Abstract
Fungal fermented foods are nutritious, environmentally friendly and sustainable protein sources. To develop fungal fermented food with acceptable sensory characteristics, it is important to assess factors that can affect the sensory characteristics of the product. The current study aimed to investigate the correlations between the chemical and microbiological characteristics and sensory characteristics of fungal fermented food. Soybeans were fermented using five local Indonesian strains of the genus Rhizopus sp. and one strain of industrial starter to mimic traditional Indonesian tempe. The chemical (amino acid and ammonia content), microbiological (lactic acid bacteria, proteolytic bacteria and yeast) and sensory characteristics of the fermented products were examined. The results showed that there is a correlation between the chemical properties, particularly glutamic acid and aspartic acid, and the overall liking of different types of tempe. In general, Rhizopus oligosporus-fermented products had better sensory characteristics than those fermented with Rhizopus oryzae and Rhizopus delemar. The sensory characteristics of the fermented products in this work made from isolates are comparable to those made with an industrial starter culture. In addition, taste and texture affect the overall liking of the product. The results of this study contribute to the development of acceptable sensory fungal fermented food and, in particular, the screening of potential starters.
1. Introduction
A major challenge of food production in the future will be providing sufficient and sustainable food for the growing population while also preserving the environment for future generations. This challenge drives an exploration of food sources that are affordable and highly nutritious, have a low carbon footprint and are associated with the efficient use of natural resources. Such food will contribute to meeting two sustainable development goals, i.e, zero hunger and climate action. Plant-based proteins generally have lower carbon footprints compared to animal-based proteins. For instance, soybeans release 0.19–0.32 kg CO2 eq/kg [1]. This is significantly lower than the emissions of beef, which is 23.4–27.2 kg CO2 eq/kg [2]. In addition, plant-based protein sources such as legumes and cereals contain high amounts of macronutrients and micronutrients.
Soybeans are one of the main plant-based protein sources, and they are an inexpensive source of dietary protein. Soybeans contain a considerable amount of protein, which can be up to 36% of their dry weight [3]. Moreover, the essential amino acids profile required for human nutrition is comparable to that of animal-based protein, with the exception of methionine and cysteine [4]. However, soybeans contain antinutritional compounds such as phytates, tannins, trypsin inhibitors and oligosaccharides [5]. These antinutritional compounds reduce the digestibility of proteins and mineral adsorption [6]. Fermentation is an ancient and traditional approach to reduce antinutritional and toxic compounds and to improve nutritional accessibility. Fermentation can also prolong shelf life and alter the sensory characteristics of products [7].
Fermented soy products have been part of staple diets in Asia for centuries and are currently becoming increasingly available worldwide due to nutritionally-related concerns regarding food products. Examples of fermented soy products include solid soybean cake, soy sauce or semisolid soy paste. Regarding the sensory acceptability of fermented products, it is important to obtain information relating to factors affecting the sensory characteristics of products. A few studies have investigated the correlations between chemical and microbiological characteristics and sensory characteristics in the context of bacteria-fermented dairy products. However, such correlations have yet to be studied in fungal-fermented soybean products.
Tempe is a product that consists of cooked and fermented soybeans that are held together by the mycelium of filamentous fungi Rhizopus spp. Recently, tempe has gained more popularity globally due to its health benefits. During fermentation, protein is degraded into amino acids and other metabolites by enzymes secreted by microorganisms [8]. Several amino acids play an important role in determining how the end product will taste, for example, umami, sweet or bitter [9]. Moreover, the traditional fermentation of soybeans may be carried out in non-sterile environments. The indigenous microflora in soybeans might thus affect the chemical composition and characteristics of the end product. This study aimed to analyze the correlations between the chemical and microbiological profiles and sensory characteristics of various fungal-fermented soybeans in order to aid the development of acceptable fungal-fermented food products.
2. Materials and Methods
2.1. Microorganism and Inoculums
The Rhizopus strains were isolated from traditional tempe starter cultures, namely Usar and Laru collected from different locations in Yogyakarta Province, Indonesia. Five strains belonging to genus Rhizopus with different chemical compositions were used in this study. The isolates were grown in PDA (Merck, Darmstadt, Germany) and kept at 4 °C until use. For comparison, an industrial starter powder (Raprima, Indonesia) containing R. microsporus var oligosporus spores was also used.
The isolation of the Rhizopus spp. were conducted according to a method previously described by Bavaro et al. [10] with minor modifications. Five grams of usar was dissolved in 45 mL of 0.05% Tween 80 (Merck, Hohenbrunn, Germany) to extract the spores, and the mixture was then homogenized using a stomacher (Seward, London, UK). One mL of spore suspension was inoculated into DRBC (Oxoid, Hampshire, UK) agar medium and incubated at 30 °C for seven days. After incubation, the colonies were transferred to a new agar medium until pure colonies were obtained. The isolates were identified based on their morphology [11,12].
The inoculum was prepared according to Azeke et al. [13]. The isolates were cultured in PDA medium, and the spores were harvested by adding 10 mL of 0.05% Tween 80 to reach 106 spores mL−1. The spore suspension was then used for making tempe starter powder [14]. The medium for the tempe starter was a mixture of rice and tofu with a ratio of 1:1. Ten milliliters of spore suspension was cultivated in 100 g of the medium and incubated for four days. Then, the medium was dried at 40 °C and milled to form a starter powder.
2.2. Fungal Fermentation
In this study, soybeans were fungal-fermented the same way that tempe is common in traditional tempe production, using solid-state fermentation method. The soybeans were soaked for 24 h and then boiled for 30 min. After this, the soybeans were dehulled and soaked for another 24 h. After the soybeans were boiled for another 30 min, excess water was drained and the soybeans were cooled down to room temperature. The boiled soybeans were then inoculated with 1% (w/w) starter powder containing 107 fungal spore/g and incubated at 29.5 °C temperature for 36 h.
2.3. Analytical Methods
2.3.1. Microflora Analysis
The microbial flora analyses consist of enumeration of lactic acid bacteria (LAB), yeast and proteolytic bacteria populations. Five grams of each sample were crushed and homogenized in 45 mL of sterile saline water (0.85% NaCl). The samples were then cultivated in PGY agar with addition of 100 ppm of chloramphenicol, 2% skim and 100 ppm of Na azide for yeast, proteolytic as well as lactic acid bacteria. They were then incubated at 37 °C for LAB and proteolytic bacteria and 30 °C for yeast for 48 h. The total number of LAB, proteolytic bacteria and yeast colonies on plates was expressed as the number of colony-forming units (CFU) per gram sample. The samples were taken at 6, 12, 18, 24, 30 and 36 h.
2.3.2. Amino Acid Composition
A 60 mg sample was ground and 4 mL of 6 M HCl was added for hydrolysis. The hydrolysis was conducted at 105 °C for 4 h. The mixture was then neutralized with 6 M NaOH and diluted to reach 100 mL. Subsequently, the mixture was filtered through 0.2 µm Whatman. Ten microliters of protein hydrolysate were diluted to 1 mL. For derivatization with OPA, 50 mg of OPA were dissolved in 4.5 mL of methanol and mixed with 0.5 mL of 0.4 M potassium borate buffer (pH = 9.5) and 50 µL 2-mercaptoethanol. The 100 µL of samples (or standard amino acid) were mixed with 200 µL OPA solution, followed by incubation at 25 °C for 1 min and used immediately for HPLC analysis.
Twenty µL of analyte were injected into HPLC (Shimadzu, Kyoto, Japan) with Eurospher C18 columns (4.6 × 250 mm, 5 μm) and Fluorescence Lab Alliance™ 1200 Series detectors (Shimadzu, Kyoto, Japan) with excitation of 340 nm wavelengths and emission wavelengths of 455 nm. The flow rate used was 1 mL min−1. The amino acid concentration was calculated using an external standard. The mobile phase was a mixture of solvent A and B. Solvent A was a mixture of 0.042 N sodium phosphate buffer pH 6.8 and 1.2% tetrahydrofuran, whereas solvent B was methanol. The ramp begins at 0 min, 86% (A) 14% (B); 14 min, 80% (A) 20% (B); 18 min, 70% (A) 30% (B); 18 min, 55% (A) 45% (B); 24 min, 50% (A) 50% (B), 26 min, 30% (A) 70% (B), 30–32 min 100% (A), 32.1–39, 86% and (A) 14% (B).
2.3.3. Analysis of Ammonia
Ammonia extraction was carried out according to a method previously described by Parris and Foglia [15]. Five grams of the fermented products was dissolved in 25 mL of distilled water and stirred for 10 min with a magnetic stirrer. The mixture was then added to 75 mL of methanol and stirred for 20 min. Next, the extract was subjected to analysis following a standardized method for determination of ammonia (Indonesian National Standard 06-6989.30-2005). One milliliter of phenol, 1 mL sodium nitroprusside and 2.5 mL of oxidizing solution was added to 25 mL tempe extract. The mixture was then homogenized and incubated for 1 h before measurement with a spectrophotometer (Shimadzu, Kyoto, Japan) at a wavelength of 640 nm. The concentration of ammonia was determined by plotting against ammonia standard curves (R2 = 0.99).
2.4. Sensory Evaluation
Traditional tempe as well as the fungal-fermented soybeans that resulted from the experiments of this study were assessed. This means that the samples used were made through the use of either a commercially available starter culture or lab-cultivated ones. All the resulting tempe products were subjected to hedonic and descriptive tests based on the method previously reported by Meilgaard et al. [16]. The hedonic test was performed with 60 untrained panelists who were asked to evaluate sensory characteristics including appearance, tempe aroma, texture, taste and overall liking. For the hedonic and the descriptive test, respectively, the panelists were asked to use a structured scale to provide a score for each attribute on a level from 1 (extremely dislike/extremely low) to 7 (extremely like/extremely high).
2.5. Statistical Analyses
Correlations between the microbial flora and the chemical composition with sensory characteristics were determined using Pearson’s test for correlation. P-values below 0.05 were considered statistically significant. The statistical analyses were performed using IBM SPSS Statistics 25. Principal Component Analysis was carried out using STATGRAPHICS Centurion XVI (Statpoint Technologies, Inc., Warrenton, VA, USA). The experiments were performed in duplicates and the results were shown as averages. The standard deviations were in the range of 1.6–7.8%.
3. Results and Discussions
Fermented food has been consumed by humans for thousands of years. The initial intention of making fermented food is for preservation; however, it is now appreciated as a healthy food with attractive sensory properties. Fermented foods, particularly made in Asia and Africa, often rely on spontaneous fermentation, even though the addition of starter culture offers better consistency, quality and safety [17]. In order to develop a starter culture, it is important to obtain information on the factors affecting the sensory of fermented products. A few studies have been conducted on this topic; however, they mainly investigated dairy fermented products using bacteria.
The current study investigated the correlations of the chemical and microbiological characteristics and sensory characteristics of Rhizopus spp.-fermented soybean products. Knowledge of such correlations is required in developing products with acceptable sensory characteristics. To achieve this, soybeans were fermented using five different strains of Rhizopus spp. using a method similar to traditional tempe production.
3.1. Isolation and Identification of the Rhizopus sp.
After a series of sub-culturing, pure colonies known as isolates were obtained. In a preliminary study, 12 isolates were obtained and used in the fermentation of soybeans. The sensory characteristics of the fungal-fermented soybean products were evaluated using hedonic tests. The products that received high scores for overall acceptability were identified and used for further studies. The result of this process is presented in Table 1 and Figure 1. Among the strains that produced products with high scores for acceptability were R. oligosporus, R. oryzae and R. delemar. Morphologically, R. delemar is similar to R. oryzae, except for the longer sporangiophore and regular shape of sporangiospore of R. oryzae. R. oligosporus strains were coded as RO1, RO2 and RO3, whereas R. delemar was coded as RD1 and the R. oryzae was coded as RZ1. The commercial starter was used with the reference code R. The massive exploitation of certain strains of Rhizopus species might threaten the diversity of Rhizopus species that can be used in the production of tempe. In the 1970s, it was reported that five Rhizopus species were used in tempe production. These include R. oligosporus, R. oryzae, R. arrhizus and R. stolonifera [11]. Despite this range, only R. oligosporus and R. delemar were found in tempe samples taken at 26 locations in Indonesia [11]. Tempe samples from Java and Sumatera contained no other strains but R. oligosporus. The decline of the diversity of Rhizopus species in tempe production might affect the supply of valuable metabolites that can affect human health as well as sensory characteristics [11]. The significance of this study may thus lay in its contribution to maintaining the diversity of Rhizopus sp. in tempe production.

Table 1.
Morphological identification of five local isolates of Rhizopus spp.
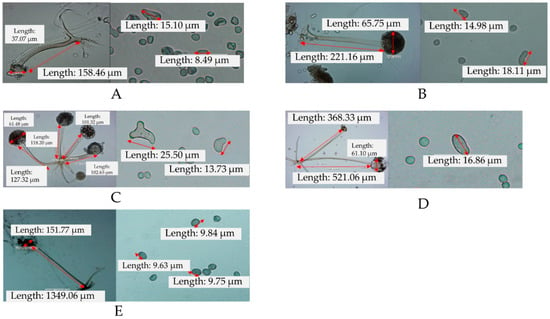
Figure 1.
Sporangiophore, sporangium, columellae (left) and sporangiospore (right) of isolates Rhizopus oligosporus 1 (A), Rhizopus oligosporus 2 (B), Rhizopus oligosporus 3 (C), Rhizopus delemar (D) and Rhizopus oryzae (E). The pictures were taken at 400× magnification, except for R. oryzae, which was taken at 40× magnification.
3.2. Fungal Fermentation
3.2.1. Chemical Profiles of Fungal-Fermented Soybean Products
The chemical composition of fungal-fermented products varies with different Rhizopus sp. because these secrete a wide range of enzymes including amylase, lipase, cellulases, proteases, polygalacturonase hemicellulases, urease, ribonuclease and pectate lyase [18]. The first five enzymes can be detected in soybeans that were fermented using different Rhizopus strains [19]. These enzymes play an important role in the penetration of the bean by fungal mycelium, which leads to changes in the physical and chemical properties of the soybeans [20]. Among the physical changes are an increase in the pH and a decrease in the hardness of the soybean. Chemical changes include an increase in crude protein, soluble protein, amino acid, antioxidant, crude fiber, ash and vitamin as well as a decrease in dry matter, crude lipid and antinutrient compounds [8]. During solid-state fermentation, 65% of the protein in soybeans is hydrolyzed into amino acids [21]. Amino acids can be grouped into triggers for sensory characteristics such as umami (glutamic acid, aspartic acid and tyrosine [22]); sweet (methionine, alanine, glycine and serine [23]); and bitter (proline, valine, leucine and phenylalanine [24]).
The results of the current study show that the amino acid compositions varied between the different fermented products derived from using different fungal strains (Table 2). Higher amounts of glutamic acid, aspartic acid and tyrosine were obtained when fermenting soybeans using R. oligosporus (samples RO1, RO2, RO3 and R) compared to the other strains. The dominant amino acids found in soybeans that were fermented using R. delemar include histidine, methionine lysin, tyrosine and valine. Soybeans fermented using R. oryzae contained a notable amount of histine, isoleucine, phenilalanine, threonine, tyrosine and valine. The total amino acid profile of the local isolates RO3 and RZ were similar to the references. Among the amino acids, glutamic acid is considered to be the most critical, and it is commercially produced as a flavor enhancer for food. Glutamic acid results in savory or umami characteristics, which are crucial attributes in tempe. The highest amounts of glutamic acid were obtained from isolate RO3, followed by R, RO1, RO2, RZ and RD. The result of the study indicates that local isolates have a similar amino acid content to that of the reference product. Yet, some isolates even contained higher amounts of total amino acids than the reference. The results therefore indicate that local isolates have the potential to be developed to become industrial starter cultures.

Table 2.
Profile of amino acids of the fermented soybean with various Rhizopus strains.
Ammonia is a derivative product of protein oxidation. Approximately a quarter of the protein in soybean cotyledons is hydrolyzed, of which approximately 10% is converted into ammonia [25]. Ammonia is also produced from the deamination of amino acids as carbon and an energy source in bacterial growth [26]. The current study found that the level of ammonia increases during fermentation for all strains (Figure 2). The presence of ammonia indicates over-fermentation. Levels of ammonia can therefore contribute to determining the shelf life of fungal-fermented soybeans. The shelf life of fresh tempe stored at ambient temperature is 1–2 days [27]. The shelf life of tempeh can be prolonged by up to five days using packaging such as food-grade wrapping paper or perforated polyethylene bags [28].
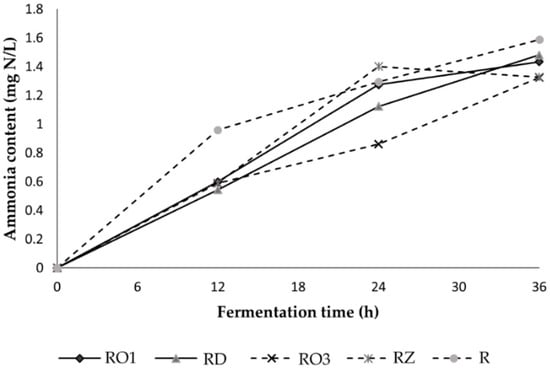
Figure 2.
Changes in the Ammonia Profile during Fermentation of Soybean with Various Rhizopus Strains.
3.2.2. Microflora Profile during Fermentation
Besides Rhizopus sp., there might be other microorganisms involved in the fermentation process when conditions are not sterile [29]. LAB and yeast are commonly found in tempe [30] and might present in the inoculum, water or soybeans. LAB can cause a decrease in pH which, in turn, inhibits the growth of pathogenic bacteria [31]. On the other hand, the role of yeast and proteolytic bacteria in tempe is rarely reported. Yet, there may be an interaction between yeast, bacteria and molds during fermentation. LAB most probably obtain nutrients from molds and yeasts. In food fermentation, it is known that yeast can synthesize vitamins, amino acids, purines and compounds derived from complex carbohydrates, which are required for the growth of LAB [32]. LAB provides lactic acid necessary for yeasts [33].
The results reveal that a shift in the microbial flora occurred during the fermentation of soybeans. LAB growth increased sharply and reached the stationary phase at 6–12 h for both local isolates and the industrial starter (Figure 3). A high level of LAB up to 109 was present in soybeans fermented using isolate RZ. Similar levels of LAB were also found in Malaysian tempe [34]. During the growth of LAB, the pH decreased as a result of acids produced by LAB. These conditions stimulate the growth of the fungi but simultaneously inhibit the growth of yeasts (Figure 4). These findings are in agreement with a previous study which reported that acidification could promote the spore germination of R. oligosporus and therefore shorten the lag phase [35]. LAB plays a vital role in tempe safety by organically producing acid, decreasing the pH and producing metabolites that inhibit pathogens [31].
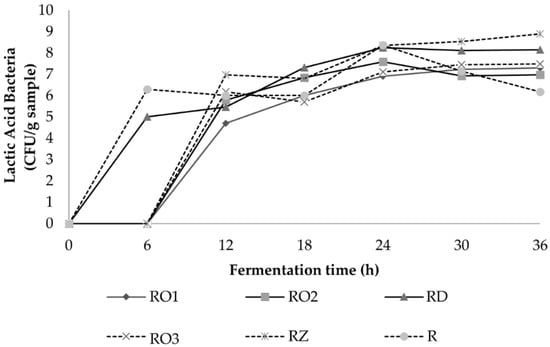
Figure 3.
Lactic acid bacteria population during fermentation of soybean with various Rhizopus Strains.
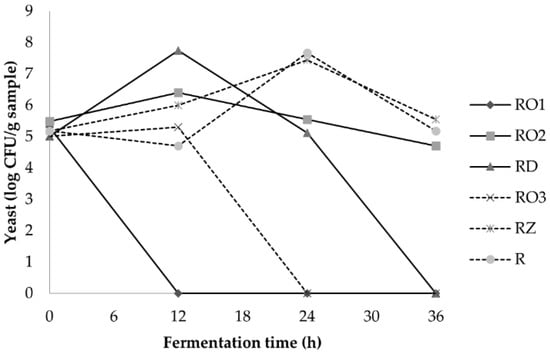
Figure 4.
Yeast population during fermentation of soybean with various Rhizopus strains.
The population of yeast in the soybeans fermented using the strains RO2, RZ and the reference was stable and started to decrease after 24 h. A decrease in yeast was already observed after 12 h in soybeans fermented using the isolates RO1, RO3 and RD. The decline of yeast was followed by an increase in proteolytic bacteria at 18–24 h (Figure 5). The increase in proteolytic bacteria is in line with the increase in ammonia and indicates that proteolytic bacteria might be involved in protein oxidation during fermentation. The microbial analysis showed that the profile of lactic acid bacteria and proteolytic bacteria are similar in all samples. However, a slight difference was observed in yeast profiles.
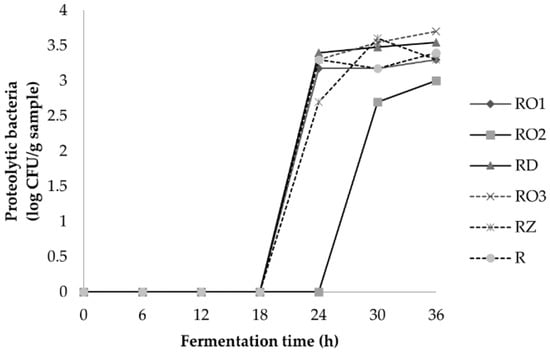
Figure 5.
Proteolytic bacteria population during fermentation of soybean with various Rhizopus strains.
3.3. Sensory Evaluation of Tempe Made from Different Fungal Strains
The results of the sensory evaluation are presented in Table 3. The results show that the appearance of the samples was similar, except for the sample of soybeans fermented using RZ. The different appearances could be due to the different fungal species. The mycelium of R. oligosporus is denser than that of R. oryzae [35], which may result in visual differences. The starter used is an important factor in determining the appearance, flavor and patty integrity of tempe [36]. In the current study, no differences between the samples were observed in terms of aroma and texture. While the participants liked the taste of the samples, the score for RD was significantly lower than that of the other samples. The overall acceptability of the local strains RO3, RO1 and RO2 were similar to that of the reference. The scores for the overall acceptability for isolates RD and RZ were lower than that of the samples made by other strains. The samples of the soybeans fermented using RZ scored lowest in terms of aroma. This might be due to the ability of R. oryzae to produce amylase and lactic acid which can cause undesirable sour-off flavors [35]. With the exception of soybeans fermented using RZ and RD, the samples based on local isolates were perceived to be as acceptable as the samples made using the commercial starter culture. The results thus indicate that a variety of Rhizopus strains have the potential to be further developed to become industrial starter cultures.

Table 3.
Sensory evaluation of soybeans fermented using various Rhizopus strains.
A principal component analysis was performed to assess the correlations between the fungal strains and the characteristics of the fermented soybean samples. Three components were extracted from the PCA results, which accounted for 86% of the variability in the original data (Figure 6). The results show that the different fungi species produced different metabolite profiles. Soybeans fermented using R. oligosporus contain glutamic and aspartic acid, which cause a pleasant aroma, taste, texture and appearance. Glutamic and aspartic acid also cause umami characteristics, which might explain the high overall liking score of R. oligosporus-fermented samples.
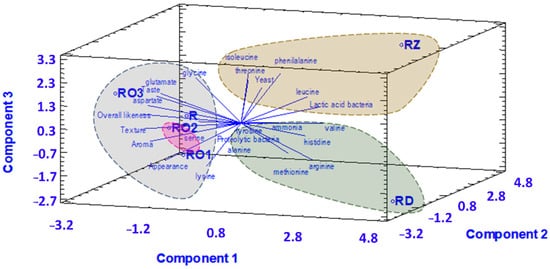
Figure 6.
Principal component analysis 3D biplot of correlation of fungal species with chemical and sensory characteristics of fermented soybean samples.
Soybeans fermented using R. oryzae contain isoleucine, threonine, leucine and phenilalanine. Leucine and phenilalanine cause bitterness. Soybeans fermented using R. oryzae also showed a strong correlation with yeast and LAB. R. oryzae and LAB both produce lactic acid, which can lead to sour off-flavor characteristics. A combination of sour off-flavor and bitterness might be the reason for the low score for taste, aroma and overall liking of the samples made with RZ.
Soybeans fermented using R. delemar contained ammonia, histidine, valine and arginine, and have a correlation with proteolytic bacteria. Valine has a bitter taste and ammonia, which is produced by proteolytic bacteria, has a strong unpleasant odor. This might explain the low taste and overall liking score that samples made with RD received. These findings are in line with a previous study that suggested that the starter culture influences the metabolite profile in tempe more than factors that relate to, e.g., the environment, soybeans and water [37]. Based on the findings of the sensory evaluation, soybeans fermented using R. oligosporus are most preferable.
3.4. Correlation between Chemical and Microbioligal Characteristic with the Sensory Characteristics of Tempe
Several factors that can affect the overall liking of tempe are important to assess when developing a tempe starter with high consumer acceptance. The results of the current study show that several factors affect the overall liking of tempe. These include glutamic acid content, aspartic acid content, texture and taste.
Glutamic and aspartic acid are amino acids responsible for umami taste characteristics and strongly correlate to the overall acceptance of tempe (0.82 and 0.90, respectively). These amino acids are most likely produced by the fungi during fermentation, given that there are no correlations between lactic acid bacteria, yeast and proteolytic bacteria with glutamic and aspartic acids. These findings are in accordance with Handoyo and Morita [38], who reported that R. oligosporus secretes a proteolytic enzyme that hydrolyzes protein into amino acids, which is then used for fungal growth.
The texture of tempe strongly correlates with overall liking (0.95). A desirable tempe texture is characterized by compactness and density in mycelium [39]. The density of the mycelium depends on the ability of the hyphae to penetrate the soybeans. Plant cell walls act as a physical barrier. However, during fermentation, the soybean cell wall is degraded by the fungi. R. oryzae is known to secrete various types of enzymes, including cell wall degrading enzymes such as cellulase, hemicellulose, polygalacturonase, chitinolytic and β-1,3-gluconolytic [18,40].
The last factor affecting the overall liking of tempe is taste. Taste is strongly positively related to the overall liking (0.93). A desirable taste of tempe is savory and mushroom-like. Savory characteristics are related to glutamic and aspartic acids. Consumers were found to dislike characteristics such as overripe taste, bitterness and acidity [41,42].
Overripe flavors are pungent odors that are associated with spoiled products and can cause a loss of appetite [43]. Overripe flavors are thus regarded as non-desirable in tempe. When fermentation is prolonged, proteins are hydrolyzed into smaller components with lower molecular weight and thus become more soluble in water [44]. Overripe flavors are derived from fat hydrolysis and protein degradation, which result in free fatty acids, free amino acids and ammonia [8]. From a nutritional point of view, overripe tempe has higher functionality compared to fresh tempe with regards to antioxidant activity [45,46]. The most dominant water-soluble component of overripe tempe is glutamic acid and amino acids, which can cause bitter taste characteristics. Bitterness might reduce the overall liking among many. However, some who are familiar with overripe tempe use it as a condiment to enhance flavors.
Acidic taste in tempe is most likely related to organic acids produced by LAB and fungi [47,48]. LAB involved in fermentation are identified as Lactobacillus plantarum, Lactobacillus fermentum, Lactobacilus brevis, Lactococcus lactis ssp. lactis and Leuconostoc mesenteroides ssp. mesenteroides [49]. These LAB are known to be able to produce up to 112.15 g/L lactic acid [50]. In addition, R. oryzae and R. delemar also produce lactic acid and fumaric-malic acid, respectively [51]. Lastly, a bitter taste in tempe might be due to the arginine content in tempe [9].
4. Conclusions
The correlation between the chemical and microbiological characteristics and sensory characteristics of fermented soybean products is an important study when developing acceptable fungal-fermented soybean products. The results of this study showed that there is a strong correlation between the glutamic and aspartic content and the overall liking of fermented soybeans. Taste and texture significantly affect the overall liking of fermented soybeans. In addition, soybeans fermented using from local isolates have similar sensory and chemical properties to soybeans fermented using a commercially available starter culture. The findings of the study suggest that chemical analysis, i.e., glutamic and aspartic content, can be used to predict the sensory acceptability of fungal-fermented soybean products.
Author Contributions
Conceptualization, R.W., S., M.J.T.; methodology, R.W., S.; software, D.A.K.; validation, R.W., C.H.; formal analysis, D.A.K., N.L.R., N.R.C., R.D.P., D.A.K., N.L.R., N.R.C., R.D.P.; writing—original draft preparation, R.W.; writing—review and editing, R.W., C.H., M.J.T. All authors have read and agreed to the published version of the manuscript.
Funding
The authors wish to thank DIKTI INDONESIA through PDUPT 2018 with contract number of 137/UN1/DITLIT/DIT-LIT/LT/2018 for financially support the project and World Class Professor Program, Ministry of Education and Culture of Indonesia 2021 for the publication.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Castanheira, É.G.; Freire, F. Greenhouse Gas Assessment of Soybean Production: Implications of Land Use Change and Different Cultivation Systems. J. Clean. Prod. 2013, 54, 49–60. [Google Scholar] [CrossRef]
- Wiedemann, S.; McGahan, E.; Murphy, C.; Yan, M.-J.; Henry, B.; Thoma, G.; Ledgard, S. Environmental Impacts and Resource Use of Australian Beef and Lamb Exported to the USA Determined Using Life Cycle Assessment. J. Clean. Prod. 2015, 94, 67–75. [Google Scholar] [CrossRef]
- Liu, K. Chemistry and Nutritional Value of Soybean Components. In Soybeans: Chemistry, Technology, and Utilization; Springer: Boston, MA, USA, 1997. [Google Scholar]
- Hassan, S. Soybean, Nutrition and Health. In Soybean—Bio-Active Compounds; El-Shemy, H.A., Ed.; InTech Open Access Publisher: Rijeka, Croatia, 2013. [Google Scholar]
- Adeyemo, S.M.; Onilude, A.A. Enzymatic Reduction of Anti-Nutritional Factors in Fermenting Soybeans by Lactobacillus Plantarum Isolates from Fermenting Cereals. Niger. Food J. 2013, 31, 84–90. [Google Scholar] [CrossRef]
- Samtiya, M.; Aluko, R.E.; Dhewa, T. Plant Food Anti-Nutritional Factors and Their Reduction Strategies: An Overview. Food Prod. Process. Nutr. 2020, 2, 6. [Google Scholar] [CrossRef]
- Hubert, J.; Berger, M.; Dayde, J. Effects of Fermentation on the Phytochemical Composition and Antioxidant Properties of Soy Germ. Food Chem. 2008, 109, 709–721. [Google Scholar] [CrossRef] [PubMed]
- Ahnan-Winarno, A.D.; Cordeiro, L.; Winarno, F.G.; Gibbons, J.; Xiao, H. Tempeh: A Semicentennial Review on Its Health Benefits, Fermentation, Safety, Processing, Sustainability, and Affordability. Compr. Rev. Food Sci. Food Saf. 2021, 20, 1717–1767. [Google Scholar] [CrossRef] [PubMed]
- Tseng, Y.-H.; Lee, Y.-L.; Li, R.-C.; Mau, J.-L. Non-Volatile Flavour Components of Ganoderma Tsugae. Food Chem. 2005, 90, 409–415. [Google Scholar] [CrossRef]
- Bavaro, S.L.; Susca, A.; Frisvad, J.C.; Tufariello, M.; Chytiri, A.; Perrone, G.; Mita, G.; Logrieco, A.F.; Bleve, G. Isolation, Characterization, and Selection of Molds Associated to Fermented Black Table Olives. Front. Microbiol. 2017, 8, 1356. [Google Scholar] [CrossRef]
- Hartanti, A.T.; Rahayu, G.; Hidayat, I. Rhizopus Species from Fresh Tempeh Collected from Several Regions in Indonesia. Hayati J. Biosci. 2015, 22, 136–142. [Google Scholar] [CrossRef]
- Samson, R.A.; Hoekstra, E.S.; Frisvad, J.C.; Filtenborg, O. Introduction to Foodborne Fungi, 4th ed.; Centraalbureau voor Schimmelcultures: Baarn, The Netherlands, 1995. [Google Scholar]
- Azeke, M.A.; Fretzdorff, B.; Buening-Pfaue, H.; Betsche, T. Comparative Effect of Boiling and Solid Substrate Fermentation Using the Tempeh Fungus (Rhizopus Oligosporus) on the Flatulence Potential of African Yambean (Sphenostylis Stenocarpa L.) Seeds. Food Chem. 2007, 103, 1420–1425. [Google Scholar] [CrossRef]
- Nurrahman, N.; Astusti, M.; Suparmo; Soesatyo, M.H.N.E. Pertumbuhan Jamur, Sifat Organoleptik Dan Aktivitas Antioksidan Tempe Kedelai Hitam Yang Diproduksi Dengan Berbagai Jenis Inokulum. Agritech 2012, 32, 60–65. [Google Scholar]
- Parris, N.; Foglia, T.A. Simplified Alcoholic Extraction Procedure for Ammonia in Meat Tissue. J. Agric. Food Chem. 1983, 31, 887–889. [Google Scholar] [CrossRef]
- Meilgaard, M.; Civille, G.V.; Carr, B.T. Sensory Evaluation Techniques, 4th ed.; CRC Press: London, UK, 2006; ISBN 9788578110796. [Google Scholar]
- Tamang, J.P.; Kort, R.; Hutkins, R.; Cotter, P.D.; Endo, A.; Liu, S.Q.; Mayo, B. Fermented Foods in a Global Age: East Meets West. Compr. Rev. Food Sci. Food Saf. 2020, 19, 184–217. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, B.; Ray, R.R. Current Commercial Perspective of Rhizopus Oryzae: A Review. J. Appl. Sci. 2011, 11, 2470–2486. [Google Scholar] [CrossRef]
- Pedizzi, C.; Lema, J.M.; Carballa, M. Enhancing Thermophilic Co-Digestion of Nitrogen-Rich Substrates by Air Side-Stream Stripping. Bioresour. Technol. 2017, 241, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Varzakas, T.H.; Pyle, D.L.; Niranjan, K. Mycelial penetration and enzymic diffusion on soybean tempe. In Advances in Solid State Fermentation; Roussos, S., Lonsane, B.K., Raimbault, M., Gonzalez, G.V., Eds.; Springer: Berlin, Germany, 1997; pp. 59–70. [Google Scholar]
- Higasa, S.; Negishi, Y.; Aoyagi, Y.; Sugahara, T. Changes in Free Amino Acids of Tempe during Preparation with Velvet Beans (Mucuna Pruriens). J. Jpn. Soc. Food Sci. Technol.—Nippon Shokuhin Kagaku Kogaku Kaishi 1996, 43, 188–193. [Google Scholar] [CrossRef]
- Schindler, A.; Dunkel, A.; Staehler, F.; Backes, M.; Ley, J.; Meyerhof, W.; Hofmann, T. Discovery of Salt Taste Enhancing Arginyl Dipeptides in Protein Digests and Fermented Fish Sauces by Means of a Sensomics Approach. J. Agric. Food Chem. 2011, 59, 12578–12588. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Shiga, K.; Kodama, Y.; Imamura, M.; Uchida, R.; Obata, A.; Fukusaki, E. Analysis of the Correlation between Dipeptides and Taste Differences among Soy Sauces by Using Metabolomics-Based Component Profiling. J. Biosci. Bioeng. 2014, 118, 56–63. [Google Scholar] [CrossRef]
- Arai, S.; Noguchi, M.; Kurosawa, S.; Kato, H.; Fujimaki, M. Applying Proteolytic Enzymes on Soybean. 6. Deodorization Effect of Aspergillopeptidase-A and Debittering Effect of Aspergillus Acid Carboxypeptidase. J. Food Sci. 1970, 35, 392–395. [Google Scholar] [CrossRef]
- Sparringa, R.A.; Owens, J.D. Protein Utilization during Soybean Tempe Fermentation. J. Agric. Food Chem. 1999, 47, 4375–4378. [Google Scholar] [CrossRef] [PubMed]
- Visessanguan, W.; Benjakul, S.; Potachareon, W.; Panya, A.; Riebroy, S. Accelerated Proteolysis of Soy Proteins during Fermentation of Thua-nao Inoculated with Bacillus Subtilis. J. Food Biochem. 2005, 29, 349–366. [Google Scholar] [CrossRef]
- Shurtleff, W.; Aoyagi, A. The Book of Tempeh; Soyinfo Center: New York, NY, USA, 2001. [Google Scholar]
- Santhirasegaram, V.; George, D.S.; Anthony, K.K.; Kaur, H.; Singh, B.; Saruan, N.M.; Razali, Z.; Somasundram, C. Effects of Soybean Processing and Packaging on the Quality of Commonly Consumed Local Delicacy Tempe. J. Food Qual. 2016, 39, 675–684. [Google Scholar] [CrossRef]
- Barus, T.; Suwanto, A.; Wahyudi, A.T.; Wijaya, H. Role of Bacteria in Tempe Bitter Taste Formation; Microbiological and Molecular Biological Analysis Based on 16S RRNA Gene. Microbiol. Indones. 2008, 2, 17–21. [Google Scholar] [CrossRef]
- Efriwati, S.A.; Rahayu, G.; Nuraida, L. Population Dynamics of Yeasts and Lactic Acid Bacteria (LAB) during Tempeh Production. Hayati J. Biosci. 2013, 20, 57–64. [Google Scholar] [CrossRef]
- Ashenafi, M.; Busse, M. Growth of Bacillus Cereus in Fermenting Tempeh Made from Various Beans and Its Inhibition by Lactobacillus Plantarum. J. Appl. Microbiol. 1991, 70, 329–333. [Google Scholar]
- Arroyo-López, F.N.; Querol, A.; Bautista-Gallego, J.; Garrido-Fernández, A. Role of Yeasts in Table Olive Production. Int. J. Food Microbiol. 2008, 128, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Cheirsilp, B.; Shimizu, H.; Shioya, S. Enhanced Kefiran Production by Mixed Culture of Lactobacillus Kefiranofaciens and Saccharomyces Cerevisiae. J. Biotechnol. 2003, 100, 43–53. [Google Scholar] [CrossRef]
- Moreno, M.R.F.; Leisner, J.J.; Tee, L.K.; Ley, C.; Radu, S.; Rusul, G.; Vancanneyt, M.; De Vuyst, L. Microbial Analysis of Malaysian Tempeh, and Characterization of Two Bacteriocins Produced by Isolates of Enterococcus Faecium. J. Appl. Microbiol. 2002, 92, 147–157. [Google Scholar] [CrossRef]
- Nout, M.J.R.; Kiers, J.L. Tempe Fermentation, Innovation and Functionality: Update into the Third Millenium. J. Appl. Microbiol. 2005, 98, 789–805. [Google Scholar] [CrossRef]
- Hachmeister, K.A.; Fung, D. Tempeh: A Mold-Modified Indigenous Fermented Food Made from Soybeans and/or Cereal Grains. Crit. Rev. Microbiol. 1993, 19, 137–188. [Google Scholar] [CrossRef]
- Kadar, A.D.; Astawan, M.; Putri, S.P.; Fukusaki, E. Metabolomics Based Study of the Effect of Raw Materials to the End Product of Tempe—an Indonesian Fermented Soybean. Metabolites 2020, 10, 367. [Google Scholar] [CrossRef]
- Handoyo, T.; Morita, N. Structural and Functional Properties of Fermented Soybean (Tempeh) by Using Rhizopus Oligosporus. Int. J. Food Prop. 2006, 9, 347–355. [Google Scholar] [CrossRef]
- Wikandari, R.; Utami, T.A.N.; Hasniah, N.; Sardjono. Chemical, Nutritional, Physical and Sensory Characterization of Tempe Made from Various Underutilized Legumes. Pakistan J. Nutr. 2020, 19, 179–190. [Google Scholar] [CrossRef]
- Battaglia, E.; Benoit, I.; van den Brink, J.; Wiebenga, A.; Coutinho, P.M.; Henrissat, B.; de Vries, R.P. Carbohydrate-Active Enzymes from the Zygomycete Fungus Rhizopus Oryzae: A Highly Specialized Approach to Carbohydrate Degradation Depicted at Genome Level. BMC Genom. 2011, 12, 38. [Google Scholar] [CrossRef]
- Radiati, A.; Sumarto, S. Analisis Sifat Fisik, Sifat Organoleptik, dan Kandungan Gizi Pada Produk Tempe dari Kacang Non-Kedelai. J. Apl. Teknol. Pangan 2015, 5, 16–22. [Google Scholar]
- Affandi, D.R.; Amanto, B.S.; Ishartani, D.; Nurani, S. Karakteristik Sensoris Tempe Koro Glinding (Phaseolus Lunatus) Pada Berbagai Waktu Fermentasi. In Proceedings of the Peranan Sumber Daya Pertanian, Perkebunan, dan Peternakan dalam Mendukung Ketahanan Pangan Nasional, Fakultas Pertanian UNS, Surakarta, Indonesia, 30 March 2017; pp. 794–799. [Google Scholar]
- Dewi, M.; Tirtaningtyas, P.; Raissha, T.; Kartika, E.; Hanny, C.; Mutukumira, A.N. Sensory Characteristics of Seasoning Powders from Overripe Tempeh, a Solid State Fermented Soybean. Procedia Chem. 2015, 14, 263–269. [Google Scholar] [CrossRef]
- Utami, R.; Wijaya, C.H.; Lioe, H.N. Taste of Water-Soluble Extracts Obtained from Over-Fermented Tempe. Int. J. Food Prop. 2016, 19, 2063–2073. [Google Scholar] [CrossRef]
- Athaillah, Z.A.; Muzdalifah, D.; Lestari, A.; Fitria, A.; Udin, L.Z.; Artanti, N.; Lioe, H.N. Phenolic Compound Profile and Functionality of Aqueous Overripe Tempe Extracts. Curr. Res. Nutr. Food Sci. 2019, 7, 382–392. [Google Scholar] [CrossRef]
- Starzyńska-Janiszewska, A.; Duliński, R.; Stodolak, B.; Mickowska, B.; Wikiera, A. Prolonged Tempe-Type Fermentation in Order to Improve Bioactive Potential and Nutritional Parameters of Quinoa Seeds. J. Cereal Sci. 2016, 71, 116–121. [Google Scholar] [CrossRef]
- Spanier, A.M.; Shahidi, F.; Parliment, T.H.; Mussinan, C.; Ho, C.-T.; Contis, E.T. Food Flavors and Chemistry: Advances of the New Millennium; The Royal Society of Chemistry: Cambridge, UK, 2007. [Google Scholar]
- Soccol, C.R.; Stonoga, V.I.; Raimbault, M. Production of L-Lactic Acid by Rhizopus Species. World J. Microbiol. Biotechnol. 1994, 10, 433–435. [Google Scholar] [CrossRef] [PubMed]
- Suliantari; Suryaatmadja, S.L.; Kusumaningrum, H. Microbial Content and Diversity in Several Tempe from Bogor Areas. In Proceedings of the Prosiding Seminar Hasil-Hasil PPM IPB, Bogor, Indonesia, 1–2 December 2015; pp. 229–237. [Google Scholar]
- de Oliveira, P.M.; Santos, L.P.; Coelho, L.F.; Avila Neto, P.M.; Sass, D.C.; Contiero, J. Production of L (+) Lactic Acid by Lactobacillus Casei Ke11: Fed Batch Fermentation Strategies. Fermentation 2021, 7, 151. [Google Scholar] [CrossRef]
- Hartanti, A.T.; Raharjo, A.; Gunawan, A.W. Rhizopus Rotting on Agricultural Products in Jakarta. Hayati J. Biosci. 2020, 27, 37–44. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).