Antimicrobial Sol–Gel Glassy Surfaces for Modification of Dental Implant Abutments to Reduce Microbial Adhesion
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimen Preparation
- Ti6Al4V: Uncoated control group;
- G: Coated group with only glassy coating;
- GAg: Coated group with a mixture of the G+ 2% Ag;
- GAgCu: Coated group with a mixture of the G+ 1% Ag+ 1% Cu.
2.2. Synthesis and Application of the Glassy Coatings
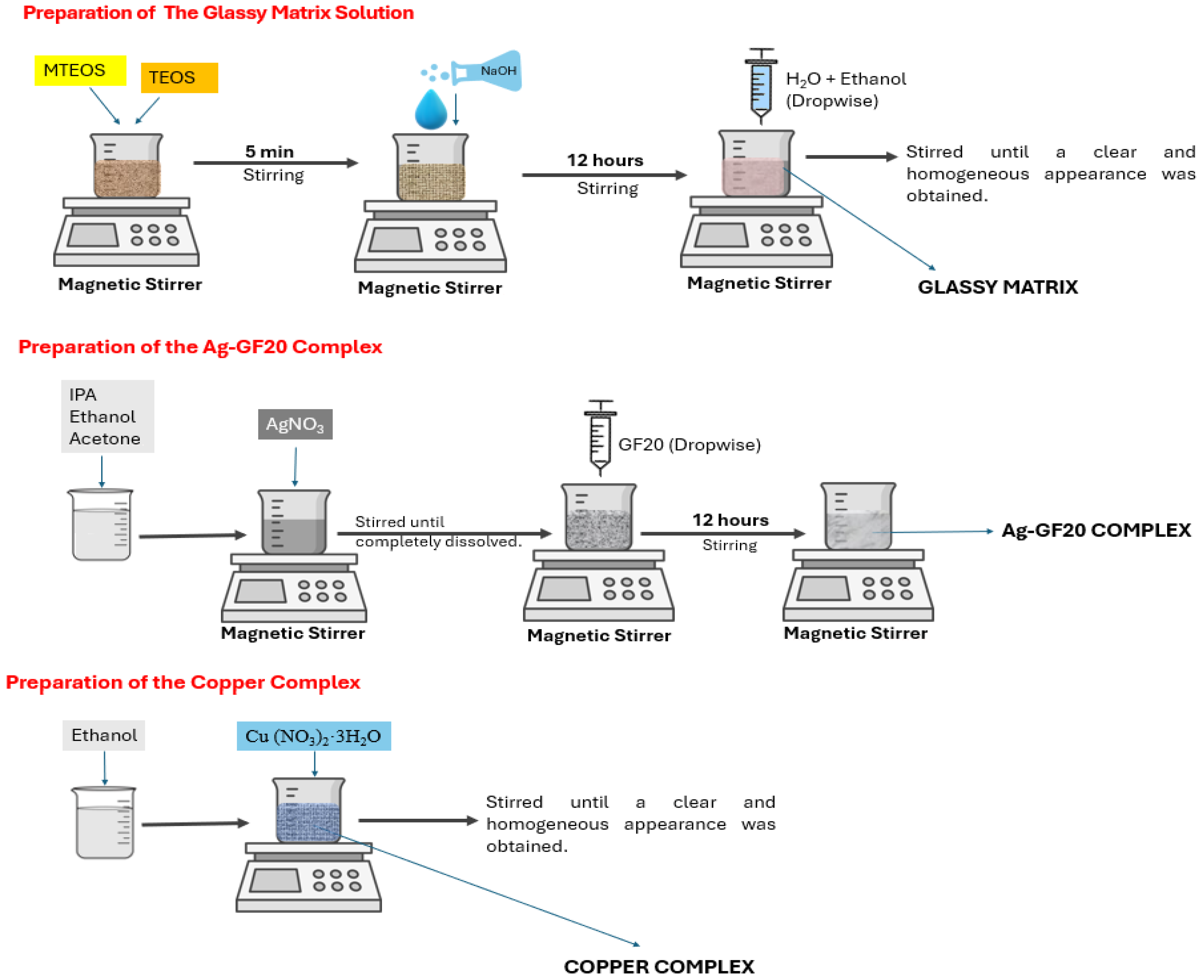
2.2.1. Preparation of the Glassy Matrix Solution
2.2.2. Preparation of the Silver-GF20 Complex and the Copper Complex
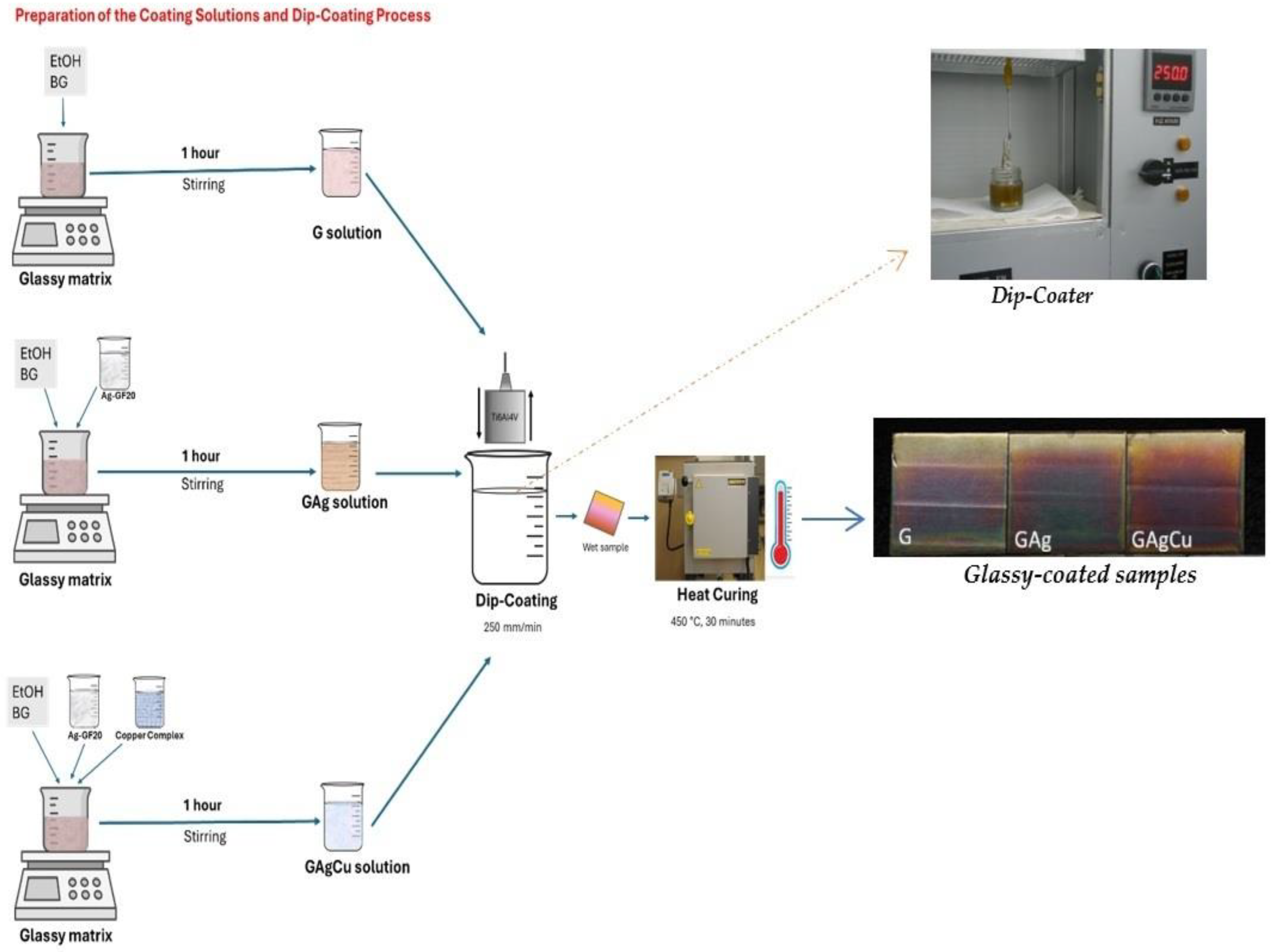
2.2.3. Preparation of the Coating Solutions
2.2.4. Dip-Coating Process
2.3. Thermal Aging Process
2.4. Mechanical and Surface Characteristics of the Coatings
2.4.1. Measurement of the Adhesion Strength
2.4.2. Measurement of the Contact Angle
2.4.3. Measurement of Surface Roughness
2.4.4. Measurement of Surface Hardness
2.5. Microbial Adhesion Tests
Microbial Assay and Fluorescence Analysis
2.6. Cytotoxicity Tests
2.6.1. Cell Cultivation
2.6.2. Agar Diffusion Test
2.6.3. MTT Assay
2.7. Statistical Analyses
3. Results
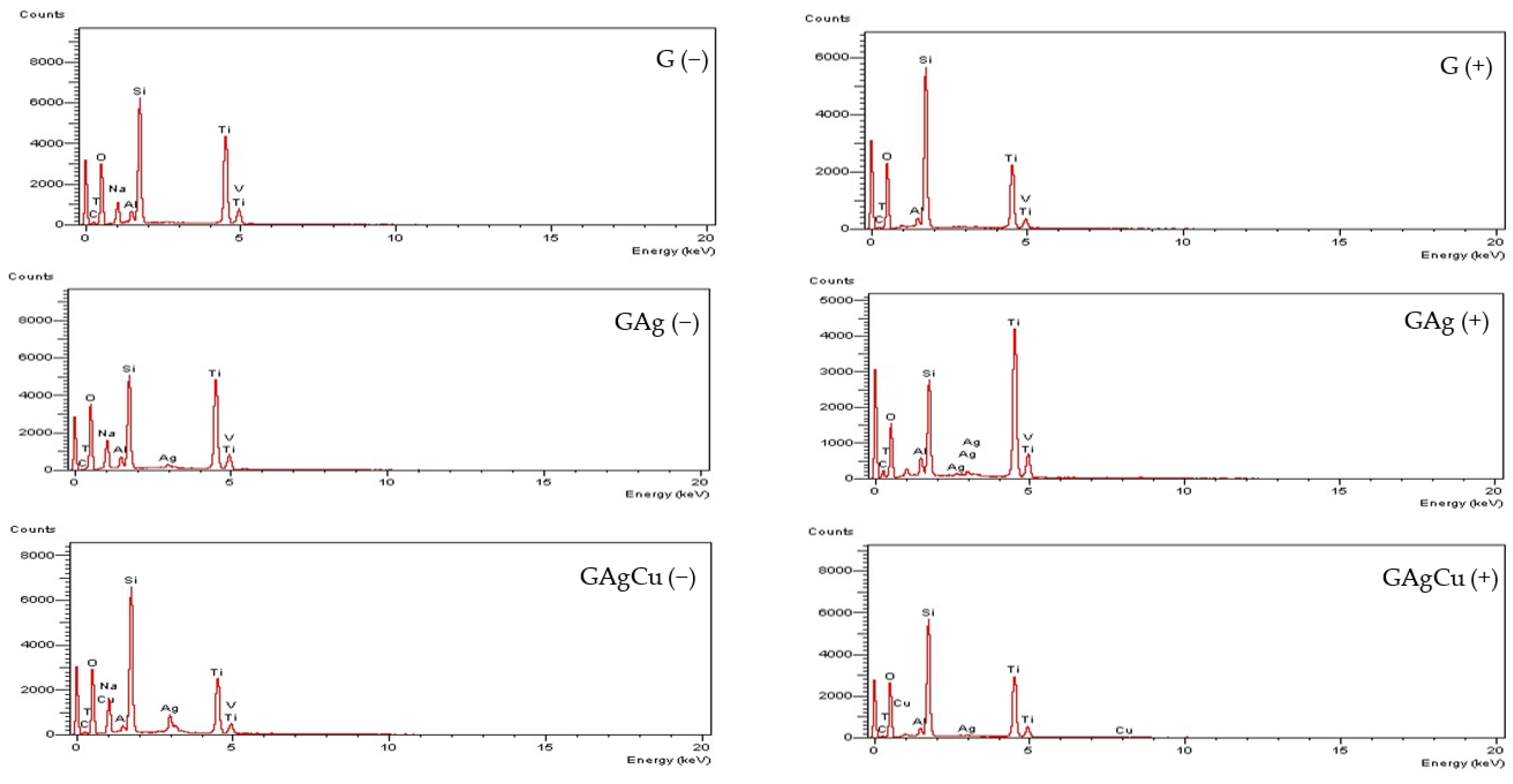
3.1. Surface Morphology and Elemental Analysis
3.2. Mechanical Test Results
3.3. Microbial Adhesion Test Results
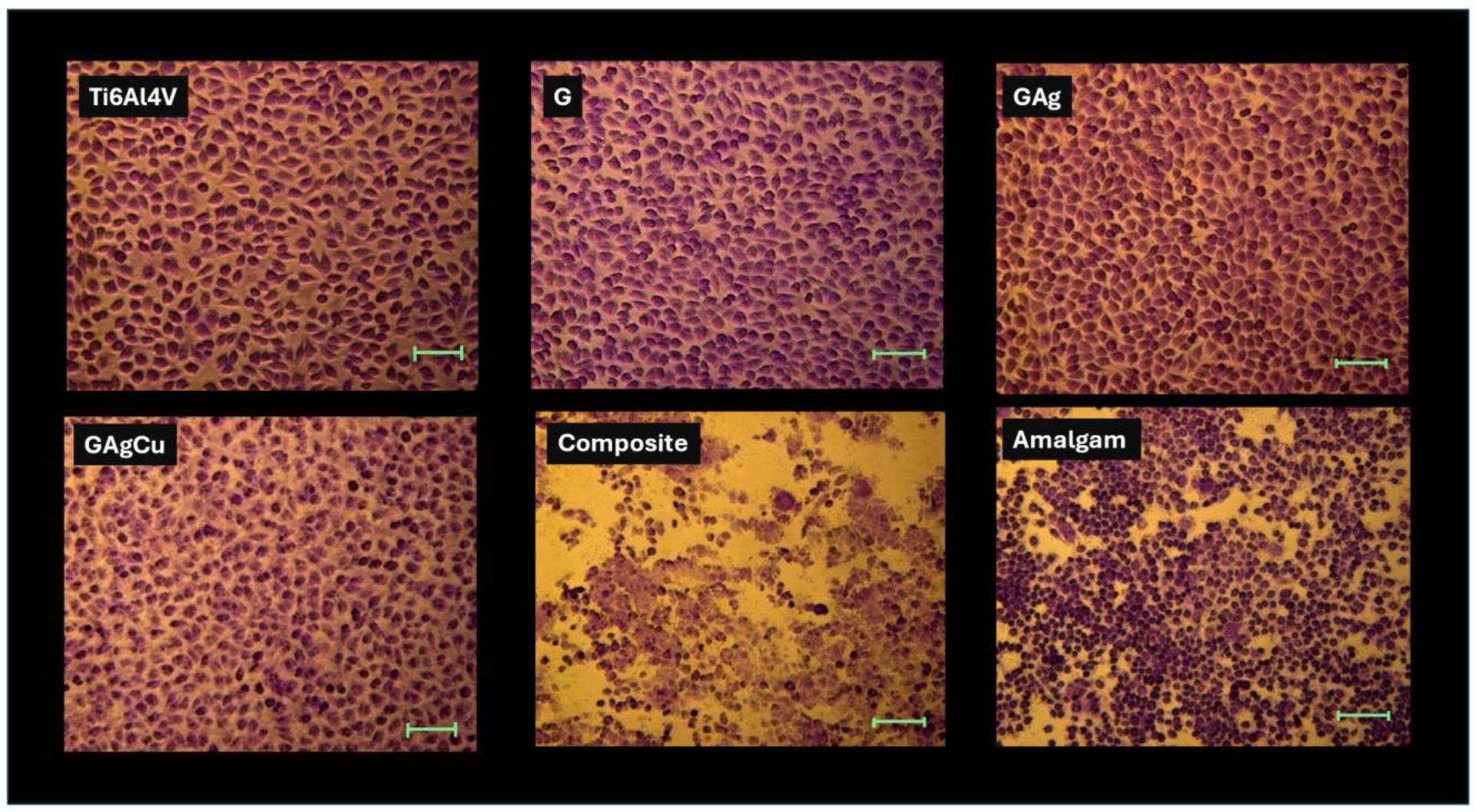
3.4. Cytotoxicity Test Results
3.4.1. Agar Diffusion Test Results
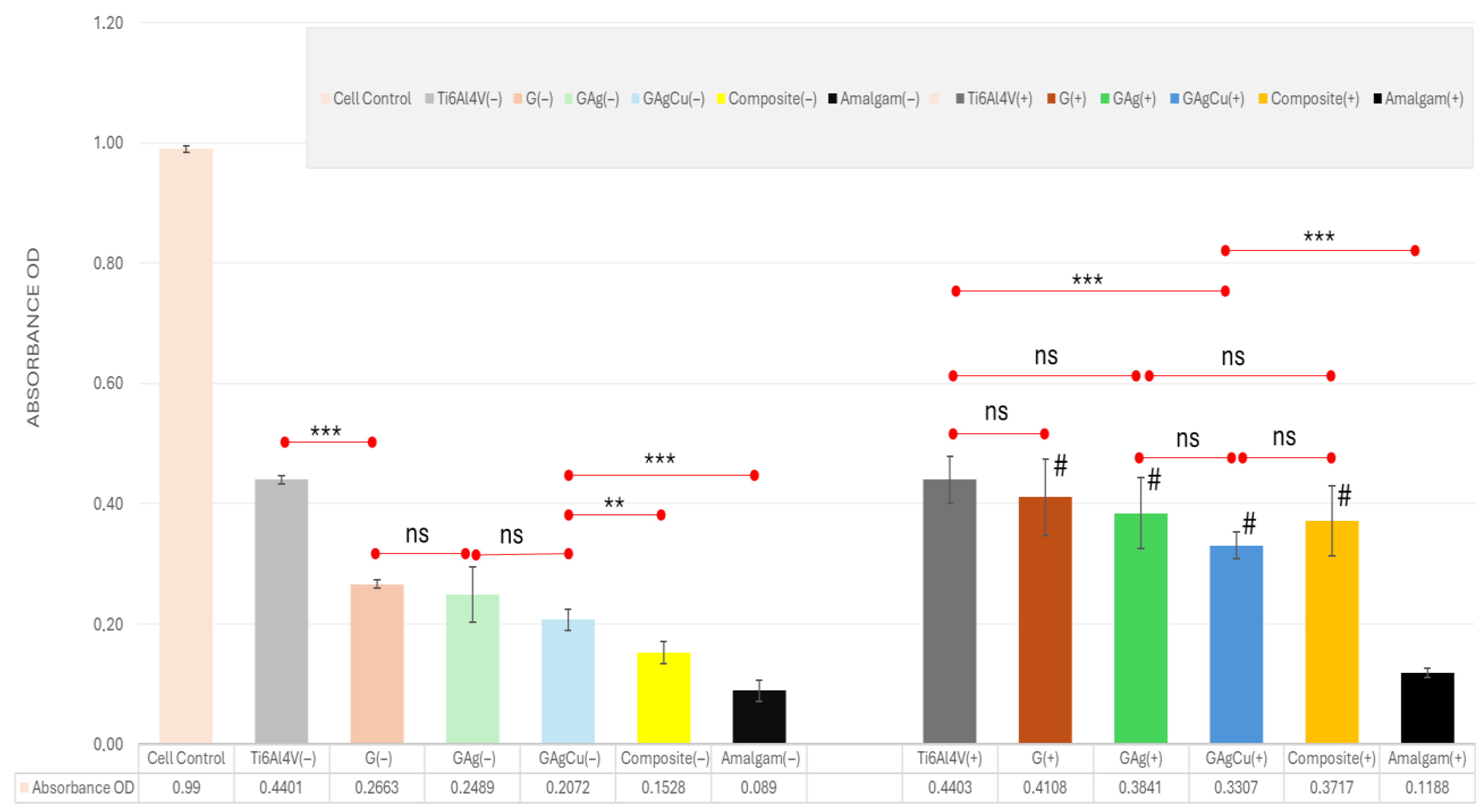
3.4.2. MTT Assay Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviations | Full Form |
| MTEOS | Methyltriethoxysilane |
| TEOS | Tetraethoxysilane |
| NaOH | Sodium hydroxide |
| Al2O3 | Aluminum oxide |
| EtOH | Ethanol |
| BG | Butyl glycol |
| Ag | Silver |
| Cu | Copper |
| DNA | Deoxyribonucleic acid |
| ASTM | American Society for Testing and Materials |
| ISO | International Organization for Standardization |
| RCF | Relative centrifugal force |
| GAM | Gifu Anaerobic Medium |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| DMEM | Dulbecco’s Modified Eagle Medium |
| SEM | Scanning Electron Microscopy |
| EDX | Energy Dispersive X-ray Spectroscopy |
| ATCC | American Type Culture Collection |
| DSM | Deutsche Sammlung von Mikroorganismen und Zellkulturen |
References
- Zhao, L.; Chu, P.K.; Zhang, Y.; Wu, Z. Antibacterial coatings on titanium implants. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 91, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Hauser-Gerspach, I.; Kulik, E.M.; Weiger, R.; Decker, E.M.; Von Ohle, C.; Meyer, J. Adhesion of Streptococcus sanguinis to dental implant and restorative materials in vitro. Dent. Mater. J. 2007, 26, 361–366. [Google Scholar] [CrossRef]
- Sardin, S.; Morrier, J.J.; Benay, G.; Barsotti, O. In vitro streptococcal adherence on prosthetic and implant materials. Interactions with physicochemical surface properties. J. Oral Rehabil. 2004, 31, 140–148. [Google Scholar] [CrossRef]
- Salihoglu, U.; Boynuegri, D.; Engin, D.; Duman, A.N.; Gokalp, P.; Balos, K. Bacterial adhesion and colonization differences between zirconium oxide and titanium alloys: An in vivo human study. Int. J. Oral Maxillofac. Implant. 2011, 26, 101–107. [Google Scholar]
- Scarano, A.; Piattelli, M.; Vrespa, G.; Caputi, S.; Piattelli, A. Bacterial adhesion on titanium nitride-coated and uncoated implants: An in vivo human study. J. Oral Implant. 2003, 29, 80–85. [Google Scholar] [CrossRef]
- Abrahamsson, I.; Berglundh, T.; Lindhe, J. Soft tissue response to plaque formation at different implant systems. A comparative study in the dog. Clin. Oral Implant. Res. 1998, 9, 73–79. [Google Scholar] [CrossRef]
- Degidi, M.; Artese, L.; Scarano, A.; Perrotti, V.; Gehrke, P.; Piattelli, A. Inflammatory Infiltrate, Microvessel Density, Nitric Oxide Synthase Expression, Vascular Endothelial Growth Factor Expression, and Proliferative Activity in Peri-Implant Soft Tissues Around Titanium and Zirconium Oxide Healing Caps. J. Periodontol. 2006, 77, 73–80. [Google Scholar] [CrossRef]
- Charyeva, O.; Altynbekov, K.; Zhartybaev, R.; Sabdanaliev, A. Long-term dental implant success and survival—A clinical study after an observation period up to 6 years. Swed. Dent. J. 2012, 36, 1–6. [Google Scholar]
- Myshin, H.L.; Wiens, J.P. Factors affecting soft tissue around dental implants: A review of the literature. J. Prosthet. Dent. 2005, 94, 440–444. [Google Scholar] [CrossRef]
- Cortés-Acha, B.; Figueiredo, R.; Blanc, V.; Soler-Ollé, A.; León, R.; Valmaseda-Castellón, E. Development and viability of biofilms grown on experimental abutments mimicking dental implants: An in vivo model. Med. Oral Patol. Oral Cir. Bucal 2019, 24, E511–E517. [Google Scholar] [CrossRef]
- Corvino, E.; Pesce, P.; Mura, R.; Marcano, E.; Canullo, L. Influence of Modified Titanium Abutment Surface on Peri-implant Soft Tissue Behavior: A Systematic Review of In Vitro Studies. Int. J. Oral Maxillofac. Implant. 2020, 35, 503–519. [Google Scholar] [CrossRef] [PubMed]
- Canullo, L.; Menini, M.; Santori, G.; Rakic, M.; Sculean, A.; Pesce, P. Titanium abutment surface modifications and peri-implant tissue behavior: A systematic review and meta-analysis. Clin. Oral Investig. 2020, 24, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Ewald, A.; Glückermann, S.K.; Thull, R.; Gbureck, U. Antimicrobial titanium/silver PVD coatings on titanium. Biomed. Eng. Online 2006, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Cao, H.; Zhao, Y.; Jin, G.; Cheng, M.; Wang, J.; Jiang, Y.; An, Z.; Zhang, X.; Liu, X. Antimicrobial and Osteogenic Properties of Silver-Ion-Implanted Stainless Steel. ACS Appl. Mater. Interfaces 2015, 7, 10785–10794. [Google Scholar] [CrossRef]
- Yoshinari, M.; Oda, Y.; Kato, T.; Okuda, K.; Hirayama, A. Influence of surface modifications to titanium on oral bacterial adhesion in vitro. J. Biomed. Mater. Res. 2000, 52, 388–394. [Google Scholar] [CrossRef]
- Chen, Q.; Li, W.; Goudouri, O.M.; Ding, Y.; Cabanas-Polo, S.; Boccaccini, A.R. Electrophoretic deposition of antibiotic loaded PHBV microsphere-alginate composite coating with controlled delivery potential. Colloids Surf. B Biointerfaces 2015, 130, 199–206. [Google Scholar] [CrossRef]
- Noda, I.; Miyaji, F.; Ando, Y.; Miyamoto, H.; Shimazaki, T.; Yonekura, Y.; Miyazaki, M.; Mawatari, M.; Hotokebuchi, T. Development of novel thermal sprayed antibacterial coating and evaluation of release properties of silver ions. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 89, 456–465. [Google Scholar] [CrossRef]
- Del Curto, B.; Brunella, M.; Giordano, C.; Pedeferri, M.; Valtulina, V.; Visai, L.; Cigada, A. Decreased bacterial adhesion to surface-treated titanium. Int. J. Artif. Organs 2005, 28, 718–730. [Google Scholar] [CrossRef]
- Unosson, E.; Rodriguez, D.; Welch, K.; Engqvist, H. Reactive combinatorial synthesis and characterization of a gradient Ag-Ti oxide thin film with antibacterial properties. Acta Biomater. 2015, 11, 503–510. [Google Scholar] [CrossRef]
- Sanchez, C.; Livage, J. Sol-gel chemistry from metal alkoxide precursors. New J. Chem. 1990, 14, 513–521. [Google Scholar]
- Advincula, M.C.; Petersen, D.; Rahemtulla, F.; Advincula, R.; Lemons, J.E. Surface analysis and biocorrosion properties of nanostructured surface sol-gel coatings on Ti6Al4V titanium alloy implants. J. Biomed. Mater. Res. Part B Appl. Biomater. 2007, 80, 107–120. [Google Scholar] [CrossRef]
- Pilotek, S.; Schmidt, H.K. Wettability of microstructured hydrophobic sol-gel coatings. J. Sol-Gel Sci. Technol. 2003, 26, 789–792. [Google Scholar] [CrossRef]
- Filiaggi, M.J.; Pilliar, R.M.; Yakubovich, R.; Shapiro, G. Evaluating sol-gel ceramic thin films for metal implant applications. I. Processing and structure of zirconia films on Ti-6Al-4V. J. Biomed. Mater. Res. 1996, 33, 225–238. [Google Scholar] [CrossRef]
- Kiraz, N.; Kesmez, Ö.; Burunkaya, E.; Budama, L.; Acar, B.; Asiltürk, M.; Çamurlu, H.E.; Arpaç, E. Antibacterial glass films prepared on metal surfaces by Sol-gel method. J. Sol-Gel Sci. Technol. 2010, 56, 227–235. [Google Scholar] [CrossRef]
- Lung, C.Y.K.; Kukk, E.; Matinlinna, J.P. The effect of silica-coating by sol-gel process on resin-zirconia bonding. Dent. Mater. J. 2013, 32, 165–172. [Google Scholar] [CrossRef]
- Kiomarsi, N.; Saburian, P.; Chiniforush, N.; Karazifard, M.J.; Hashemikamangar, S.S. Effect of thermocycling and surface treatment on repair bond strength of composite. J. Clin. Exp. Dent. 2017, 9, e945–e951. [Google Scholar] [CrossRef]
- Deste Gökay, G.; Oyar, P.; Durkan, R. Impact of various aging treatments on the microhardness and surface roughness of CAD-CAM monolithic restorative materials. J. Prosthodont. 2024. [Google Scholar] [CrossRef]
- Lange, A.; Grzenia, A.; Wierzbicki, M.; Strojny-Cieslak, B.; Kalińska, A.; Gołębiewski, M.; Radzikowski, D.; Sawosz, E.; Jaworski, S. Silver and copper nanoparticles inhibit biofilm formation by mastitis pathogens. Animals 2021, 11, 1884. [Google Scholar] [CrossRef]
- Fahmy, H.M.; Mosleh, A.M.; Elghany, A.A.; Shams-Eldin, E.; Abu Serea, E.S.; Ali, S.A.; Shalan, A.E. Coated silver nanoparticles: Synthesis, cytotoxicity, and optical properties. RSC Adv. 2019, 9, 20118–20136. [Google Scholar] [CrossRef]
- Ciołek, L.; Karaś, J.; Olszyna, A.; Zaczyńska, E.; Czarny, A.; Zywicka, B. In vitro studies of antibacterial activity of sol-gel bioglass containing silver. Eur. Cell Mater. 2011, 21, 45. [Google Scholar]
- Ma, X.; Zhou, S.; Xu, X.; Du, Q. Copper-containing nanoparticles: Mechanism of antimicrobial effect and application in dentistry-a narrative review. Front. Surg. 2022, 9, 905892. [Google Scholar] [CrossRef] [PubMed]
- Giti, R.; Zomorodian, K.; Firouzmandi, M.; Zareshahrabadi, Z.; Rahmannasab, S. Antimicrobial Activity of Thermocycled Polymethyl Methacrylate Resin Reinforced with Titanium Dioxide and Copper Oxide Nanoparticles. Int. J. Dent. 2021, 2021, 6690806. [Google Scholar] [CrossRef] [PubMed]
- Gawlik-Maj, M.; Babczyńska, A.; Gerber, H.; Kotuła, J.; Sobieszczańska, B.; Sarul, M. Cytotoxicity of Silver-Containing Coatings Used in Dentistry, a Systematic Review. Coatings 2022, 12, 1338. [Google Scholar] [CrossRef]
- Tsukamoto, M.; Miyamoto, H.; Ando, Y.; Noda, I.; Eto, S.; Akiyama, T.; Yonekura, Y.; Sonohata, M.; Mawatari, M. Acute and subacute toxicity in vivo of thermal-sprayed silver containing hydroxyapatite coating in rat tibia. BioMed Res. Int. 2014, 2014, 902343. [Google Scholar] [CrossRef]
- ASTM-F136; Standard Specification for Wrought Ti6Al4V-ELI (Extra Low Interstitial) Alloy for Surgical Implant Applications. American Society for Testing and Materials International: West Conshohocken, PA, USA, 2021.
- ISO 5832-3; Implants for Surgery—Metallic Materials—Part 3: Wrought Titanium 6-Aluminum 4-Vanadium Alloy. International Organization for Standardization: Geneva, Switzerland, 2021.
- Quirynen, M.; Bollen, C.M.L. The influence of surface roughness and surface-free energy on supra- and subgingival plaque formation in man: A review of the literature. J. Clin. Periodontol. 1995, 22, 1–14. [Google Scholar] [CrossRef]
- Arslan, S.; Balkaya, H.; Avcı, B.; Doğan, A. The Effect of Different Adhesive Resins, Composite Resins and Thermal Cycling on the Repair Bond Strength of Bulkfill Composite Resin. Meandros Med. Dent. j. 2022, 23, 31–39. [Google Scholar] [CrossRef]
- ASTM D3359; Standard Test Methods for Rating Adhesion by Tape Test. American Society for Testing and Materials International International: West Conshohocken, PA, USA, 2017.
- Buergers, R.; Rosentritt, M.; Handel, G. Bacterial adhesion of Streptococcus mutans to provisional fixed prosthodontic material. J. Prosthet. Dent. 2007, 98, 461–469. [Google Scholar] [CrossRef]
- Abidzina, V.; Deliloglu-Gürhan, I.; Özdal-Kurt, F.; Sen, B.; Tereshko, I.; Elkin, I.; Budak, S.; Muntele, C.; Ila, D. Cell adhesion study of the titanium alloys exposed to glow discharge. Nucl. Instrum. Methods Phys. Res. B 2007, 261, 624–626. [Google Scholar] [CrossRef]
- ISO 10993-5; Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity. International Organization for Standardization: Geneva, Switzerland, 2009.
- Kurt, O.; Özdal-Kurt, F.; Tuğlu, M.I.; Akçora, C.M. The cytotoxic, neurotoxic, apoptotic and antiproliferative activities of extracts of some marine algae on the MCF-7 cell line. Biotech. Histochem. 2014, 89, 568–576. [Google Scholar] [CrossRef]
- Braem, A.; Mattheys, T.; Neirinck, B.; Čeh, M.; Novak, S.; Schrooten, J.; Van der Biest, O.; Vleugels, J. Bioactive glass-ceramic coated titanium implants prepared by electrophoretic deposition. Mater. Sci. Eng. C 2012, 32, 2267–2273. [Google Scholar] [CrossRef]
- Brunello, G.; Elsayed, H.; Biasetto, L. Bioactive glass and silicate-based ceramic coatings on metallic implants: Open challenge or outdated topic? Materials 2019, 12, 2929. [Google Scholar] [CrossRef]
- Kongsuwan, P.; Brandal, G.; Yao, Y.L. Laser induced porosity and crystallinity modification of a bioactive glass coating on titanium substrates. J. Manuf. Sci. Eng. Trans. ASME 2015, 137, 031004. [Google Scholar] [CrossRef]
- López-Píriz, R.; Solá-Linares, E.; Granizo, J.J.; Díaz-Güemes, I.; Enciso, S.; Bartolomé, J.F.; Cabal, B.; Esteban-Tejeda, L.; Torrecillas, R.; Moya, J.S. Radiologic Evaluation of Bone Loss at Implants with Biocide Coated Titanium Abutments: A Study in the Dog. PLoS ONE 2012, 7, e52861. [Google Scholar] [CrossRef] [PubMed]
- Esteban-Tejeda, L.; Cabal, B.; Malpartida, F.; López-Piriz, R.; Torrecillas, R.; Saiz, E.; Tomsia, A.; Moya, J. Soda-lime glass-coating containing silver nanoparticles on Ti-6Al-4V alloy. J. Eur. Ceram. Soc. 2012, 32, 2723–2729. [Google Scholar] [CrossRef]
- Martinez, A.; Guitián, F.; López-Píriz, R.; Bartolomé, J.F.; Cabal, B.; Esteban-Tejeda, L.; Torrecillas, R.; Moya, J.S. Bone loss at implant with titanium abutments coated by soda lime glass containing silver nanoparticles: A histological study in the dog. PLoS ONE 2014, 9, e86926. [Google Scholar] [CrossRef] [PubMed]
- Mennig, M.; Jonschker, G.; Schmidt, H.K. Sol-gel derived thick coatings and their thermomechanical and optical properties. Proc SPIE. 1992, 1758, 134–145. [Google Scholar]
- Kesmez, O.; Akarsu, E.; Arpac, E.; Yesil, Z.; Kiraz, N. Odor eliminating, antibacterial, environmentally friendly hydrophobic coating composition and its production and application method. Turkish Patent TR 2011 02940 B, 2015. [Google Scholar]
- Huang, H.I.; Shih, H.Y.; Lee, C.M.; Yang, T.C.; Lay, J.J.; Lin, Y.E. In vitro efficacy of copper and silver ions in eradicating Pseudomonas aeruginosa, Stenotrophomonas maltophilia, and Acinetobacter baumannii: Implications for on-site disinfection for hospital infection control. Water Res. 2008, 42, 73–80. [Google Scholar] [CrossRef]
- Bollen, C.M.L.; Papaioanno, W.; Van Eldere, J.; Schepers, E.; Quirynen, M.; Van Steenberghe, D. The influence of abutment surface roughness on plaque accumulation and peri-implant mucositis. Clin. Oral Implant. Res. 1996, 7, 201–211. [Google Scholar] [CrossRef]
- Sukhorukova, I.V.; Sheveyko, A.N.; Shvindina, N.V.; Denisenko, E.A.; Ignatov, S.G.; Shtansky, D.V. Approaches for controlled Ag+ ion release: Influence of surface topography, roughness, and bactericide content. ACS Appl. Mater. Interfaces 2017, 9, 4259–4271. [Google Scholar] [CrossRef]
- Kaali, P.; Perez-Madrigal, M.M.; Stromberg, E.; Aune, R.E.; Czel Gy Karlsson, S. The influence of Ag+, Zn2+ and Cu2+ exchanged zeolite on antimicrobial and long-term in vitro stability of medical grade polyether polyurethane. Express Polym. Lett. 2011, 5, 1028–1040. [Google Scholar] [CrossRef]
- Guo, T.; Gulati, K.; Arora, H.; Han, P.; Fournier, B.; Ivanovski, S. Race to invade: Understanding soft tissue integration at the transmucosal region of titanium dental implants. Dent. Mater. 2021, 37, 816–831. [Google Scholar] [CrossRef]
- Riivari, S.; Shahramian, K.; Kangasniemi, I.; Willberg, J.; Närhi, T. TiO2-Modified Zirconia Surface Improves Epithelial Cell Attachment. Int. J. Oral Maxillofac. Implant. 2019, 34, 313–319. [Google Scholar] [CrossRef]
- Yen Nee, W.; Raja Awang, R.A.; Hassan, A. Effects on the Titanium Implant Surface by Different Hygiene Instrumentations: A Narrative Review. Cureus 2022, 14, e30884. [Google Scholar] [CrossRef]
- Louropoulou, A.; Slot, D.E.; van der Weijden, F. The effects of mechanical instruments on contaminated titanium dental implant surfaces: A systematic review. Clin. Oral Implant. Res. 2014, 25, 1149–1160. [Google Scholar] [CrossRef] [PubMed]
- Stefanic, M.; Krnel, K.; Pribosic, I.; Kosmac, T. Rapid biomimetic deposition of octacalcium phosphate coatings on zirconia ceramics (Y-TZP) for dental implant applications. Appl. Surf. Sci. 2012, 258, 4649–4656. [Google Scholar] [CrossRef]
- Vogkou, C.T.; Vlachogiannis, N.I.; Palaiodimos, L.; Kousoulis, A.A. The causative agents in infective endocarditis: A systematic review comprising 33,214 cases. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 1227–1245. [Google Scholar] [CrossRef] [PubMed]
- Meza-Siccha, A.S.; Aguilar-Luis, M.A.; Silva-Caso, W.; Mazulis, F.; Barragan-Salazar, C.; Del Valle-Mendoza, J. In vitro evaluation of bacterial adhesion and bacterial viability of Streptococcus mutans, Streptococcus sanguinis, and Porphyromonas gingivalis on the abutment surface of titanium and zirconium dental implants. Int. J. Dent. 2019, 2019, 4292976. [Google Scholar] [CrossRef]
- Aguilar-Perez, D.; Vargas-Coronado, R.; Cervantes-Uc, J.M.; Rodriguez-Fuentes, N.; Aparicio, C.; Covarrubias, C.; Alvarez-Perez, M.; Garcia-Perez, V.; Martinez-Hernandez, M.; Cauich-Rodriguez, J.V. Antibacterial activity of a glass ionomer cement doped with copper nanoparticles. Dent. Mater. J. 2020, 39, 389–396. [Google Scholar] [CrossRef]
- Lafuente-Ibáñez de Mendoza, I.; Cayero-Garay, A.; Quindós-Andrés, G.; Aguirre-Urizar, J.M. A systematic review on the implication of Candida in peri-implantitis. Int. J. Implant. Dent. 2021, 7, 73. [Google Scholar] [CrossRef]
- Alrabiah, M.; Alshagroud, R.S.; Alsahhaf, A.; Almojaly, S.A.; Abduljabbar, T.; Javed, F. Presence of Candida species in the subgingival oral biofilm of patients with peri-implantitis. Clin. Implant. Dent. Relat. Res. 2019, 21, 781–785. [Google Scholar] [CrossRef]
- Minagi, S.; Miyake, Y.; Inagaki, K.; Tsuru, H.; Suginaka, H. Hydrophobic interaction in Candida albicans and Candida tropicalis adherence to various denture base resin materials. Infect. Immun. 1985, 47, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Hahnel, S.; Rosentritt, M.; Handel, G.; Bürgers, R. In vitro evaluation of artificial ageing on surface properties and early Candida albicans adhesion to prosthetic resins. J. Mater. Sci. Mater. Med. 2009, 20, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Săndulescu, M.; Sîrbu, V.D.; Popovici, I.A. Bacterial species associated with peri-implant disease—A literature review. GERMS 2023, 13, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Kanagasingam, S.; Chukkapalli, S.S.; Welbury, R.; Singhrao, S.K. Porphyromonas gingivalis is a Strong Risk Factor for Alzheimer’s Disease. J. Alzheimer’s Dis. Rep. 2020, 4, 501–511. [Google Scholar] [CrossRef]
- Tsarev, V.N.; Ippolitov, E.V.; Trefilov, A.G.; Arutiunov, S.D.; Pivovarov, A.A. Features of adhesion of anaerobic periodontopathogenic bacteria and Candida albicans fungi to experimental samples of basis dental plastic depending on surface roughness and polishing method. J. Mikrobiol Epidemiol. Immunobiol. 2014, 91, 21–27. [Google Scholar]
- Ren, G.; Hu, D.; Cheng, E.W.C.; Vargas-Reus, M.A.; Reip, P.; Allaker, R.P. Characterization of copper oxide nanoparticles for antimicrobial applications. Int. J. Antimicrob. Agents 2009, 33, 587–590. [Google Scholar] [CrossRef]
- Vargas-Reus, M.A.; Memarzadeh, K.; Huang, J.; Ren, G.G.; Allaker, R.P. Antimicrobial activity of nanoparticulate metal oxides against peri-implantitis pathogens. Int. J. Antimicrob. Agents 2012, 40, 135–139. [Google Scholar] [CrossRef]
- Teughels, W.; Van Assche, N.; Sliepen, I.; Quirynen, M. Effect of material characteristics and/or surface topography on biofilm development. Clin. Oral Implants Res. 2006, 17, 68–81. [Google Scholar] [CrossRef]
- Fan, X.; Yahia, L.; Sacher, E. Antimicrobial Properties of the Ag, Cu Nanoparticle System. Biology 2021, 10, 137. [Google Scholar] [CrossRef]
- Dey, P.; Suprabha, B.S.; Suman, E.; Natarajan, S.; Shenoy, R.; Rao, A. Comparative evaluation of surface roughness and bacterial adhesion on two bioactive cements: An in-vitro study. BMC Oral Health 2024, 24, 1278. [Google Scholar] [CrossRef]
- A Mulla, S.; A Kondkari, S.; Patil, A.; Mali, S.; Jaiswal, H.C.; Jakhar, A.; Ansari, Z.M.; Agarwal, S.; Yadav, P.; Mulla, S.A.; et al. A Look Into the Cytotoxicity of Composite Fillings: Friend or Foe? Cureus 2023, 15, e46327. [Google Scholar] [CrossRef] [PubMed]
- Cao, B.; Zheng, Y.; Xi, T.; Zhang, C.; Song, W.; Burugapalli, K.; Yang, H.; Ma, Y. Concentration-dependent cytotoxicity of copper ions on mouse fibroblasts in vitro: Effects of copper ion release from TCu380A vs TCu220C intra-uterine devices. Biomed. Microdevices 2012, 14, 709–720. [Google Scholar] [CrossRef]
- Vergara-Llanos, D.; Koning, T.; Pavicic, M.F.; Bello-Toledo, H.; Díaz-Gómez, A.; Jaramillo, A.; Melendrez-Castro, M.; Ehrenfeld, P.; Sánchez-Sanhueza, G. Antibacterial and cytotoxic evaluation of copper and zinc oxide nanoparticles as a potential disinfectant material of connections in implant provisional abutments: An in-vitro study. Arch. Oral Biol. 2021, 122, 105031. [Google Scholar] [CrossRef]
- Wataha, J.C.; Rueggeberg, F.A.; Lapp, C.A.; Lewis, J.B.; Lockwood, P.E.; Ergle, J.W.; Mettenburg, D.J. In vitro cytotoxicity of resin-containing restorative materials after aging in artificial saliva. Clin. Oral Investig. 1999, 3, 144–149. [Google Scholar] [CrossRef]
- Hatipoǧlu, Ö.; Karadaş, M.; Er, H.; Turumtay, E.A. Effect of thermocycling on the amount of monomer released from bulk fill composite resins. Dent. Mater. J. 2019, 38, 1019–1025. [Google Scholar] [CrossRef]
- Subramanian, B.; Maruthamuthu, S.; Rajan, S.T. Biocompatibility evaluation of sputtered zirconium-based thin film metallic glass-coated steels. Int. J. Nanomed. 2015, 10, 17–29. [Google Scholar] [CrossRef]
- Daguano, J.K.M.F.; Strecker, K.; Ziemath, E.C.; Rogero, S.O.; Fernandes, M.H.V.; Santos, C. Effect of partial crystallization on the mechanical properties and cytotoxicity of bioactive glass from the 3CaO.P2O5-SiO2-MgO system. J. Mech. Behav. Biomed. Mater. 2012, 14, 78–88. [Google Scholar] [CrossRef]



| G | GAg | GAgCu | |
|---|---|---|---|
| Glassy matrix solution | 26.78 g | 25 g | 25 g |
| Ag-GF20 complex solution | - | 4.17 g | 2.06 g |
| Cu solution | - | - | 2.06 g |
| Butyl glycol (BG) | 2.5 g | 2.5 g | 2.41 g |
| Ethanol (EtOH) | 21.88 g | 16.89 g | 16.60 g |
| GROUP | Thermal Aging | C% | Na% | O% | Si% | Ti% | Al% | V% | Ag% | Cu% | N% |
|---|---|---|---|---|---|---|---|---|---|---|---|
| G | None | 11.56 | 4.07 | 74.62 | 37.74 | 21.19 | 1.21 | 1.17 | - | - | - |
| 3000 cycles | 4.99 | 0.01 * | 31.25 | 12.50 | 28.69 | 1.51 | 1.47 | - | - | - | |
| GAg | None | 9.22 | 4.38 | 50.97 | 10.57 | 21.25 | 1.08 | 1.20 | 8.79 | - | - |
| 3000 cycles | 5.80 | 0.01 * | 50.98 | 7.24 | 41.47 | 2.41 | 1.54 | 7.66 | - | - | |
| GAgCu | None | 6.74 | 5.70 | 52.78 | 12.67 | 21.15 | 2.00 | 1.06 | 1.21 | 0.30 | - |
| 3000 cycles | 4.72 | 0.01 * | 49.40 | 7.13 | 35.48 | 1.17 | 1.00 | 1.17 | 0.20 | - |
| Thermal Cycling | Surface Roughness Ra (µm) | Surface Contact Angle (°) | Surface Hardness (HV) | Scratch Resistance (N) | ASTM D3359 [39] | |
|---|---|---|---|---|---|---|
| Ti6Al4V | None | 0.10 ± 0.01 a | 51.6 ± 0.5 a | 136 ± 5.3 a | - | - |
| 3000 cycles | 0.14 ± 0.03 b | 51.7 ± 0.6 a | 146 ± 10.3 a | - | - | |
| G | None | 0.07 ± 0.017 c | 28.0 ± 1.6 b | 262 ± 35.7 b | 2.81 ± 0.89 a | 5B |
| 3000 cycles | 0.10 ± 0.039 a | 35.4 ± 1.9 c | 240 ± 52.9 b | 2.24 ± 0.14 a | 5B | |
| GAg | None | 0.09 ± 0.038 a | 29.6 ± 2.7 b | 277 ± 40.5 b | 3.90 ± 0.80 b | 5B |
| 3000 cycles | 0.12 ± 0.042 ab | 33.9 ± 1.4 c | 261 ± 47.7 b | 4.56 ± 0.39 b | 5B | |
| GAgCu | None | 0.08 ± 0.017 ac | 30.5 ± 1.6 b | 252 ± 51.3 b | 3.95 ± 0.55 b | 5B |
| 3000 cycles | 0.11 ± 0.027 a | 33.3 ± 2.2 c | 272 ± 55.4 b | 2.72 ± 0.53 a | 5B |
| Groups | C. albicans DSM 5817 | P. gingivalis ATCC 33277 | S. sanguinis ATCC 10556 |
|---|---|---|---|
| Ti6Al4V | 4.8 ± 0.98 a,A | 7.7 ± 0.9 a,B | 27.3 ± 0.48 a,C |
| G | 4.5 ± 1.02 a,A | 9.7 ± 1.4 b,B | 28.8 ± 12.26 a,C |
| GAg | 2.3 ± 0.48 b,A | 3.1 ± 1.04 c,A | 6.5 ± 1.42 b, B |
| GAgCu | 1.0 ± 0,12 c,A | 2.3 ± 0.29 d,B | 3.5 ± 0.38 c,B |
| Groups | C. albicans DSM 5817 | P. gingivalis ATCC 33277 | S. sanguinis ATCC 10556 |
|---|---|---|---|
| Ti6Al4V | 157.9 ± 13.2 a,A | 9.8 ± 0.84 a,B | 166.9 ± 25.98 a,A |
| G | 131.6 ± 7.6 a,A | 13.4 ± 1.12 b,B | 185.0 ± 13.90 a,C |
| GAg | 111.6 ± 8.1 b,A | 6.7 ± 0.86 c,B | 137.6 ± 14.57 b,C |
| GAgCu | 108.5 ± 10.9 b,A | 3.5 ± 0.29 d,B | 90.2 ± 16.55 c,C |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Çölgeçen, Ö.; Akarsu, M.; Akarsu, E.; Uzel, A.; Özdal Kurt, F.; Topal, E.S.; Gençer, G.M.; Keski, A.; Yavuz, E. Antimicrobial Sol–Gel Glassy Surfaces for Modification of Dental Implant Abutments to Reduce Microbial Adhesion. Gels 2025, 11, 882. https://doi.org/10.3390/gels11110882
Çölgeçen Ö, Akarsu M, Akarsu E, Uzel A, Özdal Kurt F, Topal ES, Gençer GM, Keski A, Yavuz E. Antimicrobial Sol–Gel Glassy Surfaces for Modification of Dental Implant Abutments to Reduce Microbial Adhesion. Gels. 2025; 11(11):882. https://doi.org/10.3390/gels11110882
Chicago/Turabian StyleÇölgeçen, Özlem, Murat Akarsu, Esin Akarsu, Ataç Uzel, Feyzan Özdal Kurt, Eyüp Sabri Topal, Gül Merve Gençer, Ahmet Keski, and Emre Yavuz. 2025. "Antimicrobial Sol–Gel Glassy Surfaces for Modification of Dental Implant Abutments to Reduce Microbial Adhesion" Gels 11, no. 11: 882. https://doi.org/10.3390/gels11110882
APA StyleÇölgeçen, Ö., Akarsu, M., Akarsu, E., Uzel, A., Özdal Kurt, F., Topal, E. S., Gençer, G. M., Keski, A., & Yavuz, E. (2025). Antimicrobial Sol–Gel Glassy Surfaces for Modification of Dental Implant Abutments to Reduce Microbial Adhesion. Gels, 11(11), 882. https://doi.org/10.3390/gels11110882









