Resporulation of Calcium Alginate Encapsulated Metarhizium anisopliae on Metham®-Fumigated Soil and Infectivity on Larvae of Tenebrio molitor
Abstract
1. Introduction
2. Materials and Methods
2.1. Metarhizium anisopliae
2.2. Preparation of M. anisopliae Granules
2.3. Collection of Soil for Experimentation
2.4. Soil Treatment: Fumigation, Pasteurization and Field Soil
2.5. Cultivation of Soil Microbes
2.6. Fungal Inoculation and Experimental Design
2.7. Resporulation Assessment
2.8. Infectivity Assessment
2.9. Data Analysis
3. Results
3.1. Cultivation of Soil Microbes
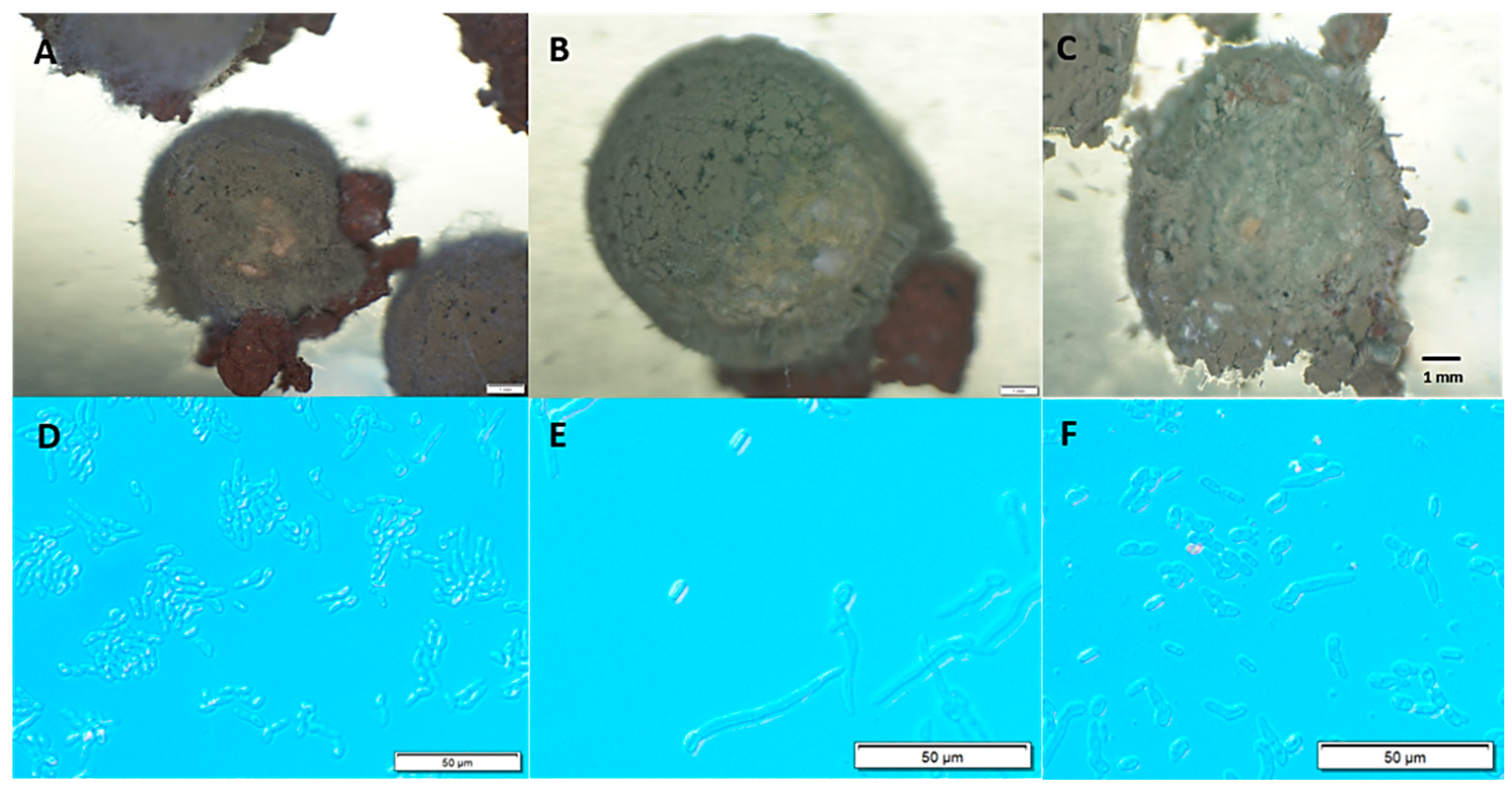
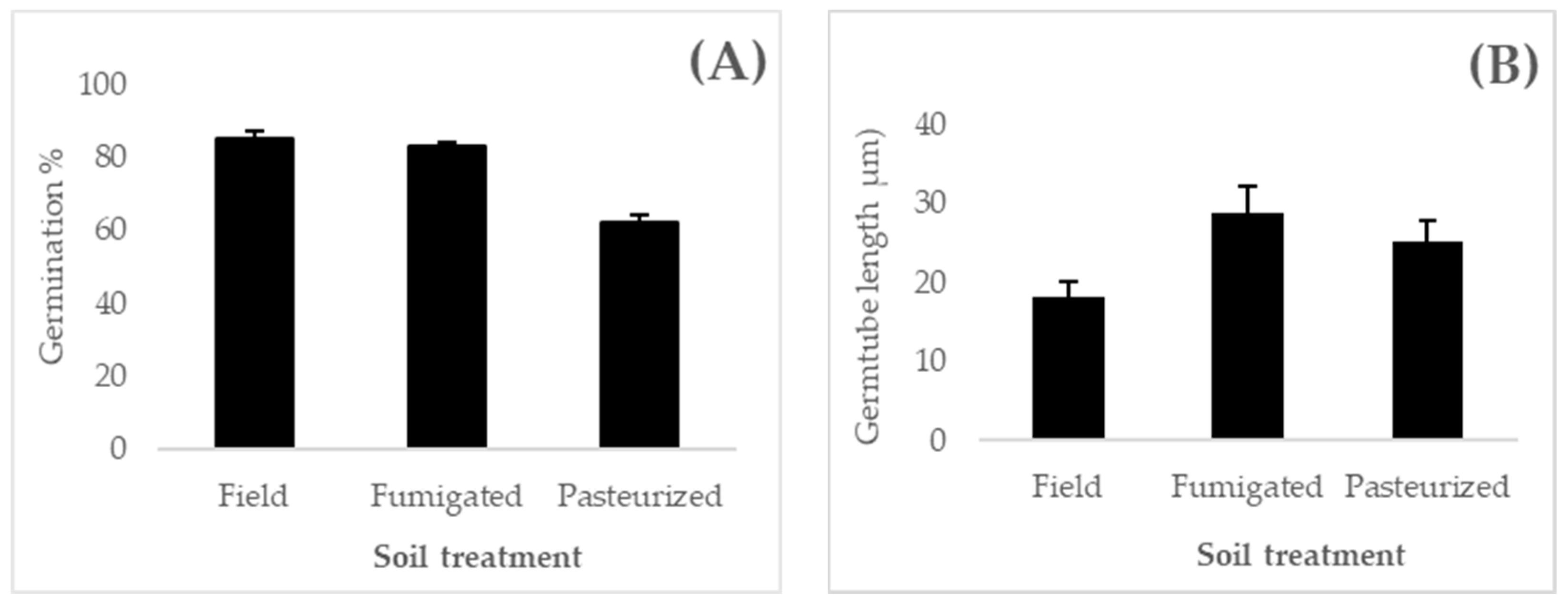
3.2. Resporulation Assessment
3.3. Infectivity Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chalfant, R.; Jansson, R.; Seal, D.; Schalk, J. Ecology, and management of sweetpotato insects. Annu. Rev. Entomol. 1990, 35, 157–180. [Google Scholar] [CrossRef]
- Johnson, A.; Gurr, G. Invertebrate pests, and diseases of sweetpotato (Ipomoea batatas): A review and identification of research priorities for smallholder production. Ann. Appl. Biol. 2016, 168, 291–320. [Google Scholar] [CrossRef]
- McCrystal, R. Improving Sweetpotato Soil Insect Pests; Horticulture Australia Limited: Sydney, Australia, 2010. [Google Scholar]
- Dotaona, R.; Wilson, B.A.L.; Stevens, M.M.; Holloway, J.; Ash, G.J. Screening of tropical isolates of Metarhizium anisopliae (Hypocreales: Clavicipitaceae) for virulence to the sweetpotato weevil, Cylas formicarius (Coleoptera: Brentidae). Int. J. Trop. Insect Sci. 2015, 35, 153–163. [Google Scholar] [CrossRef]
- Stirling, G.; Stirling, A.; Prichard, M. Sustainable sweetpotato farming systems to improve soil health and reduce losses caused by root-knot nematode. Australas. Plant Pathol. 2020, 49, 591–604. [Google Scholar] [CrossRef]
- Australia Sweetpotatoes: Strategic Agricchemical Review Process 2011–2014; Horticulture Australia: Sydney, Australia, 2014; Available online: https://ausveg.com.au/app/uploads/2017/05/Sweet_potato_SARP_report_March_14_2014.pdf (accessed on 1 October 2018).
- Medo, J.; Cagáň, Ľ. Factors affecting the occurrence of entomopathogenic fungi in soils of Slovakia as revealed using two methods. Biol. Control 2011, 59, 200–208. [Google Scholar] [CrossRef]
- Quesada-Moraga, E.; Navas-Cortes, J.A.; Maranhao, E.A.A.; Ortiz-Urquiza, A.; Santiago-Alvarez, C. Factors affecting the occurrence and distribution of entomopathogenic fungi in natural and cultivated soils. Mycol. Res. 2007, 111, 947–966. [Google Scholar] [CrossRef]
- Jabbour, R.; Barbercheck, M.E. Soil management effects on entomopathogenic fungi during the transition to organic agriculture in a feed grain rotation. Biol. Control 2009, 51, 435–443. [Google Scholar] [CrossRef]
- Castro, T.; Mayerhofer, J.; Enkerli, J.; Eilenberg, J.; Meyling, N.V.; de Andrade Moral, R.; Demétrio, C.G.B.; Delalibera, I. Persistence of Brazilian isolates of the entomopathogenic fungi Metarhizium anisopliae and M. robertsii in strawberry crop soil after soil drench application. Agric. Ecosyst. Environ. 2016, 233, 361–369. [Google Scholar] [CrossRef]
- Uzman, D.; Pliester, J.; Leyer, I.; Entling, M.H.; Reineke, A. Drivers of entomopathogenic fungi presence in organic and conventional vineyard soils. Appl. Soil Ecol. 2019, 133, 89–97. [Google Scholar] [CrossRef]
- Rath, A.; Koen, T.; Anderson, G.; Worledge, D. Field evaluation of the entomogenous fungus Metarhizium anisopliae (DAT F-001) as a biocontrol agent for the redheaded pasture cockchafer, Adoryphorus couloni (Coleoptera: Scarabaeidae). Aust. J. Agric. Res. 1995, 46, 429–440. [Google Scholar] [CrossRef]
- Ekesi, S.; Maniania, N.K.; Mohamed, S.A.; Lux, S.A. Effect of soil application of different formulations of Metarhizium anisopliae on African tephritid fruit flies and their associated endoparasitoids. Biol. Control 2005, 35, 83–91. [Google Scholar] [CrossRef]
- Kabaluk, J.T.; Vernon, R.S.; Goettel, M.S. Mortality and infection of wireworm, Agriotes obscurus (Coleoptera: Elateridae), with inundative field applications of Metarhizium anisopliae. Phytoprotection 2007, 88, 51–56. [Google Scholar] [CrossRef]
- Jaronski, S.T. Ecological factors in the inundative use of fungal entomopathogens. BioControl 2010, 55, 159–185. [Google Scholar] [CrossRef]
- Putnoky-Csicsó, B.; Tonk, S.; Szabó, A.; Márton, Z.; Tóthné Bogdányi, F.; Tóth, F.; Abod, É.; Bálint, J.; Balog, A. Effectiveness of the Entomopathogenic Fungal Species Metarhizium anisopliae Strain NCAIM 362 Treatments against Soil Inhabiting Melolontha melolontha Larvae in Sweetpotato (Ipomoea batatas L.). J. Fungi 2020, 6, 116. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.; Evans, M.; Butt, T. Identification of pathogenic strains of entomopathogenic nematodes and fungi for wireworm control. Crop Prot. 2009, 28, 269–272. [Google Scholar] [CrossRef]
- Razinger, J.; Lutz, M.; Schroers, H.-J.; Urek, G.; Grunder, J. Laboratory testing of insect associated fungi for the control of wireworms (Agriotes sp. L.). IOBC-WPRS Bull. 2013, 90, 103–107. [Google Scholar]
- Hu, G.; Leger, R.J.S. Field studies using a recombinant mycoinsecticide (Metarhizium anisopliae) reveal that it is rhizosphere competent. Appl. Environ. Microbiol. 2002, 68, 6383–6387. [Google Scholar] [CrossRef]
- Razinger, J.; Praprotnik, E.; Schroers, H.-J. Bioaugmentation of Entomopathogenic Fungi for Sustainable Agriotes Larvae (Wireworms) Management in Maize. Front. Plant Sci. 2020, 11, 535005. [Google Scholar] [CrossRef]
- Greenfield, M.; Gómez-Jiménez, M.I.; Ortiz, V.; Vega, F.E.; Kramer, M.; Parsa, S. Beauveria bassiana and Metarhizium anisopliae endophytically colonize cassava roots following soil drench inoculation. Biol. Control 2016, 95, 40–48. [Google Scholar] [CrossRef]
- Krell, V.; Jakobs-Schoenwandt, D.; Vidal, S.; Patel, A.V. Encapsulation of Metarhizium brunneum enhances endophytism in tomato plants. Biol. Control 2017, 116, 62–73. [Google Scholar] [CrossRef]
- Ahmad, I.; del Mar Jiménez-Gasco, M.; Luthe, D.S.; Shakeel, S.N.; Barbercheck, M.E. Endophytic Metarhizium robertsii promotes maize growth, suppresses insect growth, and alters plant defense gene expression. Biol. Control 2020, 144, 104167. [Google Scholar] [CrossRef]
- Mayerhofer, J.; Enkerli, J.; Zelger, R.; Strasser, H. Biological control of the European cockchafer: Persistence of Beauveria brongniartii after long-term applications in the Euroregion Tyrol. BioControl 2015, 60, 617–629. [Google Scholar] [CrossRef]
- Ekesi, S.; Maniania, N.K.; Mohamed, S.A. Efficacy of soil application of Metarhizium anisopliae and the use of GF-120 spinosad bait spray for suppression of Bactrocera invadens (Diptera: Tephritidae) in mango orchards. Biocontrol Sci. Technol. 2011, 21, 299–316. [Google Scholar] [CrossRef]
- Mayerhofer, J.; Rauch, H.; Hartmann, M.; Widmer, F.; Gschwend, F.; Strasser, H.; Leuchtmann, A.; Enkerli, J. Response of soil microbial communities to the application of a formulated Metarhizium brunneum biocontrol strain. Biocontrol Sci. Technol. 2019, 29, 547–564. [Google Scholar] [CrossRef]
- Schwarzenbach, K.; Enkerli, J.; Widmer, F. Effects of biological and chemical insect control agents on fungal community structures in soil microcosms. Appl. Soil Ecol. 2009, 42, 54–62. [Google Scholar] [CrossRef]
- Topuz, E.; Erler, F.; Gumrukcu, E. Survey of indigenous entomopathogenic fungi and evaluation of their pathogenicity against the carmine spider mite, Tetranychus cinnabarinus (Boisd.), and the whitefly, Bemisia tabaci (Genn.) biotype B. Pest Manag. Sci. 2016, 72, 2273–2279. [Google Scholar] [CrossRef] [PubMed]
- Kessler, P.; Enkerl, J.; Schweize, C.; Keller, S. Survival of Beauveria brongniartii in the soil after application as a biocontrol agent against the European cockchafer Melolontha melolontha. Biocontrol 2004, 49, 563–581. [Google Scholar] [CrossRef]
- Bonanomi, G.; Gaglione, S.A.; Incerti, G.; Zoina, A. Biochemical quality of organic amendments affects soil fungistasis. Appl. Soil Ecol. 2013, 72, 135–142. [Google Scholar] [CrossRef]
- Shah, S.; Ash, G.J.; Wilson, B.A. Resporulation on Metarhizium anisopliae granules on soil and mortality of Tenebrio molitor: Implications for wireworm management in sweetpotato. Ann. Appl. Biol. 2022, 1–12. [Google Scholar] [CrossRef]
- Gullino, M.L.; Garibaldi, A.; Gamliel, A.; Katan, J. Soil Disinfestation: From Soil Treatment to Soil and Plant Health. Plant Dis. 2022, 106, 1541–1554. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Ma, Y.; Yang, H.; Chang, Z. Effect of biofumigation and chemical fumigation on soil microbial community structure and control of pepper Phytophthora blight. World J. Microbiol. Biotechnol. 2014, 30, 507–518. [Google Scholar] [CrossRef]
- Stapleton, J.; Elmore, C.; DeVay, J. Solarization and biofumigation help disinfest soil. Calif. Agric. 2000, 54, 42–45. [Google Scholar] [CrossRef]
- Casu, S.; Fanti, A.; Lodi, M.B.; Spanu, M.; Desogus, F.; Mazzarella, G. Numerical Estimation of Agricultural Raised Bed Microwave Disinfection. Radio Sci. 2018, 53, 1176–1186. [Google Scholar] [CrossRef]
- Morra, L.; Carrieri, R.; Fornasier, F.; Mormile, P.; Rippa, M.; Baiano, S.; Cermola, M.; Piccirillo, G.; Lahoz, E. Solarization working like a “solar hot panel” after compost addition sanitizes soil in thirty days and preserves soil fertility. Appl. Soil Ecol. 2018, 126, 65–74. [Google Scholar] [CrossRef]
- Tseng, W.-H.; Juan, D.; Hsiao, W.-C.; Chan, C.-H.; Ma, H.-Y.; Lee, H.-Y. Study of solar sterilization system for organic soil treatment. In Proceedings of the 2018 IEEE International Conference on Applied System Invention (ICASI), Chiba, Japan, 13–17 April 2018; pp. 865–868. [Google Scholar]
- Omirou, M.; Rousidou, C.; Bekris, F.; Papadopoulou, K.K.; Menkissoglou-Spiroudi, U.; Ehaliotis, C.; Karpouzas, D.G. The impact of biofumigation and chemical fumigation methods on the structure and function of the soil microbial community. Microb. Ecol. 2011, 61, 201–213. [Google Scholar] [CrossRef]
- Kruger, D.; Fourie, J.; Malan, A.P. Cover crops with biofumigation properties for the suppression of plant-parasitic nematodes: A review. S. Afr. J. Enol. Vitic. 2013, 34, 287–295. [Google Scholar] [CrossRef]
- Borek, V.; Elberson, L.R.; McCaffrey, J.P.; Morra, M.J. Toxicity of isothiocyanates produced by glucosinolates in Brassicaceae species to black vine weevil eggs. J. Agric. Food Chem. 1998, 46, 5318–5323. [Google Scholar] [CrossRef]
- Di Primo, P.; Gamliel, A.; Austerweil, M.; Steiner, B.; Beniches, M.; Peretz-Alon, I.; Katan, J. Accelerated degradation of metam-sodium and dazomet in soil: Characterization and consequences for pathogen control. Crop Prot. 2003, 22, 635–646. [Google Scholar] [CrossRef]
- Klose, S.; Ajwa, H.A.; Browne, G.T.; Subbarao, K.V.; Martin, F.N.; Fennimore, S.A.; Westerdahl, B.B. Dose response of weed seeds, plant-parasitic nematodes, and pathogens to twelve rates of metam sodium in a California soil. Plant Dis. 2008, 92, 1537–1546. [Google Scholar] [CrossRef]
- Thies, J.A.; Davis, R.F.; Mueller, J.D.; Fery, R.L.; Langston, D.B.; Miller, G. Host Resistance and Metam Sodium for Managing Root-knot Nematodes in a Pepper–Cucumber Rotation. HortScience 2005, 40, 2080–2082. [Google Scholar] [CrossRef]
- Fravel, D. Interaction of biocontrol fungi with sublethal rates of metham sodium for control of Verticillium dahliae. Crop Prot. 1996, 15, 115–119. [Google Scholar] [CrossRef]
- Matthiessen, J.; Desmarchelier, J.; Vu, L.T.; Shackleton, M. Comparative efficacy of fumigants against hatchling whitefringed beetle (Coleoptera: Curculionidae) larvae and their sorption by soil. J. Econ. Entomol. 1996, 89, 1372–1378. [Google Scholar] [CrossRef]
- Kritzman, G.; Ben-Yephet, Y. Effect of metham-sodium on several bacterial diseases. Acta Hortic. 1988, 255, 49–54. [Google Scholar] [CrossRef]
- Bu, D.; Zhang, C.; Li, W.; Wang, J. Toxicity and field efficacy of metham-sodium against common soil-borne pathogens of summer squash. Acta Phytophylacica Sin. 2014, 41, 562–568. [Google Scholar]
- Li, J.; Huang, B.; Wang, Q.; Li, Y.; Fang, W.; Han, D.; Yan, D.; Guo, M.; Cao, A. Effects of fumigation with metam-sodium on soil microbial biomass, respiration, nitrogen transformation, bacterial community diversity and genes encoding key enzymes involved in nitrogen cycling. Sci. Total Environ. 2017, 598, 1027–1036. [Google Scholar] [CrossRef]
- Sederholm, M.R.; Schmitz, B.W.; Barberán, A.; Pepper, I.L. Effects of metam sodium fumigation on the abundance, activity, and diversity of soil bacterial communities. Appl. Soil Ecol. 2018, 124, 27–33. [Google Scholar] [CrossRef]
- Davis, R.; Nunez, J.; Vargas, R.; Weir, B.; Wright, S.; Munier, D. Metam-sodium kills beneficial soil fungi as well as cotton pests. Calif. Agric. 1996, 50, 42–44. [Google Scholar] [CrossRef]
- Vemmer, M.; Patel, A.V. Review of encapsulation methods suitable for microbial biological control agents. Biol. Control 2013, 67, 380–389. [Google Scholar] [CrossRef]
- BOM. Bureau of Meteorology Australia. 2021. Available online: https://www.bom.gov.au (accessed on 10 May 2021).
- Rangel, D.E.N.; Fernandes, É.K.K.; Dettenmaier, S.J.; Roberts, D.W. Thermotolerance of germlings and mycelium of the insect-pathogenic fungus Metarhizium spp. and mycelial recovery after heat stress. J. Basic Microbiol. 2010, 50, 344–350. [Google Scholar] [CrossRef]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Matthiessen, J.N.; Kirkegaard, J.A. Biofumigation and Enhanced Biodegradation: Opportunity and Challenge in Soilborne Pest and Disease Management. Crit. Rev. Plant Sci. 2006, 25, 235–265. [Google Scholar] [CrossRef]
- Wakelin, S.; Sivasithamparam, K.; Cole, A.; Skipp, R. Saprophytic growth in soil of a strain of Trichoderma koningii. N. Z. J. Agric. Res. 1999, 42, 337–345. [Google Scholar] [CrossRef]
- Zahran, E.; Sauerborn, J.; Abbasher, A.A.; Ahmed, E.A.; Mohukker, R.I.; Karlovsky, P.; Mohamed, E.A.; Müller-Stöver, D. “Pesta” and alginate delivery systems of Fusarium spp. for biological control of Striga hermonthica (Del.) Benth. under Sudanese field conditions. Biol. Control 2008, 44, 160–168. [Google Scholar] [CrossRef]
- Coombes, C.A.; Hill, M.P.; Moore, S.D.; Dames, J.F. Entomopathogenic fungi as control agents of Thaumatotibia leucotreta in citrus orchards: Field efficacy and persistence. BioControl 2016, 61, 729–739. [Google Scholar] [CrossRef]
- St. Leger, R.J.; Wang, J.B. Metarhizium: Jack of all trades, master of many. Open Biol. 2020, 10, 200307. [Google Scholar] [CrossRef]
- Braga, G.U.L.; Destefano, R.H.R.; Messias, C.L. Oxygen consumption by Metarhizium anisopliae during germination and growth of different carbon sources. J. Invertebr. Pathol. 1999, 74, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Lestan, D.; Lamar, R.T. Development of fungal inocula for bioaugmentation of contaminated soils. Appl. Environ. Microbiol. 1996, 62, 2045–2052. [Google Scholar] [CrossRef]
- Camprubí, A.; Estaún, V.; El Bakali, M.; Garcia-Figueres, F.; Calvet, C. Alternative strawberry production using solarization, metham sodium and beneficial soil microbes as plant protection methods. Agron. Sustain. Dev. 2007, 27, 179–184. [Google Scholar] [CrossRef]
- Mayerhofer, J.; Lutz, A.; Widmer, F.; Rehner, S.A.; Leuchtmann, A.; Enkerli, J. Multiplexed microsatellite markers for seven Metarhizium species. J. Invertebr. Pathol. 2015, 132, 132–134. [Google Scholar] [CrossRef]
- Inyang, E.N.; Butt, T.M.; Doughty, K.J.; Todd, A.D.; Archer, S. The effects of isothiocyanates on the growth of the entomopathogenic fungus Metarhizium anisopliae and its infection of the mustard beetle. Mycol. Res. 1999, 103, 974–980. [Google Scholar] [CrossRef]
- Lestari, A.S.; Rao, S. Laboratory bioassays of Metarhizium spp and Beauveria spp against Tenebrio molitor larvae. In Proceedings of the International Symposium for Sustainable Humanosphere, Bogor, Indonesia, 15–16 November 2016; pp. 205–211. [Google Scholar]
- Rath, A.; Worledge, D. The effect of Metarhizium anisopliae (DAT F-001) concentration and exposure time on the survival of the subterranean pasture pest, Adoryphorus couloni (Col.: Scarabaeidae). BioControl 1995, 40, 35–45. [Google Scholar]
- Kabaluk, T.; Li-Leger, E.; Nam, S. Metarhizium brunneum-An enzootic wireworm disease and evidence for its suppression by bacterial symbionts. J. Invertebr. Pathol. 2017, 150 (Suppl. C), 82–87. [Google Scholar] [CrossRef] [PubMed]
- Human, Z.R.; Slippers, B.; Wilhelm de Beer, Z.; Wingfield, M.J.; Venter, S.N. Antifungal actinomycetes associated with the pine bark beetle, Orthotomicus erosus, in South Africa. S. Afr. J. Sci. 2017, 113, 1–7. [Google Scholar] [CrossRef]
- Ericsson, J.D.; Kabaluk, J.T.; Goettel, M.S.; Myers, J.H. Spinosad interacts synergistically with the insect pathogen Metarhizium anisopliae against the exotic wireworms Agriotes lineatus and Agriotes obscurus (Coleoptera: Elateridae). J. Econ. Entomol. 2007, 100, 31–38. [Google Scholar] [CrossRef]
- Ericsson, J.D.; Kabaluk, J.T. Environmental and behavioral constraints on the infection of wireworms by Metarhizium anisopliae. Environ. Entomol. 2007, 36, 1415–1420. [Google Scholar]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shah, S.; Ash, G.J.; Wilson, B.A.L. Resporulation of Calcium Alginate Encapsulated Metarhizium anisopliae on Metham®-Fumigated Soil and Infectivity on Larvae of Tenebrio molitor. J. Fungi 2022, 8, 1114. https://doi.org/10.3390/jof8101114
Shah S, Ash GJ, Wilson BAL. Resporulation of Calcium Alginate Encapsulated Metarhizium anisopliae on Metham®-Fumigated Soil and Infectivity on Larvae of Tenebrio molitor. Journal of Fungi. 2022; 8(10):1114. https://doi.org/10.3390/jof8101114
Chicago/Turabian StyleShah, Sudhan, Gavin J. Ash, and Bree A. L. Wilson. 2022. "Resporulation of Calcium Alginate Encapsulated Metarhizium anisopliae on Metham®-Fumigated Soil and Infectivity on Larvae of Tenebrio molitor" Journal of Fungi 8, no. 10: 1114. https://doi.org/10.3390/jof8101114
APA StyleShah, S., Ash, G. J., & Wilson, B. A. L. (2022). Resporulation of Calcium Alginate Encapsulated Metarhizium anisopliae on Metham®-Fumigated Soil and Infectivity on Larvae of Tenebrio molitor. Journal of Fungi, 8(10), 1114. https://doi.org/10.3390/jof8101114





