3.2. Changes in the Algal Density and Algal Dissolution Rate—Phase 1
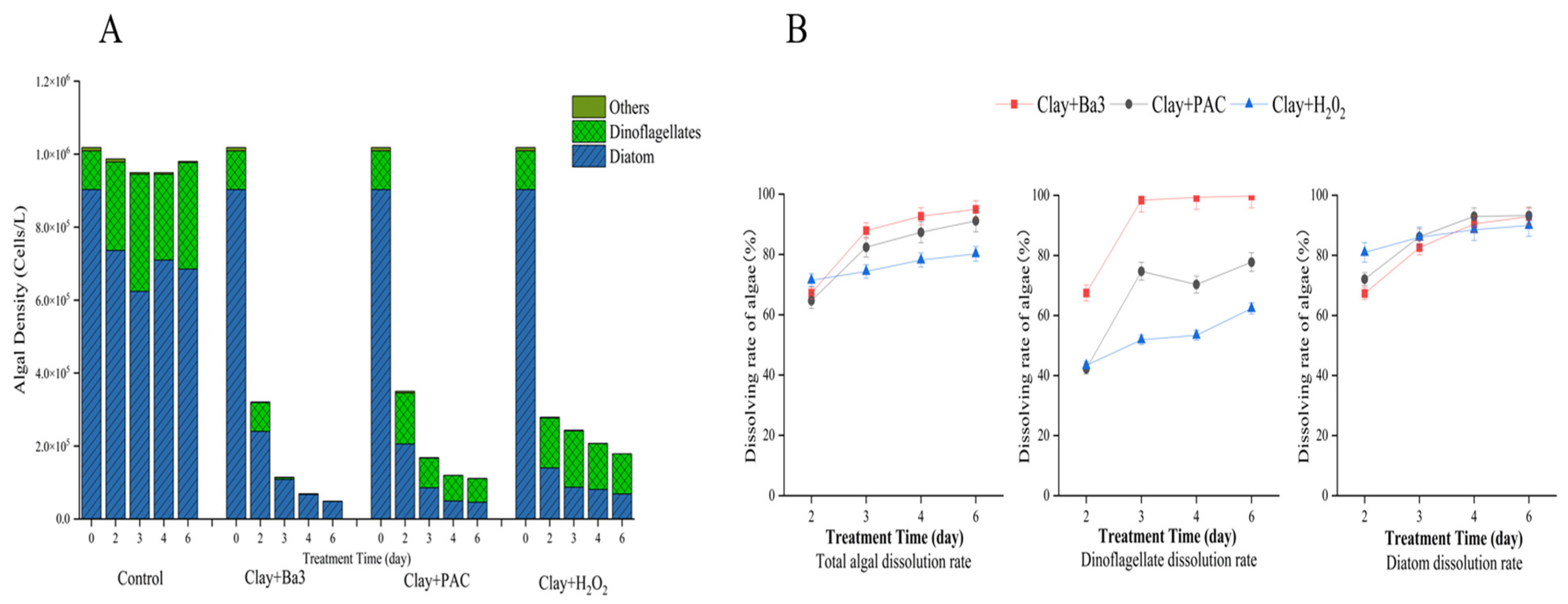
Figure 2A shows that diatoms and dinoflagellates account for 99.1% of the total density of algae; the density of chlorophyta and cryptophyta is very low. At the beginning of the experiment, the total algal density was about 1.0 × 10
6 Cells/L, the abundance of diatoms was 1.1 × 10
5 Cells/L, accounting for 88.7%, and dinoflagellates accounted for about 10.4% of the total algal density. The change in algal dissolution rate in the high-density treatment group is shown in
Figure 2B. For both total algae and dinoflagellates, the removal efficacy followed this order: clay + Ba3 broth > clay + PAC > clay + H
2O
2 and the algal dissolution rate in the clay + sterile fermentation broth group increased up to 95.0%. In the clay + H
2O
2 group, the algal dissolution rate was the highest on day 2 with a peak of 1.5%, consistent with laboratory results. The clay and H
2O
2 groups were able to rapidly precipitate algal cells in a short period. As the experiment concluded, the dinoflagellate algicidal efficiency of the clay + sterile fermentation broth group reached 99.8%. In terms of diatom removal effect, clay + PAC group > clay + sterile fermentation broth group > clay + H
2O
2. At the end of the experiment, the diatom dissolution rate of the clay + PAC group reached 93.3%. Statistical analysis results showed no significant difference in the rate of algal dissolution between total algae and diatoms in all experimental groups (
p > 0.05). However, there was a significant difference in the dissolution rate of dinoflagellates between the clay + sterile fermentation broth group and the other two groups (
p < 0.05). Compared to the other two groups, the clay + sterile fermentation broth group appeared to have the significant controlling effect. This selective mode of action that is cell wall composition-dependent is evidenced in the high removal efficiency on dinoflagellates (99.8%) as compared to that of diatoms (e.g.,
Chaetoceros spp.). Dinoflagellates that often have cellulose-containing thecae or are unarmed are more vulnerable to the enzymatic or surfactant-like metabolites of strain Ba3 than diatoms, which are defended by the rigid siliceous frustules. Specifically, the proteases and amylases present in the Ba3 broth break down the proteinaceous and polysaccharide coats of the cell wall of the dinoflagellate, and the lipopeptides initiate rapid membrane permeabilization of the cell wall. In comparison, the inflexible siliceous frustules of diatoms like
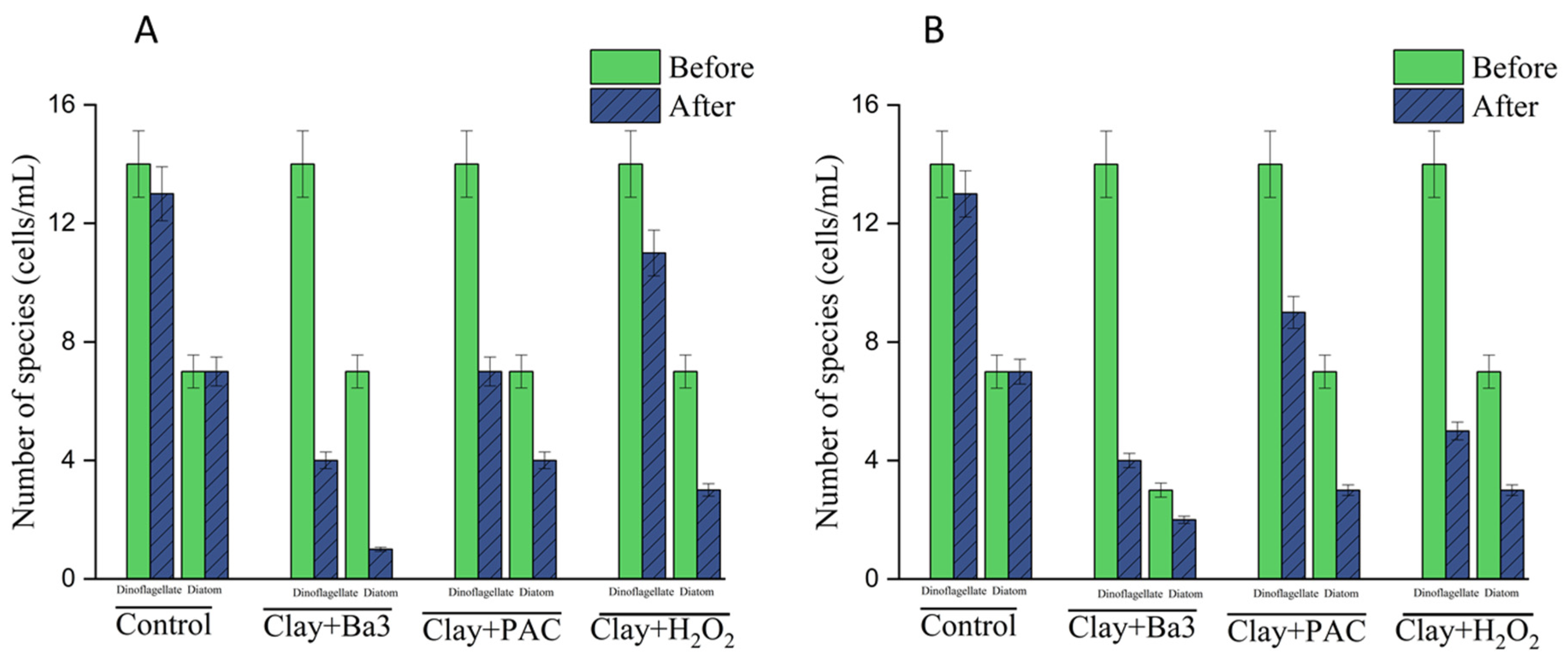
Chaetoceros spp. produce a physical shield against the cell that protects it against these particular attacks of the enzymes and surfactants, and hence the selective vulnerability observed. The synergy between clay and the Bacillus 3 sterile broth was observed and can probably be explained by the presence of clay particles used as physical carriers. By introducing flocculation, clay adsorbs bacterial metabolites and places these in direct and indirect contact with algal cell membranes, increasing the immediate and extended concentration of the algicidal compounds. Furthermore, the original mechanical stress or membrane deformation caused by the presence of clay may increase the vulnerability of dinoflagellates to the secondary metabolites contained in the broth, and this fact may lead to faster lysis when compared to the broth alone. Before the experiment, dinoflagellates accounted for 10.4% and diatoms for 88.7% of the total density of algae. After the experiment, clay + sterile fermentation broth group dinoflagellates and diatoms accounted for 11.1% and 88.9% of the total density, respectively. This stability in community structure was not observed in the other treatment groups.
The density of dinoflagellates and diatoms in the clay + PAC group was 45.9% and 54.1%. The density of dinoflagellates and diatoms in the clay + H2O2 group was 61.4% and 38.6% these percentages represent the community composition at the conclusion of the 6-day experimental period. Therefore, while total phytoplankton density decreased across all treatments, the clay + sterile fermentation broth combination more effectively maintained the diatom-dominant structure by achieving a higher selective removal rate (99.8%) for harmful dinoflagellates compared to the chemical treatments.
The density changes of each phyla of the unenriched group (group b) of algae are shown in
Figure 3A. Since the sum of the phytoplankton densities of the diatom phylum and dinoflagellate phylum accounts for 97.6% of the total algal density, and the density of the phylum chlorophyta and cryptophyta phylum is very low, and the green algae and cryptoalgae are still classified as “other” here. At the beginning of the experiment, the total algae density of group b was about 3.2 × 10
5 Cells/L, dinoflagellates accounted for about 13.2% of the total algae density, diatoms were still the absolute dominant taxa in algal water, with a density of 6.0 × 10
4 Cells/L, accounting for about 85.6%, and other algae accounted for about 1.3%.
Figure 3B shows the changes in the rate of algal dissolution in the control group. Compared with the high-density treatment group, the clay + sterile fermentation broth group still provided the best removal effect of total algae and dinoflagellates, with algicidal efficiencies of 91.8% and 98.6%, respectively. The algal lysis rate of algae and dinoflagellates showed the clay + H
2O
2 group > clay + PAC group, with the algae lysis rates of total algae and dinoflagellates at 87.0% and 63.4%, respectively. While the initial algicidal impact of the clay + H
2O
2 group was more pronounced than that of the clay + PAC group, the efficiency of the H
2O
2 combination declined significantly when applied to higher algal cell densities. In contrast, the clay + sterile fermentation broth treatment demonstrated a robust removal effect regardless of initial biomass. Notably, the algicidal efficiency observed in the alga-enriched (high-density) groups was significantly higher than that of their respective control groups, suggesting that the synergistic biological–physical method is particularly effective under the high-density conditions typical of active red tide blooms.
Before the experiment, dinoflagellates accounted for about 13.2% of the total algal density, and diatoms accounted for 85.6% of the total algal density. Following the experiment, the dinoflagellates in the clay + sterile fermentation broth group accounted for 5.7% of the total algal density, and the diatoms accounted for 94.3% of the total algal density; the clay + PAC group accounted for 69.8% of the total algal density. In the clay + H2O2 group, dinoflagellates accounted for 45.8% of the total algal density, and diatoms accounted for 54.2% of the total algal density. Compared to the proportions present before the experiment, the proportion of dinoflagellates in the total algal density in the clay + sterile fermentation broth group decreased; however, the proportion of diatoms increased. When the proportion of dinoflagellates in the clay + PAC group and clay + H2O2 group increased, the results were consistent with group a, so the combination of clay + sterile fermentation broth likely provided a desirable effect on the maintenance and improvement of community structure. In this experiment, the clay + sterile fermentation broth group had the best removal effect on dinoflagellates, especially for Karenia spp., Alexandrium spp., and Prototheca spp., which was in line with the research in the laboratory of strain Ba3 on red tide. The growth effect of dinoflagellates was consistent, but the removal effect of the clay + sterile fermentation broth group on diatoms was poor, especially for Chaetoceros spp. This absence of the removal of diatoms like Chaetoceros spp., in favor of dinoflagellates, 99.8%, suggests that the operational mechanism may be selective and seems to be linked to dissimilar cell wall compositions. The dinoflagellates (often with cellulose-containing thecae or no covering at all) can be more susceptible to the biosurfactants or extracellular enzymes of the Ba3 broth, but the diatoms, which use stone-like siliceous frustules to stiffen their cell surfaces, are relatively more resistant.
3.4. Comparative Efficacy with Benthic Interaction Analysis of Algicidals
The density changes in each category in this experiment are shown in
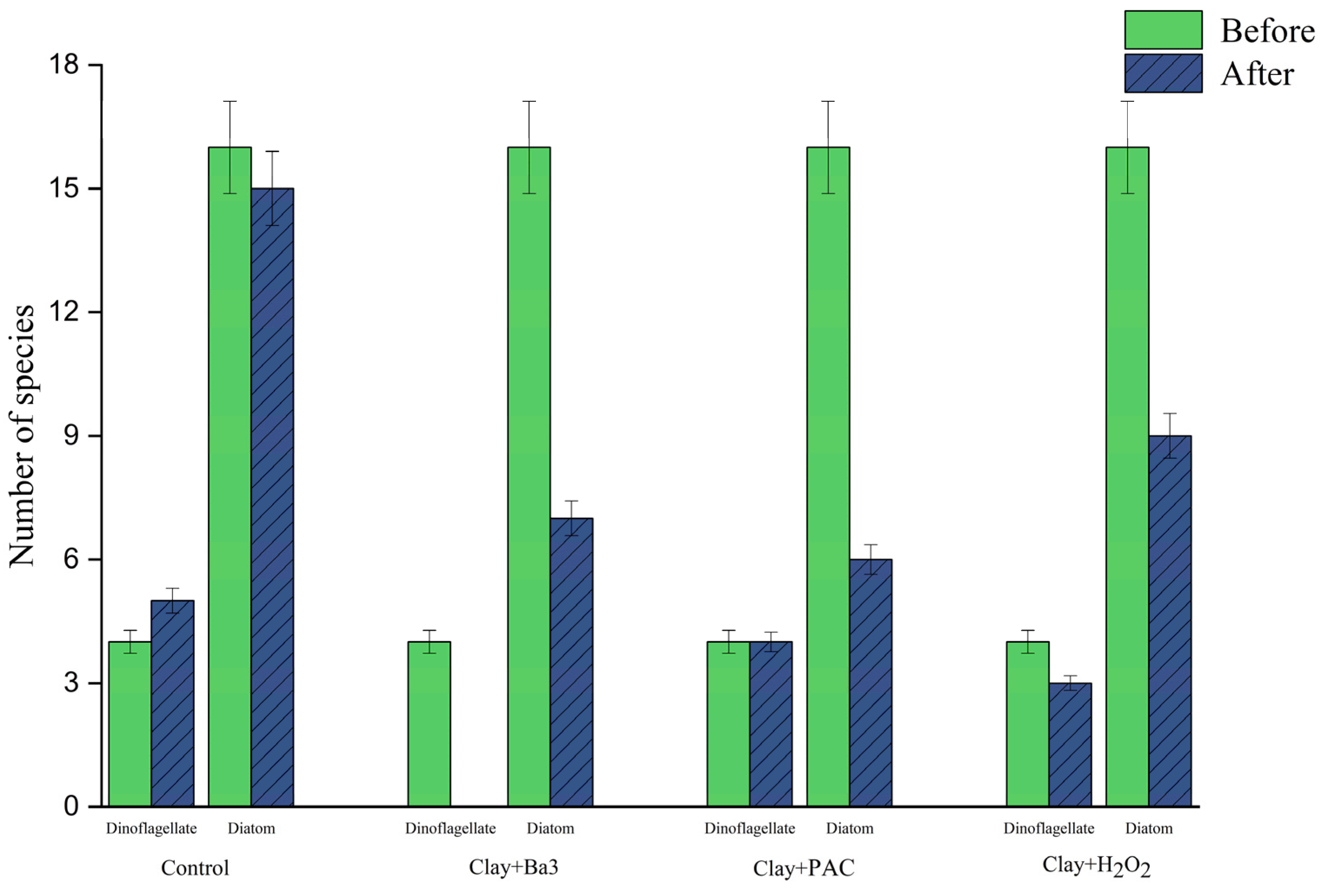
Figure 5A. The total phytoplankton density was approximately 7.3 × 10
5 Cells/L, with dinoflagellates accounting for about 4.9% of the total algal density. The diatoms dominated the algal water, with an abundance of 6.92 × 10
5 cells/L, accounting for approximately 94.4%, while other algae (Dinoflagellate, Chlorophyta, and Cryptophyta) constituted 0.7%. Because the investigational period of the in situ water sample algal removal experiment began in early June, whereas the investigational period of the in situ water sample with sediment algae removal experiment began in early July, the proportion of algae in the total algae density decreased, and the proportion of diatoms increased when compared with the in situ water sample (without the sediment experiment). The algicidal efficiency change is shown in
Figure 5A,B. In terms of the overall algal removal effect, the clay + PAC > clay + sterile fermentation broth group > clay + H
2O
2 group, and the clay + PAC group displayed the highest algicidal efficiency 88.1%, which varied significantly from the experimental results of the unassembled sediment group. However, there is the need to differentiate between the elimination of total phytoplankton and the elimination of desirable harmful species. Whereas clay + PAC was more effective in removing total algae, including beneficial diatoms, the taxonomic selectivity of the clay + Ba3 broth treatment was larger. It eliminated the target harmful dinoflagellates, both
P. donghaiense and
K. mikimotoi, with 100% success and also allowed the diatom community to flourish and eventually recover. This choosiness is desirable in aquaculture situations in order to avoid sterilizing the whole ecosystem. The clay + sterile fermentation broth was the most effective method of dissolving algae in the non-sediment experiment. The sterile fermented broth and sediment group produced a dissolving rate of algae in 6 d at a 75.4% decline, and after an 8 d lysis rate, which was only 61.3%, far below the clay + PAC group. At the same time, the algal dissolution rate of the clay + H
2O
2 group reached 68.9% on the second day and then continued to decrease to 28.2% on the eighth day. The trend of the algicidal efficiency of diatoms was almost the same as that of the total algae. Therefore, the variation trend in dinoflagellate density was consistent with the results of the algal removal experiment without sediment, and the dissolution rate of dinoflagellates was as follows: clay + sterile fermentation broth group > clay + PAC group > clay + H
2O
2 group. At the end of the experiment, the dinoflagellate dissolution rate of the clay + sterile fermentation broth group reached 100%, followed by the clay + PAC group at 66.8% and the clay + H
2O
2 group at 41.3%.
Before the experiment, dinoflagellates accounted for about 4.9% of the total algal density, and diatoms accounted for 94.4% of the total algal density. After the experiment, the dinoflagellates in the clay + sterile fermentation broth group accounted for 0% of the total algal density and the diatoms accounted for 100% of the total algal density; the clay + PAC group accounted for 15.5% of the total algal density. In the clay + H2O2 group, dinoflagellates accounted for 4.5% of the total algal density, and diatoms accounted for 95.5% of the total algal density. Compared with the data taken before the experiment, the density of dinoflagellates in the clay + sterile fermentation broth group was reduced to 0%, leaving only diatoms, while the dinoflagellate ratios of the clay + PAC group and clay + H2O2 group all differed significantly.
Compared to the experiment in which algae were removed without sediment, the clay + sterile fermentation broth group consistently removed dinoflagellates, while the clay + sterile fermentation broth group did not remove diatoms, especially when the assembled sediment was removed. In the algal experiment, the density of diatoms recovered from 1.8 × 10
5 Cells/L on the sixth day to 2.7 × 10
5 Cells/L. The recovery in this diatom density is possibly explained by two distinct processes: first, the biochemical breakdown of Ba3 metabolites by the end of day 8, thereby decreasing their inhibitory pressure, and, secondly, a local release of nutrient, silicate or nitrogen, by the benthic sediment that selectively enhances the growth of the surviving resilient diatom populations against the lysed dinoflagellate. The discriminating effect is needed to maintain a positive phytoplankton community structure and prevent the complete sterilization of the entire ecosystem in the process of HAB control. Multiple reports on the algicidal bacteria point to the existence of certain algal-lytic activities; we therefore presume that strain Ba3 will have strong lytic activity against a particular algal taxon and be ineffective against others. The co-culture with the ba3 sterile fermentation broth and clay is the only way to efficiently kill most dinoflagellates but leave a group of desirable diatoms to exist in the aqueous environment, which results in a better structure of the entire phytoplankton community. In order to support these observations, more studies are needed. Acute toxicity tests showed no objectionable effects on
Brachionus plicatilis, Artemia salina, and
Oryzias latipes; at the same time, long-term ecological safety was assessed with the help of benthic monitoring. The fact that the increase in sediment total organic carbon (TOC) and total nitrogen (TN) was not significant after the deposition of clay–alga flocs (as illustrated in
Supplementary Figure S16) shows that the treatment is not causing localized hypoxia of benthic species or nutrient overload. Furthermore, the application of biodegradable bacterial metabolites minimizes the possibility of disrupting the native microbial community in the long run when compared to the use of the indestructible chemical algaecides.
3.5. Water Safety and Environmental Quality
Temperature (avg. 27.4 °C), salinity (~30–33), and pH (~8.1) remained stable across all groups with no statistically significant differences (
p > 0.05). The changing of water temperature in the in situ water sample algal control experiment is shown in
Supplementary Figure S2. During the experiment, the water temperature in the experimental box changed from 24.1 °C to 28.8 °C, with an average water temperature of 27.4 °C. Typically, the water temperature outside the experimental box was lower than the temperature inside the box. On the sixth day of the outdoor experiment, the weather was cloudy, which resulted in a lower temperature, but no extreme weather changes affected the experiment during the investigation. The water temperature of the control group was higher than that of the experimental group, which may be due to the effect of the treatment and the algae in the experimental group or the endothermic hydrolysis reaction, resulting in a lower water temperature than that of the control group.
The temperature changes of groups a and b with different algal cell densities showed no significant variance in temperature changes between the two groups (
p > 0.05).
Supplementary Figure S3 shows the water temperature change in the in situ water sample assembly sediment algal control experiment. During the experiment, the water temperature in the experimental box changed from 26.8 °C to 28.4 °C, with an average water temperature of 27.3 °C. The water temperature outside the experimental box tended to be lower than that inside the box (experimental group).
Supplementary Figure S4. shows the change of salinity in the in situ water sample algal control experiment. The initial salinity of group a and group b was 30. The salinity outside the experimental box fluctuated greatly. There was a downward trend in the experimental period, which may have been caused by the intermittent rainfall and increased cloud cover on the sixth day and the resulting lower water temperature. The salinity variation trends in the two groups of different algal cell densities were basically the same. Therefore, there was no sharp change in salinity during the experimental period, which led to the death of algal cells.
Supplementary Figure S5 shows the change in salinity in the experiment with the in situ water sample assembly for algal control in sediment. The salinity of each group increased to 33 and overlapped during the experiment due to wind and climatic effects. There was no significant difference in the change in salinity between the control group and each experimental group (
p > 0.05), and the assembled sediment did not affect the salinity of the water body but had a significant impact.
Supplementary Figure S6 shows the change in DO concentration in the in situ water sample algal control experiment. The initial DO in the experiments of group a and group b was 8.2 mg/L and 8.5 mg/L. At the end of the experiment, the DO values of the clay + H
2O
2 algal control method increased most evidently, which is likely because the reaction products of H
2O
2 and algae were water and oxygen; the DO values of the clay + PAC group increased slightly, but the change range was small. The DO value of the clay + sterile fermentation broth group was slightly lower than that of the control group, and the a and b groups decreased to 7.9 mg/L and 7.7 mg/L, respectively, which was consistent with the laboratory algal control results.
Supplementary Figure S7 shows the change in DO concentration in the algal control experiment of in situ water sample assembly in sediment. The initial DO in the experiment was 6.9 mg/L, which was lower than the initial DO value of 8.45 mg/L in the experimental group without sediment. At the end of the experiment, the DO value of the clay + H
2O
2 algal control method most noticeably increased, reaching 8.9 mg/L, which was consistent with the conclusion of the algal control experiment without the bottom mud. In the algal control experiment involving assembling the sediment, the DO value of the clay + sterile fermentation broth group did not decrease at the end of the experiment but stayed higher than that of the control group. These conditions are closer to how sediment floats and mixes with the water body in rainy weather. This experiment shows that the combined method of clay and sterile fermentation broth to control algae does not have an adverse effect on the DO concentration in the water body at this dosage.
Supplementary Figure S8 shows the turbidity change in the in situ water sample algal control experiment. The initial turbidity of the experiments in groups a and b was 5.9 NTU and 5.2 NTU. Because the experimental area is in an aquaculture area, the turbidity of the water body is affected by the swimming of cultured organisms, in effect, so the initial turbidity is higher. At the end of the experiment, the turbidity of the experimental group was lower than that of the control group, indicating that the three algal control methods had a desirable effect on the turbidity of algal water. However, there was no significant difference in the turbidity change between the experimental groups (
p > 0.05). The turbidity removal rate of group a was as follows: clay + sterile fermentation broth group (78.3%) > clay + PAC group (76.8%) > clay + H
2O
2 (75.8%); the turbidity removal rate of group b was shown as clay + PAC group (81.9%) > clay + sterile fermentation broth group (80.7%) > clay + H
2O
2 (72.3%). It should be noted that under field conditions, turbidity may also be affected by factors such as wind power and organisms in the breeding area. Overall, the clay + sterile fermentation broth group has a better removal effect on the turbidity of algal water in the process of algal control.
Supplementary Figure S9 shows the turbidity change in the in situ water sample assembly in the sediment algal control experiment. The initial turbidity in the experimental box was 5.3 NTU. Clay + sterile fermentation broth group > clay + H
2O
2 group, the turbidity changes between clay + PAC groups were significantly different (
p < 0.05), and the turbidity removal rate was 58.6%. The diatom density in the group rebounded, so the turbidity removal rate was lower than that in the clay + PAC group, consistent with the changing trend in algal density in each experiment.
The pH of seawater generally fluctuates between 8.0 and 8.5, and the pH of surface seawater generally remains around 8.1 ± 0.2.
Supplementary Figure S10 shows the pH changes in the in situ algal control experiment. The initial pH of the experiments in groups a and b was 8.2 and 8.1. At the end of the experiment, the pH of the control group remained unchanged, while the three experimental groups decreased slightly. Among them, the clay + PAC group had the most significant pH drop in which the pH value of group a dropped to 7.7 and the pH value of group b dropped to 7.8. The smallest drop was shown by the clay + sterile fermentation broth group; the pH value of group a dropped to 7.9, and the pH value of group b dropped to 7.9. The algal water with two different algal cell densities showed the same trend of pH change in each experimental group. There was no significant difference in pH value change among each experimental group (
p > 0.05). In the laboratory simulation experiment, the effect of strain Ba3 on the pH value of the water body was negligible, and the in situ algal control experiment also showed a similar effect on algal removal compared to the other two algal control methods, clay + sterile fermentation broth.
The solution used contains nutrients suitable for the survival of certain bacteria, and the growth of some bacteria can affect the change in pH value.
Supplementary Figure S11 shows the pH value change in the in situ water sample assembly sedimentary sediment control algal control experiment. The initial pH value of the experiment was 8.2; at the end of the experiment, the pH of the control group increased slightly. There was a slight decrease in the three experimental groups. The pH value decreased the most in the clay + sterile fermentation broth, with the lowest pH value at 7.7; the pH value decreased the least in the clay + H
20
2 group, with the lowest value at 8.0. The effect on the pH value of the water body was the most limited. However, in the experiment after assembling the sediment, the pH value of the clay + sterile fermentation broth group fluctuated greatly.
Supplementary Figure S12 shows the results of changes in TP during the in situ water algal control experiment. The TP concentration of the control group fluctuated at around 0.14 mg/L in the algal water containing two different algal densities. There was no significant difference in the changes in TP concentration between the control group, the experimental group (
p > 0.05), and the other experimental groups. The TP concentration decreased most significantly in the clay + PAC group, from 0.15 mg/L to 0.09 mg/L in group a, at a removal rate of 41.7%; in group b, it decreased from 0.14 mg/L to 0.09 mg/L, at a removal rate of 41.7% and 38.0%, followed by the clay + fermentation broth group. The removal rates of TP at the end of the experiment were 23.7% and 23.2%, respectively; the clay + H
2O
2 group had the worst removal effect on TP and the TP concentration during the experiment was the same as the control group. Similarly, the changing trend in TP with different algal cell densities in the two groups was the same.
The change result of SRP is shown in
Supplementary Figure S13. During the experiment, the SRP concentration of the control group in the algal water with two different algal densities was about 0.12 mg/L; the most apparent decrease in the SRP concentration was still in the clay + PAC group, and then group a decreased from 0.122 mg/L to 0.004 mg/L with a removal rate of 96.7%; group b decreased from 0.119 mg/L to 0.007 mg/L, with a removal rate of 94.1.0%. The clay + sterile filtration group followed with a removal rate of 32.3% and 29.7%, respectively; however, the SRP in the clay + H
2O
2 group showed an upward trend, which was always slightly higher than that in the control group during the experiment. The variation trend of SRP in the two groups with different algal cell densities was basically the same.
Supplementary Figure S14 shows the results of changes in TP during the in situ water sample assembly process for the sediment control algal control experiment. The initial TP concentration of the experiment was 0.14 mg/L. At the end of the experiment, the TP concentration of the control group and each experimental group changed significantly (
p < 0.05). The TP concentration of the control group increased to 0.55 mg/L, and in each experimental group, the TP concentration in the water body increased. For the control group, each experimental group had a specific removal effect on TP, which showed that the clay + PAC group (61.2%) > the clay + fermentation broth group (58.4%) > clay + H
2O
2 group (31.4%).
The change effect of SRP is shown in
Supplementary Figure S15. The initial SRP concentration of the experiment was 0.12 mg/L. At the end of the experiment, there were significant differences in the changes in SRP concentration between the control group and the clay + PAC group and the clay + bacteria fermentation broth group (
p < 0.05), but there was no significant difference from the clay + H
2O
2 group (
p < 0.05). During the experiment, the SRP of the clay + H
2O
2 group increased more than that of the control group, which was consistent with the experimental results of the algal removal experiment without the sediment. The clay + fermentation broth and clay + PAC groups significantly impacted SRP, especially in the clay + PAC group. The SRP removal rate of the clay + PAC group was 98.3% at the end of the experiment; the SRP removal rate of the clay + fermentation broth group was 52.1% at the end of the experiment.
After the in situ water sample assembly sediment algal control experiment, the total organic carbon (TOC) and total nitrogen (TN) content of the sediment samples were measured, and the results are shown in
Supplementary Figure S16. The TOC and TN contents in marine sediments were generally less than 2.0%. In this experiment, the TOC content of the control group was 1.26%; the TOC of the clay + sterile fermentation broth group, the clay + PAC group, and the clay + H
2O
2 group was 1.23%, 1.12%, and 1.22%, respectively. The experimental group contained a TOC content lower than the control group, but there was no significant difference in the TOC content between the control group and each experimental group (
p > 0.05). The TN content of the control group was 0.1%, and the TN content of the clay + sterile fermentation broth group, the clay + PAC group, and the clay + H
2O
2 group were 0.09%, 0.07%, and 0.08%, respectively. The TN content of each experimental group was also slightly lower than that of the control group. Therefore, if maintained at the dosage used in this study, the sterile fermentation broth + clay combined algal control method will likely not increase the carbon and nitrogen load of the sediment. Although the temperature and salinity levels remained constant in the 90 L enclosures, which facilitated consistency of the experiment, there were some observed container effects, specifically, the slightly higher internal temperatures as compared to the ambient bay water, which should be taken into account when applying this technology on a larger scale. Vertical mixing and thermal dissipation in the open waters should be stronger compared to the occurrence in the closed enclosures. Subsequent large-scale experiments ought to be put in place for such hydrographic differences to ensure that the duration of contact between the clay–broth flocs and the target dinoflagellates is sufficient in varying current and temperature conditions.