Impact of Pea Fiber (Pisum sativum L.) on the Viability of Limosilactobacillus reuteri ACC27 and Quality Attributes of Fermented Milk
Abstract
1. Introduction
2. Materials and Methods
2.1. Starter Culture and Raw Milk
2.2. Experimental Design
2.3. Effect of Pea Fiber Powder on In Vitro Growth of L. reuteri ACC27
2.4. Fermented Milk Production and Monitoring of Fermentation Processes
2.5. Microbiological Analyses
2.6. Physicochemical Analyses
2.7. Apparent Viscosity and Colorimetric Properties
2.8. Organic Acid Content
2.9. Antioxidant Capacity and Total Phenolic Content
2.10. Sensory Analyses
2.11. Statistical Analyses
3. Results and Discussion
3.1. Effect of Pea Fiber Powder on In Vitro Growth of L. reuteri ACC27
3.2. Monitoring of Fermentation Processes
3.3. Microbiological Analyses
3.4. Physicochemical Analyses
3.5. Apparent Viscosity and Colorimetric Properties
3.6. Organic Acid Content
3.7. Antioxidant Capacity and Total Phenolic Content
3.8. Sensory Analyses
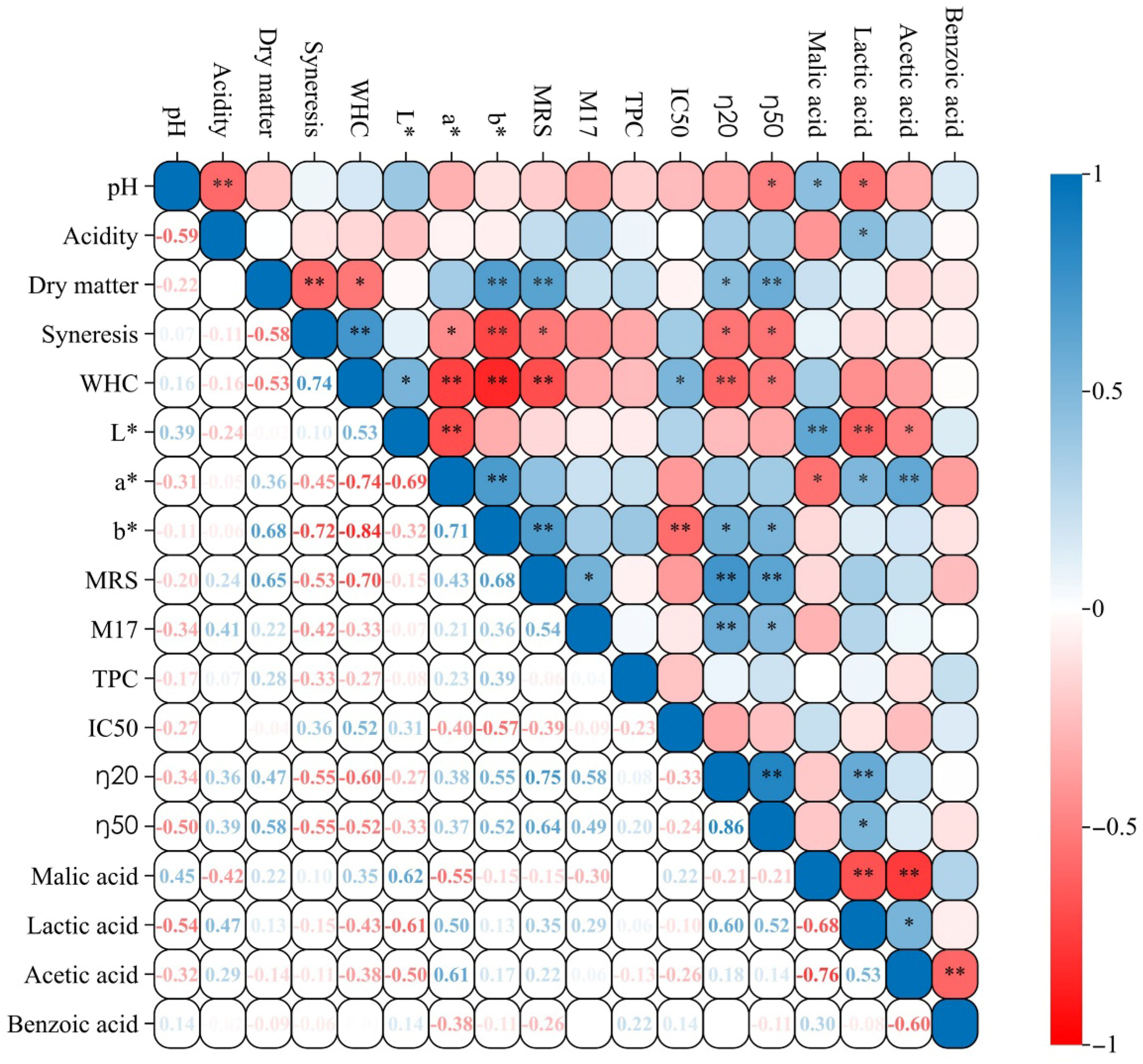
3.9. Correlation and Principal Component Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hashemi, S.M.B.; Raeisi, S.; Zanardi, E.; Paciulli, M.; Chiavaro, E. Antimicrobial and antioxidant activities of fermented meat patty with Lactobacillus strains. Ital. J. Food Sci. 2018, 30, 268–279. [Google Scholar]
- Şanlier, N.; Gökcen, B.B.; Sezgin, A.C. Health benefits of fermented foods. Crit. Rev. Food Sci. Nutr. 2017, 59, 506–527. [Google Scholar] [CrossRef]
- Kariyawasam, K.M.G.M.M.; Lee, N.K.; Paik, H.D. Fermented dairy products as delivery vehicles of novel probiotic strains isolated from traditional fermented Asian foods. J. Food Sci. Technol. 2021, 58, 2467–2478. [Google Scholar] [CrossRef]
- Çetin, B.; Aktaş, H. Monitoring probiotic properties and safety evaluation of antilisterial Enterococcus faecium strains with cholesterol-lowering potential from raw cow’s milk. Food Biosci. 2024, 61, 104532. [Google Scholar] [CrossRef]
- Şanlıbaba, P. Fermented nondairy functional foods based on probiotics. Ital. J. Food Sci. 2023, 35, 91–105. [Google Scholar] [CrossRef]
- Rayes, A.A. Enhancement of probiotic bioactivity by some prebiotics to produce bio-fermented milk. Life Sci. J. 2012, 9, 2246–2253. [Google Scholar]
- Figueroa-Gonzalez, I.; Rodriguez-Serrano, G.; Gomez-Ruiz, L.; Garcia-Garibay, M.; Cruz-Guerrero, A. Prebiotic effect of commercial saccharides on probiotic bacteria isolated from commercial products. Food Sci. Technol. 2019, 39, 747–753. [Google Scholar] [CrossRef]
- Reshetnik, E.I.; Utochkina, E.A. Healthy food products with probiotic and prebiotic properties. Foods Raw Mater. 2013, 1, 88–94. [Google Scholar] [CrossRef]
- El-Aidie, S.; Elsayed, N.; Hashem, M.; Elkashef, H. Development of fermented skimmed milk fortified with yellow sweet potato (Ipomoea batatas L.) with prebiotic and antioxidant activity. J. Food Nutr. Res. 2021, 60, 66–75. [Google Scholar]
- Barat, A.; Ozcan, T. Growth of probiotic bacteria and characteristics of fermented milk containing fruit matrices. Int. J. Dairy Technol. 2018, 71, 120–129. [Google Scholar] [CrossRef]
- Santos, R.O.; Silva, M.V.F.; Nascimento, K.O.; Batista, A.L.; Moraes, J.; Andrade, M.M.; Andrade, L.G.Z.S.; Khosravi-Darani, K.; Freitas, M.Q.; Raices, R.S.L.; et al. Prebiotic flours in dairy food processing: Technological and sensory implications. Int. J. Dairy Technol. 2018, 71, 1–10. [Google Scholar]
- Zhao, R.; Qiao, M.; Ma, Y.; Li, Q.; Li, N.; Huang, X.; Cheng, Y.; Song, L. Optimization of microwave-ultrasound assisted extraction of oligosaccharides from pea seeds and its probiotic proliferative activity. J. Food Sci. Technol. 2025; in press.
- Aktaş, H.; Çetin, B. Multidimensional evaluation of techno-functional properties of yoghurt bacteria. Int. Dairy J. 2024, 148, 105795. [Google Scholar] [CrossRef]
- Ashraf, R.; Shah, N.P. Selective and differential enumerations of Lactobacillus delbrueckii subsp. bulgaricus, Streptococcus thermophilus, Lactobacillus acidophilus, Lactobacillus casei and Bifidobacterium spp. in yoghurt—A review. Int. J. Food Microbiol. 2011, 149, 194–208. [Google Scholar]
- Aktaş, H. Characterisation of enterocins produced by antilisterial Enterococcus faecium BH04, BH12, BH84, and BH99 and in vitro/in situ inhibition of Listeria monocytogenes. Food Sci. Nutr. 2025, 13, e70142. [Google Scholar] [CrossRef]
- Aktaş, H.; Çetin, B. Effects of novel autochthonous starter cultures on quality characteristics of yoghurt. Int. J. Food Eng. 2024, 20, 799–808. [Google Scholar] [CrossRef]
- Robinson, R.K.; Lucey, J.A.; Tamime, A.Y. Manufacture of Yoghurt; Blackwell Publishing: London, UK, 2006. [Google Scholar]
- Harrigan, W.F. Laboratory Methods in Food Microbiology; Academic Press: San Diego, CA, USA, 1998. [Google Scholar]
- ISO 21527-1:2008; Microbiology of Food and Animal Feeding Stuffs-Horizontal Method for the Enumeration of Yeasts and Moulds-Part 1: Colony Count Technique in Products with Water Activity Greater than 0.95. The International Organization for Standardization: Geneva, Switzerland, 2008.
- ISO 4832:2006; Microbiology of Food and Animal Feeding Stuffs-Horizontal Method for the Enumeration of Coliforms-Colony-Count Technique. The International Organization for Standardization: Geneva, Switzerland, 2006.
- Savello, P.A.; Ernstrom, C.A.; Kalab, M. Microstructure and meltability of model process cheese made with rennet and acid casein. J. Dairy Sci. 1989, 72, 1–11. [Google Scholar] [CrossRef]
- Cemeroglu, B. General Methods in Food Analysis: Food Analysis; Food Technology Association Publications: Ankara, Türkiye, 2010. [Google Scholar]
- AOAC. Official Methods of Analysis of the AOAC, 18th ed.; AOAC: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Taşdemir, Y.; Gölge, E. Rheology and sensory properties of microencapsulated propolis-enriched stirred-type yoghurt. Ital. J. Food Sci. 2023, 35, 155–163. [Google Scholar]
- Bensmira, M.; Jiang, B. Effect of some operating variables on the microstructure and physical properties of a novel kefir formulation. J. Food Eng. 2012, 108, 579–584. [Google Scholar] [CrossRef]
- Özer, E. Characteristics of apricot yogurt fermented with different culture strains and stevia. Ital. J. Food Sci. 2024, 36, 151–163. [Google Scholar]
- Atalar, I. Functional kefir production from high pressure homogenized hazelnut milk. LWT 2019, 107, 256–263. [Google Scholar] [CrossRef]
- Ozcan, T.; Sahin, S.; Akpinar-Bayizit, A.; Yilmaz-Ersan, L. Assessment of antioxidant capacity by method comparison and amino acid characterisation in buffalo milk kefir. Int. J. Dairy Technol. 2019, 72, 65–73. [Google Scholar] [CrossRef]
- Şengül, M.; Karakütük, İ.A.; Ay, E. Effects of sweetener type and storage conditions on physicochemical properties, phenolic compounds, and antioxidant properties of cherry laurel (Prunus laurocerasus) pulp and marmalade. J. Food Meas. Charact. 2024, 18, 8622–8637. [Google Scholar] [CrossRef]
- Bodyfelt, F.W.; Tobias, J.; Trout, G.M. The Sensory Evaluation of Dairy Products; Van Nostrand Reinhold: New York, NY, USA, 1988. [Google Scholar]
- Zafar, J.; Ul Haq, I.; Nayik, G.A.; Ramniwas, S.; Mugabi, R.; Ali Alharbi, S.; Ansari, M.J. Studies on the growth of Lactobacillus reuteri, Bifidobacterium and Escherichia coli as affected by prebiotic extracted from citrus peel. Int. J. Food Prop. 2024, 27, 783–798. [Google Scholar] [CrossRef]
- Kumari, T.; Das, A.J.; Das, A.B.; Reddy, C.K.; Deka, S.C. Prebiotic activity of enzymatically modified pea peel dietary fiber: An in vitro study. Bioact. Carbohydr. Diet. Fibre 2024, 32, 100452. [Google Scholar] [CrossRef]
- de Souza Oliveira, R.P.; Perego, P.; de Oliveira, M.N.; Converti, A. Effect of inulin on the growth and metabolism of a probiotic strain of Lactobacillus rhamnosus in co-culture with Streptococcus thermophilus. LWT 2012, 47, 358–363. [Google Scholar] [CrossRef]
- Carlson, J.; Hospattankar, A.; Deng, P.; Swanson, K.; Slavin, J. Prebiotic effects and fermentation kinetics of wheat dextrin and partially hydrolyzed guar gum in an in vitro batch fermentation system. Foods 2015, 4, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, R.; Swann, J.R.; Vulevic, J.; Gibson, G.R.; Costabile, A. Fermentation properties and potential prebiotic activity of Bimuno® galacto-oligosaccharide (65% galacto-oligosaccharide content) on in vitro gut microbiota parameters. Br. J. Nutr. 2016, 116, 480–486. [Google Scholar] [CrossRef]
- Van den Abbeele, P.; Ghyselinck, J.; Marzorati, M.; Villar, A.; Zangara, A.; Smidt, C.R.; Risco, E. In vitro evaluation of prebiotic properties of a commercial artichoke inflorescence extract revealed bifidogenic effects. Nutrients 2020, 12, 1552. [Google Scholar] [CrossRef]
- Aly, E.; Ros-Berruezo, G.; López-Nicolás, R.; Darwish, A.A.; Sánchez-Moya, T.; Planes-Muñoz, D.; Frontela-Saseta, C. In vitro prebiotic activity of rhLf and galactooligosaccharides on infant intestinal microbiota. Nutr. Hosp. 2023, 40, 701–710. [Google Scholar]
- Roller, M.; Clune, Y.; Collins, K.; Rechkemmer, G.; Watzl, B. Consumption of prebiotic inulin enriched with oligofructose in combination with the probiotics Lactobacillus rhamnosus and Bifidobacterium lactis has minor effects on selected immune parameters in polypectomised and colon cancer patients. Br. J. Nutr. 2007, 97, 676–684. [Google Scholar] [CrossRef]
- Kadlec, R.; Jakubec, M. The effect of prebiotics on adherence of probiotics. J. Dairy Sci. 2014, 97, 1983–1990. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Mallet, J.F.; Graham, E.; Matar, C. Role of probiotics and prebiotics in immunomodulation. Curr. Opin. Food Sci. 2018, 20, 82–91. [Google Scholar] [CrossRef]
- You, S.; Ma, Y.; Yan, B.; Pei, W.; Wu, Q.; Ding, C.; Huang, C. The promotion mechanism of prebiotics for probiotics: A review. Front. Nutr. 2022, 9, 1000517. [Google Scholar] [CrossRef]
- Sendra, E.; Fayos, P.; Lario, Y.; Fernández-López, J.; Sayas-Barberá, E.; Pérez-Alvarez, J.A. Incorporation of citrus fibers in fermented milk containing probiotic bacteria. Food Microbiol. 2008, 25, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.P.; Florence, A.C.; Silva, R.C.; Perego, P.; Converti, A.; Gioielli, L.A.; Oliveira, M.N. Effect of different prebiotics on the fermentation kinetics, probiotic survival and fatty acids profiles in nonfat symbiotic fermented milk. Int. J. Food Microbiol. 2009, 128, 467–472. [Google Scholar] [CrossRef]
- de Souza Oliveira, R.P.; Perego, P.; de Oliveira, M.N.; Converti, A. Effect of inulin as prebiotic and synbiotic interactions between probiotics to improve fermented milk firmness. J. Food Eng. 2011, 107, 36–40. [Google Scholar] [CrossRef]
- Celestin, S.; Thorat, S.S.; Desale, R.J.; Chavan, U.D. Effect of milk supplementation with fructooligosaccharides and inulin on viable counts of probiotic bacteria in goat and cow milk yoghurts. IOSR J. Environ. Sci. Toxicol. Food Technol. 2015, 9, 6–12. [Google Scholar]
- Gopal, N.; Hill, C.; Ross, P.R.; Beresford, T.P.; Fenelon, M.A.; Cotter, P.D. The prevalence and control of Bacillus and related spore-forming bacteria in the dairy industry. Front. Microbiol. 2015, 6, 1418. [Google Scholar] [CrossRef] [PubMed]
- Mehta, D.S.; Metzger, L.E.; Hassan, A.N.; Nelson, B.K.; Patel, H.A. The ability of spore formers to degrade milk proteins, fat, phospholipids, common stabilizers, and exopolysaccharides. J. Dairy Sci. 2019, 102, 10799–10813. [Google Scholar] [CrossRef]
- Casarotti, S.N.; Monteiro, D.A.; Moretti, M.M.; Penna, A.L.B. Influence of the combination of probiotic cultures during fermentation and storage of fermented milk. Food Res. Int. 2014, 59, 67–75. [Google Scholar] [CrossRef]
- Surono, I.S.; Hosono, A. Fermented milks. In Encyclopedia of Dairy Sciences; Roginski, H., Fuquay, J.W., Fox, P.F., Eds.; Academic Press: London, UK, 2003; pp. 1018–1023. [Google Scholar]
- Xu, Z.; Li, S.; Gong, G.; Liu, Z.; Wu, Z.; Ma, C. Influence of different acidifying strains of Lactobacillus delbrueckii subsp. bulgaricus on the quality of yoghurt. Food Sci. Technol. Res. 2015, 21, 263–269. [Google Scholar] [CrossRef]
- Kalyas, A.; Ürkek, B. Effect of flaxseed powder on physicochemical, rheological, microbiological and sensory properties of yoghurt. Braz. Arch. Biol. Technol. 2022, 65, e22210012. [Google Scholar] [CrossRef]
- Tamime, A.Y.; Robinson, R.K. Tamime and Robinson’s Yoghurt: Science and Technology; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Debon, J.; Prudêncio, E.S.; Petrus, J.C.C.; Fritzen-Freire, C.B.; Müller, C.M.; Amboni, R.D.D.M.C.; Vieira, C.R.W. Storage stability of prebiotic fermented milk obtained from permeate resulting of the microfiltration process. LWT 2012, 47, 96–102. [Google Scholar] [CrossRef]
- Zare, F.; Boye, J.I.; Champagne, C.P.; Orsat, V.; Simpson, B.K. Probiotic milk supplementation with pea flour: Microbial and physical properties. Food Bioprocess Technol. 2013, 6, 1321–1331. [Google Scholar]
- Damian, C.; Olteanu, A. Influence of dietary fiber from pea on some quality characteristics of yoghurts. J. Agroaliment. Process. Technol. 2014, 20, 156–160. [Google Scholar]
- Ibrahim, A.H.; Khalifa, S.A. Improve sensory quality and textural properties of fermented camel’s milk by fortified with dietary fiber. J. Am. Sci. 2015, 11, 42–54. [Google Scholar]
- Kurbanova, I.; Lauciene, L.; Kondrotiene, K.; Zakariene, G.; Radenkovs, V.; Kiselioviene, S.; Salaseviciene, A.; Vasiliauskaite, A.; Malakauskas, M.; Musulmanova, M.; et al. Physicochemical, sensory, and microbiological analysis of fermented drinks made from white kidney bean extract and cow’s milk blends during refrigerated storage. Microorganisms 2024, 12, 1832. [Google Scholar] [CrossRef]
- Li, W.; Ghamry, M.; Xu, X.; Zhang, L.; Li, J. Organic acids: Key products of fermented foods and sources of anti-inflammatory potential. Food Rev. Int. 2025; in press.
- Özcelik, S.; Kuley, E.; Özogul, F. Formation of lactic, acetic, succinic, propionic, formic and butyric acid by lactic acid bacteria. LWT 2016, 73, 536–542. [Google Scholar] [CrossRef]
- Aktaş, H.; Meral Aktaş, H.; Ürkek, B.; Şengül, M.; Çetin, B. Evaluation of spreadable kefir produced from different milks in terms of some quality criteria. Probiotics Antimicrob. Proteins 2024, 16, 1734–1743. [Google Scholar] [CrossRef]
- Meral-Aktaş, H.; Bazu-Çırpıcı, B.; Aktaş, H.; Kadiroğlu, H.; Çetin, B. Comparative quality assessment of plant-based kefir as a vegan alternative to traditional kefir. Food Biosci. 2025, 70, 107062. [Google Scholar] [CrossRef]
- Nazir, N.; Nisar, M.; Ahmad, S.; Wadood, S.F.; Jan, T.; Zahoor, M.; Ahmad, M.; Ullah, A. Characterization of phenolic compounds in two novel lines of Pisum sativum L. along with their in vitro antioxidant potential. Environ. Sci. Pollut. Res. 2020, 27, 7639–7646. [Google Scholar]
- Nilsson, J.; Stegmark, R.; Åkesson, B. Total antioxidant capacity in different pea (Pisum sativum) varieties after blanching and freezing. Food Chem. 2004, 86, 501–507. [Google Scholar] [CrossRef]
- Zhao, T.; Su, W.; Qin, Y.; Wang, L.; Kang, Y. Phenotypic diversity of pea (Pisum sativum L.) varieties and the polyphenols, flavonoids, and antioxidant activity of their seeds. Cienc. Rural 2020, 50, e20190196. [Google Scholar]
- Vasilev, D.; Saicić, S.; Vasiljević, N. Quality and nutritive value of fermented sausages produced with inulin and pea fibre as fat replacers. Fleischwirtschaft 2013, 93, 123–127. [Google Scholar]
- Ruiz-Capillas, C.; Herrero, A.M. Sensory analysis and consumer research in new product development. Foods 2021, 10, 582. [Google Scholar] [CrossRef]
- Schneider, N.; Gerber, M. Correlation between viscosity, temperature and total solid content of algal biomass. Bioresour. Technol. 2014, 170, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Bai, M.; Kwok, L.Y.; Zhong, Z.; Sun, Z. The intricate symbiotic relationship between lactic acid bacterial starters in the milk fermentation ecosystem. Crit. Rev. Food Sci. Nutr. 2025, 65, 728–745. [Google Scholar] [CrossRef]
- Celik, S.; Bakirci, I. Some properties of yoghurt produced by adding mulberry pekmez (concentrated juice). Int. J. Dairy Technol. 2003, 56, 26–29. [Google Scholar] [CrossRef]
- Francis, M.J.; Glover, Z.J.; Yu, Q.; Povey, M.J.; Holmes, M.J. Acoustic characterisation of pH dependant reversible micellar casein aggregation. Colloids Surf. A Physicochem. Eng. Asp. 2019, 568, 259–265. [Google Scholar] [CrossRef]






| Parameters | Sample | Storage Period (Days) | Sig. | |||
|---|---|---|---|---|---|---|
| 1 | 7 | 14 | 21 | |||
| Limosilactobacillus reuteri ACC27 (log CFU/g) | FM00 | 5.57 ± 0.12 | 5.70 ± 0.00 D | 5.26 ± 0.31 | 5.89 ± 0.58 | ns |
| FM05 | 6.44 ± 0.06 | 6.65 ± 0.24 AB | 6.44 ± 0.43 | 6.19 ± 0.16 | ns | |
| FM10 | 6.21 ± 0.13 | 6.00 ± 0.00 CD | 6.29 ± 0.16 | 6.38 ± 0.51 | ns | |
| FM15 | 6.39 ± 0.55 | 6.76 ± 0.03 A | 6.20 ± 0.28 | 6.35 ± 0.49 | ns | |
| FM20 | 6.37 ± 0.52 | 6.24 ± 0.34 BC | 6.15 ± 0.21 | 6.84 ± 0.09 | ns | |
| Sig. | ns | ** | ns | ns | ||
| Streptococcus thermophilus 212S (log CFU/g) | FM00 | 8.93 ± 0.12 | 8.93 ± 0.12 | 8.92 ± 0.02 | 9.15 ± 0.21 | ns |
| FM05 | 9.07 ± 0.04 | 9.12 ± 0.20 | 9.24 ± 0.34 | 9.16 ± 0.02 | ns | |
| FM10 | 9.11 ± 0.07 | 8.98 ± 0.03 | 9.14 ± 0.01 | 9.11 ± 0.10 | ns | |
| FM15 | 9.13 ± 0.02 | 9.07 ± 0.04 | 9.10 ± 0.05 | 9.11 ± 0.10 | ns | |
| FM20 | 9.07 ± 0.17 | 9.01 ± 0.01 | 9.22 ± 0.12 | 9.05 ± 0.14 | ns | |
| Sig. | ns | ns | ns | ns | ||
| Parameters | Sample | Storage Period (Days) | Sig. | |||
|---|---|---|---|---|---|---|
| 1 | 7 | 14 | 21 | |||
| pH | FM00 | 4.68 ± 0.02 aA | 4.52 ± 0.01 c | 4.58 ± 0.02 bA | 4.55 ± 0.01 bcAB | ** |
| FM05 | 4.66 ± 0.01 aAB | 4.58 ± 0.01 b | 4.49 ± 0.01 dB | 4.52 ± 0.01 cB | *** | |
| FM10 | 4.62 ± 0.03 aB | 4.53 ± 0.01 b | 4.42 ± 0.01 cC | 4.43 ± 0.02 cC | ** | |
| FM15 | 4.54 ± 0.01 C | 4.57 ± 0.02 | 4.55 ± 0.01 A | 4.57 ± 0.02 A | ns | |
| FM20 | 4.53 ± 0.00 C | 4.57 ± 0.02 | 4.58 ± 0.02 A | 4.55 ± 0.01 AB | ns | |
| Sig. | *** | ns | *** | ** | ||
| Acidity (lactic acid, %) | FM00 | 0.90 ± 0.01 b | 0.92 ± 0.00 b | 0.91 ± 0.01 bC | 0.96 ± 0.00 aBC | ** |
| FM05 | 0.93 ± 0.01 c | 0.96 ± 0.03 bc | 1.00 ± 0.00 abA | 1.01 ± 0.00 aA | * | |
| FM10 | 0.89 ± 0.05 | 0.98 ± 0.01 | 0.98 ± 0.01 B | 0.98 ± 0.01 AB | ns | |
| FM15 | 0.95 ± 0.01 | 0.93 ± 0.04 | 1.02 ± 0.00 A | 0.96 ± 0.02 B | ns | |
| FM20 | 0.92 ± 0.01 | 0.91 ± 0.01 | 0.89 ± 0.01 C | 0.93 ± 0.01 C | ns | |
| Sig. | ns | ns | *** | ** | ||
| Total dry matter (%) | FM00 | 13.84 ± 0.06 | 14.20 ± 1.16 | 13.11 ± 1.01 | 12.30 ± 0.35 C | ns |
| FM05 | 14.91 ± 0.34 | 15.52 ± 1.62 | 14.30 ± 0.58 | 14.38 ± 0.55 B | ns | |
| FM10 | 15.20 ± 0.89 | 15.35 ± 0.67 | 15.75 ± 0.64 | 15.23 ± 0.40 AB | ns | |
| FM15 | 15.12 ± 0.12 | 14.76 ± 0.28 | 14.86 ± 0.97 | 14.61 ± 0.04 B | ns | |
| FM20 | 15.71 ± 0.16 | 14.92 ± 0.38 | 15.54 ± 0.36 | 15.66 ± 0.14 A | ns | |
| Sig. | ns | ns | ns | ** | ||
| Syneresis (%) | FM00 | 12.79 ± 0.63 | 16.73 ± 3.42 | 14.69 ± 2.20 A | 17.82 ± 2.37 | ns |
| FM05 | 11.82 ± 3.03 | 14.68 ± 0.67 | 9.82 ± 1.30 B | 11.65 ± 3.48 | ns | |
| FM10 | 11.55 ± 0.38 | 12.55 ± 1.79 | 9.77 ± 1.12 B | 12.08 ± 3.38 | ns | |
| FM15 | 10.52 ± 0.14 | 8.77 ± 1.73 | 8.31 ± 1.71 B | 6.62 ± 1.55 | ns | |
| FM20 | 10.48 ± 1.21 | 11.35 ± 5.28 | 7.53 ± 0.10 B | 9.53 ± 3.40 | ns | |
| Sig. | ns | ns | * | ns | ||
| WHC (%) | FM00 | 53.42 ± 1.05 A | 53.45 ± 0.36 A | 53.40 ± 0.01 A | 52.45 ± 1.55 A | ns |
| FM05 | 50.62 ± 0.17 aB | 48.10 ± 0.05 bB | 49.78 ± 0.99 aB | 46.98 ± 0.46 bB | ** | |
| FM10 | 49.21 ± 0.02 aBC | 48.65 ± 0.32 aB | 48.93 ± 0.43 aB | 45.82 ± 0.38 bBC | ** | |
| FM15 | 47.89 ± 0.82 aC | 41.61 ± 0.61 cC | 44.27 ± 0.43 bC | 43.19 ± 0.85 bcD | ** | |
| FM20 | 48.21 ± 0.19 aC | 42.56 ± 0.48 cC | 44.98 ± 1.01 bC | 43.84 ± 0.55 bcCD | ** | |
| Sig. | ** | *** | *** | *** | ||
| ŋ20 (cP) | FM00 | 10,817.5 ± 528.5 dD | 18,475.0 ± 884.0 aC | 11,497.5 ± 802.9 cC | 17,157.5 ± 654.8 bD | *** |
| FM05 | 19,865.0 ± 1162.9 cA | 21,612.5 ± 1227.4 bA | 25,350.0 ± 908.2 aA | 16,015.0 ± 594.5 dE | *** | |
| FM10 | 18,865.0 ± 1136.8 cBC | 19,585.0 ± 994.9 bB | 20,080.0 ± 629.0 bB | 20,672.5 ± 492.2 aC | *** | |
| FM15 | 19,457.5 ± 852.6 dAB | 21,805.0 ± 1799.3 bA | 20,587.5 ± 1832.8 cB | 26,505.0 ± 1086.3 aA | *** | |
| FM20 | 18,590.0 ± 1015.1 cC | 19,262.5 ± 1587.9 cBC | 20,482.5 ± 916.8 bB | 21,260.0 ± 961.2 aB | *** | |
| Sig. | *** | *** | *** | *** | ||
| ŋ50 (cP) | FM00 | 4917.0 ± 257.1 dD | 8263.0 ± 507.2 aCD | 6090.0 ± 478.3 bE | 5670.0 ± 236.7 cE | *** |
| FM05 | 8583.0 ± 560.2 cA | 9600.0 ± 742.9 bA | 10,994.0 ± 596.3 aA | 8232.0 ± 401.1 cD | *** | |
| FM10 | 6801.0 ± 677.0 dC | 8792.0 ± 510.6 cB | 10,051.0 ± 493.6 aB | 9348.0 ± 273.0 bC | *** | |
| FM15 | 8879.0 ± 378.6 bA | 8158.0 ± 505.8 cD | 7708.0 ± 375.5 dD | 11,354.0 ± 277.6 aA | *** | |
| FM20 | 7911.0 ± 500.8 dB | 8602.0 ± 473.1 cBC | 9230.0 ± 287.7 bC | 9559.0 ± 227.4 aB | *** | |
| Sig. | *** | *** | *** | *** | ||
| L* | FM00 | 88.59 ± 0.13 aAB | 87.48 ± 0.09 cAB | 87.72 ± 0.04 bA | 87.14 ± 0.05 dC | *** |
| FM05 | 88.82 ± 0.11 aA | 87.58 ± 0.11 bA | 87.72 ± 0.08 bA | 87.17 ± 0.01 cBC | *** | |
| FM10 | 88.76 ± 0.06 aA | 87.03 ± 0.09 cC | 87.52 ± 0.06 bAB | 87.34 ± 0.02 bA | *** | |
| FM15 | 88.36 ± 0.10 aB | 87.35 ± 0.01 bB | 87.39 ± 0.16 bB | 87.27 ± 0.10 bAB | ** | |
| FM20 | 88.43 ± 0.13 aB | 86.35 ± 0.00 cD | 86.95 ± 0.04 bC | 86.49 ± 0.01 cD | *** | |
| Sig. | * | *** | ** | *** | ||
| a* | FM00 | −3.34 ± 0.07 cC | −3.23 ± 0.01 bC | −3.24 ± 0.01 bcC | -3.09 ± 0.01 aD | * |
| FM05 | −3.39 ± 0.04 cC | −3.23 ± 0.00 bC | −3.24 ± 0.01 bC | -3.11 ± 0.00 aD | *** | |
| FM10 | −3.13 ± 0.01 bB | −3.10 ± 0.02 bB | −3.12 ± 0.03 bB | -2.99 ± 0.01 aC | ** | |
| FM15 | −3.03 ± 0.01 abA | −3.06 ± 0.03 bB | −3.13 ± 0.06 bB | -2.93 ± 0.01 aB | * | |
| FM20 | −3.07 ± 0.04 cAB | −2.86 ± 0.01 bA | −2.93 ± 0.01 bA | -2.74 ± 0.04 aA | ** | |
| Sig. | *** | *** | ** | *** | ||
| b* | FM00 | 7.07 ± 0.22 C | 7.17 ± 0.01 E | 7.50 ± 0.02 D | 7.26 ± 0.01 D | ns |
| FM05 | 7.66 ± 0.12 B | 7.84 ± 0.01 C | 7.62 ± 0.06 CD | 7.65 ± 0.02 C | ns | |
| FM10 | 7.75 ± 0.19 B | 7.63 ± 0.02 D | 7.71 ± 0.02 C | 7.74 ± 0.03 C | ns | |
| FM15 | 7.88 ± 0.19 AB | 8.03 ± 0.04 B | 7.93 ± 0.15 B | 8.05 ± 0.07 B | ns | |
| FM20 | 8.23 ± 0.09 A | 8.18 ± 0.08 A | 8.19 ± 0.04 A | 8.22 ± 0.04 A | ns | |
| Sig. | ** | *** | ** | *** | ||
| Organic Acids | Sample | Storage Period (Days) | Sig. | |||
|---|---|---|---|---|---|---|
| 1 | 7 | 14 | 21 | |||
| Malic acid | FM00 | 170.04 ± 4.48 a | 162.00 ± 3.75 a | 140.26 ± 24.35 a | 85.90 ± 4.80 b | ** |
| FM05 | 172.95 ± 13.04 a | 160.15 ± 7.30 ab | 143.38 ± 7.59 b | 97.16 ± 4.59 c | ** | |
| FM10 | 175.58 ± 12.49 a | 158.78 ± 6.67 a | 135.89 ± 3.22 b | 84.07 ± 2.79 c | *** | |
| FM15 | 172.06 ± 27.67 a | 145.30 ± 9.99 a | 133.16 ± 9.93 ab | 90.92 ± 3.56 b | * | |
| FM20 | 151.18 ± 60.23 | 140.41 ± 7.00 | 140.15 ± 20.69 | 94.84 ± 16.21 | ns | |
| Sig. | ns | ns | ns | ns | ||
| Lactic acid | FM00 | 12,054.45 ± 386.46 | 13,199.57 ± 439.39 | 11,670.45 ± 69.22 | 13,259.34 ± 772.00 | ns |
| FM05 | 12,190.02 ± 745.71 | 12,649.62 ± 457.11 | 12,940.65 ± 682.81 | 13,201.88 ± 258.74 | ns | |
| FM10 | 12,594.31 ± 660.92 | 13,308.98 ± 325.06 | 13,023.30 ± 572.64 | 13,599.24 ± 538.50 | ns | |
| FM15 | 12,172.29 ± 673.96 | 12,957.11 ± 327.71 | 12,758.84 ± 697.49 | 13,791.66 ± 1032.37 | ns | |
| FM20 | 12,403.72 ± 834.36 | 12,834.99 ± 693.17 | 12,700.44 ± 873.32 | 13,241.50 ± 621.44 | ns | |
| Sig. | ns | ns | ns | ns | ||
| Acetic acid | FM00 | 145.52 ± 10.07 | 178.84 ± 13.54 AB | 174.57 ± 36.91 | 206.50 ± 62.91 | ns |
| FM05 | 147.72 ± 8.44 | 145.12 ± 11.82 C | 150.16 ± 3.23 | 197.26 ± 47.44 | ns | |
| FM10 | 148.31 ± 18.07 | 162.24 ± 6.81 ABC | 158.50 ± 0.17 | 211.32 ± 59.54 | ns | |
| FM15 | 198.03 ± 52.69 | 189.65 ± 11.17 A | 189.85 ± 60.37 | 210.54 ± 52.73 | ns | |
| FM20 | 152.40 ± 9.98 b | 159.90 ± 7.18 bBC | 157.79 ± 18.49 b | 240.53 ± 19.09 a | * | |
| Sig. | ns | * | ns | ns | ||
| Propionic acid | FM00 | <LOD | <LOD | <LOD | <LOD | - |
| FM05 | <LOD | <LOD | <LOD | <LOD | - | |
| FM10 | <LOD | <LOD | <LOD | <LOD | - | |
| FM15 | <LOD | <LOD | <LOD | <LOD | - | |
| FM20 | <LOD | <LOD | <LOD | <LOD | - | |
| Sig. | - | - | - | - | ||
| Benzoic acid | FM00 | 19.90 ± 0.06 aA | 19.76 ± 0.16 a | 19.37 ± 0.09 b | 19.39 ± 0.03 bC | * |
| FM05 | 19.99 ± 0.00 A | 19.49 ± 0.26 | 19.71 ± 0.12 | 19.46 ± 0.04 BC | ns | |
| FM10 | 19.63 ± 0.06 B | 19.84 ± 0.20 | 19.63 ± 0.08 | 19.57 ± 0.06 B | ns | |
| FM15 | 18.65 ± 0.04 C | 20.25 ± 0.14 | 20.20 ± 0.85 | 19.75 ± 0.05 A | ns | |
| FM20 | 19.73 ± 0.02 aB | 20.34 ± 0.47 a | 20.28 ± 0.13 a | 17.07 ± 0.09 bD | *** | |
| Sig. | *** | ns | ns | *** | ||
| Parameters | Sample | Storage Period (Days) | Sig. | |||
|---|---|---|---|---|---|---|
| 1 | 7 | 14 | 21 | |||
| TPC (mg GAE/L) | FM00 | 76.35 ± 0.89 cC | 81.49 ± 0.67 bB | 92.51 ± 0.21 aA | 73.74 ± 0.21 dC | *** |
| FM05 | 80.92 ± 0.21 bBC | 79.18 ± 3.13 bB | 89.04 ± 0.67 abAB | 97.45 ± 7.31 aA | * | |
| FM10 | 93.51 ± 3.55 aA | 98.05 ± 6.25 aA | 81.24 ± 0.45 bC | 88.13 ± 1.96 abAB | * | |
| FM15 | 85.06 ± 1.11 bB | 83.25 ± 1.56 bB | 86.11 ± 1.52 bB | 91.55 ± 1.79 aA | * | |
| FM20 | 97.50 ± 4.41 A | 92.28 ± 8.33 AB | 90.21 ± 2.89 A | 80.07 ± 3.59 BC | ns | |
| Sig. | ** | * | ** | ** | ||
| Antioxidant capacity (IC50, mg/mL) | FM00 | 101.95 ± 4.46 A | 93.68 ± 21.05 | 88.93 ± 42.45 | 84.46 ± 5.87 B | ns |
| FM05 | 74.14 ± 7.34 BC | 84.46 ± 33.40 | 77.55 ± 27.62 | 67.92 ± 9.04 BC | ns | |
| FM10 | 96.25 ± 14.29 A | 92.27 ± 13.71 | 111.41 ± 18.60 | 115.01 ± 6.19 A | ns | |
| FM15 | 87.31 ± 4.98 AB | 72.30 ± 16.75 | 73.83 ± 11.37 | 56.69 ± 4.61 C | ns | |
| FM20 | 63.99 ± 6.04 bD | 58.00 ± 6.94 b | 85.96 ± 2.26 a | 57.26 ± 5.69 bC | * | |
| Sig. | * | ns | ns | ** | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hacıbayramoğlu, N.N.; Aktaş, H. Impact of Pea Fiber (Pisum sativum L.) on the Viability of Limosilactobacillus reuteri ACC27 and Quality Attributes of Fermented Milk. Fermentation 2026, 12, 189. https://doi.org/10.3390/fermentation12040189
Hacıbayramoğlu NN, Aktaş H. Impact of Pea Fiber (Pisum sativum L.) on the Viability of Limosilactobacillus reuteri ACC27 and Quality Attributes of Fermented Milk. Fermentation. 2026; 12(4):189. https://doi.org/10.3390/fermentation12040189
Chicago/Turabian StyleHacıbayramoğlu, Nisa Nur, and Haktan Aktaş. 2026. "Impact of Pea Fiber (Pisum sativum L.) on the Viability of Limosilactobacillus reuteri ACC27 and Quality Attributes of Fermented Milk" Fermentation 12, no. 4: 189. https://doi.org/10.3390/fermentation12040189
APA StyleHacıbayramoğlu, N. N., & Aktaş, H. (2026). Impact of Pea Fiber (Pisum sativum L.) on the Viability of Limosilactobacillus reuteri ACC27 and Quality Attributes of Fermented Milk. Fermentation, 12(4), 189. https://doi.org/10.3390/fermentation12040189






