Abstract
Cryptococcus neoformans (C. neoformans) is a pathogenic fungus with a global distribution. Humans become infected by inhaling the fungus from the environment, and the fungus initially colonizes the lungs. If the immune system fails to contain C. neoformans in the lungs, the fungus can disseminate to the blood and invade the central nervous system, resulting in fatal meningoencephalitis particularly in immunocompromised individuals including HIV/AIDS patients. Following brain invasion, C. neoformans will encounter host defenses involving resident as well as recruited immune cells in the brain. To overcome host defenses, C. neoformans possesses multiple virulence factors capable of modulating immune responses. The outcome of the interactions between the host and C. neoformans will determine the disease progression. In this review, we describe the current understanding of how C. neoformans migrates to the brain across the blood–brain barrier, and how the host immune system responds to the invading organism in the brain. We will also discuss the virulence factors that C. neoformans uses to modulate host immune responses.
Keywords:
fungus; Cryptococcus neoformans; cryptococcosis; fungal pathogenesis; meningoencephalitis; central nervous system; fungal dissemination; brain invasion; Trojan horse; transcytosis; paracellular crossing; microglia; monocytes; macrophages; T cells; NK cells; cytokines; virulence factors; polysaccharide capsule; laccase; melanin; phospholipase B1; urease; chitin 1. Introduction
Cryptococcus neoformans (C. neoformans) is an encapsulated pathogenic fungus that infects humans and animals [1]. The fungus exists in environments such as soil, trees, and bird droppings. Humans become infected through the inhalation of C. neoformans [2,3]. The fungal cells interact initially with alveolar macrophages in the lungs, which may lead to the phagocytosis of the organism. Macrophages may kill ingested C. neoformans; alternatively, fungal cells can survive or grow within the phagocytes, depending on the host’s immune status [4]. If C. neoformans is not contained in the lungs, the fungus can migrate to the bloodstream and cross the blood–brain barrier (BBB), leading to the infection of the central nervous system (CNS) and meningoencephalitis [1,2,3].
Most cases of cryptococcal meningoencephalitis emerge in immunocompromised patients, including patients with an HIV-infection and organ transplants, although, rarely, immunocompetent patients can also develop the illness [2,3]. Cryptococcal meningoencephalitis is fatal without timely medical care, which involves long-term treatment with amphotericin B, flucytosine, and fluconazole [5]. Even with successful therapy, survivors of this illness often develop neurologic deficits and other adverse effects [6,7]. It was estimated that this fungal disease affects nearly 223,100 people each year and leads to 181,000 deaths annually [8].
Cellular immune responses play an essential role in the clearance of a cryptococcal infection [9]. Inflammatory monocytes/macrophages, microglial cells, and antigen-specific T cells including CD4+ and CD8+ T cells are critically involved in host’s immune responses to C. neoformans infections [10,11]. Th1 immune responses, characterized by IFN-γ, and Th17 immune responses, characterized by IL-17A, mediate the protection against C. neoformans [9,12]. IFN-γ activates macrophages and promotes the classical activation of macrophages, which is correlated with fungal clearance [9,13]. In contrast, Th2 cytokines such as IL-4, IL-5, and IL-13 drive the alternative activation of macrophages, which is associated with disease progression [9,14].
C. neoformans has developed sophisticated mechanisms to enable pathogenesis by escaping host defense mechanisms and modulating immune responses [4]. Its polysaccharide capsule and melanin production are the major virulence factors of C. neoformans [15,16,17]. In addition, C. neoformans secretes a number of virulence-associated enzymes [18]. Those virulence factors are critically involved in C. neoformans’s invasion into the CNS and its suppression of host immune responses. The outcome of the interaction between the host and C. neoformans determines the progression of the disease.
2. Invasion of C. neoformans into the Central Nervous System
As a neurotropic pathogen, C. neoformans invades the brain and causes meningoencephalitis, which represents the main clinical manifestation of cryptococcosis. Two relevant entry sites have been described for neurotropic pathogens including viruses and bacteria: the BBB and the blood–cerebrospinal fluid barrier (BCSFB) [19]. C. neoformans has demonstrated limited ability to migrate through the choroid plexus to invade the CNS, based on evidence that the choroid plexus remains normal and free of fungal cells in infected mice [20,21]. Furthermore, there have been limited clinical cases of choroid plexitis reported in human patients [22,23,24,25], which supports the idea that crossing through the BCSFB plays a minor role during cryptococcal dissemination into the CNS. Instead, cryptococcal clusters have been often observed next to the cortical microvasculature of the brain and cerebellum [20,21], demonstrating that C. neoformans crosses the BBB for brain invasion.
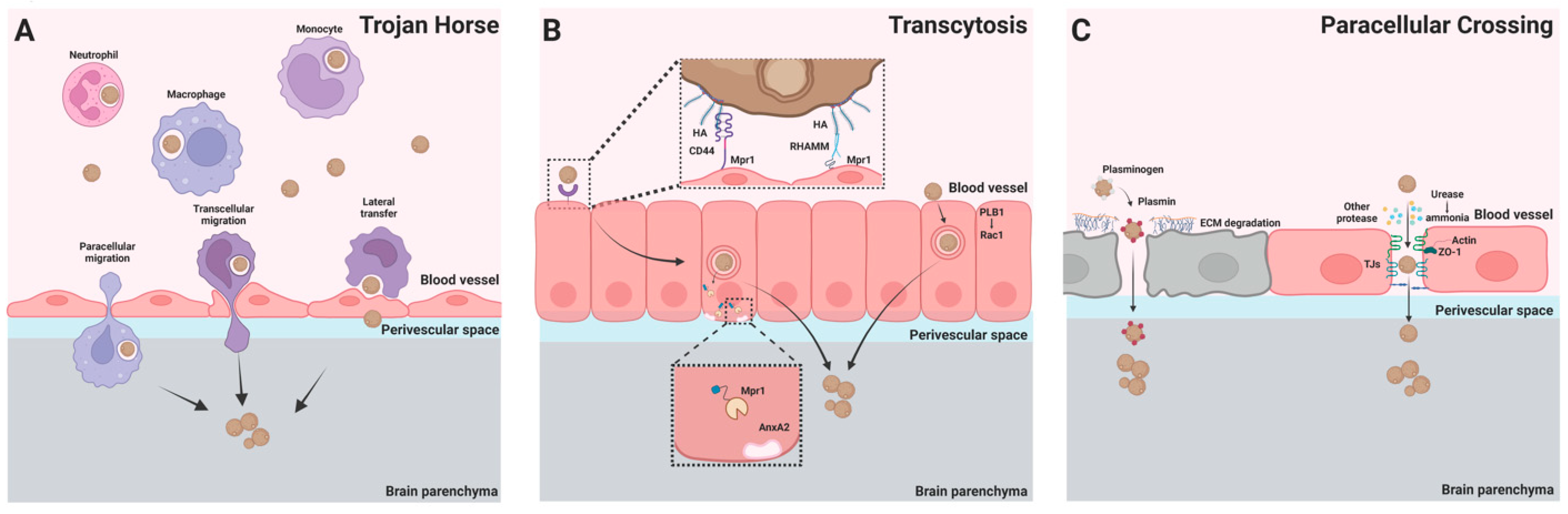
The BBB is a selectively permeable barrier that separates the brain parenchyma from the vascular compartment [26]. The major components of the BBB are endothelial cells, pericytes, and astrocytes [27]. Endothelial cells line the lumen side of the brain vasculature and are connected by tight junctions. During inflammation, circulating leukocytes can transmigrate into the brain parenchyma paracellularly through the tight junction or transcellularly through endocytosis mediated by the endothelial cells [28]. To enter the brain, C. neoformans must cross the BBB, and three major pathways have been proposed for crossing the BBB including Trojan horse, transcytosis, and paracellular crossing [29,30,31] (Figure 1).
Figure 1.
C. neoformans’ transmigration to the brain across the BBB. Three pathways have been proposed for the BBB crossing of C. neoformans. (A) Trojan horse. Phagocytes can carry ingested C. neoformans across the BBB through paracellular and transcellular migration. Phagocytes containing C. neoformans can also directly transfer the fungal cell to brain endothelial cells through lateral transfer, leading to BBB crossing of the organism. (B) Transcytosis. Interactions of cryptococcal hyaluronic acid with endothelial CD44 or receptor of hyaluronan-mediated motility (RHAMM) lead to endocytosis of the yeast cell. Following endocytosis, cryptococcal Mpr1 interacts with Annexin A2 (AnxA2) to facilitate exocytosis of the organism from the endothelial cells. In addition, cryptococcal phospholipase B1 (PLB1) promotes transcytosis through activation of host cell Rac1. (C) Paracellular crossing. Host plasminogen (white) binds to C. neoformans and is converted to the serine protease plasmin (red). Plasmin degrades the extracellular matrix (ECM), facilitating fungal crossing of the BBB. The tight junction of the BBB can be damaged by cryptococcal urease (due to the toxic effects of urease-induced ammonia) and other secreted cryptococcal proteases. C. neoformans can invade the brain across the damaged BBB.
2.1. Trojan Horse
C. neoformans is a facultative intracellular pathogen that can survive and grow inside phagocytes [32]. The expulsion of live C. neoformans (known as vomocytosis) by macrophages has been observed in vitro and in vivo following phagocytosis of the fungus [33,34,35]. In addition, C. neoformans can move directly from infected to uninfected macrophages [36,37]. These unique properties of C. neoformans support the hypothesis that phagocytes contribute to the spread and dissemination of C. neoformans from the infected lung to the CNS. Indeed, mononuclear phagocytes harboring C. neoformans have been detected in the perivascular space of the brain [38,39]. The deletion of alveolar macrophages or CD11c+ cells (alveolar macrophage and dendritic cells (DCs)) in the lungs significantly reduced the dissemination of C. neoformans to the lung lymph nodes and the brain [40,41], suggesting a role for these cells in extrapulmonary dissemination. In addition, the intravenous transfer of C. neoformans-infected macrophages to recipient mice enhanced brain colony-forming unit (CFU) levels [42,43], while the depletion of monocytes decreased brain CFU levels [39,42], providing further evidence that phagocytes are capable of transporting fungal cells to the CNS.
Using a monolayer of human cerebral microvascular endothelial cells (hCMEC/D3) cultured in vitro, monocytes infected with C. neoformans were directly seen to cross brain endothelial cells [44,45], demonstrating that these cells can facilitate neuroinvasion. Supporting this, intravital microscopy revealed that a substantial number of CX3CR1+Ly6Clow monocytes were recruited to the brain microvasculature 12 h after C. neoformans infection and were seen to engulf C. neoformans, adhere to the luminal wall of the brain microvasculature, and transmigrate to the parenchyma [46]. In contrast, CCR2+Ly6Chi monocytes were observed to accumulate in the brain starting 14 days post intravenous C. neoformans infection, and instead drive brain inflammation in mice as well as humans [47,48].
Further support of the Trojan horse mechanism of brain invasion by C. neoformans comes from clinical studies demonstrating that the efficient internalization of C. neoformans by phagocytes positively correlated with CSF fungal burdens and the risk of death in HIV-associated cryptococcosis patients [49]. Moreover, monocyte-derived macrophages from HIV/AIDS patients displayed a higher phagocytosis efficiency of C. neoformans along with more intracellular fungal growth compared to those of health individuals [50]. With the use of miRNA transcriptomics, it has been recently shown that the cytoskeleton and myocilin, encoded by MYOC, were involved in cryptococcal brain dissemination by modulating the Trojan horse pathway in mice and macaques, and provide a possible explanation for higher incidences of cryptococcal meningoencephalitis in HIV/AIDS patients, who often have dysfunctional immune cell cytoskeletons [50].
2.2. Transcytosis
In a murine model of C. neoformans infection, fungal cells were observed inside endothelial cells of the brain, suggesting that C. neoformans invades the brain through transcytosis [38]. In vitro studies using a monolayer of human brain microvascular endothelial cells (HBMECs) have shown that C. neoformans can adhere to endothelial cells and cross HBMEC monolayers via a transcellular pathway [21]. Studies have shown that the cryptococcal CPS1 gene encodes a hyaluronic acid synthase and that its product, hyaluronic acid, is critically involved in the adhesion of C. neoformans to brain endothelial cells [51]. It was further demonstrated that the adhesion of C. neoformans to HBMEC monolayers and the subsequent transcytosis was mediated by interactions between cryptococcal hyaluronic acids and CD44 expressed on HBMECs [52] and involved protein kinase C-alpha [53]. Moreover, C. neoformans enhanced the activity of EphA2, a tyrosine kinase receptor, through CD44, facilitating fungal migration into the brain [54]. In support of these in vitro observations, mice deficient in CD44 displayed lower brain fungal burdens following C. neoformans infection [55]. Despite this, CD44-deficient mice still had C. neoformans present in their brains, and in fact another hyaluronic acid receptor, RHAMM (receptor of hyaluronan-mediated motility), which is present in CD44-deficient mice, was also found to mediate the association of C. neoformans cells with mouse brain microvascular endothelial cells (BMECs) [55]. Interestingly, the brain has high levels of the sugar inositol, and inositol acquisition by C. neoformans leads to the upregulation of the cryptococcal CPS1 gene and the higher production of hyaluronic acids, thus promoting the adherence and transcytosis of C. neoformans, which may explain C. neoformans’ predilection for the brain [56].
Apart from hyaluronic acids, cryptococcal phospholipase B1 (PLB1) has been shown to promote the transcytosis of C. neoformans in cultured HBMECs through the activation of host cell Rac1 and its association with STAT3 [57]. In addition, a secreted metalloprotease (Mpr1) of C. neoformans was reported to be required for crossing hCMEC/D3 monolayers by interacting with cytoskeleton-endocytosis-associated protein Annexin A2; consequently, mice infected with a strain of the fungus lacking the gene encoding Mpr1 survived longer due to reduced brain fungal burdens [58,59]. Recently, an approach based on flow cytometry to quantitatively analyze fungal migration into the brain has been established, and using this approach, it has been confirmed that the internalization of C. neoformans by brain endothelial cells occurs in vivo [60]. Interestingly, quantitative analysis revealed that the brain endothelial cells were invaded by C. neoformans at a higher rate compared to C. deneoformans, which may reflect the higher virulence of C. neoformans during brain infection in mice [60].
2.3. Paracellular Crossing
In addition to the Trojan horse and transcytosis pathways, it has been proposed that C. neoformans can migrate into the brain through paracellular crossing between brain endothelial cells and across damaged endothelial barriers [30,31]. Early studies have shown that there was vessel damage and leakage at fungal arrest sites [20]. In addition, there is evidence that C. neoformans induces alterations of the tight junction and the cytoskeleton of endothelial cells and ultimately induces endothelial cell necrosis [61,62,63]. In this context, the cryptococcal capsule and secreted enzymes may contribute to the damage of tight junction and endothelial cells [31]. For example, cryptococcal urease is a major virulence factor and promotes fungal invasion into the CNS [29,64,65,66], likely by damaging tight junctions due to the toxic effects of urease-induced ammonia [66]. C. neoformans has also been found to secrete a number of proteases, which have been shown to disrupt the BBB during brain infection [67,68]. In addition, C. neoformans is able to bind and activate host plasminogen, leading to the conversion of plasminogen to the serine protease plasmin, which is also capable of degrading the BBB [69,70].
3. Host Immune Responses to C. neoformans in the Brain
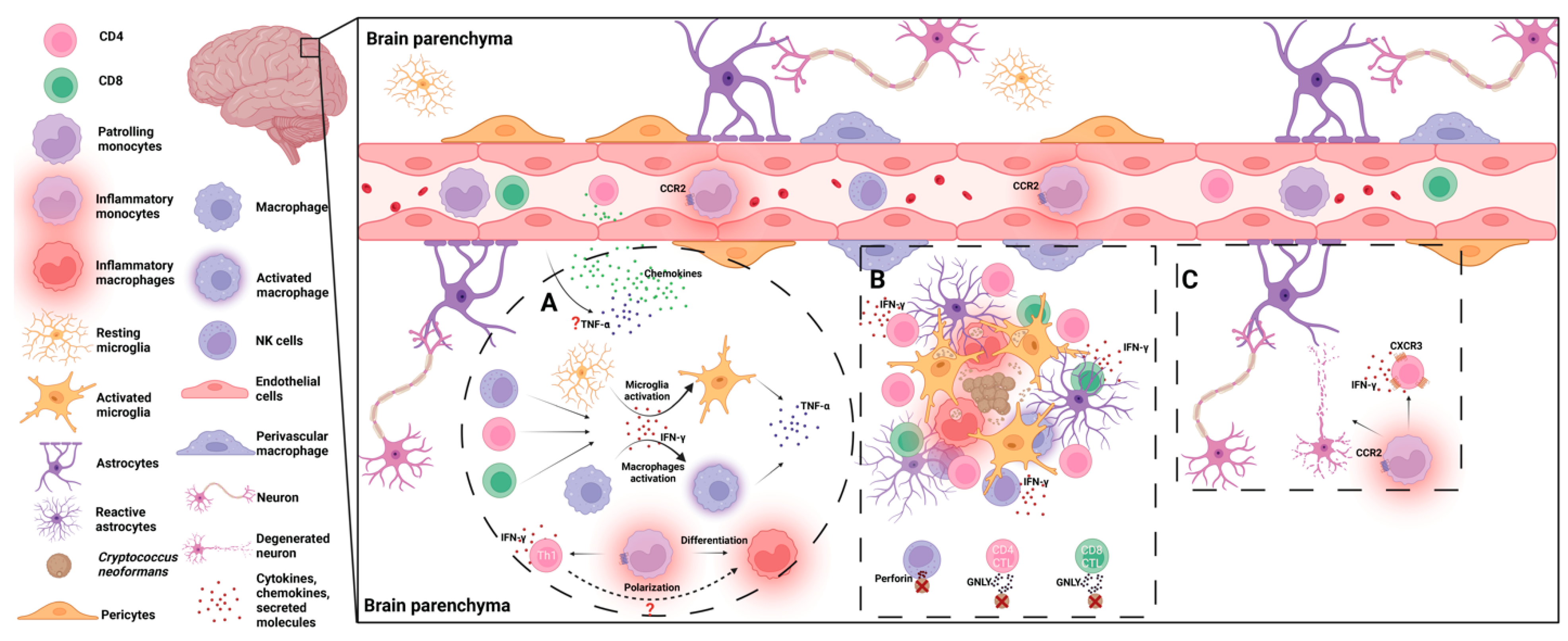
Following its invasion into the brain, C. neoformans cells begin to grow. C. neoformans will first encounter microglia, the macrophages residing in the brain. Interactions between microglia and invading C. neoformans lead to neuroinflammation including the production of proinflammatory cytokines and chemokines, which promotes the recruitment and accumulation of innate immune cells as well as adaptive immune cells. These immune cells, along with the cytokines secreted by them, are critically involved in fighting the fungal cells [9,13] (Figure 2).
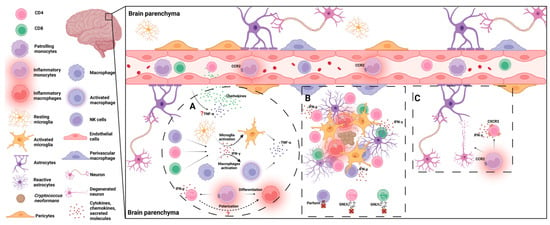
Figure 2.
Host immune responses to C. neoformans in the brain. (A) Following brain infection of C. neoformans, TNF-α and chemokines are produced, facilitating leukocytes’ migration to the brain parenchyma. IFN-γ produced by CD4+/CD8+ T cells and NK/NKT cells promotes the activation of microglia and macrophages. Activated microglia and macrophages secrete TNF-α, further driving brain inflammation. Recruited inflammatory monocytes differentiate into inflammatory macrophages and help shape Th1 immune responses, which are required for fungal clearance. (B) As the resident glial cells, microglia and astrocytes are the first cells to respond to invading yeast cells. Later, recruited leukocytes including CD4+/CD8+ T cells, NK/NKT cells, and inflammatory monocyte/macrophages accumulate in and around the fungal clusters. As effector cells, microglia and macrophages internalize the fungal cells. T cells and NK cells secrete IFN-γ to promote fungicidal activity of these phagocytes. NK cells and T cells are also involved in direct killing of the fungal cells through release of perforin or granulysin (GNLY). (C) Inflammatory responses are required for fungal clearance in the brain but must be tightly controlled. Inflammatory monocytes and IFN-γ-secreting CXCR3+ CD4+ T cells facilitate neuronal damage and cause immunopathology during cryptococcal meningoencephalitis.
3.1. Microglia
Tissue-resident macrophages are a diverse population of leukocytes that reside in mammalian tissues and play a prominent role in tissue homeostasis and host defense [71]. Tissue-resident macrophages are usually embryonic in origin and self-renewing under steady-state conditions [72,73]. As brain-resident macrophages, microglia are found throughout the brain parenchyma and play a central role in immune responses in the brain. During their resting state, microglia extend their dendrites to survey the brain [74]. Upon activation, microglia enlarge their cell bodies and adopt an “amoeboid” morphology with thicker ramifications and can engulf and clear dead neurons, pathogens, and pathogen-infected cells [75,76,77,78,79,80].
During a brain infection with the fungal pathogen Candida albicans, CARD9+ microglia mediate protection by promoting neutrophil recruitment in a IL-1β- and CXCL1-dependent manner [81]. Studies on the interactions between microglia and C. neoformans revealed that microglia can internalize C. neoformans in vitro [82,83,84,85,86] as well as in the human brain [87]. The internalization of nonopsonized C. neoformans by swine microglia was reported to involve the CD14 receptor [84]. The presence of capsule-binding antibodies was shown to enhance the phagocytosis of C. neoformans by human microglial cells [85], and in the presence of anti-cryptococcal antibodies, C. neoformans-stimulated microglia secreted proinflammatory chemokines MIP-1α and MIP-1β [88]. However, the cryptococcal capsule has been found to inhibit phagocytosis by murine microglial cells [86], as well as down-regulate the production of MIP-1α and MIP-1β [88].
The stimulation of microglia by TLR agonists also promotes phagocytosis and the killing of C. neoformans and is associated with enhanced secretions of proinflammatory cytokines including TNF-α [83]. In addition, the killing of C. neoformans by microglia was positively correlated with nitric oxide secretion [89,90]. Interestingly, the intracellular iron load of microglia, rather than the stimulation by IFN-γ, enhanced the anticryptococcal activity of microglia in vitro [91,92]. As a facultative intracellular pathogen, C. neoformans can also survive and proliferate within microglial cells depending on the activation status of the microglia [87,93].
Murine studies showed that MHC class II-positive perivascular microglial cells were involved in resistance to C. neoformans brain infection [94]. In line with this report, immunotherapy with a combined administration of anti-CD40 and IL-2 during a C. neoformans infection reduced fungal burdens in the brain, which correlated with an increase in MHC class II expression in microglia in an IFN-γ-dependent manner [95,96]. More recently, it has been shown that the number of microglial cells is significantly enhanced following the upregulation of MHC class II and CD11c in the cells during a brain infection with C. neoformans [97].
3.2. Inflammatory Monocytes/Macrophages
Monocytes consist of two major populations. These include CD14hiCD16− and CD14lowCD16hi monocytes in humans, and the corresponding CCR2hiCX3CR1lowLy6Chi inflammatory monocytes and CCR2lowCX3CR1hiLy6Clow monocytes in mice [98,99,100]. During infection and inflammation, Ly6Chi inflammatory monocytes migrate from the bone marrow to the periphery blood through CCR2 signaling and are recruited to the infected or inflamed tissues [98,101]. The recruited Ly6Chi inflammatory monocytes can differentiate to monocyte-derived Ly6ChiCD11b+ inflammatory macrophages and DCs [98]. These recruited Ly6Chi mononuclear phagocytes (i.e., Ly6Chi monocytes and their derivatives) secrete high levels of proinflammatory cytokines, promote inflammation, and act as effector cells and play an essential role in the clearance of pathogens [98].
As the signaling of CCR2 with its ligands, including CCL2 and CCL7, is required for the emigration of Ly6Chi monocytes from the bone marrow, CCR2−/− mice have been widely used to study the functions of Ly6Chi inflammatory monocytes and macrophages [98,101]. Previous studies have shown that Ly6Chi inflammatory monocytes and macrophages display a prominent role in the clearance of various fungal pathogens in the lungs, including Aspergillus fumigatus [102,103], Candida albicans [104], Histoplasma capsulatum [105,106], and Blastomyces dermatitidis [105].
During a pulmonary infection with C. neoformans, the disruption of CCR2 signaling led to higher fungal burdens and coincided with the development of detrimental Th2 responses in the infected lung [107,108]. CCR2 signaling promoted Ly6Chi inflammatory monocyte migration to the infected lung, leading to the differentiation and accumulation of Ly6Chi inflammatory macrophages and DCs [109,110,111]. These effector cells secrete nitric oxide and TNF-α and have been shown to kill C. neoformans in the lung [109]. Moreover, recent studies showed that Ly6Chi inflammatory monocytes and macrophages are critically involved in the protection generated by candidate vaccine strains of C. neoformans [112,113,114,115]. However, the fungicidal activity of Ly6Chi inflammatory monocytes and macrophages must be tightly controlled to avoid tissue destruction [98]. In this regard, more recent studies have shown that an enhanced accumulation of Ly6Chi inflammatory monocytes and macrophages mediated detrimental immune responses in the lung in a murine model of an acute infection with C. neoformans [116,117].
CCL2 is expressed by microglia, astrocytes, and endothelial cells in the brain under physiological conditions [118]. An internal crosstalk between inflammatory monocytes with microglia was observed during West Nile Virus-induced encephalitis [119], although whether it occurs during cryptococcal meningoencephalitis remains unknown. Clinical studies have shown that inflammatory monocytes are recruited to the CNS of human patients during cryptococcal meningitis [47,48]. The impaired recruitment of leukocytes to the brain in MIP-1α knockout mice correlated with an impaired clearance of C. neoformans in the brain [120], suggesting that the accumulation of leukocytes including inflammatory monocytes in the brain is required for fungal clearance. However, the brain’s inflammatory responses mediated by inflammatory monocytes/macrophages and other leukocytes must be tightly controlled to avoid neuropathology during brain infection with C. neoformans [97,121].
3.3. NK/NKT Cells
Natural killer cells (NK cells) are innate immune cells and are well known to have the capability of killing virus-infected cells and tumor cells. There are limited NK cells in the brain parenchyma under steady-state conditions, but NK cells have been shown to be recruited in a CX3CR1-dependent manner to glioblastomas, including both common and aggressive brain tumors. Both resident and recruited NK cells are activated by the cytokines IL-2, IL-15, and IL-19 to produce TNF-α and IFN-γ, as well as perforin and granzymes, which are able to lyse target cells [122]. Early studies have shown that NK cells can bind to C. neoformans and directly kill the fungus [123,124,125,126,127]. It was later found that NK cells use perforin but not granulysin to kill C. neoformans [128] and that this requires PI3K-dependent ERK1/2 signaling [129]. It was further demonstrated that β-1,3-glucan, a component of the fungal cell wall, binds to NKp30 expressed on NK cells, thereby mediating the recognition and killing of C. neoformans [130,131]. In addition to the direct killing of C. neoformans, NK cells can secrete IFN-γ upon stimulation with IL-12 and IL-18 to activate phagocytes such as macrophages, leading to the indirect killing of C. neoformans [132,133,134].
NKT cells are a unique subset of T lymphocytes and can be identified by their expression of both T cell receptors (TCR) along with NK cell lineage receptors. Invariant NKT (iNKT) cells reside in the brain parenchyma during homeostasis [135]. While iNKT cells are CD1d-restricted T cells, both the recruitment and activation of peripheral NKT cells to the CNS are CD1d-independent [136]. iNKT cells accumulate in the lung in an MCP-1-dependent manner during a pulmonary cryptococcal infection and contribute to the development of protective Th1 immune responses [137]. It was reported that α-Galactosylceramide-activated NKT cells enhanced their secretions of IFN-γ in the absence of IL-18 signaling during systematic cryptococcal infection [138]. Aged C57BL/6 mice with enhanced mature NKT cells displayed stronger fungal resistance in the lungs, indicating that NKT cells play a role in mediating protection [139]. However, their role during cryptococcal CNS infection has not yet been elucidated.
3.4. CD4/8+ T Cells
Following antigen presentation, naïve T cells proliferate and differentiate to antigen-specific T cells, which migrate to infected tissues. Cryptococcal meningoencephalitis primarily affects immunocompromised hosts such as HIV/AIDS patients whose CD4+ T cell responses are impaired [1,140], demonstrating the essential role of T cells in host defense against C. neoformans infection.
During a pulmonary infection with C. neoformans, both CD4+ and CD8+ T cells were substantially recruited to the lung, and the depletion of either CD4+ or CD8+ T cells prevented pulmonary clearance and resulted in a significant colonization of the brain in mice, demonstrating that both CD4+ and CD8+ T cells are required to clear C. neoformans infection [141,142,143,144]. The depletion of either CD4+ or CD8+ T cells markedly reduced the influx of myeloid cells, including monocytes and neutrophils, to the infected lung [142]. Interestingly, the depletion of CD4+ T cells did not affect the influx of CD8+ T cells to the infected lung [141,142,143] and CD8+ T cells functioned independently of CD4+ T cells to control fungal growth by limiting the survival of C. neoformans within macrophages through IFN-γ production [142,145].
During a brain infection with C. neoformans, a substantial accumulation of CD4+ T cells, and to a lesser extent CD8+ T cells, was observed in the brain at weeks 3 and 4 post-infection [97]. Following a cryptococcal infection, chemokines such as CCL2, CXCL1, CCL5, and CXCR3 ligands (CXCL9, 10, and 11) were secreted by astrocytes and T cells were recruited into the brain in a CXCR3-dependent manner [146]. The in vivo depletion of CD4+ T cells markedly reduced leukocyte accumulation in the brains of immunized mice and was associated with an exacerbated CNS infection [147]. Another study showed that CD4+ T cells were important for the optimal infiltration of inflammatory cells into the brain and were required for optimal regional IFN-γ secretion and iNOS expression in the C. neoformans-infected brains of immunized mice [148]. However, recent studies have shown that the depletion of CD4+ T cells prevented brain immunopathology and significantly enhanced the survival of mice despite enhanced fungal growth during a brain infection with C. neoformans [97]. It was further demonstrated that CXCR3+ CD4+ T cells mediated lethal brain pathology without contributing to fungal clearance in the brain during cryptococcal meningoencephalitis [146], emphasizing the importance of immune balance in the CNS.
In addition to regulating immune responses, T cells have been shown to bind to C. neoformans and directly kill the fungus. Human peripheral CD8+ T cells were found to use granulysin to kill C. neoformans in vitro, and this fungicidal activity was dependent on CD4+ T cells and IL-15 [149]. Likewise, cytotoxic CD4+ T cells mediated the killing of C. neoformans using granulysin and the activation of this pathway was defective in HIV+ patients [150]. Moreover, the expression of granulysin by cytotoxic CD4+ T cells required the PI3K- and STAT5-dependent expression of IL-2Rβ, which is also defective in HIV-infected patients [151].
3.5. Proinflammatory Cytokines
Cytokines play critical roles in regulating immune responses to C. neoformans [9]. Th1 immune responses, characterized by IFN-γ secretion and the classical activation of macrophages, are required to control C. neoformans infection [112,152,153,154]. In contrast, Th2 responses, characterized by IL-4, IL-5, and IL-13 production, with alternative macrophage activation worsen the disease [155,156,157,158]. As a signature cytokine of Th1 responses, IFN-γ plays a central role in combating C. neoformans infection. Early studies showed that IFN-γ activation of macrophages was critical for anticryptococcal activity [159,160,161]. During a C. neoformans pulmonary infection, IFN-γR−/− mice displayed significantly higher fungal burdens in the lungs and were markedly more susceptible to C. neoformans infection than wild-type mice [152,162]. Interestingly, the infection of mice with a murine gamma interferon-producing C. neoformans strain completely protected the mice from infection with wild-type C. neoformans [163].
During a brain infection with C. neoformans, a substantial amount of IFN-γ was detected in the brains of mice [97,164]. The in vivo neutralization of IFN-γ exacerbated a cryptococcal CNS infection [147,165]. During an intracranial infection of C. neoformans, IFN-γ was shown to mediate protection by activating microglial cells in the brains of mice [96]. Human astrocytes activated by IFN-γ and IL-1β in vitro inhibited cryptococcal growth by a nitric oxide-mediated mechanism [166]. In clinical settings, higher secretions of IFN-γ by CD4+ T cells were associated with an improved survival of patients [167] and the addition of IFN-γ to standard treatments significantly enhanced the rate of clearance of C. neoformans infection from the CSF during cryptococcal meningoencephalitis [168,169].
Th17 responses, characterized by IL-17A, also contribute to anti-cryptococcal immunity [9]. The development of Th1/Th17 responses and the classical activation of macrophages resulted in a significant containment of C. neoformans cells in the lungs of mice [170]. Using IL-17A−/− mice, it was demonstrated that IL-17A mediated protection through the promotion of leukocyte recruitment, activation, and IFN-γ secretions [171]. It was recently reported that a type I IFN induction via poly-ICLC protected mice against cryptococcosis and that the protective effect was diminished by the neutralization of IL-17A, demonstrating the protective function of IL-17A [172].
TNF-α is another cytokine that mediates protection during cryptococcosis [9]. Neutralization of TNF-α reduced leukocyte influx to the lung and resulted in higher fungal burdens during a pulmonary infection of C. neoformans [173]; TNF-α was required for the induction of IL-12 and IFN-γ, which mediated protection during a pulmonary cryptococcal infection [174]. Moreover, TNF-α mediated protection in a lung infected with C. neoformans by inducing DC1 polarization and the initial Th1/Th17 responses at the early stages of infection [175]. TNF-α expression by an engineered C. neoformans strain led to protective immune responses during pulmonary cryptococcosis [176]. The enhanced secretion of TNF-α was detected in the brain during a C. neoformans infection [164]. During a brain infection with C. neoformans, the neutralization of TNF-α led to a marked increase of brain fungal burdens, indicative of the anti-fungal activity of TNF-α in cryptococcal meningoencephalitis [165]. However, the mechanism of the protective role of TNF-α during a cryptococcal CNS infection has not been elucidated.
4. Fungal Virulence Factors as Immune Modulators
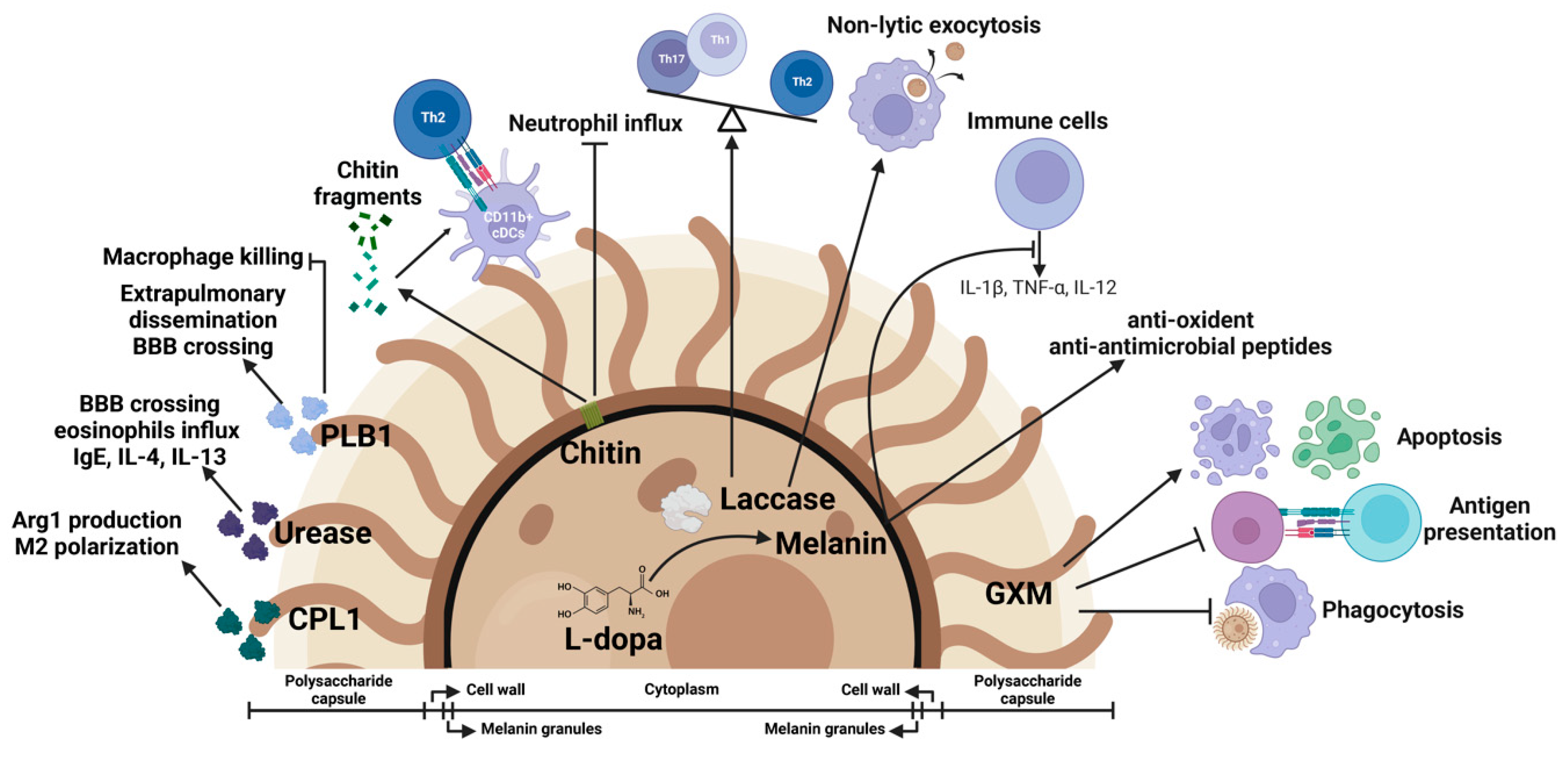
To fight against host immune defense, C. neoformans develops immune evasion strategies. The major virulence factors of C. neoformans include its polysaccharide capsule, melanin production, and the secretion of extracellular enzymes [18]. These virulence factors not only contribute to cryptococcal pathogenesis but are also involved in the modulation of host immune responses [177] (Figure 3).
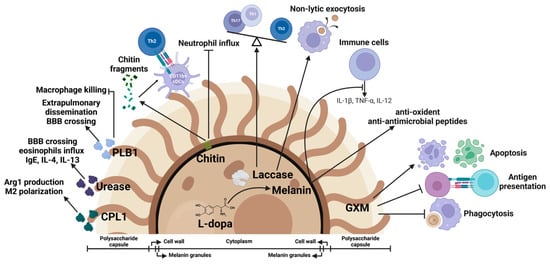
Figure 3.
Cryptococcal virulence factors as immune modulators. Fungal virulence factors mediate cryptococcal pathogenesis and function as host immune modulators. Glucuronoxylomannan (GXM) from the polysaccharide capsule inhibits phagocytosis of C. neoformans by macrophages, suppresses antigen presentation, and induces apoptosis of macrophages and T cells. Melanin protects the yeast cells from oxidants and antimicrobial peptides. Melanization suppresses the production of cytokines such as IL-1β, TNF-α, and IL-12. Laccase, an enzyme required for biosynthesis of melanin, is involved in regulation of nonlytic exocytosis of C. neoformans from macrophages and contributes to the skewing of Th1/Th17 to Th2 immune responses. Chitin fragments promote Th2 responses and inhibit neutrophil influx. PLB1 promotes fungal dissemination and inhibits macrophage killing. Urease facilitates fungal brain invasion and induces Th2 responses. Secreted protein CPL1 promotes arginase-1 production and shapes the M2 polarization.
4.1. Polysaccharide Capsule
One of the major virulence factors for C. neoformans is the polysaccharide capsule [18]. The capsule is located outside of the fungal cell wall and protects the fungus from immune recognition and destruction. Studies have shown that the capsule is mainly composed of two types of polysaccharides and that the dominant polysaccharide (>90% of the total capsule polysaccharides) is glucuronoxylomannan (GXM); another polysaccharide, known as galactoxylomannan (GalXM), represents the other 5-8% of the capsule along with nonpolysaccharide components such as mannoprotein [15,177,178]. The capsular polysaccharide is synthesized intracellularly and is believed to be translocated to the extracellular space within vesicles [178].
The cryptococcal capsule modulates host immune responses in multiple ways [15,177]. The capsule prevents the phagocytosis of the fungus by macrophages [32,179]. GXM is known to inhibit the antigen-presenting capacity of human monocytes and monocyte-derived macrophages through the induction of IL-10 secretion and the suppression of MHC-II expression, resulting in impaired T cell proliferation [180,181,182]. GXM also induces the apoptosis of macrophages and T cells through the induction of Fas ligand expression [183,184] or the induction of iNOS expression and NO production in a caspase-independent pathway [185]. GXM also inhibits DC activation and maturation [186,187]. Of note, the regulated release of capsular GXM of C. neoformans suppresses leukocyte migration to the brain, promoting fungal growth in the brain [188]. In clinical settings, capsule shedding (exocellular GXM) can be detected in the serum and CSF of patients during cryptococcal meningitis [189,190]. There is a positive correlation between serum GXM titers and mortality during HIV-associated cryptococcal meningitis [191]. Moreover, an increased size of the capsule correlated with impaired fungal clearance and reduced CSF leukocytes and proinflammatory cytokines including IL-6 and IFN-γ, confirming the immunosuppressive properties of the capsule in HIV-associated cryptococcal meningitis [189].
4.2. Laccase Activity and Melanin Formation
In addition to the capsule, laccase and melanin are known major virulence factors of C. neoformans [18,192,193,194,195,196]. Laccase is required for the biosynthesis of melanin [197], which accumulates in the C. neoformans cell wall [18]. The published work suggested that laccase facilitated the extrapulmonary dissemination of C. neoformans [195]. This is likely, at least in part, because laccase activity protected C. neoformans from being killed by alveolar macrophages [193]. Moreover, laccase activity enhanced pulmonary eosinophilia and shifted immune polarization from protective Th1/Th17 and M1 responses to deleterious Th2 and M2 responses, promoting fungal growth during a pulmonary infection with C. neoformans [196]. Recently, it has been shown that laccase activity dampened Th17 responses and neutrophil accumulation and function during the early stages of an infection with Cryptococcus gattii (C. gattii) [198]. Interestingly, laccase is also critically involved in the regulation of the nonlytic exocytosis of C. neoformans from macrophages [199]. In HIV-associated cryptococcosis patients, cryptococcal laccase activity correlated with fungal survival and poor fungal clearance in CSFs [49], arguably through the regulation of the interaction between C. neoformans and phagocytes. Thus, cryptococcal laccase modulates host defenses through multiple mechanisms involving innate and adaptive immunity.
Located in the cell wall, cryptococcal melanin is a powerful antioxidant and helps protect fungal cells against oxygen- and nitrogen-derived oxidants generated by host effector cells [17,200,201,202]. Early studies have shown that the melanization of C. neoformans correlated with higher levels of IL-4 and MCP-1 and enhanced leukocyte recruitment and interfered with phagocytosis, which is indicative of immunomodulation [203]. Due to the presence of melanin, C. neoformans is less susceptible to the cationic antimicrobial peptides released by phagocytes [204]. Solubilized melanin has been recently shown to inhibit macrophage functions [205]. In addition to modulating innate immunity, melanin inhibited antigen recognition and down-regulated T cell immunity and inflammation during a C. neoformans pulmonary infection [206]. During a brain infection with C. neoformans, melanization suppressed the production of IL-12, IL-1β TNF-α, IFN-γ, and iNOS, promoting mortality [207].
4.3. Phospholipase B1 Activity
Phospholipases are extracellular enzymes that degrade cell membrane phospholipids [18]. Amongst them, phospholipase B1 (PLB1) has been well-characterized and is known as one of the major virulence factors of C. neoformans [18,43,208,209,210,211,212,213]. The depletion of PLB1 significantly reduced fungal burdens during a pulmonary infection with C. neoformans [211]. In addition, PLB1 is essential for the extrapulmonary dissemination of C. neoformans [212,213] and the transmigration of the fungus across the BBB [43,57]. Importantly, PLB1 is required for the release of arachidonic acid from phospholipids and the production of cryptococcal eicosanoids, which down-regulates macrophages’ functions in vitro and during a pulmonary infection [213]. In line with this finding, recent work suggested that PLB1 promoted the proliferation of C. neoformans within macrophages and inhibited the killing of the fungus within the phagosome [214].
4.4. Urease Activity
Urease is an extracellular enzyme and is considered a major virulence factor of C. neoformans [64]. C. neoformans secretes urease, which catalyzes the hydrolysis of urea to ammonia and carbamate [18]. The published work suggests that urease activity promotes cryptococcal brain invasion [29,65,66]. In addition, an infection with urease-producing C. neoformans correlated with enhanced eosinophil influx; higher levels of IgE, IL-4, and IL-13; and, alternatively, the activation of macrophages, suggesting that urease activity promotes Th2 immune responses [215].
4.5. Chitin
Chitin is an essential component of the cell wall of C. neoformans, and the fungus has eight putative chitin synthases [216]. Mammalian hosts possess chitotriosidase, an enzyme that can degrade chitin into chitin fragments [217,218]. Early studies showed that degraded chitin fragments could trigger human macrophage activation [219]. In a murine model of an infection with C. neoformans, the host’s chitotriosidase degraded fungal chitin into small fragments that led to Th2 differentiation by conventional CD11b+ DCs, demonstrating that chitin recognition via chitotriosidase promoted detrimental Th2 immune responses [116]. Of note, chitotriosidase activity correlated with cryptococcal infection in humans [116]. In contrast to the negative impact of chitin on immune responses, it has been recently reported that mice infected with C. neoformans depleted of chitin synthase 3 died significantly earlier due to an excessive neutrophil influx, demonstrating a beneficial role of chitin in preventing lethal immune responses to C. neoformans [220].
4.6. CPL1
CPL1 is a secreted cryptococcal protein encoded by CNAG_02797/CPL1 [221]. A recent study showed that CPL1 induced arginase-1 production in macrophages and enhanced macrophage sensitivity to IL-4 signaling through the activation of TLR4 signaling and the promotion of the phosphorylation of STAT3. As a result, CPL1 drove the alternative activation of macrophages and promoted harmful type 2 immunity during cryptococcosis [222].
5. Concluding Remarks
Although a cryptococcal infection starts in the lungs, cryptococcosis commonly presents as meningoencephalitis [3]. Cryptococcal meningoencephalitis is among the most devastating complications of HIV/AIDS patients and is a leading cause of mortality among HIV/AIDS patients [223,224]. One of the critical steps to causing the illness is fungal dissemination and invasion into the brain by crossing the BBB [31]. A fungal invasion leads to host defenses including innate and adaptive immunity involving resident microglial cells and recruited myeloid cells and T cells [13]. In response to host defense, C. neoformans uses multiple virulence factors to suppress host immune responses [177]. In the past decades, much progress has been achieved with respect to understanding the interaction between hosts and C. neoformans. However, many questions remain unanswered. For example, which pathway is dominant during a brain invasion by C. neoformans? How do microglial cells interact with recruited leukocytes in the brain during a C. neoformans infection? What is the strategy that the fungus uses to survive and replicate in the brain? By addressing these questions, we will have a better understanding of the complex interaction between the host and C. neoformans.
Author Contributions
Conceptualization, Y.C.; writing—original draft preparation, Y.C.; writing—review and editing, Y.C., Z.W.S., A.B.S., M.S.; funding acquisition, M.S. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by National Institutes of Health (NIH) grants to M.S. (AI131219 and AI131905). Figures included in this review were created with BioRender.com accessed on 6 September 2022.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- Mitchell, T.G.; Perfect, J.R. Cryptococcosis in the Era of AIDS—100 Years after the Discovery of Cryptococcus neoformans. Clin. Microbiol. Rev. 1995, 8, 515–548. [Google Scholar] [CrossRef]
- Gottfredsson, M.; Perfect, J.R. Fungal Meningitis. Semin. Neurol. 2000, 20, 307–322. [Google Scholar] [CrossRef] [PubMed]
- Kwon-Chung, K.J.; Sorrell, T.C.; Dromer, F.; Fung, E.; Levitz, S.M. Cryptococcosis: Clinical and Biological Aspects. Med. Mycol. 2000, 38, 205–213. [Google Scholar] [CrossRef]
- Casadevall, A.; Coelho, C.; Alanio, A. Mechanisms of Cryptococcus neoformans-Mediated Host Damage. Front. Immunol. 2018, 9, 855. [Google Scholar] [CrossRef] [PubMed]
- Perfect, J.R.; Dismukes, W.E.; Dromer, F.; Goldman, D.L.; Graybill, J.R.; Hamill, R.J.; Harrison, T.S.; Larsen, R.A.; Lortholary, O.; Nguyen, M.H.; et al. Clinical Practice Guidelines for the Management of Cryptococcal Disease: 2010 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2010, 50, 291–322. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, T. Understanding and Managing the Adverse Effects of Antiretroviral Therapy. Antivir. Res. 2010, 85, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Montessori, V.; Press, N.; Harris, M.; Akagi, L.; Montaner, J.S.G. Adverse Effects of Antiretroviral Therapy for HIV Infection. CMAJ 2004, 170, 229–238. [Google Scholar] [PubMed]
- Rajasingham, R.; Smith, R.M.; Park, B.J.; Jarvis, J.N.; Govender, N.P.; Chiller, T.M.; Denning, D.W.; Loyse, A.; Boulware, D.R. Global Burden of Disease of HIV-Associated Cryptococcal Meningitis: An Updated Analysis. Lancet Infect. Dis. 2017, 17, 873–881. [Google Scholar] [CrossRef]
- Leopold Wager, C.M.; Hole, C.R.; Wozniak, K.L.; Wormley, F.L. Cryptococcus and Phagocytes: Complex Interactions That Influence Disease Outcome. Front. Microbiol. 2016, 7, 105. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.S.; Drummond, R.A. The Diverse Roles of Monocytes in Cryptococcosis. J. Fungi 2020, 6, 111. [Google Scholar] [CrossRef] [PubMed]
- Voelz, K.; May, R.C. Cryptococcal Interactions with the Host Immune System. Eukaryot. Cell 2010, 9, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Olszewski, M.A.; Zhang, Y.; Huffnagle, G.B. Mechanisms of Cryptococcal Virulence and Persistence. Future Microbiol. 2010, 5, 1269–1288. [Google Scholar] [CrossRef]
- Mohamed, S.H.; Nyazika, T.K.; Ssebambulidde, K.; Lionakis, M.S.; Meya, D.B.; Drummond, R.A. Fungal CNS Infections in Africa: The Neuroimmunology of Cryptococcal Meningitis. Front. Immunol. 2022, 13, 804674. [Google Scholar] [CrossRef]
- Nelson, B.N.; Hawkins, A.N.; Wozniak, K.L. Pulmonary Macrophage and Dendritic Cell Responses to Cryptococcus neoformans. Front. Cell. Infect. Microbiol. 2020, 10, 37. [Google Scholar] [CrossRef]
- Doering, T.L. How Sweet It Is! Cell Wall Biogenesis and Polysaccharide Capsule Formation in Cryptococcus neoformans. Annu. Rev. Microbiol. 2009, 63, 223–247. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A.; Coelho, C.; Cordero, R.J.B.; Dragotakes, Q.; Jung, E.; Vij, R.; Wear, M.P. The Capsule of Cryptococcus neoformans. Virulence 2019, 10, 822–831. [Google Scholar] [CrossRef] [PubMed]
- Williamson, P.R. Laccase and Melanin in the Pathogenesis of Cryptococcus neoformans. Front. Biosci. 1997, 2, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Almeida, F.; Wolf, J.M.; Casadevall, A. Virulence-Associated Enzymes of Cryptococcus neoformans. Eukaryot. Cell 2015, 14, 1173–1185. [Google Scholar] [CrossRef] [PubMed]
- Dando, S.J.; Mackay-Sim, A.; Norton, R.; Currie, B.J.; St. John, J.A.; Ekberg, J.A.K.; Batzloff, M.; Ulett, G.C.; Beacham, I.R. Pathogens Penetrating the Central Nervous System: Infection Pathways and the Cellular and Molecular Mechanisms of Invasion. Clin. Microbiol. Rev. 2014, 27, 691–726. [Google Scholar] [CrossRef] [PubMed]
- Charlier, C.; Chrétien, F.; Baudrimont, M.; Mordelet, E.; Lortholary, O.; Dromer, F. Capsule Structure Changes Associated with Cryptococcus neoformans crossing of the Blood-Brain Barrier. Am. J. Pathol. 2005, 166, 421–432. [Google Scholar] [CrossRef]
- Chang, Y.C.; Stins, M.F.; McCaffery, M.J.; Miller, G.F.; Pare, D.R.; Dam, T.; Paul-Satyasee, M.; Kim, K.S.; Kwon-Chung, K.J. Cryptococcal Yeast Cells Invade the Central Nervous System via Transcellular Penetration of the Blood-Brain Barrier. Infect. Immun. 2004, 72, 4985–4995. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, K.P.; Pelargos, P.E.; Milton, C.K.; Peterson, J.E.G.; Bohnstedt, B. Cryptococcal Choroid Plexitis and Non-Communicating Hydrocephalus. Cureus 2020, 12, e8512. [Google Scholar] [CrossRef]
- Kumari, R.; Raval, M.; Dhun, A. Cryptococcal Choroid Plexitis: Rare Imaging Findings of Central Nervous System Cryptococcal Infection in an Immunocompetent Individual. Br. J. Radiol. 2010, 83, e14–e17. [Google Scholar] [CrossRef] [PubMed]
- Kovoor, J.M.E.; Mahadevan, A.; Narayan, J.P.; Govindappa, S.S.; Satishchandra, P.; Taly, A.V.; Shankar, S.K. Cryptococcal Choroid Plexitis as a Mass Lesion: MR Imaging and Histopathologic Correlation Case Report. Am. J. Neuroradiol. 2002, 23, 273–276. [Google Scholar]
- Hammoud, D.A.; Mahdi, E.; Panackal, A.A.; Wakim, P.; Sheikh, V.; Sereti, I.; Bielakova, B.; Bennett, J.E.; Williamson, P.R. Choroid Plexitis and Ependymitis by Magnetic Resonance Imaging Are Biomarkers of Neuronal Damage and Inflammation in HIV-Negative Cryptococcal Meningoencephalitis. Sci. Rep. 2017, 7, 9184. [Google Scholar] [CrossRef]
- Daneman, R.; Prat, A. The Blood–Brain Barrier. Cold Spring Harb. Perspect. Biol. 2015, 7, a020412. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Calaruso, P.; Mody, C.H. Real-Time in Vivo Imaging of Fungal Migration to the Central Nervous System. Cell. Microbiol. 2012, 14, 1819–1827. [Google Scholar] [CrossRef] [PubMed]
- Kolaczkowska, E.; Kubes, P. Neutrophil Recruitment and Function in Health and Inflammation. Nat. Rev. Immunol. 2013, 13, 159–175. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Li, S.S.; Zheng, C.; Jones, G.J.; Kim, K.S.; Zhou, H.; Kubes, P.; Mody, C.H. Real-Time Imaging of Trapping and Urease-Dependent Transmigration of Cryptococcus neoformans in Mouse Brain. J. Clin. Investig. 2010, 120, 1683–1693. [Google Scholar] [CrossRef] [PubMed]
- May, R.C.; Stone, N.R.H.; Wiesner, D.L.; Bicanic, T.; Nielsen, K. Cryptococcus: From Environmental Saprophyte to Global Pathogen. Nat. Rev. Microbiol. 2016, 14, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Strickland, A.B.; Shi, M. Mechanisms of Fungal Dissemination. Cell. Mol. Life Sci. 2021, 78, 3219–3238. [Google Scholar] [CrossRef]
- Feldmesser, M.; Kress, Y.; Novikoff, P.; Casadevall, A. Cryptococcus neoformans is a Facultative Intracellular Pathogen in Murine Pulmonary Infection. Infect. Immun. 2000, 68, 4225–4237. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, M.; Casadevall, A. Phagosome Extrusion and Host-Cell Survival after Cryptococcus neoformans Phagocytosis by Macrophages. Curr. Biol. 2006, 16, 2161–2165. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Croudace, J.E.; Lammas, D.A.; May, R.C. Expulsion of Live Pathogenic Yeast by Macrophages. Curr. Biol. 2006, 16, 2156–2160. [Google Scholar] [CrossRef]
- Nicola, A.M.; Robertson, E.J.; Albuquerque, P.; da Silveira Derengowski, L.; Casadevall, A. Nonlytic Exocytosis of Cryptococcus neoformans from Macrophages Occurs in Vivo and Is Influenced by Phagosomal PH. MBio 2011, 2, e00167-11. [Google Scholar] [CrossRef]
- Ma, H.; Croudace, J.E.; Lammas, D.A.; May, R.C. Direct Cell-to-Cell Spread of a Pathogenic Yeast. BMC Immunol. 2007, 8, 15. [Google Scholar] [CrossRef]
- Alvarez, M.; Casadevall, A. Cell-to-Cell Spread and Massive Vacuole Formation after Cryptococcus neoformans Infection of Murine Macrophages. BMC Immunol. 2007, 8, 16. [Google Scholar] [CrossRef]
- Chrétien, F.; Lortholary, O.; Kansau, I.; Neuville, S.; Gray, F.; Dromer, F. Pathogenesis of Cerebral Cryptococcus neoformans Infection after Fungemia. J. Infect. Dis. 2002, 186, 522–530. [Google Scholar] [CrossRef]
- Kaufman-Francis, K.; Djordjevic, J.T.; Juillard, P.G.; Lev, S.; Desmarini, D.; Grau, G.E.R.; Sorrell, T.C. The Early Innate Immune Response to, and Phagocyte-Dependent Entry of, Cryptococcus neoformans Map to the Perivascular Space of Cortical Post-Capillary Venules in Neurocryptococcosis. Am. J. Pathol. 2018, 188, 1653–1665. [Google Scholar] [CrossRef]
- Walsh, N.M.; Botts, M.R.; McDermott, A.J.; Ortiz, S.C.; Wüthrich, M.; Klein, B.; Hull, C.M. Infectious Particle Identity Determines Dissemination and Disease Outcome for the Inhaled Human Fungal Pathogen Cryptococcus. PLoS Pathog. 2019, 15, e1007777. [Google Scholar] [CrossRef]
- Kechichian, T.B.; Shea, J.; Del Poeta, M. Depletion of Alveolar Macrophages Decreases the Dissemination of a Glucosylceramide-Deficient Mutant of Cryptococcus neoformans in Immunodeficient Mice. Infect. Immun. 2007, 75, 4792–4798. [Google Scholar] [CrossRef] [PubMed]
- Charlier, C.; Nielsen, K.; Daou, S.; Brigitte, M.; Chretien, F.; Dromer, F. Evidence of a Role for Monocytes in Dissemination and Brain Invasion by Cryptococcus neoformans. Infect. Immun. 2009, 77, 120–127. [Google Scholar] [CrossRef]
- Santangelo, R.; Zoellner, H.; Sorrell, T.; Wilson, C.; Donald, C.; Djordjevic, J.; Shounan, Y.; Wright, L. Role of Extracellular Phospholipases and Mononuclear Phagocytes in Dissemination of Cryptococcosis in a Murine Model. Infect. Immun. 2004, 72, 2229–2239. [Google Scholar] [CrossRef]
- Santiago-Tirado, F.H.; Onken, M.D.; Cooper, J.A.; Klein, R.S.; Doering, T.L. Trojan Horse Transit Contributes to Blood-Brain Barrier Crossing of a Eukaryotic Pathogen. MBio 2017, 8, e02183-16. [Google Scholar] [CrossRef]
- Sorrell, T.C.; Juillard, P.G.; Djordjevic, J.T.; Kaufman-Francis, K.; Dietmann, A.; Milonig, A.; Combes, V.; Grau, G.E.R. Cryptococcal Transmigration across a Model Brain Blood-Barrier: Evidence of the Trojan Horse Mechanism and Differences between Cryptococcus neoformans var. grubii Strain H99 and Cryptococcus gattii Strain R265. Microbes Infect. 2016, 18, 57–67. [Google Scholar] [CrossRef]
- Sun, D.; Zhang, M.; Sun, P.; Liu, G.; Strickland, A.B.; Chen, Y.; Fu, Y.; Yosri, M.; Shi, M. VCAM1/VLA4 Interaction Mediates Ly6Clow Monocyte Recruitment to the Brain in a TNFR Signaling Dependent Manner during Fungal Infection. PLoS Pathog. 2020, 16, e1008361. [Google Scholar] [CrossRef] [PubMed]
- Panackal, A.A.; Wuest, S.C.; Lin, Y.C.; Wu, T.; Zhang, N.; Kosa, P.; Komori, M.; Blake, A.; Browne, S.K.; Rosen, L.B.; et al. Paradoxical Immune Responses in Non-HIV Cryptococcal Meningitis. PLoS Pathog. 2015, 11, e1004884. [Google Scholar] [CrossRef]
- Jarvis, J.N.; Meintjes, G.; Bicanic, T.; Buffa, V.; Hogan, L.; Mo, S.; Tomlinson, G.; Kropf, P.; Noursadeghi, M.; Harrison, T.S. Cerebrospinal Fluid Cytokine Profiles Predict Risk of Early Mortality and Immune Reconstitution Inflammatory Syndrome in HIV-Associated Cryptococcal Meningitis. PLoS Pathog. 2015, 11, e1004754. [Google Scholar] [CrossRef]
- Sabiiti, W.; Robertson, E.; Beale, M.A.; Johnston, S.A.; Brouwer, A.E.; Loyse, A.; Jarvis, J.N.; Gilbert, A.S.; Fisher, M.C.; Harrison, T.S.; et al. Efficient Phagocytosis and Laccase Activity Affect the Outcome of HIV-Associated Cryptococcosis. J Clin. Investig. 2014, 124, 2000–2008. [Google Scholar] [CrossRef]
- Li, H.; Han, X.; Du, W.; Meng, Y.; Li, Y.; Sun, T.; Liang, Q.; Li, C.; Suo, C.; Gao, X.; et al. Comparative miRNA Transcriptomics of Macaques and Mice Reveals MYOC Is an Inhibitor for Cryptococcus neoformans Invasion into the Brain. Emerg. Microbes Infect. 2022, 11, 1572–1585. [Google Scholar] [CrossRef]
- Jong, A.; Wu, C.H.; Chen, H.M.; Luo, F.; Kwon-Chung, K.J.; Chang, Y.C.; LaMunyon, C.W.; Plaas, A.; Huang, S.H. Identification and Characterization of CPS1 as a Hyaluronic Acid Synthase Contributing to the Pathogenesis of Cryptococcus neoformans Infection. Eukaryot. Cell 2007, 6, 1486–1496. [Google Scholar] [CrossRef] [PubMed]
- Jong, A.; Wu, C.H.; Shackleford, G.M.; Kwon-Chung, K.J.; Chang, Y.C.; Chen, H.M.; Ouyang, Y.; Huang, S.H. Involvement of Human CD44 during Cryptococcus neoformans Infection of Brain Microvascular Endothelial Cells. Cell. Microbiol. 2008, 10, 1313–1326. [Google Scholar] [CrossRef]
- Jong, A.; Wu, C.H.; Prasadarao, N.V.; Kwon-Chung, K.J.; Chang, Y.C.; Ouyang, Y.; Shackleford, G.M.; Huang, S.H. Invasion of Cryptococcus neoformans into Human Brain Microvascular Endothelial Cells Requires Protein Kinase C-αActivation. Cell. Microbiol. 2008, 10, 1854–1865. [Google Scholar] [CrossRef] [PubMed]
- Aaron, P.A.; Jamklang, M.; Uhrig, J.P.; Gelli, A. The Blood–Brain Barrier Internalises Cryptococcus neoformans via the EphA2-Tyrosine Kinase Receptor. Cell. Microbiol. 2018, 20, e12811. [Google Scholar] [CrossRef] [PubMed]
- Jong, A.; Wu, C.H.; Gonzales-Gomez, I.; Kwon-Chung, K.J.; Chang, Y.C.; Tseng, H.K.; Cho, W.L.; Huang, S.H. Hyaluronic Acid Receptor CD44 Deficiency Is Associated with Decreased Cryptococcus neoformans Brain Infection. J. Biol. Chem. 2012, 287, 15298–15306. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.B.; Kim, J.C.; Wang, Y.; Toffaletti, D.L.; Eugenin, E.; Perfect, J.R.; Kim, K.J.; Xue, C. Brain Inositol Is a Novel Stimulator for Promoting Cryptococcus Penetration of the Blood-Brain Barrier. PLoS Pathog. 2013, 9, e1003247. [Google Scholar] [CrossRef] [PubMed]
- Maruvada, R.; Zhu, L.; Pearce, D.; Zheng, Y.; Perfect, J.; Kwon-Chung, K.J.; Kim, K.S. Cryptococcus neoformans Phospholipase B1 Activates Host Cell Rac1 for Traversal across the Blood-Brain Barrier. Cell. Microbiol. 2012, 14, 1544–1553. [Google Scholar] [CrossRef]
- Vu, K.; Tham, R.; Uhrig, J.P.; Thompson, G.R.; Na Pombejra, S.; Jamklang, M.; Bautos, J.M.; Gelli, A. Invasion of the Central Nervous System by Cryptococcus neoformans Requires a Secreted Fungal Metalloprotease. MBio 2014, 5, e01101-14. [Google Scholar] [CrossRef]
- Na Pombejra, S.; Salemi, M.; Phinney, B.S.; Gelli, A. The Metalloprotease, Mpr1, Engages AnnexinA2 to Promote the Transcytosis of Fungal Cells across the Blood-Brain Barrier. Front. Cell. Infect. Microbiol. 2017, 7, 296. [Google Scholar] [CrossRef]
- Chen, Y.; Li, C.; Sun, D.; Strickland, A.B.; Liu, G.; Shi, M. Quantitative Analysis Reveals Internalisation of Cryptococcus neoformans by Brain Endothelial Cells in Vivo. Cell. Microbiol. 2021, 23, e13330. [Google Scholar] [CrossRef]
- Vu, K.; Eigenheer, R.A.; Phinney, B.S.; Gelli, A. Cryptococcus neoformans Promotes Its Transmigration into the Central Nervous System by Inducing Molecular and Cellular Changes in Brain Endothelial Cells. Infect. Immun. 2013, 81, 3139–3147. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.H.M.; Stins, M.F.; Huang, S.H.; Chen, Y.H.; Kwon-Chung, K.J.; Chang, Y.; Kim, K.S.; Suzuki, K.; Jong, A.Y. Cryptococcus neoformans Induces Alterations in the Cytoskeleton of Human Brain Microvascular Endothelial Cells. J. Med. Microbiol. 2003, 52, 961–970. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.S.; Filler, S.G.; Alcouloumre, M.S.; Kozel, T.R.; Edwards, J.E.; Ghannoum, M.A. Adherence to and Damage of Endothelial Cells by Cryptococcus neoformans in Vitro: Role of the Capsule. Infect. Immun. 1995, 63, 4368–4374. [Google Scholar] [CrossRef]
- Cox, G.M.; Mukherjee, J.; Cole, G.T.; Casadevall, A.; Perfect, J.R. Urease as a Virulence Factor in Experimental Cryptococcosis. Infect. Immun. 2000, 68, 443–448. [Google Scholar] [CrossRef]
- Olszewski, M.A.; Noverr, M.C.; Chen, G.H.; Toews, G.B.; Cox, G.M.; Perfect, J.R.; Huffnagle, G.B. Urease Expression by Cryptococcus neoformans Promotes Microvascular Sequestration, Thereby Enhancing Central Nervous System Invasion. Am. J. Pathol. 2004, 164, 1761–1771. [Google Scholar] [CrossRef]
- Singh, A.; Panting, R.J.; Varma, A.; Saijo, T.; Waldron, K.J.; Jong, A.; Ngamskulrungroj, P.; Chang, Y.C.; Rutherford, J.C.; Kwon-Chung, K.J. Factors Required for Activation of Urease as a Virulence Determinant in Cryptococcus neoformans. MBio 2013, 4, e00220-13. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.Y.; Zhu, H.M.; Wu, J.H.; Wen, H.; Liu, C.J. Increased Permeability of Blood-Brain Barrier Is Mediated by Serine Protease during Cryptococcus Meningitis. J. Int. Med. Res. 2014, 42, 85–92. [Google Scholar] [CrossRef]
- Rodrigues, M.L.; Dos Reis, F.C.G.; Puccia, R.; Travassos, L.R.; Alviano, C.S. Cleavage of Human Fibronectin and Other Basement Membrane-Associated Proteins by a Cryptococcus neoformans Serine Proteinase. Microb. Pathog. 2003, 34, 65–71. [Google Scholar] [CrossRef]
- Stie, J.; Fox, D. Blood-Brain Barrier Invasion by Cryptococcus neoformans Is Enhanced by Functional Interactions with Plasmin. Microbiology 2012, 158, 240–258. [Google Scholar] [CrossRef]
- Stie, J.; Bruni, G.; Fox, D. Surface-Associated Plasminogen Binding of Cryptococcus neoformans Promotes Extracellular Matrix Invasion. PLoS ONE 2009, 4, e5780. [Google Scholar] [CrossRef]
- Ginhoux, F.; Guilliams, M. Tissue-Resident Macrophage Ontogeny and Homeostasis. Immunity 2016, 44, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Gomez Perdiguero, E.; Klapproth, K.; Schulz, C.; Busch, K.; Azzoni, E.; Crozet, L.; Garner, H.; Trouillet, C.; De Bruijn, M.F.; Geissmann, F.; et al. Tissue-Resident Macrophages Originate from Yolk-Sac-Derived Erythro-Myeloid Progenitors. Nature 2015, 518, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Guilliams, M.; Thierry, G.R.; Bonnardel, J.; Bajenoff, M. Establishment and Maintenance of the Macrophage Niche. Immunity 2020, 52, 434–451. [Google Scholar] [CrossRef]
- Nimmerjahn, A.; Kirchhoff, F.; Helmchen, F. Resting Microglial Cells Are Highly Dynamic Surveillants of Brain Parenchyma in Vivo. Science 2005, 308, 1314–1318. [Google Scholar] [CrossRef] [PubMed]
- Kreutzberg, G.W. Microglia: A Sensor for Pathological Events in the CNS. Trends Neurosci. 1996, 19, 312–318. [Google Scholar] [CrossRef]
- Kettenmann, H.; Hanisch, U.K.; Noda, M.; Verkhratsky, A. Physiology of Microglia. Physiol. Rev. 2011, 91, 461–553. [Google Scholar] [CrossRef]
- Nau, R.; Ribes, S.; Djukic, M.; Eiffert, H. Strategies to Increase the Activity of Microglia as Efficient Protectors of the Brain against Infections. Front. Cell. Neurosci. 2014, 8, 138. [Google Scholar] [CrossRef]
- Chhatbar, C.; Prinz, M. The Roles of Microglia in Viral Encephalitis: From Sensome to Therapeutic Targeting. Cell. Mol. Immunol. 2021, 18, 250–258. [Google Scholar] [CrossRef]
- Rock, R.B.; Gekker, G.; Hu, S.; Sheng, W.S.; Cheeran, M.; Lokensgard, J.R.; Peterson, P.K. Role of Microglia in Central Nervous System Infections. Clin. Microbiol. Rev. 2004, 17, 942. [Google Scholar] [CrossRef] [PubMed]
- Waltl, I.; Kalinke, U. Beneficial and Detrimental Functions of Microglia during Viral Encephalitis. Trends Neurosci. 2022, 45, 158–170. [Google Scholar] [CrossRef]
- Drummond, R.A.; Swamydas, M.; Oikonomou, V.; Zhai, B.; Dambuza, I.M.; Schaefer, B.C.; Bohrer, A.C.; Mayer-Barber, K.D.; Lira, S.A.; Iwakura, Y.; et al. CARD9+ Microglia Promote Antifungal Immunity via IL-1β- and CXCL1-Mediated Neutrophil Recruitment. Nat. Immunol. 2019, 20, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Tanaka, S.; Cox, D.; Lee, S.C. Fcγ Receptor Signaling in Primary Human Microglia: Differential Roles of PI-3K and Ras/ERK MAPK Pathways in Phagocytosis and Chemokine Induction. J. Leukoc. Biol. 2004, 75, 1147–1155. [Google Scholar] [CrossRef] [PubMed]
- Redlich, S.; Ribes, S.; Schütze, S.; Eiffert, H.; Nau, R. Toll-Like Receptor Stimulation Increases Phagocytosis of Cryptococcus neoformans by Microglial Cells. J. Neuroinflamm. 2013, 10, 841. [Google Scholar] [CrossRef]
- Lipovsky, M.M.; Gekker, G.; Anderson, W.R.; Molitor, T.W.; Peterson, P.K.; Hoepelman, A.I.M. Phagocytosis of Nonopsonized Cryptococcus neoformans by Swine Microglia Involves CD14 Receptors. Clin. Immunol. Immunopathol. 1997, 84, 208–211. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Kress, Y.; Dickson, D.W.; Casadevall, A. Human Microglia Mediate Anti-Cryptococcus neoformans Activity in the Presence of Specific Antibody. J. Neuroimmunol. 1995, 62, 43–52. [Google Scholar] [CrossRef]
- Barluzzi, R.; Brozzetti, A.; Delfino, D.; Bistoni, F.; Blasi, E. Role of the Capsule in Microglial Cell—Cryptococcus neoformans Interaction: Impairment of Antifungal Activity but Not of Secretory Functions. Med. Mycol. 1998, 36, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Casadevall, A.; Dickson, D.W. Immunohistochemical Localization of Capsular Polysaccharide Antigen in the Central Nervous System Cells in Cryptococcal Meningoencephalitis. Am. J. Pathol. 1996, 148, 1267–1274. [Google Scholar]
- Goldman, D.; Song, X.; Kitai, R.; Casadevall, A.; Zhao, M.L.; Lee, S.C. Cryptococcus neoformans Induces Macrophage Inflammatory Protein 1α (MIP-1α) and MIP-1β in Human Microglia: Role of Specific Antibody and Soluble Capsular Polysaccharide. Infect. Immun. 2001, 69, 1808–1815. [Google Scholar] [CrossRef]
- Blasi, E.; Barluzzi, R.; Mazzolla, R.; Tancini, B.; Saleppico, S.; Puliti, M.; Pitzurra, L.; Bistoni, F. Role of Nitric Oxide and Melanogenesis in the Accomplishment of Anticryptococcal Activity by the BV-2 Microglial Cell Line. J Neuroimmunol. 1995, 58, 111–116. [Google Scholar] [CrossRef]
- Adami, C.; Sorci, G.; Blasi, E.; Agneletti, A.L.; Bistoni, F.; Donato, R. S100b Expression in and Effects on Microglia. Glia 2001, 33, 131–142. [Google Scholar] [CrossRef]
- Saleppico, S.; Boelaert, J.R.; Salè, F.O.; Mazzolla, R.; Morucci, P.; Bistoni, F.; Blasi, E. Differential Effects of Iron Load on Basal and Interferon-Gamma plus Lipopolysaccharide Enhance Anticryptococcal Activity by the Murine Microglial Cell Line BV-2. J. Neuroimmunol. 1999, 93, 102–107. [Google Scholar] [CrossRef]
- Lipovsky, M.M.; Juliana, A.E.; Gekker, G.; Hu, S.; Hoepelman, A.I.M.; Peterson, P.K. Effect of Cytokines on Anticryptococcal Activity of Human Microglial Cells. Clin. Diagn. Lab. Immunol. 1998, 5, 410–411. [Google Scholar] [CrossRef]
- Lee, S.C.; Kress, Y.; Zhao, M.L.; Dickson, D.W.; Casadevall, A. Cryptococcus neoformans Survive and Replicate in Human Microglia. Lab. Investig. 1995, 73, 871–879. [Google Scholar]
- Aguirre, K.; Miller, S. MHC Class II-Positive Perivascular Microglial Cells Mediate Resistance to Cryptococcus neoformans Brain Infection. Glia 2002, 39, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Gault, R.A.; Kozel, T.R.; Murphy, W.J. Immunomodulation with CD40 Stimulation and Interleukin-2 Protects Mice from Disseminated Cryptococcosis. Infect. Immun. 2006, 74, 2161–2168. [Google Scholar] [CrossRef]
- Zhou, Q.; Gault, R.A.; Kozel, T.R.; Murphy, W.J. Protection from Direct Cerebral Cryptococcus Infection by Interferon-γ-Dependent Activation of Microglial Cells. J. Immunol. 2007, 178, 5753–5761. [Google Scholar] [CrossRef]
- Neal, L.M.; Xing, E.; Xu, J.; Kolbe, J.L.; Osterholzer, J.J.; Segal, B.M.; Williamson, P.R.; Olszewski, M.A. CD4+ T Cells Orchestrate Lethal Immune Pathology despite Fungal Clearance during Cryptococcus neoformans Meningoencephalitis. MBio 2017, 8, e01415-17. [Google Scholar] [CrossRef]
- Shi, C.; Pamer, E.G. Monocyte Recruitment during Infection and Inflammation. Nat. Rev. Immunol. 2011, 11, 762–774. [Google Scholar] [CrossRef] [PubMed]
- Geissmann, F.; Jung, S.; Littman, D.R. Blood Monocytes Consist of Two Principal Subsets with Distinct Migratory Properties. Immunity 2003, 19, 71–82. [Google Scholar] [CrossRef]
- Sunderkötter, C.; Nikolic, T.; Dillon, M.J.; van Rooijen, N.; Stehling, M.; Drevets, D.A.; Leenen, P.J.M. Subpopulations of Mouse Blood Monocytes Differ in Maturation Stage and Inflammatory Response. J. Immunol. 2004, 172, 4410–4417. [Google Scholar] [CrossRef]
- Serbina, N.V.; Pamer, E.G. Monocyte Emigration from Bone Marrow during Bacterial Infection Requires Signals Mediated by Chemokine Receptor CCR2. Nat. Immunol. 2006, 7, 311–317. [Google Scholar] [CrossRef]
- Espinosa, V.; Jhingran, A.; Dutta, O.; Kasahara, S.; Donnelly, R.; Du, P.; Rosenfeld, J.; Leiner, I.; Chen, C.C.; Ron, Y.; et al. Inflammatory Monocytes Orchestrate Innate Antifungal Immunity in the Lung. PLoS Pathog. 2014, 10, e1003940. [Google Scholar] [CrossRef] [PubMed]
- Hohl, T.M.; Rivera, A.; Lipuma, L.; Gallegos, A.; Shi, C.; Mack, M.; Pamer, E.G. Inflammatory Monocytes Facilitate Adaptive CD4 T Cell Responses during Respiratory Fungal Infection. Cell Host Microbe 2009, 6, 470–481. [Google Scholar] [CrossRef]
- Ngo, L.Y.; Kasahara, S.; Kumasaka, D.K.; Knoblaugh, S.E.; Jhingran, A.; Hohl, T.M. Inflammatory Monocytes Mediate Early and Organ-Specific Innate Defense during Systemic Candidiasis. J. Infect. Dis. 2014, 209, 109–119. [Google Scholar] [CrossRef]
- Wüthrich, M.; Ersland, K.; Sullivan, T.; Galles, K.; Klein, B.S. Fungi Subvert Vaccine T Cell Priming at the Respiratory Mucosa by Preventing Chemokine-Induced Influx of Inflammatory Monocytes. Immunity 2012, 36, 680–692. [Google Scholar] [CrossRef]
- Szymczak, W.A.; Deepe, G.S. The CCL7-CCL2-CCR2 Axis Regulates IL-4 Production in Lungs and Fungal Immunity. J. Immunol. 2009, 183, 1964–1974. [Google Scholar] [CrossRef] [PubMed]
- Traynor, T.R.; Herring, A.C.; Dorf, M.E.; Kuziel, W.A.; Toews, G.B.; Huffnagle, G.B. Differential Roles of CC Chemokine Ligand 2/Monocyte Chemotactic Protein-1 and CCR2 in the Development of T1 Immunity. J. Immunol. 2002, 168, 4659–4666. [Google Scholar] [CrossRef] [PubMed]
- Traynor, T.R.; Kuziel, W.A.; Toews, G.B.; Huffnagle, G.B. CCR2 Expression Determines T1 versus T2 Polarization during Pulmonary Cryptococcus neoformans Infection. J. Immunol. 2000, 164, 2021–2027. [Google Scholar] [CrossRef]
- Osterholzer, J.J.; Chen, G.H.; Olszewski, M.A.; Zhang, Y.M.; Curtis, J.L.; Huffnagle, G.B.; Toews, G.B. Chemokine Receptor 2-Mediated Accumulation of Fungicidal Exudate Macrophages in Mice That Clear Cryptococcal Lung Infection. Am. J. Pathol. 2011, 178, 198–211. [Google Scholar] [CrossRef]
- Osterholzer, J.J.; Curtis, J.L.; Polak, T.; Ames, T.; Chen, G.H.; McDonald, R.; Huffnagle, G.B.; Toews, G.B. CCR2 Mediates Conventional Dendritic Cell Recruitment and the Formation of Bronchovascular Mononuclear Cell Infiltrates in the Lungs of Mice Infected with Cryptococcus neoformans. J. Immunol. 2008, 181, 610–620. [Google Scholar] [CrossRef]
- Osterholzer, J.J.; Milam, J.E.; Chen, G.H.; Toews, G.B.; Huffnagle, G.B.; Olszewski, M.A. Role of Dendritic Cells and Alveolar Macrophages in Regulating Early Host Defense against Pulmonary Infection with Cryptococcus neoformans. Infect. Immun. 2009, 77, 3749–3758. [Google Scholar] [CrossRef] [PubMed]
- Hardison, S.E.; Herrera, G.; Young, M.L.; Hole, C.R.; Wozniak, K.L.; Wormley, F.L. Protective Immunity against Pulmonary Cryptococcosis Is Associated with STAT1-Mediated Classical Macrophage Activation. J. Immunol. 2012, 189, 4060–4068. [Google Scholar] [CrossRef] [PubMed]
- Leopold Wager, C.M.; Hole, C.R.; Campuzano, A.; Castro-Lopez, N.; Cai, H.; Van Dyke, M.C.; Wozniak, K.L.; Wang, Y.; Wormley, F.L. IFN-γ Immune Priming of Macrophages in Vivo Induces Prolonged STAT1 Binding and Protection against Cryptococcus neoformans. PLoS Pathog. 2018, 14, e1007358. [Google Scholar] [CrossRef] [PubMed]
- Leopold Wager, C.M.; Hole, C.R.; Wozniak, K.L.; Olszewski, M.A.; Mueller, M.; Wormley, F.L. STAT1 Signaling within Macrophages Is Required for Antifungal Activity against Cryptococcus neoformans. Infect Immun. 2015, 83, 4513–4527. [Google Scholar] [CrossRef] [PubMed]
- Masso-Silva, J.; Espinosa, V.; Liu, T.B.; Wang, Y.; Xue, C.; Rivera, A. The F-Box Protein Fbp1 Shapes the Immunogenic Potential of Cryptococcus neoformans. MBio 2018, 9, e01828-17. [Google Scholar] [CrossRef] [PubMed]
- Wiesner, D.L.; Specht, C.A.; Lee, C.K.; Smith, K.D.; Mukaremera, L.; Lee, S.T.; Lee, C.G.; Elias, J.A.; Nielsen, J.N.; Boulware, D.R.; et al. Chitin Recognition via Chitotriosidase Promotes Pathologic Type-2 Helper T Cell Responses to Cryptococcal Infection. PLoS Pathog. 2015, 11, e1004701. [Google Scholar] [CrossRef]
- Heung, L.J.; Hohl, T.M. Inflammatory Monocytes Are Detrimental to the Host Immune Response during Acute Infection with Cryptococcus neoformans. PLoS Pathog. 2019, 15, e1007627. [Google Scholar] [CrossRef]
- Williams, J.L.; Holman, D.W.; Klein, R.S. Chemokines in the Balance: Maintenance of Homeostasis and Protection at CNS Barriers. Front. Cell. Neurosci. 2014, 8, 154. [Google Scholar] [CrossRef]
- Getts, D.R.; Terry, R.L.; Getts, M.T.; Müller, M.; Rana, S.; Shrestha, B.; Radford, J.; Van Rooijen, N.; Campbell, I.L.; King, N.J.C. Ly6c+ “Inflammatory Monocytes” are Microglial Precursors Recruited in a Pathogenic Manner in West Nile Virus Encephalitis. J. Exp. Med. 2008, 205, 2319–2337. [Google Scholar] [CrossRef]
- Huffnagle, G.B.; McNeil, L.K. Dissemination of C. neoformans to the Central Nervous System: Role of Chemokines, Th1 Immunity and Leukocyte Recruitment. J. Neurovirol. 1999, 5, 76–81. [Google Scholar] [CrossRef]
- Xu, J.; Ganguly, A.; Zhao, J.; Ivey, M.; Lopez, R.; Osterholzer, J.J.; Cho, C.S.; Olszewski, M.A. CCR2 Signaling Promotes Brain Infiltration of Inflammatory Monocytes and Contributes to Neuropathology during Cryptococcal Meningoencephalitis. MBio 2021, 12, e01076-21. [Google Scholar] [CrossRef] [PubMed]
- Sedgwick, A.J.; Ghazanfari, N.; Constantinescu, P.; Mantamadiotis, T.; Barrow, A.D. The Role of NK Cells and Innate Lymphoid Cells in Brain Cancer. Front. Immunol. 2020, 11, 1549. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, N.; Murphy, J.W. In Vitro Binding of Natural Killer Cells to Cryptococcus neoformans Targets. Infect. Immun. 1985, 50, 50–57. [Google Scholar] [CrossRef]
- Levitz, S.M.; Dupont, M.P.; Smail, E.H. Direct Activity of Human T Lymphocytes and Natural Killer Cells against Cryptococcus neoformans. Infect. Immun. 1994, 62, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Hidore, M.R.; Nabavi, N.; Sonleitner, F.; Murphy, J.W. Murine Natural Killer Cells Are Fungicidal to Cryptococcus neoformans. Infect. Immun. 1991, 59, 1747–1754. [Google Scholar] [CrossRef]
- Murphy, J.W.; Hidore, M.R.; Wong, S.C. Direct Interactions of Human Lymphocytes with the Yeast-Like Organism, Cryptococcus neoformans. J. Clin. Investig. 1993, 91, 1553–1566. [Google Scholar] [CrossRef]
- Hidore, M.R.; Mislan, T.W.; Murphy, J.W. Responses of Murine Natural Killer Cells to Binding of the Fungal Target Cryptococcus neoformans. Infect. Immun. 1991, 59, 1489–1499. [Google Scholar] [CrossRef]
- Ma, L.L.; Wang, C.L.C.; Neely, G.G.; Epelman, S.; Krensky, A.M.; Mody, C.H. NK Cells Use Perforin Rather than Granulysin for Anticryptococcal Activity. J. Immunol. 2004, 173, 3357–3365. [Google Scholar] [CrossRef]
- Wiseman, J.C.D.; Ma, L.L.; Marr, K.J.; Jones, G.J.; Mody, C.H. Perforin-Dependent Cryptococcal Microbicidal Activity in NK Cells Requires PI3K-Dependent ERK1/2 Signaling. J. Immunol. 2007, 178, 6456–6464. [Google Scholar] [CrossRef]
- Li, S.S.; Ogbomo, H.; Mansour, M.K.; Xiang, R.F.; Szabo, L.; Munro, F.; Mukherjee, P.; Mariuzza, R.A.; Amrein, M.; Vyas, J.M.; et al. Identification of the Fungal Ligand Triggering Cytotoxic PRR-Mediated NK Cell Killing of Cryptococcus and Candida. Nat. Commun. 2018, 9, 751. [Google Scholar] [CrossRef]
- Li, S.S.; Kyei, S.K.; Timm-Mccann, M.; Ogbomo, H.; Jones, G.J.; Shi, M.; Xiang, R.F.; Oykhman, P.; Huston, S.M.; Islam, A.; et al. The NK Receptor NKp30 Mediates Direct Fungal Recognition and Killing and Is Diminished in NK Cells from HIV-Infected Patients. Cell Host Microbe 2013, 14, 387–397. [Google Scholar] [CrossRef]
- Zhang, T.; Kawakami, K.; Qureshi, M.H.; Okamura, H.; Kurimoto, M.; Saito, A. Interleukin-12 (IL-12) and IL-18 Synergistically Induce the Fungicidal Activity of Murine Peritoneal Exudate Cells against Cryptococcus neoformans through Production of Gamma Interferon by Natural Killer Cells. Infect. Immun. 1997, 65, 3594–3599. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, K.; Koguchi, Y.; Qureshi, M.H.; Miyazato, A.; Yara, S.; Kinjo, Y.; Iwakura, Y.; Takeda, K.; Akira, S.; Kurimoto, M.; et al. IL-18 Contributes to Host Resistance against Infection with Cryptococcus neoformans in Mice with Defective IL-12 Synthesis through Induction of IFN-γ Production by NK Cells. J. Immunol. 2000, 165, 941–947. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, K.; Koguchi, Y.; Qureshi, M.H.; Yara, S.; Kinjo, Y.; Uezu, K.; Saito, A. NK Cells Eliminate Cryptococcus neoformans by Potentiating the Fungicidal Activity of Macrophages Rather than by Directly Killing Them upon Stimulation with IL-12 and IL-18. Microbiol. Immunol. 2000, 44, 1043–1050. [Google Scholar] [CrossRef] [PubMed]
- Mars, L.T.; Mas, M.; Beaudoin, L.; Bauer, J.; Leite-de-Moraes, M.; Lehuen, A.; Bureau, J.F.; Liblau, R.S. Invariant NKT Cells Regulate the CD8 T Cell Response during Theiler’s Virus Infection. PLoS ONE 2014, 9, e87717. [Google Scholar] [CrossRef]
- Mars, L.T.; Gautron, A.-S.; Novak, J.; Beaudoin, L.; Diana, J.; Liblau, R.S.; Lehuen, A. Invariant NKT Cells Regulate Experimental Autoimmune Encephalomyelitis and Infiltrate the Central Nervous System in a CD1d-Independent Manner. J. Immunol. 2008, 181, 2321–2329. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, K.; Kinjo, Y.; Uezu, K.; Yara, S.; Miyagi, K.; Koguchi, Y.; Nakayama, T.; Taniguchi, M.; Saito, A. Monocyte Chemoattractant Protein-1-Dependent Increase of Vα14 NKT Cells in Lungs and Their Roles in Th1 Response and Host Defense in Cryptococcal Infection. J. Immunol. 2001, 167, 6525–6532. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, K.; Kinjo, Y.; Yara, S.; Uezu, K.; Koguchi, Y.; Tohyama, M.; Azuma, M.; Takeda, K.; Akira, S.; Saito, A. Enhanced Gamma Interferon Production through Activation of Vα14+ Natural Killer T Cells by α-Galactosylceramide in Interleukin-18-Deficient Mice with Systemic Cryptococcosis. Infect. Immun. 2001, 69, 6643–6650. [Google Scholar] [CrossRef] [PubMed]
- Blackstock, R.; Murphy, J.W. Age-Related Resistance of C57BL/6 Mice to Cryptococcus neoformans Is Dependent on Maturation of NKT Cells. Infect. Immun. 2004, 72, 5175–5180. [Google Scholar] [CrossRef] [PubMed]
- Mirza, S.A.; Phelan, M.; Rimland, D.; Graviss, E.; Hamill, R.; Brandt, M.E.; Gardner, T.; Sattah, M.; De Leon, G.P.; Baughman, W.; et al. The Changing Epidemiology of Cryptococcosis: An Update from Population-Based Active Surveillance in 2 Large Metropolitan Areas, 1992–2000. Clin. Infect. Dis. 2003, 36, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Huffnagle, G.B.; Yates, J.L.; Lipscomb, M.F. Immunity to a Pulmonary Cryptococcus neoformans Infection Requires Both CD4+ and CD8+ T Cells. J. Exp. Med. 1991, 173, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Huffnagle, G.B.; Lipscomb, M.F.; Lovchik, J.A.; Hoag, K.A.; Street, N.E. The Role of CD4+ and CD8+ T Cells in the Protective Inflammatory Response to a Pulmonary Cryptococcal Infection. J. Leukoc. Biol. 1994, 55, 35–42. [Google Scholar] [CrossRef]
- Hill, J.O.; Harmsen, A.G. Intrapulmonary Growth and Dissemination of an Avirulent Strain of Cryptococcus neoformans in Mice Depleted of CD4+ or CD8+ T Cells. J. Exp. Med. 1991, 173, 755–758. [Google Scholar] [CrossRef] [PubMed]
- Mody, C.H.; Chen, G.H.; Jackson, C.; Curtis, J.L.; Toews, G.B. Depletion of Murine CD8+ T Cells in Vivo Decreases Pulmonary Clearance of a Moderately Virulent Strain of Cryptococcus neoformans. J. Lab. Clin. Med. 1993, 121, 765–773. [Google Scholar]
- Lindell, D.M.; Moore, T.A.; McDonald, R.A.; Toews, G.B.; Huffnagle, G.B. Generation of Antifungal Effector CD8+ T Cells in the Absence of CD4+ T Cells during Cryptococcus neoformans Infection. J. Immunol. 2005, 174, 7920–7928. [Google Scholar] [CrossRef]
- Xu, J.; Neal, L.M.; Ganguly, A.; Kolbe, J.L.; Hargarten, J.C.; Elsegeiny, W.; Hollingsworth, C.; He, X.; Ivey, M.; Lopez, R.; et al. Chemokine Receptor CXCR3 Is Required for Lethal Brain Pathology but Not Pathogen Clearance during Cryptococcal Meningoencephalitis. Sci. Adv. 2020, 6, 2502–2519. [Google Scholar] [CrossRef]
- Buchanan, K.L.; Doyle, H.A. Requirement for CD4+ T Lymphocytes in Host Resistance against Cryptococcus neoformans in the Central Nervous System of Immunized Mice. Infect. Immun. 2000, 68, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Uicker, W.; McCracken, J.P.; Buchanan, K.L. Role of CD4+ T Cells in a Protective Immune Response against Cryptococcus neoformans in the Central Nervous System. Med. Mycol. 2006, 44, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.L.; Spurrell, J.C.L.; Wang, J.F.; Neely, G.G.; Epelman, S.; Krensky, A.M.; Mody, C.H. CD8 T Cell-Mediated Killing of Cryptococcus neoformans Requires Granulysin and Is Dependent on CD4 T Cells and IL-15. J. Immunol. 2002, 169, 5787–5795. [Google Scholar] [CrossRef]
- Chun, F.Z.; Ling, L.M.; Jones, G.J.; Gill, M.J.; Krensky, A.M.; Kubes, P.; Mody, C.H. Cytotoxic CD4+ T Cells Use Granulysin to Kill Cryptococcus neoformans, and Activation of This Pathway Is Defective in HIV Patients. Blood 2007, 109, 2049–2057. [Google Scholar] [CrossRef]
- Zheng, C.F.; Jones, G.J.; Shi, M.; Wiseman, J.C.D.; Marr, K.J.; Berenger, B.M.; Huston, S.M.; Gill, M.J.; Krensky, A.M.; Kubes, P.; et al. Late Expression of Granulysin by Microbicidal CD4+ T Cells Requires PI3K- and STAT5-Dependent Expression of IL-2Rβ That Is Defective in HIV-Infected Patients. J. Immunol. 2008, 180, 7221–7229. [Google Scholar] [CrossRef]
- Chen, G.H.; McDonald, R.A.; Wells, J.C.; Huffnagle, G.B.; Lukacs, N.W.; Toews, G.B. The Gamma Interferon Receptor Is Required for the Protective Pulmonary Inflammatory Response to Cryptococcus neoformans. Infect. Immun. 2005, 73, 1788–1796. [Google Scholar] [CrossRef]
- Hardison, S.E.; Ravi, S.; Wozniak, K.L.; Young, M.L.; Olszewski, M.A.; Wormley, F.L. Pulmonary Infection with an Interferon-γ-Producing Cryptococcus neoformans Strain Results in Classical Macrophage Activation and Protection. Am. J. Pathol. 2010, 176, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, K.; Kohno, S.; Kadota, J.I.; Tohyama, M.; Teruya, K.; Kudeken, N.; Saito, A.; Hara, K. T Cell-Dependent Activation of Macrophages and Enhancement of Their Phagocytic Activity in the Lungs of Mice Inoculated with Heat-Killed Cryptococcus neoformans: Involvement of IFN-γ and Its Protective Effect against Cryptococcal Infection. Microbiol. Immunol. 1995, 39, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, Y.; Arora, S.; Erb-Downward, J.R.; McDonald, R.A.; Toews, G.B.; Huffnagle, G.B. Distinct Roles for IL-4 and IL-10 in Regulating T2 Immunity during Allergic Bronchopulmonary Mycosis. J. Immunol. 2005, 174, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Müller, U.; Stenzel, W.; Köhler, G.; Werner, C.; Polte, T.; Hansen, G.; Schütze, N.; Straubinger, R.K.; Blessing, M.; McKenzie, A.N.J.; et al. IL-13 Induces Disease-Promoting Type 2 Cytokines, Alternatively Activated Macrophages and Allergic Inflammation during Pulmonary Infection of Mice with Cryptococcus neoformans. J. Immunol. 2007, 179, 5367–5377. [Google Scholar] [CrossRef]
- Stenzel, W.; Müller, U.; Köhler, G.; Heppner, F.L.; Blessing, M.; McKenzie, A.N.J.; Brombacher, F.; Alber, G. IL-4/IL-13-Dependent Alternative Activation of Macrophages but Not Microglial Cells Is Associated with Uncontrolled Cerebral Cryptococcosis. Am. J. Pathol. 2009, 174, 486–496. [Google Scholar] [CrossRef]
- Huffnagle, G.B.; Boyd, M.B.; Street, N.E.; Lipscomb, M.F. IL-5 Is Required for Eosinophil Recruitment, Crystal Deposition, and Mononuclear Cell Recruitment during a Pulmonary Cryptococcus neoformans Infection in Genetically Susceptible Mice (C57BL/6). J. Immunol. 1998, 160, 2393–2400. [Google Scholar] [PubMed]
- Mody, C.H.; Tyler, C.L.; Sitrin, R.G.; Jackson, C.; Toews, G.B. Interferon-γ Activates Rat Alveolar Macrophages for Anticryptococcal Activity. Am. J. Respir. Cell Mol. Biol. 1991, 5, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Joly, V.; Saint Julien, L.; Carbon, C.; Yeni, P. In Vivo Activity of Interferon-γ in Combination with Amphotericin B in the Treatment of Experimental Cryptococcosis. J. Infect. Dis. 1994, 170, 1331–1334. [Google Scholar] [CrossRef]
- Flesch, I.E.; Schwamberger, G.; Kaufmann, S.H. Fungicidal Activity of IFN-Gamma-Activated Macrophages. Extracellular Killing of Cryptococcus neoformans. J. Immunol. 1989, 142, 3219–3224. [Google Scholar]
- Huffnagle, G.B. Role of Cytokines in T Cell Immunity to a Pulmonary Cryptococcus neoformans Infection. Neurosignals 1996, 5, 215–222. [Google Scholar] [CrossRef]
- Wormley, F.L.; Perfect, J.R.; Steele, C.; Cox, G.M. Protection against Cryptococcosis by Using a Murine Gamma Interferon-Producing Cryptococcus neoformans Strain. Infect. Immun. 2007, 75, 1453–1462. [Google Scholar] [CrossRef]
- Uicker, W.C.; Doyle, H.A.; McCracken, J.P.; Langlois, M.; Buchanan, K.L. Cytokine and Chemokine Expression in the Central Nervous System Associated with Protective Cell-Mediated Immunity against Cryptococcus neoformans. Med. Mycol. 2005, 43, 27–38. [Google Scholar] [CrossRef]
- Aguirre, K.; Havell, E.A.; Gibson, G.W.; Johnson, L.L. Role of Tumor Necrosis Factor and Gamma Interferon in Acquired Resistance to Cryptococcus neoformans in the Central Nervous System of Mice. Infect. Immun. 1995, 63, 1725–1731. [Google Scholar] [CrossRef]
- Lee, S.C.; Dickson, D.W.; Brosnan, C.F.; Casadevall, A. Human Astrocytes Inhibit Cryptococcus neoformans Growth by a Nitric Oxide-Mediated Mechanism. J. Exp. Med. 1994, 180, 365–369. [Google Scholar] [CrossRef]
- Jarvis, J.N.; Casazza, J.P.; Stone, H.H.; Meintjes, G.; Lawn, S.D.; Levitz, S.M.; Harrison, T.S.; Koup, R.A. The Phenotype of the Cryptococcus-Specific CD4+ Memory T-Cell Response Is Associated with Disease Severity and Outcome in HIV-Associated Cryptococcal Meningitis. J. Infect. Dis. 2013, 207, 1817–1828. [Google Scholar] [CrossRef]
- Jarvis, J.N.; Meintjes, G.; Rebe, K.; Williams, G.N.; Bicanic, T.; Williams, A.; Schutz, C.; Bekker, L.G.; Wood, R.; Harrison, T.S. Adjunctive Interferon-γ Immunotherapy for the Treatment of HIV-Associated Cryptococcal Meningitis: A Randomized Controlled Trial. AIDS 2012, 26, 1105–1113. [Google Scholar] [CrossRef]
- Pappas, P.G.; Bustamante, B.; Ticona, E.; Hamill, R.J.; Johnson, P.C.; Reboli, A.; Aberg, J.; Hasbun, R.; Hsu, H.H. Recombinant Interferon-γ1b as Adjunctive Therapy for AIDS-Related Acute Cryptococcal Meningitis. J. Infect. Dis. 2004, 189, 2185–2191. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, F.; Tompkins, K.C.; McNamara, A.; Jain, A.V.; Moore, B.B.; Toews, G.B.; Huffnagle, G.B.; Olszewski, M.A. Robust Th1 and Th17 Immunity Supports Pulmonary Clearance but Cannot Prevent Systemic Dissemination of Highly Virulent Cryptococcus neoformans H99. Am. J. Pathol. 2009, 175, 2489–2500. [Google Scholar] [CrossRef]
- Murdock, B.J.; Huffnagle, G.B.; Olszewski, M.A.; Osterholzer, J.J. Interleukin-17A Enhances Host Defense against Cryptococcal Lung Infection through Effects Mediated by Leukocyte Recruitment, Activation, and Gamma Interferon Production. Infect. Immun. 2014, 82, 937–948. [Google Scholar] [CrossRef] [PubMed]
- Sionov, E.; Mayer-Barber, K.D.; Chang, Y.C.; Kauffman, K.D.; Eckhaus, M.A.; Salazar, A.M.; Barber, D.L.; Kwon-Chung, K.J. Type I IFN Induction via Poly-ICLC Protects Mice against Cryptococcosis. PLoS Pathog. 2015, 11, e1005040. [Google Scholar] [CrossRef] [PubMed]
- Huffnagle, G.B.; Toews, G.B.; Burdick, M.D.; Boyd, M.B.; McAllister, K.S.; McDonald, R.A.; Kunkel, S.L.; Strieter, R.M. Afferent Phase Production of TNF-Alpha Is Required for the Development of Protective T Cell Immunity to Cryptococcus neoformans. J. Immunol. 1996, 157, 4529–4536. [Google Scholar] [PubMed]
- Herring, A.C.; Lee, J.; McDonald, R.A.; Toews, G.B.; Huffnagle, G.B. Induction of Interleukin-12 and Gamma Interferon Requires Tumor Necrosis Factor Alpha for Protective T1-Cell-Mediated Immunity to Pulmonary Cryptococcus neoformans Infection. Infect. Immun. 2002, 70, 2959–2964. [Google Scholar] [CrossRef]
- Xu, J.; Eastman, A.J.; Flaczyk, A.; Neal, L.M.; Zhao, G.; Carolan, J.; Malachowski, A.N.; Stolberg, V.R.; Yosri, M.; Chensue, S.W.; et al. Disruption of Early Tumor Necrosis Factor Alpha Signaling Prevents Classical Activation of Dendritic Cells in Lung-Associated Lymph Nodes and Development of Protective Immunity against Cryptococcal Infection. MBio 2016, 7, e00510-16. [Google Scholar] [CrossRef]
- Fa, Z.; Xu, J.; Yi, J.; Sang, J.; Pan, W.; Xie, Q.; Yang, R.; Fang, W.; Liao, W.; Olszewski, M.A. TNF-α-Producing Cryptococcus neoformans Exerts Protective Effects on Host Defenses in Murine Pulmonary Cryptococcosis. Front. Immunol. 2019, 10, 1725. [Google Scholar] [CrossRef]
- Vecchiarelli, A.; Pericolini, E.; Gabrielli, E.; Kenno, S.; Perito, S.; Cenci, E.; Monari, C. Elucidating the Immunological Function of the Cryptococcus neoformans Capsule. Future Microbiol. 2013, 8, 1107–1116. [Google Scholar] [CrossRef]
- Zaragoza, O.; Rodrigues, M.L.; De Jesus, M.; Frases, S.; Dadachova, E.; Casadevall, A. The Capsule of the Fungal Pathogen Cryptococcus neoformans. Adv. Appl. Microbiol. 2009, 68, 133–216. [Google Scholar] [CrossRef]
- Macura, N.; Zhang, T.; Casadevall, A. Dependence of Macrophage Phagocytic Efficacy on Antibody Concentration. Infect. Immun. 2007, 75, 1904–1915. [Google Scholar] [CrossRef]
- Vecchiarelli, A.; Retini, C.; Monari, C.; Tascini, C.; Bistoni, F.; Kozel, T.R. Purified Capsular Polysaccharide of Cryptococcus neoformans Induces Interleukin-10 Secretion by Human Monocytes. Infect. Immun. 1996, 64, 2846–2849. [Google Scholar] [CrossRef]
- Retini, C.; Vecchiarelli, A.; Monari, C.; Bistoni, F.; Kozel, T.R. Encapsulation of Cryptococcus neoformans with Glucuronoxylomannan Inhibits the Antigen-Presenting Capacity of Monocytes. Infect. Immun. 1998, 66, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Monari, C.; Bistoni, F.; Casadevall, A.; Pericolini, E.; Pietrella, D.; Kozel, T.R.; Vecchiarelli, A. Glucuronoxylomannan, a Microbial Compound, Regulates Expression of Costimulatory Molecules and Production of Cytokines in Macrophages. J. Infect. Dis. 2005, 191, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Monari, C.; Pericolini, E.; Bistoni, G.; Casadevall, A.; Kozel, T.R.; Vecchiarelli, A. Cryptococcus neoformans Capsular Glucuronoxylomannan Induces Expression of Fas Ligand in Macrophages. J. Immunol. 2005, 174, 3461–3468. [Google Scholar] [CrossRef] [PubMed]
- Villena, S.N.; Pinheiro, R.O.; Pinheiro, C.S.; Nunes, M.P.; Takiya, C.M.; Dosreis, G.A.; Previato, J.O.; Mendonça-Previato, L.; Freire-de-Lima, C.G. Capsular Polysaccharides Galactoxylomannan and Glucuronoxylomannan from Cryptococcus neoformans Induce Macrophage Apoptosis Mediated by Fas Ligand. Cell. Microbiol. 2008, 10, 1274–1285. [Google Scholar] [CrossRef]
- Chiapello, L.S.; Baronetti, J.L.; Garro, A.P.; Spesso, M.F.; Masih, D.T. Cryptococcus neoformans Glucuronoxylomannan Induces Macrophage Apoptosis Mediated by Nitric Oxide in a Caspase-Independent Pathway. Int. Immunol. 2008, 20, 1527–1541. [Google Scholar] [CrossRef]
- Vecchiarelli, A.; Pietrella, D.; Lupo, P.; Bistoni, F.; McFadden, D.C.; Casadevall, A. The Polysaccharide Capsule of Cryptococcus neoformans Interferes with Human Dendritic Cell Maturation and Activation. J. Leukoc. Biol. 2003, 74, 370–378. [Google Scholar] [CrossRef]
- Huston, S.M.; Ngamskulrungroj, P.; Xiang, R.F.; Ogbomo, H.; Stack, D.; Li, S.S.; Timm-McCann, M.; Kyei, S.K.; Oykhman, P.; Kwon-Chung, K.J.; et al. Cryptococcus gattii Capsule Blocks Surface Recognition Required for Dendritic Cell Maturation Independent of Internalization and Antigen Processing. J. Immunol. 2016, 196, 1259–1271. [Google Scholar] [CrossRef]
- Denham, S.T.; Verma, S.; Reynolds, R.C.; Worne, C.L.; Daugherty, J.M.; Lane, T.E.; Brown, J.C.S. Regulated Release of Cryptococcal Polysaccharide Drives Virulence and Suppresses Immune Cell Infiltration into the Central Nervous System. Infect. Immun. 2018, 86, e00662-17. [Google Scholar] [CrossRef]
- Robertson, E.J.; Najjuka, G.; Rolfes, M.A.; Akampurira, A.; Jain, N.; Anantharanjit, J.; Von Hohenberg, M.; Tassieri, M.; Carlsson, A.; Meya, D.B.; et al. Cryptococcus neoformans Ex Vivo Capsule Size Is Associated with Intracranial Pressure and Host Immune Response in HIV-Associated Cryptococcal Meningitis. J. Infect. Dis. 2014, 209, 74–82. [Google Scholar] [CrossRef]
- Jarvis, J.N.; Percival, A.; Bauman, S.; Pelfrey, J.; Meintjes, G.; Williams, G.N.; Longley, N.; Harrison, T.S.; Kozel, T.R. Evaluation of a Novel Point-of-Care Cryptococcal Antigen Test on Serum, Plasma, and Urine from Patients with HIV-Associated Cryptococcal Meningitis. Clin. Infect. Dis. 2011, 53, 1019–1023. [Google Scholar] [CrossRef]
- Scriven, J.E.; Graham, L.M.; Schutz, C.; Scriba, T.J.; Wilkinson, K.A.; Wilkinson, R.J.; Boulware, D.R.; Urban, B.C.; Lalloo, D.G.; Meintjes, G. A Glucuronoxylomannan-Associated Immune Signature, Characterized by Monocyte Deactivation and an Increased Interleukin 10 Level, Is a Predictor of Death in Cryptococcal Meningitis. J. Infect. Dis. 2016, 213, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Salas, S.D.; Bennett, J.E.; Kwon-Chung, K.J.; Perfect, J.R.; Williamson, P.R. Effect of the Laccase Gene, CNLAC1, on Virulence of Cryptococcus neoformans. J. Exp. Med. 1996, 184, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Tewari, R.P.; Williamson, P.R. Laccase Protects Cryptococcus neoformans from Antifungal Activity of Alveolar Macrophages. Infect. Immun. 1999, 67, 6034–6039. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Williamson, P.R. Role of Laccase in the Biology and Virulence of Cryptococcus neoformans. FEMS Yeast Res. 2004, 5, 1–10. [Google Scholar] [CrossRef]
- Noverr, M.C.; Williamson, P.R.; Fajardo, R.S.; Huffnagle, G.B. CNLAC1 is Required for Extrapulmonary Dissemination of Cryptococcus neoformans but not Pulmonary Persistence. Infect. Immun. 2004, 72, 1693–1699. [Google Scholar] [CrossRef]
- Qiu, Y.; Davis, M.J.; Dayrit, J.K.; Hadd, Z.; Meister, D.L.; Osterholzer, J.J.; Williamson, P.R.; Olszewski, M.A. Immune Modulation Mediated by Cryptococcal Laccase Promotes Pulmonary Growth and Brain Dissemination of Virulent Cryptococcus neoformans in Mice. PLoS ONE 2012, 7, e47853. [Google Scholar] [CrossRef]
- Erb-Downward, J.R.; Noggle, R.M.; Williamson, P.R.; Huffnagle, G.B. The Role of Laccase in Prostaglandin Production by Cryptococcus neoformans. Mol. Microbiol. 2008, 68, 1428–1437. [Google Scholar] [CrossRef]
- Hansakon, A.; Ngamskulrungroj, P.; Angkasekwinai, P. Contribution of Laccase Expression to Immune Response against Cryptococcus gattii Infection. Infect. Immun. 2020, 88, e00712-19. [Google Scholar] [CrossRef]
- Frazão, S.D.O.; de Sousa, H.R.; da Silva, L.G.; Folha, J.D.S.; Gorgonha, K.C.d.M.; de Oliveira, G.P.; Felipe, M.S.S.; Silva-Pereira, I.; Casadevall, A.; Nicola, A.M.; et al. Laccase Affects the Rate of Cryptococcus neoformans Nonlytic Exocytosis from Macrophages. MBio 2020, 11, e02085-20. [Google Scholar] [CrossRef]
- Wang, Y.; Casadevall, A. Susceptibility of Melanized and Nonmelanized Cryptococcus neoformans to Nitrogen- and Oxygen-Derived Oxidants. Infect. Immun. 1994, 62, 3004–3007. [Google Scholar] [CrossRef]
- Jacobson, E.S.; Tinnell, S.B. Antioxidant Function of Fungal Melanin. J. Bacteriol. 1993, 175, 7102–7104. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Aisen, P.; Casadevall, A. Cryptococcus neoformans Melanin and Virulence: Mechanism of Action. Infect. Immun. 1995, 63, 3131–3136. [Google Scholar] [CrossRef] [PubMed]
- Mednick, A.J.; Nosanchuk, J.D.; Casadevall, A. Melanization of Cryptococcus neoformans Affects Lung Inflammatory Responses during Cryptococcal Infection. Infect. Immun. 2005, 73, 2012–2019. [Google Scholar] [CrossRef] [PubMed]
- Doering, T.L.; Nosanchuk, J.D.; Roberts, W.K.; Casadevall, A. Melanin as a Potential Cryptococcal Defence against Microbicidal Proteins. Med. Mycol. 1999, 37, 175–181. [Google Scholar] [CrossRef]
- Tajima, K.; Yamanaka, D.; Ishibashi, K.I.; Adachi, Y.; Ohno, N. Solubilized Melanin Suppresses Macrophage Function. FEBS Open Bio 2019, 9, 791–800. [Google Scholar] [CrossRef]
- Huffnagle, G.B.; Chen, G.H.; Curtis, J.L.; McDonald, R.A.; Strieter, R.M.; Toews, G.B. Down-Regulation of the Afferent Phase of T Cell-Mediated Pulmonary Inflammation and Immunity by a High Melanin-Producing Strain of Cryptococcus neoformans. J. Immunol. 1995, 155, 3507–3516. [Google Scholar]
- Barluzzi, R.; Brozzetti, A.; Mariucci, G.; Tantucci, M.; Neglia, R.G.; Bistoni, F.; Blasi, E. Establishment of Protective Immunity against Cerebral Cryptococcosis by Means of an Avirulent, Non Melanogenic Cryptococcus neoformans Strain. J. Neuroimmunol. 2000, 109, 75–86. [Google Scholar] [CrossRef]
- Chen, S.C.A.; Wright, L.C.; Santangelo, R.T.; Muller, M.; Moran, V.R.; Kuchel, P.W.; Sorrell, T.C. Identification of Extracellular Phospholipase B, Lysophospholipase, and Acyltransferase Produced by Cryptococcus neoformans. Infect. Immun. 1997, 65, 405–411. [Google Scholar] [CrossRef]
- Chen, S.C.A.; Muller, M.; Zhou, J.Z.; Wright, L.C.; Sorrell, T.C. Phospholipase Activity in Cryptococcus neoformans: A New Virulence Factor? J. Infect. Dis. 1997, 175, 414–420. [Google Scholar] [CrossRef]
- Djordjevic, J.T. Role of Phospholipases in Fungal Fitness, Pathogenicity, and Drug Development—Lessons from Cryptococcus neoformans. Front. Microbiol. 2010, 1, 125. [Google Scholar] [CrossRef]
- Chayakulkeeree, M.; Johnston, S.A.; Oei, J.B.; Lev, S.; Williamson, P.R.; Wilson, C.F.; Zuo, X.; Leal, A.L.; Vainstein, M.H.; Meyer, W.; et al. SEC14 is a Specific Requirement for Secretion of Phospholipase B1 and Pathogenicity of Cryptococcus neoformans. Mol. Microbiol. 2011, 80, 1088–1101. [Google Scholar] [CrossRef]
- Cox, G.M.; McDade, H.C.; Chen, S.C.A.; Tucker, S.C.; Gottfredsson, M.; Wright, L.C.; Sorrell, T.C.; Eidich, S.D.; Casadevall, A.; Ghannoum, M.A.; et al. Extracellular Phospholipase Activity Is a Virulence Factor for Cryptococcus neoformans. Mol. Microbiol. 2001, 39, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Noverr, M.C.; Cox, G.M.; Perfect, J.R.; Huffnagle, G.B. Role of PLB1 in Pulmonary Inflammation and Cryptococcal Eicosanoid Production. Infect. Immun. 2003, 71, 1538–1547. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.J.; Li, Z.; Hughes, W.S.; Djordjevic, J.T.; Nielsen, K.; May, R.C. Cryptococcal Phospholipase B1 Is Required for Intracellular Proliferation and Control of Titan Cell Morphology during Macrophage Infection. Infect. Immun. 2015, 83, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Osterholzer, J.J.; Surana, R.; Milam, J.E.; Montano, G.T.; Chen, G.H.; Sonstein, J.; Curtis, J.L.; Huffnagle, G.B.; Toews, G.B.; Olszewski, M.A. Cryptococcal Urease Promotes the Accumulation of Immature Dendritic Cells and a Non-Protective T2 Immune Response within the Lung. Am. J. Pathol. 2009, 174, 932–943. [Google Scholar] [CrossRef] [PubMed]
- Banks, I.R.; Specht, C.A.; Donlin, M.J.; Gerik, K.J.; Levitz, S.M.; Lodge, J.K. A Chitin Synthase and Its Regulator Protein Are Critical for Chitosan Production and Growth of the Fungal Pathogen Cryptococcus neoformans. Eukaryot. Cell 2005, 4, 1902–1912. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Da Silva, C.A.; Lee, J.Y.; Hartl, D.; Elias, J.A. Chitin Regulation of Immune Responses: An Old Molecule with New Roles. Curr. Opin. Immunol. 2008, 20, 684–689. [Google Scholar] [CrossRef] [PubMed]
- Gordon-Thomson, C.; Kumari, A.; Tomkins, L.; Holford, P.; Djordjevic, J.T.; Wright, L.C.; Sorrell, T.C.; Moore, G.P.M. Chitotriosidase and Gene Therapy for Fungal Infections. Cell. Mol. Life Sci. 2009, 66, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Gorzelanny, C.; Pöppelmann, B.; Pappelbaum, K.; Moerschbacher, B.M.; Schneider, S.W. Human Macrophage Activation Triggered by Chitotriosidase-Mediated Chitin and Chitosan Degradation. Biomaterials 2010, 31, 8556–8563. [Google Scholar] [CrossRef] [PubMed]
- Hole, C.R.; Lam, W.C.; Upadhya, R.; Lodge, J.K. Cryptococcus neoformans Chitin Synthase 3 Plays a Critical Role in Dampening Host Inflammatory Responses. MBio 2020, 11, e03373-19. [Google Scholar] [CrossRef]
- Liu, O.W.; Chun, C.D.; Chow, E.D.; Chen, C.; Madhani, H.D.; Noble, S.M. Systematic Genetic Analysis of Virulence in the Human Fungal Pathogen Cryptococcus neoformans. Cell 2008, 135, 174–188. [Google Scholar] [CrossRef]
- Dang, E.V.; Lei, S.; Radkov, A.; Volk, R.F.; Zaro, B.W.; Madhani, H.D. Secreted Fungal Virulence Effector Triggers Allergic Inflammation via TLR4. Nature 2022, 608, 161–167. [Google Scholar] [CrossRef]
- Coker, R.J. Cryptococcal Infection in AIDS. Int. J. STD AIDS 1992, 3, 168–172. [Google Scholar] [CrossRef] [PubMed]
- Mocsny, N. Cryptococcal Meningitis in Patients with AIDS. J. Neurosci. Nurs. 1992, 24, 265–268. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).