QU 25. RSC Adv. 2016, 6, 17659–17668. [Google Scholar] [CrossRef]
- Hu, J.; Lin, Y.; Zhang, Z.; Xiang, T.; Mei, Y.; Zhao, S.; Liang, Y.; Peng, N. High-titer lactic acid production by Lactobacillus pentosus FL0421 from corn stover using fed-batch simultaneous saccharification and fermentation. Bioresour. Technol. 2016, 214, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Pejin, J.; Radosavljević, M.; Kocić-Tanackov, S.; Mladenović, D.; Djukić-Vuković, A.; Mojović, L. Fed-batch l-(+)-lactic acid fermentation of brewer’s spent grain hydrolysate. J. Inst. Brew. 2017, 123, 537–543. [Google Scholar] [CrossRef]
- Fu, X.; Wang, Y.; Wang, J.; Garza, E.; Manow, R.; Zhou, S. Semi-industrial scale (30 m3) fed-batch fermentation for the production of d-lactate by Escherichia coli strain HBUT-D15. J. Ind. Microbiol. Biotechnol. 2017, 44, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Coelho, L.F.; Beitel, S.M.; Sass, D.C.; Neto, P.M.A.; Contiero, J. High titer and productivity of l(+) lactic acid using exponential fed batch fermentation with Bacillus coagulans arr4, a new thermotolerant bacterial strain. 3 Biotech. 2018, 8, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Brock, S.; Kuenz, A.; Prüße, U. Impact of hydrolysis methods on the utilization of agricultural residues as nutrient source for d-lactic acid production by Sporolactobacillus inulinus. Fermentation 2019, 5, 12. [Google Scholar] [CrossRef]
- Li, Z.; Lu, J.; Zhao, L.; Xiao, K.; Tan, T. Improvement of l-lactic acid production under glucose feedback controlled culture by Lactobacillus rhamnosus. Appl. Biochem. Biotechnol. 2010, 162, 1762–1767. [Google Scholar] [CrossRef]
- Romaní, A.; Yáñez, R.; Garrote, G.; Alonso, J.L. SSF production of lactic acid from cellulosic biosludges. Bioresour. Technol. 2008, 99, 4247–4254. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, B.; Li, F.; Xu, K.; Ma, C.; Tao, F.; Li, Q.; Xu, P. Highly efficient production of d-lactate by Sporolactobacillus sp. CASD with simultaneous enzymatic hydrolysis of peanut meal. Appl. Microbiol. Biotechnol. 2011, 89, 1009–1017. [Google Scholar] [CrossRef]
- Ge, X.Y.; Qian, H.; Zhang, W.G. Improvement of l-lactic acid production from Jerusalem artichoke tubers by mixed culture of Aspergillus niger and Lactobacillus sp. Bioresour. Technol. 2009, 100, 1872–1874. [Google Scholar] [CrossRef]
- Ge, X.Y.; Qian, H.; Zhang, W.G. Enhancement of l-lactic acid production in Lactobacillus casei from Jerusalem artichoke tubers by kinetic optimization and citrate metabolism. J. Microbiol. Biotechnol. 2010, 20, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Mishra, H.N. Unstructured kinetic modeling of growth and lactic acid production by Lactobacillus plantarum NCDC 414 during fermentation of vegetable juices. LWT-Food Sci. Technol. 2014, 59, 1123–1128. [Google Scholar] [CrossRef]
- Moser, A. Kinetics of batch fermentation. In Biotechnology, A Comprehensive Treatise, 1st ed.; Rehm, H.J., Reed, G., Eds.; VCH Publisher Inc.: Basel, Switzerland, 1985; pp. 246–278. [Google Scholar]
- Monteagudo, J.; Rodríguez, L.; Rincón, J.; Fuertes, J. Optimization of the conditions of the fermentation of beet molasses to lactic acid by Lactobacillus delbrueckii. Acta Biotechnologica 1994, 14, 251–260. [Google Scholar] [CrossRef]
- Ghimire, A.; Kumar, S.A.; Poudel, R. Kinetics and modeling of growth and lactic acid production in Gundruk, a Himalayan fermented vegetable dish. Food Sci. Nutr. 2020, 8, 5591–5600. [Google Scholar] [CrossRef]
- Stanbury, P.F.; Whitaker, A.; Hall, S.J. Principles of Fermentation Technology, 2nd ed.; Butterworth-Heinemann: Cornwall, UK, 2003; pp. 13–33. [Google Scholar]
- Tashiro, Y.; Kaneko, W.; Sun, Y.; Shibata, K.; Inokuma, K.; Zendo, T.; Sonomoto, K. Continuous d-lactic acid production by a novel thermotolerant Lactobacillus delbrueckii subsp. lactis QU 41. Appl. Microbiol. Biotechnol. 2011, 89, 1741–1750. [Google Scholar] [CrossRef]
- Mimitsuka, T.; Sawai, K.; Kobayashi, K.; Tsukada, T.; Takeuchi, N.; Yamada, K.; Ogino, H.; Yonehara, T. Production of d-lactic acid in a continuous membrane integrated fermentation reactor by genetically modified Saccharomyces cerevisiae: Enhancement in d-lactic acid carbon yield. J. Biosci. Bioeng. 2015, 119, 65–71. [Google Scholar] [CrossRef]
- Gao, T.; Ho, K.P. l-Lactic acid production by Bacillus subtilis MUR1 in continuous culture. J. Biotechnol. 2013, 168, 646–651. [Google Scholar] [CrossRef]
- Lee, R.K.; Ryu, H.W.; Oh, H.; Kim, M.; Wee, Y.J. Cell-recycle continuous fermentation of Enterococcus faecalis RKY1 for economical production of lactic acid by reduction of yeast extract supplementation. J. Microbiol. Biotechnol. 2014, 24, 661–666. [Google Scholar] [CrossRef]
- Wee, Y.J.; Yun, J.S.; Kim, D.; Ryu, H.W. Batch and repeated batch production of l(+)-lactic acid by Enterococcus faecalis RKY1 using wood hydrolyzate and corn steep liquor. J. Ind. Microbiol. Biotechnol. 2006, 33, 431–435. [Google Scholar] [CrossRef]
- Naritomi, T.; Kouda, T.; Yano, H.; Yoshinaga, F.; Shigematsu, T.; Morimura, S.; Kida, K. Influence of broth exchange ratio on bacterial cellulose production by repeated-batch culture. Process Biochem. 2002, 38, 41–47. [Google Scholar] [CrossRef]
- Kim, H.O.; Wee, Y.J.; Kim, J.N.; Yun, J.S.; Ryu, H.W. Production of lactic acid from cheese whey by batch and repeated batch cultures of Lactobacillus sp. RKY2. Appl. Biochem. Biotechnol. 2006, 131, 694–704. [Google Scholar] [CrossRef]
- Wee, Y.J.; Ryu, H.W. Lactic acid production by Lactobacillus sp. RKY2 in a cell-recycle continuous fermentation using lignocellulosic hydrolyzates as inexpensive raw materials. Bioresour. Technol. 2009, 100, 4262–4270. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chang, J.; Cai, D.; Wang, Z.; Qin, P.; Tan, T. Repeated-batch fermentation of l-lactic acid from acid hydrolysate of sweet sorghum juice using mixed neutralizing agent under unsterilized conditions. J. Chem. Technol. Biotechnol. 2017, 92, 1848–1854. [Google Scholar] [CrossRef]
- Tosungnoen, S.; Chookietwattana, K.; Dararat, S. Lactic acid production from repeated-batch and simultaneous saccharification and fermentation of cassava starch wastewater by amylolytic Lactobacillus plantarum MSUL 702. APCBEE Procedia 2014, 8, 204–209. [Google Scholar] [CrossRef]
- Zhao, B.; Wang, L.M.; Li, F.S.; Hua, D.; Ma, C.; Ma, Y.; Xu, P. Kinetics of d-lactic acid production by Sporolactobacillus sp. strain CASD using repeated batch fermentation. Bioresour. Technol. 2010, 101, 6499–6505. [Google Scholar] [CrossRef]
- Luo, S.; Wu, X.; Zhu, Y.; Li, X.; Jiang, S.; Zheng, Z. Fermentative intensity of l-lactic acid production using self-immobilized pelletized Rhizopus oryzae. APCBEE Procedia 2016, 15, 974–979. [Google Scholar]
- Goranov, B.; Blazheva, D.; Kostov, G.; Denkova, Z.; Germanova, Y. Lactic acid fermentation with encapsulated Lactobacillus casei ssp. rhamnosus ATCC 11979 (NBIMCC 1013) in alginate/chitosan matrices. Bulg. J. Agric. Sci. 2013, 19, 101–104. [Google Scholar]
- Thakur, A.; Panesar, P.S.; Sain, M.S. l(+)-Lactic acid production by immobilized Lactobacillus casei using low cost agro-industrial waste as carbon and nitrogen sources. Waste Biomass Valorization 2019, 10, 1119–1129. [Google Scholar] [CrossRef]
- Chang, H.N.; Kim, N.J.; Kang, J.; Jeong, C.M.; Choi, J.D.; Fei, Q.; Kim, B.J.; Kwon, S.; Lee, S.Y.; Kim, J. Multistage high cell continuous fermentation for high productivity and titer. Bioprocess Biosyst. Eng. 2011, 34, 419–431. [Google Scholar] [CrossRef]
- Kourkoutas, Y.; Bekatorou, A.; Banat, I.M.; Marchant, R.; Koutinas, A.A. Immobilization technologies and support materials suitable in alcohol beverages production: A review. Food Microbiol. 2004, 21, 377–397. [Google Scholar] [CrossRef]
- Shahri, S.Z.; Vahabzadeh, F.; Mogharei, A. Lactic acid production by loofah-immobilized Rhizopus oryzae through one-step fermentation process using starch substrate. Bioprocess Biosyst. Eng. 2020, 43, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Radosavljević, M.; Lević, S.; Belović, M.; Pejin, J.; Djukić-Vuković, A.; Mojović, L.; Nedović, V. Immobilization of Lactobacillus rhamnosus in polyvinyl alcohol/calcium alginate matrix for production of lactic acid. Bioprocess Biosyst. Eng. 2020, 43, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Radosavljević, M.; Pejin, J.; Pribić, M.; Kocić-Tanackov, S.; Romanić, R.; Mladenović, D.; Djukić-Vuković, A.; Mojović, L. Utilization of brewing and malting by-products as carrier and raw materials in l-(+)-lactic acid production and feed application. Appl. Microbiol. Biotechnol. 2019, 103, 3001–3013. [Google Scholar] [CrossRef] [PubMed]
- Djukić-Vuković, A.P.; Mojović, L.V.; Jokić, B.M.; Nikolić, S.B.; Pejin, J.D. Lactic acid production on liquid distillery stillage by Lactobacillus rhamnosus immobilized onto zeolite. Bioresour. Technol. 2013, 135, 454–458. [Google Scholar] [CrossRef]
- Bastarrachea, L.; Britt, D.W.; Ward, R.E.; Demirci, A. Development of bioactive solid support for immobilized Lactobacillus casei biofilms and the production of lactic acid. Bioprocess Biosyst. Eng. 2022, 45, 217–226. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, Y.; Yang, S.T.; Wang, R.; Ren, H. A novel honeycomb matrix for cell immobilization to enhance lactic acid production by Rhizopus oryzae. Bioresour. Technol. 2010, 101, 5557–5564. [Google Scholar] [CrossRef]
- Zhang, Y.; Cong, W.; Shi, S.Y. Repeated fed-batch lactic acid production in a packed bed-stirred fermentor system using a pH feedback feeding method. Bioprocess Biosyst. Eng. 2011, 34, 67–73. [Google Scholar] [CrossRef]
- Sirisansaneeyakul, S.; Luangpipat, T.; Vanichsriratana, W.; Srinophakun, T.; Chen, H.H.; Chisti, Y. Optimization of lactic acid production by immobilized Lactococcus lactis IO-1. J. Ind. Microbiol. Biotechnol. 2007, 34, 381–391. [Google Scholar] [CrossRef]
- Faschian, R.; De, S.; Pörtner, R. Multi-fixed-bed bioreactor system applied for bioprocess development of immobilized lactic acid bacteria. Open Biotechnol. J. 2016, 10, 1–9. [Google Scholar] [CrossRef]
- Norton, S.; Lacroix, C.; Vuillemard, J. Kinetic study of continuous whey permeate fermentation by immobilized Lactobacillus helveticus for lactic acid production. Enzyme Microb. Technol. 1994, 16, 457–466. [Google Scholar] [CrossRef]
- Thakur, A.; Panesar, P.S.; Saini, M.S. Continuous production of lactic acid in a two stage process using immobilized Lactobacillus casei MTCC 1423 cells. Int. J. Food Eng. 2018, 4, 216–222. [Google Scholar] [CrossRef]
- Pimtong, V.; Ounaeb, S.; Thitiprasert, S.; Tolieng, V.; Sooksai, S.; Boonsombat, R.; Tanasupawat, S.; Assabumrungrat, S.; Thongchul, N. Enhanced effectiveness of Rhizopus oryzae by immobilization in a static bed fermentor for l-lactic acid production. Process Biochem. 2017, 52, 44–52. [Google Scholar] [CrossRef]
- Zhao, T.; Liu, D.; Ren, H.; Shi, X.; Zhao, N.; Chen, Y.; Ying, H. d-Lactic acid production by Sporolactobacillus inulinus Y2-8 immobilized in fibrous bed bioreactor using corn flour hydrolyzate. J. Microbiol. Biotechnol. 2014, 24, 1664–1672. [Google Scholar] [CrossRef] [PubMed]
- Thongchul, N.; Yang, S.T. Controlling filamentous fungal morphology by immobilization on a rotating fibrous matrix to enhance oxygen transfer and l(+)-lactic acid production by Rhizopus oryzae. In Fermentation Biotechnology; Saha, B.C., Ed.; American Chemical Society: Washington, DC, USA, 2003; pp. 36–51. [Google Scholar]
- Lin, J.; Zhou, M.; Zhao, X.; Luo, S.; Lu, Y. Extractive fermentation of l-lactic acid with immobilized Rhizopus oryzae in a three-phase fluidized bed. Chem. Eng. Process 2007, 46, 369–374. [Google Scholar] [CrossRef]
- Patel, M.; Bassi, A.S.; Zhu, J.J.X.; Gomaa, H. Investigation of a dual-particle liquid-solid circulating fluidized bed bioreactor for extractive fermentation of lactic acid. Biotechnol. Prog. 2008, 24, 821–831. [Google Scholar] [CrossRef]
- Pal, P.; Dey, P. Process intensification in lactic acid production by three stage membrane integrated hybrid reactor system. Chem. Eng. Process 2013, 64, 1–9. [Google Scholar] [CrossRef]
- Wang, C.; Li, Q.; Wang, D.; Xing, J. Improving the lactic acid production of Actinobacillus succinogenes by using a novel fermentation and separation integration system. Process Biochem. 2014, 49, 1245–1250. [Google Scholar] [CrossRef]
- Gaspar, P.; Carvalho, A.L.; Vinga, S.; Santos, H.; Neves, A.R. From physiology to systems metabolic engineering for the production of biochemicals by lactic acid bacteria. Biotechnol. Adv. 2013, 31, 764–788. [Google Scholar] [CrossRef]
- Stefanovic, E.; Fitzgerald, G.; McAuliffe, O. Advances in the genomics and metabolomics of dairy lactobacilli: A review. Food Microbiol. 2017, 61, 33–49. [Google Scholar] [CrossRef]
- Bosma, E.F.; Forster, J.; Nielsen, A.T. Lactobacilli and pediococci as versatile cell factories—Evaluation of strain properties and genetic tools. Biotechnol. Adv. 2017, 35, 419–442. [Google Scholar] [CrossRef]
- Bron, P.A.; Wels, M.; Bongers, R.S.; van Bokhorst-van de Veen, H.; Wiersma, A.; Overmars, L.; Marco, M.L.; Kleerebezem, M. Transcriptomes reveal genetic signatures underlying physiological variations imposed by different fermentation conditions in Lactobacillus plantarum. PLoS ONE 2012, 7, e38720. [Google Scholar] [CrossRef] [PubMed]
- Mazzoli, R.; Bosco, F.; Mizrahi, I.; Bayer, E.A.; Pessione, E. Towards lactic acid bacteria-based biorefineries. Biotechnol. Adv. 2014, 32, 1216–1236. [Google Scholar] [CrossRef] [PubMed]
- Assavasirijinda, N.; Ge, D.Y.; Yu, B.; Xue, Y.F.; Ma, Y.H. Efficient fermentative production of polymer-grade d-lactate by an engineered alkaliphilic Bacillus sp. strain under non-sterile conditions. Microb. Cell Fact. 2016, 15, 3. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.Z.; Ingram, L.O.; Shanmugam, K.T. Evolution of d-lactate dehydrogenase activity from glycerol dehydrogenase and its utility for d-lactate production from lignocellulose. Proc. Natl. Acad. Sci. USA 2011, 108, 18920–18925. [Google Scholar] [CrossRef] [PubMed]
- Ou, M.S.; Awasthi, D.; Nieves, I.; Wang, L.; Erickson, J.; Vermerris, W.; Ingram, L.O.; Shanmugam, K.T. Sweet sorghum juice and bagasse as feedstocks for the production of optically pure lactic acid by native and engineered Bacillus coagulans strains. Bioenerg. Res. 2016, 9, 123–131. [Google Scholar] [CrossRef]
- Tsuge, Y.; Yamamoto, S.; Kato, N.; Suda, M.; Vertes, A.A.; Yukawa, H.; Inui, M. Overexpression of the phosphofructokinase encoding gene is crucial for achieving high production of d-lactate in Corynebacterium glutamicum under oxygen deprivation. Appl. Microbiol. Biotechnol. 2015, 99, 4679–4689. [Google Scholar] [CrossRef]
- Liu, Y.; Gao, W.; Zhao, X.; Wang, J.H.; Garza, E.; Manow, R.; Zhou, S.D. Pilot scale demonstration of d-lactic acid fermentation facilitated by Ca(OH)2 using a metabolically engineered Escherichia coli. Bioresour. Technol. 2014, 169, 559–565. [Google Scholar] [CrossRef]
- Wang, Z.W.; Saini, M.; Lin, L.J.; Chiang, C.J.; Chao, Y.P. Systematic engineering of Escherichia coli for d-lactate production from crude glycerol. J. Agric. Food Chem. 2015, 63, 9583–9589. [Google Scholar] [CrossRef]
- Lu, H.Y.; Zhao, X.; Wang, Y.Z.; Ding, X.R.; Wang, J.H.; Garza, E.; Manow, R.; Iverson, A.; Zhou, S.D. Enhancement of d-lactic acid production from a mixed glucose and xylose substrate by the Escherichia coli strain JH15 devoid of the glucose effect. BMC Biotechnol. 2016, 16, 19. [Google Scholar] [CrossRef]
- Sangproo, M.; Polyiam, P.; Jantama, S.S.; Kanchanatawee, S.; Jantama, K. Metabolic engineering of Klebsiella oxytoca M5a1 to produce optically pure d-lactate in mineral salts medium. Bioresour. Technol. 2012, 119, 191–198. [Google Scholar] [CrossRef]
- Okano, K.; Yoshida, S.; Yamada, R.; Tanaka, T.; Ogino, C.; Fukuda, H.; Kondo, A. Improved production of homo-d-lactic acid via xylose fermentation by introduction of xylose assimilation genes and redirection of the phosphoketolase pathway to the pentose phosphate pathway in l-lactate dehydrogenase gene-deficient Lactobacillus plantarum. Appl. Environ. Microbiol. 2009, 75, 7858–7861. [Google Scholar] [CrossRef] [PubMed]
- Hama, S.; Mizuno, S.; Kihara, M.; Tanaka, T.; Ogino, C.; Noda, H.; Kondo, A. Production of d-lactic acid from hardwood pulp by mechanical milling followed by simultaneous saccharification and fermentation using metabolically engineered Lactobacillus plantarum. Bioresour. Technol. 2015, 187, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.; Zhang, P.; Sun, J.E.; Tu, Y.; Gao, Q.Q.; Zhang, J.; Bao, J. Engineering wild-type robust Pediococcus acidilactici strain for high titer l- and d-lactic acid production from corn stover feedstock. J. Biotechnol. 2016, 217, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Ishida, N.; Suzuki, T.; Tokuhiro, K.; Nagamori, E.; Onishi, T.; Saitoh, S.; Kitamoto, K.; Takahashi, H. d-Lactic acid production by metabolically engineered Saccharomyces cerevisiae. J. Biosci. Bioeng. 2006, 101, 172–177. [Google Scholar] [CrossRef]
- Baek, S.H.; Kwon, E.Y.; Kim, Y.H.; Hahn, J.S. Metabolic engineering and adaptive evolution for efficient production of d-lactic acid in Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 2016, 100, 2737–2748. [Google Scholar] [CrossRef]
- Lee, J.W.; In, J.H.; Park, J.B.; Shin, J.; Park, J.H.; Sung, B.H.; Sohn, J.H.; Seo, J.H.; Park, J.B.; Kim, S.R.; et al. Co-expression of two heterologous lactate dehydrogenases genes in Kluyveromyces marxianus for l-lactic acid production. J. Biotechnol. 2017, 241, 81–86. [Google Scholar] [CrossRef]
- Angermayr, A.S.; van der Woude, A.D.; Correddu, D.; Vreugdenhil, A.; Verrone, V.; Hellingwerf, K.J. Exploring metabolic engineering design principles for the photosynthetic production of lactic acid by Synechocystis sp. PCC6803. Biotechnol. Biofuels 2014, 7, 99. [Google Scholar] [CrossRef]
- Lidan, Y.; Hua, Z.; Zhi, L.; Wu, J.C. Improved acid tolerance of Lactobacillus pentosus by error-prone whole genome amplification. Bioresour. Technol. 2013, 135, 459–463. [Google Scholar]
- Wang, Y.; Li, Y.; Pei, X.; Yu, L.; Feng, Y. Genome-shuffling improved acid tolerance and l-lactic acid volumetric productivity in Lactobacillus rhamnosus. J. Biotechnol. 2007, 129, 510–515. [Google Scholar] [CrossRef]
- Hidalgo-Cantabrana, C.; O’Flaherty, S.; Barrangou, R. CRISPR-based engineering of next-generation lactic acid bacteria. Curr. Opin. Microbiol. 2017, 37, 79–87. [Google Scholar] [CrossRef]
- Chen, P.; Zhou, H.; Liu, W.; Zhang, M.; Du, Z.; Wang, X. The synergistic effect of zinc oxide and phenylphosphonic acid zinc salt on the crystallization behavior of poly(lactic acid). Polym. Degrad. Stab. 2015, 122, 25–35. [Google Scholar] [CrossRef]
- Seader, J.D.; Henley, E.J.; Roper, D.K. Separation Process Principles, 3rd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 299–358. [Google Scholar]
- Krzyzaniak, A.; Leeman, M.; Vossebeld, F.; Visser, T.J.; Schuur, B.; de Haan, A.B. Novel extracts for the recovery of fermentation derived lactic acid. Sep. Purif. Technol. 2013, 111, 82–89. [Google Scholar] [CrossRef]
- Chen, L.; Zeng, A.; Dong, H.; Li, Q.; Niu, C. A novel process for recovery and refining of l-lactic acid from fermentation broth. Bioresour. Technol. 2012, 112, 280–284. [Google Scholar] [CrossRef]
- Sun, B.; Xu, N.; Yao, W.Y.; Zhao, C.Y.; Liu, S.C. Application of centrifugal molecular distillation equipment to l-lactic acid refining process. Chin. J. Chem. Ind. Eng. Prog. 2009, 28, 1872–1876. [Google Scholar]
- Martins, P.F.; Ito, V.M.; Batistella, C.B.; Maciel, M.R.W. Free fatty acid separation from vegetable oil deodorizer distillate using molecular distillation process. Sep. Purif. Technol. 2006, 48, 78–84. [Google Scholar] [CrossRef]
- Lopes, M.S.; Jardini, A.L.; Filho, R.M. Poly(lactic acid) production for tissue engineering applications. Procedia Eng. 2012, 42, 1402–1413. [Google Scholar] [CrossRef]
- Gupta, P.A.; Kumar, V. New emerging trends in synthetic biodegradable polymers—Polylactide: A critique. Eur. Polym. J. 2007, 43, 4053–4074. [Google Scholar] [CrossRef]
- Mecking, S. Nature or petrochemistry?—Biologically degradable materials. Angew. Chem. Int. Ed. 2004, 43, 1078–1085. [Google Scholar] [CrossRef]
- Zhao, J.; Wilkins, R.M. Low molecular weight polylactic acid as a matrix for the delayed release of pesticides. J. Agric. Food Chem. 2005, 53, 4076–4082. [Google Scholar] [CrossRef]
- Jamshidian, M.; Tehrany, E.A.; Imran, M.; Jacquot, M.; Desobry, S. Poly-lactic acid: Production, applications, nanocomposites, and release studies. Compr. Rev. Food Sci. Food Saf. 2010, 9, 552–571. [Google Scholar] [CrossRef]
- Jalil, R.; Nixon, J.R. Biodegradable poly(lactic acid) and poly(lactide-co-glycolide) microcapsules: Problems associated with preparative techniques and release properties. J. Microencapsul. 1990, 7, 297–325. [Google Scholar] [CrossRef] [PubMed]
- Proikakis, C.S.; Tarantili, P.A.; Andreopoulos, A.G. Synthesis and characterization of low molecular weight polylactic acid. J. Elastom. Plast. 2002, 34, 49–63. [Google Scholar] [CrossRef]
- Giménez, B.; Turnay, J.; Lizarbe, M.A.; Montero, P.; Gómez-Guillén, M.C. Use of lactic acid for extraction of fish skin gelatin. Food Hydrocoll. 2005, 19, 941–950. [Google Scholar] [CrossRef]
- Viljanmaa, M.; Södergård, A.; Törmälä, P. The use of lactic acid-based hot melt adhesive in the industrial lamination process. Int. J. Adhes. Adhes. 2003, 23, 151–154. [Google Scholar] [CrossRef]
- Van den Eynde, M.; Van Puyvelde, P. 3D printing of poly(lactic acid). In Industrial Applications of Poly(lactic acid). Advances in Polymer Science, 1st ed.; Di Lorenzo, M., Androsch, R., Eds.; Springer: Cham, Switzerland, 2018; pp. 139–158. [Google Scholar]
| Microorganism | Substrate | Reference |
|---|---|---|
| Lactobacillus delbrueckii sp. bulgaricus | Corn stover | [17] |
| Lactobacillus delbrueckii | Orange waste enzymatic hydrolysates | [18] |
| Lactobacillus delbrueckii | Orange peel wastes hydrolysates | [19,20] |
| Lactobacillus bulgaricus CGMCC 1.6970 | Cheese whey powder | [21] |
| Lactobacillus casei Shirota | Mixed food waste, bakery waste | [22] |
| Lactobacillus casei | Sugarcane bagasse | [23] |
| Lactobacillus rhamnosus B103 | Dairy industry waste | [24] |
| Lactobacillus rhamnosus ATCC 7469 | Recycled paper sludge | [25] |
| Lactobacillus rhamnosus | Solid carob waste | [26] |
| Lactobacillus rhamnosus PCM 489 | Cheese industry—whey | [27] |
| Lactobacillus plantarum | Brown rice | [28] |
| Lactobacillus plantarum | Hydrolysate of microalga Chlorella vulgaris | [29] |
| Lactobacillus paracasei | Molasses-enriched potato stillage | [30] |
| Lactobacillus pentosus CECT4023T | Gardening lignocellulosic residues | [31] |
| Saccharomyces cerevisiae | Food waste biomass | [32] |
| Rhizopus microsporus | Liquefied cassava starch | [33] |
| Rhizopus oryzae | Animal feeds from Sophora flavescens residues | [34] |
| Immobilization Method/Material | Microorganism | Substrate | Lactic Acid (g/L) | Productivity (g/L·h) | Reference |
|---|---|---|---|---|---|
| Loofah sponge | Rhizopus oryzae PTCC 5263 | Soluble potato starch | 5 | - | [207] |
| Alginate beads | Lactobacillus rhamnosus | Carob waste juice | 22 | 1.22 | [26] |
| Poly(vinyl alcohol)/calcium alginate (PVA/Ca-alginate) matrix | Lactobacillus rhamnosus ATCC7469 | 0.8 | [208] | ||
| Zeolite | Lactobacillus rhamnosus ATCC 7469 | Liquid distillery stillage | 42.19 | 1.69 | [209] |
| Sodium alginate | Enterococcus faecalis | Salted cheese whey | 36.95 | - | [210] |
| Chitosan-modified polypropylene | Lactobacillus casei | Synthetic media | 20.4 | - | [211] |
| Asterisk-shaped fibrous matrices in a honeycomb configuration | Rhizopus oryzae | 49.5 | 0.57 | [212] |
| Type of Bioreactor | Fermentation Method | Microorganism | Lactic Acid (g/l) | Productivity (g/L·h) | Reference |
|---|---|---|---|---|---|
| PBR | Fed-batch | L. lactis-11 | 115.0 | 2.3 | [213] |
| Batch | L. lactis IO-1 | 29.78 | 2.2 | [214] | |
| Recycled batch | L. lactis IO-1 | 23.0 | 2.4 | [214] | |
| Continuous | L. lactis IO-1 | 8.9 | 4.5 | [214] | |
| Continuous | L. bulgaricus | n.p. | 1.5 | [215] | |
| CSTR | Batch | L. casei, L. lactis | 22.5 | 0.9 | [131] |
| Repeated batch | L. casei, L. lactis | 46.0 | 1.9 | [131] | |
| Continuous | L. helveticus | n.p. | 28.5 | [216] | |
| Continuous | L. casei MTCC 1423 | 106 | 5.9 | [217] | |
| Repeated batch | R. oryzae | n.p. | 3.8 | [202] | |
| FBR | Batch | R. oryzae NRRL395 | 75.2 | 1.05 | [218] |
| Fed-batch | R. oryzae | 90.0 | 2.5 | [103] | |
| Fed-batch | S. inulinus Y2-8 | 218 | 1.65 | [219] | |
| Continuous | R. oryzae NRRL395 | 72.4 | 0.7 | [218] | |
| Repeated batch | R. oryzae | 137 | 2.1 | [220] | |
| Continuous | R. oryzae | n.p. | 11 | [221] | |
| Continuous | L. bulgaricus | 0.45 | n.p. | [222] | |
| MCRR | Continuous | L. delbrueckii NCIM-2025 | 250 | 12.4 | [223] |
| Continuous | A. succinogenes | 183.4 | 1.5 | [224] | |
| Continuous | E. faecalis RKY1 | 90.0 | 3.72 | [194] | |
| Repeated batch | E. faecalis RKY1 | 95.5 | 6.4 | [46] | |
| Continuous | E. mundtii QU 25 | 41.0 | 6.2 | [175] | |
| Fed-batch | L. rhamnosus ATCC 7469 | 58.0 | 6.9 | [25] |
| Microorganism | Modification | Substrate | Lactic Acid (g/L) | Reference |
|---|---|---|---|---|
| Bacillus sp. N16-5 | l-Lactate dehydrogenase gene (ldhL) was knocked out, and the d-lactate dehydrogenase gene (ldhD) from L. delbrueckii was introduced to construct a d-lactate producer. Exopolysaccharide biosynthesis (epsD) was subsequently disrupted. | Glucose/peanut meal | 142.1 | [230] |
| Bacillus coagulans | Deletion of native l-lactate dehydrogenase gene (ldhL) and acetolactate synthase gene (alsS) to impede anaerobic growth, and suppressor mutants that restored growth. | Glucose/LB medium | 99.8 | [231] |
| Bacillus coagulans | Deletion of native l-lactate dehydrogenase gene (ldhL) and acetolactate synthase gene (alsS) to impede anaerobic growth, and suppressor mutants that restored growth. | Sorghum juice, corn steep liquor | 124.4 | [232] |
| Corynebacterium glutamicum | Overexpression of the phosphofructokinase encoding gene. | Glucose/mineral salt medium | 195.0 | [233] |
| Escherichia coli HBUT-D | The E. coli strain HBUT-D (ΔpflB ΔfrdABCD ΔadhE Δpta ΔaldA ΔcscR) engineered from E. coli W (ATCC 9637) for d-lactic acid production. | Glucose/NBS medium/yeast extract | 127.0 | [234] |
| Escherichia coli BLac-2106 | Elimination of mgsA and enhanced expression of glpD-glpK in the glycerol catabolism and of a heterologous gene encoding d-lactate dehydrogenase. | Glucose/mineral salt medium | 105 | [235] |
| Escherichia coli JH15 | Replaced the recombinant l-lactate dehydrogenase gene (ldhL) with a d-lactate dehydrogenase gene (ldhA). | Glucose/xylose | 83.0 | [236] |
| Klebsiella oxytoca KMS002 | Deletion of alcohol dehydrogenase gene, adhE, and the phospho-transacetylase/acetate kinase A genes, pta-ackA. | Maltodextrin derived from cassava | 33.6 | [237] |
| Lactobacillus plantarum | Deletion of l-lactate dehydrogenase gene (ldhl1), expression of alpha-amylase (AmyA) from Streptococcus bovi. | Raw corn starch | 72.8 | [238] |
| Lactobacillus plantarum | Introduced xylose-assimilating xylAB operon from L. pentosus (PXylAB) and phosphoketolase 1 gene (xpk1) was replaced with the transketolase gene (tkt) from L. lactis, and the phosphoketolase 2 (xpk2) gene was deleted. | SSF/delignified hardwood pulp | 102.5 | [239] |
| Pediococcus acidilactici ZP26 | Disruption of ldhD or ldh gene. | Corn stover/peptone, yeast extract | 77.8 | [240] |
| Saccharomyces cerevisiae OC 2 | Deletion of pyruvate decarboxylase 1 and introduction of two copies of d-LDH gene. | YPD medium/peptone, yeast extract/glucose | 61.5 | [241] |
| Saccharomyces cerevisiae JHY5330 | Overexpression of d-lactate dehydrogenase gene (ldhA, LEUM_1756) of Leuconostoc mesenteroides ATCC 8293. | YPD medium/peptone, yeast extract/glucose | 112.0 | [242] |
| Kluyveromyces marxianus | l-lactate dehydrogenase (LDH) expression of Staphylococcus epidermidis, Lactobacillus acidophilus, and Bos taurus. | Glucose | 24.0 | [243] |
| Synechocystis sp. PCC6803 | Increased expression level of lactate dehydrogenase (LDH), co-expression of a heterologous pyruvate kinase, knockdown of phosphoenolpyruvate carboxylase. | CO2 | 12.9 | [244] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.; Kim, Y.-M.; Lebaka, V.R.; Wee, Y.-J. Lactic Acid for Green Chemical Industry: Recent Advances in and Future Prospects for Production Technology, Recovery, and Applications. Fermentation 2022, 8, 609. https://doi.org/10.3390/fermentation8110609
Kim J, Kim Y-M, Lebaka VR, Wee Y-J. Lactic Acid for Green Chemical Industry: Recent Advances in and Future Prospects for Production Technology, Recovery, and Applications. Fermentation. 2022; 8(11):609. https://doi.org/10.3390/fermentation8110609
Chicago/Turabian StyleKim, Jua, Young-Min Kim, Veeranjaneya Reddy Lebaka, and Young-Jung Wee. 2022. "Lactic Acid for Green Chemical Industry: Recent Advances in and Future Prospects for Production Technology, Recovery, and Applications" Fermentation 8, no. 11: 609. https://doi.org/10.3390/fermentation8110609
APA StyleKim, J., Kim, Y.-M., Lebaka, V. R., & Wee, Y.-J. (2022). Lactic Acid for Green Chemical Industry: Recent Advances in and Future Prospects for Production Technology, Recovery, and Applications. Fermentation, 8(11), 609. https://doi.org/10.3390/fermentation8110609








