Chia (Salvia hispanica L.) a Promising Alternative for Conventional and Gelled Emulsions: Technological and Lipid Structural Characteristics
Abstract
1. Introduction
2. Results and Discussion
2.1. Proximate Analysis
2.2. Thermal Emulsion Stability
2.3. Color and pH Measurements
2.4. Textural Properties
2.5. Structural Analysis
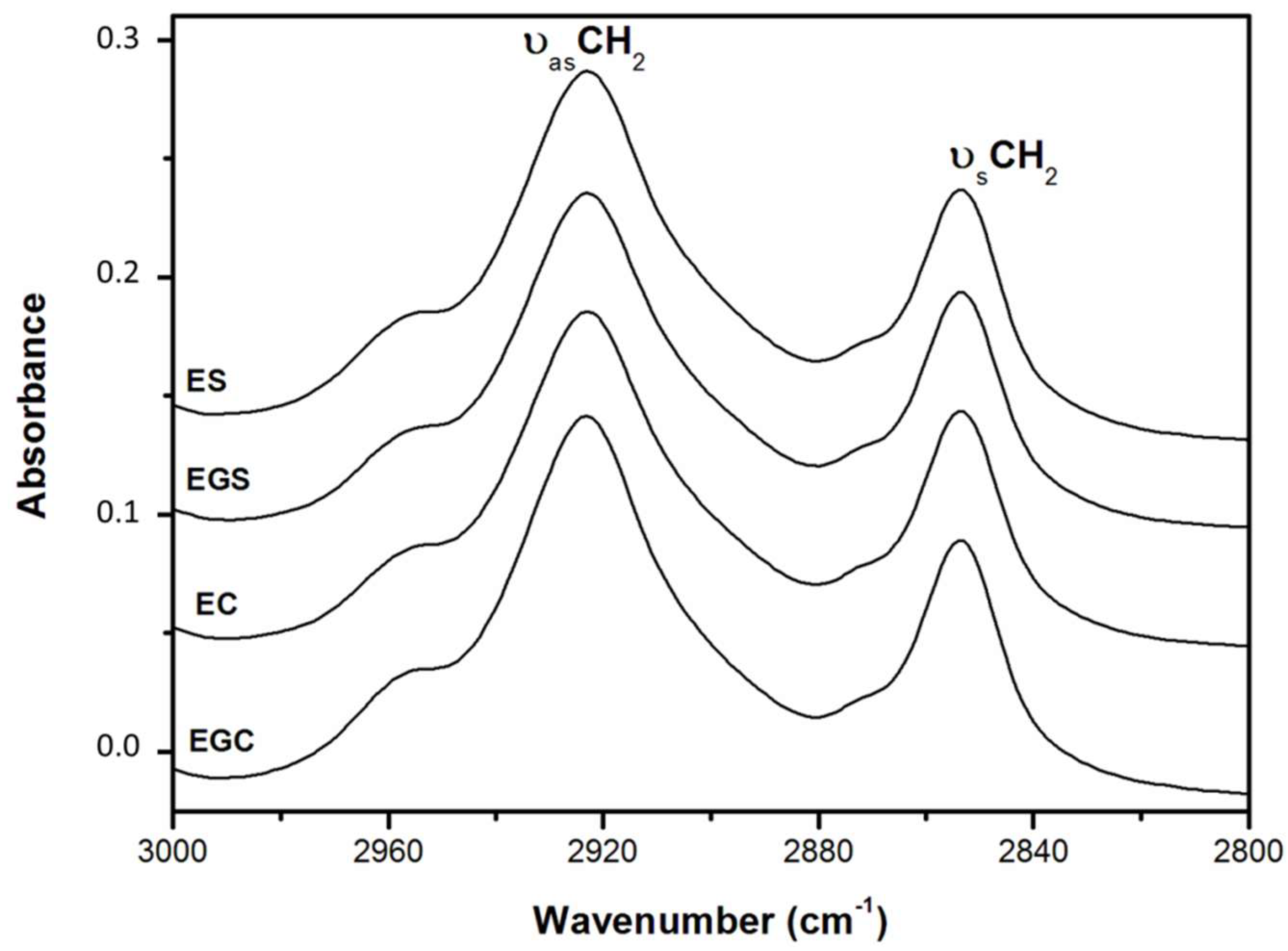
2.5.1. Lipid Structure
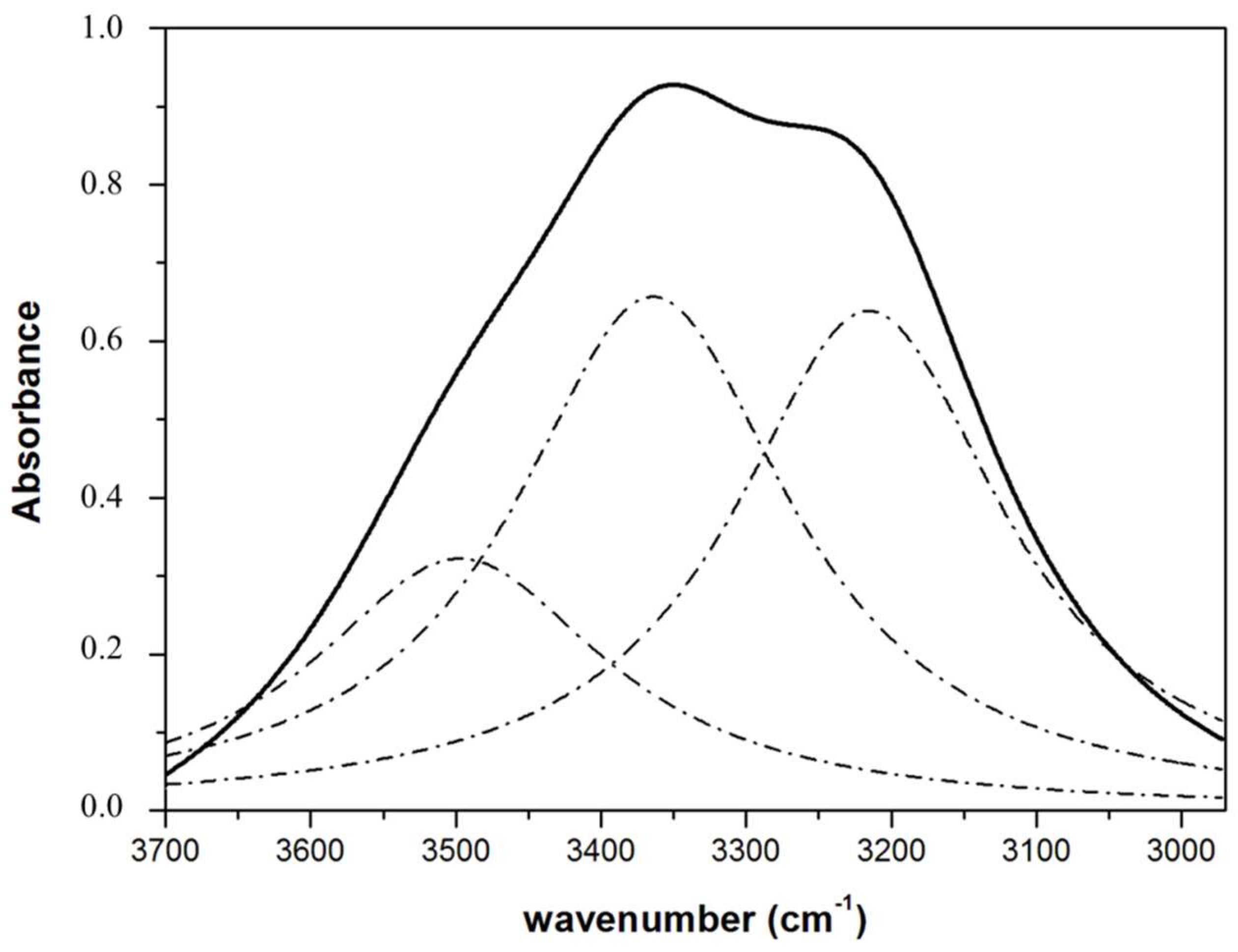
2.5.2. OH Stretching Band
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Preparation of Olive Oil-in-Water Conventional Emulsions and Emulsion Gels
4.3. Proximate Analysis
4.4. Thermal Emulsion Stability
4.5. Color and pH Measurements
4.6. Textural Properties
4.7. Lipid Structural Characteristics
4.7.1. Attenuated Total Reflectance (ATR)-FTIR Spectroscopy
Spectroscopic Measurements
Data Analysis
4.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ayerza, R.; Coates, W. Protein content, oil content and fatty acid profiles as potential criteria to determine the origin of commercially grown chia (Salvia hispanica L.). Ind. Crop. Prod. 2011, 34, 1366–1371. [Google Scholar] [CrossRef]
- Muñoz, L.A.; Cobos, A.; Diaz, O.; Aguilera, J.M. Chia Seed (Salvia hispanica): An Ancient Grain and a New Functional Food. Food Rev. Int. 2013, 29, 394–408. [Google Scholar] [CrossRef]
- Vázquez-Ovando, J.A.; Rosado-Rubio, J.L.; Chel-Guerrero, L.A.; Betancur-Ancona, D.A. Dry processing of chía (Salvia hispanica L.) flour: Chemical characterization of fiber and protein. CyTA-J. Food 2010, 8, 117–127. [Google Scholar] [CrossRef]
- Sandoval-Oliveros, M.R.; Paredes-Lopez, O. Isolation and characterization of proteins from chia seeds (Salvia hispanica L.). J. Agric. Food Chem. 2013, 61, 193–201. [Google Scholar] [CrossRef]
- Segura-Campos, M.R.; Salazar-Vega, I.M.; Chel-Guerrero, L.A.; Betancur-Ancona, D.A. Biological potential of chia (Salvia hispanica L.) protein hydrolysates and their incorporation into functional foods. LWT-Food Sci. Technol. 2013, 50, 723–731. [Google Scholar] [CrossRef]
- Olivos-Lugo, B.L.; Valdivia-Lopez, M.A.; Tecante, A. Thermal and physicochemical properties and nutritional value of the protein fraction of Mexican chia seed (Salvia hispanica L.). Food Sci. Technol. Int. 2010, 16, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Julio, L.M.; Ixtaina, V.Y.; Fernandez, M.; Torres Sanchez, R.M.; Nolasco, S.M.; Tomas, M.C. Development and characterization of functional O/W emulsions with chia seed (Salvia hispanica L.) by-products. J. Food Sci. Technol. 2016, 53, 3206–3214. [Google Scholar] [CrossRef] [PubMed]
- Coorey, R.; Tjoe, A.; Jayasena, V. Gelling properties of chia seed and flour. J. Food Sci. 2014, 79, E859–E866. [Google Scholar] [CrossRef] [PubMed]
- Valdivia-Lopez, M.A.; Tecante, A. Chia (Salvia hispanica): A Review of Native Mexican Seed and its Nutritional and Functional Properties. Adv. Food Nutr. Res. 2015, 75, 53–75. [Google Scholar] [CrossRef]
- Reyes-Caudillo, E.; Tecante, A.; Valdivia-López, M.A. Dietary fibre content and antioxidant activity of phenolic compounds present in Mexican chia (Salvia hispanica L.) seeds. Food Chem. 2008, 107, 656–663. [Google Scholar] [CrossRef]
- Bresson, J.L.; Flynn, A.; Heinonen, M.; Hulshof, K.; Korhonen, H.; Lagiou, P.; Løvik, M.; Marchelli, R.; Martin, A.; Moseley, B.; et al. Opinion on the safety of ‘Chia seeds (Salvia hispanica L.) and ground whole Chia seeds’ as a food ingredient. EFSA J. 2009, 7, 996. [Google Scholar] [CrossRef]
- Jin, F.; Nieman, D.C.; Sha, W.; Xie, G.; Qiu, Y.; Jia, W. Supplementation of milled chia seeds increases plasma ALA and EPA in postmenopausal women. Plant Foods Hum. Nutr. 2012, 67, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Mohd Ali, N.; Yeap, S.K.; Ho, W.Y.; Beh, B.K.; Tan, S.W.; Tan, S.G. The Promising Future of Chia, Salvia hispanica L. J. Biomed. Biotechnol. 2012, 2012, 9. [Google Scholar] [CrossRef]
- Iglesias-Puig, E.; Haros, M. Evaluation of performance of dough and bread incorporating chia (Salvia hispanica L.). Eur. Food Res. Technol. 2013, 237, 865–874. [Google Scholar] [CrossRef]
- Luna Pizarro, P.; Almeida, E.L.; Sammán, N.C.; Chang, Y.K. Evaluation of whole chia (Salvia hispanica L.) flour and hydrogenated vegetable fat in pound cake. LWT-Food Sci. Technol. 2013, 54, 73–79. [Google Scholar] [CrossRef]
- Inglett, G.E.; Chen, D.; Liu, S. Physical properties of sugar cookies containing chia-oat composites. J. Sci. Food Agric. 2014, 94, 3226–3233. [Google Scholar] [CrossRef] [PubMed]
- Coelho, M.S.; Salas-Mellado, M.D.L.M. Effects of substituting chia (Salvia hispanica L.) flour or seeds for wheat flour on the quality of the bread. LWT-Food Sci. Technol. 2015, 60, 729–736. [Google Scholar] [CrossRef]
- Steffolani, E.; Martinez, M.M.; León, A.E.; Gómez, M. Effect of pre-hydration of chia (Salvia hispanica L.), seeds and flour on the quality of wheat flour breads. LWT-Food Sci. Technol. 2015, 61, 401–406. [Google Scholar] [CrossRef]
- Souza, A.H.; Gohara, A.K.; Rotta, E.M.; Chaves, M.A.; Silva, C.M.; Dias, L.F.; Gomes, S.T.; Souza, N.E.; Matsushita, M. Effect of the addition of chia’s by-product on the composition of fatty acids in hamburgers through chemometric methods. J. Sci. Food Agric. 2015, 95, 928–935. [Google Scholar] [CrossRef]
- Pintado, T.; Herrero, A.M.; Jiménez-Colmenero, F.; Ruiz-Capillas, C. Strategies for incorporation of chia (Salvia hispanica L.) in frankfurters as a health-promoting ingredient. Meat Sci. 2016a, 114, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Pintado, T.; Herrero, A.M.; Ruiz-Capillas, C.; Triki, M.; Carmona, P.; Jimenez-Colmenero, F. Effects of emulsion gels containing bioactive compounds on sensorial, technological, and structural properties of frankfurters. Food Sci. Technol. Int. 2016, 22, 132–145. [Google Scholar] [CrossRef] [PubMed]
- Herrero, A.M.; Ruiz-Capillas, C.; Pintado, T.; Carmona, P.; Jimenez-Colmenero, F. Infrared spectroscopy used to determine effects of chia and olive oil incorporation strategies on lipid structure of reduced-fat frankfurters. Food Chem. 2017, 221, 1333–1339. [Google Scholar] [CrossRef] [PubMed]
- Pintado, T.; Ruiz-Capillas, C.; Jimenez-Colmenero, F.; Carmona, P.; Herrero, A.M. Oil-in-water emulsion gels stabilized with chia (Salvia hispanica L.) and cold gelling agents: Technological and infrared spectroscopic characterization. Food Chem. 2015, 185, 470–478. [Google Scholar] [CrossRef]
- Pintado, T.; Herrero, A.M.; Jiménez-Colmenero, F.; Ruiz-Capillas, C. Emulsion gels as potential fat replacers delivering β-glucan and healthy lipid content for food applications. J. Food Sci. Technol. 2016, 53, 4336–4347. [Google Scholar] [CrossRef] [PubMed]
- Capitani, M.I.; Spotorno, V.; Nolasco, S.M.; Tomás, M.C. Physicochemical and functional characterization of by-products from chia (Salvia hispanica L.) seeds of Argentina. LWT-Food Sci. Technol. 2012, 45, 94–102. [Google Scholar] [CrossRef]
- Delgado-Pando, G.; Cofrades, S.; Ruiz-Capillas, C.; Teresa Solas, M.; Jiménez-Colmenero, F. Healthier lipid combination oil-in-water emulsions prepared with various protein systems: An approach for development of functional meat products. Eur. J. Lipid Sci. Technol. 2010, 112, 791–801. [Google Scholar] [CrossRef]
- Ayerza, R.; Coates, W. Composition of chia (Salvia hispanica) grown in six tropical and subtropical ecosystems of South America. Trop. Sci. 2004, 44, 131–135. [Google Scholar] [CrossRef]
- Vazquez-Ovando, A.; Rosado-Rubio, G.; Chel-Guerrero, L.; Betancur-Ancona, D. Physicochemical properties of a fibrous fraction from chia (Salvia hispanica L.). LWT-Food Sci. Technol. 2009, 42, 168–173. [Google Scholar] [CrossRef]
- Herrero, A.M.; Carmona, P.; Pintado, T.; Jiménez-Colmenero, F.; Ruíz-Capillas, C. Infrared spectroscopic analysis of structural features and interactions in olive oil-in-water emulsions stabilized with soy protein. Food Res. Int. 2011, 44, 360–366. [Google Scholar] [CrossRef]
- Sato, A.C.K.; Moraes, K.E.F.P.; Cunha, R.L. Development of gelled emulsions with improved oxidative and pH stability. Food Hydrocoll. 2014, 34, 184–192. [Google Scholar] [CrossRef]
- Jiménez-Colmenero, F.; Herrero, A.; Pintado, T.; Solas, M.T.; Ruiz-Capillas, C. Influence of emulsified olive oil stabilizing system used for pork backfat replacement in frankfurters. Food Res. Int. 2010, 43, 2068–2076. [Google Scholar] [CrossRef]
- Dickinson, E. Stabilising emulsion-based colloidal structures with mixed food ingredients. J. Sci. Food Agric. 2013, 93, 710–721. [Google Scholar] [CrossRef]
- Roopa, B.S.; Bhattacharya, S. Texturized alginate gels: Screening experiments to identify the important variables on gel formation and their properties. LWT-Food Sci. Technol. 2010, 43, 1403–1408. [Google Scholar] [CrossRef]
- Capitani, M.I.; Nolasco, S.M.; Tomás, M.C. Stability of oil-in-water (O/W) emulsions with chia (Salvia hispanica L.) mucilage. Food Hydrocoll. 2016, 61, 537–546. [Google Scholar] [CrossRef]
- Timilsena, Y.P.; Adhikari, R.; Barrow, C.J.; Adhikari, B. Physicochemical and functional properties of protein isolate produced from Australian chia seeds. Food Chem. 2016, 212, 648–656. [Google Scholar] [CrossRef] [PubMed]
- Guillen, N.D.; Cabo, N. Characterization of edible oils and lard by fourier transform infrared spectroscopy. Relationships between composition and frequency of concrete bands in the fingerprint region. J. Ameroil. Chem. Soc. 1997, 74:10, 1281–1286. [Google Scholar] [CrossRef]
- Herrero, A.M.; Carmona, P.; Pintado, T.; Jiménez-Colmenero, F.; Ruíz-Capillas, C. Olive oil-in-water emulsions stabilized with caseinate: Elucidation of protein–lipid interactions by infrared spectroscopy. Food Hydrocoll. 2011, 25, 12–18. [Google Scholar] [CrossRef]
- Fraile, M.V.; Patrón-Gallardo, B.; López-Rodríguez, G.; Carmona, P. FT-IR study of multilamellar lipid dispersions containing cholesteryl linoleate and dipalmitoylphosphatidylcholine. Chem. Phys. Lipids 1999, 97, 119–128. [Google Scholar] [CrossRef]
- Kodati, V.R.; El-Jastimi, R.; Lafleur, M. Contribution of the Intermolecular Coupling and Librotorsional Mobility in the Methylene Stretching Modes in the Infrared Spectra of Acyl Chains. J. Phys. Chem. 1994, 98, 12191–12197. [Google Scholar] [CrossRef]
- Brubach, J.B.; Mermet, A.; Filabozzi, A.; Gerschel, A.; Lairez, D.; Krafft, M.P.; Roy, P. Dependence of Water Dynamics upon Confinement Size. J. Phys. Chem. B 2001, 105, 430–435. [Google Scholar] [CrossRef]
- Matsumura, Y.; Kang, I.J.; Sakamoto, H.; Motoki, M.; Mori, T. Filler effects of oil droplets on the viscoelastic properties of emulsion gels. Food Hydrocoll. 1993, 7, 227–240. [Google Scholar] [CrossRef]
- McClemens, D.J.; Monahan, F.J.; Kinsella, J.E. Effects of emulsion droplets on the rheology of whey protein isolate gels. J. Texture Stud. 1993, 24, 411–422. [Google Scholar] [CrossRef]
- Kim, K.H.; Gohtani, S.; Yamano, Y. Effects of oil droplets on physical and sensory properties of O/W emulsion agar gel. J. Texture Stud. 1996, 27, 655–670. [Google Scholar] [CrossRef]
- Sok Line, V.L.; Remondetto, G.E.; Subirade, M. Cold gelation of β-lactoglobulin oil-in-water emulsions. Food Hydrocoll. 2005, 19, 269–278. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method for total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Colmenero, F.; Carballo, J.; Solas, M.T. The effect of use of freeze-thawed pork on the properties of Bologna sausages with two fat levels. Int. J. Food Sci. Technol. 1995, 30, 335–345. [Google Scholar] [CrossRef]
- Lavialle, F.; Adams, R.G.; Levin, I.W. Infrared spectroscopic study of the secondary structure of melittin in water, 2-chloroethanol, and phospholipid bilayer dispersions. Biochemistry 1982, 21, 2305–2312. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.S.; Steer, C.J.; Levin, I.W. Infrared spectroscopic study of the pH-dependent secondary structure of brain clathrin. Biochemistry 1984, 23, 625–631. [Google Scholar] [CrossRef]

| Samples 2 | SPI 1 | Chia Flour | Water | Olive Oil | Gelling Agent | ||
|---|---|---|---|---|---|---|---|
| Sodium Alginate | CaSO4 | Sodium Pyrophosphate | |||||
| ES | 3.3 | 56.7 | 40 | ||||
| EC | 15.0 | 45.0 | 40 | ||||
| EGS | 3.3 | 54.7 | 40 | 0.73 | 0.73 | 0.54 | |
| EGC | 15.0 | 43.0 | 40 | 0.73 | 0.73 | 0.54 | |
| Samples 1 | Moisture | Ash | Protein | Fat |
|---|---|---|---|---|
| ES | 57.09 ± 0.43 a | 0.17 ± 0.01 d | 2.40 ± 0.19 a | 37.31 ± 1.57 a |
| EC | 45.54 ± 0.16 c | 0.64 ± 0.05 c | 2.41 ± 0.01 a | 38.93 ± 1.05 a |
| EGS | 54.65 ± 0.51 b | 1.04 ± 0.01 b | 2.42 ± 0.16 a | 38.19 ± 1.26 a |
| EGC | 43.73 ± 0.15 d | 1.55 ± 0.05 a | 2.39 ± 0.02 a | 40.69 ± 1.20 a |
| Samples 1 | L* | a* | b* | PF | GS |
|---|---|---|---|---|---|
| ES | 57.16 ± 0.29 d | −0.15 ± 0.06 d | 22.94 ± 0.44 b | Nd ** | Nd ** |
| EC | 61.72 ± 1.00 c | 0.10 ± 0.03 c | 9.51 ± 0.44 d | 0.13 ± 0.01 c | 0.25 ± 0.02 c |
| EGS | 76.62 ± 0.47 a | 0.38 ± 0.04 b | 26.14 ± 0.20 a | 0.34 ± 0.02 b | 0.79 ± 0.07 b |
| EGC | 64.56 ± 0.97 b | 1.69 ± 0.14 a | 11.47 ± 0.39 c | 1.43 ± 0.07 a | 3.12 ± 0.40 a |
| Samples 1 | Half-Bandwidth 2923 cm−1 (νasCH2) | Half-Bandwidth 2854 cm−1 (νsCH2) | Area 3220 cm−1 Band |
|---|---|---|---|
| Olive oil | 27.5 ± 0.1 c | 16.2 ± 0.2 a | - |
| Pure water | - | - | 0.427 ± 0.004 a |
| ES | 30.1 ± 0.2 a | 16.5 ± 0.3 a | 0.382 ± 0.002 b |
| EC | 31.3 ± 0.3 a | 16.5 ± 0.5 a | 0.379 ± 0.004 b |
| EGS | 28.9 ± 0.1 b | 16.9 ± 0.2 a | 0.365 ± 0.001 c |
| EGC | 29.3 ± 0.2 b | 16.4 ± 0.4 a | 0.343 ± 0.001 d |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muñoz-González, I.; Merino-Álvarez, E.; Salvador, M.; Pintado, T.; Ruiz-Capillas, C.; Jiménez-Colmenero, F.; Herrero, A.M. Chia (Salvia hispanica L.) a Promising Alternative for Conventional and Gelled Emulsions: Technological and Lipid Structural Characteristics. Gels 2019, 5, 19. https://doi.org/10.3390/gels5020019
Muñoz-González I, Merino-Álvarez E, Salvador M, Pintado T, Ruiz-Capillas C, Jiménez-Colmenero F, Herrero AM. Chia (Salvia hispanica L.) a Promising Alternative for Conventional and Gelled Emulsions: Technological and Lipid Structural Characteristics. Gels. 2019; 5(2):19. https://doi.org/10.3390/gels5020019
Chicago/Turabian StyleMuñoz-González, Irene, Esther Merino-Álvarez, Marina Salvador, Tatiana Pintado, Claudia Ruiz-Capillas, Francisco Jiménez-Colmenero, and Ana M. Herrero. 2019. "Chia (Salvia hispanica L.) a Promising Alternative for Conventional and Gelled Emulsions: Technological and Lipid Structural Characteristics" Gels 5, no. 2: 19. https://doi.org/10.3390/gels5020019
APA StyleMuñoz-González, I., Merino-Álvarez, E., Salvador, M., Pintado, T., Ruiz-Capillas, C., Jiménez-Colmenero, F., & Herrero, A. M. (2019). Chia (Salvia hispanica L.) a Promising Alternative for Conventional and Gelled Emulsions: Technological and Lipid Structural Characteristics. Gels, 5(2), 19. https://doi.org/10.3390/gels5020019







