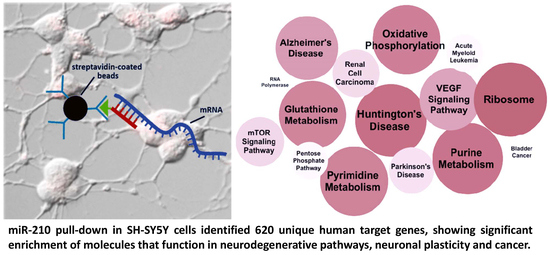
Hypoxia-Induced MicroRNA-210 Targets Neurodegenerative Pathways
Abstract
1. Introduction
2. Results
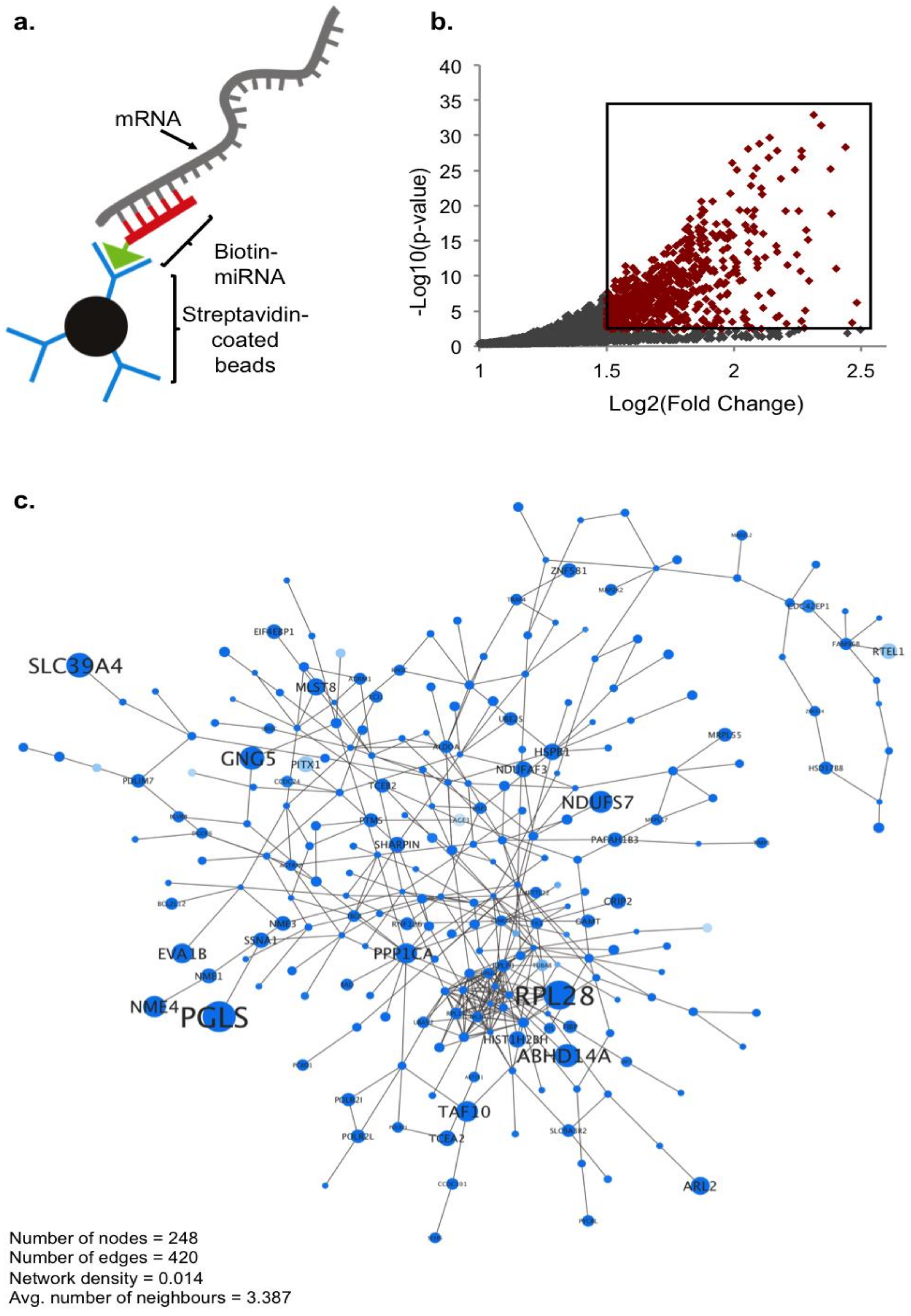
2.1. Biotin Pull-Down of miRNA-210 Targets
2.2. Global Ontology Analysis of miR-210 Targeting
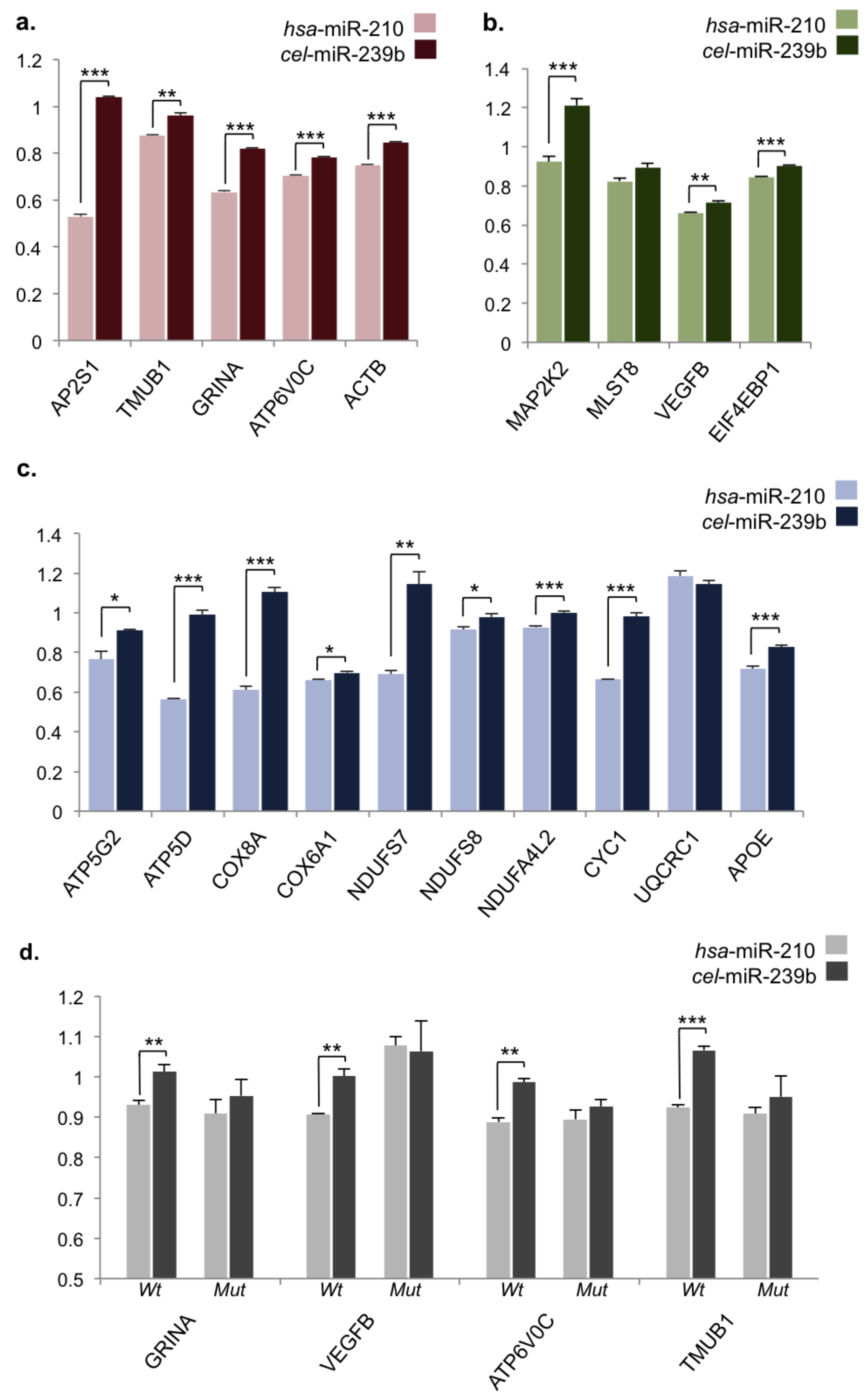
2.3. Validation of miR-210 Target Regulation
3. Discussion
4. Materials and Methods
4.1. Biotin Pull-Down Assay
4.2. RNA Extraction, Complementary DNA Synthesis, and qPCR
4.3. RNA Sequencing
4.4. Ontology Analysis
4.5. Cloning
4.6. Cell Culture and Transfection
4.7. Dual Luciferase Assays
4.8. Statistics
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Steward, O.; Fass, B. Polyribosomes associated with dendritic spines in the denervated dentate gyrus: Evidence for local regulation of protein synthesis during reinnervation. Prog. Brain Res. 1983, 58, 131–136. [Google Scholar] [PubMed]
- Lewis, B.P.; Burge, C.B.; Bartel, D.P. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 2005, 120, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Smalheiser, N.R.; Lugli, G. MicroRNA regulation of synaptic plasticity. Neuromol. Med. 2009, 11, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Sempere, L.F.; Freemantle, S.; Pitha-Rowe, I.; Moss, E.; Dmitrovsky, E.; Ambros, V. Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol. 2004, 5, R13. [Google Scholar] [CrossRef] [PubMed]
- Krichevsky, A.M.; King, K.S.; Donahue, C.P.; Khrapko, K.; Kosik, K.S. A microRNA array reveals extensive regulation of microRNAs during brain development. RNA 2003, 9, 1274–1281. [Google Scholar] [CrossRef] [PubMed]
- Cristino, A.S.; Barchuk, A.R.; Freitas, F.C.; Narayanan, R.K.; Biergans, S.D.; Zhao, Z.; Simoes, Z.L.; Reinhard, J.; Claudianos, C. Neuroligin-associated microRNA-932 targets actin and regulates memory in the honeybee. Nat. Commun. 2014, 5, 5529. [Google Scholar] [CrossRef] [PubMed]
- Saini, H.K.; Griffiths-Jones, S.; Enright, A.J. Genomic analysis of human microRNA transcripts. Proc. Natl. Acad. Sci. USA 2007, 104, 17719–17724. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Li, Y.; Zhang, H.; Huang, P.; Luthra, R. Hypoxia-regulated microRNA-210 modulates mitochondrial function and decreases ISCU and COX10 expression. Oncogene 2010, 29, 4362–4368. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Le, Q.T.; Giaccia, A.J. MiR-210—Micromanager of the hypoxia pathway. Trends Mol. Med. 2010, 16, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; He, X.; Wang, Y.; Tang, Y.; Zheng, C.; Cai, H.; Liu, J.; Wang, Y.; Fu, Y.; Yang, G.Y. MicroRNA-210 overexpression induces angiogenesis and neurogenesis in the normal adult mouse brain. Gene Ther. 2014, 21, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, A.I.; Zhang, H.; Nie, Y.; Tang, W.; Sun, T. Cdk7 and miR-210 co-regulate cell-cycle progression of neural progenitors in the developing neocortex. Stem Cell Rep. 2016, 7, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Kretschmann, A.; Danis, B.; Andonovic, L.; Abnaof, K.; van Rikxoort, M.; Siegel, F.; Mazzuferi, M.; Godard, P.; Hanon, E.; Frohlich, H.; et al. Different microRNA profiles in chronic epilepsy versus acute seizure mouse models. J. Mol. Neurosci. 2015, 55, 466–479. [Google Scholar] [CrossRef] [PubMed]
- Schouten, M.; Fratantoni, S.A.; Hubens, C.J.; Piersma, S.R.; Pham, T.V.; Bielefeld, P.; Voskuyl, R.A.; Lucassen, P.J.; Jimenez, C.R.; Fitzsimons, C.P. MicroRNA-124 and -137 cooperativity controls caspase-3 activity through BCL2L13 in hippocampal neural stem cells. Sci. Rep. 2015, 5, 12448. [Google Scholar] [CrossRef] [PubMed]
- Gorter, J.A.; Iyer, A.; White, I.; Colzi, A.; van Vliet, E.A.; Sisodiya, S.; Aronica, E. Hippocampal subregion-specific microRNA expression during epileptogenesis in experimental temporal lobe epilepsy. Neurobiol. Dis. 2014, 62, 508–520. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zheng, H.; Zhang, S. Involvement of upregulation of miR-210 in a rat epilepsy model. Neuropsychiatr. Dis. Treat. 2016, 12, 1731–1737. [Google Scholar] [PubMed]
- Hu, K.; Xie, Y.Y.; Zhang, C.; Ouyang, D.S.; Long, H.Y.; Sun, D.N.; Long, L.L.; Feng, L.; Li, Y.; Xiao, B. MicroRNA expression profile of the hippocampus in a rat model of temporal lobe epilepsy and miR-34a-targeted neuroprotection against hippocampal neurone cell apoptosis post-status epilepticus. BMC Neurosci. 2012, 13, 115. [Google Scholar] [CrossRef] [PubMed]
- Li, J.J.; Dolios, G.; Wang, R.; Liao, F.F. Soluble β-amyloid peptides, but not insoluble fibrils, have specific effect on neuronal microRNA expression. PLoS ONE 2014, 9, e90770. [Google Scholar] [CrossRef] [PubMed]
- Helwak, A.; Kudla, G.; Dudnakova, T.; Tollervey, D. Mapping the human miRNA interactome by CLASH reveals frequent noncanonical binding. Cell 2013, 153, 654–665. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Sung, Y.M.; Park, J.; Kim, S.; Kim, J.; Park, J.; Ha, H.; Bae, J.Y.; Kim, S.; Baek, D. General rules for functional microRNA targeting. Nat. Genet. 2016, 48, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Goldie, B.J.; Barnett, M.M.; Cairns, M.J. BDNF and the maturation of posttranscriptional regulatory networks in human SH-SY5Y neuroblast differentiation. Front. Cell. Neurosci. 2014, 8, 325. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Chen, S.; Liu, A.; Wan, J.; Tang, L.; Zheng, N.; Xiong, Y. Inhibition of BNDF production by MPP+ through up-regulation of miR-210-3p contributes to dopaminergic neuron damage in MPTP model. Neurosci. Lett. 2017. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.M.; Kirchner, R.; Jin, J.; Hofmann, O.; McReynolds, L.; Hide, W.; Lieberman, J. Sequencing of captive target transcripts identifies the network of regulated genes and functions of primate-specific miR-522. Cell Rep. 2014, 8, 1225–1239. [Google Scholar] [CrossRef] [PubMed]
- Orom, U.A.; Nielsen, F.C.; Lund, A.H. MicroRNA-10a binds the 5′UTR of ribosomal protein mRNAs and enhances their translation. Mol. Cell 2008, 30, 460–471. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.N.; Tilakaratne, N.; Johnson, P.S.; Allen, A.E.; Michaelis, E.K. Cloning of cDNA for the glutamate-binding subunit of an NMDA receptor complex. Nature 1991, 354, 70–73. [Google Scholar] [CrossRef] [PubMed]
- Gelinas, J.N.; Banko, J.L.; Hou, L.; Sonenberg, N.; Weeber, E.J.; Klann, E.; Nguyen, P.V. ERK and mTOR signaling couple β-adrenergic receptors to translation initiation machinery to gate induction of protein synthesis-dependent long-term potentiation. J. Biol. Chem. 2007, 282, 27527–27535. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Takagi, H.; Konishi, Y.; Ageta, H.; Ikegami, K.; Yao, I.; Sato, S.; Hatanaka, K.; Inokuchi, K.; Seog, D.H.; et al. Transmembrane and ubiquitin-like domain-containing protein 1 (Tmub1/HOPS) facilitates surface expression of GluR2-containing AMPA receptors. PLoS ONE 2008, 3, e2809. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Liu, L.; Wang, Y.T.; Sheng, M. Clathrin adaptor AP2 and NSF interact with overlapping sites of GluR2 and play distinct roles in AMPA receptor trafficking and hippocampal LTD. Neuron 2002, 36, 661–674. [Google Scholar] [CrossRef]
- Levenson, J.M.; Choi, S.; Lee, S.Y.; Cao, Y.A.; Ahn, H.J.; Worley, K.C.; Pizzi, M.; Liou, H.C.; Sweatt, J.D. A bioinformatics analysis of memory consolidation reveals involvement of the transcription factor c-Rel. J. Neurosci. Off. J. Soc. Neurosci. 2004, 24, 3933–3943. [Google Scholar] [CrossRef] [PubMed]
- Donahue, C.P.; Jensen, R.V.; Ochiishi, T.; Eisenstein, I.; Zhao, M.; Shors, T.; Kosik, K.S. Transcriptional profiling reveals regulated genes in the hippocampus during memory formation. Hippocampus 2002, 12, 821–833. [Google Scholar] [CrossRef] [PubMed]
- Robles, Y.; Vivas-Mejia, P.E.; Ortiz-Zuazaga, H.G.; Felix, J.; Ramos, X.; Pena de Ortiz, S. Hippocampal gene expression profiling in spatial discrimination learning. Neurobiol. Learn. Mem. 2003, 80, 80–95. [Google Scholar] [CrossRef]
- Katz, I.K.; Lamprecht, R. Fear conditioning leads to alteration in specific genes expression in cortical and thalamic neurons that project to the lateral amygdala. J. Neurochem. 2015, 132, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Sirri, A.; Bianchi, V.; Pelizzola, M.; Mayhaus, M.; Ricciardi-Castagnoli, P.; Toniolo, D.; D’Adamo, P. Temporal gene expression profile of the hippocampus following trace fear conditioning. Brain Res. 2010, 1308, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Ploski, J.E.; Park, K.W.; Ping, J.; Monsey, M.S.; Schafe, G.E. Identification of plasticity-associated genes regulated by Pavlovian fear conditioning in the lateral amygdala. J. Neurochem. 2010, 112, 636–650. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.M.; Mason-Parker, S.E.; Tate, W.P.; Abraham, W.C.; Williams, J.M. Rapidly induced gene networks following induction of long-term potentiation at perforant path synapses in vivo. Hippocampus 2011, 21, 541–553. [Google Scholar] [CrossRef] [PubMed]
- McNair, K.; Broad, J.; Riedel, G.; Davies, C.H.; Cobb, S.R. Global changes in the hippocampal proteome following exposure to an enriched environment. Neuroscience 2007, 145, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Park, C.S.; Gong, R.; Stuart, J.; Tang, S.J. Molecular network and chromosomal clustering of genes involved in synaptic plasticity in the hippocampus. J. Biol. Chem. 2006, 281, 30195–30211. [Google Scholar] [CrossRef] [PubMed]
- Haberman, R.P.; Lee, H.J.; Colantuoni, C.; Koh, M.T.; Gallagher, M. Rapid encoding of new information alters the profile of plasticity-related mRNA transcripts in the hippocampalCA3 region. Proc. Natl. Acad. Sci. USA 2008, 105, 10601–10606. [Google Scholar] [CrossRef] [PubMed]
- Maag, J.L.; Panja, D.; Sporild, I.; Patil, S.; Kaczorowski, D.C.; Bramham, C.R.; Dinger, M.E.; Wibrand, K. Dynamic expression of long noncoding RNAs and repeat elements in synaptic plasticity. Front. Neurosci. 2015, 9, 351. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Lou, Y.L.; Wu, J.; Ruan, Q.F.; Xie, A.; Guo, F.; Cui, S.P.; Deng, Z.F.; Wang, Y. Upregulation of microRNA-210 regulates renal angiogenesis mediated by activation of VEGF signaling pathway under ischemia/perfusion injury in vivo and in vitro. Kidney Blood Press. Res. 2012, 35, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Zhao, A.; Li, G.; Peoc’h, M.; Genin, C.; Gigante, M. Serum miR-210 as a novel biomarker for molecular diagnosis of clear cell renal cell carcinoma. Exp. Mol. Pathol. 2013, 94, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Han, Y.; Zhang, H.; Nie, L.; Jiang, Z.; Fa, P.; Gui, Y.; Cai, Z. Synthetic miRNA-mowers targeting miR-183-96-182 cluster or miR-210 inhibit growth and migration and induce apoptosis in bladder cancer cells. PLoS ONE 2012, 7, e52280. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Alafuzoff, I.; Soininen, H.; Winblad, B.; Pei, J.J. Levels of mTOR and its downstream targets 4E-BP1, eEF2, and eEF2 kinase in relationships with tau in Alzheimer’s disease brain. FEBS J. 2005, 272, 4211–4220. [Google Scholar] [CrossRef] [PubMed]
- Hebert, S.S.; Horre, K.; Nicolai, L.; Papadopoulou, A.S.; Mandemakers, W.; Silahtaroglu, A.N.; Kauppinen, S.; Delacourte, A.; De Strooper, B. Loss of microRNA cluster miR-29a/b-1 in sporadic Alzheimer’s disease correlates with increased BACE1/β-secretase expression. Proc. Natl. Acad. Sci. USA 2008, 105, 6415–6420. [Google Scholar] [CrossRef] [PubMed]
- Cogswell, J.P.; Ward, J.; Taylor, I.A.; Waters, M.; Shi, Y.; Cannon, B.; Kelnar, K.; Kemppainen, J.; Brown, D.; Chen, C.; et al. Identification of miRNA changes in Alzheimer’s disease brain and CSF yields putative biomarkers and insights into disease pathways. J. Alzheimer’s Dis. 2008, 14, 27–41. [Google Scholar] [CrossRef]
- Zhu, Y.; Li, C.; Sun, A.; Wang, Y.; Zhou, S. Quantification of microRNA-210 in the cerebrospinal fluid and serum: Implications for Alzheimer’s disease. Exp. Ther. Med. 2015, 9, 1013–1017. [Google Scholar] [CrossRef] [PubMed]
- Kwon, A.T.; Arenillas, D.J.; Worsley Hunt, R.; Wasserman, W.W. oPOSSUM-3: Advanced analysis of regulatory motif over-representation across genes or ChIP-Seq datasets. G3 Genes Genomes Genet. 2012, 2, 987–1002. [Google Scholar] [CrossRef] [PubMed]
- Berezikov, E.; Thuemmler, F.; van Laake, L.W.; Kondova, I.; Bontrop, R.; Cuppen, E.; Plasterk, R.H. Diversity of microRNAs in human and chimpanzee brain. Nat. Genet. 2006, 38, 1375–1377. [Google Scholar] [CrossRef] [PubMed]
- El Far, O.; Seagar, M. A role for v-ATPase subunits in synaptic vesicle fusion? J. Neurochem. 2011, 117, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Corder, E.H.; Saunders, A.M.; Strittmatter, W.J.; Schmechel, D.E.; Gaskell, P.C.; Small, G.W.; Roses, A.D.; Haines, J.L.; Pericak-Vance, M.A. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 1993, 261, 921–923. [Google Scholar] [CrossRef] [PubMed]
- Erecinska, M.; Silver, I.A. Tissue oxygen tension and brain sensitivity to hypoxia. Respir. Physiol. 2001, 128, 263–276. [Google Scholar] [CrossRef]
- Behura, S.K.; Whitfield, C.W. Correlated expression patterns of microRNA genes with age-dependent behavioral changes in honeybee. Insect Mol. Biol. 2010, 19, 431–439. [Google Scholar] [PubMed]
- Rampon, C.; Jiang, C.H.; Dong, H.; Tang, Y.P.; Lockhart, D.J.; Schultz, P.G.; Tsien, J.Z.; Hu, Y. Effects of environmental enrichment on gene expression in the brain. Proc. Natl. Acad. Sci. USA 2000, 97, 12880–12884. [Google Scholar] [CrossRef] [PubMed]
- Hunsberger, J.G.; Bennett, A.H.; Selvanayagam, E.; Duman, R.S.; Newton, S.S. Gene profiling the response to kainic acid induced seizures. Mol. Brain Res. 2005, 141, 95–112. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Jiao, X.; Zuzga, D.S.; Liu, Y.; Fong, D.M.; Young, D.; During, M.J. VEGF links hippocampal activity with neurogenesis, learning and memory. Nat. Genet. 2004, 36, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Attwell, D.; Laughlin, S.B. An energy budget for signaling in the grey matter of the brain. J. Cereb. Blood Flow Metab. 2001, 21, 1133–1145. [Google Scholar] [CrossRef] [PubMed]
- Roy, C.S.; Sherrington, C.S. On the regulation of the blood-supply of the brain. J. Physiol. 1890, 11, 85–158. [Google Scholar] [CrossRef] [PubMed]
- Mangia, S.; Tkac, I.; Gruetter, R.; Van de Moortele, P.F.; Maraviglia, B.; Ugurbil, K. Sustained neuronal activation raises oxidative metabolism to a new steady-state level: Evidence from 1H NMR spectroscopy in the human visual cortex. J. Cereb. Blood Flow Metab. 2007, 27, 1055–1063. [Google Scholar] [CrossRef]
- Schwarz, T.L. Mitochondrial trafficking in neurons. Cold Spring Harb. Perspect. Biol. 2013, 5. [Google Scholar] [CrossRef] [PubMed]
- Kraggerud, S.M.; Sandvik, J.A.; Pettersen, E.O. Regulation of protein synthesis in human cells exposed to extreme hypoxia. Anticancer Res. 1995, 15, 683–686. [Google Scholar] [PubMed]
- Bonaglia, M.C.; Giorda, R.; Tenconi, R.; Pessina, M.; Pramparo, T.; Borgatti, R.; Zuffardi, O. A 2.3 Mb duplication of chromosome 8q24.3 associated with severe mental retardation and epilepsy detected by standard karyotype. Eur. J. Hum. Genet. 2005, 13, 586–591. [Google Scholar] [CrossRef] [PubMed]
- Yashiro, K.; Philpot, B.D. Regulation of NMDA receptor subunit expression and its implications for LTD, LTP, and metaplasticity. Neuropharmacology 2008, 55, 1081–1094. [Google Scholar] [CrossRef] [PubMed]
- Kittler, J.T.; Delmas, P.; Jovanovic, J.N.; Brown, D.A.; Smart, T.G.; Moss, S.J. Constitutive endocytosis of GABAA receptors by an association with the adaptin AP2 complex modulates inhibitory synaptic currents in hippocampal neurons. J. Neurosci. 2000, 20, 7972–7977. [Google Scholar] [PubMed]
- Park, C.S.; Tang, S.J. Regulation of microRNA expression by induction of bidirectional synaptic plasticity. J. Mol. Neurosci. 2009, 38, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.; Kaech, S.; Knutti, D.; Matus, A. Rapid actin-based plasticity in dendritic spines. Neuron 1998, 20, 847–854. [Google Scholar] [CrossRef]
- Chen, Y.; Durakoglugil, M.S.; Xian, X.; Herz, J. ApoE4 reduces glutamate receptor function and synaptic plasticity by selectively impairing apoE receptor recycling. Proc. Natl. Acad. Sci. USA 2010, 107, 12011–12016. [Google Scholar] [CrossRef] [PubMed]
- Baldeiras, I.; Santana, I.; Proenca, M.T.; Garrucho, M.H.; Pascoal, R.; Rodrigues, A.; Duro, D.; Oliveira, C.R. Peripheral oxidative damage in mild cognitive impairment and mild Alzheimer’s disease. J. Alzheimer’s Dis. 2008, 15, 117–128. [Google Scholar] [CrossRef]
- Du, H.; Guo, L.; Yan, S.; Sosunov, A.A.; McKhann, G.M.; Yan, S.S. Early deficits in synaptic mitochondria in an Alzheimer’s disease mouse model. Proc. Natl. Acad. Sci. USA 2010, 107, 18670–18675. [Google Scholar] [CrossRef] [PubMed]
- Park, C.Y.; Jeker, L.T.; Carver-Moore, K.; Oh, A.; Liu, H.J.; Cameron, R.; Richards, H.; Li, Z.; Adler, D.; Yoshinaga, Y.; et al. A resource for the conditional ablation of microRNAs in the mouse. Cell Rep. 2012, 1, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Prosser, H.M.; Koike-Yusa, H.; Cooper, J.D.; Law, F.C.; Bradley, A. A resource of vectors and ES cells for targeted deletion of microRNAs in mice. Nat. Biotechnol. 2011, 29, 840–845. [Google Scholar] [CrossRef] [PubMed]
- White, K.; Lu, Y.; Annis, S.; Hale, A.E.; Chau, B.N.; Dahlman, J.E.; Hemann, C.; Opotowsky, A.R.; Vargas, S.O.; Rosas, I.; et al. Genetic and hypoxic alterations of the microRNA-210-ISCU1/2 axis promote iron-sulfur deficiency and pulmonary hypertension. EMBO Mol. Med. 2015, 7, 695–713. [Google Scholar] [CrossRef] [PubMed]
- Mok, Y.; Schwierzeck, V.; Thomas, D.C.; Vigorito, E.; Rayner, T.F.; Jarvis, L.B.; Prosser, H.M.; Bradley, A.; Withers, D.R.; Martensson, I.L.; et al. MiR-210 is induced by Oct-2, regulates B cells, and inhibits autoantibody production. J. Immunol. 2013, 191, 3037–3048. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Pertea, G.; Trapnell, C.; Pimentel, H.; Kelley, R.; Salzberg, S.L. TopHat2: Accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 2013, 14, R36. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R.; 1000 Genome Project Data Processing Subgroup. The sequence alignment/map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef] [PubMed]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq—A Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-Seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed]
| Gene Name | Gene ID | padj | FC |
|---|---|---|---|
| Oligosaccharyltransferase complex subunit 4 (non-catalytic) ▲ | OST4 | 1.96 × 10−91 | 7.31 |
| Ribosomal protein L28 ▲ | RPL28 | 1.08 × 10−54 | 6.02 |
| 6-Phosphogluconolactonase ▲ | PGLS | 1.74 × 10−42 | 6.27 |
| NME/NM23 Nucleoside diphosphate kinase 4 | NME4 | 3.36 × 10−30 | 4.97 |
| NADH: Ubiquinone oxidoreductase core subunit S7 ▲ | NDUFS7 | 9.47 × 10−29 | 5.07 |
| mTOR Associated protein, LST8 homolog ▲ | MLST8 | 3.47 × 10−27 | 4.41 |
| Pancreatic progenitor cell differentiation and proliferation factor | PPDPF | 2.53 × 10−26 | 4.29 |
| Solute carrier family 39 (Zinc transporter), member 4 | SLC39A4 | 7.50 × 10−26 | 5.42 |
| Glutamate receptor, ionotropic, N-methyl d-aspartate-associated protein 1 (Glutamate binding) ▲ | GRINA | 1.02 × 10−25 | 4.16 |
| Adaptor related protein complex 2 Sigma 1 subunit | AP2S1 | 1.46 × 10−25 | 4.50 |
| Phosphatidylethanolamine N-methyltransferase | PEMT | 1.46 × 10−25 | 4.80 |
| Protein phosphatase 1, catalytic subunit, alpha isozyme | PPP1CA | 9.00 × 10−25 | 4.81 |
| Vascular endothelial growth factor B | VEGFB | 1.25 × 10−24 | 4.40 |
| SH3 Domain binding glutamate-rich protein like 3 ▲ | SH3BGRL3 | 6.90 × 10−24 | 3.98 |
| Transmembrane and ubiquitin-like domain containing 1 | TMUB1 | 3.78 × 10−23 | 4.25 |
| Guanine nucleotide binding protein (G protein), gamma 5 | GNG5 | 4.70 × 10−23 | 5.21 |
| CD320 Molecule | CD320 | 4.72 × 10−23 | 4.72 |
| Eukaryotic translation initiation factor 4E binding protein 1 ▲ | EIF4EBP1 | 5.55 × 10−23 | 4.04 |
| Ganglioside induced differentiation associated protein 1-like 1 | GDAP1L1 | 3.49 × 10−22 | 4.21 |
| Lymphocyte antigen 6 complex, locus E | LY6E | 9.13 × 10−22 | 4.54 |
| Biological Process | GO Number | Count | p-Value |
| Translational elongation | GO:0006414 | 16 | 1.03 × 10−6 |
| Translation | GO:0006412 | 26 | 1.03 × 10−4 |
| Macromolecular complex assembly | GO:0065003 | 38 | 1.31 × 10−3 |
| RNA elongation | GO:0006354 | 8 | 1.33 × 10−3 |
| Protein complex assembly | GO:0006461 | 31 | 1.37 × 10−3 |
| Protein complex biogenesis | GO:0070271 | 31 | 1.37 × 10−3 |
| Generation of precursor metabolites and energy | GO:0006091 | 22 | 1.68 × 10−3 |
| Phospholipid biosynthetic process | GO:0008654 | 11 | 1.95 × 10−3 |
| Mitochondrion organization | GO:0007005 | 13 | 2.03 × 10−3 |
| Macromolecular complex subunit organization | GO:0043933 | 39 | 2.23 × 10−3 |
| KEGG Pathway | KEGG Number | Count | p-Value |
| Ribosome | hsa03010 | 15 | 3.14 × 10−6 |
| Huntington’s disease | hsa05016 | 16 | 2.76 × 10−3 |
| Purine metabolism | hsa00230 | 14 | 4.44 × 10−3 |
| Pyrimidine metabolism | hsa00240 | 10 | 8.61 × 10−3 |
| Oxidative phosphorylation | hsa00190 | 12 | 8.90 × 10−3 |
| Glutathione metabolism | hsa00480 | 7 | 1.02 × 10−2 |
| Alzheimer’s disease | hsa05010 | 13 | 1.82 × 10−2 |
| VEGF Signaling pathway | hsa04370 | 8 | 2.12 × 10−2 |
| mTOR Signaling pathway | hsa04150 | 6 | 4.35 × 10−2 |
| Renal cell carcinoma | hsa05211 | 7 | 4.56 × 10−2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watts, M.E.; Williams, S.M.; Nithianantharajah, J.; Claudianos, C. Hypoxia-Induced MicroRNA-210 Targets Neurodegenerative Pathways. Non-Coding RNA 2018, 4, 10. https://doi.org/10.3390/ncrna4020010
Watts ME, Williams SM, Nithianantharajah J, Claudianos C. Hypoxia-Induced MicroRNA-210 Targets Neurodegenerative Pathways. Non-Coding RNA. 2018; 4(2):10. https://doi.org/10.3390/ncrna4020010
Chicago/Turabian StyleWatts, Michelle E., Sarah M. Williams, Jess Nithianantharajah, and Charles Claudianos. 2018. "Hypoxia-Induced MicroRNA-210 Targets Neurodegenerative Pathways" Non-Coding RNA 4, no. 2: 10. https://doi.org/10.3390/ncrna4020010
APA StyleWatts, M. E., Williams, S. M., Nithianantharajah, J., & Claudianos, C. (2018). Hypoxia-Induced MicroRNA-210 Targets Neurodegenerative Pathways. Non-Coding RNA, 4(2), 10. https://doi.org/10.3390/ncrna4020010