Serum Levels of miR-148a and miR-21-5p Are Increased in Type 1 Diabetic Patients and Correlated with Markers of Bone Strength and Metabolism
Abstract
:1. Introduction
2. Results
2.1. A Specific Set of miRNAs Is Altered in T1D and Involved in Bone Metabolism
2.2. CirculatingLevels of miR-148a and miR-21-5p Are Increased in Sera of T1D Patients vs. Control Subjects
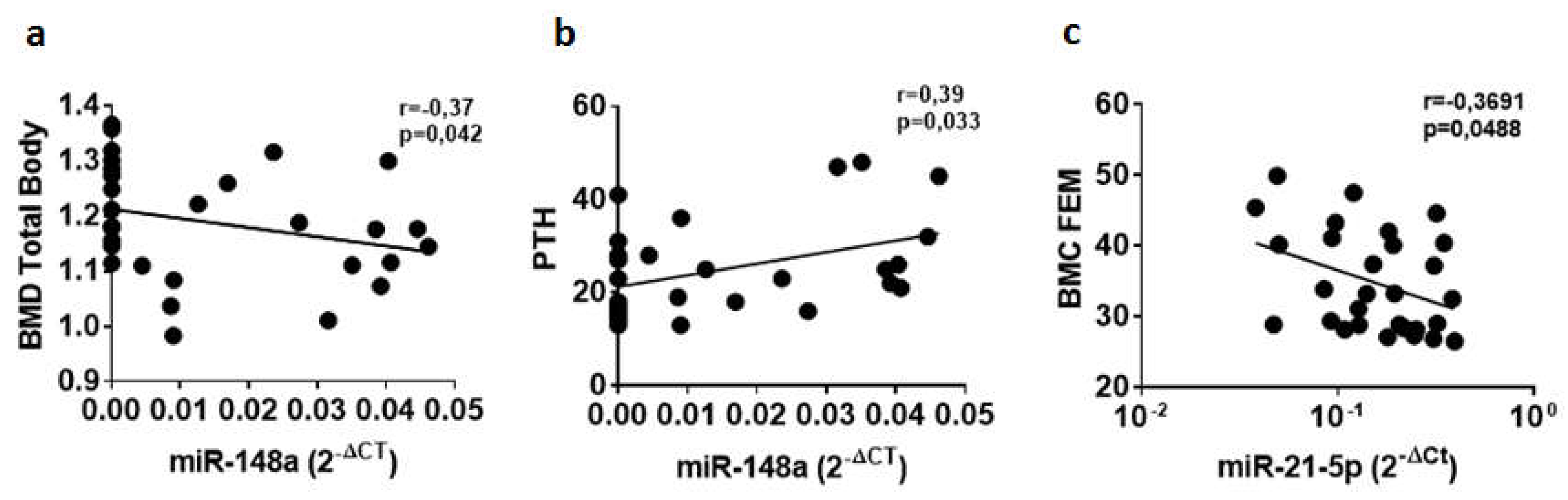
2.3. Circulating miR-148a and miR-21-5p Levels Are Associated with BMD-Total Body (TB), PTH, and BMC-FEM
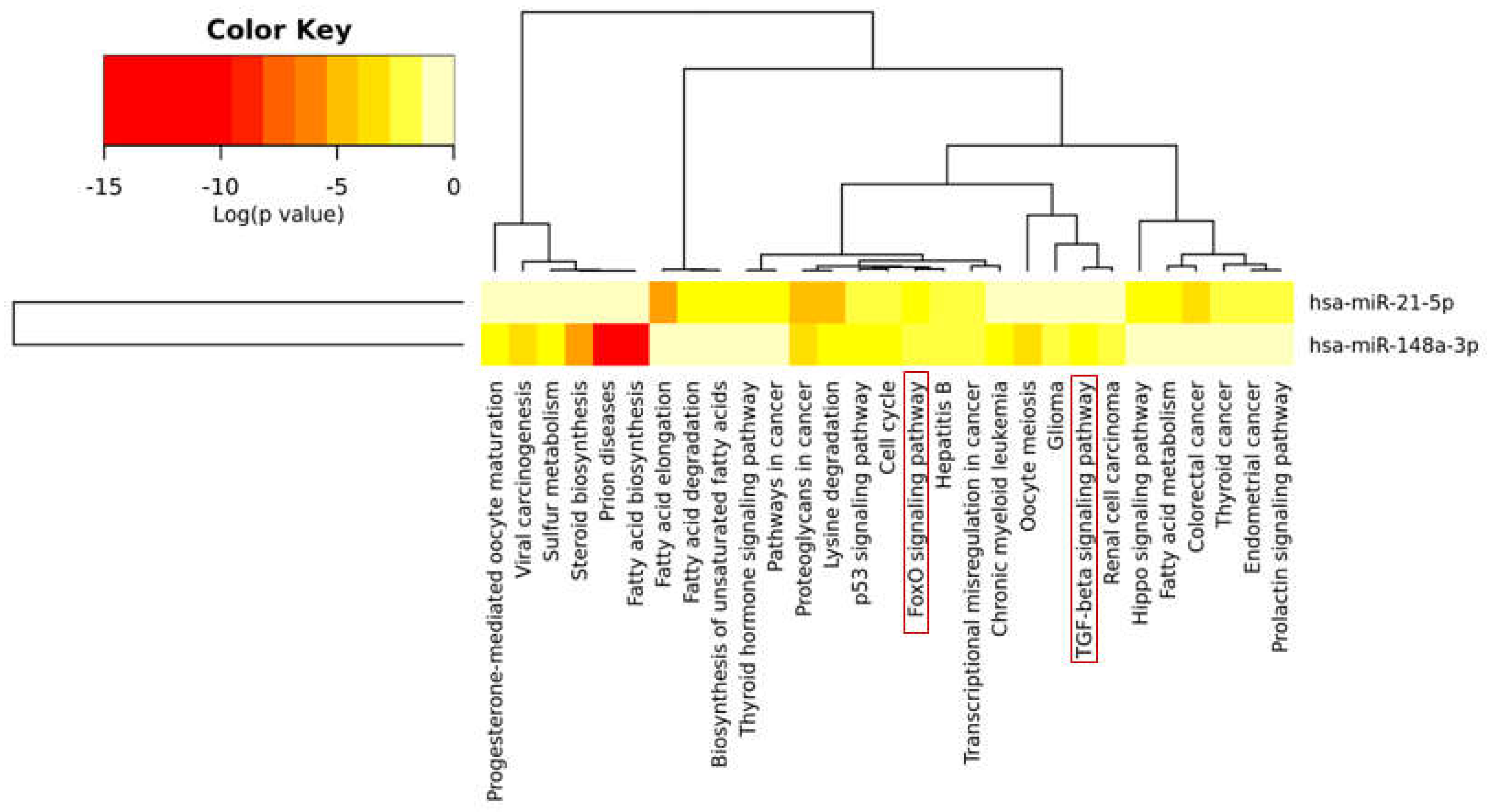
2.4. Bioinformatic Analysis Revealed that miR-148a and miR-21-5p Target Several Genes Involved in Bone Metabolism and Bone Remodeling
3. Discussion
4. Materials and Methods
4.1. Ethical Statement
4.2. Study Population
4.3. Samples Collections and Clinical Analysis
4.4. RNA Extraction
4.5. Single Assay qRT Real-Time PCR
4.6. Bioinformatic Analysis
4.7. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Janghorbani, M.; Feskanich, D.; Willett, W.C.; Hu, F. Prospective study of diabetes and risk of hip fracture: The Nurses’ Health Study. Diabetes Care 2006, 29, 1573–1578. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.R.; Haynes, K.; Leonard, M.B.; Willi, S.M.; Denburg, M.R. Type 1 diabetes is associated with an increased risk of fracture across the life span: A population-based cohort study using The Health Improvement Network (THIN). Diabetes Care 2015, 38, 1913–1920. [Google Scholar] [CrossRef] [PubMed]
- Eller-Vainicher, C.; Zhukouskaya, V.V.; Tolkachev, Y.V.; Koritko, S.S.; Cairoli, E.; Grossi, E.; Beck-Peccoz, P.; Chiodini, I.; Shepelkevich, A.P. Low bone mineral density and its predictors in type 1 diabetic patients evaluated by the classic statistics and artificial neural network analysis. Diabetes Care 2011, 34, 2186–2191. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Varthakavi, P.; Chadha, M.; Bhagwat, N. A study of bone mineral density and its determinants in type 1 diabetes mellitus. J. Osteoporos. 2013, 2013, 397814. [Google Scholar] [CrossRef] [PubMed]
- Bonds, D.E.; Larson, J.C.; Schwartz, A.V.; Strotmeyer, E.S.; Robbins, J.; Rodriguez, B.L.; Johnson, K.C.; Margolis, K.L. Risk of fracture in women with type 2 diabetes: The Women’s Health Initiative Observational Study. J. Clin. Endocrinol. Metab. 2006, 91, 3404–3410. [Google Scholar] [CrossRef]
- Ma, L.; Oei, L.; Jiang, L.; Estrada, K.; Chen, H.; Wang, Z.; Yu, Q.; Zillikens, M.C.; Gao, X.; Rivadeneira, F. Association between bone mineral density and type 2 diabetes mellitus: A meta-analysis of observational studies. Eur. J. Epidemiol. 2012, 27, 319–332. [Google Scholar] [CrossRef]
- Gilbert, L.; He, X.; Farmer, P.; Boden, S.; Kozlowski, M.; Rubin, J.; Nanes, M.S. Inhibition of osteoblast differentiation by tumor necrosis factor-α. Endocrinology 2000, 141, 3956–3964. [Google Scholar] [CrossRef]
- De Waard, E.A.C.; de Jong, J.J.A.; Koster, A.; Savelberg, H.H.C.M.; van Geel, T.A.; Houben, A.J.H.M.; Schram, M.T.; Dagnelie, P.C.; van der Kallen, C.J.; Sep, S.J.S.; et al. The association between diabetes status, HbA1c, diabetes duration, microvascular disease, and bone quality of the distal radius and tibia as measured with high-resolution peripheral quantitative computed tomography—The Maastricht Study. In Osteoporosis International; Springer: Berlin, Germany, 2018. [Google Scholar]
- Veldhuis-Vlug, A.G.; Rosen, C.J. Mechanisms of marrow adiposity and its implications for skeletal health. Metab. Clin. Exp. 2017, 67, 106–114. [Google Scholar] [CrossRef] [Green Version]
- Balint, E.; Szabo, P.; Marshall, C.F.; Sprague, S.M. Glucose-induced inhibition of in vitro bone mineralization. Bone 2001, 28, 21–28. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, X.; Wang, W.; Liu, J. Insulin stimulates osteoblast proliferation and differentiation through ERK and PI3K in MG-63 cells. Cell Biochem. Funct. 2010, 28, 334–341. [Google Scholar] [CrossRef]
- Gandhi, A.; Beam, H.A.; O’Connor, J.P.; Parsons, J.R.; Lin, S.S. The effects of local insulin delivery on diabetic fracture healing. Bone 2005, 37, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Roggli, E.; Gattesco, S.; Caille, D.; Briet, C.; Boitard, C.; Meda, P.; Regazzi, R. Changes in microRNA expression contribute to pancreatic β-cell dysfunction in prediabetic NOD mice. Diabetes 2012, 61, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
- Sebastiani, G.; Ventriglia, G.; Stabilini, A.; Socci, C.; Morsiani, C.; Laurenzi, A.; Nigi, L.; Formichi, C.; Mfarrej, B.; Petrelli, A.; et al. Regulatory T-cells from pancreatic lymphnodes of patients with type-1 diabetes express increased levels of microRNA miR-125a-5p that limits CCR2 expression. Sci. Rep. 2017, 7, 6897. [Google Scholar] [CrossRef] [PubMed]
- Sebastiani, G.; Grieco, F.A.; Spagnuolo, I.; Galleri, L.; Cataldo, D.; Dotta, F. Increased expression of microRNA miR-326 in type 1 diabetic patients with ongoing islet autoimmunity. Diabetes Metab. Res. Rev. 2011, 27, 862–866. [Google Scholar] [CrossRef] [PubMed]
- Dotta, F.; Ventriglia, G.; Snowhite, I.V.; Pugliese, A. MicroRNAs: Markers of β-cell stress and autoimmunity. Curr. Opin. Endocrinol. Diabetes Obes. 2018, 25, 237–245. [Google Scholar] [CrossRef]
- Barutta, F.; Bellini, S.; Mastrocola, R.; Bruno, G.; Gruden, G. Microrna and microvascular complications of diabetes. Int. J. Endocrinol. 2018, 2018, 6890501. [Google Scholar] [CrossRef]
- Chen, Z.; Zhang, Y.; Liang, C.; Chen, L.; Zhang, G.; Qian, A. Mechanosensitive miRNAs and Bone Formation. Int. J. Mol. Sci. 2017, 18, 1684. [Google Scholar] [CrossRef]
- Gennari, L.; Bianciardi, S.; Merlotti, D. MicroRNAs in bone diseases. Osteoporos. Int. 2017, 28, 1191–1213. [Google Scholar] [CrossRef]
- Turchinovich, A.; Weiz, L.; Langheinz, A.; Burwinkel, B. Characterization of extracellular circulating microRNA. Nucleic Acids Res. 2011, 39, 7223–7233. [Google Scholar] [CrossRef] [Green Version]
- Weiland, M.; Gao, X.-H.; Zhou, L.; Mi, Q.-S. Small RNAs have a large impact: Circulating microRNAs as biomarkers for human diseases. RNA Biol. 2012, 9, 850–859. [Google Scholar] [CrossRef] [PubMed]
- Waters, P.S.; McDermott, A.M.; Wall, D.; Heneghan, H.M.; Miller, N.; Newell, J.; Kerin, M.J.; Dwyer, R.M. Relationship between circulating and tissue microRNAs in a murine model of breast cancer. PLoS ONE 2012, 7, e50459. [Google Scholar] [CrossRef] [PubMed]
- Guay, C.; Regazzi, R. Exosomes as new players in metabolic organ cross-talk. Diabetes Obes. Metab. 2017, 19 (Suppl. 1), 137–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomou, T.; Mori, M.A.; Dreyfuss, J.M.; Konishi, M.; Sakaguchi, M.; Wolfrum, C.; Rao, T.N.; Winnay, J.N.; Garcia-Martin, R.; Grinspoon, S.K.; et al. Adipose-derived circulating miRNAs regulate gene expression in other tissues. Nature 2017, 542, 450–455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sebastiani, G.; Nigi, L.; Grieco, G.E.; Mancarella, F.; Ventriglia, G.; Dotta, F. Circulating microRNAs and diabetes mellitus: A novel tool for disease prediction, diagnosis, and staging? J. Endocrinol. Investig. 2017, 40, 591–610. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, L.B.; Wang, C.; Sørensen, K.; Bang-Berthelsen, C.H.; Hansen, L.; Andersen, M.-L.M.; Hougaard, P.; Juul, A.; Zhang, C.-Y.; Pociot, F.; Mortensen, H.B. Circulating levels of microRNA from children with newly diagnosed type 1 diabetes and healthy controls: Evidence that miR-25 associates to residual β-cell function and glycaemic control during disease progression. Exp. Diabetes Res. 2012, 2012, 896362. [Google Scholar] [PubMed]
- Seyhan, A.A.; Nunez Lopez, Y.O.; Xie, H.; Yi, F.; Mathews, C.; Pasarica, M.; Pratley, R.E. Pancreas-enriched miRNAs are altered in the circulation of subjects with diabetes: A pilot cross-sectional study. Sci. Rep. 2016, 6, 31479. [Google Scholar] [CrossRef]
- Assmann, T.S.; Recamonde-Mendoza, M.; De Souza, B.M.; Crispim, D. MicroRNA expression profiles and type 1 diabetes mellitus: Systematic review and bioinformatic analysis. Endocr. Connect. 2017, 6, 773–790. [Google Scholar] [CrossRef]
- Åkerman, L.; Casas, R.; Ludvigsson, J.; Tavira, B.; Skoglund, C. Serum miRNA levels are related to glucose homeostasis and islet autoantibodies in children with high risk for type 1 diabetes. PLoS ONE 2018, 13, e0191067. [Google Scholar] [CrossRef]
- Erener, S.; Marwaha, A.; Tan, R.; Panagiotopoulos, C.; Kieffer, T.J. Profiling of circulating microRNAs in children with recent onset of type 1 diabetes. JCI Insight 2017, 2, e89656. [Google Scholar] [CrossRef]
- Samandari, N.; Mirza, A.H.; Nielsen, L.B.; Kaur, S.; Hougaard, P.; Fredheim, S.; Mortensen, H.B.; Pociot, F. Circulating microRNA levels predict residual β cell function and glycaemic control in children with type 1 diabetes mellitus. Diabetologia 2017, 60, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Bedene, A.; Mencej Bedrač, S.; Ješe, L.; Marc, J.; Vrtačnik, P.; Preželj, J.; Kocjan, T.; Kranjc, T.; Ostanek, B. miR-148a the epigenetic regulator of bone homeostasis is increased in plasma of osteoporotic postmenopausal women. Wien. Klin. Wochenschr. 2016, 128, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Seeliger, C.; Karpinski, K.; Haug, A.T.; Vester, H.; Schmitt, A.; Bauer, J.S.; van Griensven, M. Five freely circulating miRNAs and bone tissue miRNAs are associated with osteoporotic fractures. J. Bone Miner. Res. 2014, 29, 1718–1728. [Google Scholar] [CrossRef]
- Li, H.; Wang, Z.; Fu, Q.; Zhang, J. Plasma miRNA levels correlate with sensitivity to bone mineral density in postmenopausal osteoporosis patients. Biomarkers 2014, 19, 553–556. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Wang, G.; Hu, C.; Shi, Y.; Liao, L.; Shi, S.; Cai, Y.; Cheng, S.; Wang, X.; Liu, Y.; et al. Tumor necrosis factor α suppresses the mesenchymal stem cell osteogenesis promoter miR-21 in estrogen deficiency-induced osteoporosis. J. Bone Miner. Res. 2013, 28, 559–573. [Google Scholar] [CrossRef] [Green Version]
- Zhao, C.; Sun, W.; Zhang, P.; Ling, S.; Li, Y.; Zhao, D.; Peng, J.; Wang, A.; Li, Q.; Song, J.; et al. miR-214 promotes osteoclastogenesis by targeting Pten/PI3k/Akt pathway. RNA Biol. 2015, 12, 343–353. [Google Scholar] [CrossRef] [Green Version]
- Du, F.; Wu, H.; Zhou, Z.; Liu, Y.U. microRNA-375 inhibits osteogenic differentiation by targeting runt-related transcription factor. Exp. Ther. Med. 2015, 10, 207–212. [Google Scholar] [CrossRef]
- You, L.; Pan, L.; Chen, L.; Gu, W.; Chen, J. MiR-27a is Essential for the Shift from Osteogenic Differentiation to Adipogenic Differentiation of Mesenchymal Stem Cells in Postmenopausal Osteoporosis. Cell. Physiol. Biochem. 2016, 39, 253–265. [Google Scholar] [CrossRef]
- Zeng, H.-C.; Bae, Y.; Dawson, B.C.; Chen, Y.; Bertin, T.; Munivez, E.; Campeau, P.M.; Tao, J.; Chen, R.; Lee, B.H. MicroRNA miR-23a cluster promotes osteocyte differentiation by regulating TGF-β signalling in osteoblasts. Nat. Commun. 2017, 8, 15000. [Google Scholar] [CrossRef]
- Gennari, L.; Merlotti, D.; Valenti, R.; Ceccarelli, E.; Ruvio, M.; Pietrini, M.G.; Capodarca, C.; Franci, M.B.; Campagna, M.S.; Calabrò, A.; et al. Circulating sclerostin levels and bone turnover in type 1 and type 2 diabetes. J. Clin. Endocrinol. Metab. 2012, 97, 1737–1744. [Google Scholar] [CrossRef]
- Shah, V.N.; Harrall, K.K.; Shah, C.S.; Gallo, T.L.; Joshee, P.; Snell-Bergeon, J.K.; Kohrt, W.M. Bone mineral density at femoral neck and lumbar spine in adults with type 1 diabetes: A meta-analysis and review of the literature. Osteoporos. Int. 2017, 28, 2601–2610. [Google Scholar] [CrossRef] [PubMed]
- Kaur, H.; Joshee, P.; Franquemont, S.; Baumgartner, A.; Thurston, J.; Pyle, L.; Nadeau, K.J.; Shah, V.N. Bone mineral content and bone density is lower in adolescents with type 1 diabetes: A brief report from the RESISTANT and EMERALD studies. J. Diabetes Complicat. 2018, 32, 931–933. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; Chen, C.; He, H.-B.; Hu, R.; Zhou, H.-D.; Xie, H.; Zhu, W.; Dai, R.-C.; Wu, X.-P.; Liao, E.-Y.; et al. miR-148a regulates osteoclastogenesis by targeting V-maf musculoaponeurotic fibrosarcoma oncogene homolog B. J. Bone Miner. Res. 2013, 28, 1180–1190. [Google Scholar] [CrossRef]
- Mu, J.; Zhu, D.; Shen, Z.; Ning, S.; Liu, Y.; Chen, J.; Li, Y.; Li, Z. The repressive effect of miR-148a on Wnt/β-catenin signaling involved in Glabridin-induced anti-angiogenesis in human breast cancer cells. BMC Cancer 2017, 17, 307. [Google Scholar] [CrossRef]
- Shi, C.; Zhang, M.; Tong, M.; Yang, L.; Pang, L.; Chen, L.; Xu, G.; Chi, X.; Hong, Q.; Ni, Y.; et al. miR-148a is Associated with Obesity and Modulates Adipocyte Differentiation of Mesenchymal Stem Cells through Wnt Signaling. Sci. Rep. 2015, 5, 9930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joeng, K.S.; Lee, Y.-C.; Lim, J.; Chen, Y.; Jiang, M.-M.; Munivez, E.; Ambrose, C.; Lee, B.H. Osteocyte-specific WNT1 regulates osteoblast function during bone homeostasis. J. Clin. Investig. 2017, 127, 2678–2688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J. JAK-STAT and bone metabolism. Jakstat 2013, 2, e23930. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Newnum, A.B.; Martin, J.R.; Li, P.; Nelson, M.T.; Moh, A.; Fu, X.-Y.; Yokota, H.; Li, J. Osteoblast/osteocyte-specific inactivation of Stat3 decreases load-driven bone formation and accumulates reactive oxygen species. Bone 2011, 49, 404–411. [Google Scholar] [CrossRef]
- Rached, M.-T.; Kode, A.; Xu, L.; Yoshikawa, Y.; Paik, J.-H.; Depinho, R.A.; Kousteni, S. FoxO1 is a positive regulator of bone formation by favoring protein synthesis and resistance to oxidative stress in osteoblasts. Cell Metab. 2010, 11, 147–160. [Google Scholar] [CrossRef]
- Dou, C.; Li, N.; Ding, N.; Liu, C.; Yang, X.; Kang, F.; Cao, Z.; Quan, H.; Hou, T.; Xu, J.; et al. HDAC2 Regulates FOXO1 during RANKL-Induced Osteoclastogenesis. Am. J. Physiol. Cell Physiol. 2016, 310, C780–C787. [Google Scholar] [CrossRef]
- Iyer, S.; Ambrogini, E.; Bartell, S.M.; Han, L.; Roberson, P.K.; de Cabo, R.; Jilka, R.L.; Weinstein, R.S.; O’Brien, C.A.; Manolagas, S.C.; et al. FOXOs attenuate bone formation by suppressing Wnt signaling. J. Clin. Investig. 2013, 123, 3409–3419. [Google Scholar] [CrossRef] [Green Version]
- Kode, A.; Mosialou, I.; Silva, B.C.; Joshi, S.; Ferron, M.; Rached, M.T.; Kousteni, S. FoxO1 protein cooperates with ATF4 protein in osteoblasts to control glucose homeostasis. J. Biol. Chem. 2012, 287, 8757–8768. [Google Scholar] [CrossRef]
- Wang, Y.; Dong, G.; Jeon, H.H.; Elazizi, M.; La, L.B.; Hameedaldeen, A.; Xiao, E.; Tian, C.; Alsadun, S.; Choi, Y.; et al. FOXO1 mediates RANKL-induced osteoclast formation and activity. J. Immunol. 2015, 194, 2878–2887. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Vander Ark, A.; Lee, P.; Hostetter, G.; Bhowmick, N.A.; Matrisian, L.M.; Williams, B.O.; Miranti, C.K.; Li, X. Myeloid-specific TGF-β signaling in bone promotes basic-FGF and breast cancer bone metastasis. Oncogene 2016, 35, 2370–2378. [Google Scholar] [CrossRef]
- Meng, X.; Vander Ark, A.; Daft, P.; Woodford, E.; Wang, J.; Madaj, Z.; Li, X. Loss of TGF-β signaling in osteoblasts increases basic-FGF and promotes prostate cancer bone metastasis. Cancer Lett. 2018, 418, 109–118. [Google Scholar] [CrossRef]
- Wu, M.; Chen, G.; Li, Y.-P. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 2016, 4, 16009. [Google Scholar] [CrossRef]
- Lamora, A.; Talbot, J.; Mullard, M.; Brounais-Le Royer, B.; Redini, F.; Verrecchia, F. TGF-β Signaling in Bone Remodeling and Osteosarcoma Progression. J. Clin. Med. 2016, 5, 96. [Google Scholar] [CrossRef]
- Regazzi, R. MicroRNAs as therapeutic targets for the treatment of diabetes mellitus and its complications. Expert Opin. Ther. Targets 2018, 22, 153–160. [Google Scholar] [CrossRef]
- Wang, C.; Wan, S.; Yang, T.; Niu, D.; Zhang, A.; Yang, C.; Cai, J.; Wu, J.; Song, J.; Zhang, C.-Y.; et al. Increased serum microRNAs are closely associated with the presence of microvascular complications in type 2 diabetes mellitus. Sci. Rep. 2016, 6, 20032. [Google Scholar] [CrossRef] [Green Version]
- Weber, D.R.; Schwartz, G. Epidemiology of skeletal health in type 1 diabetes. Curr. Osteoporos. Rep. 2016, 14, 327–336. [Google Scholar] [CrossRef]
- Sugatani, T.; Vacher, J.; Hruska, K.A. A microRNA expression signature of osteoclastogenesis. Blood 2011, 117, 3648–3657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hackl, M.; Heilmeier, U.; Weilner, S.; Grillari, J. Circulating microRNAs as novel biomarkers for bone diseases—Complex signatures for multifactorial diseases? Mol. Cell. Endocrinol. 2016, 432, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Yang, T.; Han, J.; Yan, K.; Qiu, X.; Zhou, Y.; Fan, Q.; Ma, B. MicroRNA expression during osteogenic differentiation of human multipotent mesenchymal stromal cells from bone marrow. J. Cell. Biochem. 2011, 112, 1844–1856. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.-F.; Yang, G.-H.; Pan, X.-H.; Zhang, S.-J.; Zhao, C.; Qiu, B.-S.; Gu, H.-F.; Hong, J.-F.; Cao, L.; Chen, Y.; et al. Altered microRNA expression profile in exosomes during osteogenic differentiation of human bone marrow-derived mesenchymal stem cells. PLoS ONE 2014, 9, e114627. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.-H.; Sui, B.-D.; Du, F.-Y.; Shuai, Y.; Zheng, C.-X.; Zhao, P.; Yu, X.-R.; Jin, Y. miR-21 deficiency inhibits osteoclast function and prevents bone loss in mice. Sci. Rep. 2017, 7, 43191. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z.; Liu, X.; Wang, H.; Li, J.; Dai, L.; Li, J.; Dong, C. Lung adenocarcinoma cell-derived exosomal miR-21 facilitates osteoclastogenesis. Gene 2018, 666, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, T.; Watanabe, K.; Hara, E.S.; Ono, M.; Kuboki, T.; Calderwood, S.K. OstemiR: A novel panel of microRNA biomarkers in osteoblastic and osteocytic differentiation from mesencymal stem cells. PLoS ONE 2013, 8, e58796. [Google Scholar] [CrossRef]
- Shilo, V.; Mor-Yosef Levi, I.; Abel, R.; Mihailović, A.; Wasserman, G.; Naveh-Many, T.; Ben-Dov, I.Z. Let-7 and MicroRNA-148 Regulate Parathyroid Hormone Levels in Secondary Hyperparathyroidism. J. Am. Soc. Nephrol. 2017, 28, 2353–2363. [Google Scholar] [CrossRef]
- Pizzamiglio, S.; Zanutto, S.; Ciniselli, C.M.; Belfiore, A.; Bottelli, S.; Gariboldi, M.; Verderio, P. A methodological procedure for evaluating the impact of hemolysis on circulating microRNAs. Oncol. Lett. 2017, 13, 315–320. [Google Scholar] [CrossRef]
- Kirschner, M.B.; Edelman, J.J.B.; Kao, S.C.-H.; Vallely, M.P.; van Zandwijk, N.; Reid, G. The Impact of Hemolysis on Cell-Free microRNA Biomarkers. Front. Genet. 2013, 4, 94. [Google Scholar] [CrossRef]
- Murray, M.J.; Watson, H.L.; Ward, D.M.; Bailey, S.; Ferraresso, M.; Nicholson, J.C.; Gnanapragasam, V.J.; Thomas, B.; Scarpini, C.G.; Coleman, N. “Future-proofing” blood processing for measurement of circulating microRNAs in samples from biobanks and prospective clinical trials. Cancer Epidemiol. Biomarkers Prev. 2017, 27, 208–218. [Google Scholar] [CrossRef]
- Murray, M.J.; Bell, E.; Raby, K.L.; Rijlaarsdam, M.A.; Gillis, A.J.M.; Looijenga, L.H.J.; Brown, H.; Destenaves, B.; Nicholson, J.C.; Coleman, N. A pipeline to quantify serum and cerebrospinal fluid microRNAs for diagnosis and detection of relapse in paediatric malignant germ-cell tumours. Br. J. Cancer 2015, 114, 151–162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Zhu, Y.; Jin, F.; Tang, L.; He, Z.; He, Z. Differential expression of circulating microRNAs in blood and haematoma samples from patients with intracerebral haemorrhage. J. Int. Med. Res. 2016, 44, 419–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rice, J.; Roberts, H.; Rai, S.N.; Galandiuk, S. Housekeeping genes for studies of plasma microRNA: A need for more precise standardization. Surgery 2015, 158, 1345–1351. [Google Scholar] [CrossRef]
- Marabita, F.; de Candia, P.; Torri, A.; Tegnér, J.; Abrignani, S.; Rossi, R.L. Normalization of circulating microRNA expression data obtained by quantitative real-time RT-PCR. Brief Bioinform. 2016, 17, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Gevaert, A.B.; Witvrouwen, I.; Vrints, C.J.; Heidbuchel, H.; Van Craenenbroeck, E.M.; Van Laere, S.J.; Van Craenenbroeck, A.H. MicroRNA profiling in plasma samples using qPCR arrays: Recommendations for correct analysis and interpretation. PLoS ONE 2018, 13, e0193173. [Google Scholar] [CrossRef] [PubMed]

| Selected miRNAs | Evidences of Circulating miRNAs Differentially Expressed in T1D Patients | Evidences of miRNAs Involved in Bone Remodeling |
|---|---|---|
| hsa-miR-148a | Nielsen et al. 2012 [27] Seyan et al.2016 [28] Assman et al. 2017 [29] Akerman 2018 [30] | Bedene et al. 2016 [33] Seeliger et al. 2015 [34] |
| hsa-miR-21-5p | Seyan et al.2016 [28] Assman et al. 2017 [29] | Li et al. 2014 [35] Seeliger et al. 2015 [34] Yang et al. 2013 [36] |
| hsa-miR-214 | Erener et al. 2017 [31] | Zhao et al. 2015 [37] |
| hsa-miR-375 | Seyan et al.2016 [28] | Du et al. 2015 [38] |
| Samandari et al.2016 [32] | ||
| Assman et al. 2017 [29] | ||
| hsa-miR-27a | Nielsen et al.2012 [27] | You et al. 2016 [39] |
| Zeng et al. 2017 [40] | ||
| hsa-miR-24 | Seyan et al.2016 [28] | Seeliger et al. 2015 [34] |
| Samandari et al.2016 [32] | ||
| Assman et al. 2017 [29] | ||
| Nielsen et al. 2012 [27] | ||
| Erener et al. 2017 [31] |
| Non-Diabetic Subjects | Type 1 Diabetic Patients | |
|---|---|---|
| Subjects (n) | 14 | 15 |
| Male/Female (n/n) | 10/4 | 10/5 |
| Age (years) | 28 ± 7.93 | 32 ± 7.9 |
| Disease duration (years) | / | 15.71 ± 11.33 |
| HbA1c (%) | N/A | 7.36 ± 0.80593 |
| Chronic complications (n/n) | N/A | 2/15 |
| Smoking habits (n/n) | N/A | 3/15 |
| Other therapies (n/n) | N/A | 6/15 |
| Chronic inflammatory diseases (n/n) | N/A | 4/15 |
| BMI # | 24 ± 4.34 | 21 ± 1.73 # |
| Ca (mg/dL) | 9.37 ± 0.50 | 9.54 ± 0.46 |
| Osteocalcin (ng/mL) | 4.24 ± 1.82 | 4.50 ± 2.21 |
| bALP (μg/L) | 10.5 ± 7.33 | 10.2 ± 4.72 |
| 25OHD (ng/mL) # | 27.9 ± 10.33 | 14.3 ± 9.83 # |
| PTH (pg/mL) # | 18 ± 9.36 | 26 ± 10.23 # |
| BMD TB (g/cm2) | 1.23 ± 0.11 | 1.15 ± 0.09 |
| T score TB | 0.7 ± 1.18 | −0.2 ± 1.15 |
| Z score TB | 0.43 ± 1.10 | 0.08 ± 1.12 |
| BMC TB (g/cm2) | 2.82 ± 0.46 | 2.61 ± 0.38 |
| BMD FEM (g/cm2) # | 1.156 ± 0.17 | 0.967 ± 0.13 # |
| T score FEM # | 0.8 ± 1.18 | −0.8 ± 1.10 # |
| Z score FEM # | 0.6 ± 1.04 | −0.8 ± 1.11 # |
| BMC FEM (g/cm2) * | 37.86 ± 8.13 | 32.04 ± 4.82* |
| BMD N (g/cm2) * | 1.09 ± 0.18 | 0.94 ± 0.12* |
| T score N * | 0.3 ± 1.27 | −0.80 ± 1.08* |
| Z score N # | 0.2 ± 1.13 | −0.8 ± 1.10 # |
| BMC N (g/cm2) | 5.34 ± 1.35 | 4.40 ± 1.04 |
| BMD L (g/cm2) | 1.20 ± 0.15 | 1.14 ± 0.17 |
| T score L | −0.04 ± 1.24 | −0.57 ± 1.48 |
| Z score L | −0.14 ± 1.26 | −0.42 ± 1.59 |
| Target Gene | Gene Name | Function | Targeting miRNA |
|---|---|---|---|
| MAFB | V-mafavianmusculoaponeurotic fibrosarcoma oncogenehomolog B | Negatively regulates RANKL-induced osteoclast differentiation | miR-148a |
| WNT1 | Wingless-type MMTV integration site family, member 1 | Canonical pathwayWnt/β catenin: stimulates osteoblast differentiation and proliferation | miR-148a |
| BCL2L11 | BCL2-like 11 (apoptosis facilitator) | Induces apoptosis | miR-148a |
| PTEN | Phosphatase and tensin homolog | Interacts with Canonical pathwayWnt/β catenin | miR-148a |
| TGFB2 | Transforming growth factor, β 2 | Stimulates osteoblast differentiation and proliferation and bone formation | miR-148a |
| IGF1 | Insulin-like growth factor 1 (somatomedin C) | Stimulates bone mineralization and positively regulates osteoblast differentiation | miR-148a |
| GADD45A | Growth arrest and DNA-damage-inducible, α | Positively regulates apoptosis | miR-148a |
| STAT3 | Signal transducer and activator of transcription 3 | Specifically inactivates osteoblast/osteocytes | miR-21-5p |
| TGFBR1 | Transforming Growth Factor β Receptor 1 | Regulates bone resorption | miR-21-5p |
| IGF1R | Insulin-like growth factor 1 receptor | Involved in bone remodeling | miR-21-5p |
| BCL6 | B-Cell lymphoma 6 | Regulates osteoclast differentiation | miR-21-5p |
| TGFB2 | Transforming growth factor-β 2 | Involved in osteoblast differentiation and bone remodeling | miR-21-5p |
| PDCD4 | Programmed Cell Death 4 | Involved in osteoclastogenesis through cFos regulation | miR-21-5p |
| SOD2 | Superoxide dismutase 2 | Controls osteoclastogenesis and maintains osteoblast differentiation | miR-21-5p |
| FOXO3 | Forkhead box O-3 | Its overexpression increases bone resorption | miR-21-5p |
| USP7 | Ubiquitin-specific protease 7 | Involved in osteogenic differentiation | miR-21-5p |
| PTEN | Phosphatase and tensin homolog | Involved in adiponectin-induced osteogenesis | miR-21-5p |
| IL-10 | Interleukin 10 | Inhibits bone resorption | miR-21-5p |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grieco, G.E.; Cataldo, D.; Ceccarelli, E.; Nigi, L.; Catalano, G.; Brusco, N.; Mancarella, F.; Ventriglia, G.; Fondelli, C.; Guarino, E.; et al. Serum Levels of miR-148a and miR-21-5p Are Increased in Type 1 Diabetic Patients and Correlated with Markers of Bone Strength and Metabolism. Non-Coding RNA 2018, 4, 37. https://doi.org/10.3390/ncrna4040037
Grieco GE, Cataldo D, Ceccarelli E, Nigi L, Catalano G, Brusco N, Mancarella F, Ventriglia G, Fondelli C, Guarino E, et al. Serum Levels of miR-148a and miR-21-5p Are Increased in Type 1 Diabetic Patients and Correlated with Markers of Bone Strength and Metabolism. Non-Coding RNA. 2018; 4(4):37. https://doi.org/10.3390/ncrna4040037
Chicago/Turabian StyleGrieco, Giuseppina E., Dorica Cataldo, Elena Ceccarelli, Laura Nigi, Giovanna Catalano, Noemi Brusco, Francesca Mancarella, Giuliana Ventriglia, Cecilia Fondelli, Elisa Guarino, and et al. 2018. "Serum Levels of miR-148a and miR-21-5p Are Increased in Type 1 Diabetic Patients and Correlated with Markers of Bone Strength and Metabolism" Non-Coding RNA 4, no. 4: 37. https://doi.org/10.3390/ncrna4040037
APA StyleGrieco, G. E., Cataldo, D., Ceccarelli, E., Nigi, L., Catalano, G., Brusco, N., Mancarella, F., Ventriglia, G., Fondelli, C., Guarino, E., Crisci, I., Sebastiani, G., & Dotta, F. (2018). Serum Levels of miR-148a and miR-21-5p Are Increased in Type 1 Diabetic Patients and Correlated with Markers of Bone Strength and Metabolism. Non-Coding RNA, 4(4), 37. https://doi.org/10.3390/ncrna4040037






