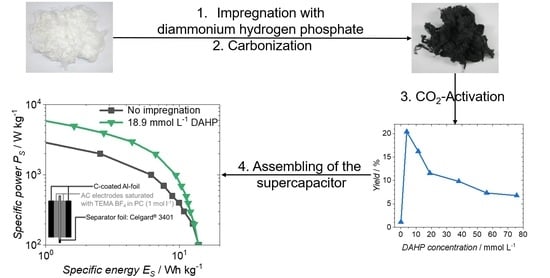
Supercapacitor Electrodes from Viscose-Based Activated Carbon Fibers: Significant Yield and Performance Improvement Using Diammonium Hydrogen Phosphate as Impregnating Agent
Abstract
1. Introduction
2. Materials and Methods
2.1. Acitvated Carbon Fiber Preparation
2.2. Electrode Fabrication and Electrochemical Characterization
2.3. Structural Characterization
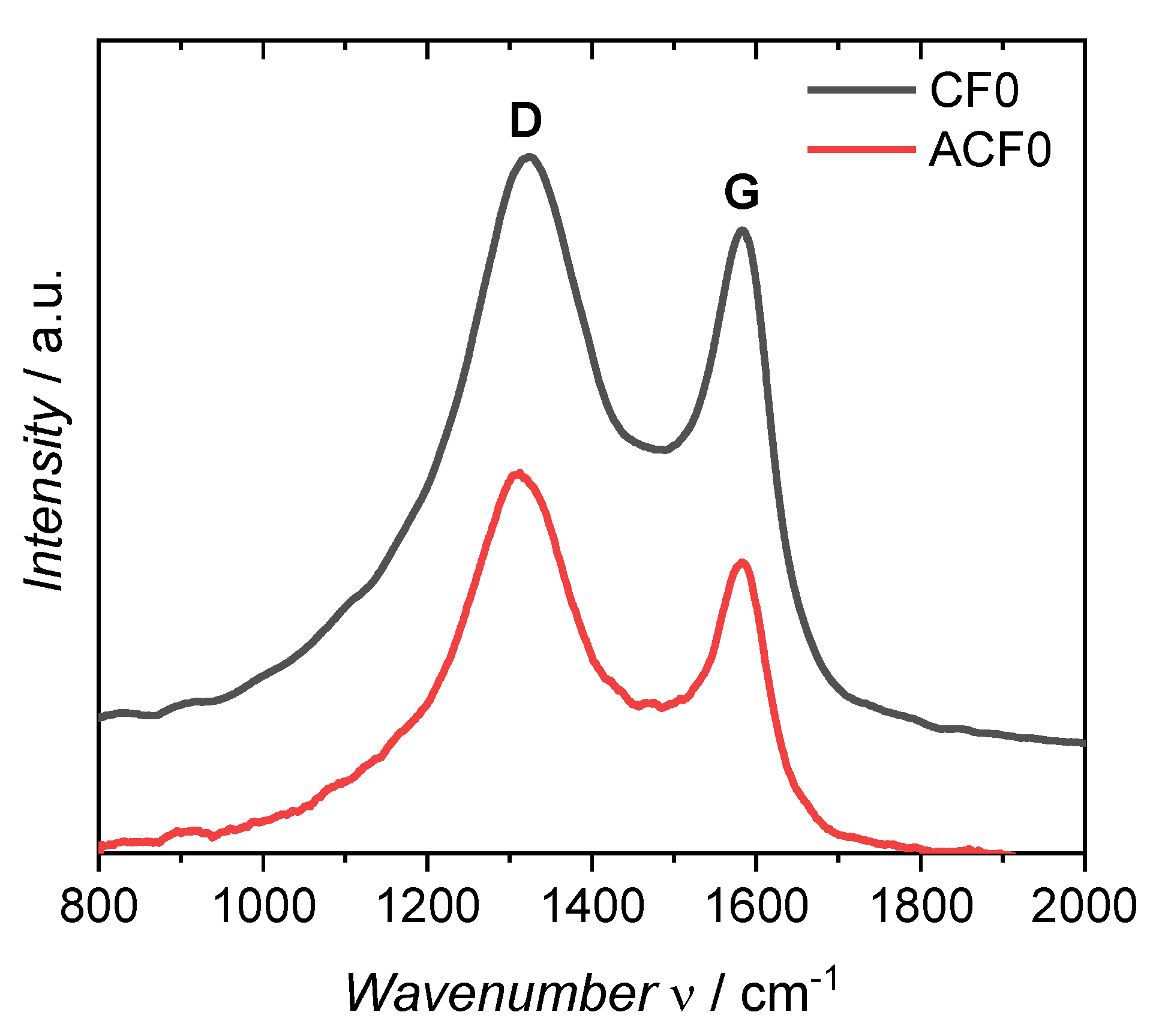
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Enock, T.K.; King’ondu, C.K.; Pogrebnoi, A.; Jande, Y.A.C. Status of Biomass Derived Carbon Materials for Supercapacitor Application. Int. J. Electrochem. 2017, 2017, 1–14. [Google Scholar] [CrossRef]
- Hao, L.; Li, X.; Zhi, L. Carbonaceous electrode materials for supercapacitors. Adv. Mater. 2013, 25, 3899–3904. [Google Scholar] [CrossRef] [PubMed]
- Sevilla, M.; Mokaya, R. Energy storage applications of activated carbons: Supercapacitors and hydrogen storage. Energy Environ. Sci. 2014, 7, 1250–1280. [Google Scholar] [CrossRef]
- Wang, H.; Yu, W.; Shi, J.; Mao, N.; Chen, S.; Liu, W. Biomass derived hierarchical porous carbons as high-performance anodes for sodium-ion batteries. Electrochim. Acta 2016, 188, 103–110. [Google Scholar] [CrossRef]
- Wang, J.; Kaskel, S. KOH activation of carbon-based materials for energy storage. J. Mater. Chem. 2012, 22, 23710. [Google Scholar] [CrossRef]
- Wang, Y.; Song, Y.; Xia, Y. Electrochemical capacitors: Mechanism, materials, systems, characterization and applications. Chem. Soc. Rev. 2016, 45, 5925–5950. [Google Scholar] [CrossRef]
- Yan, J.; Wang, Q.; Wei, T.; Fan, Z. Recent Advances in Design and Fabrication of Electrochemical Supercapacitors with High Energy Densities. Adv. Energy Mater. 2014, 4, 1300816. [Google Scholar] [CrossRef]
- Zhang, L.L.; Zhao, X.S. Carbon-based materials as supercapacitor electrodes. Chem. Soc. Rev. 2009, 38, 2520–2531. [Google Scholar] [CrossRef]
- Zhong, C.; Deng, Y.; Hu, W.; Qiao, J.; Zhang, L.; Zhang, J. A review of electrolyte materials and compositions for electrochemical supercapacitors. Chem. Soc. Rev. 2015, 44, 7484–7539. [Google Scholar] [CrossRef]
- Yee Liew, S.; Thielemans, W.; Freunberger, S.; Spirk, S. Polysaccharide Based Supercapacitors; Springer International Publishing: Cham, Switzerland, 2017. [Google Scholar]
- Murray, D.B.; Hayes, J.G. Cycle Testing of Supercapacitors for Long-Life Robust Applications. IEEE Trans. Power Electron. 2015, 30, 2505–2516. [Google Scholar] [CrossRef]
- Stavropoulos, G.G.; Zabaniotou, A.A. Minimizing activated carbons production cost. Fuel Process. Technol. 2009, 90, 952–957. [Google Scholar] [CrossRef]
- Pandolfo, A.G.; Hollenkamp, A.F. Carbon properties and their role in supercapacitors. J. Power Sources 2006, 157, 11–27. [Google Scholar] [CrossRef]
- Dumanlı, A.G.; Windle, A.H. Carbon fibres from cellulosic precursors: A review. J. Mater. Sci. 2012, 47, 4236–4250. [Google Scholar] [CrossRef]
- Zeng, F.; Pan, D.; Pan, N. Choosing the Impregnants by Thermogravimetric Analysis for Preparing Rayon-Based Carbon Fibers. J. Inorg. Organomet. Polym. 2005, 15, 261–267. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, X.; Yang, R.; Li, G.; Hu, C. The role of H3PO4 in the preparation of activated carbon from NaOH-treated rice husk residue. RSC Adv. 2015, 5, 32626–32636. [Google Scholar] [CrossRef]
- Akita, K.; Kase, M. Determination of kinetic parameters for pyrolysis of cellulose and cellulose treated with ammonium phosphate by differential thermal analysis and thermal gravimetric analysis. J. Polym. Sci. A-1 Polym. Chem. 1967, 5, 833–848. [Google Scholar] [CrossRef]
- Hirata, T.; Nishimoto, T. DSC, DTA, and TG of cellulose untreated and treated with flame-retardants. Thermochim. Acta 1991, 193, 99–106. [Google Scholar] [CrossRef]
- Frank, E.; Steudle, L.M.; Ingildeev, D.; Spörl, J.M.; Buchmeiser, M.R. Carbon fibers: Precursor systems, processing, structure, and properties. Angew. Chem. Int. Ed. Engl. 2014, 53, 5262–5298. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Landers, J.; Gor, G.Y.; Neimark, A.V. Density functional theory methods for characterization of porous materials. Colloids Surf. A: Physicochem. Eng. Asp. 2013, 437, 3–32. [Google Scholar] [CrossRef]
- Wheeler, E. The Manufacture of Artificial Silk with Special Reference to the Viscose Process; D. Van Nostrand Company: New York, NY, USA, 1928. [Google Scholar]
- Karacan, I.; Gül, A. Carbonization behavior of oxidized viscose rayon fibers in the presence of boric acid–phosphoric acid impregnation. J. Mater. Sci. 2014, 49, 7462–7475. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Interpretation of Raman spectra of disordered and amorphous carbon. Phys. Rev. B 2000, 61, 14095–14107. [Google Scholar] [CrossRef]
- Ranaweera, C.K.; Kahol, P.K.; Ghimire, M.; Mishra, S.R.; Gupta, R.K. Orange-Peel-Derived Carbon: Designing Sustainable and High-Performance Supercapacitor Electrodes. C J. Carbon Res. 2017, 3, 25. [Google Scholar] [CrossRef]
- Lewin, M.; Sello, S. Handbook of Fiber Science and Technology Volume 2. Functional Finishes; Part B; Marcel Dekker, Inc.: New York, NY, USA; Basel, Switzerland, 1984; ISBN 9780203752920. [Google Scholar]
- Simon, P.; Gogotsi, Y. Charge storage mechanism in nanoporous carbons and its consequence for electrical double layer capacitors. Proc. R. Soc. A 2010, 368, 3457–3467. [Google Scholar] [CrossRef]
- Yang, S.; Kim, I.-J.; Choi, I.-S.; Bae, M.-K.; Kim, H.-S. Influence of electrolytes (TEABF4 and TEMABF4) on electrochemical performance of graphite oxide derived from needle coke. J. Nanosci. Nanotechnol. 2013, 13, 3747–3751. [Google Scholar] [CrossRef]
- Simon, P.; Gogotsi, Y. Materials for electrochemical capacitors. Nat. Mater. 2008, 7, 845–854. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, L.; Zhang, J. A review of electrode materials for electrochemical supercapacitors. Chem. Soc. Rev. 2012, 41, 797–828. [Google Scholar] [CrossRef]
- Supercapacitors. Materials, Systems, and Applications; Beguin, F., Frackowiak, E., Eds.; Wiley: Weinheim, Germany, 2013; ISBN 978-3-527-64668-5. [Google Scholar]
- Fic, K.; Lota, G.; Meller, M.; Frackowiak, E. Novel insight into neutral medium as electrolyte for high-voltage supercapacitors. Energy Environ. Sci. 2012, 5, 5842–5850. [Google Scholar] [CrossRef]
- Lee, J.H.; Park, N.; Kim, B.G.; Jung, D.S.; Im, K.; Hur, J.; Choi, J.W. Restacking-inhibited 3D reduced graphene oxide for high performance supercapacitor electrodes. ACS Nano 2013, 7, 9366–9374. [Google Scholar] [CrossRef]
- Sun, W.; Zheng, R.; Chen, X. Symmetric redox supercapacitor based on micro-fabrication with three-dimensional polypyrrole electrodes. J. Power Sources 2010, 195, 7120–7125. [Google Scholar] [CrossRef]
- Ates, M. Review study of electrochemical impedance spectroscopy and equivalent electrical circuits of conducting polymers on carbon surfaces. Prog. Org. Coat. 2011, 71, 1–10. [Google Scholar] [CrossRef]
- Pina, A.; Amaya, A.; Marcuzzo, J.; Rodrigues, A.; Baldan, M.; Tancredi, N.; Cuña, A. Supercapacitor Electrode Based on Activated Carbon Wool Felt. C J. Carbon Res. 2018, 4, 24. [Google Scholar] [CrossRef]
- Qi, F.; Xia, Z.; Sun, R.; Sun, X.; Xu, X.; Wei, W.; Wang, S.; Sun, G. Graphitization induced by KOH etching for the fabrication of hierarchical porous graphitic carbon sheets for high performance supercapacitors. J. Mater. Chem. A 2018, 6, 14170–14177. [Google Scholar] [CrossRef]
- Simon, P.; Burke, A.F. Nanostructured carbons: Double-layer capacitance and more. Electrochem. Soc. Interface 2008, 17, 38–43. [Google Scholar]
- Chen, X.Y.; Chen, C.; Zhang, Z.J.; Xie, D.H.; Deng, X.; Liu, J.W. Nitrogen-doped porous carbon for supercapacitor with long-term electrochemical stability. J. Power Sources 2013, 230, 50–58. [Google Scholar] [CrossRef]

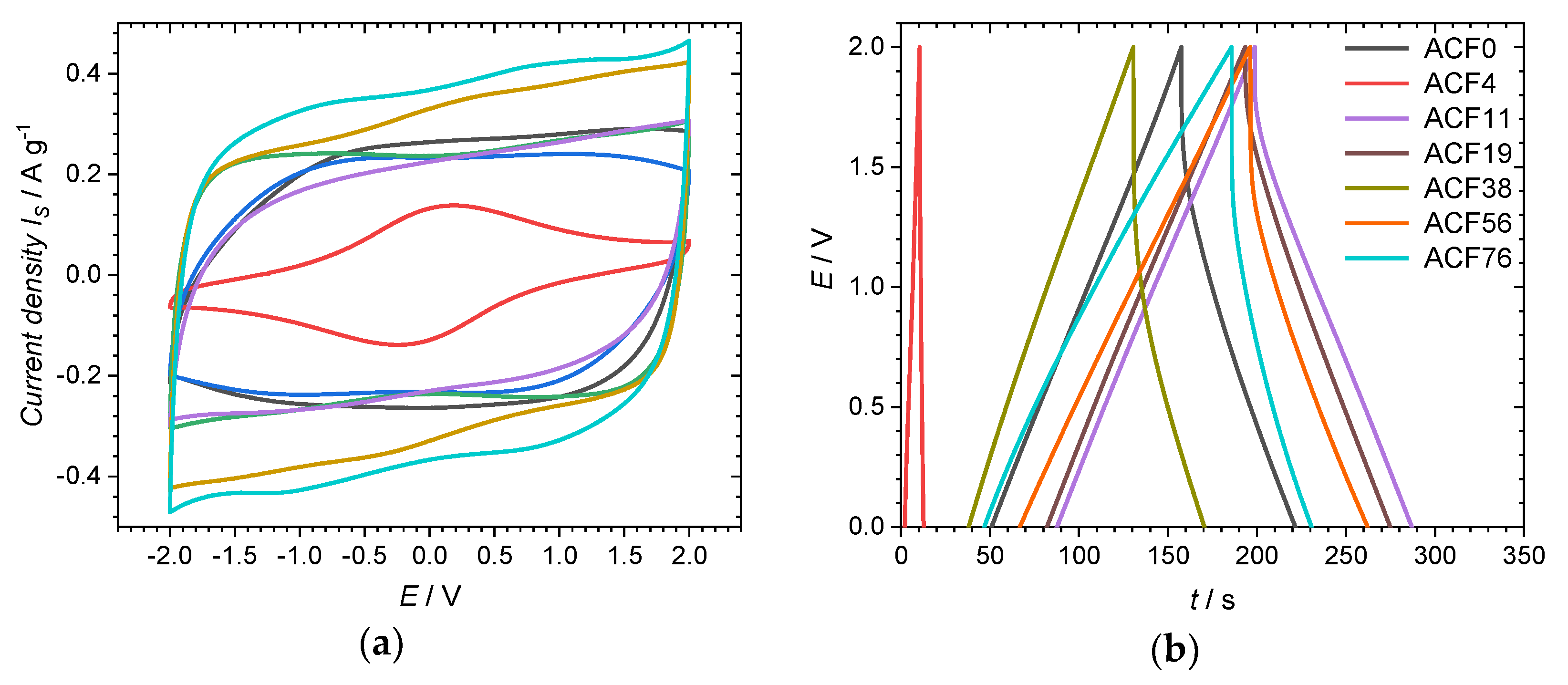
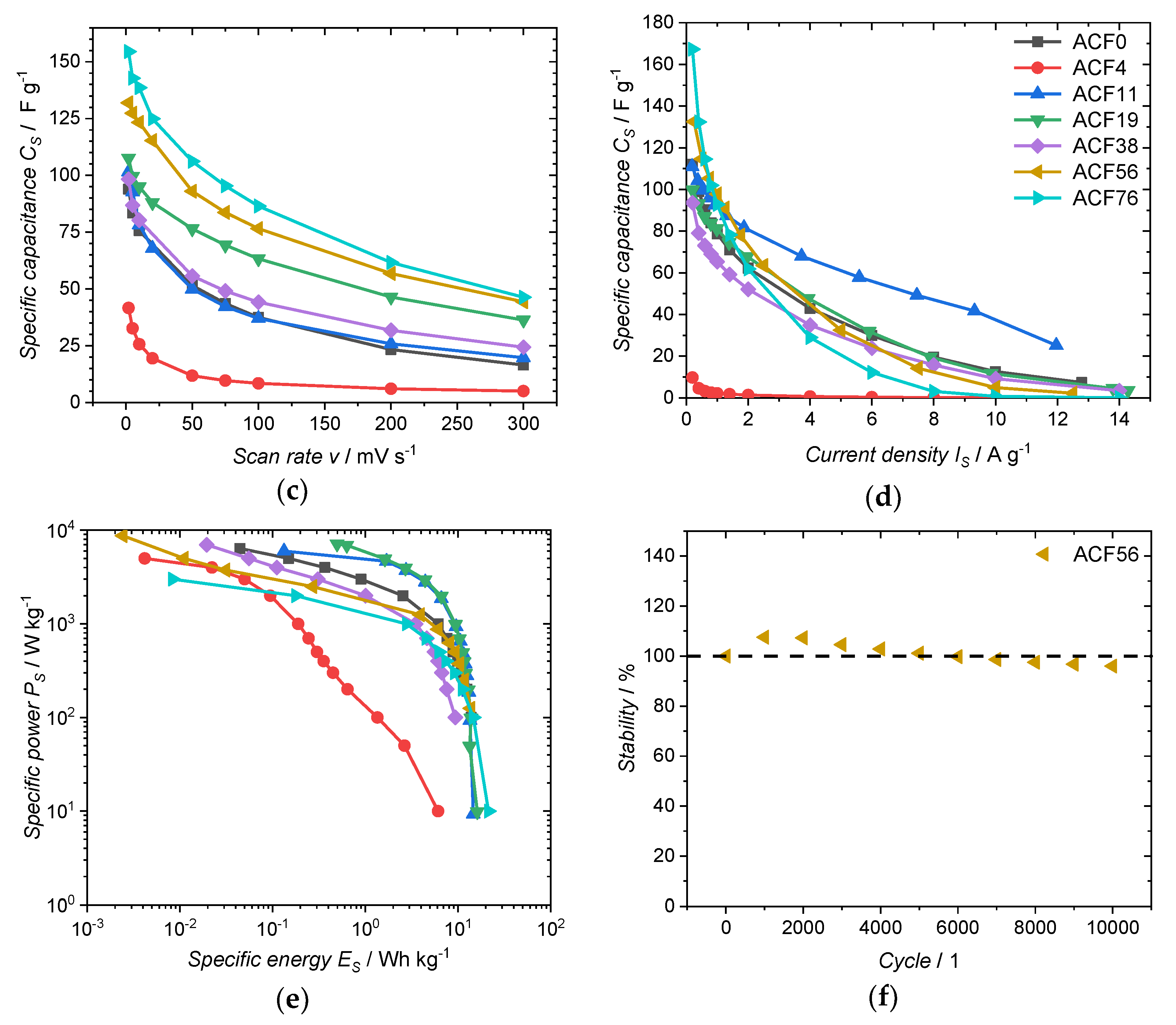
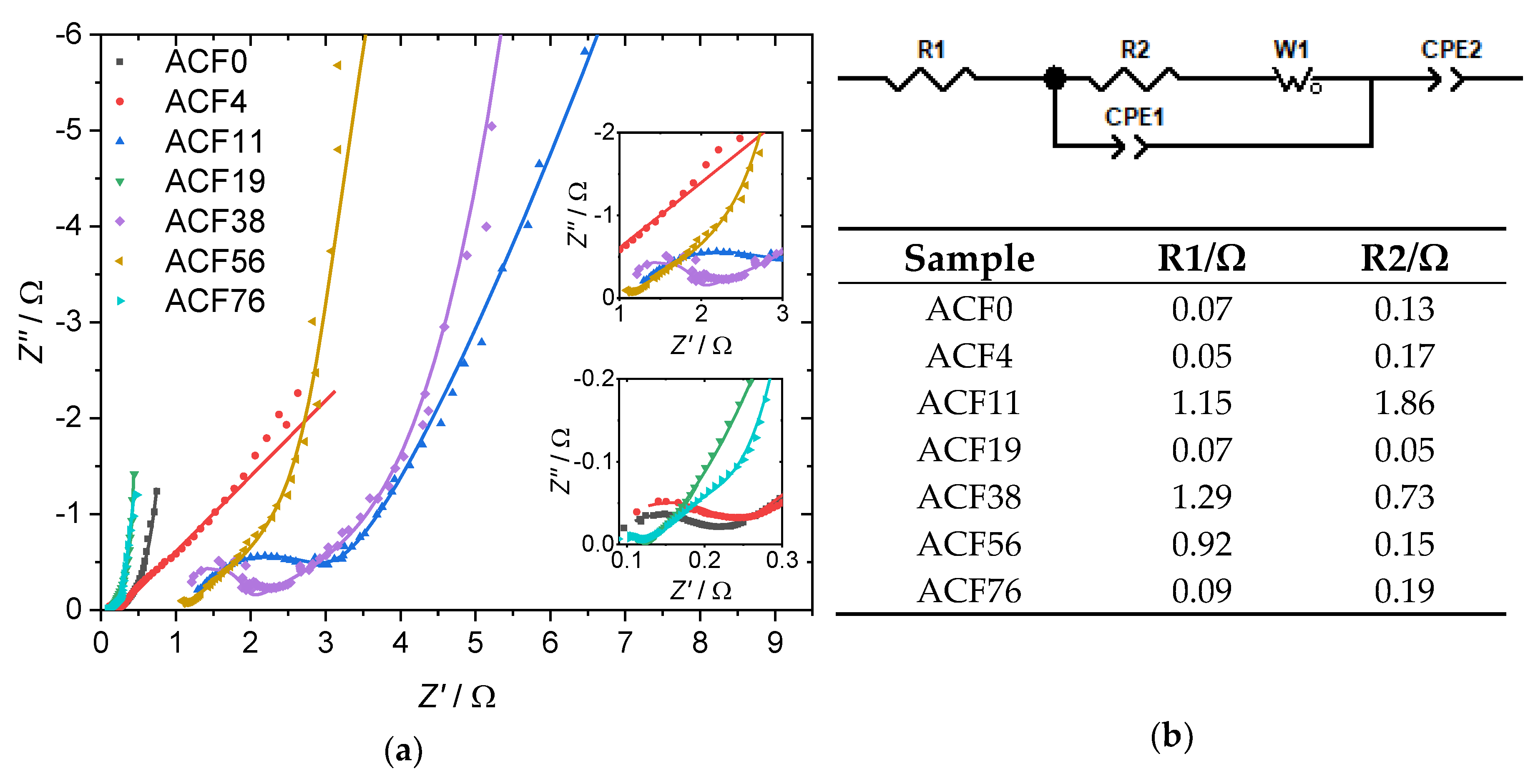
| Sample | DAHP Concentration/mmol·L−1 | C/at.% | O/at.% | P/at.% |
|---|---|---|---|---|
| CF0 | 0.0 | 98.54 ± 0.13 | 1.25 ± 0.14 | - |
| CF38 | 37.9 | 97.12 ± 0.13 | 2.18± 0.17 | 0.70 ± 0.06 |
| ACF0 | 0.0 | 98.74 ± 0.34 | 1.01 ± 0.36 | - |
| ACF4 | 3.8 | 98.44 ± 0.38 | 1.41 ± 0.33 | 0.17 ± 0.06 |
| ACF11 | 11.4 | 97.64 ± 0.33 | 1.05 ± 0.15 | 1.08 ± 0.25 |
| ACF19 | 18.9 | 95.60 ± 1.16 | 2.15 ± 0.46 | 1.76 ± 0.50 |
| ACF38 | 37.9 | 92.57 ± 1.25 | 3.24 ± 0.39 | 2.27 ± 0.75 |
| ACF56 | 56.1 | 93.38 ± 1.17 | 3.45 ± 0.74 | 2.06 ± 0.32 |
| ACF76 | 75.7 | 89.27 ± 1.31 | 4.92 ± 0.34 | 3.73 ± 0.34 |
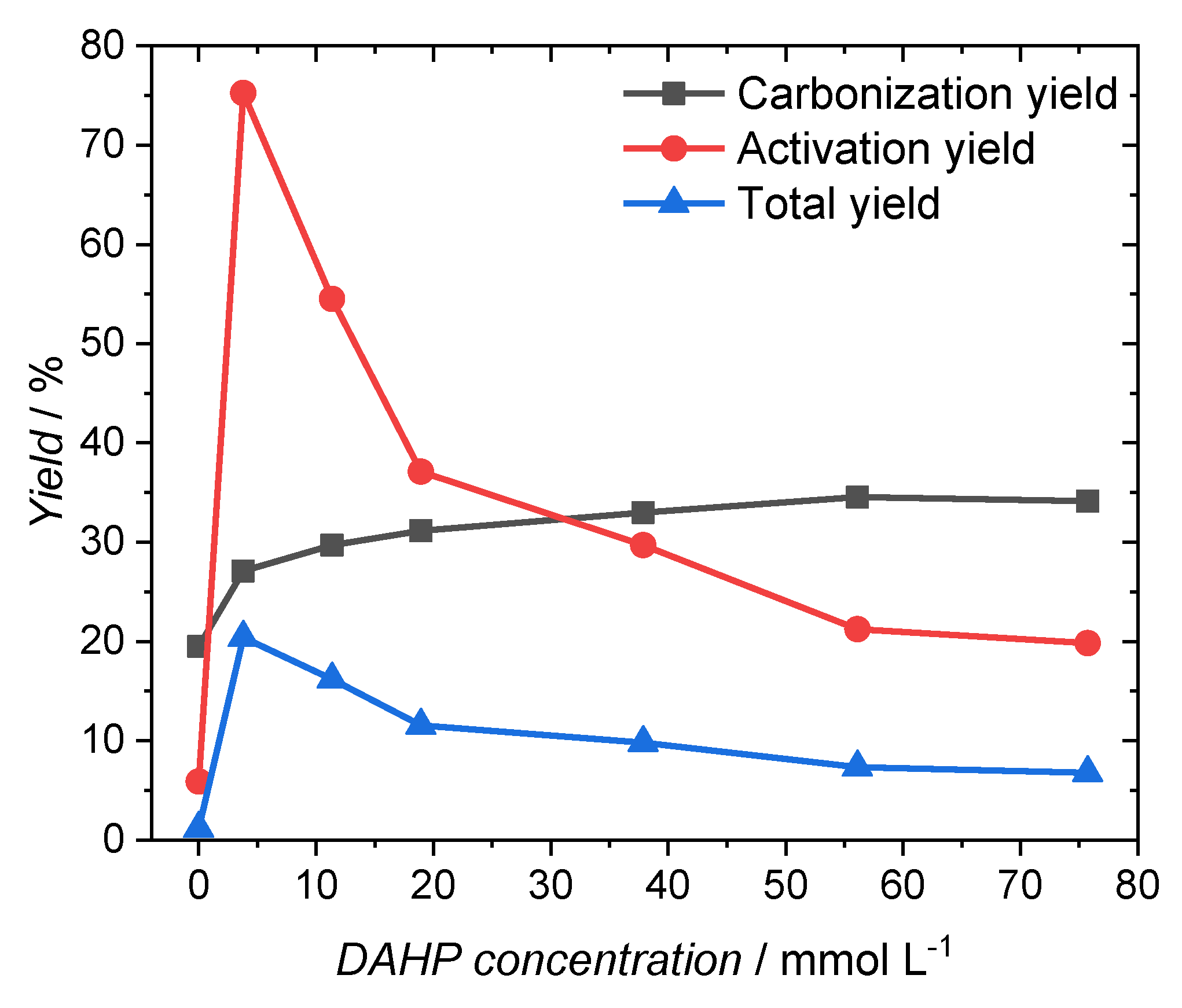
| DAHP Concentration/mmol·L−1 | Yield of Carbonization/% | Yield of Activation/% | Total Yield/% |
|---|---|---|---|
| 0.0 | 19.5 | 5.9 | 1.1 |
| 3.8 | 27.1 | 75.3 | 20.4 |
| 11.4 | 29.7 | 54.5 | 16.2 |
| 18.9 | 31.1 | 37.1 | 11.6 |
| 37.9 | 33.0 | 29.7 | 9.8 |
| 56.1 | 34.6 | 21.2 | 7.3 |
| 75.7 | 34.1 | 19.8 | 6.8 |
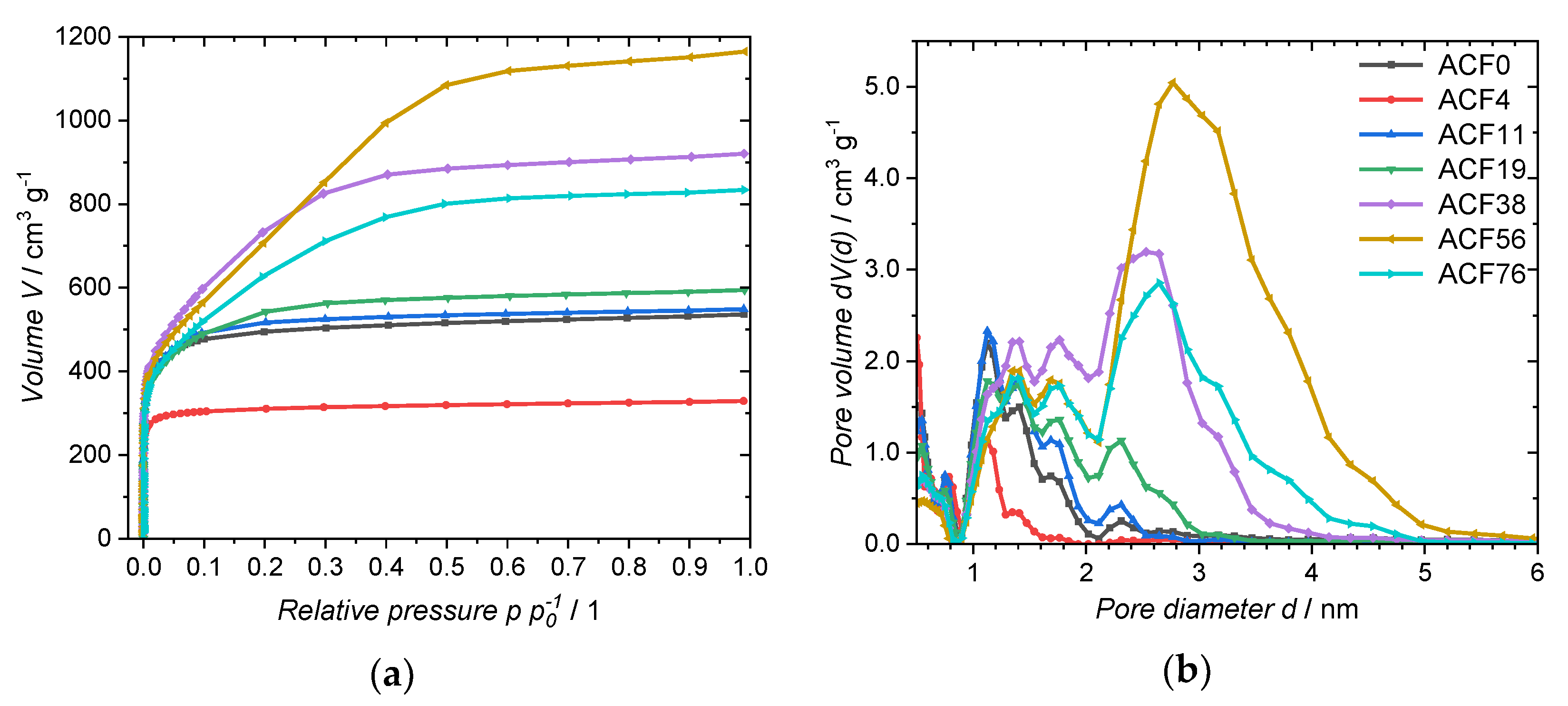
| Sample | SBET m2·g−1 | Vtot cm3·g−1 | Ddom nm | CS,CV @ 10 mV·s−1 F·g−1 | CS,GDC @ 1.0 A·g−1 F·g−1 |
|---|---|---|---|---|---|
| ACF0 | 1932 | 0.83 | 1.1; 1.4 | 76 | 79 |
| ACF4 | 1250 | 0.51 | 0.5; 1.1 | 26 | 2 |
| ACF11 | 2002 | 0.85 | 1.1; 1.4; 1.7 | 78 | 93 |
| ACF19 | 2018 | 0.95 | 1.1; 1.4; 1.7; 2.3 | 95 | 81 |
| ACF38 | 2718 | 1.42 | 1.4; 1.8; 3.0 | 80 | 65 |
| ACF56 | 2763 | 1.83 | 1.4; 1.7; 2.8 | 115 | 98 |
| ACF76 | 2301 | 1.29 | 1.4; 1.8; 2.7 | 139 | 93 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Breitenbach, S.; Lumetzberger, A.; Hobisch, M.A.; Unterweger, C.; Spirk, S.; Stifter, D.; Fürst, C.; Hassel, A.W. Supercapacitor Electrodes from Viscose-Based Activated Carbon Fibers: Significant Yield and Performance Improvement Using Diammonium Hydrogen Phosphate as Impregnating Agent. C 2020, 6, 17. https://doi.org/10.3390/c6020017
Breitenbach S, Lumetzberger A, Hobisch MA, Unterweger C, Spirk S, Stifter D, Fürst C, Hassel AW. Supercapacitor Electrodes from Viscose-Based Activated Carbon Fibers: Significant Yield and Performance Improvement Using Diammonium Hydrogen Phosphate as Impregnating Agent. C. 2020; 6(2):17. https://doi.org/10.3390/c6020017
Chicago/Turabian StyleBreitenbach, Stefan, Alexander Lumetzberger, Mathias Andreas Hobisch, Christoph Unterweger, Stefan Spirk, David Stifter, Christian Fürst, and Achim Walter Hassel. 2020. "Supercapacitor Electrodes from Viscose-Based Activated Carbon Fibers: Significant Yield and Performance Improvement Using Diammonium Hydrogen Phosphate as Impregnating Agent" C 6, no. 2: 17. https://doi.org/10.3390/c6020017
APA StyleBreitenbach, S., Lumetzberger, A., Hobisch, M. A., Unterweger, C., Spirk, S., Stifter, D., Fürst, C., & Hassel, A. W. (2020). Supercapacitor Electrodes from Viscose-Based Activated Carbon Fibers: Significant Yield and Performance Improvement Using Diammonium Hydrogen Phosphate as Impregnating Agent. C, 6(2), 17. https://doi.org/10.3390/c6020017