Involvement of the Autophagy Protein Atg1 in Development and Virulence in Botryosphaeria dothidea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungal Strains and Culture Conditions
2.2. Gene Deletion and Complementation
2.3. Staining and Microscopy
2.4. Protein Manipulation and Western Blotting
2.5. Pathogenicity Tests
2.6. Transcriptome Sequencing and Analysis
3. Results
3.1. Identification of BdATG1 in B. dothidea
3.2. Deletion and Complementation of BdATG1 in B. dothidea
3.3. BdAtg1 Is Indispensable for Autophagy
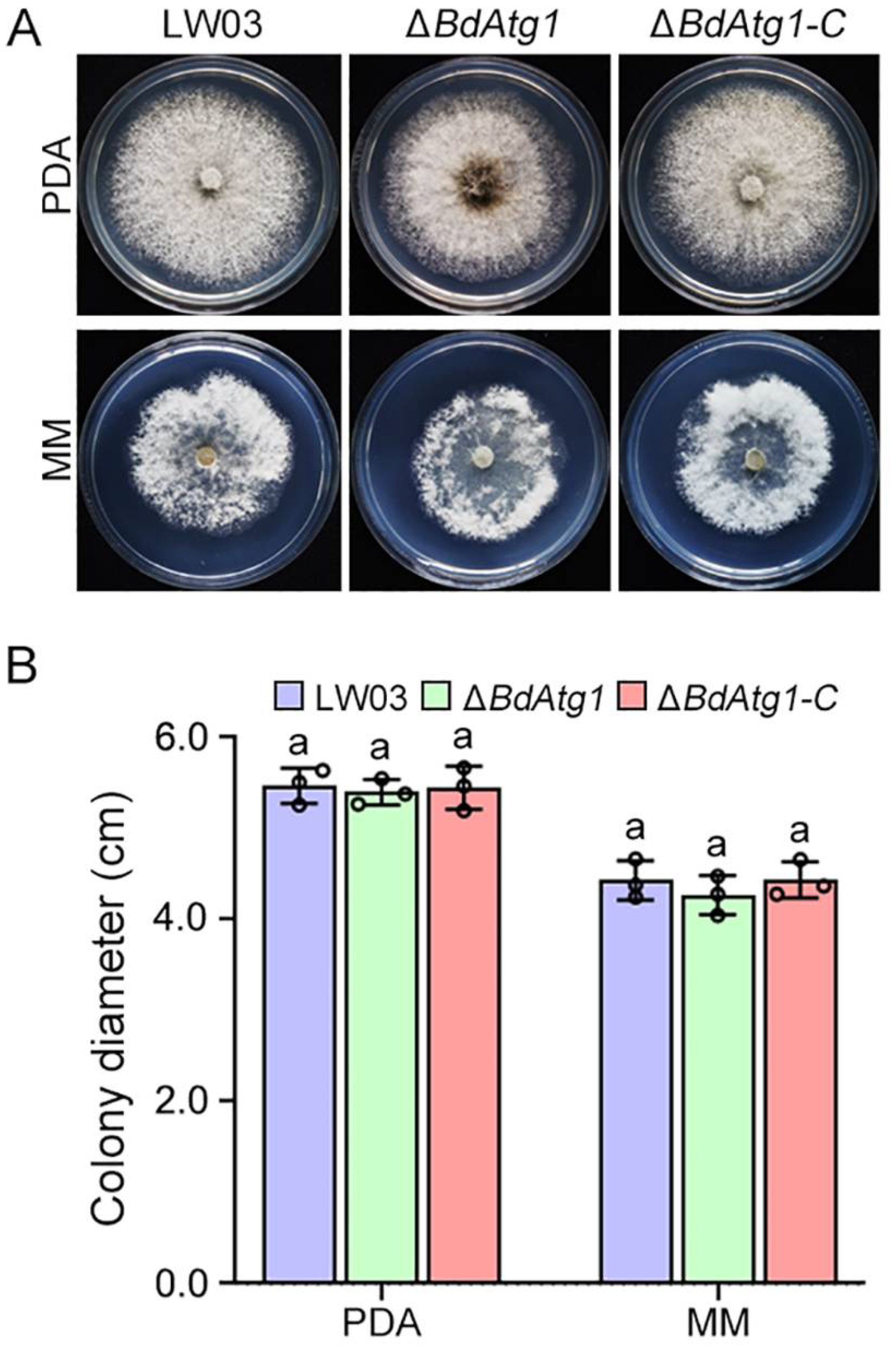
3.4. BdAtg1 Is Involved in Vegetative Growth
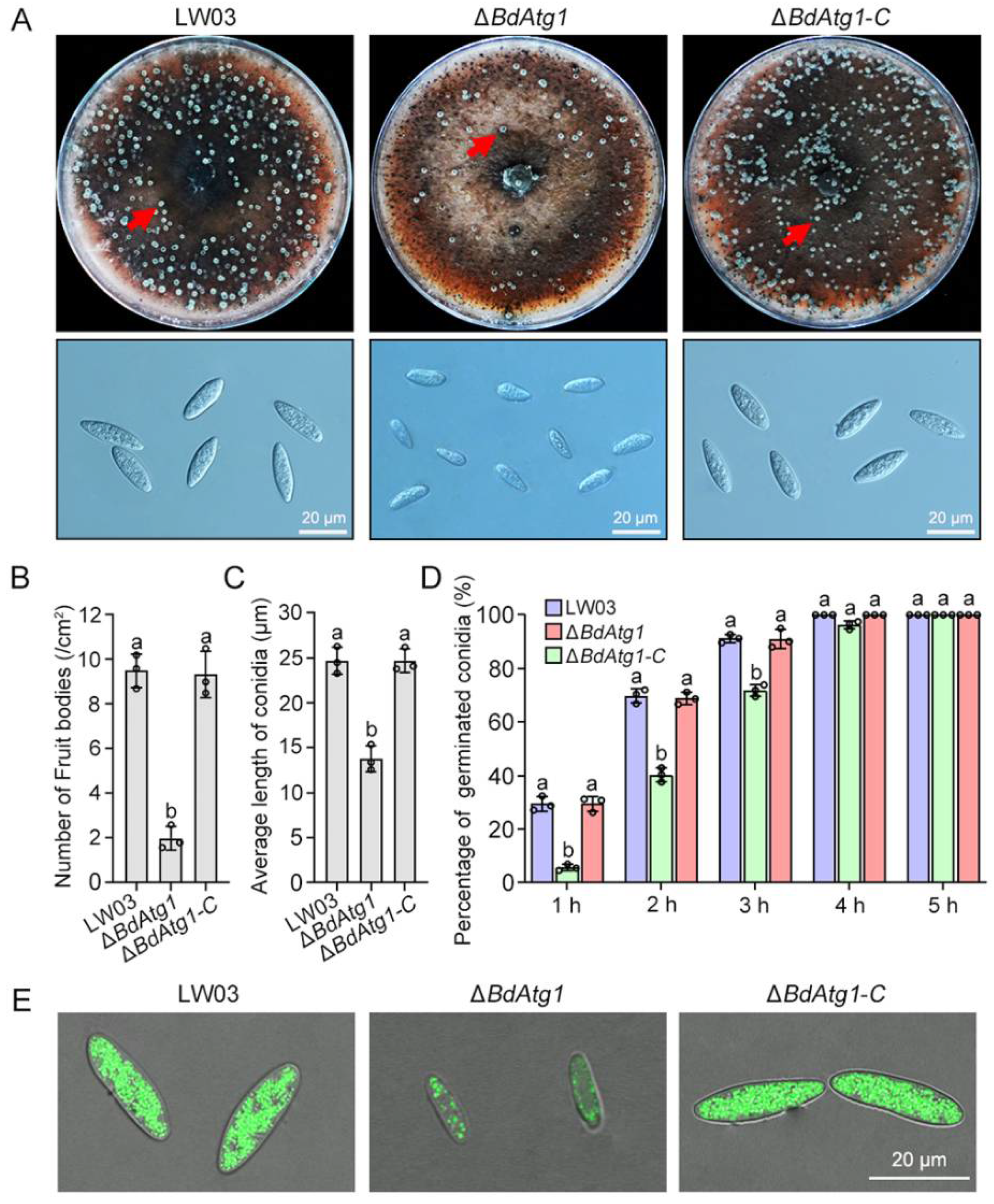
3.5. BdAtg1 Is Crucial for Asexual and Sexual Reproduction
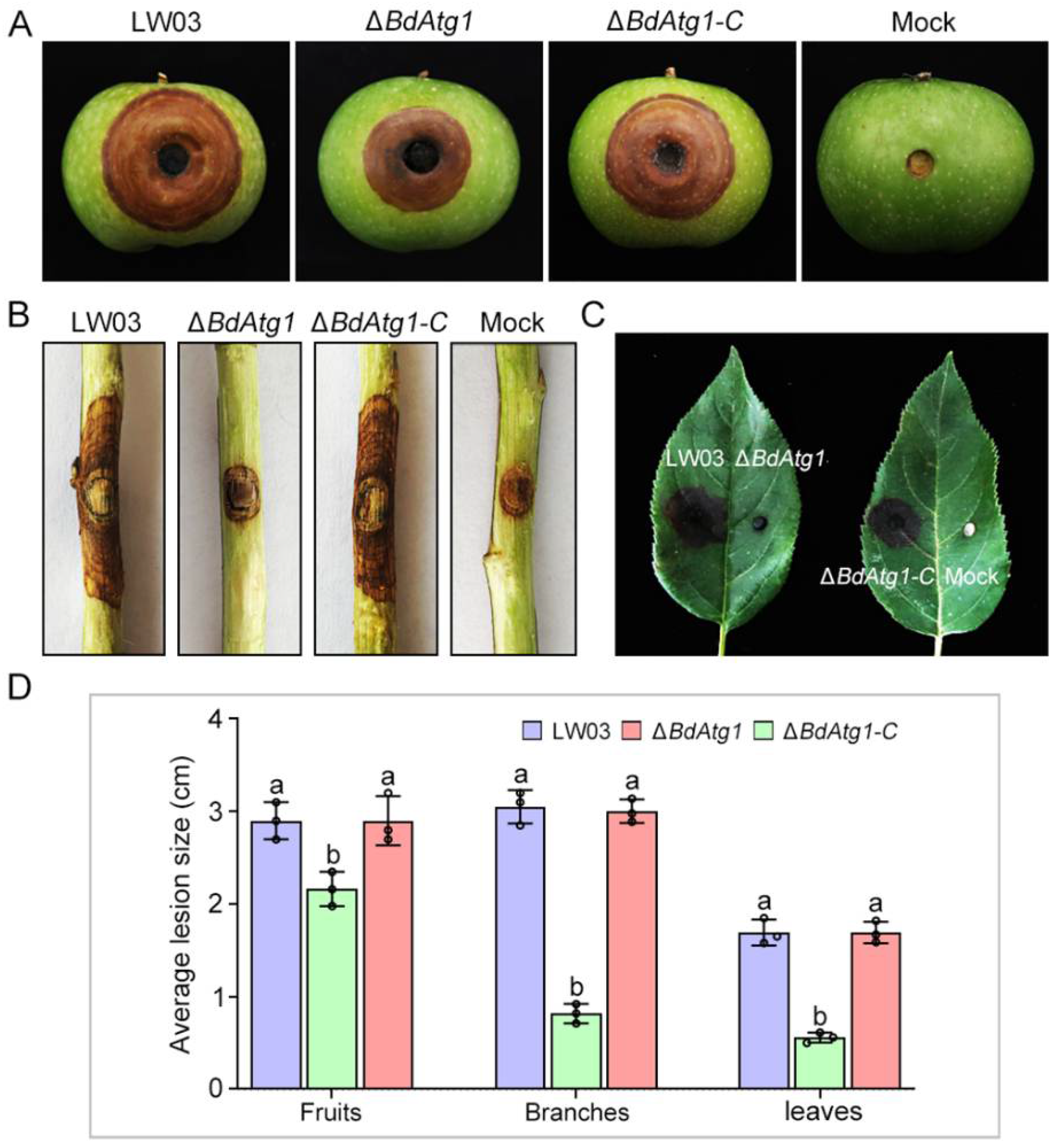
3.6. BdAtg1 Is Required for Full Virulence
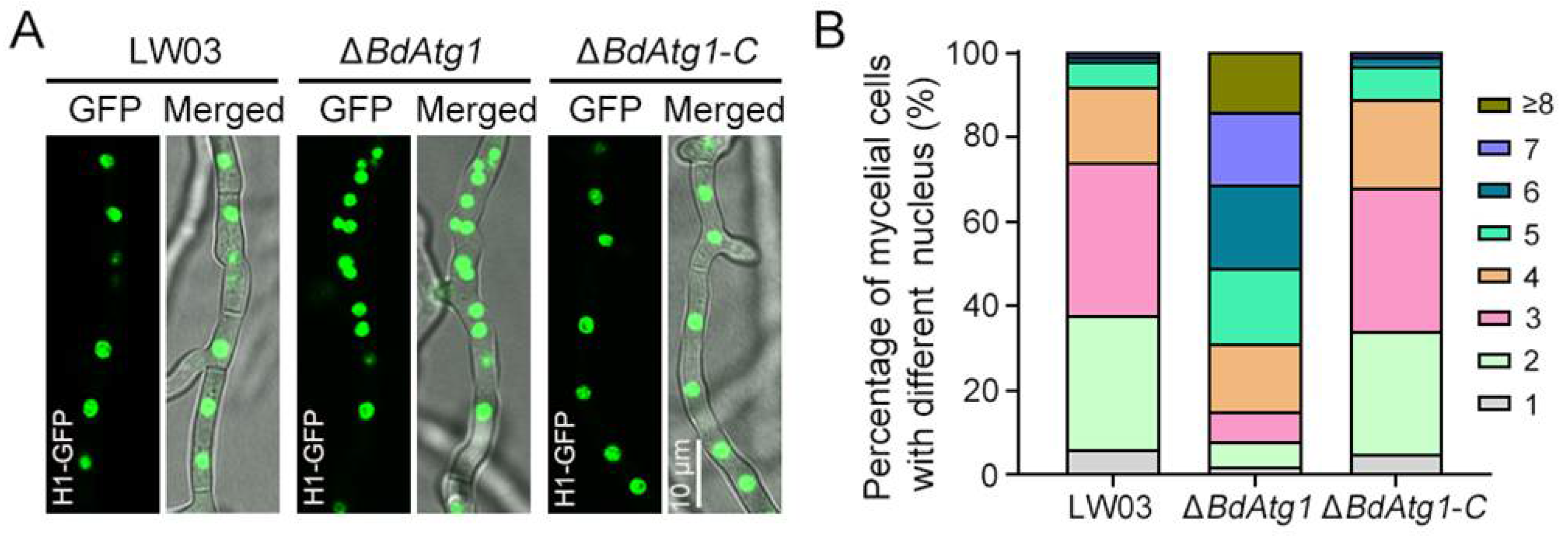
3.7. BdAtg1 Mediates Nuclei Distribution
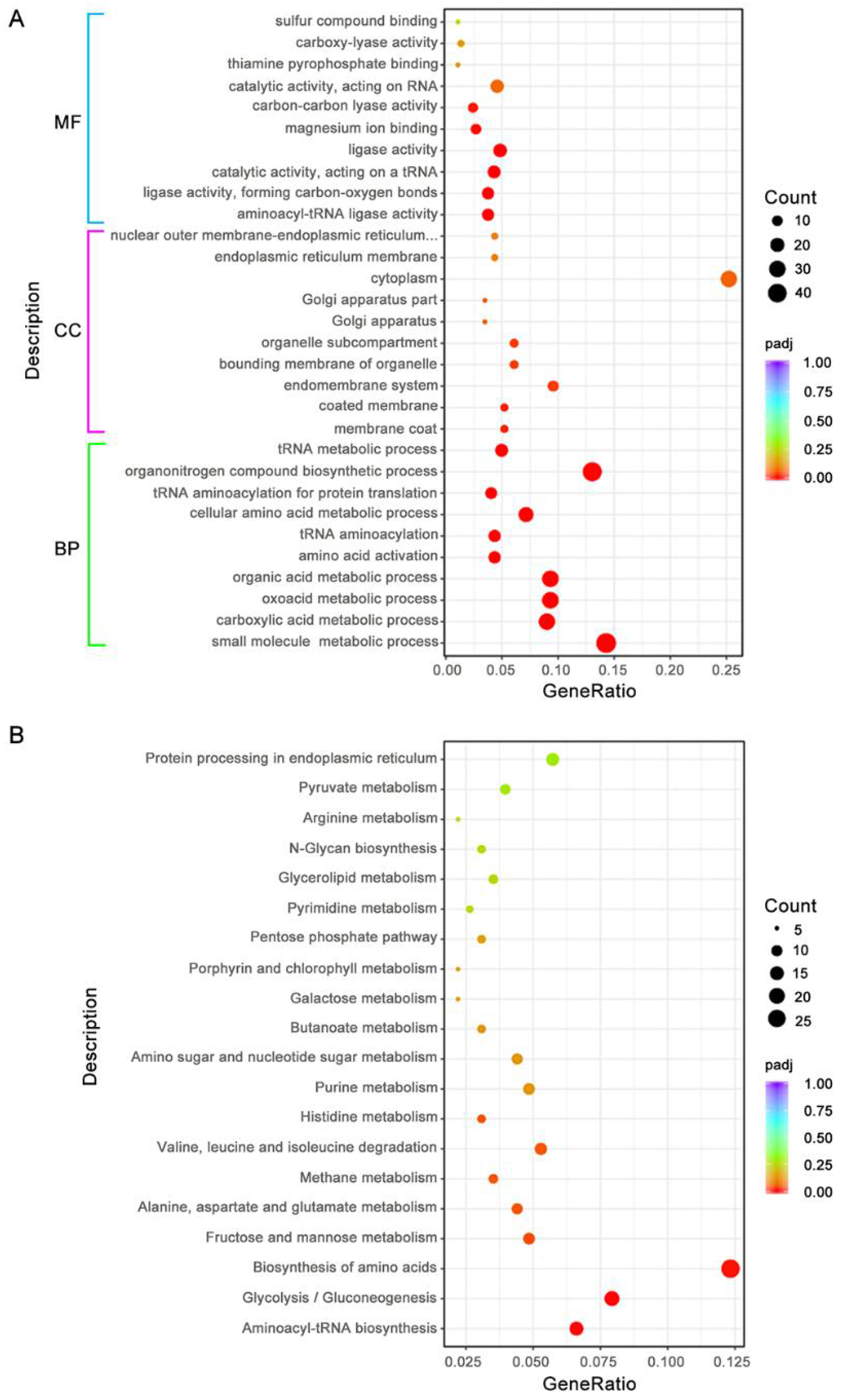
3.8. BdAtg1 Influences Multiple Metabolic Pathways
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dong, X.; Cheng, Z.; Leng, W.; Li, B.; Xu, X.; Lian, S.; Wang, C. Progression of symptoms caused by Botryosphaeria dothidea on apple branches. Phytopathology 2021, 111, 1551–1559. [Google Scholar] [CrossRef] [PubMed]
- Marsberg, A.; Kemler, M.; Jami, F.; Nagel, J.H.; Postma-Smidt, A.; Naidoo, S.; Wingfield, M.J.; Crous, P.W.; Spatafora, J.W.; Hesse, C.N.; et al. Botryosphaeria dothidea: A latent pathogen of global importance to woody plant health. Mol. Plant Pathol. 2017, 18, 477–488. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Sun, C.; Guan, X.; Lian, S.; Li, B.; Wang, C. Biocontrol efficiency of Meyerozyma guilliermondii Y-1 against apple postharvest decay caused by Botryosphaeria dothidea and the possible mechanisms of action. Int. J. Food Microbiol. 2021, 338, 108957. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Ding, Z.; Zhou, Z.; Wang, Y.; Guo, L. Phylogenetic and pathogenic analyses show that the causal agent of apple ring rot in China is Botryosphaeria dothidea. Plant Dis. 2012, 96, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tu, H.; Hou, H.; Zhou, Z.; Yuan, H.; Luo, C.; Gu, Q. Occurrence and detection of carbendazim resistance in Botryosphaeria dothidea from apple orchards in China. Plant Dis. 2022, 106, 207–214. [Google Scholar] [CrossRef]
- Boya, P.; Reggiori, F.; Codogno, P. Emerging regulation and functions of autophagy. Nat. Cell Biol. 2013, 15, 713–720. [Google Scholar] [CrossRef]
- Sedwick, C. Yoshinori Ohsumi: Autophagy from beginning to end. J. Cell Biol. 2012, 197, 164–165. [Google Scholar] [CrossRef]
- Jiang, G.; Tan, Y.; Wang, H.; Peng, L.; Chen, H.; Meng, X.; Li, L.; Liu, Y.; Li, W.; Shan, H. The relationship between autophagy and the immune system and its applications for tumor immunotherapy. Mol. Cancer 2019, 18, 17. [Google Scholar] [CrossRef]
- Mizushima, N. Autophagy: Process and function. Gene. Dev. 2007, 21, 2861–2873. [Google Scholar] [CrossRef]
- Chan, E.Y.; Tooze, S.A. Evolution of Atg1 function and regulation. Autophagy 2009, 5, 758–765. [Google Scholar] [CrossRef] [Green Version]
- Mizushima, N. The role of the Atg1/ULK1 complex in autophagy regulation. Curr. Opin. Cell Biol. 2010, 22, 132–139. [Google Scholar] [CrossRef]
- Noda, N.N.; Fujioka, Y. Atg1 family kinases in autophagy initiation. Cell. Mol. Life Sci. 2015, 72, 3083–3096. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Kundu, M. Canonical and noncanonical functions of ULK/Atg1. Curr. Opin. Cell Biol. 2017, 45, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lu, J.; Zhang, L.; Dong, B.; Min, H.; Lin, F. Involvement of a Magnaporthe grisea serine/threonine kinase gene, MgATG1, in appressorium turgor and pathogenesis. Eukaryot. Cell 2007, 6, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Zhang, Z.; Shao, W.; Yang, Y.; Zhou, M.; Chen, C. The autophagy-related gene BcATG1 is involved in fungal development and pathogenesis in Botrytis cinerea. Mol. Plant Pathol. 2017, 18, 238–248. [Google Scholar] [CrossRef]
- Lv, W.; Wang, C.; Yang, N.; Que, Y.; Talbot, N.J.; Wang, Z. Genome-wide functional analysis reveals that autophagy is necessary for growth, sporulation, deoxynivalenol production and virulence in Fusarium graminearum. Sci. Rep. 2017, 7, 11062. [Google Scholar] [CrossRef]
- Yu, J.H.; Hamari, Z.; Han, K.H.; Seo, J.A.; Reyes-Domínguez, Y.; Scazzocchio, C. Double-joint PCR: A PCR-based molecular tool for gene manipulations in filamentous fungi. Fungal Genet. Biol. 2004, 41, 973–981. [Google Scholar] [CrossRef]
- Gronover, C.S.; Kasulke, D.; Tudzynski, P.; Tudzynski, B. The role of G protein alpha subunits in the infection process of the gray mold fungus Botrytis cinerea. Mol. Plant Microbe Interact. 2001, 14, 1293–1302. [Google Scholar] [CrossRef]
- Yu, F.; Gu, Q.; Yun, Y.; Yin, Y.; Xu, J.R.; Shim, W.B.; Ma, Z. The TOR signaling pathway regulates vegetative development and virulence in Fusarium graminearum. New phytol. 2014, 203, 219–232. [Google Scholar] [CrossRef]
- Nair, U.; Thumm, M.; Klionsky, D.J.; Krick, R. GFP-Atg8 protease protection as a tool to monitor autophagosome biogenesis. Autophagy 2011, 7, 1546–1550. [Google Scholar] [CrossRef] [Green Version]
- Corral-Ramos, C.; Roca, M.G.; Di Pietro, A.; Roncero, M.I.; Ruiz-Roldán, C. Autophagy contributes to regulation of nuclear dynamics during vegetative growth and hyphal fusion in Fusarium oxysporum. Autophagy 2015, 11, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Chun, Y.; Kim, J. Autophagy: An essential degradation program for cellular homeostasis and life. Cells 2018, 7, 278. [Google Scholar] [CrossRef] [PubMed]
- Pollack, J.K.; Harris, S.D.; Marten, M.R. Autophagy in filamentous fungi. Fungal Genet. Biol. 2009, 46, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.A.; Lu, J.; Liu, X.; Rehman, A.; Lin, F. Multifunction of autophagy-related genes in filamentous fungi. Microbiol. Res. 2012, 167, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Shao, G.; Liu, X.; Chen, Q.; Dong, M.; Du, L. Atg1 kinase in fission yeast is activated by Atg11-mediated dimerization and cis-autophosphorylation. eLife 2020, 9, e58073. [Google Scholar] [CrossRef]
- Yanagisawa, S.; Kikuma, T.; Kitamoto, K. Functional analysis of Aoatg1 and detection of the Cvt pathway in Aspergillus oryzae. FEMS Microbiol. Lett. 2013, 338, 168–176. [Google Scholar] [CrossRef]
- Shoji, J.; Arioka, M.; Kitamoto, K. Possible involvement of pleiomorphic vacuolar networks in nutrient recycling in filamentous fungi. Autophagy 2006, 2, 226–227. [Google Scholar] [CrossRef]
- Liu, X.; Gao, H.; Xu, F.; Lu, J.; Devenish, R.J.; Lin, F. Autophagy vitalizes the pathogenicity of pathogenic fungi. Autophagy 2012, 8, 1415–1425. [Google Scholar] [CrossRef]
- Zhu, X.; Li, L.; Wu, M.; Liang, S.; Shi, H.; Liu, X.; Lin, F. Current opinions on autophagy in pathogenicity of fungi. Virulence 2019, 10, 481–489. [Google Scholar] [CrossRef] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, N.; Zhu, M.; Zhang, Y.; Wang, Z.; Li, B.; Ren, W. Involvement of the Autophagy Protein Atg1 in Development and Virulence in Botryosphaeria dothidea. J. Fungi 2022, 8, 904. https://doi.org/10.3390/jof8090904
Liu N, Zhu M, Zhang Y, Wang Z, Li B, Ren W. Involvement of the Autophagy Protein Atg1 in Development and Virulence in Botryosphaeria dothidea. Journal of Fungi. 2022; 8(9):904. https://doi.org/10.3390/jof8090904
Chicago/Turabian StyleLiu, Na, Meiqi Zhu, Yihan Zhang, Zhongqiang Wang, Baohua Li, and Weichao Ren. 2022. "Involvement of the Autophagy Protein Atg1 in Development and Virulence in Botryosphaeria dothidea" Journal of Fungi 8, no. 9: 904. https://doi.org/10.3390/jof8090904
APA StyleLiu, N., Zhu, M., Zhang, Y., Wang, Z., Li, B., & Ren, W. (2022). Involvement of the Autophagy Protein Atg1 in Development and Virulence in Botryosphaeria dothidea. Journal of Fungi, 8(9), 904. https://doi.org/10.3390/jof8090904




