Indigenous Yeast, Lactic Acid Bacteria, and Acetic Acid Bacteria from Cocoa Bean Fermentation in Indonesia Can Inhibit Fungal-Growth-Producing Mycotoxins
Abstract
:1. Introduction
2. Materials and Methods
2.1. Media and Equipment
2.2. Sample and Microorganisms
2.3. Preparation of Microorganisms for Cocoa Bean Fermentation
2.4. Fermentation of Cocoa Beans
2.5. Extraction and Analysis of Ochratoxin A
2.6. Statistical Analysis
3. Results
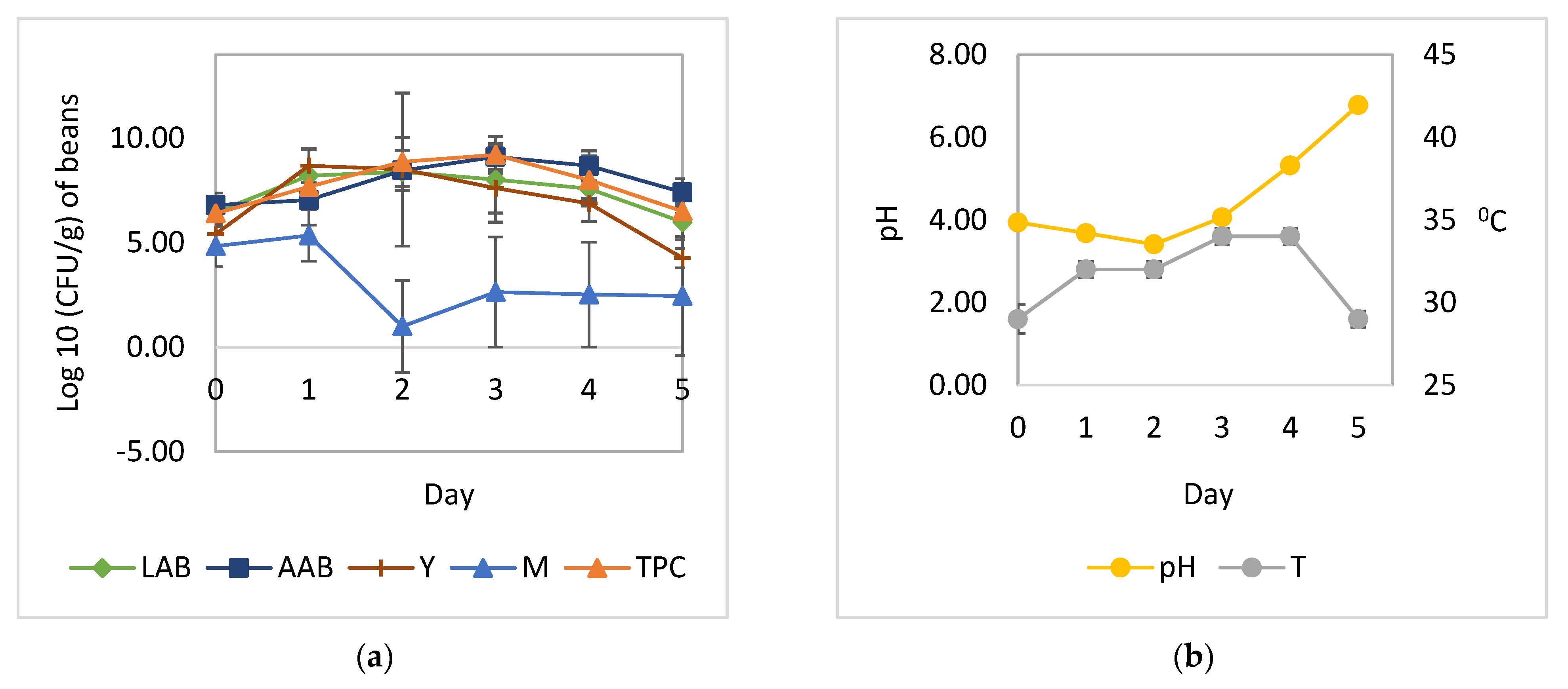
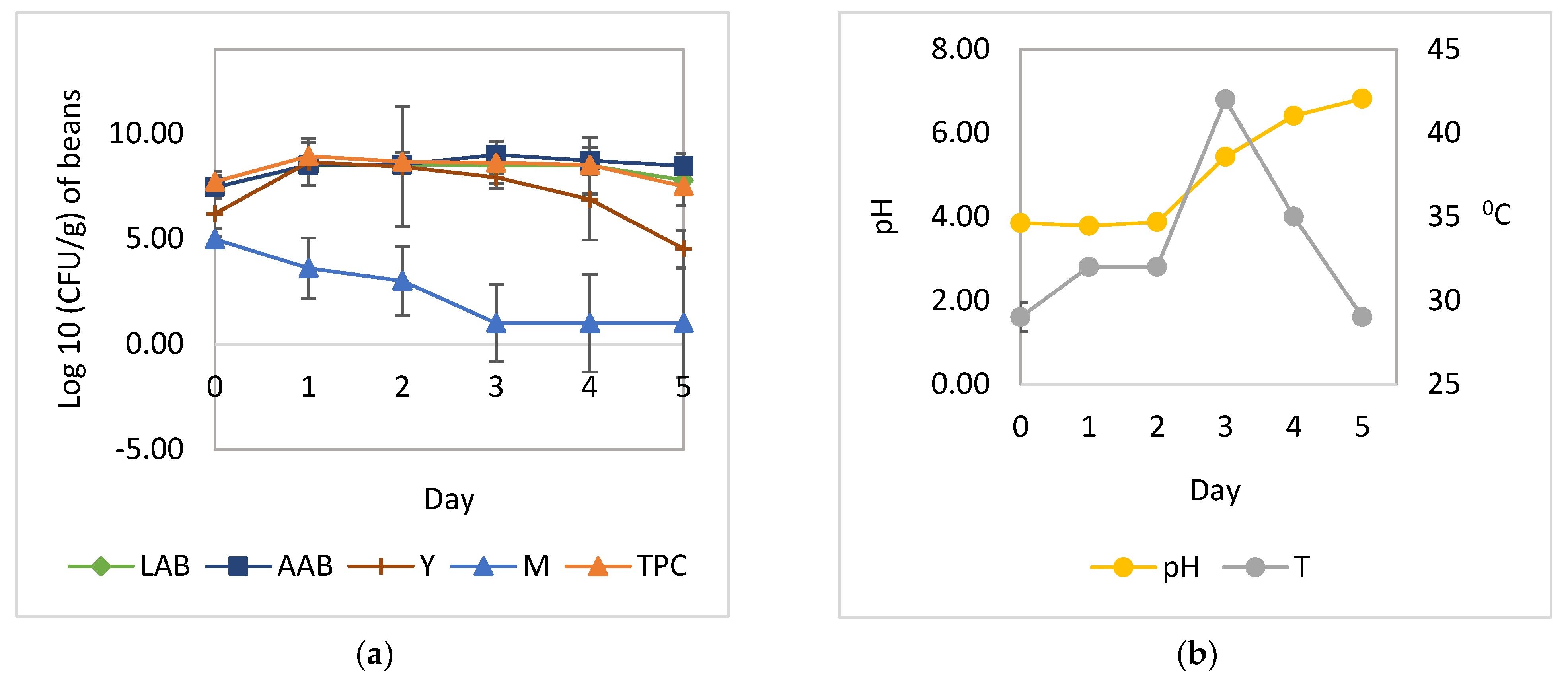
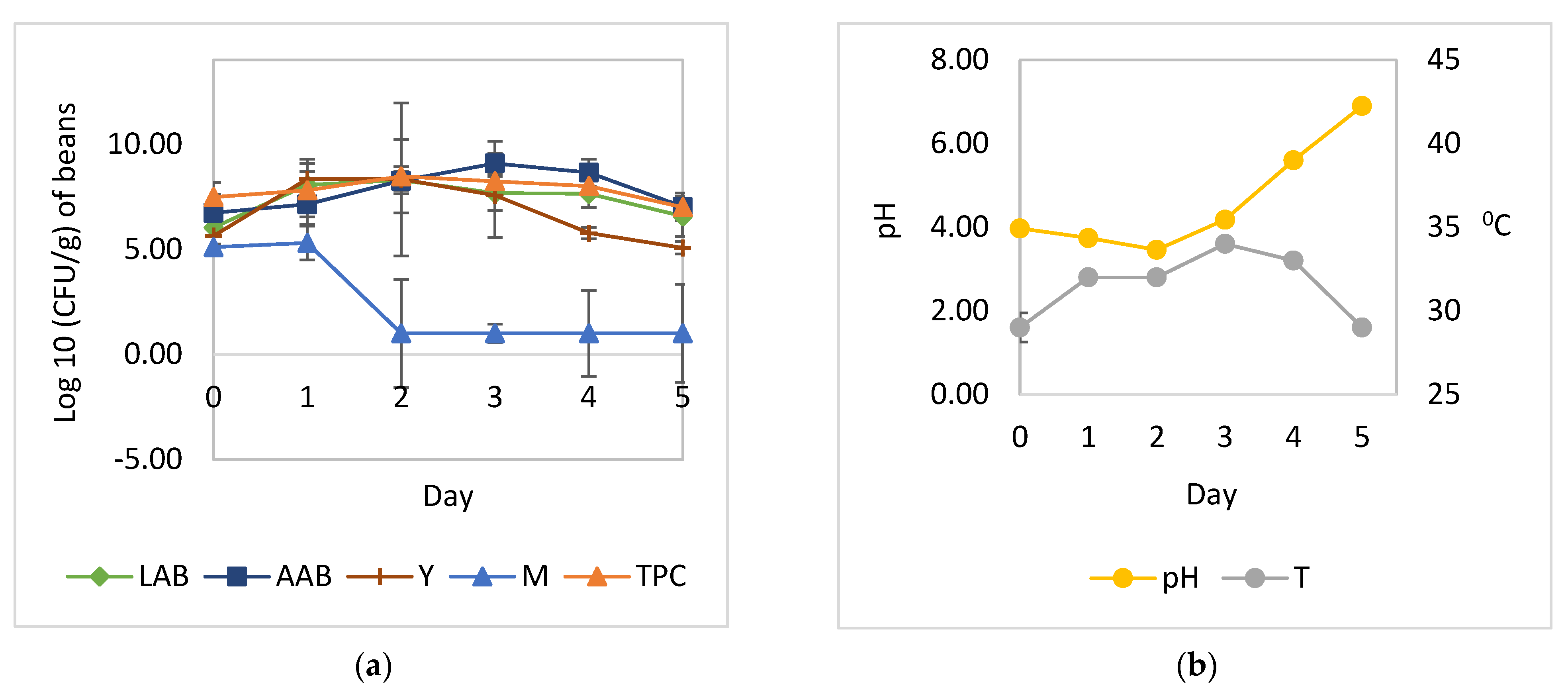
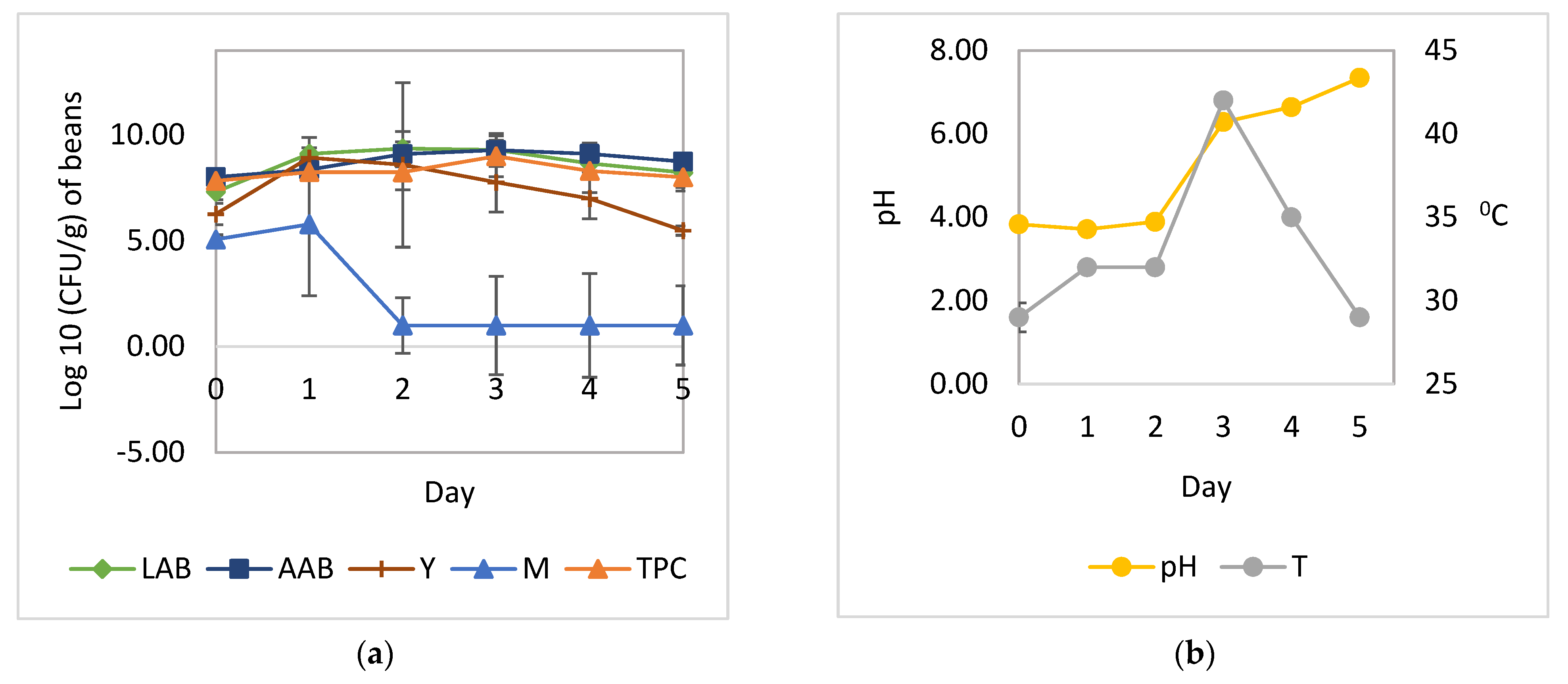
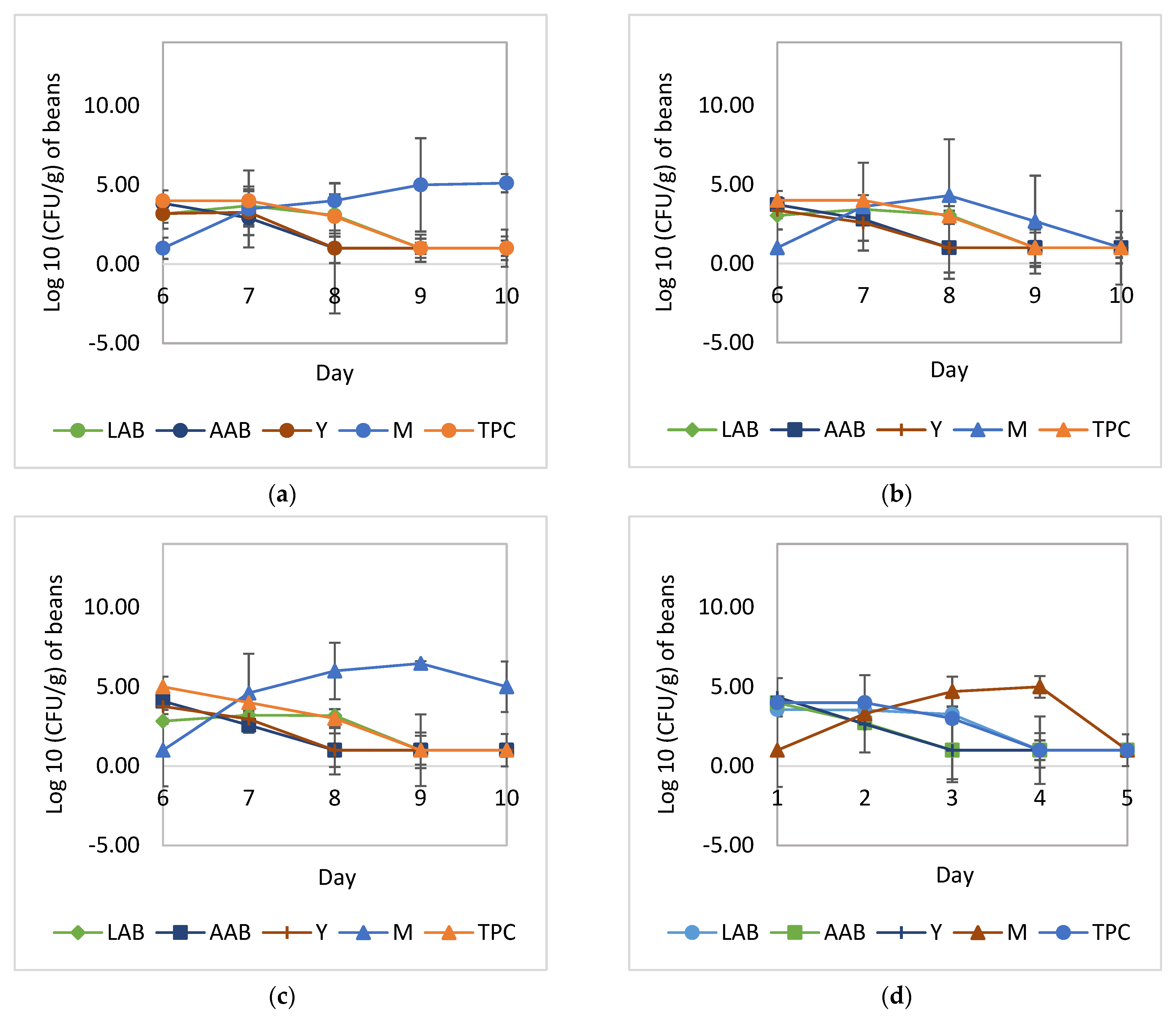
3.1. Cocoa Bean Fermentation
3.2. Drying of Fermented Cocoa Beans
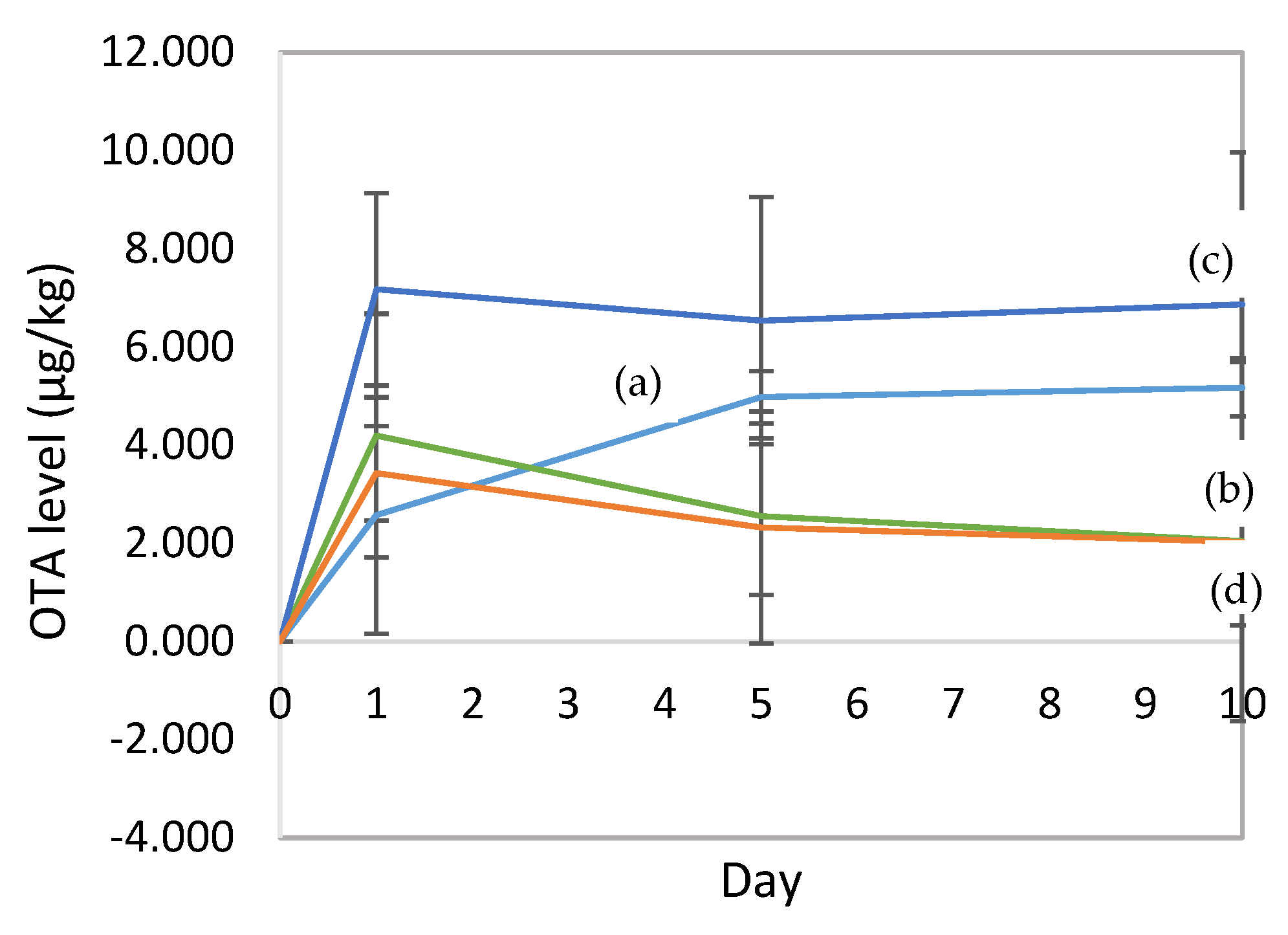
3.3. Ochratoxin A Level in Fermented Cocoa Beans
4. Discussion
5. Conclusions
6. Patents
- Patent: Dry Starter Manufacturing Process of L. plantarum HLM-15 for Mycotoxin-Producing Fungus Control, 2017 (IDS000001851) (in Indonesian)
- Patent Cocoa Bean Fermentation Process with the Addition of Starter L. plantarum HL-15 to Inhibit Fungal Growth, 2019 (IDS000002554) (in Indonesian)
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bortolini, C.; Patrone, V.; Puglisi, E.; Morelli, L. Detailed analyses of the bacterial populations in processed cocoa beans of different geographic origin, subject to varied fermentation conditions. Int. J. Food Microbiol. 2016, 236, 98–106. [Google Scholar] [CrossRef]
- Hamdouche, Y.; Meile, J.C.; Lebrun, M.; Guehi, T.; Boulanger, R.; Teyssier, C.; Montet, D. Impact of turning, pod storage and fermentation time on microbial ecology and volatile composition of cocoa beans. Food Res. Int. 2019, 119, 477–491. [Google Scholar] [CrossRef]
- Ramos, S.; Salazar, M.; Nascimento, L.; Carazzolle, M.; Pereira, G.; Delforno, T.; Nascimento, M.; de Aleluia, T.; Celeghini, R.; Efraim, P. Influence of pulp on the microbial diversity during cupuassu fermentation. Int. J. Food Microbiol. 2020, 318, 108465. [Google Scholar] [CrossRef]
- Lima, C.O.; Vaz, A.B.M.; De Castro, G.M.; Lobo, F.; Solar, R.; Rodrigues, C.; Martins Pinto, L.R.; Vandenberghe, L.; Pereira, G.; Miúra da Costa, A.; et al. Integrating microbial metagenomics and physicochemical parameters and a new perspective on starter culture for fine cocoa fermentation. Food Microbiol. 2021, 93, 103608. [Google Scholar] [CrossRef]
- Miguel, M.G.; da Castro, L.V.; Efraim, P.; Santos, C.; Lima, N.; Schwan, R.F. Cocoa fermentation: Microbial identification by MALDI-TOF MS, and sensory evaluation of produced chocolate. LWT Food Sci. Technol. 2017, 77, 362–369. [Google Scholar] [CrossRef] [Green Version]
- Ho, V.T.T.; Fleet, G.H.; Zhao, J. Unravelling the contribution of lactic acid bacteria and acetic acid bacteria to cocoa fermentation using inoculated organisms. Int. J. Food Microbiol. 2018, 279, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Taniwaki, M.H.; Pitt, J.I.; Magan, N. Aspergillus species and mycotoxins: Occurrence and importance in major food commodities. Curr. Opin. Food Sci. 2018, 23, 38–43. [Google Scholar] [CrossRef] [Green Version]
- Ruggirello, M.; Nucera, D.; Cannoni, M.; Peraino, A.; Rosso, F.; Fontana, M.; Cocolin, L.; Dolci, P. Antifungal activity of yeasts and lactic acid bacteria isolated from cocoa bean fermentations. Food Res. Int. 2019, 115, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Akinfala, T.O.; Houbraken, J.; Sulyok, M.; Adedeji, A.R.; Odebode, A.C.; Krska, R.; Ezekiel, C.N. Moulds and their secondary metabolites associated with the fermentation and storage of two cocoa bean hybrids in Nigeria. Int. J. Food Microbiol. 2020, 316, 108490. [Google Scholar] [CrossRef] [PubMed]
- Cabañes, F.J.; Bragulat, M.R. Black aspergilli and ochratoxin A-producing species in foods. Curr. Opin. Food Sci. 2018, 23, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Alves da Silva, S.; Fonseca Alvarenga Pereira, R.G.; de Azevedo Lira, N.; Micotti da Glória, E.; Chalfoun, S.M.; Batista, L.R. Fungi associated to beans infested with coffee berry borer and the risk of ochratoxin A. Food Control. 2020, 113, 107204. [Google Scholar] [CrossRef]
- Da Silva, A.R.P.; Fungaro, M.H.P.; Silva, J.J.; Martins, L.M.; Taniwaki, M.H.; Iamanaka, B.T. Ochratoxin A and related fungi in Brazilian black pepper (Piper nigrum L.). Food Res. Int. 2021, 142, 110207. [Google Scholar] [CrossRef] [PubMed]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef] [PubMed]
- Makhuvele, R.; Naidu, K.; Gbashi, S.; Thipe, V.C.; Adebo, O.A.; Njobeh, P.B. The use of plant extracts and their phytochemicals for control of toxigenic fungi and mycotoxins. Heliyon 2020, 6, e05291. [Google Scholar] [CrossRef]
- Mousavi Khaneghah, A.; Hashemi Moosavi, M.; Oliveira, C.A.F.; Vanin, F.; Sant’Ana, A.S. Electron beam irradiation to reduce the mycotoxin and microbial contaminations of cereal-based products: An overview. Food Chem. Toxicol. 2020, 143, 111557. [Google Scholar] [CrossRef]
- Shehata, M.G.; Badr, A.N.; El Sohaimy, S.A.; Asker, D.; Awad, T.S. Characterization of antifungal metabolites produced by novel lactic acid bacterium and their potential application as food biopreservatives. Ann. Agric. Sci. 2019, 64, 71–78. [Google Scholar] [CrossRef]
- Gavahian, M.; Mathad, G.N.; Oliveira, C.A.F.; Mousavi Khaneghah, A. Combinations of emerging technologies with fermentation: Interaction effects for detoxification of mycotoxins? Food Res. Int. 2021, 141, 110104. [Google Scholar] [CrossRef] [PubMed]
- Du, G.; Liu, L.; Guo, Q.; Cui, Y.; Chen, H.; Yuan, Y.; Wang, Z.; Gao, Z.; Sheng, Q.; Yue, T. Microbial community diversity associated with Tibetan kefir grains and its detoxification of Ochratoxin A during fermentation. Food Microbiol. 2021, 99, 103803. [Google Scholar] [CrossRef]
- Romanens, E.; Freimüller Leischtfeld, S.; Volland, A.; Stevens, M.; Krähenmann, U.; Isele, D.; Fischer, B.; Meile, L.; Miescher Schwenninger, S. Screening of lactic acid bacteria and yeast strains to select adapted anti-fungal co-cultures for cocoa bean fermentation. Int. J. Food Microbiol. 2019, 290, 262–272. [Google Scholar] [CrossRef] [PubMed]
- Sadiq, F.A.; Yan, B.; Tian, F.; Zhao, J.; Zhang, H.; Chen, W. Lactic Acid Bacteria as Antifungal and Anti-Mycotoxigenic Agents: A Comprehensive Review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1403–1436. [Google Scholar] [CrossRef] [Green Version]
- Marwati, T.; Khusna, R.N.B.; Djaafar, T.F.; Rahayu, E.S. Inhibition Growth of Mycotoxin Producing Fungi by Lactic Acid Bacteria Isolated from Fermented Cocoa Bean (Theobroma cacao L.) in Indonesia. In Proceedings of the 15th ASEAN Conference on Food Science and Technology, Ho Chi Minh City, Vietnam, 14–17 November 2017; Volume 2, pp. 295–302. [Google Scholar]
- Djaafar, T.F.; Marwati, T.; Rahayu, E.S. Isolation and Identification of Yeast and Acetic Acid Bacteria from Cocoa Bean (Theobroma cacao Linn.) Fermentation in Gunung Kidul Regency, Yogyakarta, Indonesia Titiek. In Proceedings of the 15th ASEAN Conference on Food Science and Technology, Ho Chi Minh City, Vietnam, 14–17 November 2017; Volume 1, pp. 189–194. [Google Scholar]
- Nugroho, A.D.; Setyabudi, F.M.C.S.; Salleh, B.; Rahayu, E.S. Ochratoxingenic Black Aspergilli Isolated from Dried Agricultural Products in Yogyakarta, Indonesia. J. Food Sci. Biotechnol. 2013, 3, 472–480. [Google Scholar]
- da Silva Duarte, V.; Carlot, M.; Pakroo, S.; Tarrah, A.; Lombardi, A.; Santiago, H.; Corich, V.; Giacomini, A. Comparative evaluation of cheese whey microbial composition from four Italian cheese factories by viable counts and 16S rRNA gene amplicon sequencing. Int. Dairy J. 2020, 104, 104656. [Google Scholar] [CrossRef]
- De Vuyst, L.; Weckx, S. The cocoa bean fermentation process: From ecosystem analysis to starter culture development. J. Appl. Microbiol. 2016, 121, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Fossi, B.; Ngah, B.; Nchanji, G.; Wanji, S.; Ndjouenkeu, R. Lactobacilli Cultures against Ochratoxin A Producing Moulds Isolated from Cocoa in the South West Region of Cameroon. Br. Microbiol. Res. J. 2016, 15, 1–16. [Google Scholar] [CrossRef]
- Santos, D.S.; Rezende, R.P.; Dos Santos, T.F.; Marques, E.D.; Ferreira, A.C.; e Silva, A.B.; Romano, C.C.; da Cruz Santos, D.W.; Dias, J.C.; Bisneto, J.D. Fermentation in fine cocoa type Scavina: Change in standard quality as the effect of use of starters yeast in fermentation. Food Chem. 2020, 328, 7–12. [Google Scholar] [CrossRef]
- Essia Ngang, J.J.; Yadang, G.; Sado Kamdem, S.L.; Kouebou, C.P.; Youte Fanche, S.A.; Tsochi Kougan, D.L.; Tsoungui, A.; Etoa, F.X. Antifungal properties of selected lactic acid bacteria and application in the biological control of ochratoxin A producing fungi during cocoa fermentation. Biocontrol Sci. Technol. 2015, 25, 245–259. [Google Scholar] [CrossRef]
- Ouattara, D.H.; Ouattara, H.G.; Goualie, B.G.; Kouame, L.M.; Niamke, S.L. Biochemical and functional properties of lactic acid bacteria isolated from Ivorian cocoa fermenting beans. J. Appl. Biosci. 2014, 77, 6489. [Google Scholar] [CrossRef]
- Schwan, R.F.; Wheals, A.E. The microbiology of cocoa fermentation and its role in chocolate quality. Crit. Rev. Food Sci. Nutr. 2004, 44, 205–221. [Google Scholar] [CrossRef] [PubMed]
- Blagojev, N.; Škrinjar, M.; Vesković-Moračanin, S.; Šošo, V. Control of mould growth and mycotoxin production by lactic acid bacteria metabolites. Rom. Biotechnol. Lett. 2012, 17, 7219–7226. [Google Scholar]
- Copetti, M.V.; Iamanaka, B.T.; Mororó, R.C.; Pereira, J.L.; Frisvad, J.C.; Taniwaki, M.H. The effect of cocoa fermentation and weak organic acids on growth and ochratoxin A production by Aspergillus species. Int. J. Food Microbiol. 2012, 155, 158–164. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahayu, E.S.; Triyadi, R.; Khusna, R.N.B.; Djaafar, T.F.; Utami, T.; Marwati, T.; Hatmi, R.U. Indigenous Yeast, Lactic Acid Bacteria, and Acetic Acid Bacteria from Cocoa Bean Fermentation in Indonesia Can Inhibit Fungal-Growth-Producing Mycotoxins. Fermentation 2021, 7, 192. https://doi.org/10.3390/fermentation7030192
Rahayu ES, Triyadi R, Khusna RNB, Djaafar TF, Utami T, Marwati T, Hatmi RU. Indigenous Yeast, Lactic Acid Bacteria, and Acetic Acid Bacteria from Cocoa Bean Fermentation in Indonesia Can Inhibit Fungal-Growth-Producing Mycotoxins. Fermentation. 2021; 7(3):192. https://doi.org/10.3390/fermentation7030192
Chicago/Turabian StyleRahayu, Endang Sutriswati, Rokhmat Triyadi, Rosyida N. B. Khusna, Titiek Farianti Djaafar, Tyas Utami, Tri Marwati, and Retno Utami Hatmi. 2021. "Indigenous Yeast, Lactic Acid Bacteria, and Acetic Acid Bacteria from Cocoa Bean Fermentation in Indonesia Can Inhibit Fungal-Growth-Producing Mycotoxins" Fermentation 7, no. 3: 192. https://doi.org/10.3390/fermentation7030192
APA StyleRahayu, E. S., Triyadi, R., Khusna, R. N. B., Djaafar, T. F., Utami, T., Marwati, T., & Hatmi, R. U. (2021). Indigenous Yeast, Lactic Acid Bacteria, and Acetic Acid Bacteria from Cocoa Bean Fermentation in Indonesia Can Inhibit Fungal-Growth-Producing Mycotoxins. Fermentation, 7(3), 192. https://doi.org/10.3390/fermentation7030192




