Fungal Keratitis: Clinical Features, Risk Factors, Treatment, and Outcomes
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Castano, G.; Elnahry, A.G.; Mada, P.K. Fungal Keratitis; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Acharya, Y.; Acharya, B.; Karki, P. Fungal keratitis: Study of increasing trend and common determinants. Nepal J. Epidemiol. 2017, 7, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Zhai, H.; Zhao, J.; Sun, S.; Shi, W.; Dong, X. Antifungal Susceptibility for Common Pathogens of Fungal Keratitis in Shandong Province, China. Am. J. Ophthalmol. 2008, 146, 260–265.e1. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, R.P.; Nayyar, S.V.; Romanowski, E.G.; Shanks, R.M.; Mammen, A.; Dhaliwal, D.K.; Jhanji, V. The Prevalence of Bacteria, Fungi, Viruses, and Acanthamoeba from 3004 Cases of Keratitis, Endophthalmitis, and Conjunctivitis. Eye Contact Lens 2020, 46, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, S.; Masoomi, A.; Ahmadikia, K.; Tabatabaei, S.A.; Soleimani, M.; Rezaie, S.; Ghahvechian, H.; Banafsheafshan, A. Fungal keratitis: An overview of clinical and laboratory aspects. Mycoses 2018, 61, 916–930. [Google Scholar] [CrossRef]
- Ritterband, D.C.; Seedor, J.A.; Shah, M.K.; Koplin, R.S.; McCormick, S.A. Fungal Keratitis at the New York Eye and Ear Infirmary. Cornea 2006, 25, 264–267. [Google Scholar] [CrossRef]
- Tanure, M.A.G.; Cohen, E.J.; Sudesh, S.; Rapuano, C.J.; Laibson, P.R. Spectrum of Fungal Keratitis at Wills Eye Hospital, Philadelphia, Pennsylvania. Cornea 2000, 19, 307–312. [Google Scholar] [CrossRef]
- Manikandan, P.; Abdel-Hadi, A.; Randhir Babu Singh, Y.; Banawas, S.; Dukhyil, A.A.B.; Alshehri, B.; Shobana, C.S.; Selvam, K.P.; Narendran, V. Fungal Keratitis: Epidemiology, Rapid Detection, and Antifungal Susceptibilities of Fusarium and Aspergillus Isolates from Corneal Scrapings. Biomed. Res. Int. 2019, 2019, 6395840. [Google Scholar] [CrossRef]
- Bourcier, T.; Sauer, A.; Dory, A.; Denis, J.; Sabou, M. Fungal keratitis. J. Fr. Ophtalmol. 2017, 40, e307–e313. [Google Scholar] [CrossRef]
- Austin, A.; Lietman, T.; Rose-Nussbaumer, J. Update on the Management of Infectious Keratitis. Ophthalmology 2017, 124, 1678–1689. [Google Scholar] [CrossRef]
- Jhanji, V.; Sharma, N.; Mannan, R.; Titiyal, J.S.; Vajpayee, R.B. Management of tunnel fungal infection with voriconazole. J. Cataract Refract. Surg. 2007, 33, 915–917. [Google Scholar] [CrossRef]
- Arnfield, A.J. Köppen Climate Classification. Encyclopedia Britannica. Climatology Web Site. Available online: https://www.britannica.com/science/Koppen-climate-classification (accessed on 12 September 2022).
- Tiew, S.; Lim, C.; Sivagnanasithiyar, T. Using an excel spreadsheet to convert Snellen visual acuity to LogMAR visual acuity. Eye 2020, 34, 2148–2149. [Google Scholar] [CrossRef]
- Estopinal, C.B.; Ewald, M.D. Geographic Disparities in the Etiology of Bacterial and Fungal Keratitis in the United States of America. Semin. Ophthalmol. 2016, 31, 345–352. [Google Scholar] [CrossRef]
- Inoue, Y.; Ohashi, Y.; Suzuki, T.; Shimomura, Y.; Fukuda, M.; Sotozono, C.; Hatano, H.; Eguchi, H.; Araki-Sasaki, K.; Hoshi, S.; et al. Multicenter Prospective Observational Study of Fungal Keratitis—Current Status of Patients’ Background, Clinical Findings, Treatment and Prognosis. Nippon Ganka Gakkai Zasshi 2016, 120, 5–16. [Google Scholar]
- Liesegang, T.J. Contact lens-related microbial keratitis: Part I: Epidemiology. Cornea 1997, 16, 125–131. [Google Scholar] [CrossRef]
- Liesegang, T.J. Contact lens-related microbial keratitis: Part II: Pathophysiology. Cornea 1997, 16, 265–273. [Google Scholar] [CrossRef]
- Knutsson, K.; Iovieno, A.; Matuska, S.; Fontana, L.; Rama, P. Topical Corticosteroids and Fungal Keratitis: A Review of the Literature and Case Series. J. Clin. Med. 2021, 10, 1178. [Google Scholar] [CrossRef]
- Doughman, D.J.; Leavenworth, N.M.; Campbell, R.C.; Lindstrom, R.L. Fungal keratitis at the University of Minnesota: 1971–1981. Trans. Am. Ophthalmol. Soc. 1982, 80, 235–247. [Google Scholar]
- Asbell, P.; Stenson, S. Ulcerative keratitis. Survey of 30 years’ laboratory experience. Arch. Ophthalmol. 1982, 100, 77–80. [Google Scholar] [CrossRef]
- Durrani, A.F.; Atta, S.; Bhat, A.K.; Mammen, A.; Dhaliwal, D.; Kowalski, R.P.; Jhanji, V. Methicillin-Resistant Staphylococcus aureus Keratitis: Initial Treatment, Risk Factors, Clinical Features, and Treatment Outcomes. Am. J. Ophthalmol. 2020, 214, 119–126. [Google Scholar] [CrossRef]
- Atta, S.; Perera, C.; Nayyar, S.; Kowalski, R.P.; Jhanji, V. An 18-Year Overview of Serratia marcescens Ocular Infection. Eye Contact Lens Sci. Clin. Pr. 2021, 47, 471–475. [Google Scholar] [CrossRef]
- Dahlgren, M.A.; Lingappan, A.; Wilhelmus, K.R. The Clinical Diagnosis of Microbial Keratitis. Am. J. Ophthalmol. 2007, 143, 940–944.e1. [Google Scholar] [CrossRef]
- Prajna, N.V.; Krishnan, T.; Mascarenhas, J.; Rajaraman, R.; Prajna, L.; Srinivasan, M.; Raghavan, A.; Oldenburg, C.E.; Ray, K.J.; Zegans, M.E.; et al. The mycotic ulcer treatment trial: A randomized trial comparing natamycin vs voriconazole. JAMA Ophthalmol. 2013, 131, 422–429. [Google Scholar] [CrossRef]
- Retamal, J.; Ordenes-Cavieres, G.; Grau-Diez, A. Natamycin versus voriconazole for fungal keratitis. Medwave 2018, 18, e7388. [Google Scholar] [CrossRef]
- Prajna, N.V.; Krishnan, T.; Rajaraman, R.; Patel, S.; Srinivasan, M.; Das, M.; Ray, K.J.; O’Brien, K.S.; Oldenburg, C.E.; McLeod, S.D.; et al. Effect of Oral Voriconazole on Fungal Keratitis in the Mycotic Ulcer Treatment Trial II (MUTT II): A Randomized Clinical Trial. JAMA Ophthalmol. 2016, 134, 1365–1372. [Google Scholar] [CrossRef]
- Ho, J.W.; Fernandez, M.M.; Rebong, R.A.; Carlson, A.N.; Kim, T.; Afshari, N.A. Microbiological profiles of fungal keratitis: A 10-year study at a tertiary referral center. J. Ophthalmic Inflamm. Infect. 2016, 6, 5. [Google Scholar] [CrossRef] [Green Version]
| Feature | N = 28 |
|---|---|
| Demographic/General Information | |
| Female (%, n) | 46.4 (13) |
| Age at presentation (median, IQR) | 58.5 (18.5) years |
| Laterality (left eye) (%, n) | 57.1 (16) |
| Symptom duration prior to presentation (median, IQR) | 10 (35.8) |
| Referred from outside provider (%, n) | 78.5 (22) |
| Warm season (spring/summer months) (%, n) | 60.7 (17) |
| Ocular Risk Factors (%, n) | |
| Contact lens use | 67.8 (19) |
| Poor contact lens hygiene | 57.8 (11/19) |
| Recent ocular trauma/abrasion | 42.8 (12) |
| Water exposure to eye | 14.2 (4) |
| History of: | |
| Corneal ulcer/keratitis | 35.7 (10) |
| Ocular surface disease * | 32.1 (9) |
| Glaucoma | 10.7 (3) |
| Ocular surgery | 42.8 (12) |
| PKP | 7.1 (2) |
| Cataract surgery | 17.8 (5) |
| LASIK surgery | 17.8 (5) |
| Glaucoma/retina surgery | 10.7 (3) |
| Topical steroid use | 32.1 (9) |
| Topical medication use | 35.7 (10) |
| Systemic Comorbidities (%, n) | |
| Hypertension | 35.7 (10) |
| Smoker | 21.4 (6) |
| Systemic Atopy | 57.1 (16) |
| Clinical Features at Presentation | |
| Initial visual acuity (median, IQR) | 1.35 (1.72) |
| Initial intraocular pressure (median, IQR) | 13.0 (8.0) mmHg |
| Central ulcer (%, n) | 42.8 (12/28) |
| Infiltrate size (median, IQR) | 6.7 (6.3) |
| Corneal thinning (%, n) | 50.0 (14/28) |
| Hypopyon (%, n) | 32.1 (9/28) |
| Feature | N = 28 |
|---|---|
| Medical Management (%, n) | |
| Anti-fungal | |
| Natamycin drops | 53.5 (15) |
| Amphotericin B drops | 28.5 (8) |
| Intrastromal amphotericin B | 10.7 (3) |
| Voriconazole drops | 71.4 (20) |
| Oral voriconazole | 64.2 (18) |
| Intrastromal voriconazole | 17.8 (5) |
| Intravitreal voriconazole | 3.5 (1) |
| Oral ketoconazole | 3.5 (1) |
| Oral diflucan | 17.8 (5) |
| Anti-bacterial | |
| Fluoroquinolone drops | 89.2 (25) |
| Gatifloxacin | 17.8 (5) |
| Moxifloxacin | 67.8 (19) |
| Ciprofloxacin | 21.4 (6) |
| Ofloxacin | 25.0 (7) |
| Besifloxacin | 17.8 (5) |
| Fortified drops | 67.8 (19) |
| Fortified tobramycin | 67.8 (19) |
| Fortified cefazolin | 57.1 (16) |
| Fortified vancomycin | 21.4 (6) |
| Anti-viral | |
| Valacyclovir | 32.1 (9) |
| Acyclovir | 3.5 (1) |
| Ganciclovir | 7.1 (2) |
| Other | |
| Any steroids | 64.2 (18) |
| Topical steroids prior to anti-fungal | 39.2 (11) |
| Topical steroids later in management | 42.8 (12) |
| Vitamin C | 64.2 (18) |
| Doxycycline | 71.4 (20) |
| Cyclopentolate | 71.4 (20) |
| Atropine | 25.0 (7) |
| Adjunctive Management (%, n) | |
| Any adjunctive treatment | 78.5 (22) |
| Serum drops | 32.1 (9) |
| Bandage contact lens | 28.5 (8) |
| Tarsorrhaphy | 7.14 (2) |
| Debridement | 25.0 (7) |
| PKP | 32.1 (9) |
| Enucleation | 3.5 (1) |
| Other * | 14.2 (4) |
| Clinical Outcomes | |
| Treatment duration (median, IQR) | 65.6 (46.3) days |
| Time to defect resolution (median, IQR) | 42.5 (47.0) days |
| Final visual acuity (median, IQR) | 0.5 (1.84) LogMAR |
| Final intraocular pressure (median, IQR) | 12.5 (6.5) mmHg |
| Complication during management (%, n) | 46.4 (13) |
| Microbiology Collection Type, Findings | Percentage (n), N = 28 |
|---|---|
| Gram stain collected | 10.7 (3) |
| with fungal elements | 33.3 (1/3) |
| Smear collected (corneal) | 100 (28) |
| with fungal elements | 50.0 (14/28) |
| Fungal culture collected | 92.8 (26) |
| Positive culture for fungus | 57.6 (15/26) |
| Corneal culture collected | 89.2 (25) |
| Positive culture for fungus | 32.0 (8/25) |
| Conjunctival culture collected | 67.8 (19) |
| Positive culture for fungus | 10.5 (2/19) |
| Total culture-positive cases | 75 (21) |
| Concurrent bacterial infection | 50.0 (14) |
| Concurrent herpes simplex virus infection | 3.5 (1) |
| Isolated fungus types | |
| Filamentous | |
| Aspergillus spp. | 10.7 (3) |
| Fusarium spp. | 10.7 (3) |
| Acremonium spp. | 7.1 (2) |
| Scedosporium spp. | 7.1 (2) |
| Unspecified “hyphae” | 7.1 (2) |
| Alternaria spp. | 3.5 (1) |
| Bipolaris spp. | 3.5 (1) |
| Exserohilum spp. | 3.5 (1) |
| Yeast | |
| Unspecified “yeast” | 10.7 (3) |
| Candida spp. | 7.1 (2) |
| Unspecified “fungal elements” | 3.5 (1) |
| Clinical Feature | Poor VA Outcome | Better VA Outcome | p-Value |
|---|---|---|---|
| Presentation | |||
| Cool season at time of presentation (fall/winter) (%, n) | 52.3% (11/21) | 0% (0/7) | 0.044 |
| Warm season at time of presentation (spring/summer) (%, n) | 47.6% (10/21) | 100% (7/7) | 0.044 |
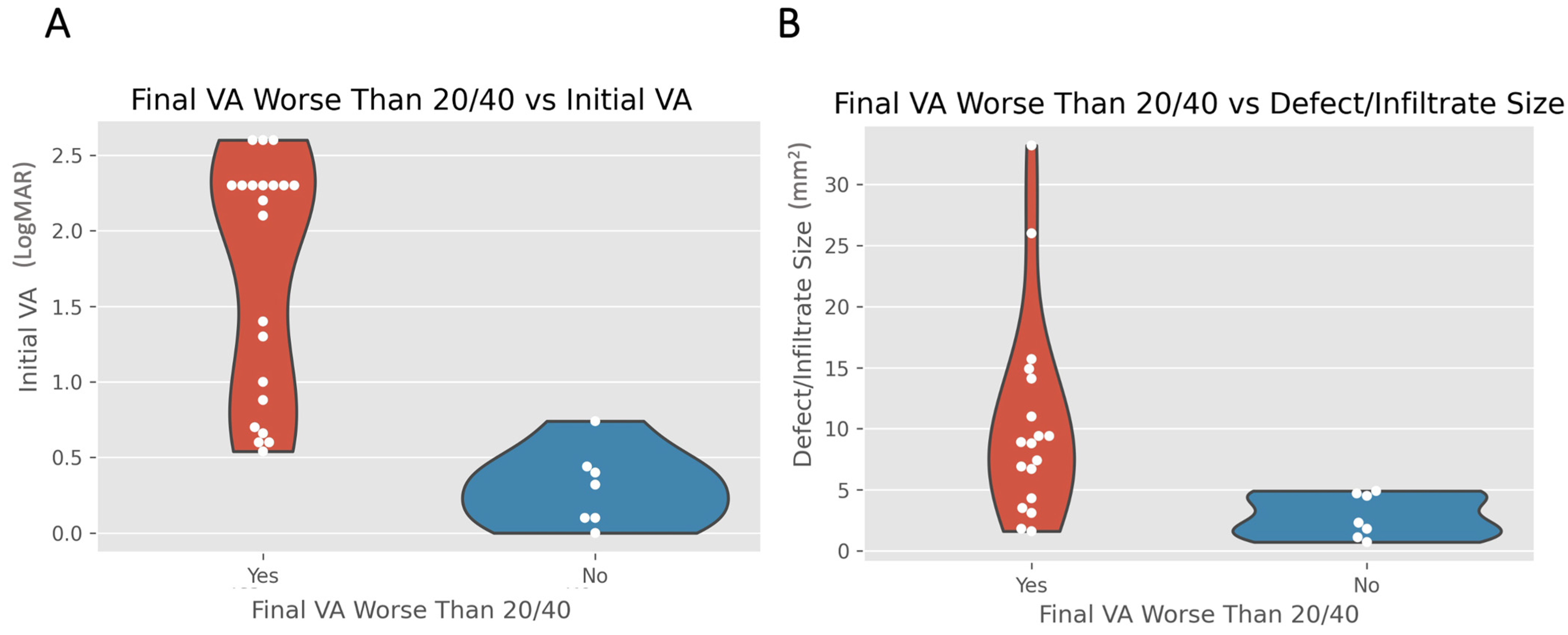
| Initial VA (mean ± SD) | 1.7 ± 0.8 LogMAR (21/28) | 0.3 ± 0.3 LogMAR (7/28) | <0.001 |
| Defect size (mean ± SD) | 10.3 ± 8.0 (18/28) | 2.8 ± 1.6 (7/28) | 0.002 |
| Central defect location (%, n) | 57.1% (12/21) | 0% (0/7) | 0.027 |
| Microbiology | |||
| Smear-positive for fungal elements (%, n) | 33.3% (7/21) | 100% (7/7) | 0.009 |
| Smear-negative for fungal elements (%, n) | 57.1% (12/21) | 0% (0/7) | 0.027 |
| Aspergillus on culture (%, n) | 0% (0/21) | 42.8% (3/7) | 0.039 |
| Treatment | |||
| Oral voriconazole (%, n) | 80.9% (17/21) | 14.2% (1/7) | 0.038 |
| Debridement (%, n) | 9.5% (2/21) | 71.4% (5/7) | 0.030 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atta, S.; Perera, C.; Kowalski, R.P.; Jhanji, V. Fungal Keratitis: Clinical Features, Risk Factors, Treatment, and Outcomes. J. Fungi 2022, 8, 962. https://doi.org/10.3390/jof8090962
Atta S, Perera C, Kowalski RP, Jhanji V. Fungal Keratitis: Clinical Features, Risk Factors, Treatment, and Outcomes. Journal of Fungi. 2022; 8(9):962. https://doi.org/10.3390/jof8090962
Chicago/Turabian StyleAtta, Sarah, Chandrashan Perera, Regis P. Kowalski, and Vishal Jhanji. 2022. "Fungal Keratitis: Clinical Features, Risk Factors, Treatment, and Outcomes" Journal of Fungi 8, no. 9: 962. https://doi.org/10.3390/jof8090962
APA StyleAtta, S., Perera, C., Kowalski, R. P., & Jhanji, V. (2022). Fungal Keratitis: Clinical Features, Risk Factors, Treatment, and Outcomes. Journal of Fungi, 8(9), 962. https://doi.org/10.3390/jof8090962







