Management of Filamentous Fungal Keratitis: A Pragmatic Approach
Abstract
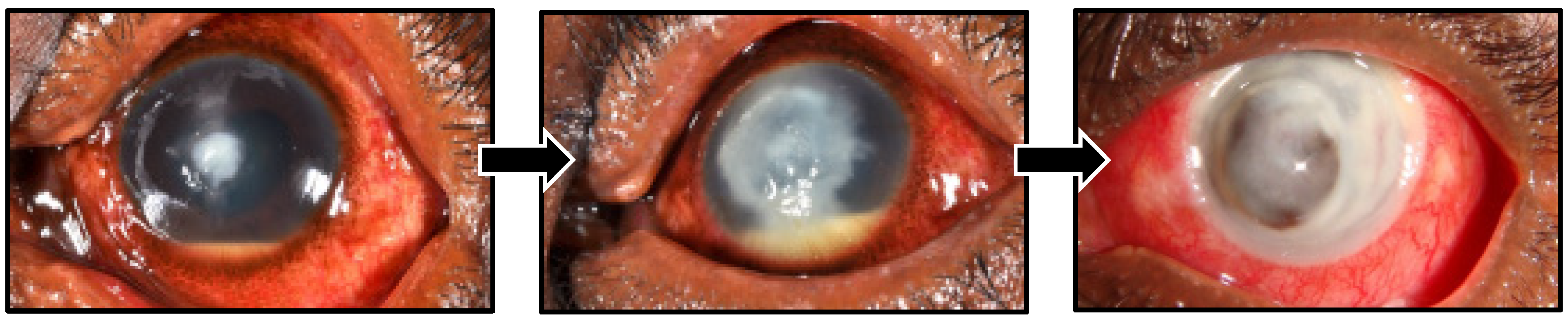
1. General Management of Fungal Keratitis
2. Medical Management of FK
2.1. Topical Treatment
2.2. Natamycin, Voriconazole and Econazole
2.3. Amphotericin B, Fluconazole, and Echinocandins
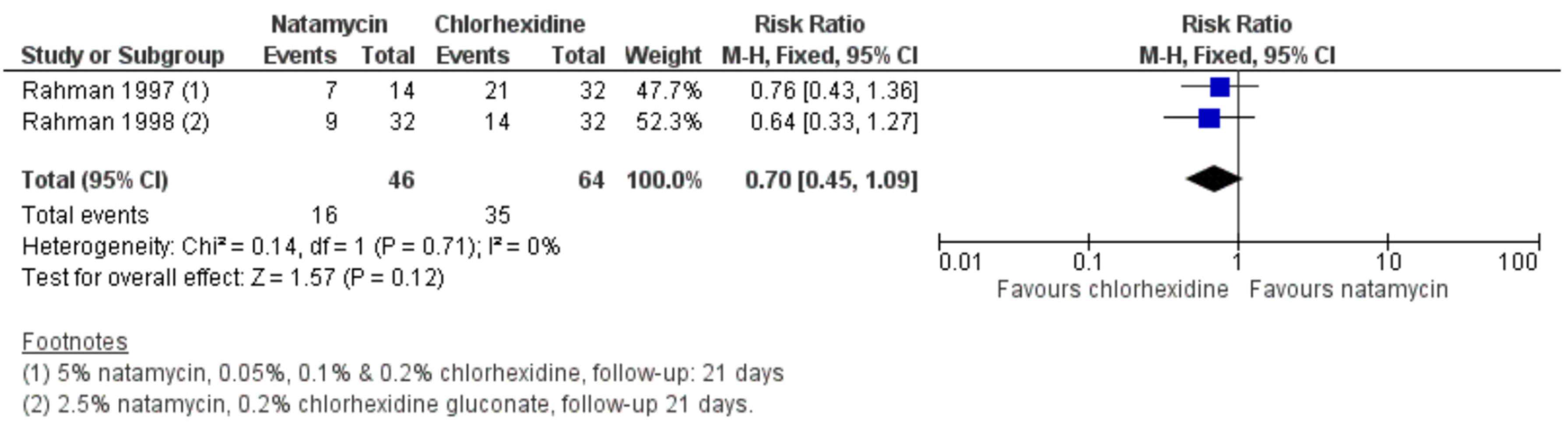
2.4. Chlorhexidine for Fungal Keratitis
2.5. Oral Treatment
2.6. Injected Drug Delivery
3. Surgical Treatment
3.1. Therapeutic Penetrating Keratoplasty
3.2. Lamellar Keratoplasty
3.3. Amniotic Membrane Grafts and Conjunctival Flaps
3.4. Corneal Collagen Cross-Linking
3.5. Argon Laser for Fungal Keratitis
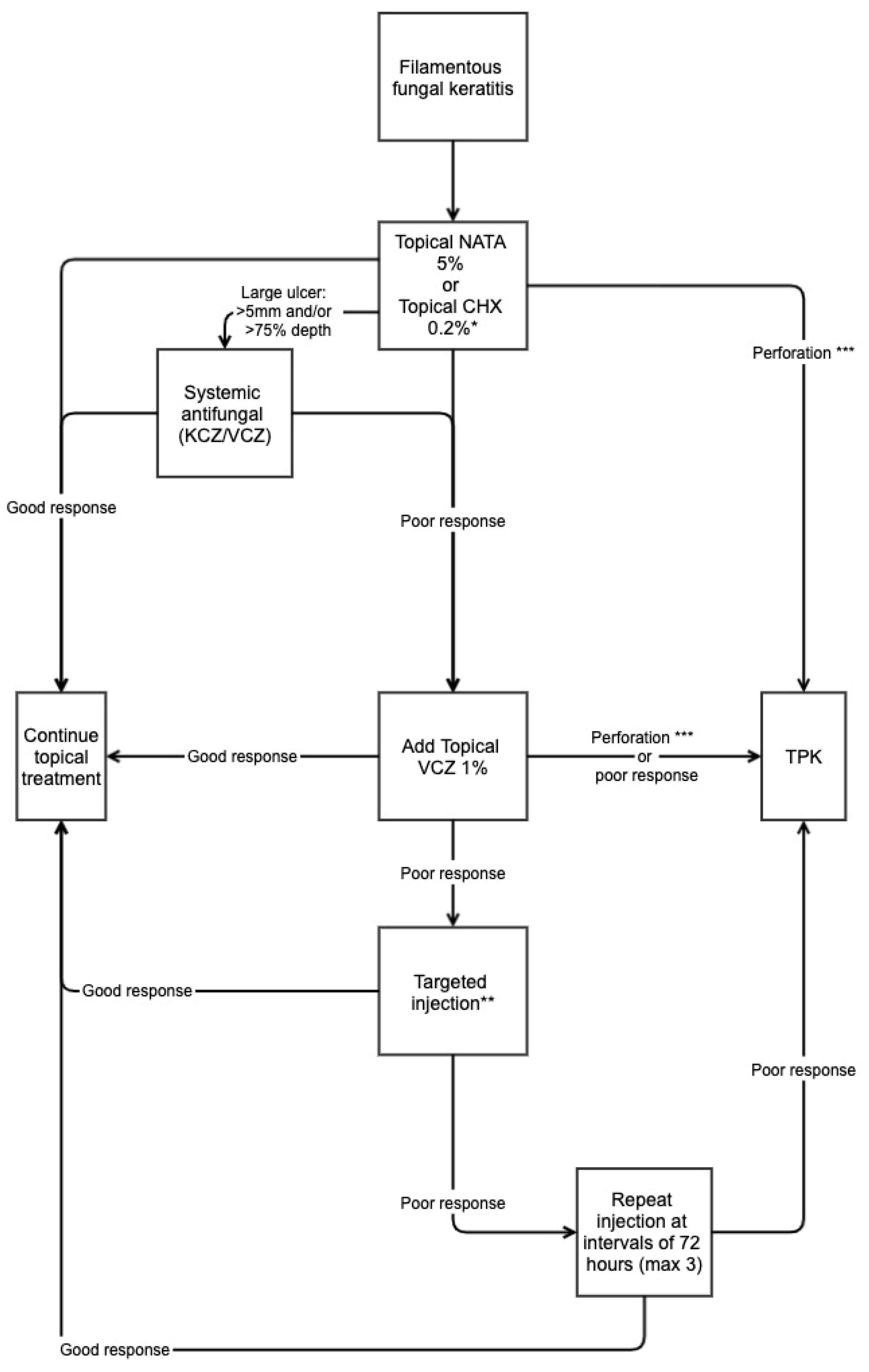
4. Management Strategy: A Pragmatic Approach
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Patil, A.; Lakhani, P.; Majumdar, S. Current perspectives on natamycin in ocular fungal infections. J. Drug Deliv. Sci. Technol. 2017, 41, 206–212. [Google Scholar] [CrossRef]
- Ghannoum, M.A.; Rice, L.B. Antifungal agents: Mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin. Microbiol. Rev. 1999, 12, 501–517. [Google Scholar] [CrossRef] [PubMed]
- Borelli, C.; Schaller, M.; Niewerth, M.; Nocker, K.; Baasner, B.; Berg, D.; Tiemann, R.; Tietjen, K.; Fugmann, B.; Lang-Fugmann, S.; et al. Modes of action of the new arylguanidine abafungin beyond interference with ergosterol biosynthesis and in vitro activity against medically important fungi. Chemotherapy 2008, 54, 245–259. [Google Scholar] [CrossRef]
- Lakhani, P.; Patil, A.; Majumdar, S. Challenges in the polyene- and azole-based pharmacotherapy of ocular fungal infections. J. Ocul. Pharmacol. Ther. 2019, 35, 6–22. [Google Scholar] [CrossRef]
- FlorCruz, N.V.; Evans, J.R. Medical interventions for fungal keratitis. Cochrane Database Syst. Rev. 2015, 4, CD004241. [Google Scholar] [CrossRef] [PubMed]
- Schein, O.D. Evidence-based treatment of fungal keratitis. JAMA Ophthalmol. 2016, 134, 1372–1373. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Zhao, G.Q.; Lin, J.; Wang, X.; Hu, L.T.; Du, Z.D.; Wang, Q.; Zhu, C.C. Natamycin in the treatment of fungal keratitis: A systematic review and Meta-analysis. Int. J. Ophthalmol. 2015, 8, 597–602. [Google Scholar] [CrossRef]
- Lalitha, P.; Vijaykumar, R.; Prajna, N.V.; Fothergill, A.W. In vitro natamycin susceptibility of ocular isolates of Fusarium and Aspergillus species: Comparison of commercially formulated natamycin eye drops to pharmaceutical-grade powder. J. Clin. Microbiol. 2008, 46, 3477–3478. [Google Scholar] [CrossRef]
- Sharma, N.; Bagga, B.; Singhal, D.; Nagpal, R.; Kate, A.; Saluja, G.; Maharana, P.K. Fungal keratitis: A review of clinical presentations, treatment strategies and outcomes. Ocul. Surf. 2022, 24, 22–30. [Google Scholar] [CrossRef]
- Parchand, S.; Gupta, A.; Ram, J.; Gupta, N.; Chakrabarty, A. Voriconazole for fungal corneal ulcers. Ophthalmology 2012, 119, 1083–1083.e3. [Google Scholar] [CrossRef]
- Prajna, N.V.; Mascarenhas, J.; Krishnan, T.; Reddy, P.R.; Prajna, L.; Srinivasan, M.; Vaitilingam, C.M.; Hong, K.C.; Lee, S.M.; McLeod, S.D.; et al. Comparison of natamycin and voriconazole for the treatment of fungal keratitis. Arch. Ophthalmol. (Chic. Ill. 1960) 2010, 128, 672–678. [Google Scholar] [CrossRef]
- Prajna, N.V.; Krishnan, T.; Mascarenhas, J.; Rajaraman, R.; Prajna, L.; Srinivasan, M.; Raghavan, A.; Oldenburg, C.E.; Ray, K.J.; Zegans, M.E.; et al. The Mycotic Ulcer Treatment Trial: A randomized trial comparing natamycin vs. voriconazole. JAMA Ophthalmol. 2013, 131, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Prajna, N.V.; Krishnan, T.; Rajaraman, R.; Patel, S.; Srinivasan, M.; Das, M.; Ray, K.J.; O’Brien, K.S.; Oldenburg, C.E.; McLeod, S.D.; et al. Effect of oral voriconazole on fungal keratitis in the Mycotic Ulcer Treatment Trial II (MUTT II): A randomized clinical trial. JAMA Ophthalmol. 2016, 134, 1365–1372. [Google Scholar] [CrossRef]
- Sharma, S.; Das, S.; Virdi, A.; Fernandes, M.; Sahu, S.K.; Koday, N.K.; Ali, M.H.; Garg, P.; Motukupally, S.R. Re-appraisal of topical 1% voriconazole and 5% natamycin in the treatment of fungal keratitis in a randomised trial. Br. J. Ophthalmol. 2015, 99, 1190–1195. [Google Scholar] [CrossRef] [PubMed]
- Prajna, N.V.; John, R.K.; Nirmalan, P.K.; Lalitha, P.; Srinivasan, M. A randomised clinical trial comparing 2% econazole and 5% natamycin for the treatment of fungal keratitis. Br. J. Ophthalmol. 2003, 87, 1235–1237. [Google Scholar] [CrossRef]
- Bunya, V.Y.; Hammersmith, K.M.; Rapuano, C.J.; Ayres, B.D.; Cohen, E.J. Topical and oral voriconazole in the treatment of fungal keratitis. Am. J. Ophthalmol. 2007, 143, 151–153. [Google Scholar] [CrossRef]
- Diekema, D.J.; Messer, S.A.; Hollis, R.J.; Jones, R.N.; Pfaller, M.A. Activities of caspofungin, itraconazole, posaconazole, ravuconazole, voriconazole, and amphotericin b against 448 recent clinical isolates of filamentous fungi. J. Clin. Microbiol. 2003, 41, 3623–3626. [Google Scholar] [CrossRef] [PubMed]
- McDonald, E.M.; Ram, F.S.; Patel, D.V.; McGhee, C.N. Effectiveness of topical antifungal drugs in the management of fungal keratitis: A systematic review and meta-analysis of randomized controlled trials. Asia Pac. J. Ophthalmol. 2014, 3, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Arora, R.; Gupta, D.; Goyal, J.; Kaur, R. Voriconazole versus natamycin as primary treatment in fungal corneal ulcers. Clin. Exp. Ophthalmol. 2011, 39, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Burton, M.J.; Pithuwa, J.; Okello, E.; Afwamba, I.; Onyango, J.J.; Oates, F.; Chevallier, C.; Hall, A.B. Microbial keratitis in East Africa: Why are the outcomes so poor? Ophthalmic Epidemiol. 2011, 18, 158–163. [Google Scholar] [CrossRef]
- Velpandian, T.; Nirmal, J.; Sharma, H.P.; Sharma, S.; Sharma, N.; Halder, N. Novel water soluble sterile natamycin formulation (Natasol) for fungal keratitis. Eur. J. Pharm. Sci. 2021, 163, 105857. [Google Scholar] [CrossRef]
- World Health Organization. World Health Organization Model List of Essential Medicines, 21st List, 2019; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Lotery, A.J.; Kerr, J.R.; Page, B.A. Fungal keratitis caused by Scopulariopsis brevicaulis: Successful treatment with topical amphotericin B and chloramphenicol without the need for surgical debridement. Br. J. Ophthalmol. 1994, 78, 730. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mahdy, R.A.; Nada, W.M.; Wageh, M.M. Topical amphotericin B and subconjunctival injection of fluconazole (combination therapy) versus topical amphotericin B (monotherapy) in treatment of keratomycosis. J. Ocul. Pharmacol. Ther. 2010, 26, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.L.; Jones, D.B.; Wilhelmus, K.R. Clinical characteristics and outcome of Candida keratitis. Am. J. Ophthalmol. 2007, 143, 1043–1045. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, T.P. Therapy of ocular fungal infections. Ophthalmol. Clin. N. Am. 1999, 12, 33–50. [Google Scholar] [CrossRef]
- Rao, S.K.; Madhavan, H.N.; Rao, G.; Padmanabhan, P. Fluconazole in filamentous fungal keratitis. Cornea 1997, 16, 700. [Google Scholar] [PubMed]
- Sonego-Krone, S.; Martino, D.S.-D.; Ayala-Lugo, R.; Torres-Alvariza, G.; Ta, C.N.; Barbosa, L.; de Kaspar, H.M. Clinical results of topical fluconazole for the treatment of filamentous fungal keratitis. Graefes Arch. Clin. Exp. Ophthalmol. 2006, 244, 782–787. [Google Scholar] [CrossRef]
- Patil, A.; Majumdar, S. Echinocandins in ocular therapeutics. J. Ocul. Pharmacol. Ther. 2017, 33, 340–352. [Google Scholar] [CrossRef]
- Chen, S.C.; Slavin, M.A.; Sorrell, T.C. Echinocandin antifungal drugs in fungal infections: A comparison. Drugs 2011, 71, 11–41. [Google Scholar] [CrossRef]
- Denning, D.W. Echinocandins: A new class of antifungal. J. Antimicrob. Chemother. 2002, 49, 889–891. [Google Scholar] [CrossRef]
- Goldblum, D.; Frueh, B.E.; Sarra, G.M.; Katsoulis, K.; Zimmerli, S. Topical caspofungin for treatment of keratitis caused by Candida albicans in a rabbit model. Antimicrob. Agents Chemother. 2005, 49, 1359–1363. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kusbeci, T.; Avci, B.; Cetinkaya, Z.; Ozturk, F.; Yavas, G.; Ermis, S.S.; Inan, U.U. The effects of caspofungin and voriconazole in experimental Candida endophthalmitis. Curr. Eye Res. 2007, 32, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Vorwerk, C.K.; Tuchen, S.; Streit, F.; Binder, L.; Hofmüller, W.; Behrens-Baumann, W. Aqueous humor concentrations of topically administered caspofungin in rabbits. Ophthalmic Res. 2009, 41, 102–105. [Google Scholar] [CrossRef]
- Shen, Y.C.; Liang, C.Y.; Wang, C.Y.; Lin, K.H.; Hsu, M.Y.; Yuen, H.L.; Wei, L.C. Pharmacokinetics and safety of intravitreal caspofungin. Antimicrob. Agents Chemother. 2014, 58, 7234–7239. [Google Scholar] [CrossRef]
- Kernt, M.; Kampik, A. Intraocular caspofungin: In vitro safety profile for human ocular cells. Mycoses 2011, 54, e110–e121. [Google Scholar] [CrossRef]
- Tu, E.Y. Alternaria Keratitis: Clinical presentation and resolution with topical fluconazole or intrastromal voriconazole and topical caspofungin. Cornea 2009, 28, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Neoh, C.F.; Leung, L.; Vajpayee, R.B.; Stewart, K.; Kong, D.C. Treatment of Alternaria keratitis with intrastromal and topical caspofungin in combination with intrastromal, topical, and oral voriconazole. Ann. Pharmacother. 2011, 45, 681. [Google Scholar] [CrossRef]
- Mitani, A.; Shiraishi, A.; Miyamoto, H.; Sunada, A.; Ueda, A.; Asari, S.; Zheng, X.; Yamamoto, Y.; Hara, Y.; Ohashi, Y. Fungal keratitis caused by Beauveria bassiana: Drug and temperature sensitivity profiles: A case report. BMC Res. Notes 2014, 7, 677. [Google Scholar] [CrossRef]
- Kokuzawa, S.; Suemori, S.; Mochizuki, K.; Hirose, Y.; Yaguchi, T. Aspergillus tubingenesis endophthalmitis after cataract surgery with implantation of preloaded intraocular lens. Semin. Ophthalmol. 2014, 29, 218–221. [Google Scholar] [CrossRef]
- Kamoshita, M.; Matsumoto, Y.; Nishimura, K.; Katono, Y.; Murata, M.; Ozawa, Y.; Shimmura, S.; Tsubota, K. Wickerhamomyces anomalus fungal keratitis responds to topical treatment with antifungal micafungin. J. Infect. Chemother. 2015, 21, 141–143. [Google Scholar] [CrossRef]
- McDonnell, G.; Russell, A.D. Antiseptics and disinfectants: Activity, action, and resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar] [CrossRef] [PubMed]
- Shariff, J.A.; Lee, K.C.; Leyton, A.; Abdalal, S. Neonatal mortality and topical application of chlorhexidine on umbilical cord stump: A meta-analysis of randomized control trials. Public Health 2016, 139, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Hu, B.; Liu, Y.; Yang, Z.; Song, J. The efficacy of intra-alveolar 0.2% chlorhexidine gel on alveolar osteitis: A meta-analysis. Oral Dis. 2017, 23, 598–608. [Google Scholar] [CrossRef] [PubMed]
- James, P.; Worthington, H.V.; Parnell, C.; Harding, M.; Lamont, T.; Cheung, A.; Whelton, H.; Riley, P. Chlorhexidine mouthrinse as an adjunctive treatment for gingival health. Cochrane Database Syst. Rev. 2017, 3, CD008676. [Google Scholar] [CrossRef] [PubMed]
- Nittayananta, W.; DeRouen, T.A.; Arirachakaran, P.; Laothumthut, T.; Pangsomboon, K.; Petsantad, S.; Vuddhakul, V.; Sriplung, H.; Jaruratanasirikul, S.; Martin, M.D. A randomized clinical trial of chlorhexidine in the maintenance of oral candidiasis-free period in HIV infection. Oral Dis. 2008, 14, 665–670. [Google Scholar] [CrossRef]
- Ellepola, A.N.; Samaranayake, L.P. Adjunctive use of chlorhexidine in oral candidoses: A review. Oral Dis. 2001, 7, 11–17. [Google Scholar] [CrossRef]
- Ong, H.S.; Fung, S.S.M.; Macleod, D.; Dart, J.K.G.; Tuft, S.J.; Burton, M.J. Altered patterns of fungal keratitis at a London ophthalmic referral hospital: An eight-year retrospective observational study. Am. J. Ophthalmol. 2016, 168, 227–236. [Google Scholar] [CrossRef]
- Rahman, M.R.; Minassian, D.C.; Srinivasan, M.; Martin, M.J.; Johnson, G.J. Trial of chlorhexidine gluconate for fungal corneal ulcers. Ophthalmic Epidemiol. 1997, 4, 141–149. [Google Scholar] [CrossRef]
- Rahman, M.R.; Johnson, G.J.; Husain, R.; Howlader, S.A.; Minassian, D.C. Randomised trial of 0.2% chlorhexidine gluconate and 2.5% natamycin for fungal keratitis in Bangladesh. Br. J. Ophthalmol. 1998, 82, 919–925. [Google Scholar] [CrossRef]
- Dart, J.K.G.; Saw, V.P.J.; Kilvington, S. Acanthamoeba keratitis: Diagnosis and treatment update 2009. Am. J. Ophthalmol. 2009, 148, 487–499.e482. [Google Scholar] [CrossRef]
- Kosrirukvongs, P.; Wanachiwanawin, D.; Visvesvara, G.S. Treatment of Acanthamoeba keratitis with chlorhexidine. Ophthalmology 1999, 106, 798–802. [Google Scholar] [CrossRef]
- Seal, D.; Hay, J.; Kirkness, C.; Morrell, A.; Booth, A.; Tullo, A.; Ridgway, A.; Armstrong, M. Successful medical therapy of Acanthamoeba keratitis with topical chlorhexidine and propamidine. Eye 1996, 10, 413–421. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Martin, M.J.; Rahman, M.R.; Johnson, G.J.; Srinivasan, M.; Clayton, Y.M. Mycotic keratitis: Susceptibility to antiseptic agents. Int. Ophthalmol. 1995, 19, 299–302. [Google Scholar] [CrossRef]
- Hoffman, J.J.; Yadav, R.; Das Sanyam, S.; Chaudhary, P.; Roshan, A.; Singh, S.K.; Arunga, S.; Matayan, E.; Macleod, D.; Weiss, H.A.; et al. Topical chlorhexidine 0.2% versus topical natamycin 5% for fungal keratitis in Nepal: Rationale and design of a randomised controlled non-inferiority trial. BMJ Open 2020, 10, e038066. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.J.; Yadav, R.; Sanyam, S.D.; Chaudhary, P.; Roshan, A.; Singh, S.K.; Singh, S.K.; Mishra, S.K.; Arunga, S.; Hu, V.H.; et al. Topical chlorhexidine 0·2% versus topical natamycin 5% for the treatment of fungal keratitis in Nepal: A randomised controlled non-inferiority trial. Ophthalmology 2022, 129, 530–541. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.; Leck, A.K.; Gichangi, M.; Burton, M.J.; Denning, D.W. The global incidence and diagnosis of fungal keratitis. Lancet Infect. Dis. 2021, 21, e49–e57. [Google Scholar] [CrossRef]
- Arunga, S.; Mbarak, T.; Ebong, A.; Mwesigye, J.; Kuguminkiriza, D.; Mohamed-Ahmed, A.H.A.; Hoffman, J.J.; Leck, A.; Hu, V.; Burton, M. Chlorhexidine gluconate 0.2% as a treatment for recalcitrant fungal keratitis in Uganda: A pilot study. BMJ Open Ophthalmol. 2021, 6, e000698. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.; Arunga, S.; Matayan, E.; Burton, M.J. A Comparison of Two Treatment Regimes for the Treatment of Fungal Eye Infections in East Africa. In ISRCTN Registry; BMC: London, UK, 2021. [Google Scholar] [CrossRef]
- Agarwal, P.K.; Roy, P.; Das, A.; Banerjee, A.; Maity, P.K.; Banerjee, A.R. Efficacy of topical and systemic itraconazole as a broad-spectrum antifungal agent in mycotic corneal ulcer. A preliminary study. Indian J. Ophthalmol. 2001, 49, 173–176. [Google Scholar] [PubMed]
- Sharma, N.; Singhal, D.; Maharana, P.K.; Sinha, R.; Agarwal, T.; Upadhyay, A.D.; Velpandian, T.; Satpathy, G.; Titiyal, J.S. Comparison of oral voriconazole versus oral ketoconazole as an adjunct to topical natamycin in severe fungal keratitis: A randomized controlled trial. Cornea 2017, 36, 1521–1527. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Clinical Excellence. British National Formulary. Available online: https://bnf.nice.org.uk/drugs/voriconazole/ (accessed on 10 June 2022).
- Purkins, L.; Wood, N.; Ghahramani, P.; Greenhalgh, K.; Allen, M.J.; Kleinermans, D. Pharmacokinetics and safety of voriconazole following intravenous- to oral-dose escalation regimens. Antimicrob. Agents Chemother. 2002, 46, 2546–2553. [Google Scholar] [CrossRef] [PubMed]
- Purkins, L.; Wood, N.; Greenhalgh, K.; Allen, M.J.; Oliver, S.D. Voriconazole, a novel wide-spectrum triazole: Oral pharmacokinetics and safety. Br. J. Clin. Pharmacol. 2003, 56, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Medicines and Healthcare Products Regulatory Agency. Voriconazole: Reminder of Risk of Liver Toxicity, Phototoxicity, and Squamous Cell Carcinoma. In Drug Safety Update; HM Government: London, UK, 2014; Volume 7, p. A2. [Google Scholar]
- Sahay, P.; Singhal, D.; Nagpal, R.; Maharana, P.K.; Farid, M.; Gelman, R.; Sinha, R.; Agarwal, T.; Titiyal, J.S.; Sharma, N. Pharmacologic therapy of mycotic keratitis. Surv. Ophthalmol. 2019, 64, 380–400. [Google Scholar] [CrossRef]
- Prakash, G.; Sharma, N.; Goel, M.; Titiyal, J.S.; Vajpayee, R.B. Evaluation of intrastromal injection of voriconazole as a therapeutic adjunctive for the management of deep recalcitrant fungal keratitis. AJOPHT 2008, 146, 56–59. [Google Scholar] [CrossRef]
- Sharma, N.; Agarwal, P.; Sinha, R.; Titiyal, J.S.; Velpandian, T.; Vajpayee, R.B. Evaluation of intrastromal voriconazole injection in recalcitrant deep fungal keratitis: Case series. Br. J. Ophthalmol 2011, 95, 1735–1737. [Google Scholar] [CrossRef]
- Maharana, P.K.; Sharma, N.; Nagpal, R.; Jhanji, V.; Das, S.; Vajpayee, R.B. Recent advances in diagnosis and management of Mycotic Keratitis. Indian J. Ophthalmol. 2016, 64, 346. [Google Scholar] [PubMed]
- Mimouni, M.; Tam, G.; Paitan, Y.; Kidron, D.; Segev, F. Safety and efficacy of intrastromal injection of 5% natamycin in experimental Fusarium keratitis. J. Ocul. Pharmacol. Ther. 2014, 30, 543–547. [Google Scholar] [CrossRef]
- Saluja, G.; Sharma, N.; Agarwal, R.; Sharma, H.P.; Maharana, P.; Satpathy, G.; Titiyal, J.S.; Velpandian, T. Determination of surgical outcomes with a novel formulation of intrastromal natamycin in recalcitrant fungal keratitis: A pilot study. Indian J. Ophthalmol. 2021, 69, 2670–2674. [Google Scholar] [CrossRef]
- Sharma, N.; Chacko, J.; Velpandian, T.; Titiyal, J.S.; Sinha, R.; Satpathy, G.; Tandon, R.; Vajpayee, R.B. Comparative evaluation of topical versus intrastromal voriconazole as an adjunct to natamycin in recalcitrant fungal keratitis. Ophthalmology 2013, 120, 677–681. [Google Scholar] [CrossRef]
- Narayana, S.; Krishnan, T.; Ramakrishnan, S.; Samantaray, P.P.; Austin, A.; Pickel, J.; Porco, T.; Lietman, T.; Rose-Nussbaumer, J. Mycotic Antimicrobial Localized Injection: A randomized clinical trial evaluating intrastromal injection of voriconazole. Ophthalmology 2019, 126, 1084–1089. [Google Scholar] [CrossRef]
- Konar, P.; Joshi, S.; Mandhare, S.J.; Thakur, R.; Deshpande, M.; Dayal, A. Intrastromal voriconazole: An adjuvant approach for recalcitrant mycotic keratitis. Indian J. Ophthalmol. 2020, 68, 35–38. [Google Scholar] [CrossRef]
- Kalaiselvi, G.; Narayana, S.; Krishnan, T.; Sengupta, S. Intrastromal voriconazole for deep recalcitrant fungal keratitis: A case series. Br. J. Ophthalmol. 2015, 99, 195–198. [Google Scholar] [CrossRef]
- Sharma, N.; Sahay, P.; Maharana, P.K.; Singhal, D.; Saluja, G.; Bandivadekar, P.; Chako, J.; Agarwal, T.; Sinha, R.; Titiyal, J.S.; et al. Management algorithm for fungal keratitis: The TST (Topical, Systemic, and Targeted Therapy) Protocol. Cornea 2019, 38, 141–145. [Google Scholar] [CrossRef]
- Mittal, V.; Mittal, R. Intracameral and topical voriconazole for fungal corneal endoexudates. Cornea 2012, 31, 366–370. [Google Scholar] [CrossRef]
- Shen, Y.-C.; Wang, C.-Y.; Tsai, H.-Y.; Lee, H.-N. Intracameral voriconazole injection in the treatment of fungal endophthalmitis resulting from keratitis. Am. J. Ophthalmol. 2010, 149, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.T. Clinical curative effect of irrigating the anterior chamber with solution of amphotericin B to treat the fungal keratitis. Int. J. Ophthalmol. 2011, 11, 1194–1196. [Google Scholar] [CrossRef]
- Sharma, N.; Sankaran, P.; Agarwal, T.; Arora, T.; Chawla, B.; Titiyal, J.S.; Tandon, R.; Satapathy, G.; Vajpayee, R.B. Evaluation of intracameral amphotericin B in the management of fungal keratitis: Randomized controlled trial. Ocul. Immunol. Inflamm. 2016, 24, 493–497. [Google Scholar] [CrossRef]
- Gupta, A.; Thakur, A.; Gupta, S.; Icchpuchany, P.; Tandon, M.; Ram, J.; Chakrabarti, A.; Gosh, A. Early versus delayed intervention with intracameral liposomal amphotericin B in recalcitrant keratomycosis: Experience of a large case series. J. Clin. Diagn. Res. 2019, 13, NC05–NC09. [Google Scholar] [CrossRef]
- Sharma, B.; Kataria, P.; Anand, R.; Gupta, R.; Kumar, K.; Kumar, S.; Gupta, R. Efficacy profile of intracameral amphotericin B. The Often Forgotten Step. Asia Pac. J. Ophthalmol. 2015, 4, 360–366. [Google Scholar] [CrossRef]
- Shao, Y.; Yu, Y.; Pei, C.G.; Tan, Y.H.; Zhou, Q.; Yi, J.L.; Gao, G.P. Therapeutic efficacy of intracameral amphotericin B injection for 60 patients with keratomycosis. Int. J. Ophthalmol. 2010, 3, 257–260. [Google Scholar] [CrossRef]
- Yoon, K.C.; Jeong, I.Y.; Im, S.K.; Chae, H.J.; Yang, S.Y. Therapeutic effect of intracameral amphotericin B injection in the treatment of fungal keratitis. Cornea 2007, 26, 814–818. [Google Scholar] [CrossRef]
- Yilmaz, S.; Ture, M.; Maden, A. Efficacy of intracameral amphotericin B injection in the management of refractory keratomycosis and endophthalmitis. Cornea 2007, 26, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhang, J.; Li, Y.; Han, X.; Zheng, W.; Yang, J.; Xu, G. A combination of intrastromal and intracameral injections of amphotericin B in the treatment of severe fungal keratitis. J. Ophthalmol. 2016, 2016, 3436415. [Google Scholar] [CrossRef] [PubMed]
- Nada, W.M.; Aswad, M.A.A.; El-Haig, W.M. Combined intrastromal injection of amphotericin B and topical fluconazole in the treatment of resistant cases of keratomycosis: A retrospective study. Clin. Ophthalmol. 2017, 11, 871–874. [Google Scholar] [CrossRef] [PubMed]
- Aydin, B.; Cubuk, M.O.; Ucgul, A.; Ertop, M.; Ozmen, M.C.; Atalay, T.; Akata, F. Combined intrastromal voriconazole and amphotericin B treatment for persistent fungal keratitis. Eye Contact Lens 2020, 46, 269–273. [Google Scholar] [CrossRef]
- Saluja, G.; Sharma, N.; Agarwal, R.; Sharma, H.P.; Singhal, D.; Maharana, P.K.; Sinha, R.; Agarwal, T.; Velpandian, T.; Titiyal, J.S.; et al. Comparison of safety and efficacy of intrastromal injections of voriconazole, amphotericin b and natamycin in cases of recalcitrant fungal keratitis: A randomized controlled trial. Clin. Ophthalmol. 2021, 15, 2437–2446. [Google Scholar] [CrossRef] [PubMed]
- Mundra, J.; Dhakal, R.; Mohamed, A.; Jha, G.; Joseph, J.; Chaurasia, S.; Murthy, S. Outcomes of therapeutic penetrating keratoplasty in 198 eyes with fungal keratitis. Indian J. Ophthalmol. 2019, 67, 1599–1605. [Google Scholar] [CrossRef]
- Rautaraya, B.; Sharma, S.; Kar, S.; Das, S.; Sahu, S.K. Diagnosis and treatment outcome of mycotic keratitis at a tertiary eye care center in eastern India. BMC Ophthalmol. 2011, 11, 39. [Google Scholar] [CrossRef]
- Xie, L.; Dong, X.; Shi, W. Treatment of fungal keratitis by penetrating keratoplasty. Br. J. Ophthalmol. 2001, 85, 1070–1074. [Google Scholar] [CrossRef]
- Xie, L.; Zhai, H.; Shi, W. Penetrating keratoplasty for corneal perforations in fungal keratitis. Cornea 2007, 26, 158–162. [Google Scholar] [CrossRef]
- Saha, S.; Sengupta, J.; Banerjee, D.; Saha, S.; Khetan, A.; Mandal, S.M. Systemic evaluation on antifungal susceptibility of keratitis associated fungal pathogens in Eastern India. J. Med. Microbiol. Diagn. 2014, 3, 1. [Google Scholar]
- Prajna, N.V.; Krishnan, T.; Rajaraman, R.; Patel, S.; Shah, R.; Srinivasan, M.; Das, M.; Ray, K.J.; Oldenburg, C.E.; McLeod, S.D.; et al. Predictors of corneal perforation or need for therapeutic keratoplasty in severe fungal keratitis: A secondary analysis of the Mycotic Ulcer Treatment Trial II. JAMA Ophthalmol. 2017, 135, 987–991. [Google Scholar] [CrossRef] [PubMed]
- Bajracharya, L.; Gurung, R. Outcome of therapeutic penetrating keratoplasty in a tertiary eye care center in Nepal. Clin. Ophthalmol. 2015, 9, 2299–2304. [Google Scholar] [CrossRef] [PubMed]
- Selver, O.B.; Egrilmez, S.; Palamar, M.; Arici, M.; Polat, S.H.; Yagci, A. Therapeutic corneal transplant for fungal keratitis refractory to medical therapy. Exp. Clin. Transplant. 2015, 13, 355–359. [Google Scholar] [CrossRef][Green Version]
- Liu, Y.; Jia, H.; Shi, X.; Wang, J.; Ning, Y.; He, B.; Wang, C.; Zheng, X. Minimal trephination penetrating keratoplasty for severe fungal keratitis complicated with hypopyon. Can. J. Ophthalmol. 2013, 48, 529–534. [Google Scholar] [CrossRef]
- Chen, W.L.; Wu, C.Y.; Hu, F.R.; Wang, I.J. Therapeutic penetrating keratoplasty for microbial keratitis in Taiwan from 1987 to 2001. Am. J. Ophthalmol. 2004, 137, 736–743. [Google Scholar] [CrossRef]
- Killingsworth, D.W.; Stern, G.A.; Driebe, W.T.; Knapp, A.; Dragon, D.M. Results of therapeutic penetrating keratoplasty. Ophthalmology 1993, 100, 534–541. [Google Scholar] [CrossRef]
- Yao, Y.F.; Zhang, Y.M.; Zhou, P.; Zhang, B.; Qiu, W.Y.; Tseng, S.C. Therapeutic penetrating keratoplasty in severe fungal keratitis using cryopreserved donor corneas. Br. J. Ophthalmol. 2003, 87, 543–547. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Jain, M.; Sehra, S.V.; Maharana, P.; Agarwal, T.; Satpathy, G.; Vajpayee, R.B. Outcomes of therapeutic penetrating keratoplasty from a tertiary eye care centre in northern India. Cornea 2014, 33, 114–118. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, M.; Xu, M.; Gu, F.; Liu, Q.; Chen, Y.; Zhang, H.; Kijlstra, A. Outcomes of therapeutic keratoplasty for severe infectious keratitis in Chongqing, a 16-year experience. Infect. Drug Resist. 2019, 12, 2487–2493. [Google Scholar] [CrossRef]
- Raj, N.; Vanathi, M.; Ahmed, N.H.; Gupta, N.; Lomi, N.; Tandon, R. Recent perspectives in the management of fungal keratitis. J. Fungi 2021, 7, 907. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, X.; Zhang, X.; Guo, X.; Qi, X.; Li, S.; Shi, W.; Gao, H. Comparison of complications and visual outcomes between big-bubble deep anterior lamellar keratoplasty and penetrating keratoplasty for fungal keratitis. Clin. Exp. Ophthalmol. 2021, 49, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Hu, J.; Shi, W. Treatment failure after lamellar keratoplasty for fungal keratitis. Ophthalmology 2008, 115, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Shi, W.; Liu, Z.; Li, S. Lamellar keratoplasty for the treatment of fungal keratitis. Cornea 2002, 21, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.J.; Xie, L.X. Changing indications for lamellar keratoplasty in Shandong, 1993–2008. Chin. Med. J. 2010, 123, 3268–3271. [Google Scholar] [PubMed]
- Sabatino, F.; Sarnicola, E.; Sarnicola, C.; Tosi, G.M.; Perri, P.; Sarnicola, V. Early deep anterior lamellar keratoplasty for fungal keratitis poorly responsive to medical treatment. Eye 2017, 31, 1639–1646. [Google Scholar] [CrossRef]
- Anwar, M.; Teichmann, K.D. Deep lamellar keratoplasty: Surgical techniques for anterior lamellar keratoplasty with and without baring of Descemet’s membrane. Cornea 2002, 21, 374–383. [Google Scholar] [CrossRef]
- Anwar, M.; Teichmann, K.D. Big-bubble technique to bare Descemet’s membrane in anterior lamellar keratoplasty. J. Cataract. Refract. Surg. 2002, 28, 398–403. [Google Scholar] [CrossRef]
- Zhang, M.C.; Liu, X.; Jin, Y.; Jiang, D.L.; Wei, X.S.; Xie, H.T. Lamellar keratoplasty treatment of fungal corneal ulcers with acellular porcine corneal stroma. Am. J. Transplant. 2015, 15, 1068–1075. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhang, Y.; Ren, Y.; Zhao, Z.; Hua, S.; Li, J.; Wang, H.; Ye, C.; Kim, A.D.; Wang, L.; et al. Deep anterior lamellar keratoplasty with cross-linked acellular porcine corneal stroma to manage fungal keratitis. Xenotransplantation 2021, 28, e12655. [Google Scholar] [CrossRef]
- Reinhart, W.J.; Musch, D.C.; Jacobs, D.S.; Lee, W.B.; Kaufman, S.C.; Shtein, R.M. Deep anterior lamellar keratoplasty as an alternative to penetrating keratoplasty: A report by the American Academy of Ophthalmology. Ophthalmology 2011, 118, 209–218. [Google Scholar] [CrossRef]
- Singh, G.; Malik, S.R. Therapeutic keratoplasty in fungal corneal ulcers. Br. J. Ophthalmol. 1972, 56, 41–45. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Anshu, A.; Parthasarathy, A.; Mehta, J.S.; Htoon, H.M.; Tan, D.T. Outcomes of therapeutic deep lamellar keratoplasty and penetrating keratoplasty for advanced infectious keratitis: A comparative study. Ophthalmology 2009, 116, 615–623. [Google Scholar] [CrossRef]
- Van Herendael, B.J.; Oberti, C.; Brosens, I. Microanatomy of the human amniotic membranes. A light microscopic, transmission, and scanning electron microscopic study. Am. J. Obstet. Gynecol. 1978, 131, 872–880. [Google Scholar] [CrossRef]
- Dua, H.S.; Gomes, J.A.; King, A.J.; Maharajan, V.S. The amniotic membrane in ophthalmology. Surv. Ophthalmol. 2004, 49, 51–77. [Google Scholar] [CrossRef] [PubMed]
- Jirsova, K.; Jones, G.L.A. Amniotic membrane in ophthalmology: Properties, preparation, storage and indications for grafting-a review. Cell Tissue Bank. 2017, 18, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Ting, D.S.J.; Henein, C.; Said, D.G.; Dua, H.S. Amniotic membrane transplantation for infectious keratitis: A systematic review and meta-analysis. Sci. Rep. 2021, 11, 13007. [Google Scholar] [CrossRef]
- Miri, A.; Al-Deiri, B.; Dua, H.S. Long-term outcomes of autolimbal and allolimbal transplants. Ophthalmology 2010, 117, 1207–1213. [Google Scholar] [CrossRef]
- Berguiga, M.; Mameletzi, E.; Nicolas, M.; Rivier, D.; Majo, F. Long-term follow-up of multilayer amniotic membrane transplantation (MLAMT) for non-traumatic corneal perforations or deep ulcers with descemetocele. Klin. Monbl. Augenheilkd. 2013, 230, 413–418. [Google Scholar] [CrossRef]
- Bourcier, T.; Patteau, F.; Borderie, V.; Baudrimont, M.; Rondeau, N.; Bonnel, S.; Chaumeil, C.; Laroche, L. Amniotic membrane transplantation for the treatment severe Acanthamoeba keratitis. Can. J. Ophthalmol. 2004, 39, 621–631. [Google Scholar] [CrossRef]
- Sharma, A.; Mohan, K.; Sharma, R.; Nirankari, V.S. Repositioning of pedicle conjunctival flap performed for refractory corneal ulcer. Middle East Afr. J. Ophthalmol. 2014, 21, 89–91. [Google Scholar] [CrossRef]
- Gundersen, T. Conjunctival flaps in the treatment of corneal disease with reference to a new technique of application. AMA Arch. Ophthalmol. 1958, 60, 880–888. [Google Scholar] [CrossRef] [PubMed]
- Abdulhalim, B.E.; Wagih, M.M.; Gad, A.A.; Boghdadi, G.; Nagy, R.R. Amniotic membrane graft to conjunctival flap in treatment of non-viral resistant infectious keratitis: A randomised clinical study. Br. J. Ophthalmol. 2015, 99, 59–63. [Google Scholar] [CrossRef]
- Zhong, J.; Wang, B.; Li, S.; Deng, Y.; Huang, H.; Chen, L.; Yuan, J. Full-thickness conjunctival flap covering surgery combined with amniotic membrane transplantation for severe fungal keratitis. Exp. Ther. Med. 2018, 15, 2711–2718. [Google Scholar] [CrossRef]
- Garg, P.; Das, S.; Roy, A. Collagen cross-linking for microbial keratitis. Middle East Afr. J. Ophthalmol. 2017, 24, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Alshehri, J.M.; Caballero-Lima, D.; Hillarby, M.C.; Shawcross, S.G.; Brahma, A.; Carley, F.; Read, N.D.; Radhakrishnan, H. Evaluation of corneal cross-linking for treatment of fungal keratitis: Using confocal laser scanning microscopy on an ex vivo human corneal model. Investig. Ophthalmol. Vis. Sci. 2016, 57, 6367–6373. [Google Scholar] [CrossRef]
- Prajna, N.V.; Radhakrishnan, N.; Lalitha, P.; Austin, A.; Ray, K.J.; Keenan, J.D.; Porco, T.C.; Lietman, T.M.; Rose-Nussbaumer, J. Cross-Linking-Assisted Infection Reduction: A randomized clinical trial evaluating the effect of adjuvant cross-linking on outcomes in fungal keratitis. Ophthalmology 2020, 127, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Wei, A.; Wang, K.; Wang, Y.; Gong, L.; Xu, J.; Shao, T. Evaluation of corneal cross-linking as adjuvant therapy for the management of fungal keratitis. Graefes Arch. Clin. Exp. Ophthalmol. 2019, 257, 1443–1452. [Google Scholar] [CrossRef]
- Uddaraju, M.; Mascarenhas, J.; Das, M.R.; Radhakrishnan, N.; Keenan, J.D.; Prajna, L.; Prajna, V.N. Corneal cross-linking as an adjuvant therapy in the management of recalcitrant deep stromal fungal keratitis: A randomized trial. Am. J. Ophthalmol. 2015, 160, 131–134.e135. [Google Scholar] [CrossRef] [PubMed]
- Prajna, N.V.; Radhakrishnan, N.; Lalitha, P.; Liu, Z.; Keenan, J.D.; Arnold, B.F.; Rose-Nussbaumer, J. Mediators of the effect of corneal cross-linking on visual acuity for fungal ulcers: A prespecified secondary analysis from the Cross-Linking-Assisted Infection Reduction Trial. Cornea 2022, 10. [Google Scholar] [CrossRef]
- Mani, V.J.; Parthasarathy, D.; Padmanabhan, P.; Narayanan, N.; Lakshmipathy, M.; Pachayappan, S.K.; Jayavel, P.; Therese, K.L.; Madhavan, H.N.R.; Jambulingam, M. Therapeutic effect of corneal crosslinking on fungal keratitis: Efficacy of corneal collagen crosslinking as an adjuvant therapy for fungal keratitis in a tertiary eye hospital in South India. Ocul. Immunol. Inflamm. 2020, 10, 1–8. [Google Scholar] [CrossRef]
- Ting, D.S.J.; Henein, C.; Said, D.G.; Dua, H.S. Re: Prajna et al.: Cross-Linking-Assisted Infection Reduction (CLAIR): A randomized clinical trial evaluating the effect of adjuvant cross-linking on outcomes in fungal keratitis. Ophthalmology 2020, 127, e55–e56. [Google Scholar] [CrossRef]
- Ting, D.S.J.; Henein, C.; Said, D.G.; Dua, H.S. Photoactivated chromophore for infectious keratitis—Corneal cross-linking (PACK-CXL): A systematic review and meta-analysis. Ocul. Surf. 2019, 17, 624–634. [Google Scholar] [CrossRef]
- Pellegrino, F.; Carrasco, M.A. Argon laser phototherapy in the treatment of refractory fungal keratitis. Cornea 2013, 32, 95–97. [Google Scholar] [CrossRef]
- Fromer, C.; L’Esperance, F. Argon laser phototherapy of pseudomonas corneal ulcers. Investig. Ophthalmol. 1971, 10, 1–8. [Google Scholar]
- Khater, M.M.; El-Shorbagy, M.S.; Selima, A.A. Argon laser photocoagulation versus intrastromal voriconazole injection in treatment of mycotic keratitis. Int. J. Ophthalmol. 2016, 9, 225–229. [Google Scholar] [CrossRef]
- Khater, M.M. Amniotic membrane graft with argon laser photocoagulation versus amniotic membrane graft with tissue debridement for treatment of mycotic keratitis. Semin. Ophthalmol. 2017, 32, 348–352. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoffman, J.J.; Arunga, S.; Mohamed Ahmed, A.H.A.; Hu, V.H.; Burton, M.J. Management of Filamentous Fungal Keratitis: A Pragmatic Approach. J. Fungi 2022, 8, 1067. https://doi.org/10.3390/jof8101067
Hoffman JJ, Arunga S, Mohamed Ahmed AHA, Hu VH, Burton MJ. Management of Filamentous Fungal Keratitis: A Pragmatic Approach. Journal of Fungi. 2022; 8(10):1067. https://doi.org/10.3390/jof8101067
Chicago/Turabian StyleHoffman, Jeremy J., Simon Arunga, Abeer H. A. Mohamed Ahmed, Victor H. Hu, and Matthew J. Burton. 2022. "Management of Filamentous Fungal Keratitis: A Pragmatic Approach" Journal of Fungi 8, no. 10: 1067. https://doi.org/10.3390/jof8101067
APA StyleHoffman, J. J., Arunga, S., Mohamed Ahmed, A. H. A., Hu, V. H., & Burton, M. J. (2022). Management of Filamentous Fungal Keratitis: A Pragmatic Approach. Journal of Fungi, 8(10), 1067. https://doi.org/10.3390/jof8101067





