3.1.3. Fourier Transform Infrared Spectroscopy (FTIR)
FTIR was employed to elucidate the chemical structure of bacterial cellulose (BC) before and after filtration of synthetic effluents, providing complementary insights to the structural data obtained from XRD and EDS. The transmittance spectrum of the untreated (as-synthesized) BC, presented in
Figure 3, exhibits distinct absorption bands that confirm the preservation of both the structural integrity and functional groups characteristic of the cellulose matrix. The peak at 3340 cm
−1 corresponds to the O–H stretching vibration associated with hydrogen bonding, reflecting the material’s hydrophilic nature and its capacity for water interaction [
40]. The band at 2895 cm
−1 is attributed to the asymmetric stretching of –CH
2 groups, characteristic of the polymeric backbone [
41]. Additionally, the absorption observed at 1654 cm
−1 is likely related to the bending vibrations of adsorbed water molecules [
42]. In the spectral region between 1500 and 1000 cm
−1, a series of well-resolved absorption bands characteristic of cellulose is evident. These correspond to vibrational modes associated with the glycosidic ring structure, including C=C stretching, C–H and O–H bending, as well as C=O and C–O–C stretching vibrations. A detailed assignment of these bands is provided in
Table 2 [
43].
Following the filtration of the ES1 effluent, distinct changes were observed in the FTIR spectrum, characterized by more intense absorption peaks and reduced transmittance in specific bands (
Figure 4). In addition to changes in transmittance at 3343 and 2854 cm
−1, a new absorption band emerged at 2922 cm
−1, attributed to the asymmetric stretching vibration of C–H groups. This feature suggests the incorporation of lipophilic residues into the BC matrix during the filtration process (
Table 3). According to Balistreri et al. [
41], increases in peak intensity, area, and sharpness are indicative of a higher density of chemical bonds, which is consistent with the fact that OCB2 contains functional groups similar to those found in native BC [
14]. Furthermore, previous studies indicate that BC membranes are capable of assembling at the oil–water interface, promoting micelle stabilization and influencing the interactions between the polymer and surrounding components. This interfacial behavior may have promoted a reorganization of the hydrogen-bonding network within the BC structure [
44].
A comparison between the FTIR spectra of membranes exposed to ES1 and ES2 revealed no significant differences, as both effluents contained the same oil (OCB2) as the primary contaminant. Sodium chloride (NaCl), a simple ionic compound, does not exhibit characteristic absorption bands in the mid-infrared region due to the absence of oscillating dipole moments in its ionic bonds [
45].
As such, NaCl is commonly used as an infrared-transparent window material in FTIR spectroscopy. Any spectral features related to NaCl can typically only be observed indirectly, such as through its interactions with water molecules or organic compounds. Consequently, the direct detection of NaCl via FTIR is inherently limited [
45]. Therefore, the spectral modifications observed are more directly attributed to the presence of oil in the system, which is likely responsible for the interactions with the polymer matrix. The corresponding peaks are illustrated in
Figure 5 and listed in
Table 4.
Considering the spectral changes observed after filtration of oily effluents and the absence of significant structural alterations in the membrane despite the presence of surfactants or saline components, it can be concluded that BC demonstrates structural resilience under such conditions. This highlights the membrane’s substantial potential for treating industrial effluents containing synthetic surfactants and for use in high-salinity environments such as seawater, where it ensures effective separation of oil residues.
3.1.4. Thermogravimetric Analysis (TGA)
TGA was conducted to assess the thermal stability of BC membranes before and after the filtration of synthetic effluents containing oil (BCO) and oil with salt (BCOS). The curves shown in
Figure 6 illustrate mass loss as a function of temperature under an inert atmosphere, revealing distinct thermal profiles among the samples.
The BC membrane (green), composed solely of purified cellulose, exhibited a thermal profile characteristic of cellulose-based materials. An initial mass loss of approximately 5% was observed up to 120 °C, attributed to the evaporation of physically adsorbed moisture. The main degradation phase occurred between 250 and 370 °C, with a significant mass loss of around 52%, associated with the cleavage of glycosidic bonds and the subsequent pyrolysis of the cellulose structure [
46]. The final residual mass at 600 °C was 25.2%, consistent with its predominantly organic composition and the absence of inorganic components.
Thermal analysis of the BCO sample (blue), following filtration of fuel oil-containing effluent (70 mg/L), revealed a shifted degradation pattern. The main mass loss began at an earlier temperature, around 220 °C, resulting in a total weight loss of approximately 96.6%. The final residual mass was only 3.4%, highlighting the high volatility of the retained hydrocarbons. These compounds act as thermal plasticizers, increasing molecular mobility and facilitating the degradation of the cellulose matrix. According to Rutkowski and Kubacki [
47], the presence of oil promotes a more pronounced and homogeneous decomposition, favoring complete pyrolysis of the composite material. This finding is particularly relevant for potential post-filtration applications involving thermal regeneration or energy recovery of membranes saturated with hydrocarbon-based contaminants.
In contrast, the BCOS sample (red), which filtered the effluent containing oil and 35 g/L of sea salt, retained high thermal stability throughout the heating process.
Degradation of the cellulose matrix occurred more gradually, with the main mass loss taking place between 260 °C and 400 °C. This behavior is attributed to the presence of NaCl, an inorganic compound with a high melting point (801 °C), which remains stable at elevated temperatures and contributes directly to the residual mass. Additionally, NaCl may promote the formation of carbonaceous residues by hindering the diffusion of volatile degradation products, thereby limiting their release during pyrolysis [
48,
49]. This synergistic effect between oil and salt results in a membrane that is more resistant to thermal degradation, yet also more persistent in terms of solid residue accumulation.
The low residual mass of the BCO sample indicates potential for efficient thermal disposal, whereas the high thermal stability of the BCOS sample underscores the need for specific treatment routes, such as controlled incineration or reuse in composite matrices [
50].
Moreover, the multiple degradation stages in the BCO and BCOS curves indicate that contaminants significantly alter the structure and degradation kinetics of BC, possibly promoting the formation of intermediate or residual phases. These findings contribute to a deeper understanding of the saturation mechanisms and may inform the development of environmentally appropriate strategies for the regeneration or disposal of BC membranes used in contaminated industrial settings.
3.1.5. X-Ray Diffractometry (XRD)
This analysis revealed significant structural changes in the BC membranes following their use in the filtration of oily and saline emulsions. As shown in
Figure 7, the pure BC sample exhibited well-defined diffraction peaks at 2θ ≈ 15.16°, 17.44°, and 23.35°, corresponding to the (100), (010), and (110) planes of cellulose Iα. These reflections confirm the high initial crystallinity of the membrane. The CI of 67.36% further confirms the organized structure of the pure membrane, which is consistent with values typically observed in BC samples synthesized under homogeneous conditions and in the absence of contaminants [
51].
In the sample filtered with ES1 effluent, the characteristic cellulose peaks showed a pronounced decrease in intensity, accompanied by the emergence of new reflections at 2θ ≈ 14.55°, 16.68°, 23.35°, 28.82°, and 31.45°, consistent with the incorporation of lipid fractions into the polymeric matrix. The 10% reduction in CI reflects a structural alteration within the crystalline domains. This change likely arises from interactions between polymer chains and bulky molecules, such as oil, which disrupt the packing of ordered regions and lead to a partial loss of structural organization [
17]. Similarly, Medeiros et al. [
15] demonstrated that reduced crystallinity compromises membrane hydraulic permeability, as contaminant accumulation within the pores decreases water affinity and hinders fluid transport through the membrane matrix.
The membrane used for filtering the ES2 effluent exhibited, in addition to cellulose and oil-associated peaks, a high-intensity peak at 2θ ≈ 31.8°, consistent with crystalline salts, likely chlorides or metallic carbonates, originating from the effluent itself [
52]. The presence of these salts suggests that inorganic contaminants have deposited on the surface and within the pores of the membrane, potentially accelerating structural degradation during prolonged use. This structural alteration aligns with the findings of Barud et al. [
51], who reported that the introduction of bulky or nonpolar groups hinders cellulose chain packing, increases interfibrillar distance, and results in less-defined diffraction patterns, an effect analogous to that induced by the adsorption of ions or hydrophobic molecules within the matrix.
The reduction in crystallinity, coupled with lipid retention and salt accumulation, substantially alters the membrane’s microstructure, impairing both water transport capacity and mechanical integrity. These combined effects compromise long-term functional performance, underscoring the importance of implementing periodic regeneration strategies or planned replacement to ensure efficiency and extend the service life of oil–water separation systems.
3.1.6. Optical Microscopy
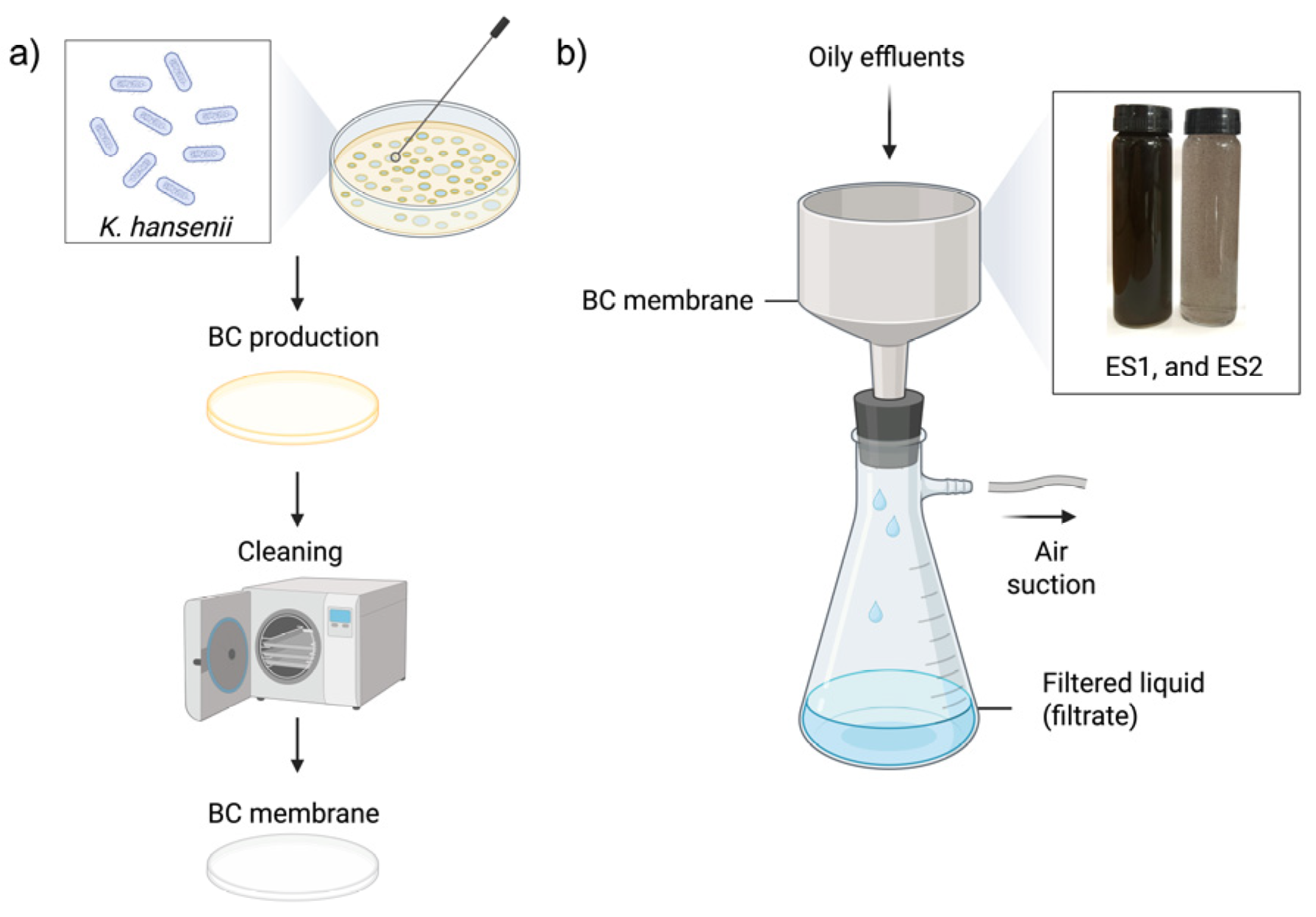
Upon completion of the filtration process, the membranes were analyzed for signs of water loss, mechanical deformation, and retained contaminants. The membranes were then cut to specific dimensions for mechanical testing (7.0 × 2.5 cm) as shown in
Figure 8 and taken for optical analysis.
Macroscopic evaluation of the “BC” sample revealed that drying occurred in a non-uniform manner. As moisture was lost, localized shrinkage occurred, leading to fiber bundling and the formation of elevated, darker regions across the membrane surface. These aggregated zones became increasingly evident under higher magnification, suggesting microstructural reorganization during dehydration (
Figure 9).
The progression of the images reveals an initially homogeneous surface with slight porosity and subtle undulations, evolving into a more compact and wrinkled structure at higher magnifications. The presence of parallel lines and grooves indicates the superficial entanglement of cellulose nanofibrils, characteristic of the three-dimensional organization of bacterial cellulose (BC). The topography becomes more evident, with marked elevations and depressions formed during the drying process, highlighting the structural complexity of the membrane and its potential for applications in filtration and molecular load support [
14,
53].
In the BCO membrane (
Figure 10), the effect of vacuum pressure is evident, as shown by the grooves matching the shape and dimensions of the Büchner funnel pores. Additionally, the membrane exhibits a significantly darker coloration, particularly within these deformed regions, suggesting the localized accumulation of contaminants driven by suction during filtration.
The region selected for higher magnification (
Figure 10A) corresponds to an imprint left by one of the pores of the Büchner funnel on the BCO. In this region, the cellulose fibers appear more densely packed, likely due to the mechanical pressure exerted during the vacuum filtration process. In these regions, smaller oil molecules likely became trapped between cellulose fibers during filtration and, upon drying, acted as adhesive agents, reinforcing the local compaction caused by mechanical deformation and further reducing inter-fiber spacing [
14].
In
Figure 10B, a micro-tear in the membrane’s superficial layer is evident, likely caused by vacuum-induced mechanical stress during filtration. The torn segment remained compact during filtration, entrapping contaminants, and only became evident during the subsequent drying step.
Figure 10C corroborates localized fouling: particulate material is confined within the interfibrillar network, and the more pronounced yellow discoloration indicates residual oil. Despite partial surface damage (see
Figure 10B), the BC nanofibrillar architecture retains its selective retention capability via capillary confinement of hydrophobic species [
54]. In addition, the localized yellowish discoloration observed in
Figure 10C, more pronounced than in the preceding images, can serve as a qualitative marker of membrane saturation, supporting the need for regeneration or scheduled replacement after a defined number of operating cycles.
When examining the BCOS membrane (
Figure 11), distinct patterns are observed. The principal effect reflects the effluent composition rather than intrinsic membrane properties. The emulsion’s high salinity, compounded by concentration during filtration and drying, drives NaCl crystallization and the accumulation of oil–salt deposits on the membrane [
55].
Figure 11A shows whitish surface deposits consistent with crystalline NaCl aggregates concentrated at high-salinity sites on the membrane. Unlike BCO, the Büchner-imprint grooves are not evident, indicating that in situ salt crystallization with oil co-deposition dominates the post-filtration surface topography. These localized deposits induce internal stresses and compromise surface integrity [
56,
57,
58]. High-magnification images (
Figure 11C) confirm NaCl crystals with adherent oil forming localized fouling deposits, which explains the altered morphology after drying [
58,
59].
Oil–salt micro-aggregates appear to deposit within the cellulose fiber network, hindering uniform water evaporation and creating locally compacted, stiff regions. This heterogeneous fouling produces opaque, textured surface zones and can degrade performance in subsequent filtration cycles by altering porosity, flexibility, and rehydration capacity [
60].
The micrographs show that BC membranes retain particles spanning diverse sizes, morphologies, and chemistries, even under vacuum-induced pressure. Superficial micro-tears associated with deformation at Büchner-pore imprints were observed, but no through-thickness rupture occurred. Oil, alone or combined with salts, promotes localized pore occlusion and fiber compaction without compromising overall structural integrity [
61,
62].
3.1.7. Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS)
To better observe the surface morphology of the membranes before and after the filtration process, scanning electron microscopy (SEM) was performed. This test enabled us to visualize how the presence of contaminants affected the fibers and the 3D spatial organization of the membrane, as well as how the permeability of the effluents penetrated the membrane fibers in different ways, thereby allowing us to trace a membrane saturation profile in the presence of various types of effluents.
As can be seen in
Figure 12A–H, the lower the applied magnitude, the more uniform the membrane appears. However, at 200× magnification (B), we can already perceive the overlapping nature of the membrane fibers, with a rough, wave-like appearance [
63].
The wide-field images (
Figure 12E–H) confirm the nanometric profile of the bacterial cellulose membrane, with an average fiber diameter of approximately 87 nm. The non-uniform fiber formation, a result of the microorganisms’ metabolic activity and growth patterns, leads to the development of an interwoven three-dimensional network. This unique morphology promotes the creation of a porous mesh with high surface area, enabling efficient retention of larger molecular contaminants such as oils and greases, even under flow and pressure conditions typical of filtration systems [
15,
64].
Microscopy combined with energy-dispersive X-ray spectroscopy (EDS) analysis (
Table 5) revealed that, although the membranes underwent a purification process, residual traces of elements from the original culture medium remain embedded within the cellulose matrix. The composition was predominantly carbon and oxygen, as expected for bacterial cellulose. However, low concentrations of sulfur, magnesium, and phosphorus were detected, indicating residual compounds of the culture medium [
14].
Although these concentrations are minor, the residual presence of such elements may subtly influence membrane performance, particularly under critical filtration conditions. Phosphorus, magnesium, and sulfur may serve as anchoring points for salts or polar molecules, promoting localized accumulation and potentially contributing to pore blockage over time. While they are not the leading cause of membrane saturation, these residues underscore the importance of strict purification protocols, particularly for applications that require high selectivity or extended reuse cycles [
65,
66].
Upon analyzing the micrographs of the BCO membranes, significant alterations in surface morphology become evident, especially in the lower magnification images (
Figure 13A,B), when compared to the pure BC membranes. The BCO surface appears notably smoother, exhibiting fewer protrusions and irregularities across various regions. This more homogeneous appearance is accompanied by the presence of granules of differing sizes, irregularly distributed. Such uniformity is likely the result of mechanical pressure applied during the filtration process, which compresses the bacterial cellulose fibers. This compression is further enhanced by the presence of oil in the effluent, which, once retained between the fibers, acts as a natural adhesive. The lipophilic molecules infiltrate the porous structure of the membrane, facilitating a stronger entanglement of the nanofibrils and thereby reinforcing the cohesion between pressure-affected regions [
65,
67].
The observed granules of varying sizes correspond to oil particles retained after the internal saturation of the membrane. Once the internal porosity is filled, excess oil begins to accumulate on the external surface. This accumulation is a direct consequence of the composition of the ES1 effluent, which contains a mixture of fuel oil and surfactant, resulting in partially emulsified structures that are difficult to separate. The granules visible on the surface are the result of coalescence of these oil particles, forming clusters of different sizes depending on local emulsification density and the extent of oil penetration into the fibrillar network [
14].
In the intermediate magnification images (
Figure 13C,D), the morphological profile of the retained contaminant becomes more evident. A heterogeneous distribution of oily material is observed across the surface, with some areas showing higher accumulation while others remain relatively clean. This irregularity may be attributed to local variations in porosity or the interaction of the effluent flow with the membrane’s three-dimensional structure [
67].
In the high-magnification images (
Figure 13E,F), the entangled fibrillar arrangement becomes more pronounced, clearly revealing how the combination of filtration pressure and oil content resulted in denser compaction of the cellulose fibers. These fibers appear to be pressed tightly together, forming a more compact network in which the contaminant is interwoven and embedded within the membrane structure. This observation supports the hypothesis that oil, in addition to acting as a contaminant, also physically modifies the membrane’s topography, directly affecting its surface configuration and potential for reuse [
65].
EDS analysis after the filtration of Synthetic Effluent 1 (ES1) revealed the presence of residual elements on the bacterial cellulose membranes (
Table 6). These elements are not intrinsic to cellulose and were incorporated from the effluent contaminants, particularly surfactants, salts, and components of the fuel oil. Their detection reinforces the membrane’s ability to act as a selective barrier, retaining unwanted ionic and molecular species even after drying [
68].
Elemental ratios also indicated a consistently higher oxygen content compared to carbon across all samples. This can be explained by the natural composition of cellulose, which contains hydroxyl groups, as well as the accumulation of oxygen-rich compounds on the membrane surface following filtration of oil-based emulsions [
69].
The adsorption of fuel oil (OCB2) promotes the retention of compounds rich in oxygenated groups, such as esters, alcohols, and carboxylates. These molecules, combined with mild surface oxidation from air exposure and thermal drying, increase the detectable oxygen signal in EDS, which probes only the upper surface layer (up to 2 µm). Furthermore, as EDS does not detect hydrogen—an abundant element in the cellulose backbone—the apparent oxygen-to-carbon ratio becomes skewed. The O > C trend observed reflects the surface modification of the membrane caused by interactions with oil and surfactant residues from the effluent [
70,
71].
When analyzing the membranes used to filter the synthetic effluent ES2, even more pronounced structural differences become evident compared to the other samples. The high salt concentration in the effluent directly interfered with the scanning electron microscopy (SEM) imaging process, requiring a second round of metal coating to improve contrast and surface resolution. Despite this additional step, it was not possible to achieve magnifications beyond 1000×, likely due to the increased conductivity and charging effects caused by the accumulation of crystalline salts on the sample [
72].
The presence of NaCl significantly altered the surface texture of the membrane. The resulting structure appeared rough, brittle, and uneven, as seen in
Figure 14. This is most likely due to the infiltration and retention of NaCl crystals between the cellulose fibers, which, after the drying process, remained embedded and formed raised crystalline structures. These formations disrupted the otherwise smooth topography of the membrane, leading to a rugged relief. Such surface irregularities not only modify the physical appearance of the membrane but may also influence its mechanical behavior and reduce its potential for reuse in subsequent filtration cycles.
In the images obtained at 400× and 1000× magnifications (
Figure 14C,D), the crystals present on the membrane surface become clearly visible. Their shapes appear distinctly irregular, with asymmetric contours and an absence of the sharp edges typically associated with crystalline salts such as sodium chloride (NaCl). Rather than displaying well-defined geometric features, these crystals are covered by a denser, amorphous layer of material, suggesting partial encapsulation by oily compounds. The interaction between salt and oil, both present in the effluent, likely results in a surface coating that obscures the usual morphological characteristics of the crystals, making their direct identification under electron microscopy more difficult [
73].
This lipophilic coating directly interferes with the visualization of crystalline features at high magnifications, giving the surface a more diffuse and rough appearance. Furthermore, the coalescent behavior of the oil may contribute to the formation of heterogeneous aggregates, where different sizes and densities of material overlap. This complex configuration supports the hypothesis that the residual oil retained in the membrane acts not only as a contaminant but also as a surface-modifying agent, altering the way crystals organize and interact with the cellulose matrix after the drying process [
73].
After the filtration of ES2, the EDS analysis revealed notable changes in the elemental composition of the sample. Although carbon (C) and oxygen (O) remained the major constituents, their contents slightly decreased compared to the pure membrane. This reduction is attributed to the occupation of the cellulose network by other elements from the effluent, suggesting the incorporation of both organic and inorganic contaminants into the BC matrix during the filtration process.
The notable presence of sodium (Na) and chlorine (Cl), confirmed by EDS analysis (see
Table 7), indicates the retention of salts from the marine salt used in the effluent formulation. The significant capture of these ions shows the membrane’s effectiveness in trapping saline components, likely due to its high porosity and the combined effects of capillarity and surface adsorption.
This retention suggests that salt ions are partially immobilized within the porous matrix, especially in regions of lower fluid mobility. Although partial ion occupation of pores could theoretically reduce the available surface area for organic retention, the membrane still exhibited high removal efficiency for organic contaminants (up to 98% of BOD and COD), indicating that the structural and chemical properties of bacterial cellulose remained active and functional even in saline conditions. This reinforces the potential of BC membranes to operate effectively in complex effluents with both oily and saline loads.
Small amounts of magnesium, silicon, phosphorus, sulfur, and calcium were also detected. Although present in trace levels, these elements reflect the complexity of the membrane after filtration of the effluent. The non-uniform distribution of these residuals may be linked to the BC’s three-dimensional structure and the affinity of the contaminants for the cellulose’s functional groups.
3.1.8. Mechanical Testing
Flexible membranes accommodate bending and handling-induced strain and sustain greater tensile elongation without brittle fracture, reducing crack initiation and extending service life. Their compliance also improves conformal contact under sealing pressure, although in-plane compressive loads generally produce wrinkling rather than increased load-bearing [
74]. During tensile testing, the specimens exhibited distinct failure behaviors, indicating that retained contaminants modified the mechanical response of the BC network (
Figure 15).
Mechanical analyses of the bacterial cellulose (BC) membranes revealed that the presence and type of retained contaminants significantly influenced tensile strength and deformation behavior (
Figure 16). The control membrane (BC), composed solely of pure cellulose, exhibited a clear transverse fracture, breaking entirely from one edge to the other, with an average tensile strength of 90.925 ± 13.817 N and an elongation at break of 17.175 ± 1.746%. This profile reflects a homogeneous and porous structure, rigid and free from plasticizing agents, which promotes uniform stress distribution across the matrix and results in complete, brittle failure [
64,
75].
In contrast, the BCO membrane, saturated with effluent containing only oil, demonstrated the highest elongation at break (70.894 ± 4.821%) but intermediate tensile strength (93.225 ± 12.441 N), and did not fully rupture during the test, having been automatically interrupted by the equipment. The surface of these samples appeared smoother, with fewer irregularities. This behavior suggests that residual oil acted as a plasticizer, enhancing internal mobility and delaying crack propagation. While this increased ductility, it also reduced the membrane’s structural resistance under increasing load [
76,
77].
The BCOS membrane, used for filtering effluent containing oil and 35 g/L of NaCl, showed the highest tensile strength (193.315 ± 14.975 N) with an intermediate elongation of 49.381 ± 5.106%. Despite the oil content, its rupture behavior was distinctly abrupt and concentrated, indicating a more compact and rigid matrix. This can be attributed to salt crystallization between the cellulose fibers, which may have created ionic bridges or points of stiffness, temporarily increasing mechanical resistance while reducing flexibility. The visual aspect of these samples was rougher and more brittle, consistent with the presence of residual salt crystals on the surface [
56].
Therefore, the fracture patterns observed align closely with the quantitative data from the mechanical tests, reflecting the complex interaction between retained contaminants and the three-dimensional fibrillar network of the membranes. Oil acts as a natural plasticizer, increasing flexibility and deformation capacity, whereas salt promotes rigidity and compactness, enhancing tensile strength at the expense of ductility. The chemical nature of the adsorbed contaminants has a direct impact on the structural performance of the membranes, particularly in applications that require prolonged mechanical loading or reuse over multiple filtration cycles [
78].