On the Behaviour of Living Cells under the Influence of Ultrasound
Abstract
1. Introduction
1.1. Cells
1.2. Ultrasound
1.3. Mechanical Cell Response to Ultrasound
2. Overview of Scientific Publications on Cellular Ultrasonics
2.1. Early Studies
2.2. Damage
2.3. Translation
2.4. Proliferation
2.5. Internal Changes
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CHO | Chinese hamster ovary; |
| COS-7 | CV-1 in origin simian line 7 |
| DNA | deoxyribonucleic acid |
| DOX | doxorubicin |
| HASM | human airway smooth muscle |
| HEK | human embryonic kidney |
| HeLa | Henrietta Lacks |
| HIFU | high-intensity focussed ultrasound |
| HL | human leukaemia |
| MCF-7 | Michigan Cancer Foundation cell line 7 |
| MCLW | metal-clad leaky waveguide |
| MDCK | Madin–Darby canine kidney |
| MET | mesenchymal-epithelial |
| MI | mechanical index |
| MRC-5 | Medical Research Council cell strain 5 |
| PCR | polymerase chain reaction |
| pDNA | plasmid DNA |
| PNP | peak-negative pressure |
| PPP | peak-positive pressure |
| PRF | pulse-repetition frequency |
| PRT | pulse-repetition time |
| P2P | peak-to-peak pressure |
References
- ter Haar, G.R. HIFU tissue ablation: Concept and devices. In Therapeutic Ultrasound; Escoffre, J.M., Bouakaz, A., Eds.; Springer: Berlin, Germany, 2016; Volume 880, pp. 3–20. [Google Scholar]
- Eisenmenger, W. The mechanisms of stone fragmentation in ESWL. Ultrasound Med. Biol. 2001, 27, 683–693. [Google Scholar] [CrossRef]
- Dimcevski, G.; Kotopoulis, S.; Bjånes, T.; Hoem, D.; Schjøtt, J.; Gjertsen, B.T.; Biermann, M.; Molven, A.; Sorbye, H.; McCormack, E.; et al. A human clinical trial using ultrasound and microbubbles to enhance gemcitabine treatment of inoperable pancreatic cancer. J. Control. Release 2016, 243, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, Y.; Maruyama, T.; Takegami, K.; Watanabe, T.; Mitsui, H.; Hanajiri, K.; Nagawa, H.; Matsumoto, Y. Use of a microbubble agent to increase the effects of high intensity focused ultrasound on liver tissue. Eur. Radiol. 2005, 15, 1415–1420. [Google Scholar] [CrossRef] [PubMed]
- van Wamel, A.; Sontum, P.C.; Healey, A.J.; Kvåle, S.; Bush, N.; Bamber, J.; de Lange Davies, C. Acoustic cluster therapy (ACT) enhances the therapeutic efficacy of paclitaxel and Abraxane for treatment of human prostate adenocarcinoma in mice. J. Control. Release 2016, 236, 15–21. [Google Scholar] [CrossRef] [PubMed]
- van der Windt, D.A.; van der Heijden, G.J.; van den Berg, S.G.; ter Riet, G.; de Winter, A.F.; Bouter, L.M. Ultrasound therapy for musculoskeletal disorders: A systematic review. Pain 1999, 81, 257–271. [Google Scholar] [CrossRef]
- Desmeules, F.; Boudreault, J.; Roy, J.S.; Dionne, C.; Frémont, P.; MacDermid, J.C. The efficacy of therapeutic ultrasound for rotator cuff tendinopathy: A systematic review and meta-analysis. Phys. Ther. Sport 2015, 16, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Harle, J.; Salih, V.; Mayia, F.; Knowles, J.C.; Olsen, I. Effects of ultrasound on the growth and function of bone and periodontal ligament cells in vitro. Ultrasound Med. Biol. 2001, 27, 579–586. [Google Scholar] [CrossRef]
- Lim, K.; Kim, J.; Seonwoo, H.; Park, S.H.; Choung, P.H.; Chung, J.H. In vitro effects of low-intensity pulsed ultrasound stimulation on the osteogenic differentiation of human alveolar bone-derived mesenchymal stem cells for tooth tissue engineering. BioMed Res. Int. 2013, 2013, 269724. [Google Scholar] [CrossRef] [PubMed]
- Nakao, J.; Fujii, Y.; Kusuyama, J.; Bandow, K.; Kakimoto, K.; Ohnishi, T.; Matsuguchi, T. Low-intensity pulsed ultrasound (LIPUS) inhibits LPS-induced inflammatory responses of osteoblasts through TLR4–MyD88 dissociation. Bone 2014, 58, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Schumann, D.; Kujat, R.; Zellner, J.; Angele, M.K.; Nerlich, M.; Mayr, E.; Angele, P. Treatment of human mesenchymal stem cells with pulsed low intensity ultrasound enhances the chondrogenic phenotype in vitro. Biorheology 2006, 43, 431–443. [Google Scholar] [PubMed]
- Dalla-Bona, D.A.; Tanaka, E.; Inubushi, T.; Oka, H.; Ohta, A.; Okada, H.; Miyauchi, M.; Takata, T.; Tanne, K. Cementoblast response to low- and high-intensity ultrasound. Arch. Oral Biol. 2008, 53, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, R.; Ryo, A.; Komitsu, N.; Mikuni-Takagaki, Y.; Fukui, A.; Takagi, Y.; Shiraishi, T.; Morishita, S.; Yamazaki, Y.; Kumagai, K.; et al. Low-intensity pulsed ultrasound activates the phosphatidylinositol 3 kinase/Akt pathway and stimulates the growth of chondrocytes in three-dimensional cultures: A basic science study. Arthrit. Res. Ther. 2008, 10, R77. [Google Scholar] [CrossRef] [PubMed]
- Bazou, D.; Maimon, N.; Munn, L.L.; Gonzalez, I. Effects of low intensity continuous ultrasound (LICU) on mouse pancreatic tumor explants. Appl. Sci. 2017, 7, 1275. [Google Scholar] [CrossRef]
- Wiklund, M. Acoustofluidics 12: Biocompatibility and cell viability in microfluidic acoustic resonators. Lab Chip 2012, 12, 2018–2028. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, W.D., Jr. Ultrasound–biophysics mechanisms. Prog. Biophys. Mol. Biol. 2007, 93, 212–255. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Nyborg, W.L. Ultrasound, cavitation bubbles and their interaction with cells. Adv. Drug Deliv. Rev. 2008, 60, 1103–1116. [Google Scholar] [CrossRef] [PubMed]
- Shankar, H.; Pagel, P.S. Potential adverse ultrasound-related biological effects: a critical review. Anesthesiology 2011, 115, 1109–1124. [Google Scholar] [CrossRef] [PubMed]
- Izadifar, Z.; Babyn, P.; Chapman, D. Mechanical and biological effects of ultrasound: A review of present knowledge. Ultrasound Med. Biol. 2017, 43, 1085–1104. [Google Scholar] [CrossRef] [PubMed]
- Tortora, G.J.; Derrickson, B.H. Principles of Anatomy and Physiology, 13th ed.; Wiley: Philadelphia, PA, USA, 2008. [Google Scholar]
- Yang, T.; Bragheri, F.; Nava, G.; Chiodi, I.; Mondello, C.; Osellame, R.; Berg-Sørensen, K.; Cristiani, I.; Minzioni, P. A comprehensive strategy for the analysis of acoustic compressibility and optical deformability on single cells. Sci. Rep. 2016, 6, 23946. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, M.J.; Tomlinson, S.; Yang, X.B.; Kirkham, J. Cell separation: Terminology and practical considerations. J. Tissue Eng. 2013, 4, 2041–7314. [Google Scholar] [CrossRef] [PubMed]
- Postema, M. Fundamentals of Medical Ultrasonics; Spon: London, UK, 2011; ISBN 978-1135-179-36-6. [Google Scholar]
- Nowicki, A. Ultradźwięki w Medycynie: Wprowadzenie do Współczesnej Ultrasonografii; Instytut Podstawowych Problemów Techniki, Polska Akademia Nauk: Warszawa, Poland, 2010. [Google Scholar]
- Postema, M.; Kotopoulis, S.; Jenderka, K.V. Basic physical principles of medical ultrasound. In EFSUMB Course Book on Ultrasound; Dietrich, C.F., Ed.; EFSUMB: London, UK, 2012; pp. 9–37. [Google Scholar]
- Geffen, C.; Kimmel, E. The effect of low intensity ultrasound on adhesion molecules, actin monomers and membrane permeability in endothelial cells. In Micro-Acoustics in Marine and Medical Research; Kotopoulis, S., Delalande, A., Godø, O.R., Postema, M., Eds.; Institutt for fysikk og teknologi, Universitetet i Bergen: Bergen, Norway, 2012; pp. 131–170. [Google Scholar]
- Dehoux, T.; Abi Ghanem, M.; Zouani, O.F.; Rampnoux, J.M.; Guillet, Y.; Dilhaire, S.; Durrieu, M.C.; Audoin, B. All-optical broadband ultrasonography of single cells. Sci. Rep. 2015, 5, 8650. [Google Scholar] [CrossRef] [PubMed]
- Blase, C.; Bereiter-Hahn, J. Ultrasonic characterization of biological cells. In Ultrasonic and Electromagnetic NDE for Structure and Material Characterization: Engineering and Biomedical Applications; Kundu, T., Ed.; CRC: Boca Raton, FL, USA, 2012; pp. 689–722. [Google Scholar]
- Silberrad, O. Propeller erosion. Engineering 1912, 33, 33–35. [Google Scholar]
- Brennen, C.E. Cavitation and Bubble Dynamics; Oxford: New York, NY, USA, 1995. [Google Scholar]
- Young, F.R. Cavitation; McGraw-Hill: New York, NY, USA, 1989. [Google Scholar]
- Kondo, T.; Fukushima, Y.; Kon, H.; Riesz, P. Effect of shear stress and free radicals induced by ultrasound on erythrocytes. Arch. Biochem. Biophys. 1989, 269, 381–389. [Google Scholar] [CrossRef]
- Riesz, P.; Kondo, T. Free radical formation induced by ultrasound and its biological implications. Free Radic. Biol. Med. 1992, 13, 247–270. [Google Scholar] [CrossRef]
- Carstensen, E.L.; Kelly, P.; Church, C.C.; Brayman, A.A.; Child, S.Z.; Raeman, C.H.; Schery, L. Lysis of erythrocytes by exposure to CW ultrasound. Ultrasound Med. Biol. 1993, 19, 147–165. [Google Scholar] [CrossRef]
- Dalecki, D. Mechanical bioeffects of ultrasound. Cancer Res. 2004, 6, 229–248. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, I.; Sostaric, J.Z.; Riesz, P. Sonodynamic therapy—A review of the synergistic effects of drugs and ultrasound. Ultrason. Sonochem. 2004, 11, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Harvey, E.N.; Loomis, A.L. High frequency sound waves of small intensity and their biological effects. Nature 1928, 121, 622–624. [Google Scholar] [CrossRef]
- Harvey, E.N.; Harvey, E.B.; Loomis, A.L. Further observations on the effect of high frequency sound waves on living matter. Biol. Bull. 1928, 55, 459–469. [Google Scholar] [CrossRef]
- British Medical Ultrasound Society. Guidelines for the Safe Use of Diagnostic Ultrasound Equipment. SAGE J. 2000, 18, 52–59. [Google Scholar]
- ter Haar, G. Safety and bio-effects of ultrasound contrast agents. Med. Biol. Eng. Comput. 2009, 47, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Postema, M.; Gilja, O.H. Contrast-enhanced and targeted ultrasound. World J. Gastroenterol. 2011, 17, 28–41. [Google Scholar] [CrossRef] [PubMed]
- Voigt, J.U. Ultrasound molecular imaging. Methods 2009, 48, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Postema, M.; van Wamel, A.; Lancée, C.T.; de Jong, N. Ultrasound-induced encapsulated microbubble phenomena. Ultrasound Med. Biol. 2004, 30, 827–840. [Google Scholar] [CrossRef] [PubMed]
- Heppner, P.; Lindner, J.R. Contrast ultrasound assessment of angiogenesis by perfusion and molecular imaging. Expert Rev. Mol. Diagn. 2005, 5, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Prentice, P.; Cuschieri, A.; Dholakia, K.; Prausnitz, M.; Campbell, P. Membrane disruption by optically controlled microbubble cavitation. Nat. Phys. 2005, 1, 107–110. [Google Scholar] [CrossRef]
- Sundaram, J.; Mellein, B.R.; Mitragotri, S. An experimental and theoretical analysis of ultrasound-induced permeabilization of cell membranes. Biophys. J. 2003, 84, 3087–3101. [Google Scholar] [CrossRef]
- Kudo, N.; Okada, K.; Yamamoto, K. Sonoporation by single-shot pulsed ultrasound with microbubbles adjacent to cells. Biophys. J. 2009, 96, 4866–4876. [Google Scholar] [CrossRef] [PubMed]
- Keyhani, K.; Guzmán, H.R.; Parsons, A.; Lewis, T.N.; Prausnitz, M.R. Intracellular drug delivery using low-frequency ultrasound: Quantification of molecular uptake and cell viability. Pharm. Res. 2001, 18, 1514–1520. [Google Scholar] [CrossRef] [PubMed]
- Postema, M.; Gilja, O.H. Ultrasound-directed drug delivery. Curr. Pharm. Biotechnol. 2007, 8, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.G.; Berry, J.L.; Lee, T.C.; Wang, A.T.; Honowitz, S.; Murphree, A.L.; Varshney, N.; Hinton, D.R.; Fawzi, A.A. Sonoporation enhances chemotherapeutic efficacy in retinoblastoma cells in vitro. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3868–3873. [Google Scholar] [CrossRef] [PubMed]
- Delalande, A.; Postema, M.; Mignet, N.; Midoux, P.; Pichon, C. Ultrasound and microbubble-assisted gene delivery: Recent advances and ongoing challenges. Therap. Deliv. 2012, 3, 1199–1215. [Google Scholar] [CrossRef]
- Kaufman, G.E.; Miller, M.W.; Griffiths, T.D.; Ciaravino, V.; Carstensen, E.L. Lysis and viability of cultured mammalian cells exposed to 1 MHz ultrasound. Ultrasound Med. Biol. 1977, 3, 21–25. [Google Scholar] [CrossRef]
- Brayman, A.A.; Coppage, M.L.; Vaidya, S.; Miller, M.W. Transient poration and cell surface receptor removal from human lymphocytes in vitro by 1 MHz ultrasound. Ultrasound Med. Biol. 1999, 25, 999–1008. [Google Scholar] [CrossRef]
- Firestein, F.; Rozenszajn, L.A.; Shemesh-Darvish, L.; Elimelech, R.; Radnay, J.; Rosenschein, U. Induction of apoptosis by ultrasound application in human malignant lymphoid cells: role of mitochondria-caspase pathway activation. Ann. N. Y. Acad. Sci. 2003, 1010, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Duvshani-Eshet, M.; Machluf, M. Therapeutic ultrasound optimization for gene delivery: A key factor achieving nuclear DNA localization. J. Control. Release 2005, 108, 513–528. [Google Scholar] [CrossRef] [PubMed]
- Delalande, A.; Kotopoulis, S.; Rovers, T.; Pichon, C.; Postema, M. Sonoporation at a low mechanical index. Bubble Sci. Eng. Technol. 2011, 3, 3–12. [Google Scholar] [CrossRef]
- Johansen, K.; Kimmel, E.; Postema, M. Theory of red blood cell oscillations in an ultrasound field. Arch. Acoust. 2017, 42, 121–126. [Google Scholar] [CrossRef]
- Krasovitski, B.; Frenkel, V.; Shoham, S.; Kimmel, E. Intramembrane cavitation as a unifying mechanism for ultrasound-induced bioeffects. Proc. Natl Acad. Sci. USA 2011, 108, 3258–3263. [Google Scholar] [CrossRef] [PubMed]
- Johansen, K.; Postema, M. Lagrangian formalism for computing oscillations of spherically symmetric encapsulated acoustic antibubbles. Hydroacoustics 2016, 19, 197–208. [Google Scholar]
- Shamloo, A.; Boodaghi, M. Design and simulation of a microfluidic device for acoustic cell separation. Ultrasonics 2018, 84, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Leighton, T.G. The Acoustic Bubble; Academic: London, UK, 1994. [Google Scholar]
- Shi, J.; Ahmed, D.; Mao, X.; Lin, S.C.S.; Lawit, A.; Huang, T.J. Acoustic tweezers: Patterning cells and microparticles using standing surface acoustic waves (SSAW). Lab Chip 2009, 9, 2890–2895. [Google Scholar] [CrossRef] [PubMed]
- Dayton, P.A.; Morgan, K.E.; Klibanov, A.L.; Brandenburger, G.; Nightingale, K.R.; Ferrara, K.W. A preliminary evaluation of the effects of primary and secondary radiation forces on acoustic contrast agents. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 1997, 44, 1264–1277. [Google Scholar] [CrossRef]
- Duckhouse, H.; Mason, T.J.; Phull, S.S.; Lorimer, J.P. The effect of sonication on microbial disinfection using hypochlorite. Ultrason. Sonochem. 2004, 11, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Trampuz, A.; Piper, K.E.; Hanssen, A.D.; Osmon, D.R.; Cockerill, F.R.; Steckelberg, J.M.; Patel, R. Sonication of explanted prosthetic components in bags for diagnosis of prosthetic joint infection is associated with risk of contamination. J. Clin. Microbiol. 2006, 44, 628–631. [Google Scholar] [CrossRef] [PubMed]
- Selman, G.G.; Jurand, A. An electron microscope study of the endoplasmic reticulum in Newt notochord cells after disturbance with ultrasonic treatment and subsequent regeneration. J. Cell Biol. 1964, 20, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Macintosh, I.J.C.; Davey, D.A. Chromosome aberrations induced by an ultrasonic fetal pulse detector. Br. Med. J. 1970, 4, 92–93. [Google Scholar] [CrossRef] [PubMed]
- Ravitz, M.J.; Schnitzler, R.M. Morphological changes induced in the frog semitendinosus muscle fiber by localized ultrasound. Exp. Cell Res. 1970, 60, 78–85. [Google Scholar] [CrossRef]
- Miller, M.W.; Voorhees, S.M.; Carstensen, E.L.; Eames, F.A. An histological study of the effect of ultrasound on growth of Vicia faba roots. Radiat. Bot. 1974, 14, 201–206. [Google Scholar] [CrossRef]
- Miller, M.W. Comparison of micronuclei induction for X-ray and ultrasound exposures of Vicia faba root meristem cells. Ultrasound Med. Biol. 1978, 4, 267. [Google Scholar] [CrossRef]
- Miller, D.L. Cell death thresholds in Elodea for 0.45–10 MHz ultrasound compared to gas-body resonance theory. Ultrasound Med. Biol. 1979, 5, 351–357. [Google Scholar] [CrossRef]
- Miller, D.L. A cylindrical-bubble model for the response of plant-tissue gas bodies to ultrasound. J. Acoust. Soc. Am. 1979, 65, 1313–1321. [Google Scholar] [CrossRef]
- Dyer, H.J.; Nyborg, W.L. Ultrasonically-induced movements in cells and cell models. IRE Trans. Med. Electron. 1960, 163–165. [Google Scholar] [CrossRef]
- Gershoy, A.; Nyborg, W.L. Perturbation of plant-cell contents by ultrasonic micro-irradiation. J. Acoust. Soc. Am. 1973, 54, 1356–1367. [Google Scholar] [CrossRef]
- Trenchard, P.M. Ultrasound-induced orientation of discoid platelets and simultaneous changes in light transmission: preliminary characterisation of the phenomenon. Ultrasound Med. Biol. 1987, 13, 183–195. [Google Scholar] [CrossRef]
- Williams, A.R.; Sykes, S.M.; O’Brien, W.D., Jr. Ultrasonic exposure modifies platelet morphology and function in vitro. Ultrasound Med. Biol. 1977, 2, 311–317. [Google Scholar] [CrossRef]
- Miller, M.W.; Azadniv, M.; Doida, Y.; Brayman, A.A. Effect of a stabilized microbubble contrast agent on CW ultrasound induced red blood cell lysis in vitro. Echocardiogr. J. 1995, 12, 1–11. [Google Scholar] [CrossRef]
- Liu, J.; Lewis, T.N.; Prausnitz, M.R. Non-invasive assessment and control of ultrasound-mediated membrane permeabilization. Pharm. Res. 1998, 15. [Google Scholar]
- Tachibana, K.; Uchida, T.; Ogawa, K.; Yamashita, N.; Tamura, K. Induction of cell-membrane porosity by ultrasound. Lancet 1999, 353, 1409. [Google Scholar] [CrossRef]
- Belgrader, P.; Hansford, D.; Kovacs, G.T.A.; Venkateswaran, K.; Mariella, R., Jr.; Milanovich, F.; Nasarabadi, S.; Okuzumi, M.; Pourahmadi, F.; Northrup, M.A. A minisonicator to rapidly disrupt bacterial spores for DNA analysis. Anal. Chem. 1999, 71, 4232–4236. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.T.; Belgrader, P.; Furman, B.J.; Pourahmadi, F.; Kovacs, G.T.A.; Northrup, M.A. Lysing bacterial spores by sonication through a flexible interface in a microfluidic system. Anal. Chem. 2001, 73, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Ashush, H.; Rozenszajn, L.A.; Blass, M.; Barda-Saad, M.; Azimov, D.; Radnay, J.; Zipori, D.; Rosenschein, U. Apoptosis induction of human myeloid leukemic cells by ultrasound exposure. Cancer Res. 2000, 60, 1014–1020. [Google Scholar] [PubMed]
- Taniyama, Y.; Tachibana, K.; Hiraoka, K.; Namba, T.; Yamasaki, K.; Hashiya, N.; Aoki, M.; Ogihara, T.; Yasufumi, K.; Morishita, R. Local delivery of plasmid DNA into rat carotid artery using ultrasound. Circulation 2002, 105, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Lagneaux, L.; Delforge, A.; Dejeneffe, M.; Massy, M.; Moerman, C.; Hannecart, B.; Canivet, Y.; Lepeltier, M.F.; Bron, D. Ultrasonic low-energy treatment: A novel approach to induce apoptosis in human leukemic cells. Exp. Hematol. 2002, 30, 1293–1301. [Google Scholar] [CrossRef]
- Li, G.; Xiao, H.; Glo, M.; Cheg, J. Miniaturized cell lysis device using spherically focused ultrasound. Tsinghua Sci. Technol. 2003, 8, 487–492. [Google Scholar]
- Song, Y.; Hahn, T.; Thompson, I.P.; Mason, T.J.; Preston, G.M.; Li, G.; Paniwnyk, L.; Huang, W.E. Ultrasound-mediated DNA transfer for bacteria. Nucleic Acids Res. 2007, 35, e129-1–e129-9. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Kondo, T.; Ogawa, R.; Feril, L.B.; Zhao, Q.L.; Watanabe, A.; Tsukada, K. Combination of doxorubicin and low-intensity ultrasound causes a synergistic enhancement in cell killing and an additive enhancement in apoptosis induction in human lymphoma U937 cells. Cancer Chemother. Pharmacol. 2008, 61, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Monsen, T.; Lövgren, E.; Widerström, M.; Wallinder, L. In vitro effect of ultrasound on bacteria and suggested protocol for sonication and diagnosis of prosthetic infections. J. Clin. Microbiol. 2009, 47, 2496–2501. [Google Scholar] [CrossRef] [PubMed]
- Drakopoulou, S.; Terzakis, S.; Fountoulakis, M.S.; Mantzavinos, D.; Manios, T. Ultrasound-induced inactivation of gram-negative and gram-positive bacteria in secondary treated municipal wastewater. Ultrason. Sonochem. 2009, 16, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Kotopoulis, S.; Schommartz, A.; Postema, M. Sonic cracking of blue-green algae. Appl. Acoust. 2009, 70, 1306–1312. [Google Scholar] [CrossRef]
- Zhang, G.; Zhang, P.; Liu, H.; Wang, B. Ultrasonic damages on cyanobacterial photosynthesis. Ultrason. Sonochem. 2006, 13, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, Y.; Fujiwara, Y.; Campbell, P.; Zhao, Q.L.; Ogawa, R.; Hassan, M.A.; Tabuchi, Y.; Takasaki, I.; Takahashi, A.; Kondo, T. DNA double-strand breaks induced by cavitational mechanical effects of ultrasound in cancer cell lines. PLoS ONE 2012, 7, e29012. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.A.; Ahmed, I.S.; Campbell, P.; Kondo, T. Enhanced gene transfection using calcium phosphate co-precipitates and low-intensity pulsed ultrasound. Eur. J. Pharm. Sci. 2012, 47, 768–773. [Google Scholar] [CrossRef] [PubMed]
- Ly, M.; Lu, F.; Maheshwari, G.; Subramanian, S. Microscale acoustic disruption of mammalian cells for intracellular product release. J Biotechnol. 2014, 184, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Samal, A.B.; Adzerikho, I.D.; Mrochek, A.G.; Loiko, E.N. Platelet aggregation and change in intracellular Ca2+ induced by low frequency ultrasound in vitro. Eur. J. Ultrasound 2000, 11, 53–59. [Google Scholar] [CrossRef]
- Zourob, M.; Hawkes, J.J.; Coakley, W.T.; Treves Brown, B.J.; Fielden, P.R.; McDonnell, M.B.; Goddard, N.J. Optical leaky waveguide sensor for detection of bacteria with ultrasound attractor force. Anal. Chem. 2005, 77, 6163–6168. [Google Scholar] [CrossRef] [PubMed]
- Mizrahi, N.; Zhou, E.H.; Lenormand, G.; Krishnan, R.; Weihs, D.; Butler, J.P.; Weitz, D.A.; Fredberg, J.J.; Kimmel, E. Low intensity ultrasound perturbs cytoskeleton dynamics. Soft Matter 2012, 8, 2438–2443. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, J.J.; Limaye, M.S.; Coakley, W.T. Filtration of bacteria and yeast by ultrasound-enhanced sedimentation. J. Appl. Microbiol. 1997, 82, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Mazzawi, N.; Postema, M.; Kimmel, E. Bubble-like response of living blood cells and microparticles in an ultrasound field. Acta Phys. Pol. A 2015, 127, 103–105. [Google Scholar] [CrossRef]
- Ding, X.; Peng, Z.; Lin, S.C.S.; Geri, M.; Li, S.; Li, P.; Chen, Y.; Dao, M.; Suresh, S.; Huang, T.J. Cell separation using tilted-angle standing surface acoustic waves. Proc. Natl Acad. Sci. USA 2014, 11, 12992–12997. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Mao, Z.; Peng, Z.; Zhou, L.; Chen, Y.; Huang, P.H.; Truica, C.I.; Drabick, J.J.; El-Deiry, W.S.; Dao, M.; et al. Acoustic separation of circulating tumor cells. Proc. Natl Acad. Sci. USA 2015, 112, 4970–4975. [Google Scholar] [CrossRef] [PubMed]
- Walther, T.; Postema, M. Device for the Identification, Separation and/or Cell Type-Specific Manipulation of at Least One Cell of a Cellular System. U.S. Patent 14476187, 3 September 2014. [Google Scholar]
- Pitt, W.G.; Ross, S.A. Ultrasound increases the rate of bacterial cell growth. Biotechnol. Prog. 2003, 19, 1038–1044. [Google Scholar] [CrossRef] [PubMed]
- Raz, D.; Zaretsky, U.; Einav, S.; Elad, D. Cellular alterations in cultured endothelial cells exposed to therapeutic ultrasound irradiation. Endothelium 2005, 12, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Hultström, J.; Manneberg, O.; Dopf, K.; Hertz, H.M.; Brismar, H.; Wiklund, M. Proliferation and viability of adherent cells manipulated by standing-wave ultrasound in a microfluidic chip. Ultrasound Med. Biol. 2007, 33, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Zderic, V.; Clark, J.I.; Vaezy, S. Drug delivery into the eye with the use of ultrasound. J. Ultrasound Med. 2004, 23, 1349–1359. [Google Scholar] [CrossRef] [PubMed]
- Or, M.; Kimmel, E. Modeling linear vibration of cell nucleus in low intensity ultrasound field. Ultrasound Med. Biol. 2009, 35, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Frenkel, V.; Kimmel, E.; Iger, Y. Ultrasound-facilitated transport of silver chloride (AgCl) particles in fish skin. J. Control. Release 2000, 68, 251–261. [Google Scholar] [CrossRef]
- Mazzawi, N.; Kimmel, E.; Tsarfaty, I. The effect of low-intensity ultrasound and met signaling on cellular motility and morphology. Appl. Acoust. 2019, 143, 1–6. [Google Scholar] [CrossRef]
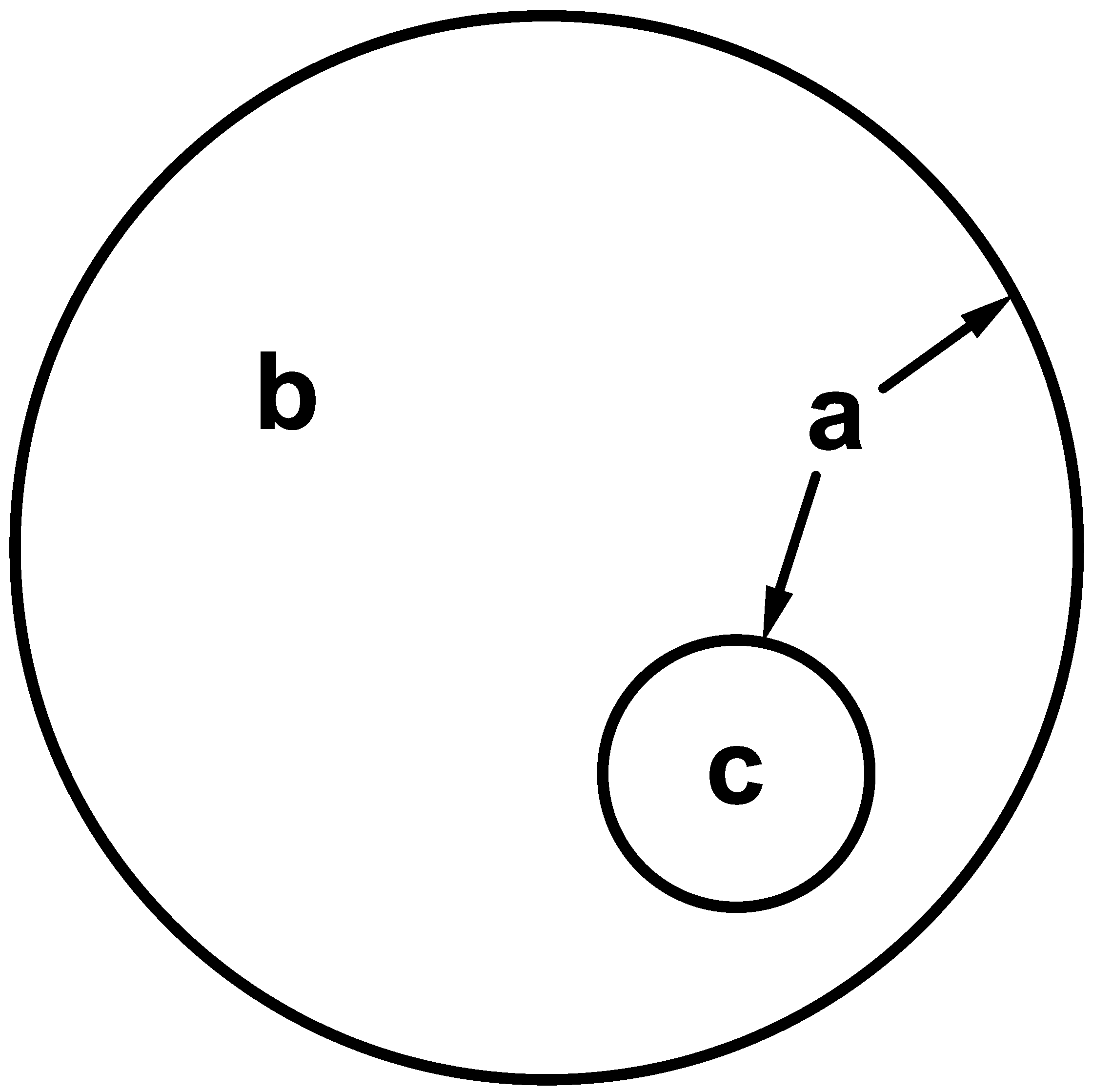
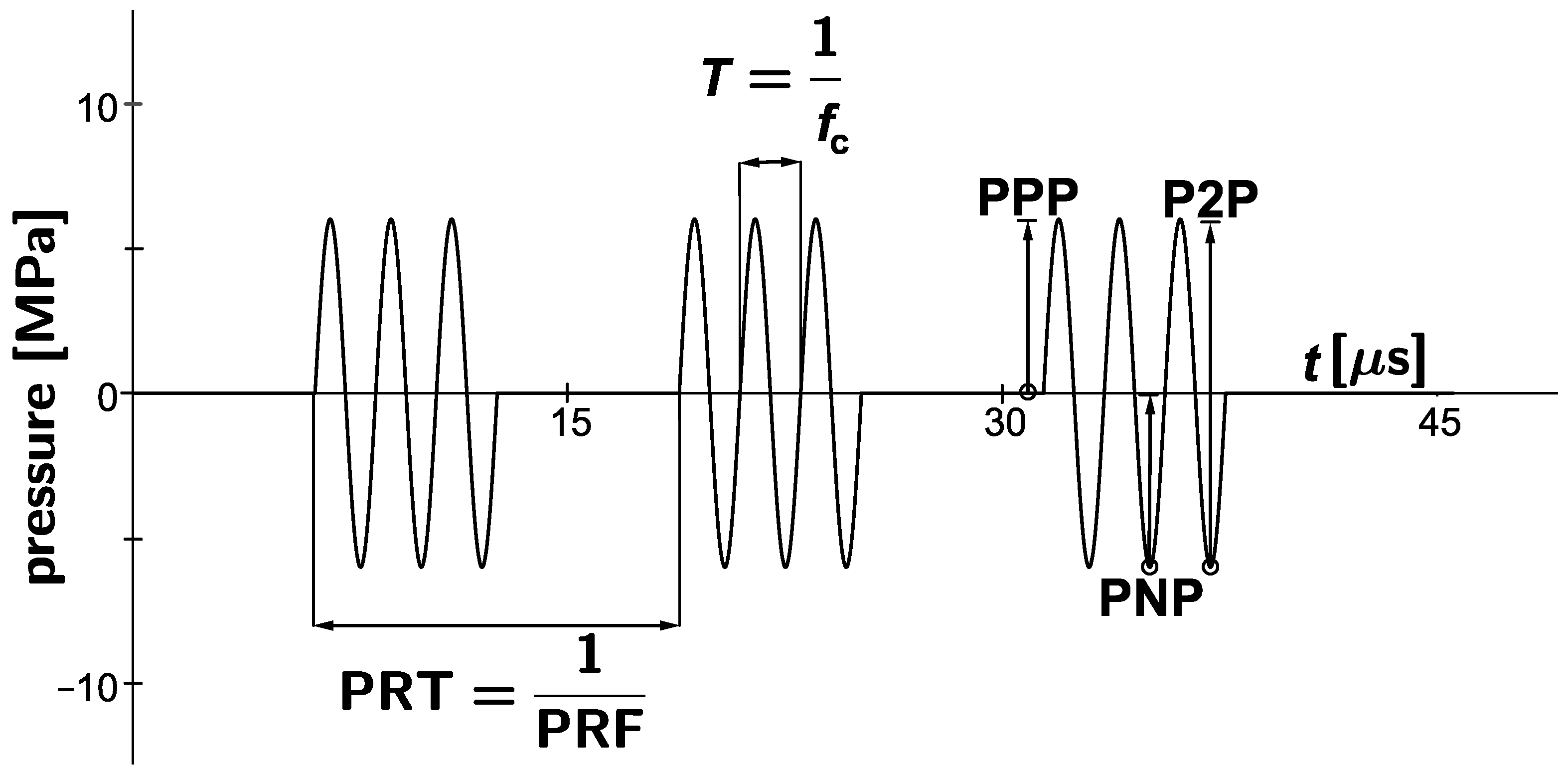
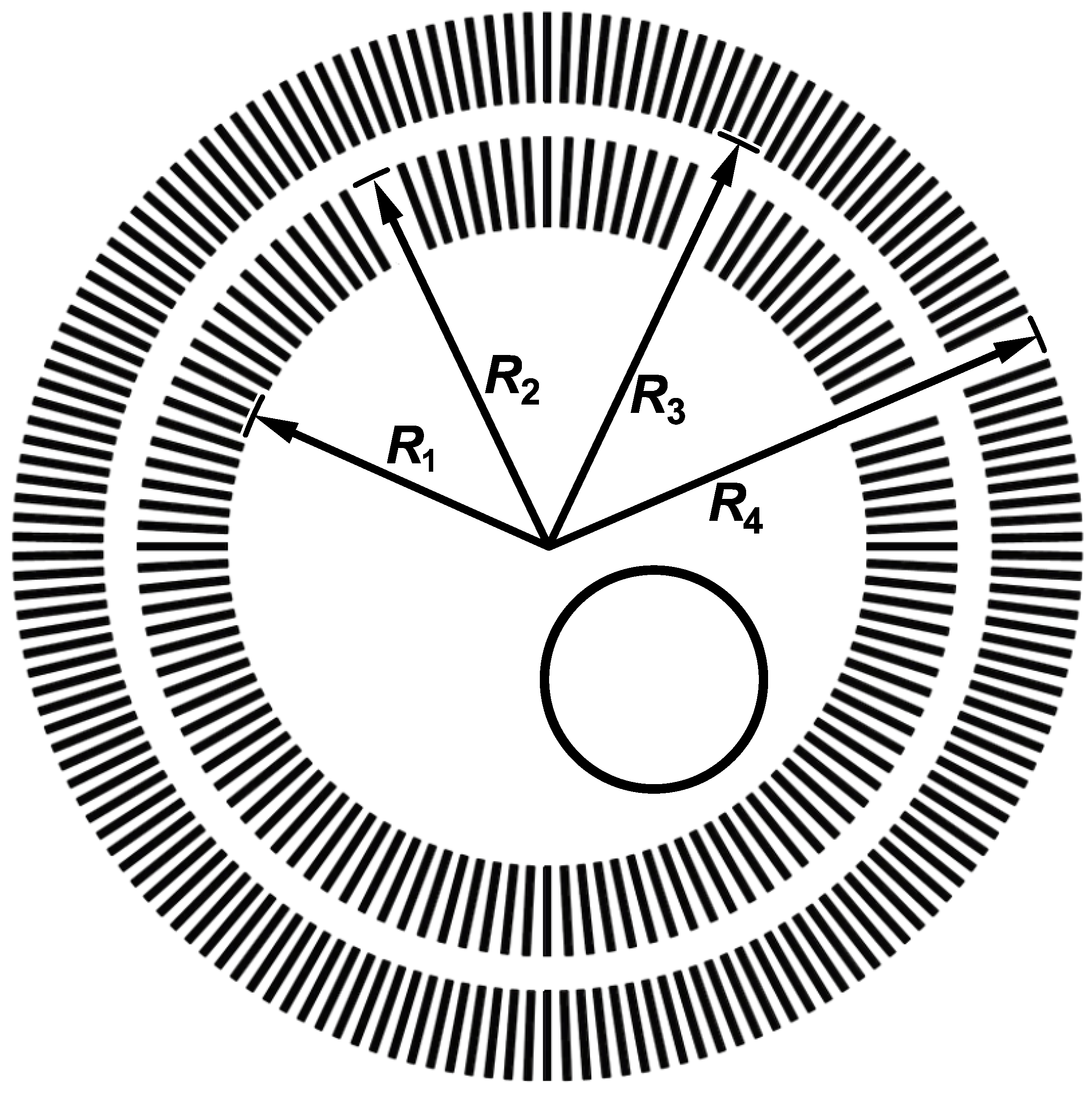
| Cell Type | Frequency | Amplitude | Main Effect | Ref. |
|---|---|---|---|---|
| Newt notochord | 1 MHz | 8–15 W cm | Disruption | [66] |
| Blood | 2.25 MHz | 30 mW | Chromosomal damage | [67] |
| Frog muscle | 85 kHz | 1–5 m | Structure disruption | [68] |
| Elodea | 0.45–10 MHz | 75–180 mW cm | Cell death | [71,72] |
| Platelets | 1–10 MHz | 0.5–76 kPa | Streaming | [75] |
| Platelets | 1 MHz | 0.2, 0.6 W cm | Disruption | [76] |
| Erythrocytes | 1 MHz | 0–5 W cm | Lysis | [77] |
| Erythrocytes | 24 kHz | 100 kPa–1 MPa | Permeation | [78] |
| HL-60 | 255 kHz | 0.4 W cm | Membrane changes | [79] |
| B. subtilis | 22–67 kHz | 20–60 W | Lysis | [80] |
| B. subtilis | 40 kHz | 25–38 m | Lysis | [81] |
| Leukaemic | 750 kHz | 22.4–103.7 W cm | Apoptosis | [82] |
| Platelets | 22 kHz | 1–8.8 W cm | Aggregation | [95] |
| Endothelial | 2.5 W cm | Permeation | [83] | |
| Leukaemic | 1.8 MHz | 7 mW mL | Apoptosis | [84] |
| E. coli | 1 MHz | 5.2 W cm | Eradication | [85] |
| S. cerevisiae | 1 MHz | 5.2 W cm | Eradication | [85] |
| S. epidermidis | 75 kHz | 2 W cm | Proliferation | [103] |
| E. coli | 75 kHz | 2 W cm | Proliferation | [103] |
| P. aeruginosa | 75 kHz | 2 W cm | Proliferation | [103] |
| Cornea | 880 kHz | 0.19–0.56 W cm | Structural changes | [106] |
| Endothelial | 0.5–5 MHz | 1.2 W cm | Proliferation | [104] |
| Bacterial spores | 2–4 MHz | Translation | [96] | |
| COS-7 | 3 MHz | 0.85 MPa | Proliferation | [105] |
| Bacteria | 40 kHz | 240 mW cm | Permeation | [86] |
| U937 | 1 MHz | 0.3–0.5 W cm | Apoptosis | [87] |
| Bacteria | 40 kHz | Eradication | [88] | |
| Bacteria | 24 kHz | 1500 W L | Eradication | [89] |
| Anabaena sphaerica | 200 kHz–2.2 MHz | 40–68 kPa | Disruption | [90] |
| Microcystis aeruginosa | 25 kHz | 0.32 W mL | Proliferation | [91] |
| Fish epidermis | 3 MHz | 2.2 W cm | Organelle changes | [58,108] |
| HASM | 1 MHz | 1 W cm, 2 W cm | Cytoskeletal changes | [97] |
| Endothelial | 1 MHz | 50–300 kPa | Translation | [26] |
| Leukaemic | 1 MHz | 0.3–0.4 W cm | DNA breakage | [92] |
| HeLa | 1 MHz | 0.3 W cm | Permeation | [93] |
| MRC-5 | 0.1–10 W cm | Lysis | [94] | |
| Erythrocytes | 1 MHz, 3 MHz | 1–10 V | Translation | [99] |
| MCF-7, leukocytes | 19.4 MHz | 2 W cm | Separation | [100,101] |
| CHO, HEK | 7 MHz | Separation | [102] | |
| MDCK | 960 kHz | 200 kPa | Morphology changes | [109] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rubin, D.M.; Anderton, N.; Smalberger, C.; Polliack, J.; Nathan, M.; Postema, M. On the Behaviour of Living Cells under the Influence of Ultrasound. Fluids 2018, 3, 82. https://doi.org/10.3390/fluids3040082
Rubin DM, Anderton N, Smalberger C, Polliack J, Nathan M, Postema M. On the Behaviour of Living Cells under the Influence of Ultrasound. Fluids. 2018; 3(4):82. https://doi.org/10.3390/fluids3040082
Chicago/Turabian StyleRubin, David M., Nicole Anderton, Charl Smalberger, Jethro Polliack, Malavika Nathan, and Michiel Postema. 2018. "On the Behaviour of Living Cells under the Influence of Ultrasound" Fluids 3, no. 4: 82. https://doi.org/10.3390/fluids3040082
APA StyleRubin, D. M., Anderton, N., Smalberger, C., Polliack, J., Nathan, M., & Postema, M. (2018). On the Behaviour of Living Cells under the Influence of Ultrasound. Fluids, 3(4), 82. https://doi.org/10.3390/fluids3040082





