Anti-SARS-CoV-2 Agents in Artemisia Endophytic Fungi and Their Abundance in Artemisia vulgaris Tissue
Abstract
1. Introduction
2. Materials and Methods
2.1. Host Plant and Its Endophytic Fungi
2.2. Preparation of Endophytic Culture Extracts
2.3. Cell Culture
2.4. Infection Assay
2.5. Quantification of Viral RNA
2.6. Cell Viability
2.7. The Ethanol Extract of A. vulgaris
2.8. SARS-CoV-2 3C-like Protease Inhibitory Activity Test
2.9. Metabarcoding Analysis of Fungi Abundance in A. vulgaris and Their Cultivar Soil Sample Collection and DNA Extraction
2.10. Analysis of the Mycobiome Composition
2.11. Statistical Analyses
3. Results
3.1. Anti-SARS-CoV-2 Activity of Artemisia Endophyte Extract
3.1.1. Effect of Endophytic Extracts on the Cytopathology of SARS-CoV-2-Infected Cells
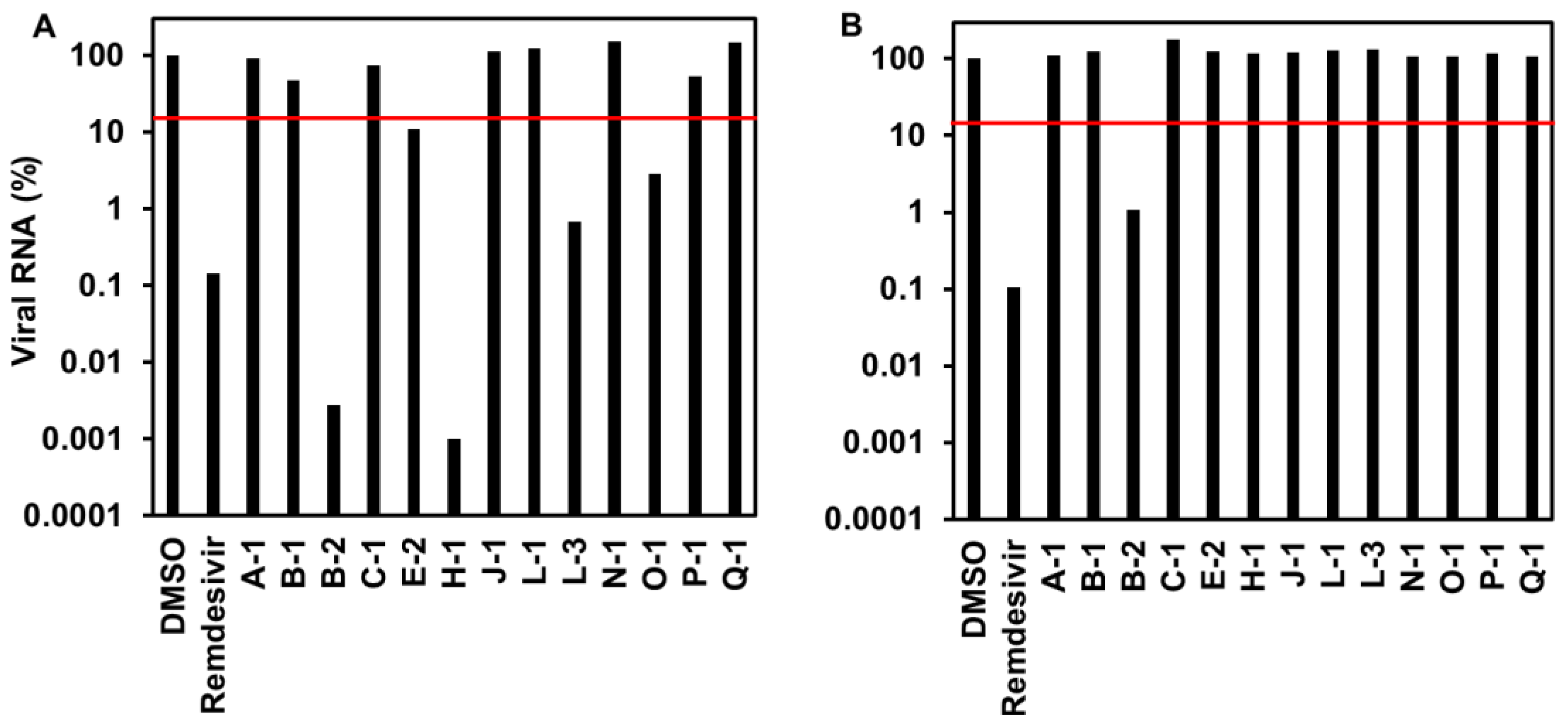
3.1.2. Viral RNA Level of Endophytic Extracts to SARS-CoV-2-Infected Cells
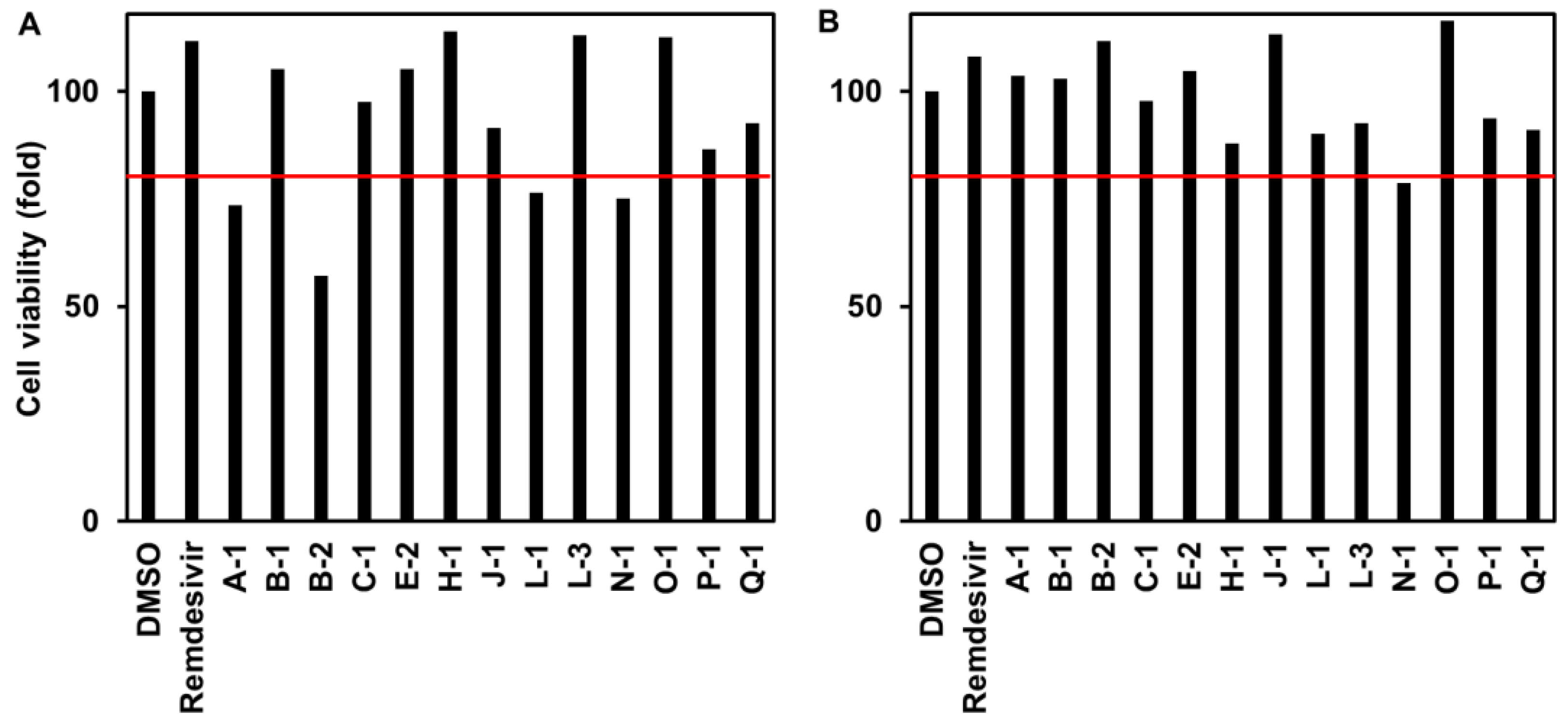
3.1.3. Cytotoxicity to VeroE6/TMPRSS2 Cells
3.1.4. SARS-CoV-2 3CL Protease Inhibitory Activity Test
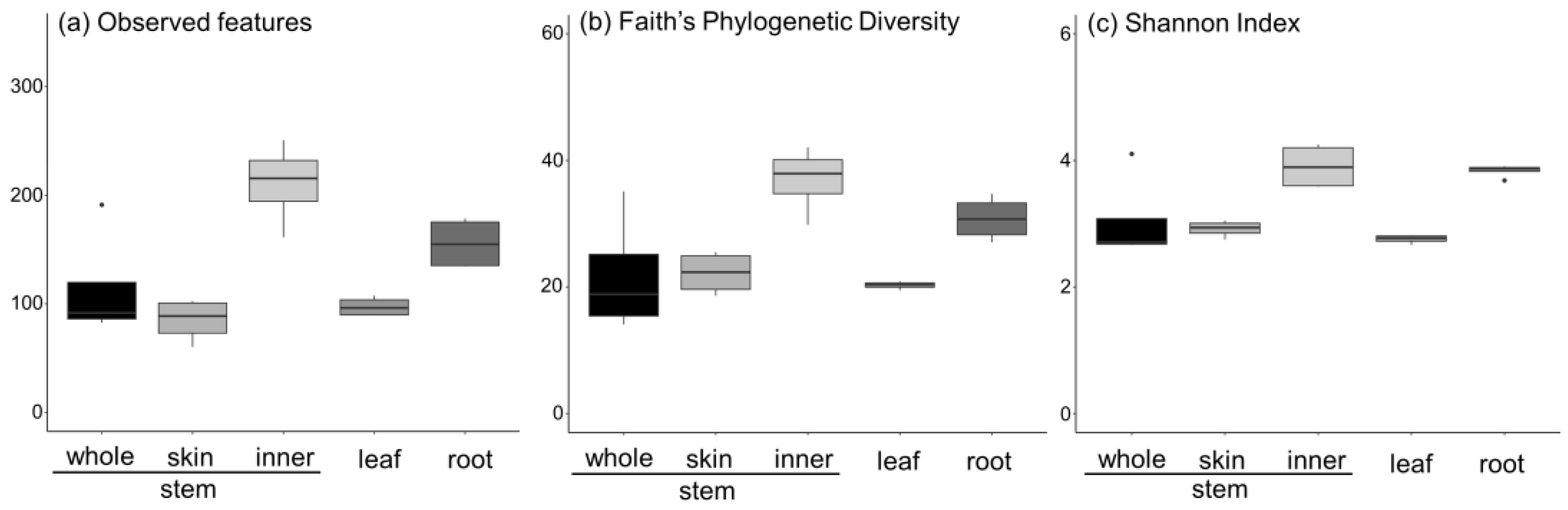
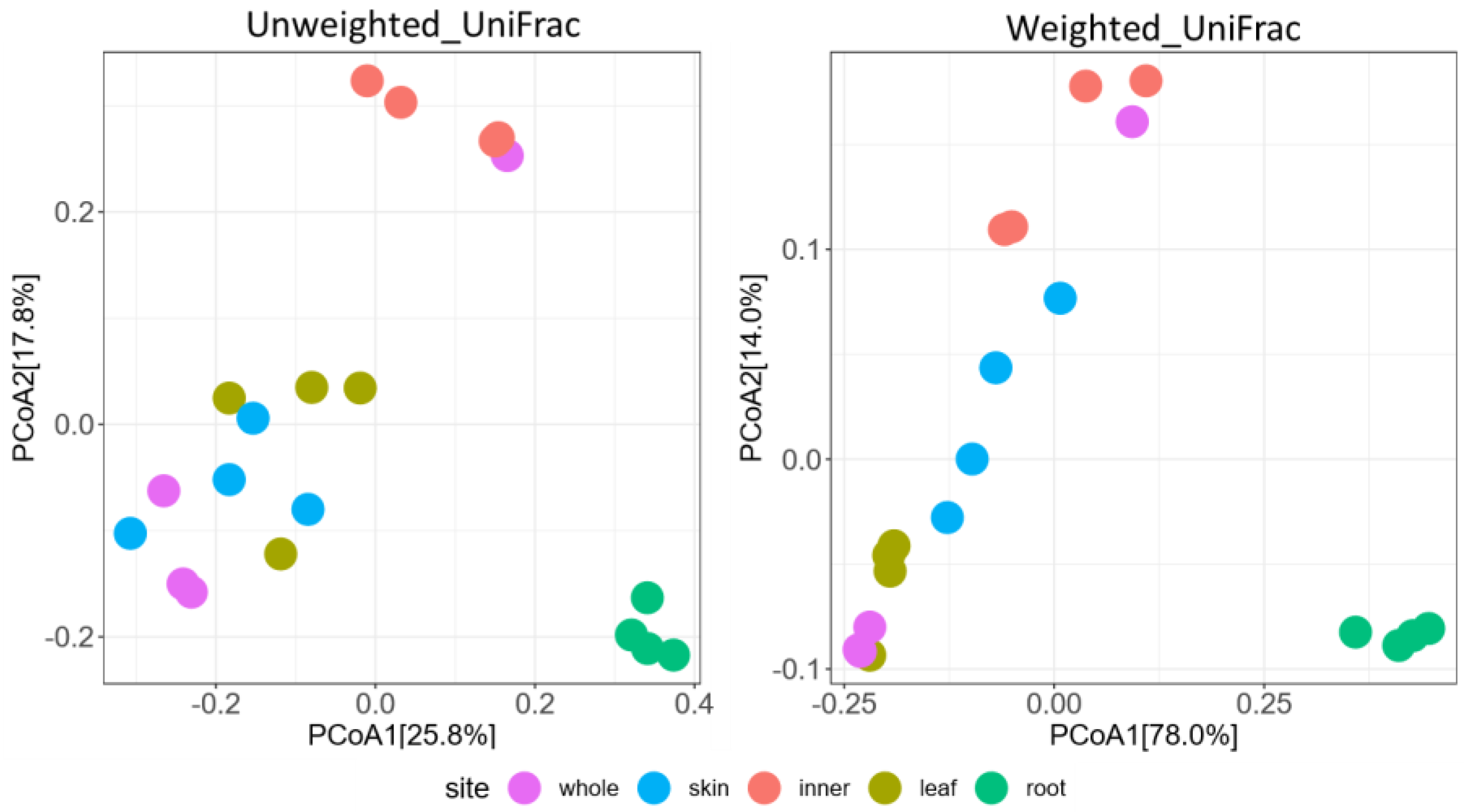
3.2. Metabarcoding Analysis of Fungi Abundance in A. vulgaris and Their Cultivar Soil
- (a)
- Aspergillus spp. are more abundant on the stem and leaves and skin than on the inner part of the stem.
- (b)
- Cladosporium spp. are more abundant on the stem and leaves, with a tenth of the abundance of Aspergillus, and are present in the inner part of the stem and the skin to the same extent.
- (c)
- Colletotrichum spp. are more abundant on the stem and leaves and account for the largest abundance of the six genera. They are more abundant on the skin than on the inner part of the stem.
- (d)
- Cryptococcus spp. are abundant in the leaves and have the lowest abundance (approximately 0.05%).
- (e)
- Diaporthe spp. are abundant in the leaves and roots, with a particularly high percentage in the roots.
- (f)
- Penicillium spp. are abundant in the stem and leaves, with a higher percentage abundance in the skin than in the inner part of the stem.

4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Saikkonen, K.; Wäli, P.; Helander, M.; Faeth, S.H. Evolution of endophyte-plant symbioses. Trends Plant Sci. 2004, 9, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Wani, Z.A.; Ashraf, N.; Mohiuddin, T.; Riyaz-Ul-Hassan, S. Plant-Endophyte Symbiosis, an Ecological Perspective. Appl. Microbiol. 2015, 99, 2955–2965. [Google Scholar] [CrossRef]
- Arachevaleta, M.; Bacon, C.W.; Hoveland, C.S.; Radcliffe, D.E. Effect of the Tall Fescue Endophyte on Plant Response to Environmental Stress. Agron. J. 1989, 81, 83–90. [Google Scholar] [CrossRef]
- Siegel, M.R.; Latch, G.C.M.; Johnson, M.C. Fungal Endophytes of Grasses. Annu. Rev. Phytopathol. 1987, 25, 293–315. [Google Scholar] [CrossRef]
- Kanda, K.; Hirai, Y.; Koga, H.; Hasegawa, K. Endophyte-Enhanced Resistance in Perennial Ryegrass and Tall Fescue to Bluegrass Webworm, Parapediasia Teterrella. Jpn. J. Appl. Entomol. Zool. 1994, 38, 141–145. [Google Scholar] [CrossRef][Green Version]
- Stierle, A.; Strobel, G.; Stierle, D. Taxol and Taxane Production by Taxomyces Andreanae, an Endophytic Fungus of Pacific Yew. Science 1993, 260, 214–216. [Google Scholar] [CrossRef]
- Eyberger, A.L.; Dondapati, R.; Porter, J.R. Endophyte Fungal Isolates from Podophyllum peltatum Produce Podophyllotoxin. J. Nat. Prod. 2006, 69, 1121–1124. [Google Scholar] [CrossRef]
- Kusari, S.; Zühlke, S.; Spiteller, M. An Endophytic Fungus from Camptotheca acuminata That Produces Camptothecin and Analogues. J. Nat. Prod. 2009, 72, 2–7. [Google Scholar] [CrossRef]
- Kusari, S.; Lamshöft, M.; Zühlke, S.; Spiteller, M. An Endophytic Fungus from Hyperucyn Perforatum That Produces Hypericin. J. Nat. Prod. 2008, 71, 159–162. [Google Scholar]
- Maehara, S.; Simanjuntak, P.; Maetani, Y.; Kitamura, C.; Ohashi, K.; Shibuya, H. Ability of Endophytic Filamentous Fungi Associated with Cinchona Ledgeriana to Produce Cinchona Alkaloids. J. Nat. Med. 2013, 67, 421–423. [Google Scholar] [CrossRef]
- Nair, M.S.; Huang, Y.; Fidock, D.A.; Polyak, S.J.; Wagoner, J.; Towler, M.J.; Weathers, P.J. Artemisia annua L. Extracts Inhibit the In Vitro Replication of SARS-CoV-2 and Two of Its Variants. J. Ethnopharmacol. 2021, 274, 14016. [Google Scholar] [CrossRef]
- Khiralla, A.; Spina, R.; Varbanov, M.; Philippot, S.; Lemiere, P.; Slezack-Deschaumes, S.; André, P.; Mohamed, I.; Yagi, S.M.; Laurain-Mattar, D. Evaluation of Antiviral, Antibacterial and Antiproliferative Activities of the Endophytic Fungus Curvularia papendorfii, and Isolation of a New Polyhydroxyacid. Microorganisms 2020, 8, 1353. [Google Scholar] [CrossRef] [PubMed]
- Cosoveanu, A.; Cabrera, R. Endophytic Fungi in Species of Artemisia. J. Fungi 2018, 4, 53. [Google Scholar] [CrossRef]
- Maehara, S.; Fathoni, A.; Tagawa, M.; Shiose, M.; Yamasaki, H.; Kikuchi, M.; Evana, E.; Ilyas, M.; Adriyani, M.; Hata, T.; et al. Environmental Differences between Japan and Indonesia Provide Endophyte Diversity Associated with Artemisia Plant and Variety of Artemisinin Derivatives in Microbial Conversion. J. Nat. Med. 2023, 77, 916–927. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, S.; Ohashi, H.; Akazawa, D.; Torii, S.; Suzuki, R.; Fukuhara, T.; Watashi, K. Antiviral Activity of Micafungin and Its Derivatives against SARS-CoV-2 RNA Replication. Viruses 2023, 15, 452. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, Interactive, Scalable and Extensible Microbiome Data Science Using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar]
- Martin, M. Cutadapt Removes Adapter Sequences from High-Throughput Sequencing Reads. EMBnet. J. 2011, 17, 10. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-Resolution Sample Inference from Illumina Amplicon Data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Kõljalg, U.; Larsson, K.H.; Abarenkov, K.; Nilsson, R.H.; Alexander, I.J.; Eberhardt, U.; Erland, S.; Høiland, K.; Kjøller, R.; Larsson, E.; et al. UNITE: A Database Providing Web-Based Methods for the Molecular Identification of Ectomycorrhizal Fungi. New Phytol. 2005, 166, 1063–1068. [Google Scholar] [CrossRef]
- Kim, J.W.; Shim, S.H. The Fungus Colletotrichum as a Source for Bioactive Secondary Metabolites. Arch. Pharm. Res. 2019, 42, 735–753. [Google Scholar] [CrossRef]
- Durán, P.; Thiergart, T.; Garrido-Oter, R.; Agler, M.; Kemen, E.; Schulze-Lefert, P.; Hacquard, S. Microbial Interkingdom Interactions in Roots Promote Arabidopsis Survival. Cell 2018, 175, 973–983. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.C.; Lu, Y.H.; Wang, J.F.; Song, Z.Q.; Hou, Y.G.; Liu, S.S.; Liu, C.S.; Wu, S.H. Bioactive Secondary Metabolites of the Genus Diaporthe and Anamorph Phomopsis from Terrestrial and Marine Habitats and Endophytes: 2010–2019. Microorganisms 2021, 9, 217. [Google Scholar] [CrossRef] [PubMed]




| Collection Name | Accession Number | Genus | Extract Weight (mg) |
|---|---|---|---|
| yomogi fungi A-1 | LC719230 | Aspergillus sp. | 80 |
| yomogi fungi B-1 | LC719228 | Cladosporium sp. | 80 |
| yomogi fungi B-2 | LC719244 | Cryptococcus sp. | 40 |
| yomogi fungi C-1 | LC719242 | Penicillium sp. | 70 |
| yomogi fungi E-2 | LC719209 | Colletotrichum sp. | 50 |
| yomogi fungi H-1 | LC719243 | Mollisia sp. | 40 |
| yomogi fungi J-1 | LC719231 | Aspergillus sp. | 130 |
| yomogi fungi L-1 | LC719219 | Diaporthe sp. | 150 |
| yomogi fungi L-3 | LC719210 | Colletotrichum sp. | 80 |
| yomogi fungi N-1 | LC719232 | Aspergillus sp. | 70 |
| yomogi fungi O-1 | LC719233 | Aspergillus sp. | 40 |
| yomogi fungi P-1 | LC775239 | Hypomontagnella sp. | 120 |
| yomogi fungi Q-1 | LC719229 | Cladosporium sp. | 40 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maehara, S.; Nakajima, S.; Watashi, K.; Agusta, A.; Kikuchi, M.; Hata, T.; Takayama, K. Anti-SARS-CoV-2 Agents in Artemisia Endophytic Fungi and Their Abundance in Artemisia vulgaris Tissue. J. Fungi 2023, 9, 905. https://doi.org/10.3390/jof9090905
Maehara S, Nakajima S, Watashi K, Agusta A, Kikuchi M, Hata T, Takayama K. Anti-SARS-CoV-2 Agents in Artemisia Endophytic Fungi and Their Abundance in Artemisia vulgaris Tissue. Journal of Fungi. 2023; 9(9):905. https://doi.org/10.3390/jof9090905
Chicago/Turabian StyleMaehara, Shoji, Shogo Nakajima, Koichi Watashi, Andria Agusta, Misato Kikuchi, Toshiyuki Hata, and Kento Takayama. 2023. "Anti-SARS-CoV-2 Agents in Artemisia Endophytic Fungi and Their Abundance in Artemisia vulgaris Tissue" Journal of Fungi 9, no. 9: 905. https://doi.org/10.3390/jof9090905
APA StyleMaehara, S., Nakajima, S., Watashi, K., Agusta, A., Kikuchi, M., Hata, T., & Takayama, K. (2023). Anti-SARS-CoV-2 Agents in Artemisia Endophytic Fungi and Their Abundance in Artemisia vulgaris Tissue. Journal of Fungi, 9(9), 905. https://doi.org/10.3390/jof9090905





