Biochemical Approaches on Commercial Strains of Agaricus subrufescens Growing under Two Environmental Cultivation Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Mushroom Samples and Cultivation Conditions
2.2. Proximal Composition
2.3. Chemical Composition Regarding Hydrophilic Compounds
2.3.1. Free Sugars
2.3.2. Organic Acids
2.4. Chemical Composition Regarding Lipophilic Compounds
2.4.1. Fatty Acids
2.4.2. Tocopherols
2.5. Phenolic Acids and Related Compounds
2.6. Bioactive Properties
2.6.1. Antioxidant Activity Evaluation
2.6.2. Antimicrobial Activity
2.7. Statistical Analysis
3. Results and Discussion
3.1. Proximal Composition
3.2. Chemical Composition Regarding Hydrophilic Compounds
3.3. Chemical Composition Regarding Lipophilic Compounds
3.4. Phenolic Acids and Related Compounds
3.5. Bioactive Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Royse, D.J.; Baars, J.; Tan, Q. Current Overview of Mushroom Production in the World. In Edible and Medicinal Mushrooms; Diego, C.Z., Pardo-Giménez, A., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2017; pp. 5–13. ISBN 978-1-119-14944-6. [Google Scholar]
- FAOSTAT. Value of Agricultural Production—Mushroom and Truffles. Available online: https://www.fao.org/faostat/en/#data/QV (accessed on 18 February 2022).
- Dias, E.S. Mushroom Cultivation in Brazil: Challenges and Potential for Growth. Ciênc. Agrotecnol. 2010, 34, 795–803. [Google Scholar] [CrossRef]
- Ferrari, A.B.S.; Galo Marcheafave, G.; Mannochio-Russo, H.; da Silva Bolzani, V.; Cunha Zied, D.; Spacino Scarminio, I.; Zeraik, M.L. Chemical Composition and Chromatographic Fingerprint of Three Strains of Agaricus subrufescens Cultivated with Handmade and Commercial Supplements. Food Chem. 2021, 363, 130227. [Google Scholar] [CrossRef] [PubMed]
- Kerrigan, R.W. Agaricus subrufescens, a Cultivated Edible and Medicinal Mushroom, and Its Synonyms. Mycologia 2005, 97, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Mleczek, M.; Siwulski, M.; Jasińska, A.; Budka, A.; Niedzielski, P.; Kalač, P.; Gąsecka, M.; Budzyńska, S. Multielemental Analysis of Fruit Bodies of Three Cultivated Commercial Agaricus species. J. Food Compos. Anal. 2017, 59, 170–178. [Google Scholar] [CrossRef]
- Wisitrassameewong, K.; Karunarathna, S.C.; Thongklang, N.; Zhao, R.; Callac, P.; Moukha, S.; Férandon, C.; Chukeatirote, E.; Hyde, K.D. Agaricus subrufescens: A Review. Saudi J. Biol. Sci. 2012, 19, 131–146. [Google Scholar] [CrossRef]
- Bilal, A.; Wani, B.A.; Bodha, R.H.; Wani, A.H. Nutritional and Medicinal Importance of Mushrooms. J. Med. Plants Res. 2010, 4, 2598–2604. [Google Scholar] [CrossRef]
- Lima, C.U.J.O.; Gris, E.F.; Karnikowski, M.G.O. Antimicrobial Properties of the Mushroom Agaricus blazei—Integrative Review. Rev. Bras. Farmacogn. 2016, 26, 780–786. [Google Scholar] [CrossRef][Green Version]
- Wang, H.; Fu, Z.; Han, C. The Medicinal Values of Culinary-Medicinal Royal Sun Mushroom (Agaricus blazei Murrill). Evid. Based Complement. Altern. Med. 2013, 2013, 842619. [Google Scholar] [CrossRef][Green Version]
- Rózsa, S.; Măniuțiu, D.N.; Poșta, G.; Gocan, T.M.; Andreica, I.; Bogdan, I.; Rózsa, M.; Lazăr, V. Influence of the Culture Substrate on the Agaricus blazei Murrill Mushrooms Vitamins Content. Plants 2019, 8, 316. [Google Scholar] [CrossRef]
- Win, T.T.; Ohga, S. Study on the Cultivation of Agaricus blazei (Almond Mushroom) Grown on Compost Mixed with Selected Agro-Residues. AiM 2018, 8, 778–789. [Google Scholar] [CrossRef][Green Version]
- Zied, D.C.; Junior, W.G.V.; Soares, D.M.M.; Stevani, C.V.; Dias, E.S.; Iossi, M.R.; Pardo-Giménez, A. Overview of Agaricus Subrufescens Strains Used in the Last 15 Years in Brazil and Other Countries and Current Potential Materials for the Future. Mycol. Prog. 2021, 20, 953–966. [Google Scholar] [CrossRef]
- Bellettini, M.B.; Fiorda, F.A.; Maieves, H.A.; Teixeira, G.L.; Ávila, S.; Hornung, P.S.; Júnior, A.M.; Ribani, R.H. Factors Affecting Mushroom Pleurotus spp. Saudi J. Biol. Sci. 2019, 26, 633–646. [Google Scholar] [CrossRef] [PubMed]
- Dias, E.S.; Abe, C.; Schwan, R.F. Truths and Myths about the Mushroom Agaricus blazei. Sci. Agric. 2004, 61, 545–549. [Google Scholar] [CrossRef]
- Junior, W.G.V.; Centeio Cardoso, R.V.; Fernandes, Â.; Ferreira, I.C.F.R.; Barros, L.; Pardo-Giménez, A.; Mendel Soares, D.M.; Stevani, C.V.; Zied, D.C. Influence of Strains and Environmental Cultivation Conditions on the Bioconversion of Ergosterol and Vitamin D2 in the Sun Mushroom. J. Sci Food Agric. 2021, 102, 1699–1706. [Google Scholar] [CrossRef] [PubMed]
- Suwannarach, N.; Kumla, J.; Zhao, Y.; Kakumyan, P. Impact of Cultivation Substrate and Microbial Community on Improving Mushroom Productivity: A Review. Biology 2022, 11, 569. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.F.; Azevedo, R.S.; Braga, C.; da Silva, R.; Dias, E.S.; Schwan, R.F. Microbial Diversity in a Bagasse-Based Compost Prepared for the Production of Agaricus brasiliensis. Braz. J. Microbiol. 2009, 40, 590–600. [Google Scholar] [CrossRef][Green Version]
- Ryckeboer, J.; Mergaert, J.; Vaes, K.; Klammer, S.; De Clercq, D.; Coosemans, J.; Insam, H.; Swings, J. A Survey of Bacteria and Fungi Occurring during Composting and Self-Heating Processes. Ann. Microbiol. 2003, 53, 349–410. [Google Scholar]
- Eger, G. Experiments and Comments on the Action of Bacteria on Sporophore Initiation in Agaricus bisporus. Mushroom Sci. 1972, 8, 719–725. [Google Scholar]
- AOAC. Official Methods of Analysis of AOAC International, 20th ed.; AOAC International: Gaithersburg, MD, USA, 2016; ISBN 978-0-935584-87-5. [Google Scholar]
- Spréa, R.M.; Fernandes, Â.; Calhelha, R.C.; Pereira, C.; Pires, T.C.S.P.; Alves, M.J.; Canan, C.; Barros, L.; Amaral, J.S.; Ferreira, I.C.F.R. Chemical and Bioactive Characterization of the Aromatic Plant Levisticum officinale, W.D.J. Koch: A Comprehensive Study. Food Funct. 2020, 11, 1292–1303. [Google Scholar] [CrossRef]
- Barros, L.; Pereira, C.; Ferreira, I.C.F.R. Optimized Analysis of Organic Acids in Edible Mushrooms from Portugal by Ultra Fast Liquid Chromatography and Photodiode Array Detection. Food Anal. Methods 2013, 6, 309–316. [Google Scholar] [CrossRef]
- Obodai, M.; Narh Mensah, D.; Fernandes, Â.; Kortei, N.; Dzomeku, M.; Teegarden, M.; Schwartz, S.; Barros, L.; Prempeh, J.; Takli, R.; et al. Chemical Characterization and Antioxidant Potential of Wild Ganoderma Species from Ghana. Molecules 2017, 22, 196. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, Â.; Barros, L.; Antonio, A.L.; Barreira, J.C.M.; Oliveira, M.B.P.P.; Martins, A.; Ferreira, I.C.F.R. Using Gamma Irradiation to Attenuate the Effects Caused by Drying or Freezing in Macrolepiota procera Organic Acids and Phenolic Compounds. Food Bioprocess. Technol. 2014, 7, 3012–3021. [Google Scholar] [CrossRef][Green Version]
- Sarmento, A.; Barros, L.; Fernandes, Â.; Carvalho, A.M.; Ferreira, I.C. Valorization of Traditional Foods: Nutritional and Bioactive Properties of Cicer arietinum L. and Lathyrus sativus L. Pulses: Valorization of Traditional Foods. J. Sci. Food Agric. 2015, 95, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Lockowandt, L.; Pinela, J.; Roriz, C.L.; Pereira, C.; Abreu, R.M.V.; Calhelha, R.C.; Alves, M.J.; Barros, L.; Bredol, M.; Ferreira, I.C.F.R. Chemical Features and Bioactivities of Cornflower (Centaurea cyanus L.) Capitula: The Blue Flowers and the Unexplored Non-Edible Part. Ind. Crops Prod. 2019, 128, 496–503. [Google Scholar] [CrossRef]
- Pires, T.C.S.P.; Dias, M.I.; Barros, L.; Calhelha, R.C.; Alves, M.J.; Oliveira, M.B.P.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Edible Flowers as Sources of Phenolic Compounds with Bioactive Potential. Food Res. Int. 2018, 105, 580–588. [Google Scholar] [CrossRef]
- Carneiro, A.A.J.; Ferreira, I.C.F.R.; Dueñas, M.; Barros, L.; da Silva, R.; Gomes, E.; Santos-Buelga, C. Chemical Composition and Antioxidant Activity of Dried Powder Formulations of Agaricus blazei and Lentinus edodes. Food Chem. 2013, 138, 2168–2173. [Google Scholar] [CrossRef]
- Thatoi, H.; Singdevsachan, S.K. Diversity, Nutritional Composition and Medicinal Potential of Indian Mushrooms: A Review. Afr. J. Biotechnol. 2014, 13, 598–603. [Google Scholar] [CrossRef]
- Cho, S.-M.; Jang, K.-Y.; Park, H.-J.; Park, J.-S. Analysis of the Chemical Constituents of Agaricus brasiliensis. Mycobiology 2008, 36, 50–54. [Google Scholar] [CrossRef]
- Tsai, S.-Y.; Tsai, H.-L.; Mau, J.-L. Non-Volatile Taste Components of Agaricus blazei, Agrocybe cylindracea and Boletus edulis. Food Chem. 2008, 107, 977–983. [Google Scholar] [CrossRef]
- Carvajal, A.E.S.S.; Koehnlein, E.A.; Soares, A.A.; Eler, G.J.; Nakashima, A.T.A.; Bracht, A.; Peralta, R.M. Bioactives of Fruiting Bodies and Submerged Culture Mycelia of Agaricus brasiliensis (A. Blazei) and Their Antioxidant Properties. LWT-Food Sci. Technol. 2012, 46, 493–499. [Google Scholar] [CrossRef]
- Gąsecka, M.; Magdziak, Z.; Siwulski, M.; Mleczek, M. Profile of Phenolic and Organic Acids, Antioxidant Properties and Ergosterol Content in Cultivated and Wild Growing Species of Agaricus. Eur. Food Res. Technol. 2018, 244, 259–268. [Google Scholar] [CrossRef]
- Bentsen, H. Dietary Polyunsaturated Fatty Acids, Brain Function and Mental Health. Microb. Ecol. Health Dis. 2017, 28, 1281916. [Google Scholar] [CrossRef]
- Tsai, S.-Y.; Tsai, H.-L.; Mau, J.-L. Antioxidant Properties of Agaricus blazei, Agrocybe cylindracea, and Boletus edulis. LWT-Food Sci. Technol. 2007, 40, 1392–1402. [Google Scholar] [CrossRef]
- Barouh, N.; Bourlieu-Lacanal, C.; Figueroa-Espinoza, M.C.; Durand, E.; Villeneuve, P. Tocopherols as Antioxidants in Lipid-based Systems: The Combination of Chemical and Physicochemical Interactions Determines Their Efficiency. Compr. Rev. Food Sci. Food Saf. 2022, 21, 642–688. [Google Scholar] [CrossRef]
- Frankel, E.N. The Antioxidant and Nutritional Effects of Tocopherols, Ascorbic Acid and Beta-Carotene in Relation to Processing of Edible Oils. Bibl. Nutr. Dieta 1989, 43, 297–312. [Google Scholar] [CrossRef]
- Stojković, D.; Reis, F.S.; Glamočlija, J.; Ćirić, A.; Barros, L.; Van Griensven, L.J.L.D.; Ferreira, I.C.F.R.; Soković, M. Cultivated Strains of Agaricus bisporus and A. brasiliensis: Chemical Characterization and Evaluation of Antioxidant and Antimicrobial Properties for the Final Healthy Product—Natural Preservatives in Yoghurt. Food Funct. 2014, 5, 1602. [Google Scholar] [CrossRef]
- Wei, Q.; Zhan, Y.; Chen, B.; Xie, B.; Fang, T.; Ravishankar, S.; Jiang, Y. Assessment of Antioxidant and Antidiabetic Properties of Agaricus blazei Murill Extracts. Food Sci. Nutr. 2020, 8, 332–339. [Google Scholar] [CrossRef]
- Khatua, S.; Paul, S.; Acharya, K. Mushroom as the Potential Source of New Generation of Antioxidant: A Review. Res. J. Pharm. Technol. 2013, 6, 496–505. [Google Scholar]
- Morales, D.; Piris, A.J.; Ruiz-Rodriguez, A.; Prodanov, M.; Soler-Rivas, C. Extraction of bioactive compounds against cardiovascular diseases from Lentinula edodes using a sequential extraction method. Biotechnol. Prog. 2018, 34, 746–755. [Google Scholar] [CrossRef]
- Mazzutti, S.; Ferreira, S.R.S.; Riehl, C.A.S.; Smania, A.; Smania, F.A.; Martínez, J. Supercritical Fluid Extraction of Agaricus brasiliensis: Antioxidant and Antimicrobial Activities. J. Supercrit. Fluids 2012, 70, 48–56. [Google Scholar] [CrossRef]
- Alves, M.J.; Ferreira, I.C.F.R.; Froufe, H.J.C.; Abreu, R.M.V.; Martins, A.; Pintado, M. Antimicrobial Activity of Phenolic Compounds Identified in Wild Mushrooms, SAR Analysis and Docking Studies. J. Appl. Microbiol. 2013, 115, 346–357. [Google Scholar] [CrossRef] [PubMed]



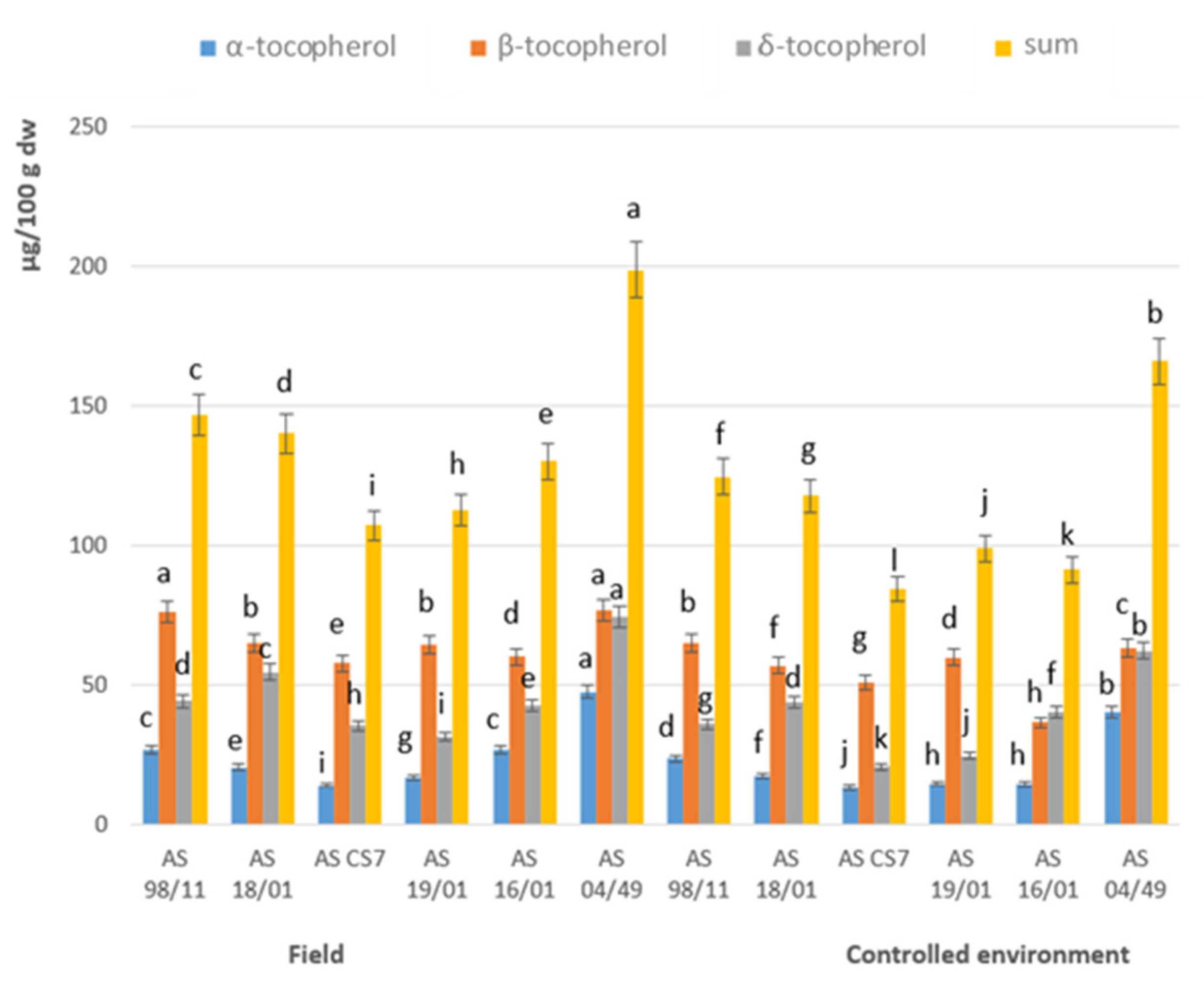
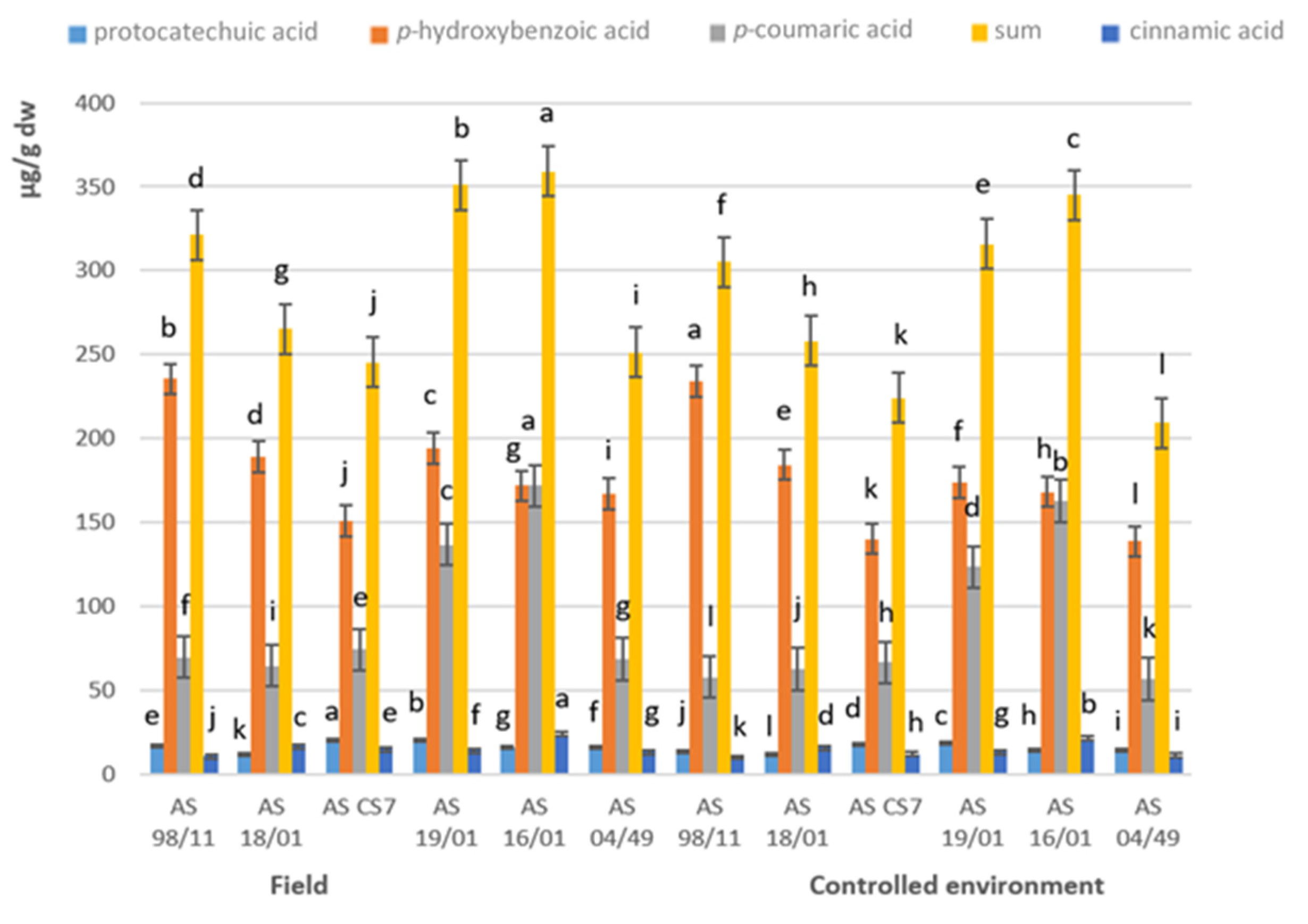
| Field | Controlled Environment | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AS 98/11 | AS 18/01 | AS CS7 | AS 19/01 | AS 16/01 | AS 04/49 | AS 98/11 | AS 18/01 | AS CS7 | AS 19/01 | AS 16/01 | AS 04/49 | |
| Nutritional value (g/100 g dw) | ||||||||||||
| Fat | 1.96 ± 0.03 a | 1.8 ± 0.1 b | 1.54 ± 0.05 d | 1.42 ± 0.04 e | 1.9 ± 0.1 a | 1.94 ± 0.02 a | 1.47 ± 0.02 e | 1.6 ± 0.1 c | 1.44 ± 0.04 e | 1.6 ± 0.1 c | 1.73 ± 0.01 b | 1.93 ± 0.04 a |
| Proteins | 29 ± 1 b | 26.8 ± 0.1 c | 27 ± 1 c | 22.3 ± 0.2 g | 28.83 ± 0.04 b | 25.0 ± 0.1 e | 26.7 ± 0.4 c | 22.46 ± 0.01 g | 24.5 ± 0.4 f | 18.8 ± 0.2 h | 35.8 ± 0.1 a | 25.6 ± 0.2 d |
| Ash | 10 ± 1 b | 9.4 ± 0.1 c | 9.3 ± 0.3 c | 8.1 ± 0.2 f | 10.66 ± 0.03 a | 8.67 ± 0.05 e | 8.4 ± 0.3 f | 7.2 ± 0.3 h | 7.5 ± 0.2 g | 7.13 ± 0.02 h | 9.0 ± 0.3 d | 7.3 ± 0.2 gh |
| Carbohydrates | 59 ± 1 i | 62.0 ± 0.1 h | 62.3 ± 0.6 h | 68.11 ± 0.03 c | 58.6 ± 0.1 j | 64.4 ± 0.1 f | 63.4 ± 0.1 g | 68.8 ± 0.1 b | 66.6 ± 0.5 d | 72.6 ± 0.2 a | 53.4 ± 0.3 k | 65.2 ± 0.1 e |
| Energy (Kcal/100 g dw) | 368 ± 2 g | 371 ± 1 f | 370 ± 1 f | 375 ± 1 c,d | 367.0 ± 0.5 h | 375.0 ± 0.1 c | 374 ± 1 d | 380 ± 1 a | 377.1 ± 0.5 b | 379.3 ± 0.3 a | 373 ± 1 e | 380 ± 1 a |
| Free sugars (g/100 g dw) | ||||||||||||
| Fructose | 0.270 ± 0.004 b | 0.280 ± 0.008 a | 0.280 ± 0.02 a | 0.220 ± 0.004 e | 0.240 ± 0.004 d | 0.260 ± 0.004 c | 0.170 ± 0.02 g | 0.210 ± 0.003 f | 0.240 ± 0.003 d | 0.170 ± 0.006 g | 0.270 ± 0.005 b | 0.220 ± 0.002 e |
| Glucose | 19.6 ± 0.2 i | 25.8 ± 0.1 h | 28.3 ± 0.2 f | 29.6 ± 0.3 e | 18.6 ± 0.1 j | 16.8 ± 0.5 k | 27.8 ± 0.4 g | 31.66 ± 0.02 c | 34.1 ± 0.3 b | 39.2 ± 0.3 a | 27.6 ± 0.1 g | 30.8 ± 0.5 d |
| Trehalose | 1.03 ± 0.01 e | 0.950 ± 0.002 f | 0.90 ± 0.01 g | 2.06 ± 0.02 b | 0.79 ± 0.02 i | 0.21 ± 0.01 j | 0.8 ± 0.1 h,i | 1.21 ± 0.04 d | 0.87 ± 0.02 g, h | 3.7 ± 0.1 a | 1.5 ± 0.1 c | 0.85 ± 0.02 g,h |
| Sum | 20.9 ± 0.2 h | 27.1 ± 0.1 g | 29.5 ± 0.2 e | 31.9 ± 0.2 d | 19.63 ± 0.04 i | 17.3 ± 0.5 j | 28.8 ± 0.5 f | 33.09 ± 0.02 c | 35.2 ± 0.3 b | 43.1 ± 0.4 a | 29.4 ± 0.2 e | 31.9 ± 0.5 d |
| Organic acids (g/100 g dw) | ||||||||||||
| Oxalic acid | 2.71 ± 0.07 e | 2.75 ± 0.01 e | 2.509 ± 0.003 f | 1.83 ± 0.04 i | 4.4 ± 0.1 a | 3.07 ± 0.04 c | 2.2 ± 0.1 h | 2.53 ± 0.03 f | 2.32 ± 0.03 g | 1.77 ± 0.02 j | 3.96 ± 0.01 b | 2.80 ± 0.02 d |
| Malic acid | 24.0 ± 0.2 c | 26.2 ± 0.2 b | 27.52 ± 0.02 a | 18.2 ± 0.3 f | 3.6 ± 0.1 h | 3.22 ± 0.03 k | 19.7 ± 0.1 d | 19.3 ± 0.1 e | 23.9 ± 0.2 c | 14.4 ± 0.1 g | 3.42 ± 0.03 i | 3.05 ± 0.03 j |
| Citric acid | 16.42 ± 0.01 c | 19.0 ± 0.1 a | 16.6 ± 0.1 b | 12.33 ± 0.05 k | 16.0 ± 0.2 d | 14.2 ± 0.2 h | 14.6 ± 0.2 f | 14.38 ± 0.01 g | 15.75 ± 0.03 e | 10.7 ± 0.1 l | 13.8 ± 0.1 j | 13.98 ± 0.04 i |
| Fumaric acid | tr | tr | tr | tr | tr | tr | tr | tr | tr | tr | tr | tr |
| Sum | 43.2 ± 0.2 c | 48.0 ± 0.4 a | 46.7 ± 0.1 b | 32.3 ± 0.2 g | 24.0 ± 0.3 i | 20.5 ± 0.1 k | 36.5 ± 0.4 e | 36.2 ± 0.1 f | 42.0 ± 0.3 d | 26.9 ± 0.2 h | 21.2 ± 0.1 j | 19.83 ± 0.02 l |
| Field | Controlled Environment | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AS 98/11 | AS 18/01 | AS CS7 | AS 19/01 | AS 16/01 | AS 04/49 | AS 98/11 | AS 18/01 | AS CS7 | AS 19/01 | AS 16/01 | AS 04/49 | |
| Fatty Acids (%) | ||||||||||||
| C16:0 | 12.7 ± 0.2 | 15 ± 1 | 14.3 ± 0.1 | 17.60 ± 0.03 | 13.5 ± 0.1 | 13.4 ± 0.3 | 12.31 ± 0.01 | 15.0 ± 0.1 | 17 ± 1 | 16.5 ± 0.2 | 15 ± 1 | 14 ± 1 |
| C18:0 | 3.8 ± 0.1 | 4.9 ± 0.3 | 4.42 ± 0.07 | 4.97 ± 0.02 | 5.0 ± 0.1 | 4.4 ± 0.2 | 4.250 ± 0.001 | 4.6 ± 0.3 | 5.23 ± 0.01 | 5.15 ± 0.05 | 4.9 ± 0.3 | 4.9 ± 0.3 |
| C18:1n9c | 1.2 ± 0.1 | 2.1 ± 0.2 | 1.54 ± 0.03 | 1.5 ± 0.1 | 1.6 ± 0.1 | 1.28 ± 0.03 | 1.19 ± 0.03 | 1.5 ± 0.1 | 1.9 ± 0.1 | 2.0 ± 0.1 | 1.6 ± 0.1 | 1.4 ± 0.1 |
| C18:2n6c | 73.4 ± 0.2 | 68 ± 1 | 66.2 ± 0.2 | 64 ± 1 | 70.0 ± 0.3 | 71 ± 1 | 72.8 ± 0.1 | 68.1 ± 0.1 | 64 ± 1 | 64 ± 1 | 67 ± 2 | 67.5 ± 0.4 |
| C20:0 | 1.48 ± 0.01 | 1.69 ± 0.03 | 1.38 ± 0.01 | 1.69 ± 0.01 | 1.7 ± 0.1 | 1.7 ± 0.1 | 1.50 ± 0.01 | 1.34 ± 0.04 | 1.83 ± 0.03 | 1.7 ± 0.2 | 1.6 ± 0.1 | 1.6 ± 0.1 |
| C22:0 | 2.83 ± 0.03 | 3.1 ± 0.1 | 3.06 ± 0.04 | 3.1 ± 0.1 | 3.08 ± 0.05 | 3.04 ± 0.02 | 3.19 ± 0.03 | 3.3 ± 0.1 | 3.3 ± 0.1 | 3.5 ± 0.3 | 3.06 ± 0.03 | 3.5 ± 0.3 |
| SFA | 24.7 ± 0.3 i | 29 ± 1 d | 31.2 ± 0.2 b | 33 ± 1 a | 27.8 ± 0.2 f | 27 ± 1 g | 25.3 ± 0.1 h | 29.7 ± 0.2 c | 33 ± 1 a | 33 ± 1 a | 30 ± 2 b | 30.4 ± 0.4 b,c |
| MUFA | 1.7 ± 0.1 i | 2.8 ± 0.2 a | 2.44 ± 0.01 c | 2.2 ± 0.1 d | 2.1 ± 0.1 e | 1.8 ± 0.1 h | 1.62 ± 0.04 j | 2.0 ± 0.2 f | 2.2 ± 0.1 d | 2.6 ± 0.1 b | 2.1 ± 0.1 e | 1.94 ± 0.03 g |
| PUFA | 73.6 ± 0.2 a | 68 ± 1 c | 66.3 ± 0.2 e | 65 ± 1 f | 70.1 ± 0.3b | 71 ± 1 b | 73.0 ± 0.1 a | 68.3 ± 0.1 c | 65 ± 1 f | 64 ± 1 g | 67 ± 2 d | 67.7 ± 0. 4 c,d |
| TBARS (EC50; mg/mL) * | OxHLIA (IC50; µg/mL, Δt = 60 min) ** | ||
|---|---|---|---|
| Field | AS 98/11 | 1.91 ± 0.05 a | 221 ± 6 c |
| AS 18/01 | 0.60 ± 0.04 h | 88 ± 4 h,i | |
| AS CS7 | 0.44 ± 0.03 i | 95 ± 3 g,h | |
| AS 19/01 | 1.89 ± 0.04 a | 296 ± 9 b | |
| AS 16/01 | 0.50 ± 0.01 i | 96 ± 2 g | |
| AS 04/49 | 0.93 ± 0.03 c,d | 93 ± 4 g,h | |
| Controlled Environment | AS 98/11 | 1.7 ± 0.1 b | 310 ± 10 a |
| AS 18/01 | 1.9 ± 0.1 a | 111 ± 4 f | |
| AS CS7 | 0.96 ± 0.02 c | 199 ± 1 d | |
| AS 19/01 | 0.72 ± 0.04 g | 133 ± 5 e | |
| AS 16/01 | 0.88 ± 0.03 d,e | 89 ± 3 g,h,i | |
| AS 04/49 | 0.8 ± 0.1 f | 83 ± 2 j |
| Gram-Negative Bacteria | Gram-Positive Bacteria | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Escherichia coli | Klebsiella pneumoniae | Morganella morganii | Proteus mirabilis | Pseudomonas aeruginosa | Enterococcus faecalis | Listeria monocytogenes | MRSA | |||
| Field | AS 98/11 | MIC | 20 | >20 | >20 | >20 | >20 | >20 | 20 | 20 |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS 18/01 | MIC | 20 | >20 | >20 | >20 | >20 | >20 | >20 | 20 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS CS7 | MIC | 20 | >20 | >20 | >20 | >20 | 20 | 20 | 20 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS 19/01 | MIC | 10 | >20 | >20 | >20 | >20 | 10 | 10 | 10 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS 16/01 | MIC | 20 | >20 | >20 | >20 | >20 | 20 | 20 | 20 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS 04/49 | MIC | 20 | >20 | >20 | >20 | >20 | 20 | 20 | 20 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| Controlled environment | AS 98/11 | MIC | 20 | >20 | >20 | >20 | >20 | >20 | >20 | 20 |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS 18/01 | MIC | 20 | >20 | >20 | >20 | >20 | 20 | 20 | 20 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS CS7 | MIC | 20 | >20 | >20 | >20 | >20 | 20 | 20 | 20 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS 19/01 | MIC | 10 | >20 | >20 | >20 | >20 | 10 | 10 | 10 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS 16/01 | MIC | 20 | >20 | >20 | >20 | >20 | 20 | 20 | 20 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| AS 04/49 | MIC | 20 | >20 | >20 | >20 | >20 | 20 | 20 | 20 | |
| MBC | >20 | >20 | >20 | >20 | >20 | >20 | >20 | >20 | ||
| Negative controls | Ampicillin (20 mg/mL) | MIC | <0.15 | 10 | >20 | >20 | >20 | <0.15 | <0.15 | <0.15 |
| MBC | <0.15 | 20 | >20 | >20 | >20 | <0.15 | <0.15 | <0.15 | ||
| Imipenem (1 mg/mL) | MIC | <0.0078 | <0.0078 | <0.0078 | <0.0078 | 0.5 | n.t. | <0.0078 | n.t. | |
| MBC | <0.0078 | <0.0078 | <0.0078 | <0.0078 | 1 | n.t. | <0.0078 | n.t. | ||
| Vancomycin (1 mg/mL) | MIC | n.t. | n.t. | n.t. | n.t. | n.t. | <0.0078 | n.t. | 0.25 | |
| MBC | n.t. | n.t. | n.t. | n.t. | n.t. | <0.0078 | n.t. | 0.5 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almeida, D.; Cardoso, R.V.C.; Pereira, C.; Alves, M.J.; Ferreira, I.C.F.R.; Zied, D.C.; Junior, W.G.V.; Caitano, C.E.C.; Fernandes, Â.; Barros, L. Biochemical Approaches on Commercial Strains of Agaricus subrufescens Growing under Two Environmental Cultivation Conditions. J. Fungi 2022, 8, 616. https://doi.org/10.3390/jof8060616
Almeida D, Cardoso RVC, Pereira C, Alves MJ, Ferreira ICFR, Zied DC, Junior WGV, Caitano CEC, Fernandes Â, Barros L. Biochemical Approaches on Commercial Strains of Agaricus subrufescens Growing under Two Environmental Cultivation Conditions. Journal of Fungi. 2022; 8(6):616. https://doi.org/10.3390/jof8060616
Chicago/Turabian StyleAlmeida, Daiana, Rossana V. C. Cardoso, Carla Pereira, Maria José Alves, Isabel C. F. R. Ferreira, Diego Cunha Zied, Wagner G. Vieira Junior, Cinthia E. C. Caitano, Ângela Fernandes, and Lillian Barros. 2022. "Biochemical Approaches on Commercial Strains of Agaricus subrufescens Growing under Two Environmental Cultivation Conditions" Journal of Fungi 8, no. 6: 616. https://doi.org/10.3390/jof8060616
APA StyleAlmeida, D., Cardoso, R. V. C., Pereira, C., Alves, M. J., Ferreira, I. C. F. R., Zied, D. C., Junior, W. G. V., Caitano, C. E. C., Fernandes, Â., & Barros, L. (2022). Biochemical Approaches on Commercial Strains of Agaricus subrufescens Growing under Two Environmental Cultivation Conditions. Journal of Fungi, 8(6), 616. https://doi.org/10.3390/jof8060616










