Nickel (II) and Cobalt (II) Alginate Biopolymers as a “Carry and Release” Platform for Polyhistidine-Tagged Proteins
Abstract
:1. Introduction
2. Results and Discussion
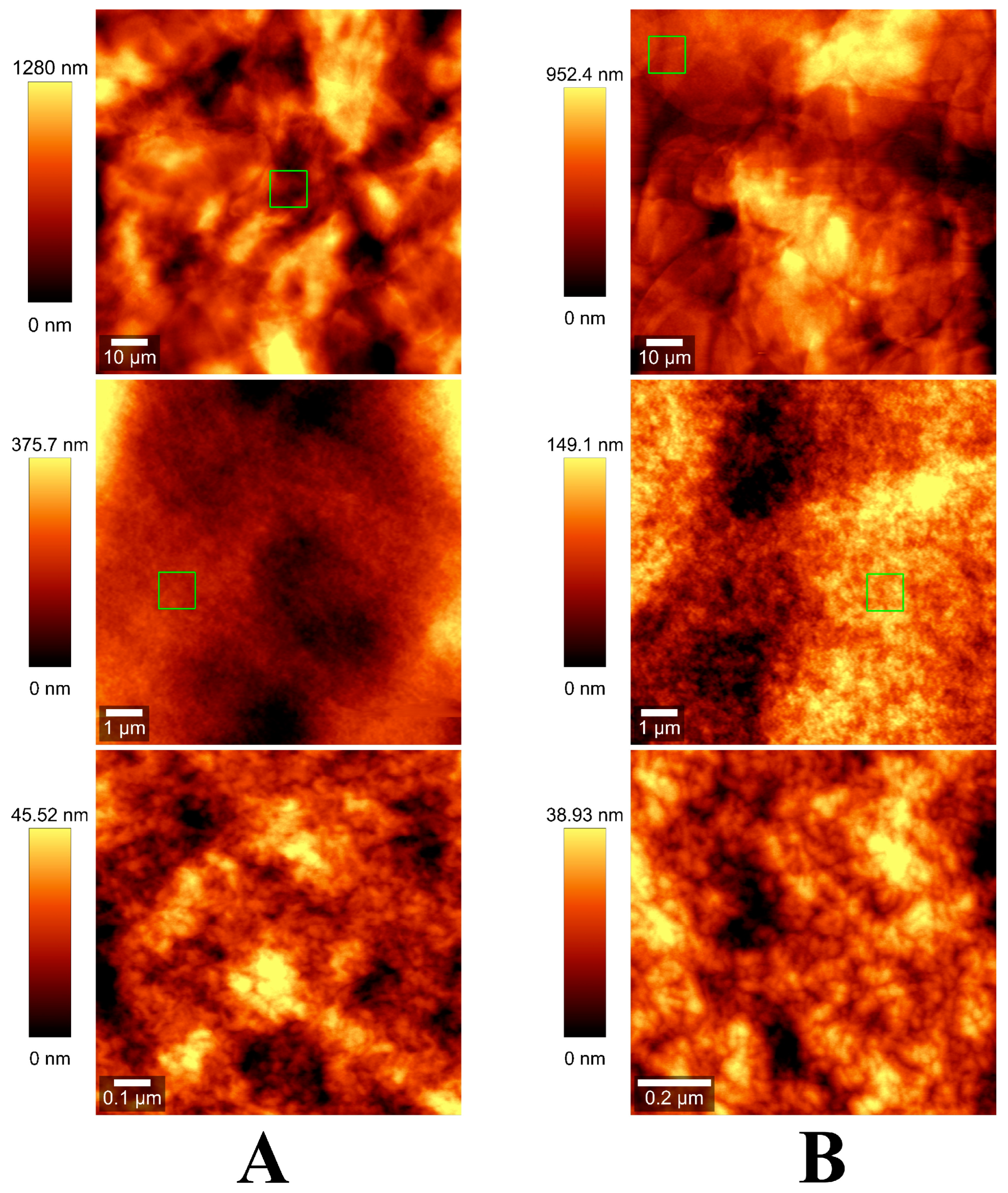
2.1. Characterization of Alginate Hydrogels
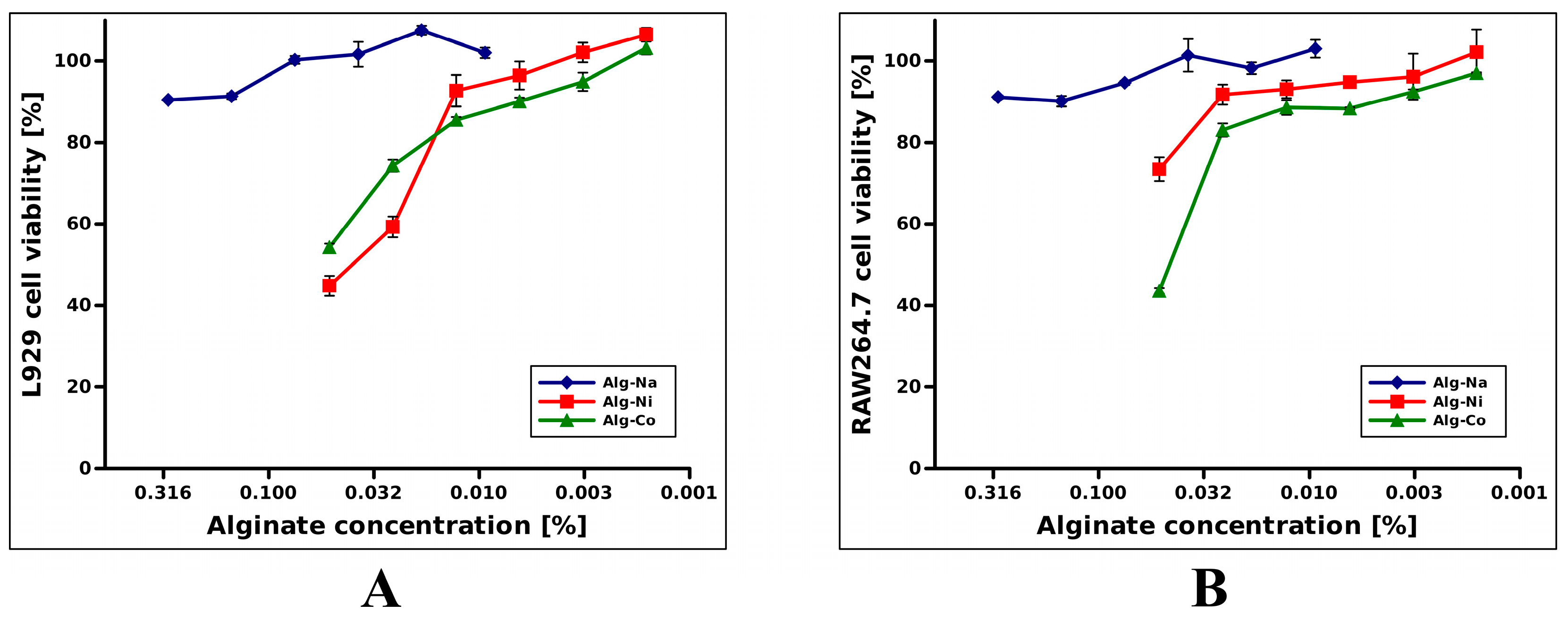
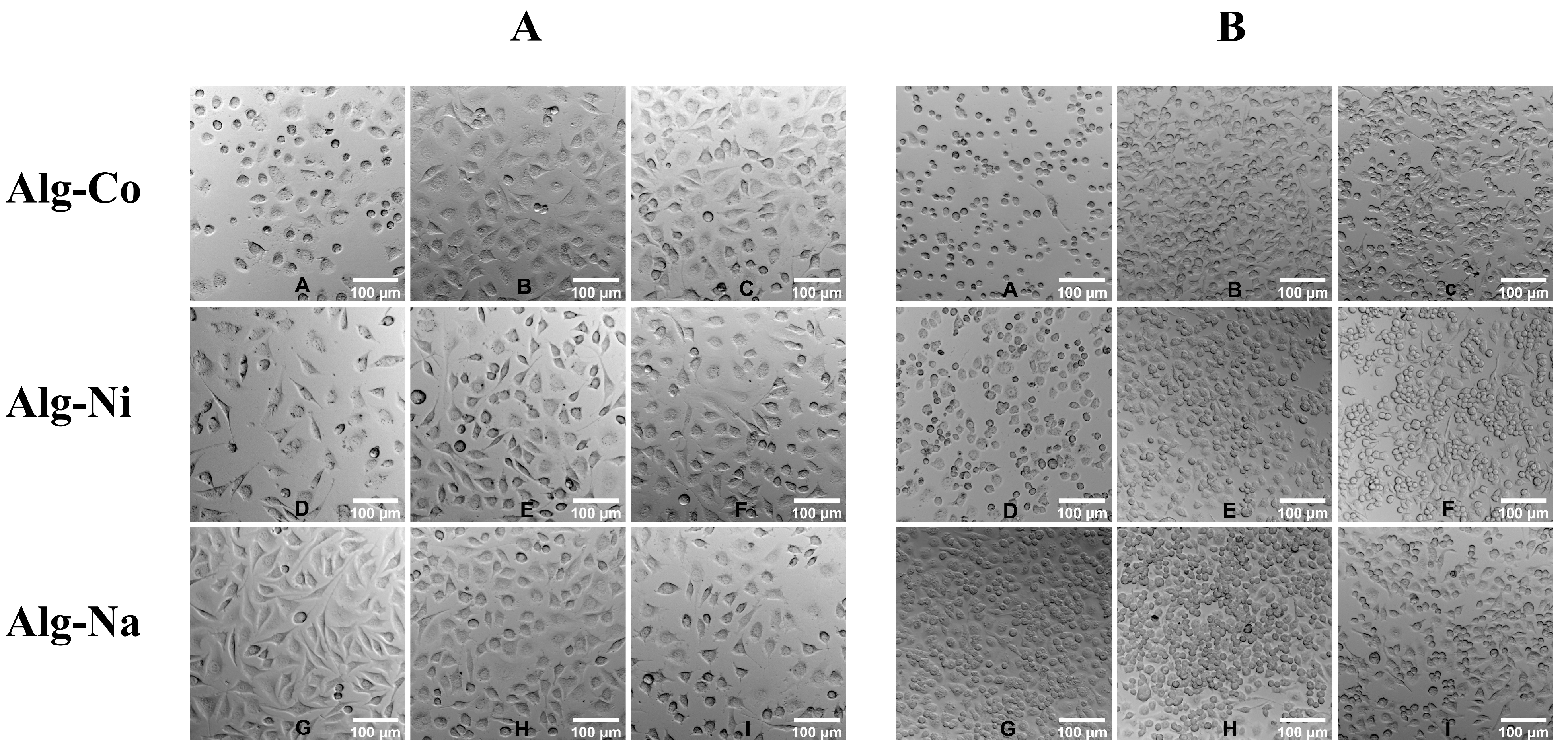
2.2. Cytotoxicity Assessment of Alginate Hydrogels
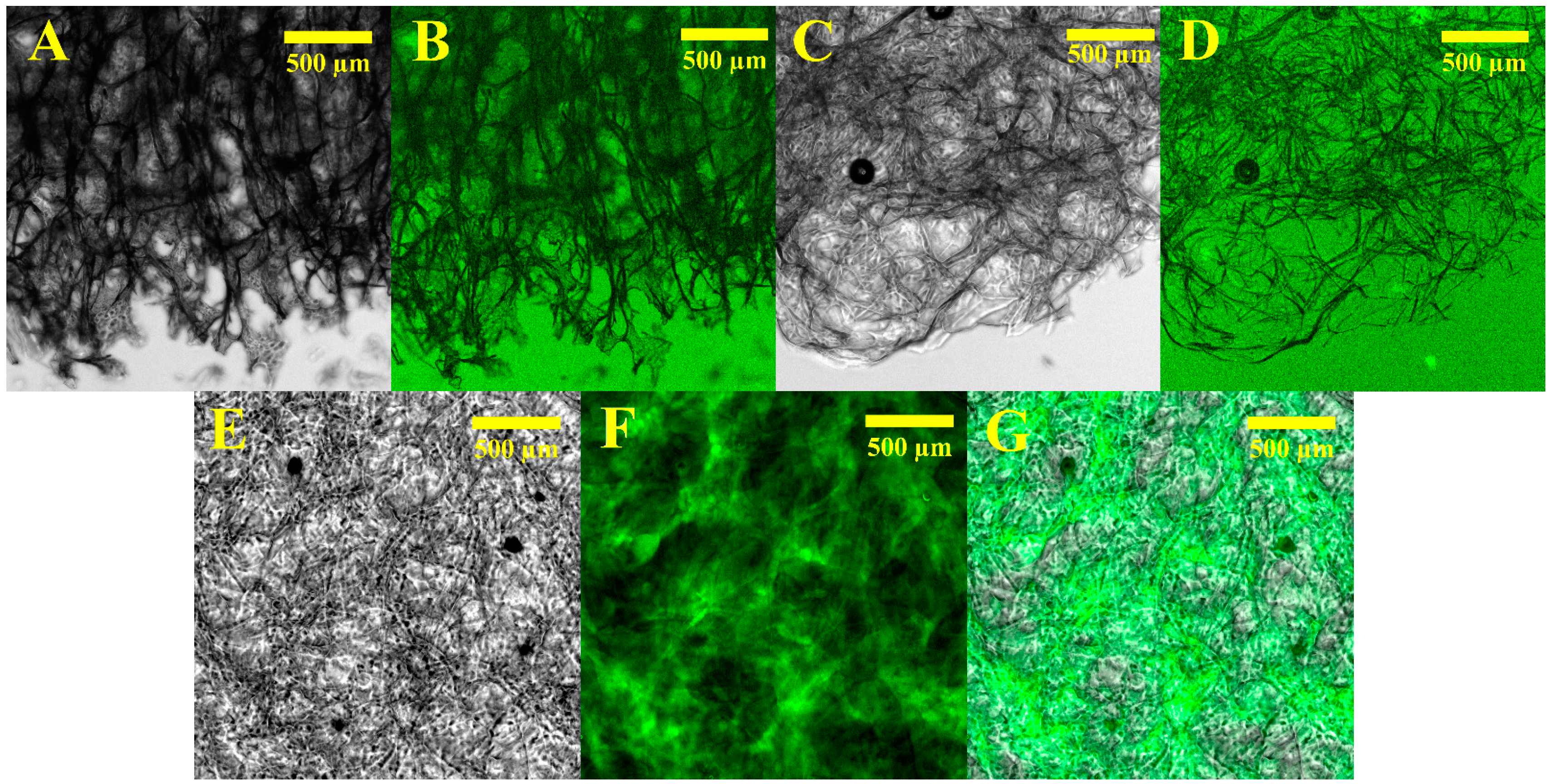
2.3. Alginate Hydrogels Specific Binding of Proteins
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Alginate Hydrogel Synthesis
4.3. Cell Culture and Cytotoxicity Assay
4.4. Specific His-Tagged Protein Binding Assessment
4.5. Protein Binding Capacity Assay
4.6. Optical and Fluorescence Imaging Investigation of Dried or Lyophilized Hydrogels
4.7. AFM Surface Characterization
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Reddy, M.S.B.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A Comparative Review of Natural and Synthetic Biopolymer Composite Scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef]
- McLachlan, J. Macroalgae (seaweeds): Industrial resources and their utilization. Plant Soil 1985, 89, 137–157. [Google Scholar] [CrossRef]
- Kirkpatrick, F.H. Overview of agarose gel properties. Curr. Commun. Cell Mol. Biol. 1990, 1, 9–22. [Google Scholar]
- Mourão, C.A.; Carmignotto, G.P.; Bueno, S.M.A. Separation of human IgG fragments using copper, nickel, zinc, and cobalt chelated to CM-Asp-agarose by positive and negative chromatography. J. Chromatogr. B 2016, 1017–1018, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Jerome, P. Alginate submicron beads prepared through w/o emulsification and gelation with CaCl2 nanoparti-cles. Food Hydrocoll. 2013, 31, 428–434. [Google Scholar]
- Wylie, A. Alginates as Food Additives. R. Soc. Health J. 1973, 93, 309–313. [Google Scholar] [CrossRef]
- Feng, P.; Luo, Y.; Ke, C.; Qiu, H.; Wang, W.; Zhu, Y.; Hou, R.; Xu, L.; Wu, S. Chitosan-Based Functional Materials for Skin Wound Repair: Mechanisms and Applications. Front. Bioeng. Biotechnol. 2021, 9, 111. [Google Scholar] [CrossRef]
- Li, Z.; Ramay, H.R.; Hauch, K.D.; Xiao, D.; Zhang, M. Chitosan–alginate hybrid scaffolds for bone tissue engineering. Biomaterials 2005, 26, 3919–3928. [Google Scholar] [CrossRef]
- Li, Y.; Ogorzalek, T.L.; Wei, S.; Zhang, X.; Yang, P.; Jasensky, J.; Brooks, C.L.; Marsh, E.N.G.; Chen, Z. Effect of immobilization site on the orientation and activity of surface-tethered enzymes. Phys. Chem. Chem. Phys. 2018, 20, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Pagel, M.; Annette, G. Multifunctional biomaterial coatings: Synthetic challenges and biological activity. Biol. Chem. 2017, 398, 3–22. [Google Scholar] [CrossRef]
- Malhotra, I.; Basir, S.F. Immobilization of invertase in calcium alginate and calcium alginate-kappa-carrageenan beads and its application in bioethanol production. Prep. Biochem. Biotechnol. 2020, 50, 494–503. [Google Scholar] [CrossRef]
- Zawawi, F.S.M. Enzyme activity and stability of lactase immobilized on two different supports: Calci-um alginate and magnetic chitosan. Malays. J. Fundam. Appl. Sci. 2020, 16, 413–417. [Google Scholar] [CrossRef]
- Welch, N.G.; Scoble, J.; Muir, B.; Pigram, P.J. Orientation and characterization of immobilized antibodies for improved immunoassays (Review). Biointerphases 2017, 12, 02D301. [Google Scholar] [CrossRef] [Green Version]
- Fathi, Z.; Doustkhah, E.; Ebrahimipour, G.; Darvishi, F. Noncovalent Immobilization of Yarrowia lipolytica Lipase on Dendritic-Like Amino Acid-Functionalized Silica Nanoparticles. Biomolecules 2019, 9, 502. [Google Scholar] [CrossRef] [Green Version]
- Ana, I.; Martina, L.C. Agarose vs. Methacrylate as Material Supports for Enzyme Immobilization and Continuous Processing. Catalysts 2021, 11, 814. [Google Scholar]
- Wawrzyńska, E.; Kubies, D. Alginate Matrices for Protein Delivery—A Short Review. Physiol. Res. 2018, 67, S319–S334. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, R.; Zou, L.; McClements, D.J. Protein encapsulation in alginate hydrogel beads: Effect of pH on microgel stability, protein retention and protein release. Food Hydrocoll. 2016, 58, 308–315. [Google Scholar] [CrossRef] [Green Version]
- Bresolin, I.T.L. Evaluation of immobilized metal-ion affinity chromatography (IMAC) as a technique for IgG 1 monoclonal antibodies purification: The effect of chelating ligand and support. Appl. Biochem. Biotechnol. 2010, 160, 2148–2165. [Google Scholar] [CrossRef] [PubMed]
- Bauer, W.S.; Richardson, K.A.; Adams, N.M.; Ricks, K.M.; Gasperino, D.J.; Ghionea, S.J.; Rosen, M.; Nichols, K.P.; Weigl, B.H.; Haselton, F.R.; et al. Rapid concentration and elution of malarial antigen histidine-rich protein II using solid phase Zn(II) resin in a simple flow-through pipette tip format. Biomicrofluidics 2017, 11, 034115. [Google Scholar] [CrossRef]
- Block, H.; Maertens, B.; Spriestersbach, A.; Brinker, N.; Kubicek, J.; Fabis, R.; Labahn, J.; Schäferm, F. Withdrawn: Reprint of: Immobilized-Metal Affinity Chromatography (IMAC): A Review. Protein Expr. Purif. 2009, 463, 439–473. [Google Scholar] [CrossRef]
- Young, C.L.; Zachary, T.; Robinson, A.S. Recombinant protein expression and purification: A comprehensive review of affinity tags and microbial applications. Biotechnol. J. 2012, 7, 620–634. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.-Y.; Narayanan, J.; Liu, X.-Y.; Chong, T.K.; Chen, S.B.; Chung, T.-S. Topology Evolution and Gelation Mechanism of Agarose Gel. J. Phys. Chem. B 2005, 109, 5638–5643. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, X.; Liu, Y.; Li, H.; Li, H. Single-stranded structure of alginate and its conformation evolvement after an interaction with calcium ions as revealed by electron microscopy. RSC Adv. 2016, 6, 114779–114782. [Google Scholar] [CrossRef]
- Rosiak, P.; Latanska, I.; Paul, P.; Sujka, W.; Kolesinska, B. Modification of Alginates to Modulate Their Physic-Chemical Properties and Obtain Biomaterials with Different Functional Properties. Molecules 2021, 26, 7264. [Google Scholar] [CrossRef] [PubMed]
- Abasalizadeh, F.; Moghaddam, S.V.; Alizadeh, E.; Akbari, E.; Kashani, E.; Fazljou, S.M.B.; Torbati, M.; Akbarzadeh, A. Alginate-based hydrogels as drug delivery vehicles in cancer treatment and their applications in wound dressing and 3D bioprinting. J. Biol. Eng. 2020, 14, 8, Correction in 2020, 14, 17. [Google Scholar] [CrossRef]
- Kulanthaivel, S. Cobalt doped nano-hydroxyapatite incorporated gum tragacanth-alginate beads as angio-genic-osteogenic cell encapsulation system for mesenchymal stem cell based bone tissue engineering. Int. J. Biol. Macromol. 2021, 179, 101–115. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, X.; Ding, H.; He, Y.; Yang, H.; Chen, Y.; Chen, X.; Luo, X. Novel alginate particles decorated with nickel for enhancing ciprofloxacin removal: Characterization and mechanism analysis. Ecotoxicol. Environ. Saf. 2019, 169, 392–401. [Google Scholar] [CrossRef]
- Liu, C.; Tao, Y.; Xu, Y.; Liu, Y.; Zhu, P.; Wang, Y. Effect of Bio-Based Cobalt Alginate on the Fire Safety and Mechanical Properties for Epoxy Resin. Macromol. Mater. Eng. 2021, 306, 2100466. [Google Scholar] [CrossRef]
- Dalal, S.; Smita, R.; Guptam, M.N. Single-step purification of recombinant green fluorescent protein on expanded beds of immobilized metal affinity chromatography media. Biochem. Eng. J. 2008, 42, 301–307. [Google Scholar] [CrossRef]
- Silverio, G.B.; Sakanaka, L.S.; Alvim, I.D.; Shirai, M.; Grosso, C.R.F. Production and characterization of alginate microparticles obtained by ionic gelation and electrostatic adsorption of concentrated soy protein. Ciência Rural 2018, 48. [Google Scholar] [CrossRef]
- Paques, J.P.; van der Linden, E.; van Rijn, C.J.; Sagis, L.M. Preparation methods of alginate nanoparticles. Adv. Colloid Interface Sci. 2014, 209, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Tam, S.; Bilodeau, S.; Dusseault, J.; Langlois, G.; Hallé, J.-P.; Yahia, L. Biocompatibility and physicochemical characteristics of alginate–polycation microcapsules. Acta Biomater. 2011, 7, 1683–1692. [Google Scholar] [CrossRef]
- Genchi, G.; Carocci, A.; Lauria, G.; Sinicropi, M.S.; Catalano, A. Nickel: Human Health and Environmental Toxicology. Int. J. Environ. Res. Public Health 2020, 17, 679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahlström, M.G. Nickel allergy and allergic contact dermatitis: A clinical review of immunology, epidemiology, exposure, and treatment. Contact Dermat. 2019, 81, 227–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leyssens, L.; Vinck, B.; Van Der Straeten, C.; Wuyts, F.; Maes, L. Cobalt toxicity in humans—A review of the potential sources and systemic health effects. Toxicology 2017, 387, 43–56. [Google Scholar] [CrossRef]
- Cannella, V.; Altomare, R.; Chiaramonte, G.; Di Bella, S.; Mira, F.; Russotto, L.; Pisano, P.; Guercio, A. Cytotoxicity Evaluation of Endodontic Pins on L929 Cell Line. BioMed Res. Int. 2019, 2019, 3469525. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Zhang, Y.; Liu, J.; Wei, L.; Song, B.; Shao, L. Exposure of the murine RAW 264.7 macrophage cell line to dicalcium silicate coating: Assessment of cytotoxicity and pro-inflammatory effects. J. Mater. Sci. Mater. Med. 2016, 27, 59. [Google Scholar] [CrossRef]
- Stepanenko, O.V.; Verkhusha, V.V.; Kazakov, V.I.; Shavlovsky, M.M.; Kuznetsova, I.M.; Uversky, V.N.; Turoverov, K.K. Comparative Studies on the Structure and Stability of Fluorescent Proteins EGFP, zFP506, mRFP1, “dimer2”, and DsRed1. Biochemistry 2004, 43, 14913–14923. [Google Scholar] [CrossRef]
- Stavaru, C.; Onu, A.; Lupulescu, E.; Tucureanu, C.; Rasid, O.; Vlase, E.; Coman, C.; Caras, I.; Ghiorghisor, A.; Berbecila, L.; et al. Technology transfer of oil-in-water emulsion adjuvant manufacturing for pandemic influenza vaccine production in Romania: Preclinical evaluation of split virion inactivated H5N1 vaccine with adjuvant. Hum. Vaccines Immunother. 2016, 12, 1009–1026. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Guo, Y.; Zhao, Y.; Yu, L.; Chang, Z.; Pei, H.; Huang, J.; Chen, C.; Xue, H.; Xu, X.; et al. Expression of a recombinant FLT3 ligand and its emtansine conjugate as a therapeutic candidate against acute myeloid leukemia cells with FLT3 expression. Microb. Cell Factories 2021, 20, 67. [Google Scholar] [CrossRef] [PubMed]
- Vadana, M.; Cecoltan, S.; Ciortan, L.; Macarie, R.D.; Tucureanu, M.M.; Mihaila, A.C.; Droc, I.; Butoi, E.; Manduteanu, I. Molecular mechanisms involved in high glucose-induced valve calcification in a 3D valve model with human valvular cells. J. Cell. Mol. Med. 2020, 24, 6350–6361. [Google Scholar] [CrossRef] [PubMed]


| EGFP-HisTAG (µg) | Incubation Time (h) | |
|---|---|---|
| t1 = 1 h | t2 = 24 h | |
| 12.5 µg | 98.45% | 95.05% |
| 25 µg | 97.15% | 87.01% |
| 37.5 µg | 93.52% | 67.16% |
| 50 µg | 87.28% | 56.11% |
| 62.5 µg | 73.56% | 43.14% |
| EGFP-HisTAG (µg) | Incubation Time (h) | |
|---|---|---|
| t1 = 1 h | t2 = 24 h | |
| 12.5 µg | 96.87% | 78.50% |
| 25 µg | 85.91% | 67.50% |
| 37.5 µg | 77.24% | 59.77% |
| 50 µg | 71.92% | 57.44% |
| 62.5 µg | 66.40% | 52.35% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dumitrașcu, A.-M.; Caraș, I.; Țucureanu, C.; Ermeneanu, A.-L.; Tofan, V.-C. Nickel (II) and Cobalt (II) Alginate Biopolymers as a “Carry and Release” Platform for Polyhistidine-Tagged Proteins. Gels 2022, 8, 66. https://doi.org/10.3390/gels8020066
Dumitrașcu A-M, Caraș I, Țucureanu C, Ermeneanu A-L, Tofan V-C. Nickel (II) and Cobalt (II) Alginate Biopolymers as a “Carry and Release” Platform for Polyhistidine-Tagged Proteins. Gels. 2022; 8(2):66. https://doi.org/10.3390/gels8020066
Chicago/Turabian StyleDumitrașcu, Andrei-Mihai, Iuliana Caraș, Cătălin Țucureanu, Andreea-Laura Ermeneanu, and Vlad-Constantin Tofan. 2022. "Nickel (II) and Cobalt (II) Alginate Biopolymers as a “Carry and Release” Platform for Polyhistidine-Tagged Proteins" Gels 8, no. 2: 66. https://doi.org/10.3390/gels8020066
APA StyleDumitrașcu, A.-M., Caraș, I., Țucureanu, C., Ermeneanu, A.-L., & Tofan, V.-C. (2022). Nickel (II) and Cobalt (II) Alginate Biopolymers as a “Carry and Release” Platform for Polyhistidine-Tagged Proteins. Gels, 8(2), 66. https://doi.org/10.3390/gels8020066






