3.2. Taxonomy
A key to the species of Clavascidium and Placidium is listed in
Table 1.
ClavascidiumBreuss, Annln naturh. Mus. Wien, Ser. B, Bot. Zool. 98 (Suppl.): 41, 1996
The genus
Clavascidium is characterized by squamulose thallus, the presence of rhizines, and clavate to (sub) cylindrical asci containing biseriate ascospores. Gueidan [
2,
9] introduced
Clavascidium as a sister genus of
Placidium based on morphological characters and a phylogenetic analysis, between which
Clavascidium has clavate asci with biseriate ascospores, whereas
Placidium has cylindrical asci with uniseriate ascospores. However, uniseriate or both biseriate and uniseriate ascospores were also reported [
30].
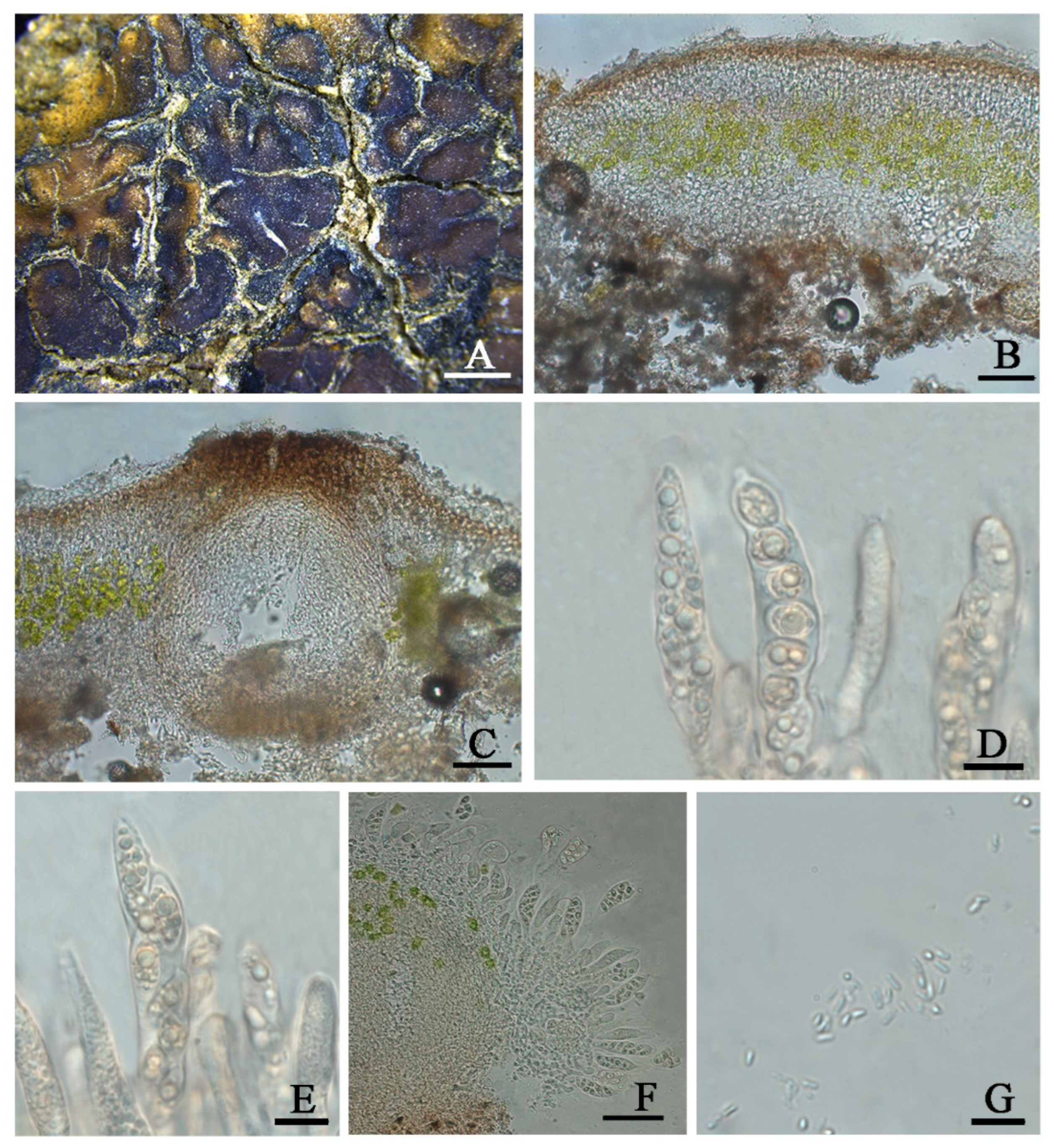
Clavascidium lacinulatum (Ach.) M. Prieto, in Prieto et al., Am. J. Bot. 99 (1): 28, 2012 (
Figure 3)
≡ Endocarpon hepaticum var. Lacinulatum Ach Lich. univ.: 299, 1810.
Description: Thallus squamulose, terricolous, 300 µm thick; lobes 1–4 mm wide, roundish to deeply lobed, contiguous, rarely overlapping; upper surface dark brown, dull; lower surface pale ± rhizines; epinecral layer transparent, up to 15 µm thick; upper cortex paraplectenchymous, 50–70 µm thick, cells 4 × 10 µm in diam.; uppmost layer brown, 25–30 µm thick; photobiont layer 50–100 µm thick, algal cells 6–10 µm in diam; medullar tissue paraplectenchymous; lower cortex not delimited from the medulla, 40 µm thick, paraplectenchymous, composed of irregularly arranged roundish cells up to 7.5 µm in diam. Rhizohyphae hyaline, 4–6 µm wide. Pycnidia laminal, Dermatocarpon–type, immersed, subglobose, light brown, conidia oblong–ellipsoid to bacilliform, 1–1.3 × 3–3.7 µm in size.
Perithecia immersed, broadly pyriform (150 × 180 µm) to subglobose (up to 230 µm) wide; perithecia wall bright, 30–35 µm thick; hymenium bright, 35–45 µm thick; involucrellum brown, 42–44 µm thick; pyriphyses 35–40 × 2.5–3 μm; asci cylindrical to clavate, 14–18 × 40–52 µm, ascospores 8 per ascus, uniseriate to biseriate, ellipsoid to fusiform to ovoid, 5–7 × 10–12 µm.
Chemistry: All the spot tests were negative, and no substances were detected by TLC.
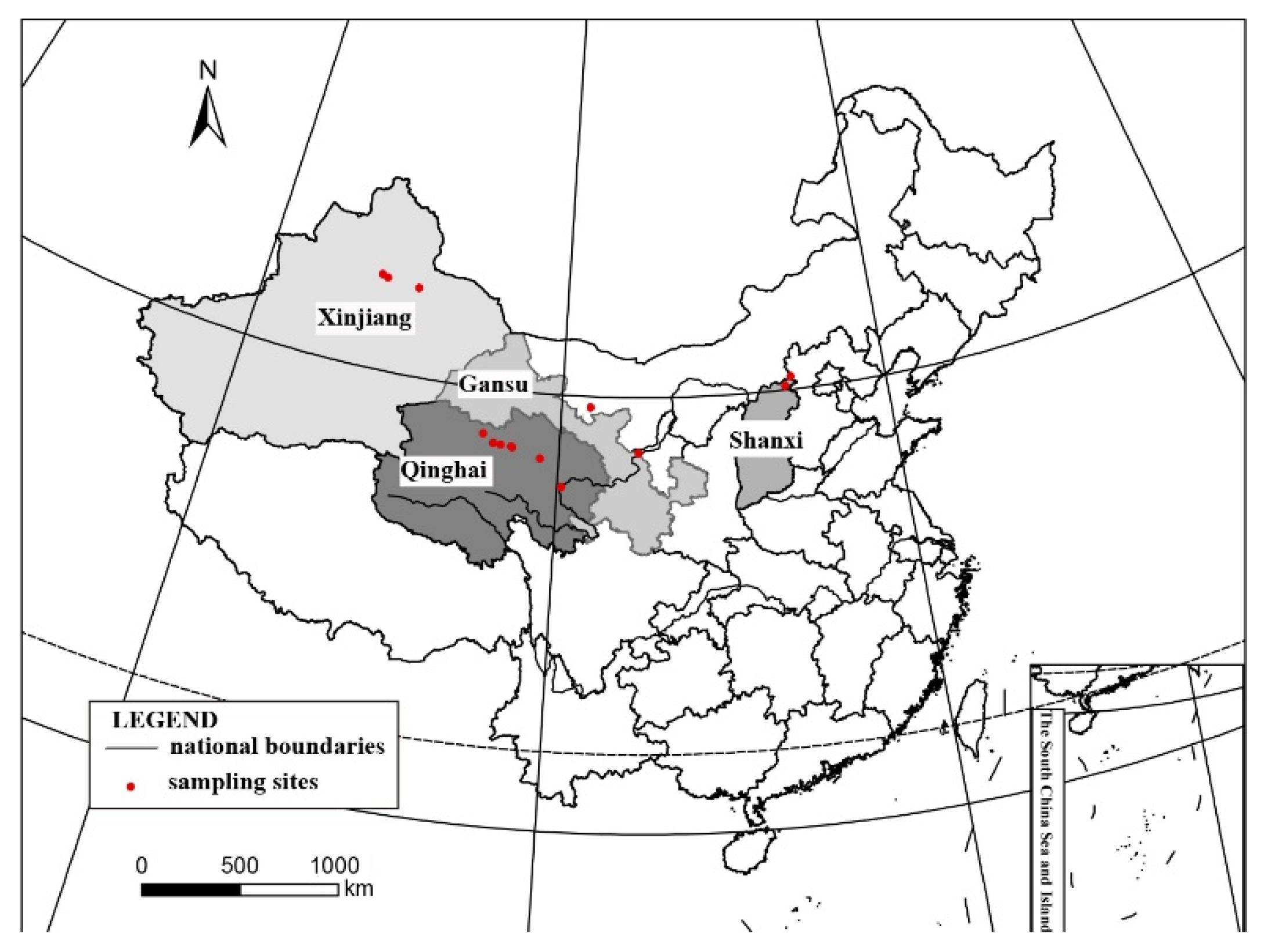
Habitat and distribution: These species grow on the surface of sandy soil in the semi-arid and arid region of Northwest China, located in the open areas with sun exposure. The surrounding environment is characterized by interlace of meadows, fixed undulating sand dunes under shrubs, and the Gobi Desert, with the elevations greatly varying from 923 to 3175 m. It distributes worldwide [
30] and is new to China.
Specimens examined: CHINA. SHANXI: Datong City, Yanggao County. 40.98° N 113.88° E, 1264 m alt., on the sand, 14 April 2021, X. Qian & T.T. Zhang 20210273 (HMAS–L 153955), 20210274 (HMAS–L 153956), 20210283 (HMAS–L 153957); 40°25′51″N 113°44′19″E, 923 m alt., on the sand, 15 April 2021, X. Qian & T.T. Zhang 20210299 (HMAS–L 153962), 20210300 (HMAS–L 153958), 20210302 (HMAS–L 153959), 20210306 (HMAS–L 153960), 20210307 (HMAS–L 153961). GANSU: Baiyin City, Jingtai County. 37°19′41″ N 104°35′37″ E, 1524 m alt., on the sand, 21 October 2020, X. Qian et al. 20201529 (HMAS–L 153781). QINGHAI: Haixi Mongolian and Tibetan Autonomous Prefecture, Delhi City. 37°23′02″ N 97°08′44″ E, 3041 m alt., on the sand, 24 April 2021, X.L. Wei & T.T. Zhang 20210436 (HMAS–L 151942); 37°24′29″ N 96°30′43″ E, 3175 m alt., on the sand, 24 April 2021, X.L. Wei & T.T. Zhang 20210499 (HMAS–L 153954); Hainan Tibetan Autonomous Prefecture, Guinan County. 35°40′54″ N 100°15′08″ E, 1606 m alt., on the sand, 16 October 2020, X. Qian et al. QX20200026 (HMAS–L 153953).
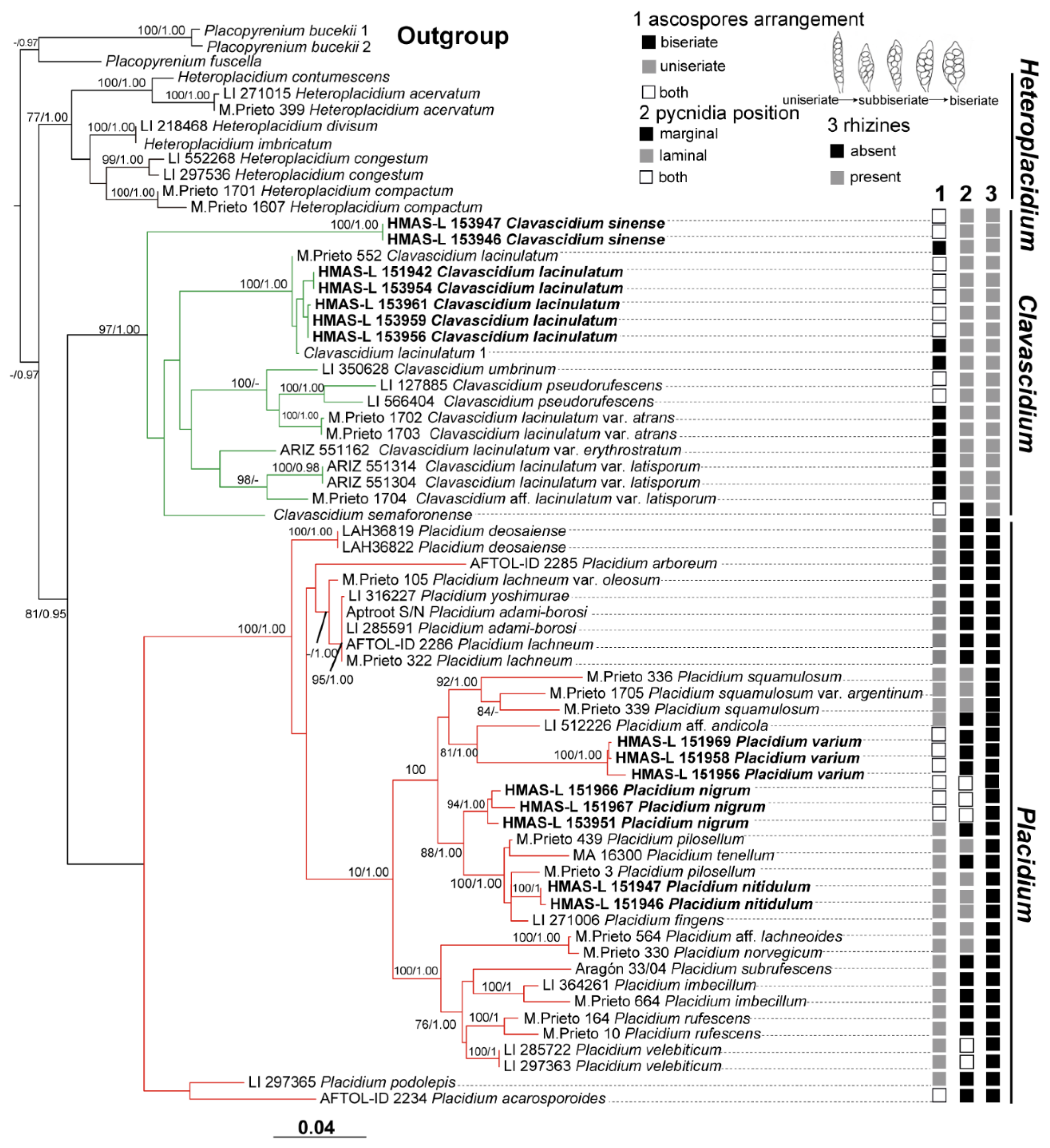
Notes: This species is widespread in the desert regions of China as a crust (
Figure 1).Our specimens are well-clustered with
Cl. lacinulatum in the phylogeny [
4] (
Figure 2 and
Figures S1–S3) and, also, with a high consistency of the phenotype described by Nash et al. [
30]. Although this species is variable in the external morphology of thallus and squamules, several important taxonomic characters such as laminal pycnidia, oblong-ellipsoidal to subcylindrical conidia, and rhizines are constantly present. Breuss described uniseriate ascospores in this species [
31]; however, we found this character is variable (
Figure 3).
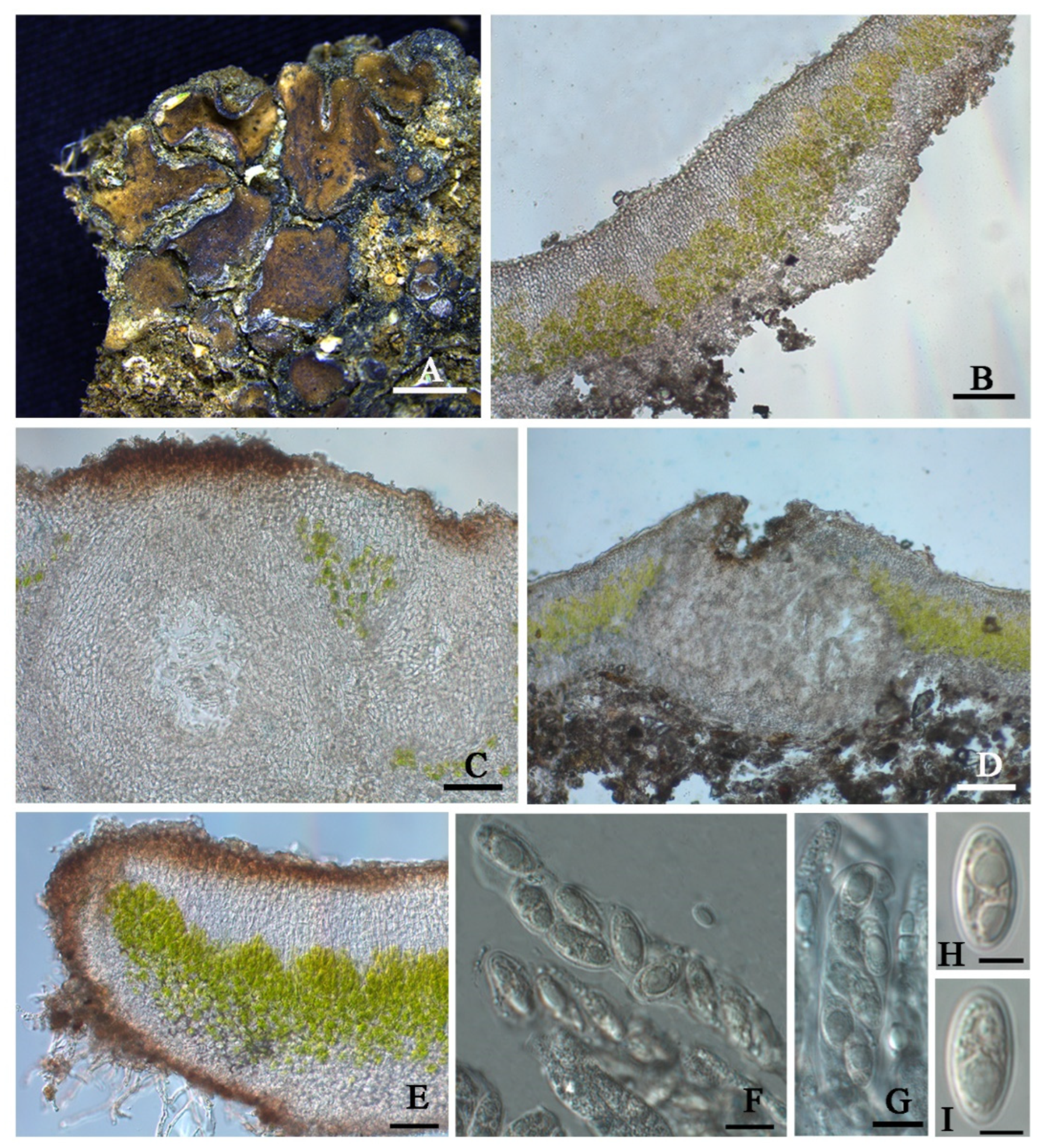
Clavascidium sinense T.T. Zhang & X.L. Wei,
sp. nov. (
Figure 4)
Fungal Names No.: FN571022
Etymology: The epithet ‘sinense’ refers to the Chinese distribution of this species.
Typus: CHINA. SHANXI: Datong City, Yanggao County. 40°25′28″ N 113°46′07″ E, 1177 m alt., on soil, 14 April 2021, X. Qian & T.T. Zhang 20210245 (holotype, HMAS–L 153946, ITS ON712842, nuLSU ON712829).
Diagnosis: It is characterized by the co-existence of both uniseriate and biseriate ascospores in asci, laminal pycnidia, and mixed-type medulla.
Description: Thallus squamulose, terricolous; lobes 1.5–4 mm wide, (194) 255 ± 32 (314) µm thick, deeply lobed, contiguous, rarely overlapping, full appressed to the substrate or with raised margins free from the substrate; upper surface medium to dark brown, dull; lower surface pale ± rhizines; epinecral layer often absent, if present up to 10 µm thick; upper cortex paraplectenchymatous, (57) 81 ± 19 (109) µm thick, cells (7.8) 9.9 ± 1.7 (12.6) µm in diam., uppmost layer bright brown, 15–25 µm thick; algal layer (70) 107 ± 24 (157) µm thick, composed of rounded cells (7.4) 9.5 ± 1.6 (12) µm in diam., globose to sub-globose; medulla mixed type, 60–100 µm thick; lower cortex not clearly delimited from the medulla, transparent, outer layer of lower cortex bright brown, (37) 51 ± 8.5 (59) µm thick, composed of rounded cells (7.2) 8.2 ± 0.9 (10.2) µm diam. Rhizohyphae 4–5 µm thick, colorless. Pycnidia laminal, Dermatocarpon-type, subglobose, light brown, conidia oblong-ellipsoid to bacilliform, (3.0) 3.4 ± 0.29 (3.7) × (1.26) 1.4 ± 0.18 (1.75) µm.
Perithecia laminal, immersed, subglobular, up to 400 µm in diam.; perithecia wall 40–55 µm thick; hymenium pale brown, 65–100 µm thick; involucrellum brown, 38–50 µm thick; pyriphyses 30–40 µm long; asci (sub)cylindrical to clavate, 64–84 × 12–15 µm, 8 spored, ascospores uniseriate to biseriate, hyaline, narrow ellipsoid, (10.2) 13 ± 1.2 (13.9) × (4.8) 6.3 ± 0.7 (7.3) µm.
Chemistry: All the spot tests were negative, and no substances were detected by TLC.
Habitat and distribution: On the surface of a broken great wall built by clay in the semi-arid region of Northwest China. It has been known only in China up to now.
Additional specimens examined: CHINA. SHANXI: Datong City, Yanggao County. 40°25′28″N 113°46′07″E, 1177 m alt., on soil, 14 April 2021, X. Qian & T.T. Zhang 20210246 (HMAS-L 153947).
Notes: Biseriate and uniseriate ascospores in asci are generally diagnostic characters of
Clavascidium and
Placidium, respectively [
2,
9]. However, uniseriate or both biseriate and uniseriate ascospores in asci were also found in
Clavascidium [
30], including
Cl. sinense, indicating ascospores arrangement is a continuously changing character between these two closely related genera in phylogeny. The phylogenetic analysis clearly supported
Cl. sinense obviously separated from other
Clavascidium species with both types of ascospores arrangements, such as
Cl. lacinulatum,
Cl. pseudorufescens (Breuss) M. Prieto, and
Cl. semaforonense (Breuss) M. Prieto. However,
Cl. sinense has mixed-type medulla, distinct from
Cl. lacinulatum with paraplectenchymous medulla and
Cl. pseudorufescens with filamentous-hyphae medulla [
30].
Cl. sinense has laminal pycnidia, but
Cl. semaforoense has marginal pycnidia, which is an exception within the genus
Clavascidium [
8]. The phylogenetic trees (
Figure 2 and
Figures S1–S3) provided further support, indicating
Cl. semaforoense formed a separate clade, far from all the other species of
Clavascidium. However, the new species
Cl. sinense situated in the outermost clade, and
Cl. semaforoense situated in the secondary outermost, indicating that laminal or marginal pycnidia is not an absolutely unchanging character within this genus but can be used in delimitation species. There are three
Clavascidium species absence of DNA sequences:
Clavascidium antillarum (Breuss) Breuss,
Clavascidium imitans (Breuss) M. Prieto, and
Clavascidium krylovianum (Tomin) M. Prieto. However,
Cl. sinense has distinct traits compared with these three species, for example,
Cl. antillarum is characterized by dark brown to black lower surface and rhizines [
5], while
Cl. sinense is characterized by a pale lower surface and rhizines.
Cl. imitans and
Cl. krylovianum can be distinguished by the morphology of medulla; the former medullary hyphae divide into many short and swollen cells, but the latter medullary hyphae are filamentous [
5]; in comparison,
Cl. sinense has a mixed-type medulla.
Placidium A. Massal. Symmict. Lich.: 75, 1855
The genus
Placidium has squamulose thallus, laminal or marginal
Dermatocarpon-type pycnidia, cylindrical asci, uniseriate ascospores, and rhizohyphae [
8]. Two
Placidium species have been reported from Taiwan by Dr. Aptroot [
13]. This genus is firstly reported from Mainland China in this study, and we found some additional morphological characters in
Placidium such as a glossy upper surface and aggregated pycnidia, which can be used to distinguish some species.
Placidium nitidulum T.T. Zhang & X.L. Wei
sp. nov. (
Figure 5)
Fungal Names No.: FN571023
Etymology: The epithet ‘nitidulum’ refers to the glossy upper surface of the thallus in this species.
Typus: CHINA. QINGHAI: Haixi Mongolian and Tibetan Autonomous Prefecture, Wulan County. 36°57′31″ N 98°54′03″ E, 3314 m alt., on the sand, 25 April 2021, X.L. Wei & T.T. Zhang 20210560 (holotype, HMAS–L 151947, ITS ON712844, nuLSU ON712835).
Diagnosis: It is characterized by a glossy upper surface, tiny lobes, and a thick algal layer.
Description: Thallus squamulose; terricolous; lobes roundish, tiny, 0.7–2 mm wide, (356) 442 ± 55 (523) µm thick; contiguous to densely aggregated, tightly adnate to the substrate; upper surface medium brown, glossy, brown rimmed; lower surface pale to pale yellow; epinecral layer transparent, (33.4) 35.4 ± 1.6 (37.5) µm thick; upper cortex (41) 58 ± 15 (78) µm thick, paraplectenchymatous, cells 8–10 µm in diam.; uppermost layer pale yellow to light brown, 33–36 µm thick; algal layer (104) 159 ± 34 (195) µm thick, algal cells globular, (7.3) 7.8 ± 0.5 (8.7) µm in diam. dispersed over the whole medulla; medulla zone not clear; lower cortex not delimited from the medulla, (42) 63 ± 16 (88) µm thick, composed of densely aggregated globular cells, cells (8.4) 10.6 ± 1.5 (12.6) µm in diam. Rhizohyphae (4.0) 5.2 ± 0.7 (5.9) µm thick, colorless. Pycnidia laminal, immersed, Dermatocarpon-type, immature, subglobular, 250 µm in dim., conidia not seen.
Perithecia laminal, immersed, occasionally aggregated, narrowly pyriform, up to 300 µm wide; perithecia wall grey, 28–42 µm thick; hymenium pale yellow, 45–75 µm thick; involucrellum brown or absence, 68–90 µm thick; paraphyses branched, 35–55 × 1.5–2.5 µm; asci cylindrical, 45–65 × 5–8 µm, 8 spored, uniseriate, ascospores hyaline, narrow ellipsoid, (8.4) 8.7 ± 1.5 (11.5) × (5.1) 6.1 ± 0.5 (7.0) µm; paraphyses well-developed.
Chemistry: All the spot tests were negative, and no substances were detected by TLC.
Ecology and distribution: On the surface of sandy soil in high altitude areas of the Qinghai–Tibet plateau. It has been known only in China up to now.
Additional specimens examined: CHINA. QINGHAI: Haixi Mongolian and Tibetan Autonomous Prefecture, Delhi City. 37°23′02″ N 97°08′44″ E, 3041 m alt., on the sand, 24 April 2021, X.L. Wei & T.T. Zhang 20210421 (HMAS-L 151962); Wulan County. 36°57′31″ N 98°54′03″ E, 3314 m alt., on the sand, 25 April 2021, X.L. Wei & T.T. Zhang 20210552 (HMAS–L 151946).
Notes: This new species can be easily recognized by its glossy appearance of the upper surface and tiny lobes, which is very distinctive and different from all the other known
Placidium species. The medulla zone is obscure due to being fully covered by the thick algal layer, so the lower cortex is difficult to delimit from the medulla, similar to
Placidium tenellum (Breuss) Breuss in this character. However, the upper surface of
Pl. tenellum is matt, the algal layer is thinner ((40) 93 ± 24 (155) µm), and the pycnidia is much broader (up to 500 µm). The distribution is more frequent in coastal areas [
8]. Based on the phylogenetic trees (
Figure 2 and
Figures S1–S3), the new species is close to
Placidium fingens (Breuss) Breuss,
Pl. pilosellum, and
Pl. tenellum, among which the first two are more intimate than the last one to the new species in phylogeny; however, the lobe widths of
Pl. fingens and
Pl. pilosellum (up to 6 mm) are nearly three times the new species (0.7–2 mm).
Pl. nitidulum is closer to
Pl. fingens, both of which have laminal pycnidia, while the species
Pl. pilosellum within this subclade with a little far distance has marginal pycnidia.
Pl. nitidulum has more slender asci (45–65 × 5–8 µm) than the species
Pl. pilosellum (70–90 × 10–15 µm). Additionally,
Pl. nitidulum has squamules with a smooth margin, but
Pl. pilosellum has squamules with hairy margins [
8].
Placidiumnigrum T.T. Zhang & X.L. Wei,
sp. nov. (
Figure 6)
Fungal Names No.: FN571024
Etymology: The epithet ‘nigrum’ refers to the surrounding black area due to the aggregation of abundant pycnidia of this species.
Typus: CHINA. QINGHAI: Haixi Mongolian and Tibetan Autonomous Prefecture, Dachaidan Region. 37°52′11″ N 95°15′54″ E, 3515 m alt., on the sand, 25 April 2021, X.L. Wei & T.T. Zhang 20210516 (holotype, HMAS–L 151944, ITS ON712851).
Diagnosis: It is characterized by both laminal and marginal pycnidia, with abundant aggregation forming into the surrounding black area.
Description: Thallus squamulose, terricolous; lobes 1–3 mm wide, (211) 336 ± 81 (462) µm thick, tumid roundish to lobate, scattered or contiguous to densely aggregated, tightly adnate to the substrate or with slightly raised margins; upper surface pale to medium brown, dull, pruinose; epinecral layer transparent, (12) 20.4 ± 7.2 (33) µm thick; upper cortex paraplectenchymatous, (31) 52 ± 11 (70) µm thick, cells (8.5) 10.5 ± 1.9 (13.8) µm in diam., uppermost layer brown, 8–12 µm thick; algal layer (37) 67 ± 14 (85) µm thick, composed of spherical cells, cells (7.4) 13.3 ± 2.9 (17.6) µm in diam., globose to subglobose, in clusters of 1–3 cells; medulla mixed type, (37) 69 ± 17 (102) µm thick; lower cortex delimited from the medulla, hyaline, (32) 44 ± 9.8 (73) µm thick, composed of spherical cells, cells (8.1) 9.8 ± 1.4 (12.4) µm in diam. Rhizohyphae (3.3) 4.4 ± 0.77 (5.7) µm thick, colorless. Pycnidia laminal and marginal, abundant, aggregated into all-round black area, immersed, Dermatocarpon-type, subglobular to irregular, up to (230) 274 ± 33 (317) µm wide; conidia bacilliform, (2.7) 3 ± 0.39 (3.5) × (0.96) 1.3 ± 0.29 (1.89) µm.
Perithecia laminal, immersed, subglobular, 320 × 350 µm; perithecia wall bright, 28–50 µm thick; hymenium 70–85 µm thick; involucrellum brown, 43–55 µm thick; periphyses 40–55 µm long; asci cylindrical to clavate, 45–65 × 9–18 µm, 8 spored, uniseriate to (sub)biseriate, ascospores hyaline, ellipsoid, (8.4) 9.9 ± 1 (11.5) × (4.0) 5.7 ± 0.9 (8.0).
Chemistry: All the spot tests were negative, and no substances were detected by TLC.
Habitat and distribution: On the surface of sandy soil in the semi-arid and arid region of Northwest China and the Qinghai–Tibet plateau. The distribution range is relatively wide, from low altitude to high altitude (438–3515 m alt.). The environment in which they grow is also more varied, including naturally formed bush bottoms and artificial vegetation fix sand forests (i.e., Ammopiptanthus mongolicus (Maxim. ex Kom.) Cheng f. and Populus alba var. pyramidalis Bge.), and the Gobi Desert, common in dry and open habitats. It has been known only in China up to now.
Additional specimens examined: CHINA. QINGHAI: Haixi Mongolian and Tibetan Autonomous Prefecture, Dachaidan Region. 37°52′11″ N 95°15′54″ E, 3515 m alt., on sand, 25 April 2021, X.L. Wei & T.T. Zhang 20210535 (HMAS–L 153951), 20210522 (HMAS–L 151945), 20210523 (HMAS–L 151965), 20210524 (HMAS–L 151966), 20210534 (HMAS–L 153950); Delhi City. 37°23′02″ N 97°08′44″ E, 3041 m alt., on the sand, 24 April 2021, X.L. Wei & T.T. Zhang 20210461 (HMAS–L 151963); Hainan Tibetan Autonomous Prefecture, Guinan County. 35°40′54″ N 100°15′08″ E, 1606 m alt., on the sand, 16 October 2020, X. Qian et al. 20201384 (HMAS–L 153732). GANSU: Baiyin City, Jingtai County. 37°25′06″ N 104°34′56″ E, 1591 m alt., on the sand, 21 October 2020, X. Qian et al. 20201486 (HMAS–L 153786). XINJIANG: Changji Hui Autonomous Prefecture, Fukang County. 44°22′59″ N 87°52′34″ E, 438 m alt., on the sand, 10 May 2021, X. Qian & T.T. Zhang 20210666 (HMAS–L 151952); Qitai County. 44°13′32″ N 90°02′32″ E, 723 m alt., on the sand, 10 May 2021, X. Qian & T.T. Zhang 20210746 (HMAS–L 151941); Wujiaqu City. 44°29′54″ N 87°28′21″ E, 408 m alt., on the sand, 9 May 2021, X. Qian & T.T. Zhang 20210606 (HMAS–L 151967). INNER MONGOLIA: Alxa Right Banner, 39°28′24″ N 101°04′03″ E, 1564 m alt., on sand, 22 July 2017, D.L. Liu & R.D. Liu XL2017267 (HMAS–L 140940); 39°28′22″ N 101°04′04″ E, 1563 m alt., on sand, 5 June 2018, D.L. Liu et al. ALS2018022 (HMAS–L 143912); 39°32′30″ N 101°06′34″ E, 1478 m alt., on sand, 5 June 2018, D.L. Liu et al. ALS2018040 (HMAS–L 153952).
Notes: This species is distinctive by having both uniseriate and (sub) biseriate ascospores arrangement, both laminal and marginal pycnidia, and a surrounding black area due to the aggregation of abundant pycnidia, which well-separate it from all the other
Placidium species.
Placidium is generally known as the only genus comprising species with laminal or marginal pycnidia [
31]. Within this genus, very few species have both laminal and marginal pycnidia such as
Pl. velebiticum (Zahlbr. ex Zschacke) Breuss [
4,
8]; however,
Pl. velebiticum only has a uniseriate ascospores arrangement, which is different from
Pl. nigrum.
Placidium varium T.T. Zhang & X.L. Wei,
sp. nov. (
Figure 7)
Fungal Names No.: FN571025
Etymology: The epithet ‘varium’ refers to the morphology and arrangement of the variable ascospores in this species.
Typus: CHINA. XINJIANG: Changji Hui Autonomous Prefecture, Qitai County. 44°13′32″ N 90°02′32″ E, 723 m alt., on the sand, 10 May 2021, X. Qian & T.T. Zhang 20210751 (holotype, HMAS–L 151970, nuLSU ON712841).
Diagnosis: This new species is characterized chiefly by both (sub)biseriate and uniseriate ascospores arrangement and having macro guttule in the center of each ascospore.
Description: Thallus squamulose, terricolous; lobes up to 4 mm wide, (183) 202 ± 10 (224) µm thick, deeply lobate, scattered or contiguous to densely aggregated, sometimes overlapped or imbricated, adpressed to the substrate or with raised margins free from the substrate, sometimes with a black margin; upper surface red-brown to dark brown, pruinose, dull; lower surface greyish brown; epinecral layer transparent, very thin, up to 20 µm thick; upper cortex (31) 46 ± 9 (65) µm thick, paraplectenchymatous cells (7.8) 8.2 ± 0.3 (8.6) µm diam, uppermost layer light brown, 10–13 µm thick; algal layer (73) 93 ± 14 (112) µm thick, algal cells globular, (7.2) 8.5 ± 0.9 (10) µm in diam., in clusters of 1–3 cells; medulla mixed type, (46) 54 ± 5(62) µm thick; lower cortex not delimited from the medulla, hyaline, (28) 33 ± 5.9 (43) µm thick, composed of spherical cells, (5.9) 7.7 ± 1.5 (10) µm in diam. Rhizohyphae (3.2) 4.4 ± 0.78 (5.7) µm thick, colorless. Pycnidia marginal, immersed, Dermatocarpon-type, subglobular to pyriform, up to 256–320 × 220–288 µm in size; conidia bacilliform to oblong-ellipsoid, (3.1) 3.5 ± 0.21 (3.9) × (1.0) 1.5 ± 0.2 (1.9) µm.
Perithecia immersed, subglobular to broad pyriform, up to 260 µm wide; perithecia wall bright, 19–25 µm thick; hymenium bright, 22–46 µm thick; involucrellum abscence; periphyses 18–26 µm long; asci cylindrical to clavate, 55–73 × 8–21 µm, 8 spored, uniseriate to (sub)biseriate, ascospores hyaline, ellipsoid to ovoid, (8.5) 11.8 ± 1.8 (15.3) × (7.6) 8.6 ± 1.0 (11.6) µm in size, central guttule (6.6) 8.0 ± 1.1 (10.8) × (4.9) 5.8 ± 0.68 (7.0) µm.
Chemistry: All the spot tests were negative, and no substances were detected by TLC.
Ecology and distribution: On the surface of sandy soil in the arid region of Xinjiang Autonomous, Northwest China. The specimens were collected at the bottom of a fixed sand area of cultivated shrubs. The habitat is a low-altitude open area with long daylight exposure, low precipitation and high evaporation. It has been known only in China up to now.
Additional specimens examined: CHINA. XINJIANG: Changji Hui Autonomous Prefecture, Fukang County. 44°22′59″ N 87°52′34″ E, 438 m alt., on the sand, 10 May 2021, X. Qian & T.T. Zhang 20210639 (HMAS–L 153949), 20210676 (HMAS–L 151953), 20210689 (HMAS–L 151955), 20210692 (HMAS–L 1519561), 20210698 (HMAS–L 151958), 20210696 (HMAS–L 151957); Wujiaqu City. 44°29′54″ N 87°28′21″ E, 408 m alt., on the sand, 9 May 2021, X. Qian & T.T. Zhang 20210576 (HMAS–L 153948), 20210599 (HMAS–L 151948), 20210608 (HMAS–L 151949), 20210611 (HMAS–L 151940), 20210623 (HMAS–L 151951); Qitai County. 44°13′32″ N 90°02′32″ E, 723 m alt., on the sand, 10 May 2021, X. Qian & T.T. Zhang 20210725 (HMAS–L 151969), 20210716 (HMAS–L 152826).
Notes: The ascospores arrangement is variable in this new species. Compared with other
Placidium species with small ascospores, such as
Placidium arboretum (Schwein. ex E. Michener) Lendemer (syn.
Placidium tuckermanii (Rav. ex Mont.) Breuss),
Placidium andicola (Breuss) Breuss,
Placidium chilense (Breuss) Breuss,
Placidium corticola (Räsänen) Breuss, and
Placidium ruiz-lealii (Räsänen) Breuss, characterized by oval and uniseriate ascospores [
30,
32], the new species has more variable ascospores in shape and arrangement. Moreover, several species are not terricolous like this new species but corticolous in
Pl. corticola and
Pl. arboretum or saxicolous in
Pl. ruiz-lealii [
30]. Although
Pl. andicola and
Pl. chilense are terricolous, they are different from the new species in morphology besides the different shapes of ascospores; for example,
Pl. andicola has brown but not red-brown thallus and only uniseriate ascospores in asci [
30].
Pl. chilense has much more extensive (4–10 (−20) mm wide) and thicker (600 µm thick) lobes and much larger perithecia (up to 600 µm wide) than
Pl. varium [
30]. In the phylogenetic analyses, due to the absence of DNA sequences, among the species mentioned earlier related to
Pl. varium, only
Pl. andicola and
Pl. arboretum can be further compared. It can be seen from the phylogenetic trees (
Figure 2 and
Figures S1–S3)
Pl. varium clustered more closely to
Pl. andicola than other species, although it is also pronounced there is a much distant relationship between these two species, and
Pl. varium formed a well-supported separate clade, indicating it is a new species. As comparison, there is farther phylogenetic relationship between terricolous
Pl. varium and corticolous
Pl. arboretum, indicating the substrate type contributes to the species delimitation in
Placidium.