Fabrication and Characterizations of Pharmaceutical Emulgel Co-Loaded with Naproxen-Eugenol for Improved Analgesic and Anti-Inflammatory Effects
Abstract
1. Introduction
2. Results and Discussion
2.1. Physical Appearance and Thermodynamic Stability
2.2. Evaluation of pH
2.3. Viscosity Determination
2.4. Spreadability Study
2.5. Extrudability Study
2.6. Drug Content Assays
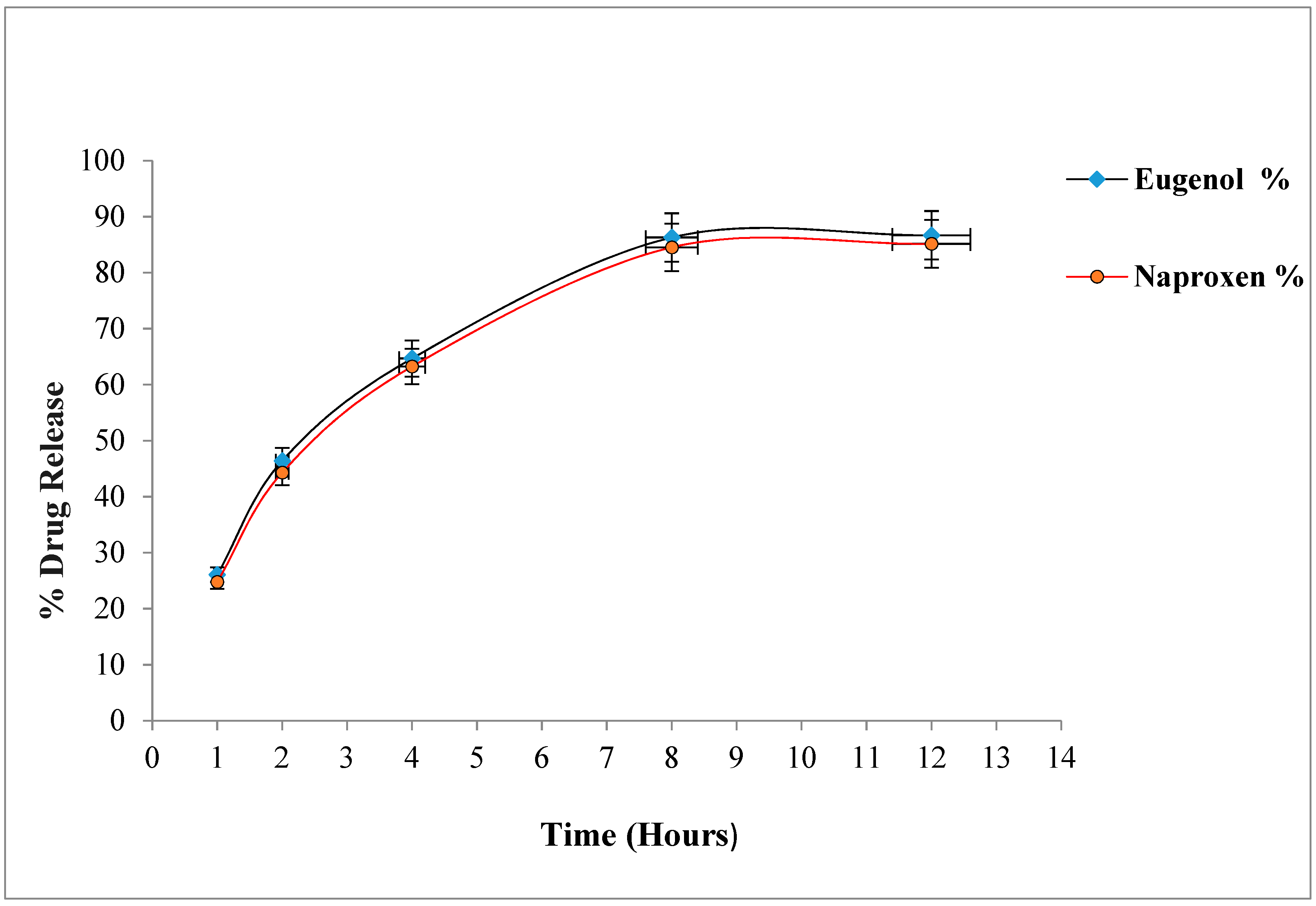
2.7. In-Vitro Drug Release Study
Drug Release Kinetics
2.8. Ex-Vivo Permeation Study
2.9. Drug Retention
2.10. In-Vivo Characterization of Naproxen-Eugenol Emulgel
2.10.1. Skin Irritation Study
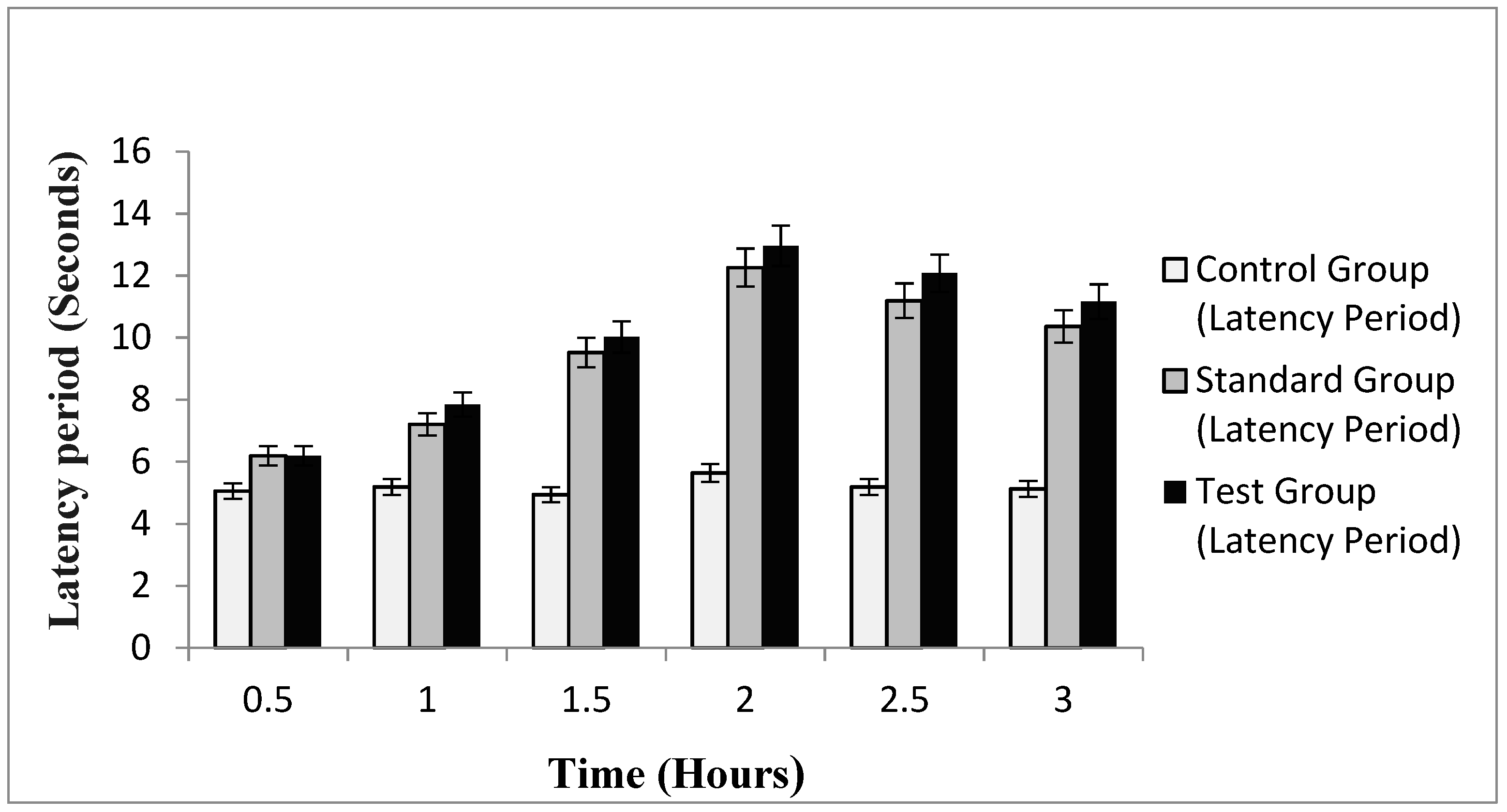
2.10.2. In-Vivo Analgesic Activity
2.10.3. In-Vivo Anti-Inflammatory Activity
3. Conclusions
4. Materials and Methods
4.1. Chemicals
4.2. Apparatus
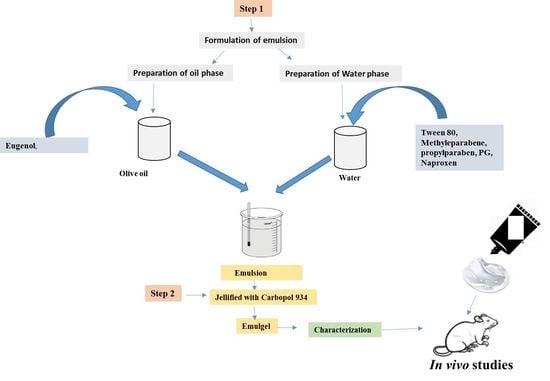
4.3. Preparation of Naproxen-Eugenol Emulsion
4.4. Preparation of Gel
4.5. Preparation of Naproxen-Eugenol Emulgel (NEE)
4.6. Physical Appearance
4.7. Determination of pH
4.8. Rheological Study
4.9. Determination of Drug Assays
4.9.1. Preparation of Standard Naproxen Solution
4.9.2. Preparation of Sample Naproxen Solution
4.9.3. Preparation of Mobile Phase for Eugenol
4.9.4. Preparation of Eugenol Standard Solution
4.9.5. Preparation of Eugenol Test Solution
4.9.6. Procedure
4.10. Spreadability Study
- M = weight tied to upper glass slide,
- L = length of glass slides,
- T = time taken to separate the glass slides from each other.
4.11. Extrudability Study
4.12. In-Vitro Drug Release
4.13. Drug Release Kinetics
4.14. Ethical Consideration
4.15. Ex-Vivo Permeation Study
4.16. Skin Retention Study
4.17. Skin Irritation Study
4.18. In-Vivo Anti-Inflammatory Activity
- Vc = inflammatory increase in paw volume of the control group,
- Vt = inflammatory increase in paw volume of the test group.
Treatment Protocols
- Group 1: Control group, Treated with 0.1 mL of 1% carrageenan injection only,
- Group 2: Test group, Treated with optimized naproxen-eugenol emulgel + 0.1 mL of 1% carrageenan injection.
- Group 3: Standard group, Treated with marketed emulgel (Voltral®) + 0.1 mL of 1% carrageenan injection.
4.19. In-Vivo Analgesic Activity
- Group 1: Control group, no treatment was given,
- Group 2: Test group, Treated with optimized naproxen-eugenol emulgel,
- Group 3: Standard group, Treated with marketed diclofenac emulgel (Voltral®).
5. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abdulkhaleq, L.A.; Assi, M.A.; Abdullah, R.; Zamri-Saad, M.; Taufiq-Yap, Y.H.; Hezmee, M.N.M. The crucial roles of inflammatory mediators in inflammation: A review. Vet. World 2018, 11, 627. [Google Scholar] [CrossRef]
- Adegbenro, O.O.; Opeyemi, O.T. Development and estimation of anti-inflammatory activity of topical etoricoxib emulgel by carrageenan induced paw oedema method. Univers. J. Pharm. Res. 2019, 4, 23–28. [Google Scholar] [CrossRef]
- Akram, A.; Khalid Khan, M.; Ali Khan, B. Preparation, Characterizations and Evaluation of Antifungal Activity of Eugenol-Linalool Emulgel against Anthrophilic Dermatophytic Trichophyton Rubrum; Main Group Chemistry; IOS Press: Amsterdam, The Netherlands, 2021; pp. 1–15. [Google Scholar]
- Akram, M.A.; Khan, B.A.; Khan, M.K.; Alqahtani, A.; Alshahrani, S.M.; Hosny, K.M. Fabrication and Characterization of Polymeric Pharmaceutical Emulgel Co-Loaded with Eugenol and Linalool for the Treatment of Trichophyton rubrum Infections. Polymers 2021, 13, 3904. [Google Scholar] [CrossRef]
- Akram, M.; Naqvi, S.B.S.; Khan, A. Design and development of insulin emulgel formulation for transdermal drug delivery and its evaluation. Pak. J. Pharm. Sci. 2013, 26, 323–332. [Google Scholar] [PubMed]
- Alexander, A.; Dwivedi, S.; Giri, T.K.; Saraf, S.; Saraf, S.; Tripathi, D.K. Approaches for breaking the barriers of drug permeation through transdermal drug delivery. J. Control. Release 2012, 164, 26–40. [Google Scholar] [CrossRef] [PubMed]
- Al-saraf, M.F.; Khalil, Y.I. Formulation and evaluation of topical itraconazole emulgel. Int. J. Pharm. Ther. J. 2016, 7, 9–17. [Google Scholar]
- Al-Suwayeh, S.A.; Taha, E.I.; Al-Qahtani, F.M.; Ahmed, M.O.; Badran, M.M. Evaluation of skin permeation and analgesic activity effects of carbopol lornoxicam topical gels containing penetration enhancer. Sci. World J. 2014, 23, 234–245. [Google Scholar] [CrossRef]
- Ambhore, N.P.; Dandagi, P.M.; Gadad, A.P.; Mandora, P. Formulation and characterization of tapentadol loaded emulgel for topical application. Indian J. Pharm. Educ. Res. 2017, 51, 525–535. [Google Scholar] [CrossRef]
- Angiolillo, D.J.; Weisman, S.M. Clinical pharmacology and cardiovascular safety of naproxen. Am. J. Cardiovasc. Drugs 2017, 17, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Arany, E.; Szabó, R.K.; Apáti, L.; Alapi, T.; Ilisz, I.; Mazellier, P.; Gajda-Schrantz, K. Degradation of naproxen by UV, VUV photolysis and their combination. J. Hazard. Mater. 2013, 262, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Arulselvan, P.; Fard, M.T.; Tan, W.S.; Gothai, S.; Fakurazi, S.; Norhaizan, M.E.; Kumar, S.S. Role of antioxidants and natural products in inflammation. Oxidative Med. Cell. Longev. Polym. 2016, 11, 3904–3912. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, S.; Whiteman, A.; Brandner, B. Transdermal drug delivery in pain management. Contin. Educ. Anaesth. Crit. Care Pain 2011, 11, 39–43. [Google Scholar] [CrossRef]
- Burki, I.K.; Khan, M.K.; Khan, B.A.; Uzair, B.; Braga, V.A.; Jamil, Q.A. Formulation development, characterization, and evaluation of a novel dexibuprofen-capsaicin skin emulgel with improved in vivo anti-inflammatory and analgesic effects. AAPS PharmSciTech 2020, 21, 211. [Google Scholar] [CrossRef] [PubMed]
- Bennett, J.M.; Reeves, G.; Billman, G.E.; Sturmberg, J.P. Inflammation–nature’s way to efficiently respond to all types of challenges: Implications for understanding and managing “the epidemic” of chronic diseases. Front. Med. 2018, 5, 316. [Google Scholar]
- Bonacucina, G.; Cespi, M.; Palmieri, G.F. Characterization and stability of emulsion gels based on acrylamide/sodium acryloyldimethyl taurate copolymer. AAPS PharmSciTech 2009, 10, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Brutzkus, J.C.; Shahrokhi, M.; Varacallo, M. Naproxen; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Banyal, M.; Joshi, S. EMULGEL: An Enormous Approach for Topical Delivery of Hydrophobic Drugs. AAPS PharmSciTech 2020, 18, 8–17. [Google Scholar]
- Cao, M.; Ren, L.; Chen, G. Formulation optimization and ex vivo and in vivo evaluation of celecoxib microemulsion-based gel for transdermal delivery. AAPS PharmSciTech 2017, 18, 1960–1971. [Google Scholar] [CrossRef]
- Chan, F.K.; Ching, J.Y.; Tse, Y.K.; Lam, K.; Wong, G.L.; Ng, S.C.; Kyaw, M.H. Gastrointestinal safety of celecoxib versus naproxen in patients with cardiothrombotic diseases and arthritis after upper gastrointestinal bleeding (CONCERN): An industry-independent, double-blind, double-dummy, randomised trial. Lancet 2017, 389, 2375–2382. [Google Scholar] [CrossRef]
- Chavda, V.; Vishal, R. Formulation and evaluation of naproxen emulgel for topical delivery by a modified method. Int. J. Compr. Pharm. 2013, 7, 1–4. [Google Scholar]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Zhao, L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarge 2018, 9, 7204. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.P.; Vase, L.; Hooten, W.M. Chronic pain: An update on burden, best practices, and new advances. Lancet 2021, 397, 2082–2097. [Google Scholar] [CrossRef]
- Cong, H.; Khaziakhmetova, V.; Zigashina, L. Rat paw oedema modeling and NSAIDs: Timing of effects. Int. J. Risk Saf. Med. 2015, 27, S76–S77. [Google Scholar] [CrossRef] [PubMed]
- Daniel, A.N.; Sartoretto, S.M.; Schmidt, G.; Caparroz-Assef, S.M.; Bersani-Amado, C.A.; Cuman, R.K.N. Anti-inflammatory and antinociceptive activities A of eugenol essential oil in experimental animal models. Rev. Bras. Farmacogn. 2009, 19, 212–217. [Google Scholar] [CrossRef]
- De Araújo Lopes, A.; da Fonseca, F.N.; Rocha, T.M.; de Freitas, L.B.; Araújo, E.V.O.; Wong, D.V.T.; Leal, L.K.A.M. Eugenol as a promising molecule for the treatment of dermatitis: Antioxidant and anti-inflammatory activities and its nanoformulation. Oxidative Med. Cell. Longev. Clin. Med. Res. 2018, 9, 34–42. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Pereira, F.; Mendes, J.M.; de Oliveira Lima, E. Investigation on mechanism of antifungal activity of eugenol against Trichophyton rubrum. Med. Mycol. 2013, 51, 507–513. [Google Scholar] [CrossRef]
- De Souza Ferreira, S.B.; Slowik, K.M.; de Castro Hoshino, L.V.; Baesso, M.L.; Murdoch, C.; Colley, H.E.; Bruschi, M.L. Mucoadhesive emulgel systems containing curcumin for oral squamous cell carcinoma treatment: From pre-formulation to cytotoxicity in tissue-engineering oral mucosa. Eur. J. Pharm. Sci. 2020, 151, 105372. [Google Scholar] [CrossRef]
- Dehdashtian, A.; Stringer, T.P.; Warren, A.J.; Mu, E.W.; Amirlak, B.; Shahabi, L. Anatomy and Physiology of the Skin. In Melanoma; Springer: Cham, Switzerland, 2018; pp. 15–26. [Google Scholar]
- Dharmalingam, S.R.; Ramamurthy, S.; Chidambaram, K.; Nadaraju, S. Development and validation of UV spectrophotometric method for the estimation of naproxen in bulk and semi-solid formulation. Int. J. Anal. Pharm. Biomed. Sci. 2013, 2, 49–55. [Google Scholar]
- Du, B.; Daniels, V.R.; Vaksman, Z.; Boyd, J.L.; Crady, C.; Putcha, L. Evaluation of physical and chemical changes in pharmaceuticals flown on space missions. AAPS J. 2011, 13, 299–308. [Google Scholar] [CrossRef]
- Elmataeeshy, M.E.; Sokar, M.S.; Bahey-El-Din, M.; Shaker, D.S. Enhanced transdermal permeability of Terbinafine through novel nanoemulgel formulation; Development, in vitro and in vivo characterization. Future J. Pharm. Sci. 2018, 4, 18–28. [Google Scholar] [CrossRef]
- Esmaeili, F.; Rajabnejhad, S.; Partoazar, A.R.; Mehr, S.E.; Faridi-Majidi, R.; Sahebgharani, M.; Amani, A. Anti-inflammatory effects of eugenol nanoemulsion as a topical delivery system. Pharm. Dev. Technol. 2016, 21, 887–893. [Google Scholar] [CrossRef]
- Gaikwad, A.K. Transdermal drug delivery system: Formulation aspects and evaluation. Compr. J. Pharm. Sci. 2013, 1, 1–10. [Google Scholar]
- Gaikwad, D.T.; Jadhav, N.R. Formulation design and evaluation of an emulgel containing Terminalia arjuna bark extract for transdermal delivery. Pharmacogn. Mag. 2018, 14, 249. [Google Scholar] [CrossRef]
- Giuliani, F. The composition, structure, sources, and applications of eugenol. ESSAI 2014, 12, 19. [Google Scholar]
- Gul, R.; Ahmed, N.; Ullah, N.; Khan, M.I.; Elaissari, A.; Rehman, A. Biodegradable ingredient-based emulgel loaded with ketoprofen nanoparticles. AAPS PharmSciTech 2018, 19, 1869–1881. [Google Scholar] [CrossRef]
- Bakhsh, S.; Khan, G.M.; Menaa, F.; Khan, B.A. Parmacokinetic evaluation of ibuprofen controlled release matrix tablets using hydrophilic Eudragit® polymer and co-excipients. Pak. J. Pharm. Sci. 2015, 28, 1745–1755. [Google Scholar]
- Gupta, R.; Gupta, G.D. Formulation development and evaluation of anti-inflammatory potential of Cordia obliqua topical gel on animal model. Pharmacogn. J. 2017, 9, s93–s98. [Google Scholar] [CrossRef]
- Taher, Y.A.; Samud, A.M.; El-Taher, F.E.; Ben-Hussin, G.; Elmezogi, J.S.; Al-Mehdawi, B.F.; Salem, H.A. Experimental evaluation of anti-inflammatory, antinociceptive and antipyretic activities of clove oil inmice. Libyan J. Med. 2015, 10, 425–435. [Google Scholar] [CrossRef]
- Akhtar, N.; Akhtar, N.; Menaa, F.; Alharbi, W.; Alaryani, F.S.S.; Alqahtani, A.M.; Ahmad, F. Fabrication of Ethosomes Containing Tocopherol Acetate to Enhance Transdermal Permeation: In Vitro and Ex Vivo Characterizations. Gels 2022, 8, 335. [Google Scholar] [CrossRef]
- Khan, Z.U.; Razzaq, A.; Khan, A.; Rehman, N.U.; Khan, H.; Khan, T.; Khan, A.U.; Althobaiti, N.A.; Menaa, F.; Iqbal, H.; et al. Physicochemical Characterizations and Pharmacokinetic Evaluation of Pentazocine Solid Lipid Nanoparticles against Inflammatory Pain Model. Pharmaceutics 2022, 14, 409. [Google Scholar] [CrossRef]


| Stability Parameters | Day 0 | Day 1 | Day 2 | Day 7 | Day 14 | Day 28 | |
|---|---|---|---|---|---|---|---|
| Color | A | LY | LY | LY | LY | LY | LY |
| B | LY | LY | LY | LY | LY | LY | |
| C | LY | LY | LY | LY | LY | LY | |
| D | LY | LY | LY | LY | LY | LY | |
| Homogeneity | A | *** | *** | *** | *** | *** | *** |
| B | *** | *** | *** | *** | *** | *** | |
| C | *** | *** | *** | *** | ** | ** | |
| D | *** | *** | *** | ** | ** | ** | |
| Smell | A | -ve | -ve | -ve | -ve | -ve | -ve |
| B | -ve | -ve | -ve | -ve | -ve | -ve | |
| C | -ve | -ve | -ve | -ve | -ve | -ve | |
| D | -ve | -ve | -ve | -ve | -ve | -ve | |
| Phase separation | A | -ve | -ve | -ve | -ve | -ve | -ve |
| B | -ve | -ve | -ve | -ve | -ve | -ve | |
| C | -ve | -ve | -ve | -ve | -ve | -ve | |
| D | -ve | -ve | -ve | -ve | -ve | -ve | |
| Grittiness | A | -ve | -ve | -ve | -ve | -ve | -ve |
| B | -ve | -ve | -ve | -ve | -ve | -ve | |
| C | -ve | -ve | -ve | -ve | -ve | -ve | |
| D | -ve | -ve | -ve | -ve | -ve | -ve | |
| Time (Days) | pH at 8 °C | pH at 25 °C | pH at 40 °C |
|---|---|---|---|
| 0 | 6.10 ± 0.04 | 6.10 ± 0.04 | 6.10 ± 0.04 |
| 1 | 6.08 ± 0.07 | 6.09 ± 0.40 | 6.06 ± 0.94 |
| 2 | 6.07 ± 0.01 | 6.07 ± 0.23 | 6.02 ± 0.36 |
| 7 | 6.03 ± 0.28 | 6.04 ± 0.34 | 5.94 ± 0.68 |
| 14 | 6.03 ± 0.97 | 6.02 ± 0.45 | 5.88 ± 0.24 |
| 28 | 6.01 ± 0.18 | 5.98 ± 0.12 | 5.86 ± 0.38 |
| Time (Days) | Viscosity (mPa) at 8 °C | Viscosity (mPa) at 25 °C | Viscosity (mPa) at 40 °C |
|---|---|---|---|
| 0 | 19,687 ± 0.17 | 19,687 ± 0.17 | 19,687 ± 0.17 |
| 1 | 19676 ± 0.18 | 19,594 ± 0.40 | 19,260 ± 0.94 |
| 2 | 19,650 ± 0.18 | 19,390 ± 0.91 | 19,120 ± 0.32 |
| 7 | 19,638 ± 0.12 | 19,318 ± 0.34 | 18,680 ± 0.68 |
| 14 | 19,630 ± 0.23 | 19,290 ± 0.45 | 18,320 ± 0.28 |
| 28 | 19,622 ± 0.23 | 19,240 ± 0.12 | 18,080 ± 0.40 |
| S.No | Temperature | Spreadability (gm·cm/s) |
|---|---|---|
| 1 | 8 °C | 14.36 ± 0.92 |
| 2 | 25 °C | 16.12 ± 1.36 |
| 3 | 40 °C | 20.08 ± 1.43 |
| S.No | Temperatures | Extrudability (gm/cm2) |
|---|---|---|
| 1 | 8 °C | 20.03± 1.08 |
| 2 | 25 °C | 19.89 ± 0.74 |
| 3 | 40 °C | 19.21 ± 0.53 |
| Drugs | Standard (Mean ± sd) | Sample (Mean ± sd) | Drug Content (Mean ± sd) |
|---|---|---|---|
| Naproxen (Absorbance) | 0.242 ± 0.006 | 0.234 ± 0.01 | 96.69% |
| Eugenol (Area under the peak) | 1,036,891 ± 1.26 | 1,008,324 ± 1.79 | 97.24% |
| Drugs | Zero Order Kinetic | 1st Order Kinetic | Higuchi | Hixson–Crowell | Korsmeyer–Peppas | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| K | R2 | K | R2 | K | R2 | K | R2 | K | R2 | N | |
| Eugenol | 1.29 | 0.98 | 1.46 | 0.59 | 1.14 | 0.99 | 1.0 | 0.991 | 1.14 | 0.99 | 0.71 |
| Naproxen | 1.86 | 0.98 | 1.65 | 0.65 | 1.14 | 0.99 | 1.2 | 0.993 | 1.01 | 0.99 | 0.78 |
| S.No | Drugs | % Drug Retention |
|---|---|---|
| 1 | Naproxen | 6.98 ± 0.24 |
| 2 | Eugenol | 8.52 ± 0.22 |
| Days | Primary Irritation Index | |
|---|---|---|
| Control Group | Test Group | |
| 0 | 0.00 | 0.00 |
| 1 | 0.00 | 0.04 |
| 2 | 0.00 | 0.02 |
| 3 | 0.00 | 0.00 |
| S.No | Ingredients | Quantity (w/w) |
|---|---|---|
| 1 | Naproxen | 10 |
| 2 | Eugenol | 2 |
| 3 | Tween-80 | 10 |
| 4 | PG | 8 |
| 5 | Olive oil | 6 |
| 6 | Ethanol | 4 |
| 7 | Liquid paraffin | 6 |
| 8 | Methyl paraben | 0.03 |
| 9 | Propyl paraben | 0.05 |
| 10 | Sepineo-P600 | 6 |
| 12 | Distilled water q.s | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, B.A.; Ahmad, S.; Khan, M.K.; Hosny, K.M.; Bukhary, D.M.; Iqbal, H.; Murshid, S.S.; Halwani, A.A.; Alissa, M.; Menaa, F. Fabrication and Characterizations of Pharmaceutical Emulgel Co-Loaded with Naproxen-Eugenol for Improved Analgesic and Anti-Inflammatory Effects. Gels 2022, 8, 608. https://doi.org/10.3390/gels8100608
Khan BA, Ahmad S, Khan MK, Hosny KM, Bukhary DM, Iqbal H, Murshid SS, Halwani AA, Alissa M, Menaa F. Fabrication and Characterizations of Pharmaceutical Emulgel Co-Loaded with Naproxen-Eugenol for Improved Analgesic and Anti-Inflammatory Effects. Gels. 2022; 8(10):608. https://doi.org/10.3390/gels8100608
Chicago/Turabian StyleKhan, Barkat Ali, Sajeel Ahmad, Muhammad Khalid Khan, Khaled M. Hosny, Deena M. Bukhary, Haroon Iqbal, Samar S. Murshid, Abdulrahman A. Halwani, Mohammed Alissa, and Farid Menaa. 2022. "Fabrication and Characterizations of Pharmaceutical Emulgel Co-Loaded with Naproxen-Eugenol for Improved Analgesic and Anti-Inflammatory Effects" Gels 8, no. 10: 608. https://doi.org/10.3390/gels8100608
APA StyleKhan, B. A., Ahmad, S., Khan, M. K., Hosny, K. M., Bukhary, D. M., Iqbal, H., Murshid, S. S., Halwani, A. A., Alissa, M., & Menaa, F. (2022). Fabrication and Characterizations of Pharmaceutical Emulgel Co-Loaded with Naproxen-Eugenol for Improved Analgesic and Anti-Inflammatory Effects. Gels, 8(10), 608. https://doi.org/10.3390/gels8100608