The Use of Life Cycle Assessment in the Support of the Development of Fungal Food Products from Surplus Bread
Abstract
:1. Introduction
2. Materials and Methods
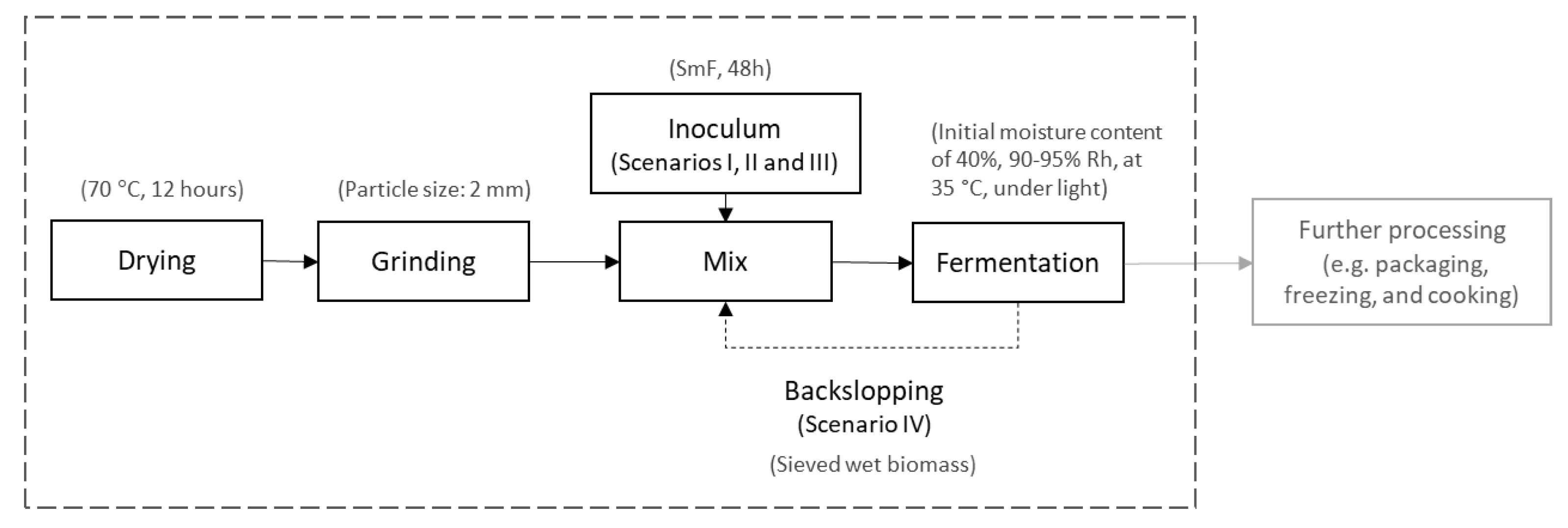
2.1. Process Description
2.1.1. Substrate and Fungus
2.1.2. Design of the Inoculation
2.1.3. Solid State Fermentation
2.2. Analyses
2.3. Statistical Analysis
2.4. Life Cycle Assessment
3. Results
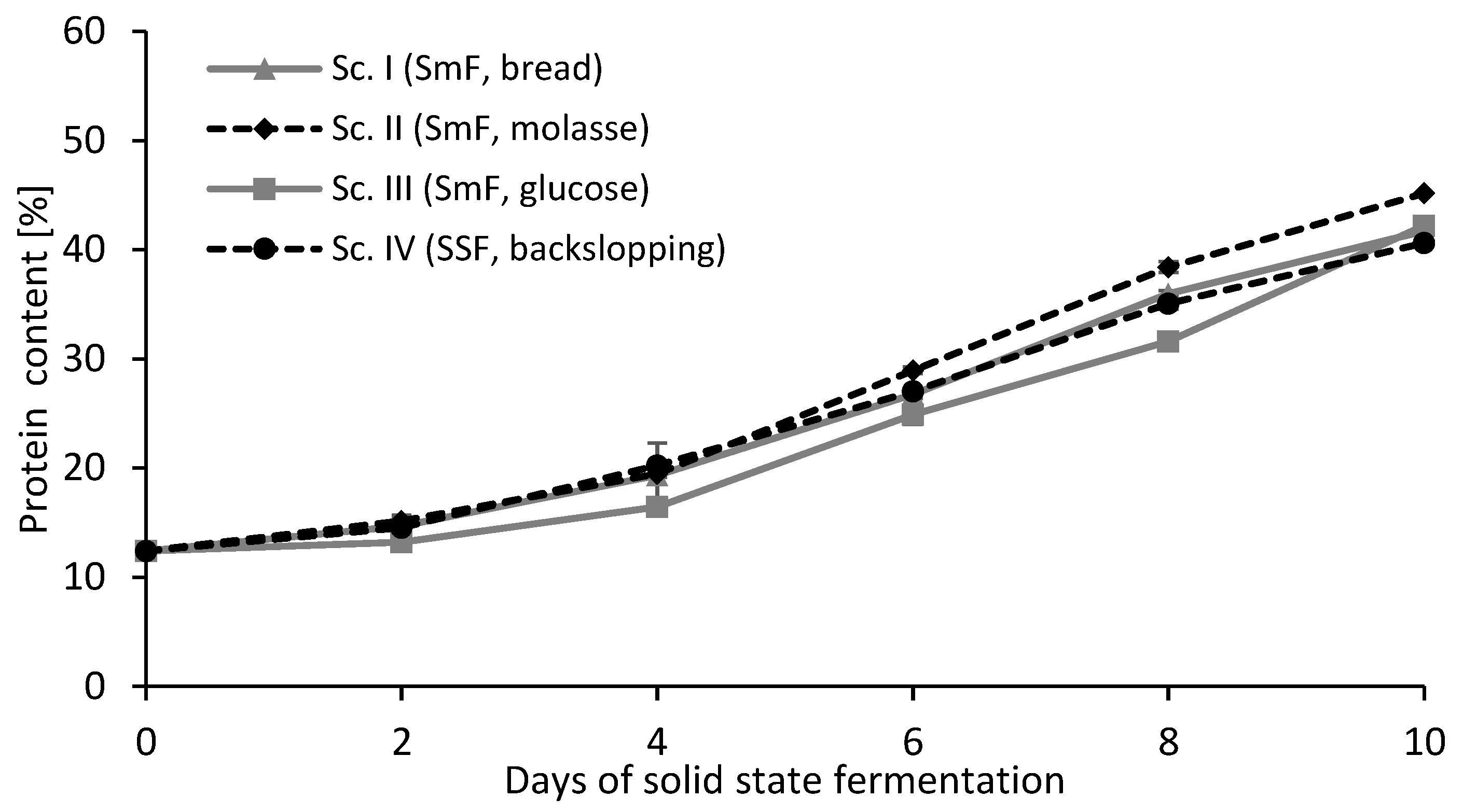
3.1. Experimental Results
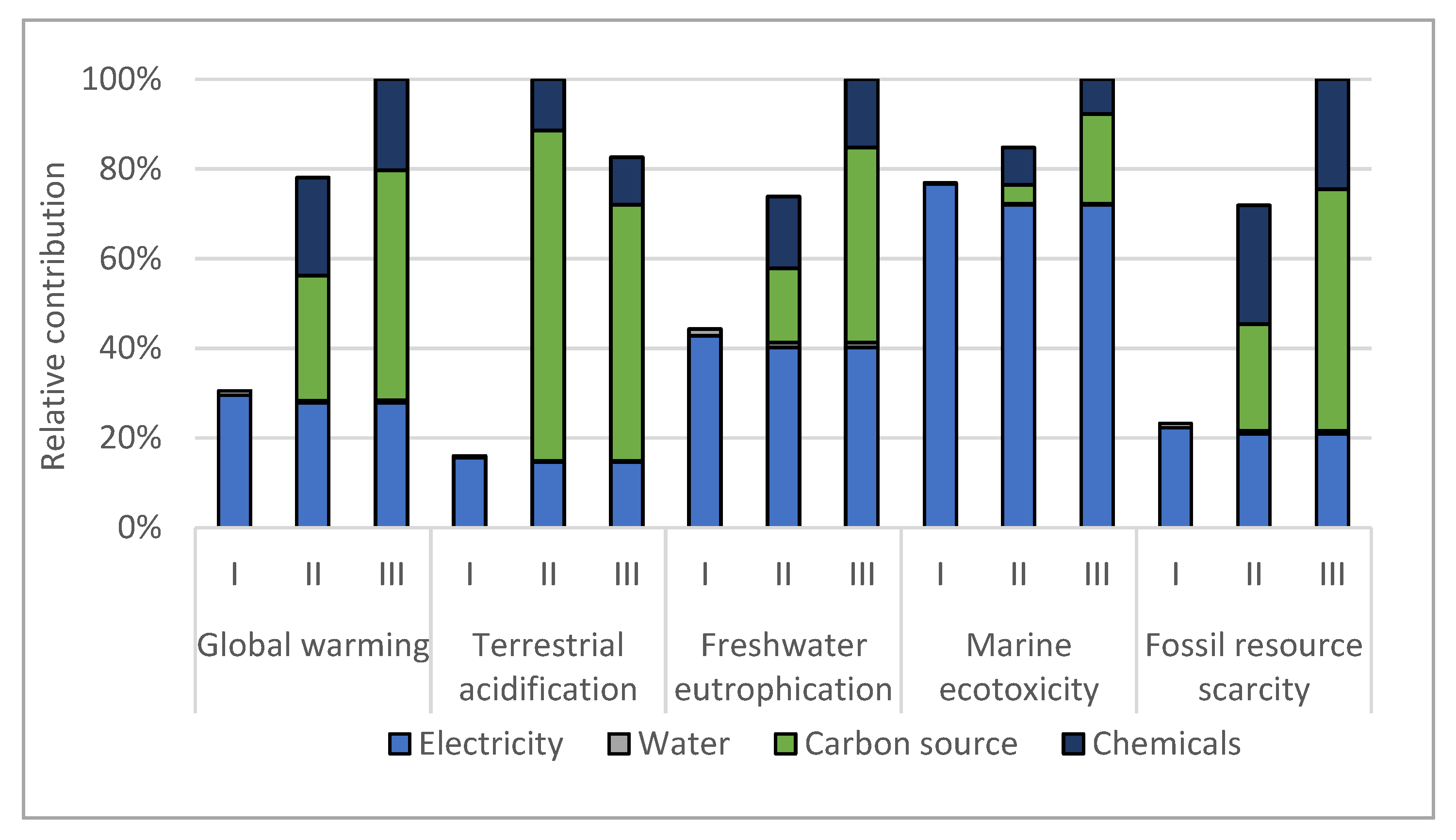
3.2. Life Cycle Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- FAO. The State of Food and Agriculture 2019: Moving Forward on Food Loss and Waste Reduction; Food and Agriculture Organization of the United Nations: Rome, Italy, 2019. [Google Scholar]
- Hartmann, C.; Siegrist, M. Consumer perception and behaviour regarding sustainable protein consumption: A systematic review. Trends Food Sci. Technol. 2017, 61, 11–25. [Google Scholar] [CrossRef]
- Godfray, H.C.J.; Aveyard, P.; Garnett, T.; Hall, J.W.; Key, T.J.; Lorimer, J.; Pierrehumbert, R.T.; Scarborough, P.; Springmann, M.; Jebb, S.A. Meat consumption, health, and the environment. Science 2018, 361, eaam5324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Filho, P.F.S.; Nair, R.B.; Andersson, D.; Lennartsson, P.R.; Taherzadeh, M.J. Vegan-mycoprotein concentrate from pea-processing industry byproduct using edible filamentous fungi. Fungal Biol. Biotechnol. 2018, 5, 5. [Google Scholar] [CrossRef]
- Moore, D.; Chiu, S.W. Fungal products as food. Fungal Divers. Res. Ser. 2001, 6, 223–251. [Google Scholar]
- Gmoser, R.; Fristedt, R.; Larsson, K.; Undeland, I.; Taherzadeh, M.J.; Lennartsson, P.R. From stale bread and brewers spent grain to a new food source using edible filamentous fungi. Bioengineered 2020, 11, 582–598. [Google Scholar] [CrossRef] [PubMed]
- Shurtleff, W.; Aoyagi, A. The Book of Tempeh; Harper & Row, Publishers: New York, NY, USA, 1979. [Google Scholar]
- Wiebe, M. Myco-protein from Fusarium venenatum: A well-established product for human consumption. Appl. Microbiol. Biotechnol. 2002, 58, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Smetana, S.; Mathys, A.; Knoch, A.; Heinz, V. Meat alternatives: Life cycle assessment of most known meat substitutes. Int. J. Life Cycle Assess. 2015, 20, 1254–1267. [Google Scholar] [CrossRef]
- Melikoglu, M.; Webb, C. Chapter 4—Use of Waste Bread to Produce Fermentation Products. In Food Industry Wastes; Kosseva, M.R., Webb, C., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 63–76. [Google Scholar]
- WRAP, H. Link Consumer Strategies, Campden BRI. In Reducing Household Bakery Waste; WRAP, Waste and Resource Action Programme: Banbury, UK, 2011. [Google Scholar]
- Brancoli, P.; Rousta, K.; Bolton, K. Life cycle assessment of supermarket food waste. Resour. Conserv. Recycl. 2017, 118, 39–46. [Google Scholar] [CrossRef]
- Brancoli, P.; Lundin, M.; Bolton, K.; Eriksson, M. Bread loss rates at the supplier-retailer interface—Analysis of risk factors to support waste prevention measures. Resour. Conserv. Recycl. 2019, 147, 128–136. [Google Scholar] [CrossRef]
- Ghosh, R.; Eriksson, M. Food waste due to retail power in supply chains: Evidence from Sweden. Glob. Food Sec. 2019, 20, 1–8. [Google Scholar] [CrossRef]
- Eriksson, M.; Ghosh, R.; Mattsson, L.; Ismatov, A. Take-back agreements in the perspective of food waste generation at the supplier-retailer interface. Resour. Conserv. Recycl. 2017, 122, 83–93. [Google Scholar] [CrossRef]
- Papargyropoulou, E.; Lozano, R.; Steinberger, J.K.; Wright, N.; bin Ujang, Z. The food waste hierarchy as a framework for the management of food surplus and food waste. J. Clean. Prod. 2014, 76, 106–115. [Google Scholar] [CrossRef]
- Brancoli, P.; Bolton, K.; Eriksson, M. Environmental impacts of waste management and valorisation pathways for surplus bread in Sweden. Waste Manag. 2020, 117, 136–145. [Google Scholar] [CrossRef]
- Cucurachi, S.; van der Giesen, C.; Guinée, J. Ex-ante LCA of Emerging Technologies. Procedia CIRP 2018, 69, 463–468. [Google Scholar] [CrossRef]
- Jeswiet, J.; Hauschild, M. EcoDesign and future environmental impacts. Mater. Des. 2005, 26, 629–634. [Google Scholar] [CrossRef]
- McAloone, T.C.; Bey, N. Environmental Improvement through Product Development: A Guide; Danish Environmental Protection Agency: Copenhagen, Denmark, 2009. [Google Scholar]
- European Commission. Innovating for Sustainable Growth: A Bioeconomy for Europe; Publications Office of the European Union: Brussels, Belgium, 2012. [Google Scholar]
- Eriksson, M.; Strid, I.; Hansson, P.A. Carbon footprint of food waste management options in the waste hierarchy—A Swedish case study. J. Clean. Prod. 2015, 93, 115–125. [Google Scholar] [CrossRef]
- Vandermeersch, T.; Alvarenga, R.A.F.; Ragaert, P.; Dewulf, J. Environmental sustainability assessment of food waste valorization options. Resour. Conserv. Recycl. 2014, 87, 57–64. [Google Scholar] [CrossRef]
- Lam, C.-M.; Yu, I.K.M.; Hsu, S.-C.; Tsang, D.C.W. Life-cycle assessment on food waste valorisation to value-added products. J. Clean. Prod. 2018, 199, 840–848. [Google Scholar] [CrossRef]
- Gmoser, R.; Sintca, C.; Taherzadeh, M.J.; Lennartsson, P.R. Combining submerged and solid state fermentation to convert waste bread into protein and pigment using the edible filamentous fungus N. intermedia. Waste Manag. 2019, 97, 63–70. [Google Scholar] [CrossRef]
- Melikoglu, M.; Lin, C.S.K.; Webb, C. Solid state fermentation of waste bread pieces by Aspergillus awamori: Analysing the effects of airflow rate on enzyme production in packed bed bioreactors. Food Bioprod. Process. 2015, 95, 63–75. [Google Scholar] [CrossRef]
- Buyle, M.; Audenaert, A.; Billen, P.; Boonen, K.; Van Passel, S. The future of ex-ante LCA? Lessons learned and practical recommendations. Sustainability 2019, 11, 5456. [Google Scholar] [CrossRef] [Green Version]
- Wiloso, E.I.; Sinke, P.; Setiawan, A.A.R.; Sari, A.A.; Waluyo, J.; Putri, A.M.H.; Guinée, J. Hotspot identification in the Indonesian tempeh supply chain using life cycle assessment. Int. J. Life Cycle Assess. 2019, 24, 1948–1961. [Google Scholar] [CrossRef]
- Dunn, J.B.; Mueller, S.; Wang, M.; Han, J. Energy consumption and greenhouse gas emissions from enzyme and yeast manufacture for corn and cellulosic ethanol production. Biotechnol. Lett. 2012, 34, 2259–2263. [Google Scholar] [CrossRef]
- Jungbluth, N.; Nowack, K.; Eggenberger, S.; König, A.; Keller, R. Untersuchungen zur umweltfreundlichen Eiweissversorgung–Pilotstudie; ESU-services GmbH für das Bundesamt für Umwelt (BAFU): Bern, Switzerland, 2016. [Google Scholar]
- Wang, R.; Gmoser, R.; Taherzadeh, M.J.; Lennartsson, P.R. Solid-state fermentation of stale bread by an edible fungus in a semi-continuous plug-flow bioreactor. Biochem. Eng. J. 2021, 169, 107959. [Google Scholar] [CrossRef]
- Huijbregts, M.A.J.; Steinmann, Z.J.N.; Elshout, P.M.F.; Stam, G.; Verones, F.; Vieira, M.D.M.; Hollander, A.; Zijp, M.; Van Zelm, R. ReCiPe 2016: A harmonized life cycle impact assessment method at midpoint and endpoint level Report I: Characterization. Int. J. Life Cycle Assess. 2017, 22, 138–147. [Google Scholar] [CrossRef]
- Gmoser, R.; Lennartsson, P.R.; Taherzadeh, M.J. From surplus bread to burger using filamentous fungi at bakeries: Techno-economical evaluation. Clean. Environ. Syst. 2021, 2, 100020. [Google Scholar] [CrossRef]
- Ingledew, W.M.; Austin, G.D.; Kraus, J.K. Commercial yeast production for the fuel ethanol and distilled beverage industries. In The Alcohol Textbook, 5th ed.; Nottingham University Press: Nottingham, UK, 2009; pp. 127–144. [Google Scholar]
- Piccinno, F.; Hischier, R.; Seeger, S.; Som, C. From laboratory to industrial scale: A scale-up framework for chemical processes in life cycle assessment studies. J. Clean. Prod. 2016, 135, 1085–1097. [Google Scholar] [CrossRef]
- Ward, O.P. Production of recombinant proteins by filamentous fungi. Biotechnol. Adv. 2012, 30, 1119–1139. [Google Scholar] [CrossRef]
- Nair, R.B. Integration of first and second generation bioethanol processes. In Swedish Centre for Resource Recovery; University of Borås: Borås, Sweden, 2017. [Google Scholar]
- Ferreira, J.A.; Lennartsson, P.R.; Edebo, L.; Taherzadeh, M.J. Zygomycetes-based biorefinery: Present status and future prospects. Bioresour. Technol. 2013, 135, 523–532. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Unit | Scenario I | Scenario II | Scenario. III | Scenario IV | |
|---|---|---|---|---|---|
| Inputs | |||||
| Bread | kg | 2.0 | 2.0 | 2.0 | 2.0 |
| Inoculum | g | 10 | 10 | 10 | 29 * |
| Water | l | 2.63 | 1.81 | 1.92 | - |
| Glucose | g | - | - | 38.50 | - |
| NH4Cl | g | - | 16.28 | 14.50 | - |
| NaOH | g | - | - | 0.62 | - |
| Molasses | g | - | 0.36 | - | - |
| Bread | g | 52.60 | - | - | - |
| Electricity | kWh | 0.065 | 0.033 | 0.033 | 0.033 |
| Water for mixing | l | 1.34 | 1.34 | 1.34 | 1.34 |
| Electricity for drying | kWh | 0.53 | 0.53 | 0.53 | 0.53 |
| Electricity for grinding | kWh | 0.024 | 0.024 | 0.024 | 0.024 |
| Electricity for SSF | kWh | 1.55 | 1.55 | 1.55 | 1.55 |
| Outputs | |||||
| Fermented fungal product * | kg | 1 | 1 | 1 | 1 |
| Impact Category | Unit | Sc. I | Sc. II | Sc. III | Sc. IV |
|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 1.4 × 10−1 | 1.9 × 10−1 | 2.1 × 10−1 | 1.1 × 10−1 |
| Stratospheric ozone depletion | kg CFC-11 eq | 3.0 × 10−7 | 6.2 × 10−7 | 5.2 × 10−7 | 2.4 × 10−7 |
| Ionizing radiation | kBq Co-60 eq | 8.0 × 10−1 | 7.9 × 10−1 | 7.9 × 10−1 | 6.4 × 10−1 |
| Ozone formation, Human health | kg NOx eq | 3.5 × 10−4 | 4.8 × 10−4 | 5.1 × 10−4 | 2.8 × 10−4 |
| Fine particulate matter formation | kg PM2.5 eq | 2.3 × 10−4 | 3.7 × 10−4 | 3.8 × 10−4 | 1.9 × 10−4 |
| Ozone formation, TE | kg NOx eq | 3.6 × 10−4 | 4.9 × 10−4 | 5.2 × 10−4 | 2.8 × 10−4 |
| Terrestrial acidification | kg SO2 eq | 5.8 × 10−4 | 1.2 × 10−3 | 1.1 × 10−3 | 4.6 × 10−4 |
| Freshwater eutrophication | kg p eq | 9.4 × 10−5 | 1.1 × 10−4 | 1.2 × 10−4 | 7.54 × 10−5 |
| Marine eutrophication | kg n eq | 1.8 × 10−5 | 1.0 × 10−4 | 8.5 × 10−5 | 1.5 × 10−5 |
| Terrestrial ecotoxicity | kg 1,4-DCB | 2.2 × 100 | 2.4 × 100 | 2.7 × 100 | 1.8 × 100 |
| Freshwater ecotoxicity | kg 1,4-DCB | 9.4 × 10−2 | 9.6 × 10−2 | 9.9 × 10−2 | 7.5 × 10−2 |
| Marine ecotoxicity | kg 1,4-DCB | 1.1 × 10−1 | 1.2 × 10−1 | 1.2 × 10−1 | 9.1 × 10−2 |
| Human carcinogenic toxicity | kg 1,4-DCB | 1.9 × 10−2 | 2.0 × 10−2 | 2.1 × 10−2 | 1.5 × 10−2 |
| Human non-carcinogenic toxicity | kg 1,4-DCB | 6.0 × 10−1 | 6.4 × 10−1 | 6.5 × 10−1 | 4.8 × 10−1 |
| Land use | m2 a crop eq | 6.1 × 10−2 | 9.2 × 10−2 | 8.7 × 10−2 | 4.8 × 10−2 |
| Mineral resource scarcity | kg Cu eq | 2.0 × 10−3 | 2.4 × 10−3 | 2.5 × 10−3 | 1.6 × 10−3 |
| Fossil resource scarcity | kg oil eq | 2.6 × 10−2 | 3.8 × 10−2 | 4.5 × 10−2 | 2.1 × 10−2 |
| Water consumption | m3 | 2.1 × 10−2 | 2.3 × 10−2 | 2.2 × 10−2 | 1.5 × 10−2 |
| Scenarios | Environmental Impact (mPt) | Protein Content | Lag Phase | Morphology |
|---|---|---|---|---|
| Scenario I | 9.7 | 27% | Short | Dispersed mycelium |
| Scenario II | 12.5 | 29% | Short | Pellets |
| Scenario III | 13.0 | 25% | Long | Dispersed mycelium |
| Scenario IV | 7.6 | 27% | Short | Dispersed mycelium |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brancoli, P.; Gmoser, R.; Taherzadeh, M.J.; Bolton, K. The Use of Life Cycle Assessment in the Support of the Development of Fungal Food Products from Surplus Bread. Fermentation 2021, 7, 173. https://doi.org/10.3390/fermentation7030173
Brancoli P, Gmoser R, Taherzadeh MJ, Bolton K. The Use of Life Cycle Assessment in the Support of the Development of Fungal Food Products from Surplus Bread. Fermentation. 2021; 7(3):173. https://doi.org/10.3390/fermentation7030173
Chicago/Turabian StyleBrancoli, Pedro, Rebecca Gmoser, Mohammad J. Taherzadeh, and Kim Bolton. 2021. "The Use of Life Cycle Assessment in the Support of the Development of Fungal Food Products from Surplus Bread" Fermentation 7, no. 3: 173. https://doi.org/10.3390/fermentation7030173
APA StyleBrancoli, P., Gmoser, R., Taherzadeh, M. J., & Bolton, K. (2021). The Use of Life Cycle Assessment in the Support of the Development of Fungal Food Products from Surplus Bread. Fermentation, 7(3), 173. https://doi.org/10.3390/fermentation7030173







