Long Non-Coding RNA in Vascular Disease and Aging
Abstract
1. Introduction
1.1. Age and Cardiovascular Disease
1.2. LncRNAs in Vascular Disease
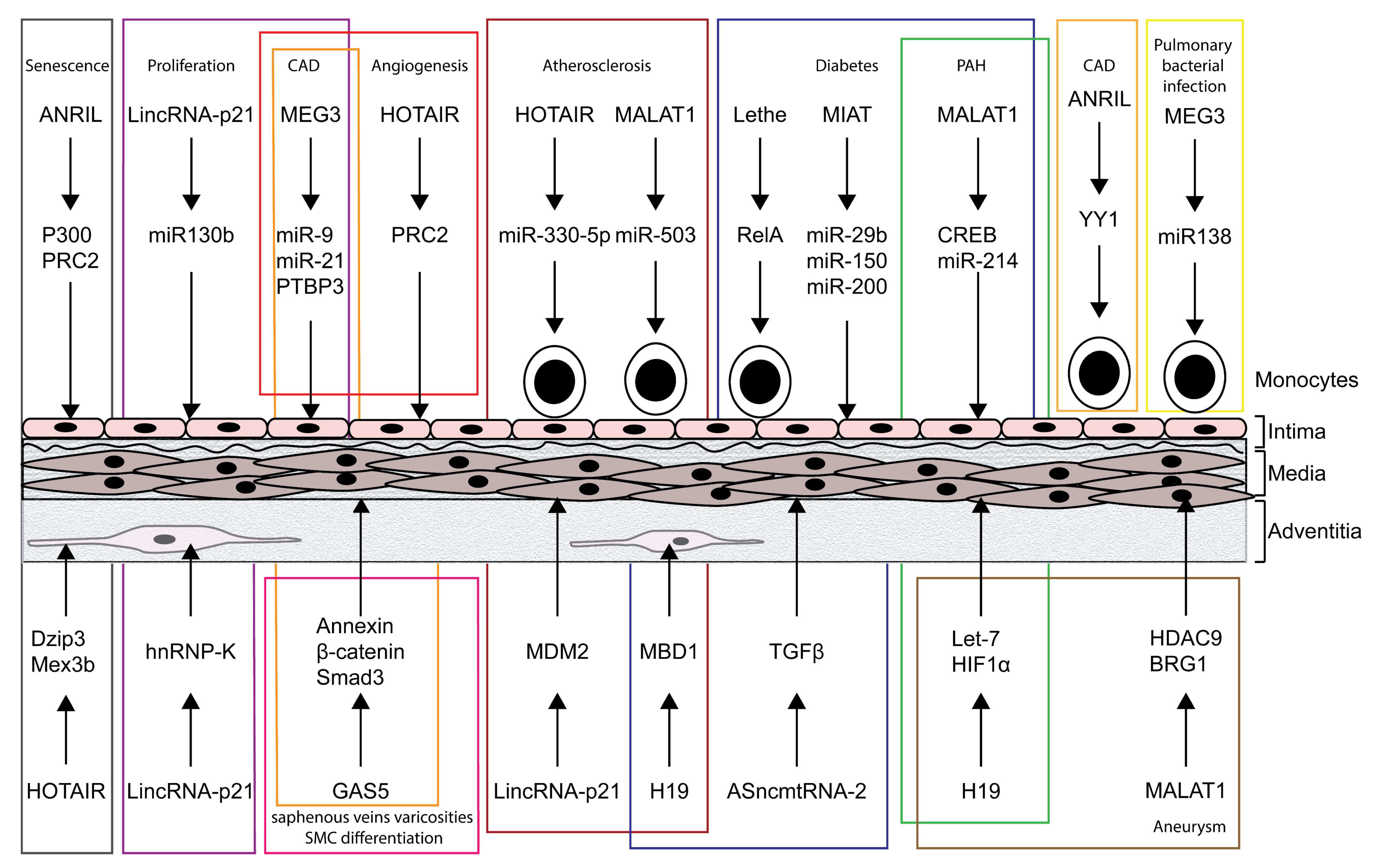
2. LncRNA Function in Vascular Diseases
- Decoy lncRNA: These are RNP-binding lncRNAs, which inhibit the biological function of the RNP by preventing them from binding to their targets. This group also includes competing endogenous RNAs (ceRNAs) which bind to miRNAs and are often claimed to “sponge” off the miRNA. However, because lncRNAs are expressed at low levels and miRNAs are expressed at high levels, it is not completely clear how a lncRNA sponge would have such a big effect on miRNA levels [3].
- Guide lncRNA: These lncRNAs bind to a transcription factor or a chromatin modifier and guide it to the target promoter thereby altering the transcription of the target gene.
- Scaffold lncRNA: These lncRNAs bind to several RNPs, bringing them together to form ribonucleoprotein complexes, which in some cases can lead to transcriptional activation or repression.
- Enhancer lncRNAs: these lncRNAs are transcribed from enhancer regions and bring together enhancers and promoters in the genome through chromosomal looping to activate expression. We did not find published evidence for enhancer lncRNAs that play a role in vascular aging.
2.1. Decoy lncRNAs
2.2. Guide lncRNAs
2.3. Scaffold lncRNAs
2.4. Other lncRNAs Regulated in Vascular Diseases and Aging
3. LncRNAs as Biomarkers
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ANRIL | antisense non-coding RNA in the INK4 locus |
| CAD | coronary artery disease |
| ceRNAs | competing endogenous RNAs |
| ECs | endothelial cells |
| GAS5 | Growth Arrest Specific 5 |
| HOTAIR | HOX Antisense Intergenic RNA |
| HUVECs | human umbilical vein endothelial cells |
| lncRNA | long non-coding RNA |
| MALAT1 | Metastasis Associated Lung Adenocarcinoma Transcript 1 |
| MDM2 | Mouse double minute 2 homolog |
| Meg3 | Maternally expressed gene 3 |
| MIAT | Myocardial infarction associated transcript |
| miRNA | microRNA |
| ncRNA | non-coding RNA |
| PAH | pulmonary arterial hypertension |
| RNP | ribonucleoprotein |
| ROS | reactive oxygen species |
| SMCs | smooth muscle cells. |
References
- Venter, J.C.; Adams, M.D.; Myers, E.W.; Li, P.W.; Mural, R.J.; Sutton, G.G.; Smith, H.O.; Yandell, M.; Evans, C.A.; Holt, R.A.; et al. The sequence of the human genome. Science 2001, 291, 1304–1351. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, Y.; Furuno, M.; Kasukawa, T.; Adachi, J.; Bono, H.; Kondo, S.; Nikaido, I.; Osato, N.; Saito, R.; Suzuki, H.; et al. Analysis of the mouse transcriptome based on functional annotation of 60,770 full-length cdnas. Nature 2002, 420, 563–573. [Google Scholar] [PubMed]
- Boon, R.A.; Jae, N.; Holdt, L.; Dimmeler, S. Long noncoding rnas: From clinical genetics to therapeutic targets? J. Am. Coll. Cardiol. 2016, 67, 1214–1226. [Google Scholar] [CrossRef] [PubMed]
- Kapranov, P.; Cheng, J.; Dike, S.; Nix, D.A.; Duttagupta, R.; Willingham, A.T.; Stadler, P.F.; Hertel, J.; Hackermuller, J.; Hofacker, I.L.; et al. Rna maps reveal new rna classes and a possible function for pervasive transcription. Science 2007, 316, 1484–1488. [Google Scholar] [CrossRef] [PubMed]
- Christensen, K.; Doblhammer, G.; Rau, R.; Vaupel, J.W. Ageing populations: The challenges ahead. Lancet 2009, 374, 1196–1208. [Google Scholar] [CrossRef]
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y.; et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the global burden of disease study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- Zarrinkoob, L.; Ambarki, K.; Wahlin, A.; Birgander, R.; Carlberg, B.; Eklund, A.; Malm, J. Aging alters the dampening of pulsatile blood flow in cerebral arteries. J. Cereb. Blood Flow Metab. 2016, 36, 1519–1527. [Google Scholar] [CrossRef]
- Steppan, J.; Barodka, V.; Berkowitz, D.E.; Nyhan, D. Vascular stiffness and increased pulse pressure in the aging cardiovascular system. Cardiol. Res. Pract. 2011, 2011, 263585. [Google Scholar] [CrossRef]
- Hong, Y.M. Atherosclerotic cardiovascular disease beginning in childhood. Korean Circ. J. 2010, 40, 1–9. [Google Scholar] [CrossRef]
- Nakae, I.; Fujita, M.; Miwa, K.; Hasegawa, K.; Kihara, Y.; Nohara, R.; Miyamoto, S.; Ueda, K.; Tamaki, S.; Sasayama, S. Age-dependent impairment of coronary collateral development in humans. Heart Vessels 2000, 15, 176–180. [Google Scholar] [CrossRef]
- Rivard, A.; Fabre, J.E.; Silver, M.; Chen, D.; Murohara, T.; Kearney, M.; Magner, M.; Asahara, T.; Isner, J.M. Age-dependent impairment of angiogenesis. Circulation 1999, 99, 111–120. [Google Scholar] [CrossRef]
- Madonna, R.; Balistreri, C.R.; Geng, Y.J.; De Caterina, R. Diabetic microangiopathy: Pathogenetic insights and novel therapeutic approaches. Vasc. Pharmacol. 2017, 90, 1–7. [Google Scholar] [CrossRef]
- Simion, V.; Haemmig, S.; Feinberg, M.W. Lncrnas in vascular biology and disease. Vasc. Pharmacol. 2018. [Google Scholar] [CrossRef]
- Li, H.; Zhu, H.; Ge, J. Long noncoding rna: Recent updates in atherosclerosis. Int. J. Biol. Sci. 2016, 12, 898–910. [Google Scholar] [CrossRef]
- Yu, B.; Wang, S. Angio-lncrs: Lncrnas that regulate angiogenesis and vascular disease. Theranostics 2018, 8, 3654–3675. [Google Scholar] [CrossRef]
- Zhou, T.; Ding, J.W.; Wang, X.A.; Zheng, X.X. Long noncoding rnas and atherosclerosis. Atherosclerosis 2016, 248, 51–61. [Google Scholar] [CrossRef]
- Duggirala, A.; Delogu, F.; Angelini, T.G.; Smith, T.; Caputo, M.; Rajakaruna, C.; Emanueli, C. Non coding rnas in aortic aneurysmal disease. Front. Genet. 2015, 6, 125. [Google Scholar] [CrossRef]
- Leung, A.; Amaram, V.; Natarajan, R. Linking diabetic vascular complications with lncrnas. Vasc. Pharmacol. 2018. [Google Scholar] [CrossRef]
- He, X.; Ou, C.; Xiao, Y.; Han, Q.; Li, H.; Zhou, S. Lncrnas: Key players and novel insights into diabetes mellitus. Oncotarget 2017, 8, 71325–71341. [Google Scholar] [CrossRef]
- Wang, K.C.; Chang, H.Y. Molecular mechanisms of long noncoding rnas. Mol. Cell 2011, 43, 904–914. [Google Scholar] [CrossRef]
- Devaux, Y.; Zangrando, J.; Schroen, B.; Creemers, E.E.; Pedrazzini, T.; Chang, C.P.; Dorn, G.W., 2nd; Thum, T.; Heymans, S.; Cardiolinc, n. Long noncoding rnas in cardiac development and ageing. Nat. Rev. Cardiol. 2015, 12, 415–425. [Google Scholar]
- Boon, R.A.; Hofmann, P.; Michalik, K.M.; Lozano-Vidal, N.; Berghauser, D.; Fischer, A.; Knau, A.; Jae, N.; Schurmann, C.; Dimmeler, S. Long noncoding rna meg3 controls endothelial cell aging and function: Implications for regenerative angiogenesis. J. Am. Coll. Cardiol. 2016, 68, 2589–2591. [Google Scholar] [CrossRef]
- White, R.R.; Milholland, B.; MacRae, S.L.; Lin, M.; Zheng, D.; Vijg, J. Comprehensive transcriptional landscape of aging mouse liver. BMC Genom. 2015, 16, 899. [Google Scholar] [CrossRef]
- Wu, Z.; He, Y.; Li, D.; Fang, X.; Shang, T.; Zhang, H.; Zheng, X. Long noncoding rna meg3 suppressed endothelial cell proliferation and migration through regulating mir-21. Am. J. Transl. Res. 2017, 9, 3326–3335. [Google Scholar]
- Sun, Z.; Nie, X.; Sun, S.; Dong, S.; Yuan, C.; Li, Y.; Xiao, B.; Jie, D.; Liu, Y. Long non-coding rna meg3 downregulation triggers human pulmonary artery smooth muscle cell proliferation and migration via the p53 signaling pathway. Cell. Physiol. Biochem. 2017, 42, 2569–2581. [Google Scholar] [CrossRef]
- Liu, H.Z.; Wang, Q.Y.; Zhang, Y.; Qi, D.T.; Li, M.W.; Guo, W.Q.; Ma, Y.H.; Wang, L.Y.; Chen, Y.; Gao, C.Y. Pioglitazone up-regulates long non-coding rna meg3 to protect endothelial progenitor cells via increasing hdac7 expression in metabolic syndrome. Biomed. Pharmacother. 2016, 78, 101–109. [Google Scholar] [CrossRef]
- Sathishkumar, C.; Prabu, P.; Mohan, V.; Balasubramanyam, M. Linking a role of lncrnas (long non-coding rnas) with insulin resistance, accelerated senescence, and inflammation in patients with type 2 diabetes. Hum. Genom. 2018, 12, 41. [Google Scholar] [CrossRef]
- Wu, H.; Zhao, Z.A.; Liu, J.; Hao, K.; Yu, Y.; Han, X.; Li, J.; Wang, Y.; Lei, W.; Dong, N.; et al. Long noncoding rna meg3 regulates cardiomyocyte apoptosis in myocardial infarction. Gene Ther. 2018, 25, 511–523. [Google Scholar] [CrossRef]
- He, C.; Yang, W.; Yang, J.; Ding, J.; Li, S.; Wu, H.; Zhou, F.; Jiang, Y.; Teng, L.; Yang, J. Long noncoding rna meg3 negatively regulates proliferation and angiogenesis in vascular endothelial cells. DNA Cell Biol. 2017, 36, 475–481. [Google Scholar] [CrossRef]
- Michalik, K.M.; You, X.; Manavski, Y.; Doddaballapur, A.; Zornig, M.; Braun, T.; John, D.; Ponomareva, Y.; Chen, W.; Uchida, S.; et al. Long noncoding rna malat1 regulates endothelial cell function and vessel growth. Circ. Res. 2014, 114, 1389–1397. [Google Scholar] [CrossRef]
- Li, R.; Fang, L.; Pu, Q.; Bu, H.; Zhu, P.; Chen, Z.; Yu, M.; Li, X.; Weiland, T.; Bansal, A.; et al. Meg3-4 is a mirna decoy that regulates il-1beta abundance to initiate and then limit inflammation to prevent sepsis during lung infection. Sci. Signal. 2018, 11. [Google Scholar] [CrossRef]
- Voellenkle, C.; Garcia-Manteiga, J.M.; Pedrotti, S.; Perfetti, A.; De Toma, I.; Da Silva, D.; Maimone, B.; Greco, S.; Fasanaro, P.; Creo, P.; et al. Implication of long noncoding rnas in the endothelial cell response to hypoxia revealed by rna-sequencing. Sci. Rep. 2016, 6, 24141. [Google Scholar] [CrossRef]
- Tripathi, V.; Shen, Z.; Chakraborty, A.; Giri, S.; Freier, S.M.; Wu, X.; Zhang, Y.; Gorospe, M.; Prasanth, S.G.; Lal, A.; et al. Long noncoding rna malat1 controls cell cycle progression by regulating the expression of oncogenic transcription factor b-myb. PLoS Genet. 2013, 9, e1003368. [Google Scholar] [CrossRef]
- Cremer, S.; Michalik, K.M.; Fischer, A.; Pfisterer, L.; Jae, N.; Winter, C.; Boon, R.A.; Muhly-Reinholz, M.; John, D.; Uchida, S.; et al. Hematopoietic deficiency of the long non-coding rna malat1 promotes atherosclerosis and plaque inflammation. Circulation 2018, 139, 1320–1334. [Google Scholar] [CrossRef]
- Wang, G.; Li, Y.; Peng, Y.; Tang, J.; Li, H. Association of polymorphisms in malat1 with risk of coronary atherosclerotic heart disease in a Chinese population. Lipids Health Dis. 2018, 17, 75. [Google Scholar] [CrossRef]
- Zhuo, Y.; Zeng, Q.; Zhang, P.; Li, G.; Xie, Q.; Cheng, Y. Functional polymorphism of lncrna malat1 contributes to pulmonary arterial hypertension susceptibility in Chinese people. Clin. Chem. Lab. Med. 2017, 55, 38–46. [Google Scholar] [CrossRef]
- Yao, J.; Wang, X.Q.; Li, Y.J.; Shan, K.; Yang, H.; Wang, Y.N.; Yao, M.D.; Liu, C.; Li, X.M.; Shen, Y.; et al. Long non-coding rna malat1 regulates retinal neurodegeneration through creb signaling. EMBO Mol. Med. 2016, 8, 346–362. [Google Scholar] [CrossRef]
- Liu, J.Y.; Yao, J.; Li, X.M.; Song, Y.C.; Wang, X.Q.; Li, Y.J.; Yan, B.; Jiang, Q. Pathogenic role of lncrna-malat1 in endothelial cell dysfunction in diabetes mellitus. Cell Death Dis. 2014, 5, e1506. [Google Scholar] [CrossRef]
- Abdelmohsen, K.; Panda, A.; Kang, M.J.; Xu, J.; Selimyan, R.; Yoon, J.H.; Martindale, J.L.; De, S.; Wood, W.H., 3rd; Becker, K.G.; et al. Senescence-associated lncrnas: Senescence-associated long noncoding rnas. Aging Cell 2013, 12, 890–900. [Google Scholar] [CrossRef]
- Yan, B.; Yao, J.; Liu, J.Y.; Li, X.M.; Wang, X.Q.; Li, Y.J.; Tao, Z.F.; Song, Y.C.; Chen, Q.; Jiang, Q. Lncrna-miat regulates microvascular dysfunction by functioning as a competing endogenous rna. Circ. Res. 2015, 116, 1143–1156. [Google Scholar] [CrossRef]
- Zhu, M.; Li, N.; Luo, P.; Jing, W.; Wen, X.; Liang, C.; Tu, J. Peripheral blood leukocyte expression of lncrna miat and its diagnostic and prognostic value in ischemic stroke. J. Stroke Cerebrovasc. Dis. 2018, 27, 326–337. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, M.; Chen, J.; Lin, S.; Cai, D.; Chen, C.; Chen, Z. Long non-coding rna miat acts as a biomarker in diabetic retinopathy by absorbing mir-29b and regulating cell apoptosis. Biosci. Rep. 2017, 37. [Google Scholar] [CrossRef]
- Wang, H.; Ding, X.G.; Yang, J.J.; Li, S.W.; Zheng, H.; Gu, C.H.; Jia, Z.K.; Li, L. Lncrna miat facilitated bm-mscs differentiation into endothelial cells and restored erectile dysfunction via targeting mir-200a in a rat model of erectile dysfunction. Eur. J. Cell Biol. 2018, 97, 180–189. [Google Scholar] [CrossRef]
- Brannan, C.I.; Dees, E.C.; Ingram, R.S.; Tilghman, S.M. The product of the h19 gene may function as an rna. Mol. Cell. Biol. 1990, 10, 28–36. [Google Scholar] [CrossRef]
- Hofmann, P.; Sommer, J.; Theodorou, K.; Kirchhof, L.; Fischer, A.; Li, Y.; Perisic, L.; Hedin, U.; Maegdefessel, L.; Dimmeler, S.; et al. Long non-coding rna h19 regulates endothelial cell aging via inhibition of stat3 signaling. Cardiovasc. Res. 2018, 115, 230–242. [Google Scholar] [CrossRef]
- Poirier, F.; Chan, C.T.; Timmons, P.M.; Robertson, E.J.; Evans, M.J.; Rigby, P.W. The murine h19 gene is activated during embryonic stem cell differentiation in vitro and at the time of implantation in the developing embryo. Development 1991, 113, 1105–1114. [Google Scholar]
- Kim, D.K.; Zhang, L.; Dzau, V.J.; Pratt, R.E. H19, a developmentally regulated gene, is reexpressed in rat vascular smooth muscle cells after injury. J. Clin. Investig. 1994, 93, 355–360. [Google Scholar] [CrossRef]
- Han, D.K.; Khaing, Z.Z.; Pollock, R.A.; Haudenschild, C.C.; Liau, G. H19, a marker of developmental transition, is reexpressed in human atherosclerotic plaques and is regulated by the insulin family of growth factors in cultured rabbit smooth muscle cells. J. Clin. Investig. 1996, 97, 1276–1285. [Google Scholar] [CrossRef]
- Li, D.Y.; Busch, A.; Jin, H.; Chernogubova, E.; Pelisek, J.; Karlsson, J.; Sennblad, B.; Liu, S.; Lao, S.; Hofmann, P.; et al. H19 induces abdominal aortic aneurysm development and progression. Circulation 2018, 138, 1551–1568. [Google Scholar] [CrossRef]
- Hadji, F.; Boulanger, M.C.; Guay, S.P.; Gaudreault, N.; Amellah, S.; Mkannez, G.; Bouchareb, R.; Marchand, J.T.; Nsaibia, M.J.; Guauque-Olarte, S.; et al. Altered DNA methylation of long noncoding rna h19 in calcific aortic valve disease promotes mineralization by silencing notch1. Circulation 2016, 134, 1848–1862. [Google Scholar] [CrossRef]
- Zhang, Z.; Gao, W.; Long, Q.Q.; Zhang, J.; Li, Y.F.; Liu, D.C.; Yan, J.J.; Yang, Z.J.; Wang, L.S. Increased plasma levels of lncrna h19 and lipcar are associated with increased risk of coronary artery disease in a chinese population. Sci. Rep. 2017, 7, 7491. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhao, H.; Fan, Z.; Li, G.; Ma, Q.; Tao, Z.; Wang, R.; Feng, J.; Luo, Y. Long noncoding rna h19 promotes neuroinflammation in ischemic stroke by driving histone deacetylase 1-dependent m1 microglial polarization. Stroke 2017, 48, 2211–2221. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Zhu, M.; Wang, H.; Zhao, S.; Zhao, D.; Yang, Y.; Wang, Z.M.; Wang, F.; Yang, Z.J.; Lu, X.; et al. Association of polymorphisms in long non-coding rna h19 with coronary artery disease risk in a Chinese population. Mutat. Res. 2015, 772, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cao, B.; Han, D.; Sun, M.; Feng, J. Long non-coding rna h19 induces cerebral ischemia reperfusion injury via activation of autophagy. Aging Dis. 2017, 8, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Kallen, A.N.; Zhou, X.B.; Xu, J.; Qiao, C.; Ma, J.; Yan, L.; Lu, L.; Liu, C.; Yi, J.S.; Zhang, H.; et al. The imprinted h19 lncrna antagonizes let-7 micrornas. Mol. Cell 2013, 52, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Xu, X.; Yan, C.; Shi, Y.; Hu, Y.; Dong, L.; Ying, S.; Ying, K.; Zhang, R. Lncrna h19 promotes the proliferation of pulmonary artery smooth muscle cells through at1r via sponging let-7b in monocrotaline-induced pulmonary arterial hypertension. Respir. Res. 2018, 19, 254. [Google Scholar] [CrossRef]
- Groff, A.F.; Sanchez-Gomez, D.B.; Soruco, M.M.L.; Gerhardinger, C.; Barutcu, A.R.; Li, E.; Elcavage, L.; Plana, O.; Sanchez, L.V.; Lee, J.C.; et al. In vivo characterization of linc-p21 reveals functional cis-regulatory DNA elements. Cell Rep. 2016, 16, 2178–2186. [Google Scholar] [CrossRef]
- Tang, S.S.; Cheng, J.; Cai, M.Y.; Yang, X.L.; Liu, X.G.; Zheng, B.Y.; Xiong, X.D. Association of lincrna-p21 haplotype with coronary artery disease in a chinese han population. Dis. Mark. 2016, 2016, 9109743. [Google Scholar]
- Wu, G.; Cai, J.; Han, Y.; Chen, J.; Huang, Z.P.; Chen, C.; Cai, Y.; Huang, H.; Yang, Y.; Liu, Y.; et al. Lincrna-p21 regulates neointima formation, vascular smooth muscle cell proliferation, apoptosis, and atherosclerosis by enhancing p53 activity. Circulation 2014, 130, 1452–1465. [Google Scholar] [CrossRef]
- Cekin, N.; Ozcan, A.; Goksel, S.; Arslan, S.; Pinarbasi, E.; Berkan, O. Decreased fendrr and lincrna-p21 expression in atherosclerotic plaque. Anatol. J. Cardiol. 2018, 19, 131–136. [Google Scholar]
- Hu, W.; Wang, Z.; Li, Q.; Wang, J.; Li, L.; Jiang, G. Upregulation of lincrna-p21 in thoracic aortic aneurysms is involved in the regulation of proliferation and apoptosis of vascular smooth muscle cells by activating tgf-beta1 signaling pathway. J. Cell. Biochem. 2018, 120, 4113–4120. [Google Scholar] [CrossRef] [PubMed]
- Huarte, M.; Guttman, M.; Feldser, D.; Garber, M.; Koziol, M.J.; Kenzelmann-Broz, D.; Khalil, A.M.; Zuk, O.; Amit, I.; Rabani, M.; et al. A large intergenic noncoding rna induced by p53 mediates global gene repression in the p53 response. Cell 2010, 142, 409–419. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Ding, J.W.; Li, S.; Wu, H.; Jiang, Y.R.; Yang, W.; Teng, L.; Yang, J.; Yang, J. The role of long intergenic noncoding rna p21 in vascular endothelial cells. DNA Cell Biol. 2015, 34, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.H.; Abdelmohsen, K.; Kim, J.; Yang, X.; Martindale, J.L.; Tominaga-Yamanaka, K.; White, E.J.; Orjalo, A.V.; Rinn, J.L.; Kreft, S.G.; et al. Scaffold function of long non-coding rna hotair in protein ubiquitination. Nat. Commun. 2013, 4, 2939. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, M.; Chen, W.; Wang, R.; Ye, Z.; Wang, Y.; Li, X.; Cai, C. Lncrna-hotair inhibition aggravates oxidative stress-induced h9c2 cells injury through suppression of mmp2 by mir-125. Acta Biochim. Biophys. Sin. (Shanghai) 2018, 50, 996–1006. [Google Scholar] [CrossRef]
- Guo, X.; Chang, Q.; Pei, H.; Sun, X.; Qian, X.; Tian, C.; Lin, H. Long non-coding rna-mrna correlation analysis reveals the potential role of hotair in pathogenesis of sporadic thoracic aortic aneurysm. Eur. J. Vasc. Endovasc. Surg. 2017, 54, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Meng, K.; Jiang, L.; Zhong, Y.; Yang, Y.; Lan, Y.; Zeng, Q.; Cheng, L. Thymic stromal lymphopoietin-induced hotair activation promotes endothelial cell proliferation and migration in atherosclerosis. Biosci. Rep. 2017, 37. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.L.; Wang, J.W.; Hu, P.Y.; Jiang, J.S.; Yu, C. Hotair alleviates ox-ldl-induced inflammatory response in raw264.7 cells via inhibiting nf-kappab pathway. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 6991–6998. [Google Scholar]
- Jiang, Y.; Mo, H.; Luo, J.; Zhao, S.; Liang, S.; Zhang, M.; Yuan, J. Hotair is a potential novel biomarker in patients with congenital heart diseases. Biomed. Res. Int. 2018, 2018, 2850657. [Google Scholar] [CrossRef]
- Yamada, Y.; Horibe, H.; Oguri, M.; Sakuma, J.; Takeuchi, I.; Yasukochi, Y.; Kato, K.; Sawabe, M. Identification of novel hyper- or hypomethylated cpg sites and genes associated with atherosclerotic plaque using an epigenome-wide association study. Int. J. Mol. Med. 2018, 41, 2724–2732. [Google Scholar] [CrossRef]
- Liu, J.; Huang, G.Q.; Ke, Z.P. Silence of long intergenic noncoding rna hotair ameliorates oxidative stress and inflammation response in ox-ldl-treated human macrophages by upregulating mir-330-5p. J. Cell. Physiol. 2019, 234, 5134–5142. [Google Scholar] [CrossRef]
- Rapicavoli, N.A.; Qu, K.; Zhang, J.; Mikhail, M.; Laberge, R.M.; Chang, H.Y. A mammalian pseudogene lncrna at the interface of inflammation and anti-inflammatory therapeutics. Elife 2013, 2, e00762. [Google Scholar] [CrossRef] [PubMed]
- Zgheib, C.; Hodges, M.M.; Hu, J.; Liechty, K.W.; Xu, J. Long non-coding rna lethe regulates hyperglycemia-induced reactive oxygen species production in macrophages. PLoS ONE 2017, 12, e0177453. [Google Scholar] [CrossRef] [PubMed]
- Meier, I.; Fellini, L.; Jakovcevski, M.; Schachner, M.; Morellini, F. Expression of the snorna host gene gas5 in the hippocampus is upregulated by age and psychogenic stress and correlates with reduced novelty-induced behavior in c57bl/6 mice. Hippocampus 2010, 20, 1027–1036. [Google Scholar] [CrossRef]
- Cuomo, D.; Porreca, I.; Ceccarelli, M.; Threadgill, D.W.; Barrington, W.T.; Petriella, A.; D’Angelo, F.; Cobellis, G.; De Stefano, F.; D’Agostino, M.N.; et al. Transcriptional landscape of mouse-aged ovaries reveals a unique set of non-coding rnas associated with physiological and environmental ovarian dysfunctions. Cell Death Discov. 2018, 4, 112. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; She, Q. Association between the deletion allele of ins/del polymorphism (rs145204276) in the promoter region of gas5 with the risk of atherosclerosis. Cell. Physiol. Biochem. 2018, 49, 1431–1443. [Google Scholar] [CrossRef]
- Chen, L.; Yao, H.; Hui, J.Y.; Ding, S.H.; Fan, Y.L.; Pan, Y.H.; Chen, K.H.; Wan, J.Q.; Jiang, J.Y. Global transcriptomic study of atherosclerosis development in rats. Gene 2016, 592, 43–48. [Google Scholar] [CrossRef]
- Wang, Y.N.; Shan, K.; Yao, M.D.; Yao, J.; Wang, J.J.; Li, X.; Liu, B.; Zhang, Y.Y.; Ji, Y.; Jiang, Q.; et al. Long noncoding rna-gas5: A novel regulator of hypertension-induced vascular remodeling. Hypertension 2016, 68, 736–748. [Google Scholar] [CrossRef]
- Li, L.; Li, X.; The, E.; Wang, L.J.; Yuan, T.Y.; Wang, S.Y.; Feng, J.; Wang, J.; Liu, Y.; Wu, Y.H.; et al. Low expression of lncrna-gas5 is implicated in human primary varicose great saphenous veins. PLoS ONE 2015, 10, e0120550. [Google Scholar] [CrossRef]
- Li, X.; Hou, L.; Cheng, Z.; Zhou, S.; Qi, J.; Cheng, J. Overexpression of gas5 inhibits abnormal activation of wnt/beta-catenin signaling pathway in myocardial tissues of rats with coronary artery disease. J. Cell. Physiol. 2018. [Google Scholar] [CrossRef]
- Tang, R.; Zhang, G.; Wang, Y.C.; Mei, X.; Chen, S.Y. The long non-coding rna gas5 regulates transforming growth factor beta (tgf-beta)-induced smooth muscle cell differentiation via rna smad-binding elements. J. Biol. Chem. 2017, 292, 14270–14278. [Google Scholar] [CrossRef]
- Shihabudeen Haider Ali, M.S.; Cheng, X.; Moran, M.; Haemmig, S.; Naldrett, M.J.; Alvarez, S.; Feinberg, M.W.; Sun, X. Lncrna meg3 protects endothelial function by regulating the DNA damage response. Nucleic Acids Res. 2018, 47, 1505–1522. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, Z.; Trottier, J.; Barbier, O.; Wang, L. Long noncoding rna meg3 induces cholestatic liver injury by interaction with ptbp1 to facilitate shp mrna decay. Hepatology 2017, 65, 604–615. [Google Scholar] [CrossRef]
- Zhu, J.; Liu, S.; Ye, F.; Shen, Y.; Tie, Y.; Zhu, J.; Wei, L.; Jin, Y.; Fu, H.; Wu, Y.; et al. Long noncoding rna meg3 interacts with p53 protein and regulates partial p53 target genes in hepatoma cells. PLoS ONE 2015, 10, e0139790. [Google Scholar] [CrossRef]
- Lino Cardenas, C.L.; Kessinger, C.W.; Cheng, Y.; MacDonald, C.; MacGillivray, T.; Ghoshhajra, B.; Huleihel, L.; Nuri, S.; Yeri, A.S.; Jaffer, F.A.; et al. An hdac9-malat1-brg1 complex mediates smooth muscle dysfunction in thoracic aortic aneurysm. Nat. Commun. 2018, 9, 1009. [Google Scholar] [CrossRef]
- Betancur, J.G.; Tomari, Y. Cryptic rna-binding by prc2 components ezh2 and suz12. RNA Biol. 2015, 12, 959–965. [Google Scholar] [CrossRef]
- Tsai, M.C.; Manor, O.; Wan, Y.; Mosammaparast, N.; Wang, J.K.; Lan, F.; Shi, Y.; Segal, E.; Chang, H.Y. Long noncoding rna as modular scaffold of histone modification complexes. Science 2010, 329, 689–693. [Google Scholar] [CrossRef]
- Rinn, J.L.; Kertesz, M.; Wang, J.K.; Squazzo, S.L.; Xu, X.; Brugmann, S.A.; Goodnough, L.H.; Helms, J.A.; Farnham, P.J.; Segal, E.; et al. Functional demarcation of active and silent chromatin domains in human hox loci by noncoding rnas. Cell 2007, 129, 1311–1323. [Google Scholar] [CrossRef]
- Boudreau, N.; Andrews, C.; Srebrow, A.; Ravanpay, A.; Cheresh, D.A. Induction of the angiogenic phenotype by hox d3. J. Cell Biol. 1997, 139, 257–264. [Google Scholar] [CrossRef]
- Myers, C.; Charboneau, A.; Cheung, I.; Hanks, D.; Boudreau, N. Sustained expression of homeobox d10 inhibits angiogenesis. Am. J. Pathol. 2002, 161, 2099–2109. [Google Scholar] [CrossRef]
- Monnier, P.; Martinet, C.; Pontis, J.; Stancheva, I.; Ait-Si-Ali, S.; Dandolo, L. H19 lncrna controls gene expression of the imprinted gene network by recruiting mbd1. Proc. Natl. Acad. Sci. USA 2013, 110, 20693–20698. [Google Scholar] [CrossRef]
- Zaina, S.; Pettersson, L.; Ahren, B.; Branen, L.; Hassan, A.B.; Lindholm, M.; Mattsson, R.; Thyberg, J.; Nilsson, J. Insulin-like growth factor ii plays a central role in atherosclerosis in a mouse model. J. Biol. Chem. 2002, 277, 4505–4511. [Google Scholar] [CrossRef]
- Casellas, A.; Mallol, C.; Salavert, A.; Jimenez, V.; Garcia, M.; Agudo, J.; Obach, M.; Haurigot, V.; Vila, L.; Molas, M.; et al. Insulin-like growth factor 2 overexpression induces beta-cell dysfunction and increases beta-cell susceptibility to damage. J. Biol. Chem. 2015, 290, 16772–16785. [Google Scholar] [CrossRef]
- Kalwa, M.; Hanzelmann, S.; Otto, S.; Kuo, C.C.; Franzen, J.; Joussen, S.; Fernandez-Rebollo, E.; Rath, B.; Koch, C.; Hofmann, A.; et al. The lncrna hotair impacts on mesenchymal stem cells via triple helix formation. Nucleic Acids Res. 2016, 44, 10631–10643. [Google Scholar] [CrossRef]
- Yap, K.L.; Li, S.; Munoz-Cabello, A.M.; Raguz, S.; Zeng, L.; Mujtaba, S.; Gil, J.; Walsh, M.J.; Zhou, M.M. Molecular interplay of the noncoding rna anril and methylated histone h3 lysine 27 by polycomb cbx7 in transcriptional silencing of ink4a. Mol. Cell 2010, 38, 662–674. [Google Scholar] [CrossRef]
- Broadbent, H.M.; Peden, J.F.; Lorkowski, S.; Goel, A.; Ongen, H.; Green, F.; Clarke, R.; Collins, R.; Franzosi, M.G.; Tognoni, G.; et al. Susceptibility to coronary artery disease and diabetes is encoded by distinct, tightly linked snps in the anril locus on chromosome 9p. Hum. Mol. Genet. 2008, 17, 806–814. [Google Scholar] [CrossRef]
- Yari, M.; Bitarafan, S.; Broumand, M.A.; Fazeli, Z.; Rahimi, M.; Ghaderian, S.M.H.; Mirfakhraie, R.; Omrani, M.D. Association between long noncoding rna anril expression variants and susceptibility to coronary artery disease. Int. J. Mol. Cell. Med. 2018, 7, 1–7. [Google Scholar]
- Holdt, L.M.; Beutner, F.; Scholz, M.; Gielen, S.; Gabel, G.; Bergert, H.; Schuler, G.; Thiery, J.; Teupser, D. Anril expression is associated with atherosclerosis risk at chromosome 9p21. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 620–627. [Google Scholar] [CrossRef]
- Yasuno, K.; Bilguvar, K.; Bijlenga, P.; Low, S.K.; Krischek, B.; Auburger, G.; Simon, M.; Krex, D.; Arlier, Z.; Nayak, N.; et al. Genome-wide association study of intracranial aneurysm identifies three new risk loci. Nat. Genet. 2010, 42, 420–425. [Google Scholar] [CrossRef]
- Scott, L.J.; Mohlke, K.L.; Bonnycastle, L.L.; Willer, C.J.; Li, Y.; Duren, W.L.; Erdos, M.R.; Stringham, H.M.; Chines, P.S.; Jackson, A.U.; et al. A genome-wide association study of type 2 diabetes in finns detects multiple susceptibility variants. Science 2007, 316, 1341–1345. [Google Scholar] [CrossRef]
- Kotake, Y.; Nakagawa, T.; Kitagawa, K.; Suzuki, S.; Liu, N.; Kitagawa, M.; Xiong, Y. Long non-coding rna anril is required for the prc2 recruitment to and silencing of p15(ink4b) tumor suppressor gene. Oncogene 2011, 30, 1956–1962. [Google Scholar] [CrossRef]
- Thomas, A.A.; Feng, B.; Chakrabarti, S. Anril: A regulator of vegf in diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2017, 58, 470–480. [Google Scholar] [CrossRef]
- Zhou, X.; Han, X.; Wittfeldt, A.; Sun, J.; Liu, C.; Wang, X.; Gan, L.M.; Cao, H.; Liang, Z. Long non-coding rna anril regulates inflammatory responses as a novel component of nf-kappab pathway. RNA Biol. 2016, 13, 98–108. [Google Scholar] [CrossRef]
- Boekholdt, S.M.; Peters, R.J.; Hack, C.E.; Day, N.E.; Luben, R.; Bingham, S.A.; Wareham, N.J.; Reitsma, P.H.; Khaw, K.T. Il-8 plasma concentrations and the risk of future coronary artery disease in apparently healthy men and women: The epic-norfolk prospective population study. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1503–1508. [Google Scholar] [CrossRef]
- Zhang, B.; Ma, Y.; Xiang, C. Sirt2 decreases atherosclerotic plaque formation in low-density lipoprotein receptor-deficient mice by modulating macrophage polarization. Biomed. Pharmacother. 2018, 97, 1238–1242. [Google Scholar] [CrossRef]
- Seki, N.; Bujo, H.; Jiang, M.; Tanaga, K.; Takahashi, K.; Yagui, K.; Hashimoto, N.; Schneider, W.J.; Saito, Y. Lrp1b is a negative modulator of increased migration activity of intimal smooth muscle cells from rabbit aortic plaques. Biochem. Biophys. Res. Commun. 2005, 331, 964–970. [Google Scholar] [CrossRef]
- Sun, J.; Ermann, J.; Niu, N.; Yan, G.; Yang, Y.; Shi, Y.; Zou, W. Histone demethylase lsd1 regulates bone mass by controlling wnt7b and bmp2 signaling in osteoblasts. Bone Res. 2018, 6, 14. [Google Scholar] [CrossRef]
- Bianchessi, V.; Badi, I.; Bertolotti, M.; Nigro, P.; D’Alessandra, Y.; Capogrossi, M.C.; Zanobini, M.; Pompilio, G.; Raucci, A.; Lauri, A. The mitochondrial lncrna asncmtrna-2 is induced in aging and replicative senescence in endothelial cells. J. Mol. Cell. Cardiol. 2015, 81, 62–70. [Google Scholar] [CrossRef]
- Burzio, V.A.; Villota, C.; Villegas, J.; Landerer, E.; Boccardo, E.; Villa, L.L.; Martinez, R.; Lopez, C.; Gaete, F.; Toro, V.; et al. Expression of a family of noncoding mitochondrial rnas distinguishes normal from cancer cells. Proc. Natl. Acad. Sci. USA 2009, 106, 9430–9434. [Google Scholar] [CrossRef]
- Gao, Y.; Chen, Z.Y.; Wang, Y.; Liu, Y.; Ma, J.X.; Li, Y.K. Long non-coding rna asncmtrna-2 is upregulated in diabetic kidneys and high glucose-treated mesangial cells. Exp. Ther. Med. 2017, 13, 581–587. [Google Scholar] [CrossRef]
- Paloschi, V.; Kurtovic, S.; Folkersen, L.; Gomez, D.; Wagsater, D.; Roy, J.; Petrini, J.; Eriksson, M.J.; Caidahl, K.; Hamsten, A.; et al. Impaired splicing of fibronectin is associated with thoracic aortic aneurysm formation in patients with bicuspid aortic valve. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 691–697. [Google Scholar] [CrossRef]
- Zhang, K.; Luo, Z.; Zhang, Y.; Zhang, L.; Wu, L.; Liu, L.; Yang, J.; Song, X.; Liu, J. Circulating lncrna h19 in plasma as a novel biomarker for breast cancer. Cancer Biomark. 2016, 17, 187–194. [Google Scholar] [CrossRef]
- Zhou, X.; Yin, C.; Dang, Y.; Ye, F.; Zhang, G. Identification of the long non-coding rna h19 in plasma as a novel biomarker for diagnosis of gastric cancer. Sci. Rep. 2015, 5, 11516. [Google Scholar] [CrossRef]
- Yin, Q.; Wu, A.; Liu, M. Plasma long non-coding rna (lncrna) gas5 is a new biomarker for coronary artery disease. Med. Sci. Monit. 2017, 23, 6042–6048. [Google Scholar] [CrossRef]
- Liang, W.; Lv, T.; Shi, X.; Liu, H.; Zhu, Q.; Zeng, J.; Yang, W.; Yin, J.; Song, Y. Circulating long noncoding rna gas5 is a novel biomarker for the diagnosis of nonsmall cell lung cancer. Medicine (Baltimore) 2016, 95, e4608. [Google Scholar] [CrossRef]
- Yu, F.; Zhou, G.; Huang, K.; Fan, X.; Li, G.; Chen, B.; Dong, P.; Zheng, J. Serum lincrna-p21 as a potential biomarker of liver fibrosis in chronic hepatitis b patients. J. Viral Hepat. 2017, 24, 580–588. [Google Scholar] [CrossRef]
- Zhao, W.; Song, M.; Zhang, J.; Kuerban, M.; Wang, H. Combined identification of long non-coding rna ccat1 and hotair in serum as an effective screening for colorectal carcinoma. Int. J. Clin. Exp. Pathol. 2015, 8, 14131–14140. [Google Scholar]
- Liu, M.; Xing, L.Q.; Liu, Y.J. A three-long noncoding rna signature as a diagnostic biomarker for differentiating between triple-negative and non-triple-negative breast cancers. Medicine (Baltimore) 2017, 96, e6222. [Google Scholar] [CrossRef]
- McPherson, R.; Pertsemlidis, A.; Kavaslar, N.; Stewart, A.; Roberts, R.; Cox, D.R.; Hinds, D.A.; Pennacchio, L.A.; Tybjaerg-Hansen, A.; Folsom, A.R.; et al. A common allele on chromosome 9 associated with coronary heart disease. Science 2007, 316, 1488–1491. [Google Scholar] [CrossRef]
- Aleyasin, S.A.; Navidi, T.; Davoudi, S. Association between rs10757274 and rs2383206 snps as genetic risk factors in iranian patients with coronary artery disease. J. Tehran Heart Cent. 2017, 12, 114–118. [Google Scholar]
- Nawaz, S.K.; Noreen, A.; Rani, A.; Yousaf, M.; Arshad, M. Association of the rs10757274 snp with coronary artery disease in a small group of a pakistani population. Anatol. J. Cardiol. 2015, 15, 709–715. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bink, D.I.; Lozano-Vidal, N.; Boon, R.A. Long Non-Coding RNA in Vascular Disease and Aging. Non-Coding RNA 2019, 5, 26. https://doi.org/10.3390/ncrna5010026
Bink DI, Lozano-Vidal N, Boon RA. Long Non-Coding RNA in Vascular Disease and Aging. Non-Coding RNA. 2019; 5(1):26. https://doi.org/10.3390/ncrna5010026
Chicago/Turabian StyleBink, Diewertje I., Noelia Lozano-Vidal, and Reinier A. Boon. 2019. "Long Non-Coding RNA in Vascular Disease and Aging" Non-Coding RNA 5, no. 1: 26. https://doi.org/10.3390/ncrna5010026
APA StyleBink, D. I., Lozano-Vidal, N., & Boon, R. A. (2019). Long Non-Coding RNA in Vascular Disease and Aging. Non-Coding RNA, 5(1), 26. https://doi.org/10.3390/ncrna5010026




