Variation in Fungal Community in Grapevine (Vitis vinifera) Nursery Stock Depends on Nursery, Variety and Rootstock
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Necrosis Evaluation
2.3. Fungal Isolates
2.4. Library Preparation for Metabarcoding
2.5. Quality Control and Bioinformatic Analysis of Metabarcoding Data
2.6. Statistical Analysis
2.6.1. Necrosis
2.6.2. Mapping Fungal Communities
2.7. Indicator Species
3. Results
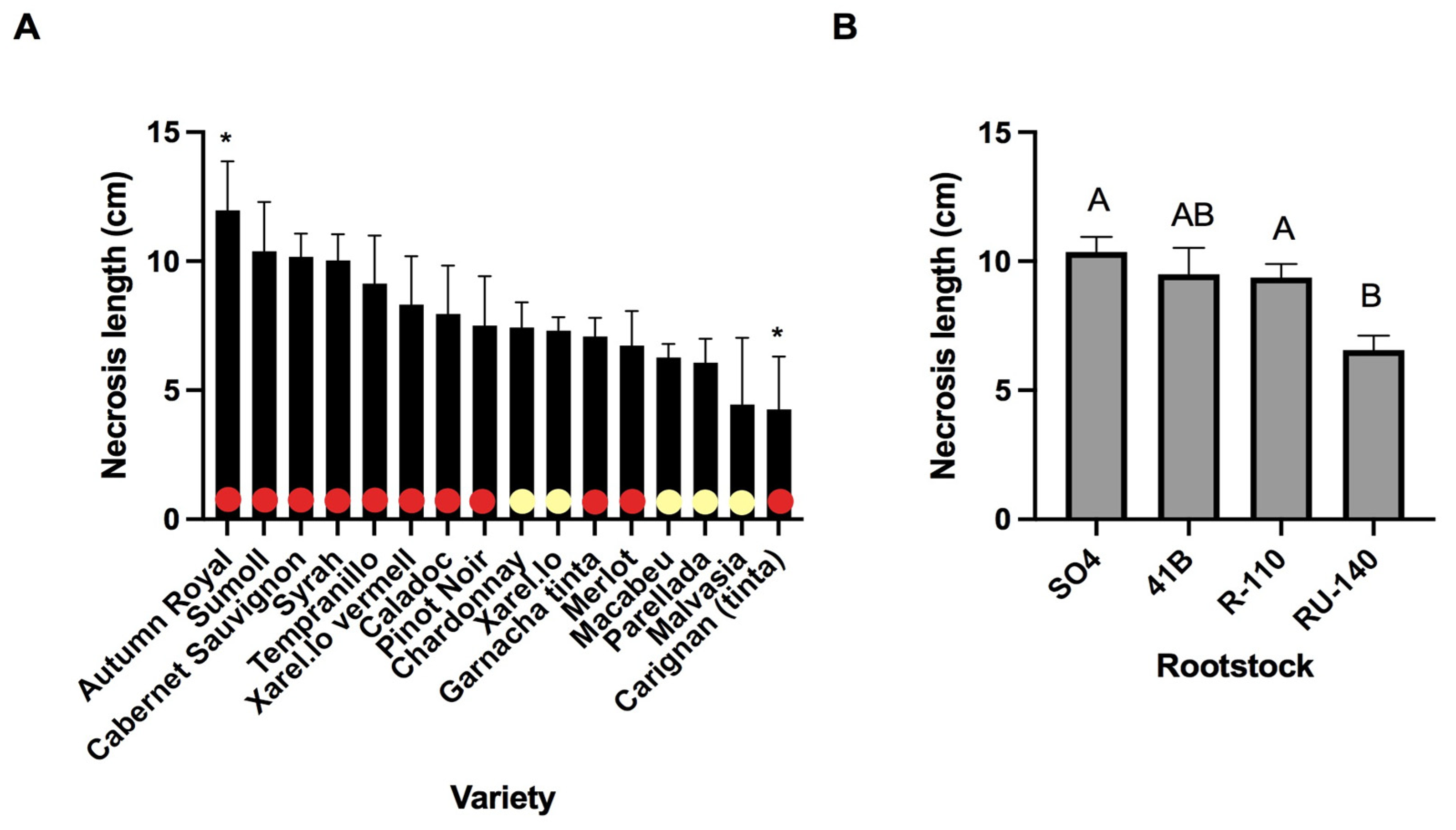
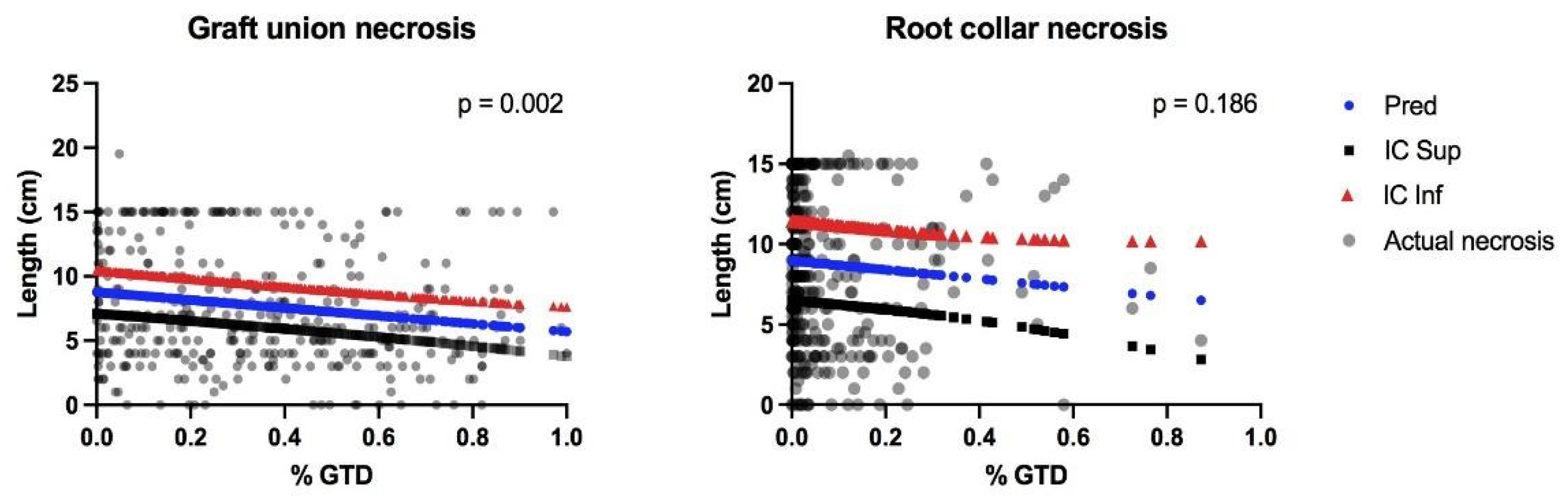
3.1. Plant Attributes and Conditions Contributing to Necrosis
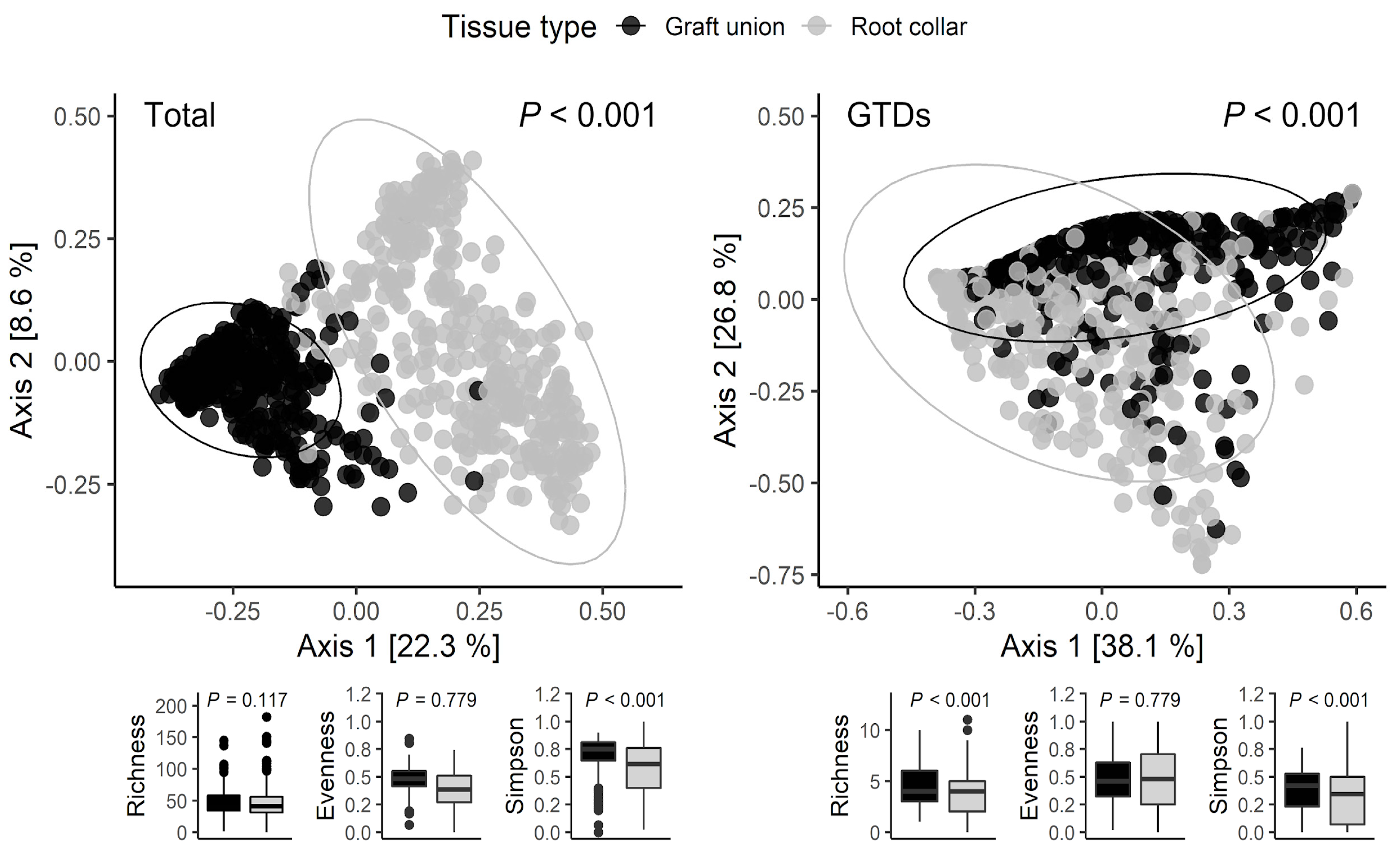
3.2. Fungal Community Distribution
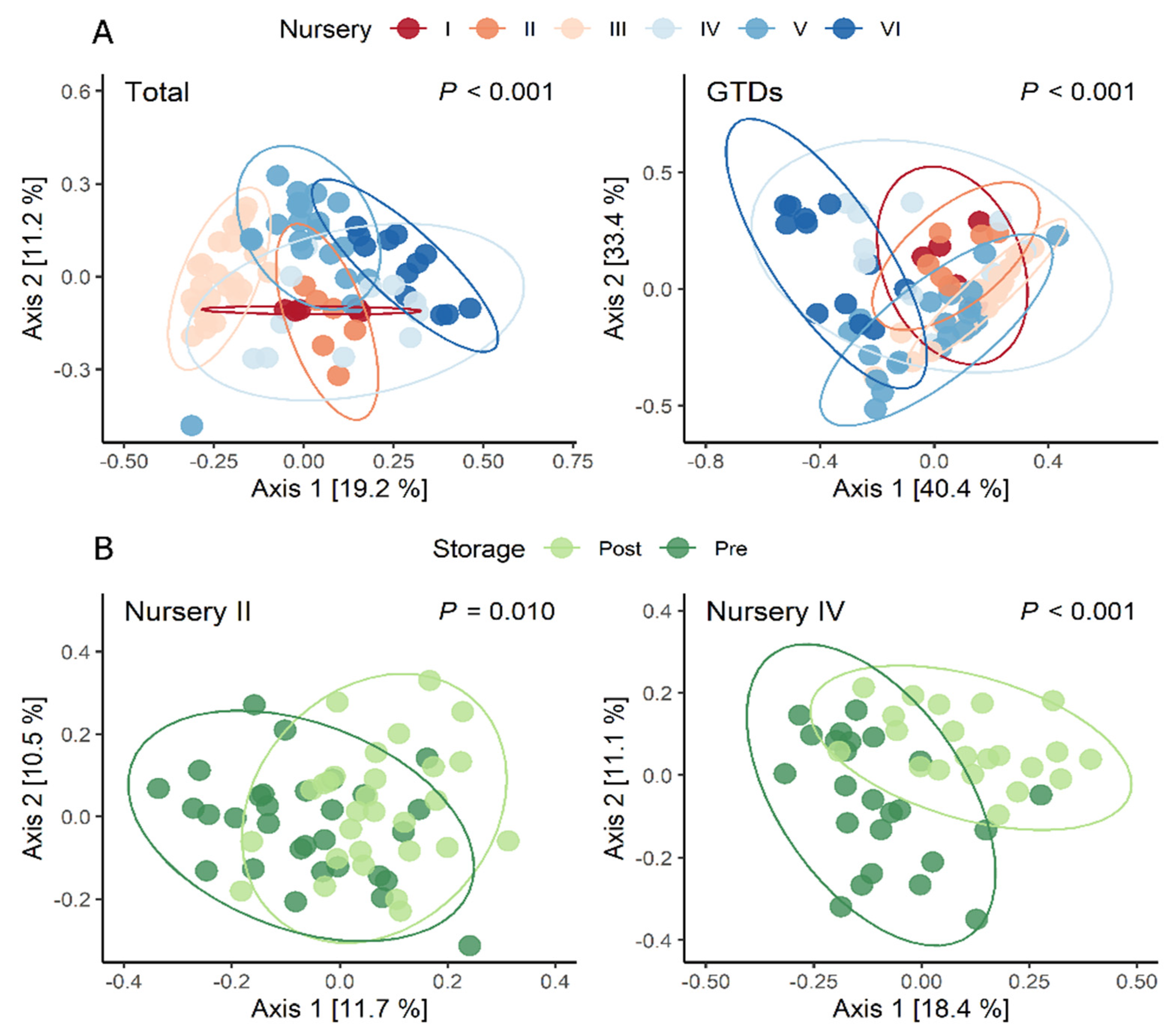
3.3. Fungal Community Distribution within and between Nurseries and Varieties
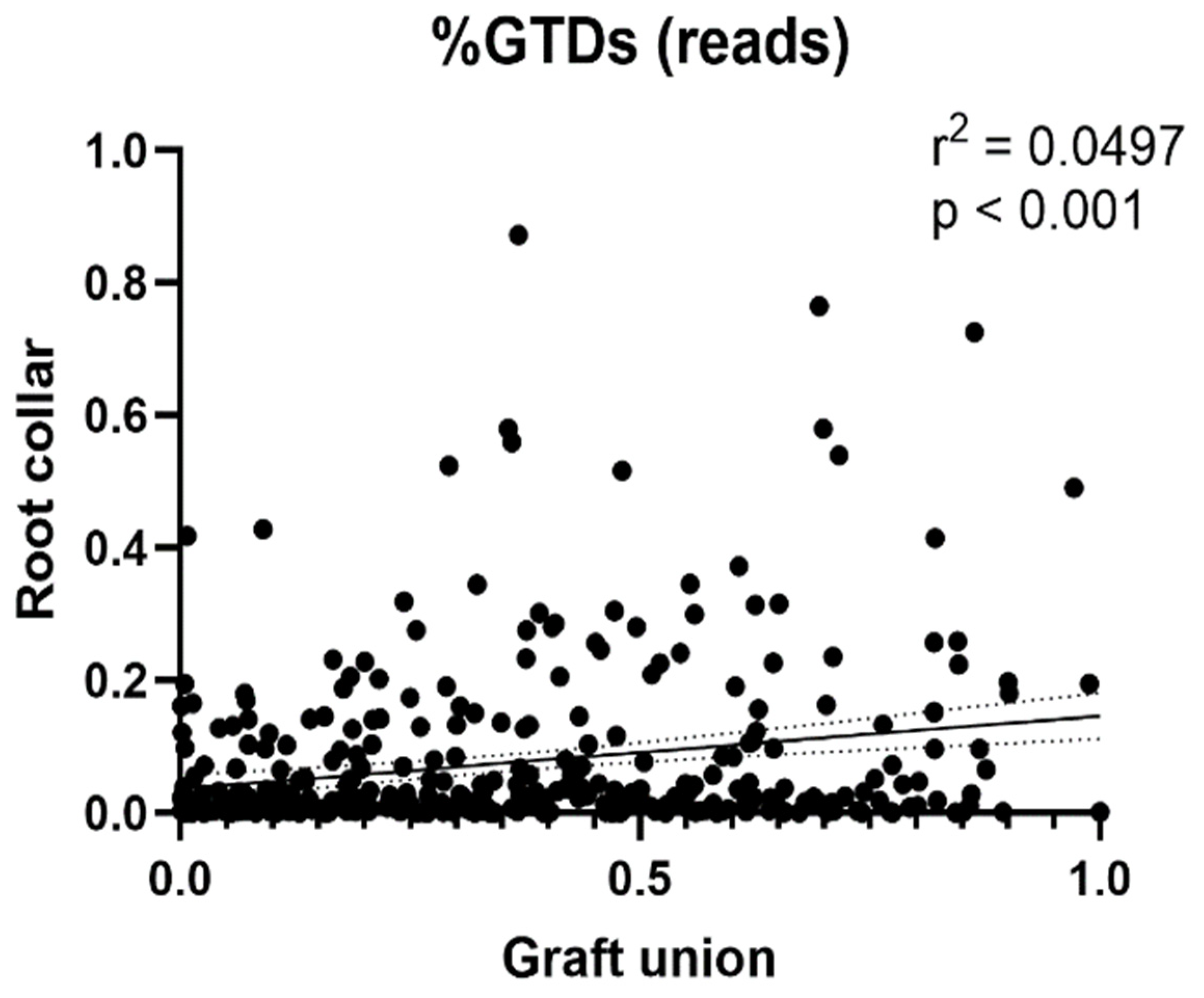
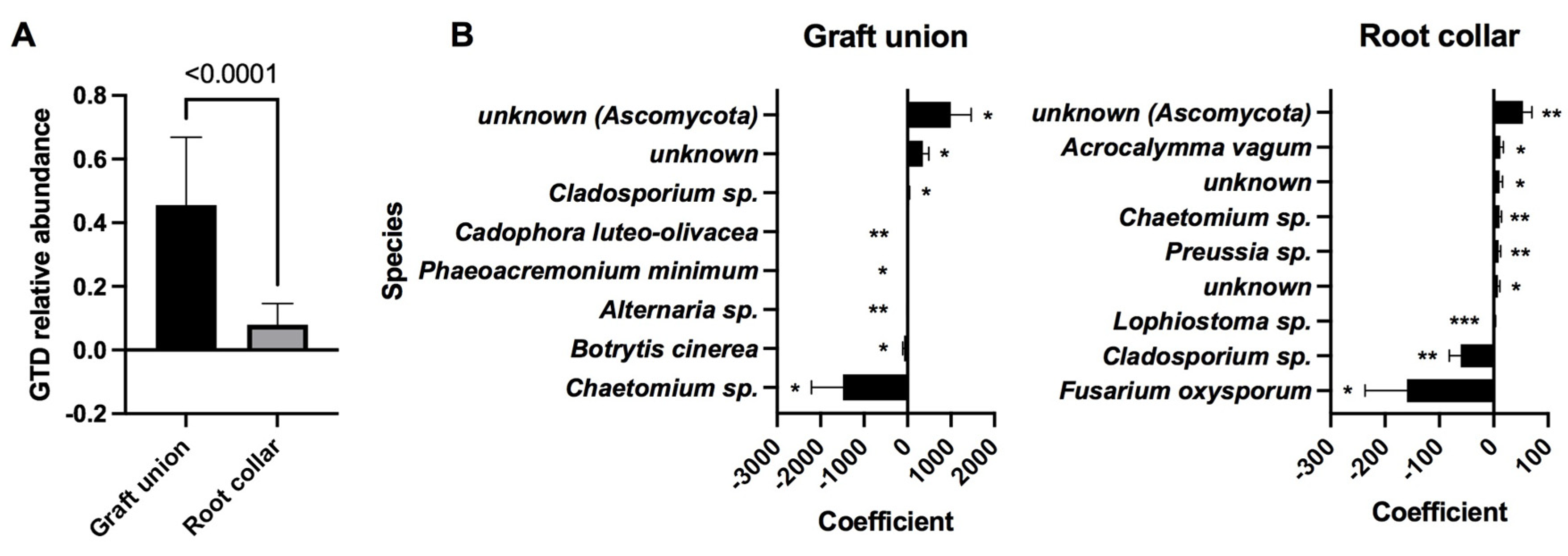
3.4. Relative Abundance of GTD Fungal Isolates and OTU Clusters
3.5. Indicator Species Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gramaje, D.; Úrbez-Torres, J.R.; Sosnowski, M.R. Managing grapevine trunk diseases with respect to etiology and epidemiology: Current strategies and future prospects. Plant Dis. 2018, 102, 12–39. [Google Scholar] [CrossRef]
- Gramaje, D.; Armengol, J. Fungal Trunk Pathogens in the Grapevine Propagation Process: Potential Inoculum Sources, Detection, Identification, and Management Strategies. Plant Dis. 2011, 95, 1040–1055. [Google Scholar] [CrossRef]
- Waite, H.; Whitelaw-Weckert, M.; Torley, P. Grapevine propagation: Principles and methods for the production of high-quality grapevine planting material. N. Z. J. Crop Hortic. Sci. 2014, 43, 144–161. [Google Scholar] [CrossRef]
- Stamp, J.A. The contribution of imperfections in nursery stock to the decline of young vines in California. Phytopathol. Mediterr. 2001, 40, S369–S375. [Google Scholar]
- Waite, H.; Morton, L. Hot Water Treatment, Trunk Diseases and Other Critical Factors in the Production of High-Quality Grapevine Planting Material. Phytopathol. Mediterr. 2007, 46, 5–17. [Google Scholar] [CrossRef]
- Decoin, M. Grapevine products: News on withdrawals and restrictions. Phytoma 2001, 543, 28–33. [Google Scholar]
- US EPA. The Montreal Amendment (1997) to the Montreal Protocol Agreement. 1987. Available online: https://ozone.unep.org/treaties/montreal-protocol/amendments/montreal-amendment-1997-amendment-montreal-protocol-agreed (accessed on 28 June 2021).
- Mondello, V.; Songy, A.; Battiston, E.; Pinto, C.; Coppin, C.; Trotel-Aziz, P.; Clement, C.; Mugnai, L.; Fontaine, F. Grapevine Trunk Diseases: A Review of Fifteen Years of Trials for Their Control with Chemicals and Biocontrol Agents. Plant Dis. 2018, 102, 1189–1217. [Google Scholar] [CrossRef] [PubMed]
- DelFrari, G.; Gobbi, A.; Aggerbeck, M.R.; Oliveira, H.; Hansen, L.H.; Ferreira, R.B. Characterization of the Wood Mycobiome of Vitis vinifera in a Vineyard Affected by Esca. Spatial Distribution of Fungal Communities and Their Putative Relation With Leaf Symptoms. Front. Plant Sci. 2019, 10, 910. [Google Scholar] [CrossRef] [PubMed]
- Pinto, C.; Gomes, A.C. Vitis vinifera microbiome: From basic research to technological development. BioControl 2016, 61, 243–256. [Google Scholar] [CrossRef]
- Bertsch, C.; Ramirez-Suero, M.; Magnin-Robert, M.; Larignon, P.; Chong, J.; Abou-Mansour, E.; Spagnolo, A.; Clement, C.; Fontaine, F. Grapevine trunk diseases: Complex and still poorly understood. Plant Pathol. 2012, 12, 1–23. [Google Scholar] [CrossRef]
- Mugnai, L.; Graniti, A.; Surico, G. Esca (Black Measles) and Brown Wood-Streaking: Two Old and Elusive Diseases of Grapevines. Plant Dis. 1999, 83, 404–418. [Google Scholar] [CrossRef]
- Larignon, P. Maladies Cryptogamiques du Bois de la Vigne: Symptomatologie et Agents Pathogènes, 2nd ed.; Institut Francais de la Vigne et du Vin: Le Grau-du-Roi, France, 2016. [Google Scholar]
- Bruno, G.; Sparapano, L. Effects of three esca-associated fungi on Vitis vinifera L.: V. Changes in the chemical and biological profile of xylem sap from diseased cv. Sangiovese vines. Physiol. Mol. Plant Pathol. 2007, 71, 210–229. [Google Scholar] [CrossRef]
- Cloete, M.; Fischer, M.; Mostert, L.; Halleen, F. A novel Fomitiporia species associated with esca on grapevine in South Africa. Mycol. Prog. 2014, 13, 303–311. [Google Scholar] [CrossRef]
- Diaz, G.A.; Latorre, B.A. Efficacy of paste and liquid fungicide formulations to protect pruning wounds against pathogens associated with grapevine trunk diseases in Chile. Crop Prot. 2013, 46, 106–112. [Google Scholar] [CrossRef]
- Úrbez-Torres, J.R.; Peduto, F.; Smith, R.J.; Gubler, W. The status of Botryosphaeriaceae species infecting grapevines. Phytopathol. Mediterr. 2011, 50, S5–S45. [Google Scholar]
- Úrbez-Torres, J.R.; Peduto, F.; Smith, R.J.; Gubler, W. Phomopsis Dieback: A Grapevine Trunk Disease Caused by Phomopsis viticola in California. Plant Dis. 2013, 97, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Larignon, P.; Fontaine, F.; Farine, S.; Clement, C.; Bertsch, C. Esca et Black Dead Arm: Deux acteurs majeurs des maladies du bois chez la Vigne. C. R. Biol. 2009, 332, 765–783. [Google Scholar] [CrossRef] [PubMed]
- Catalunya, G.D. Catalan Wines: Opening up to New Markets. Available online: http://catalonia.com/industries-in-catalonia/sectors/food-industries/catalan-wines.jsp (accessed on 20 June 2021).
- Prodeca. The Catalan Wine Sector. Available online: https://www.prodeca.cat/en/sectors/the-catalan-wine-sector (accessed on 28 June 2021).
- Barber, V. Cuanto Cuesta Plantar una Hectárea de Viñedo en España? Plantación Viñedo 2014. Available online: https://www.vitivinicultura.net/cuanto-cuesta-plantar-una-hectarea-de.html (accessed on 25 June 2021).
- Surico, G.; Mugnai, L.; Marchi, G. Older and more recent observations on esca: A critical overview. Phytopathol. Mediterr. 2006, 45, S68–S86. [Google Scholar]
- Guan, X.; Essakhi, S.; Laloue, H.; Nick, P.; Bertsch, C.; Chong, J. Mining new resources for grape resistance against Botryosphaeriaceae: A focus on Vitis vinifera subsp. sylvestris. Plant Pathol. 2016, 65, 273–284. [Google Scholar] [CrossRef]
- Quaglia, M.; Covarelli, L.; Zazzerini, A. Epidemiological survey on esca disease in Umbria, central Italy. Phytopathol. Mediterr. 2009, 48, 84–91. [Google Scholar]
- Gubler, W.D.; Baumgartner, K.; Browne, G.T.; Eskalen, A.; Rooney Latham, S.; Petit, E.; Bayramian, L.A. Root diseases of grapevines in California and their control. Australas. Plant Pathol. 2004, 33, 157–165. [Google Scholar] [CrossRef]
- Gramaje, D.; Alaniz, S.; Abad-Campos, P.; Garcia-Jimenez, J.; Armengol, J. Effect of hot-water treatments in vitro on conidial germination and mycelial growth of grapevine trunk pathogens. Ann. Appl. Biol. 2010, 156, 231–241. [Google Scholar] [CrossRef]
- Marsais, P. L’Esca. Rev. Vitic. 1923, 59, 8–14. [Google Scholar]
- Murolo, S.; Romanazzi, G. Effects of grapevine cultivar, rootstock and clone on esca disease. Australas. Plant Pathol. 2014, 43, 215–221. [Google Scholar] [CrossRef]
- Ferreira, J.H.S.; van Wyk, P.S.; Calitz, F.J. Slow dieback of grapevine in South Africa: Stress-related disposition of young vines for infection by Phaeoacremonium chlamydosporum. S. Afr. J. Enol. Vitic. 1999, 20, 43–46. [Google Scholar]
- Peay, K.; Kennedy, P.G.; Talbot, J. Dimensions of biodiversity in the Earth mycobiome. Nature 2016, 14, 434–447. [Google Scholar] [CrossRef] [PubMed]
- Morgan, H.H.; du Toit, M.; Setati, M.E. The grapevine and wine microbiome: Insights from high-throughput amplicon sequencing. Front. Microbiol. 2017, 8, 820. [Google Scholar] [CrossRef] [PubMed]
- Jayawardena, R.S.; Purahong, W.; Zhang, W.; Wubet, T.; Li, X.; Liu, M.; Zhao, W.; Hyde, K.D.; Liu, J.; Yan, J. Biodiversity of fungi on Vitis vinifera L. revealed by traditional and high-resolution culture-independent approaches. Fungal Divers. 2018, 90, 1–84. [Google Scholar] [CrossRef]
- Dissanayake, A.J.; Purahong, W.; Wubet, T.; Hyde, K.D.; Zhang, W.; Zu, H.; Zhang, G.; Fu, C.; Liu, M.; Xing, Q.; et al. Direct comparison of culture-dependent and culture-independent molecular approaches reveal the diversity of fungal endophytic communities in stems of grapevine (Vitis vinifera). Fungal Divers. 2018, 90, 85–107. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Thorngate, J.H.; Richardson, P.M.; Mills, D.A. Microbial biogeography of wine grapes is conditioned by cultivar, vintage and climate. Proc. Natl. Acad. Sci. USA 2014, a111, E139–E148. [Google Scholar] [CrossRef]
- Santamaría, O.; Smith, D.R.; Stanosz, G.R. Interaction between Diplodia pinea or Diplodia scrobiculata and fungal endophytes isolated from pine shoots. Can. J. For. Res. 2012, 42, 1819–1826. [Google Scholar] [CrossRef]
- Oliva, J.; Messal, M.; Wendt, L.; Elfstrand, M. Quantitative interactions between the biocontrol fungus Phlebiopsis gigantea, the forest pathogen Heterobasidion annosum and the fungal community inhabiting Norway spruce stumps. For. Ecol. Manag. 2017, 402, 253–264. [Google Scholar] [CrossRef]
- Ihrmark, K.; Bödeker, I.T.M.; Cruz-Martinez, K.; Friberg, H.; Kubartova, A.; Schenck, J.; Strid, Y.; Stenlid, J.; Brandström-Durling, M.; Clemmensen, K.E.; et al. New primers to amplify the fungal ITS2 region—Evaluation by 454-sequencing of artificial and natural communities. FEMS Microbiol. 2012, 82, 666–677. [Google Scholar] [CrossRef]
- Somervuo, P.; Koskela, S.; Pennanen, J.; Nilsson, R.H.; Ovaskainen, O. Unbiased probabilistic taxonomic classification for DNA barcoding. Bioinformatics 2016, 32, 2920–2927. [Google Scholar] [CrossRef]
- Abarenkov, K.; Somervuo, P.; Nilsson, R.H. Protax-fungi: A web-based tool for probabilistic taxonomic placement of fungal internal transcribed spacer sequences. N. Phytol. 2018, 220, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Abrego, N.; Norros, V.; Halme, P.; Somervuo, P.; Ali-Kovero, H.; Ovaskainen, O. Give me a sample of air and I will tell which species are found from your region: Molecular identification of fungi from airborne spore samples. Mol. Ecol. Resour. 2018, 18, 511–524. [Google Scholar] [CrossRef] [PubMed]
- Huson, D.H.; Auch, A.F.; Qi, J.; Schuster, S.C. MEGAN analysis of metagenomic data. Genome Res. 2007, 17, 377–386. [Google Scholar] [CrossRef]
- Frøslev, T.; Kjøller, R.; Bruun, H.H. Algorithm for post-clustering curation of DNA amplicon data yields reliable biodiversity estimates. Nat. Commun. 2017, 8, 1188. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. 2020. Available online: https://www.R-project.org/ (accessed on 20 June 2021).
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGLinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. vegan: Community Ecology Package. 2018. Available online: https://CRAN.R-project.org/package=vegan (accessed on 20 June 2021).
- De Cáceres, M.; Legendre, P. Associations between species and groups of sites: Indices and statistical inference. Ecology 2009, 90, 3566–3574. [Google Scholar] [CrossRef]
- Pantidou, M.E. Fungus-Host Index for Greece; Benaki Phytopathological Institute: Athens, Greece, 1973; 382p. [Google Scholar]
- Benavides, P.G.; Maistrello, L.; Martin Zamorano, P.; Ocete Perez, C.A.; Ocete Rubio, R. Biodiversity of pathogenic wood fungi isolated from Xylotrechus arvicola (Olivier) galleries in vine shoots. J. Int. Des Sci. Vigne Vin 2013, 47, 73–81. [Google Scholar] [CrossRef]
- Summerbell, R.C.; Gueidan, C.; Schroers, H.-J.; de Hoog, G.S.; Starink, M.; Arocha Rosete, Y.; Guarro, J.; Scott, J.A. Acremonium phylogenetic overview and revision of Gliomastix, Sarocladium, and Trichothecium. Stud. Mycol. 2011, 68, 139–162. [Google Scholar] [CrossRef]
- Gatica, M.; Cesari, C.; Magnin, S.; Dupont, J. Phaeoacremonium species and Phaeomoniella chlamydospora in vines showing “hoja de malvón” and young vine decline symptoms in Argentina. Phytopathol. Mediterr. 2001, 40, S317–S324. [Google Scholar]
- Halleen, F.; Crous, R.W.; Petrin, O. Fungi associated with healthy grapevine cuttings in nurseries, with special reference to pathogens involved in the decline of young vines. Australas. Plant Pathol. 2003, 32, 47–52. [Google Scholar] [CrossRef]
- Luque, J.; Martos, S.; Aroca, A.; Raposo, R.; Garcia-Figueres, F. Symptoms and fungi associated with declining mature grapevine plants in northeast Spain. J. Plant Pathol. 2009, 91, 381–390. [Google Scholar]
- Mohammadi, H.; Banihashemi, Z. First report of Phaeoacremonium inflatipes and Phaeoacremonium mortoniae associated with grapevine Petri disease in Iran. J. Agric. Sci. Technol. 2012, 14, 1405–1414. [Google Scholar]
- Mondello, V.; Lo Piccolo, S.; Conigliaro, G.; Alfonzo, A.; Torta, L.; Burruano, S. First report of Neofusiccoccum vitifusiforme and presence of other Botryosphaeriaceae species associated with Botryosphaeria dieback of grapevine in Sicily (Italy). Phytopathol. Mediterr. 2013, 52, 388–396. [Google Scholar]
- Oh, S.Y.; Nam, K.W.; Yoon, D.H. Identification of Acremonium acutatum and Trichothecium roseum isolated from Grape with White Stain Symptom in Korea. Mycobiology 2014, 42, 269–273. [Google Scholar] [CrossRef]
- Phillips, A.J. L, Botryosphaeria dothidea and Other Fungi Associated with Excoriose and Dieback of Grapevines in Portugal. J. Phytopathol. Mediterr. 1998, 146, 327–332. [Google Scholar] [CrossRef]
- Phillips, A.J.L. Excoriose, cane blight and related diseases of grapevines: A taxonomic review of the pathogens. Phytopathol. Mediterr. 2000, 39, 341–356. [Google Scholar]
- Slippers, B.; Smit, W.A.; Crous, P.W.; Coutinho, T.A.; Wingfield, B.D.; Wingfield, M.J. Taxonomy, phylogeny and identification of Botryosphaeriaceae associated with pome and stone fruit trees in South Africa and other regions of the world. Plant Pathol. 2007, 56, 128–139. [Google Scholar] [CrossRef]
- Larignon, P.; Dubos, B. The villainy of Black Dead Arm. Wines Vines 2001, 82, 86–89. [Google Scholar]
- van Niekerk, J.M.; Fourie, P.H.; Halleen, F.; Crous, P.W. Botryosphaeria spp. as grapevine trunk disease pathogens. Phytopathol. Mediterr. 2006, 45, S43–S54. [Google Scholar]
- Kobayashi, T. Index of Fungi Inhabiting Woody Plants in Japan; Zenkoku-Noson-Kyoiku Kyokai Publishing: Tokyo, Japan, 2007. [Google Scholar]
- Pitt, W.M.; Huang, R.; Steel, C.C.; Savocchia, S. Identification, distribution and current taxonomy of Botryosphaeriaceae species associated with grapevine decline in New South Wales and South Australia. Aust. J. Grape Wine Res. 2010, 16, 258–271. [Google Scholar] [CrossRef]
- Qiu, Y.; Steel, C.C.; Ash, G.J.; Savocchia, S. Survey of Botryosphaeriaceae associated with grapevine decline in the Hunter Valley and Mudgee grape growing regions of New South Wales. Australas. Plant. Pathol. 2011, 40, 1–11. [Google Scholar] [CrossRef]
- Úrbez-Torres, J.R.; Peduto, F.; Striegler, R.K.; Urrea-Romero, K.E.; Rupe, J.C.; Cartwright, R.D.; Gubler, W.D. Characterization of fungal pathogens associated with grapevine trunk diseases in Arkansas and Missouri. Fungal Divers. 2012, 52, 169–189. [Google Scholar] [CrossRef]
- Abreo, E.; Lupo, S.; Bettucci, L. Fungal community of grapevine trunk diseases: A continuum of symptoms? Sydowia 2012, 64, 1–12. [Google Scholar]
- Arzanlou, M.; Moshari, S.; Bakhshi, M.; Khodaie, S. Botryosphaeria dothidea associated with grapevine decline disease in Iran. Australas. Plant Dis. Notes 2012, 7, 197–200. [Google Scholar] [CrossRef][Green Version]
- Baskarathevan, J.; Jaspers, M.V.; Jones, E.E.; Ridgway, H.J. Incidence and distribution of botryosphaeriaceous species in New Zealand vineyards. Eur. J. Plant Pathol 2012, 132, 549–560. [Google Scholar] [CrossRef]
- Yan, J.; Xie, Y.; Yao, S.; Wang, Z.; Li, X. Characterization of Botryosphaeria dothidea, the causal agent of grapevine canker in China. Australas 2012, 41, 351–357. [Google Scholar] [CrossRef]
- Akgul, D.S.; Savas, N.G.; Eskalen, A. First report of wood canker caused by Botryosphaeria dothidea, Diplodia seriata, Neofusicoccum parvum, and Lasiodiplodia theobromae on grapevine in Turkey. Plant Dis. 2014, 98, 568. [Google Scholar] [CrossRef]
- Chebil, S.; Fersi, R.; Yakoub, A.; Chenenaoui, S.; Chattaoui, M.; Melki, I.; Zemni, H.; Rhouma, A.; Durante, G.; Zacchi, E.; et al. First report of Botryosphaeria dothidea, Diplodia seriata, and Neofusicoccum luteum associated with canker and dieback of grapevines in Tunisia. Plant Dis. 2014, 98, 420. [Google Scholar] [CrossRef] [PubMed]
- Carlucci, A.; Cibelli, F.; Lops, F.; Raimondo, M.L. Characterization of Botryosphaeriaceae species as causal agents of trunk disease on Grapevines. Plant Dis. 2015, 99, 1678–1688. [Google Scholar] [CrossRef]
- Fischer, M.; Schneider, P.; Kraus, C.; Molnar, M.; Dubois, C.; D’Aguiar, D.; Haag, N. Grapevine trunk disease in German viticulture: Occurence of lesser known fungi and first report of Phaeoacremonium viticola and P. fraxinopennsylvanicum. Vitis 2016, 55, 145–156. [Google Scholar]
- Fourie, P.H.; Halleen, F. Investigation on the occurrence of Phaeomoniella chlamydospora in canes of rootstock mother vines. Australas 2002, 31, 425–426. [Google Scholar] [CrossRef]
- Giménez-Jaime, A.; Aroca, A.; Raposo, R.; García-Jiménez, J.; Armengol, J. Occurrence of fungal pathogens associated with grapevine nurseries and the decline of young vines in Spain. J. Phytopathol. 2006, 154, 598–602. [Google Scholar] [CrossRef]
- Martin, M.T.; Cobos, R. Identification of fungi associated with grapevine decline in Castilla y Leon (Spain). Phytopathol. Mediterr. 2007, 46, 18–25. [Google Scholar]
- Sosnowski, M.R.; Lardner, R.; Wicks, T.J.; Scott, E.S. The influence of grapevine cultivar and isolate of Eutypa lata on wood and foliar symptoms. Plant Dis. 2007, 91, 924–931. [Google Scholar] [CrossRef] [PubMed]
- Casieri, L.; Hofstetter, V.; Viret, O.; Gindro, K. Fungal communities living in the wood of different cultivars of young Vitis vinifera plants. Phytopathol. Mediterr. 2009, 48, 73–83. [Google Scholar]
- Nakaune, R.; Tatsuki, M.; Matsumoto, H.; Ikoma, Y. First report of a new postharvest disease of grape caused by Cadophora luteo-olivacea. J. Gen. Plant Pathol. 2016, 82, 116–119. [Google Scholar] [CrossRef]
- Travadon, R.; Lawrence, D.P.; Rooney-Latham, S.; Gubler, W.D.; Wilcox, W.F.; Rolshausen, P.E.; Baumgartner, K. Cadophora species associated with wood-decay of grapevine in North America. Fungal Biol. 2015, 119, 53–66. [Google Scholar] [CrossRef]
- Blanchette, R.A.; Held, B.W.; Arenz, B.E.; Jurgens, J.A.; Baltes, N.J.; Duncan, S.M.; Farrell, R.L. An Antarctic Hot Spot for Fungi at Shackleton’s Historic Hut on Cape Royds. Microb. Ecol. 2010, 60, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Abreo, E.; Martinez, S.; Bettucci, L.; Lupo, S. Morphological and molecular characterisation of Campylocarpon and Cylindrocarpon spp. associated with black foot disease of grapevines in Uruguay. Australas. Plant Pathol. 2010, 39, 446–452. [Google Scholar] [CrossRef]
- Petit, E.; Barriault, E.; Baumgartner, K.; Wilcox, W.F.; Rolshausen, P.E. Cylindrocarpon Species Associated with Black-Foot of Grapevine in Northeastern United States and Southeastern Canada. Am. J. Enol. Vit. 2011, 62, 177–183. [Google Scholar] [CrossRef]
- Correia, K.C.; Camara, M.P.S.; Barbosa, M.A.G.; Sales, R.; Agustí-Brisach, C.; Gramaje, D.; León, M.; García-Jiménez, J.; Abad-Campos, P.; Armengol, J.; et al. Fungal trunk pathogens associated with table grape decline in Northeastern Brazil. Phytopathol. Mediterr. 2013, 52, 380–387. [Google Scholar]
- Akgul, D.S.; Savas, N.G.; Onder, S.; Ozben, S.; Kaymak, S. First report of Campylocarpon fasciculare causing black foot disease of grapevine in Turkey. Plant Dis. 2014, 98, 1277. [Google Scholar] [CrossRef] [PubMed]
- Úrbez-Torres, J.R.; Haag, P.; Bowen, P.; O’Gorman, D.T. Grapevine trunk diseases in British Columbia: Incidence and characterization of the fungal pathogens associated with black foot disease of Grapevine. Plant Dis. 2014, 98, 456–468. [Google Scholar] [CrossRef]
- Carlucci, A.; Lops, F.; Mostert, L.; Halleen, F.; Raimondo, M.L. Occurrence fungi causing black foot on young grapevines and nursery rootstock plants in Italy. Phytopathol. Mediterr. 2017, 56, 10–39. [Google Scholar]
- González, C.D.; Chaverri, P. Corinectria, a new genus to accommodate Neonectria fuckeliana and C. constricta sp. nov. from Pinus radiata in Chile. Mycol. Prog. 2017, 16, 1015–1027. [Google Scholar] [CrossRef]
- Lombard, L.; Van Der Merwe, N.A.; Groenewald, J.Z.; Crous, P.W. Lineages in Nectriaceae: Re-evaluating the generic status of Ilyonectria and allied genera. Phytopathol. Mediterr. 2014, 53, 515–532. [Google Scholar]
- Fontaine, F.; Gramaje, D.; Armengol, J.; Smart, R.; Nagy, Z.A.; Borgo, M.; Rego, C.; Corio-Costet, M.-F. Grapevine Trunk Diseases: A Review; International Organisation of Vine and Wine: Le Grau-du-Roi, France, 2016; Volume 1, pp. 1–24. [Google Scholar]
- Whitelaw-Weckert, M.A.; Nair, N.G.; Lamont, R.; Alonso, M.; Priest, M.J.; Huang, R. Root infection of Vitis vinifera by Cylindrocarpon liriodendri in Australia. Australas. Plant Pathol. 2007, 36, 403–406. [Google Scholar] [CrossRef]
- Choueiri, E.; Jreijiri, F.; El Amil, R.; Chlela, P.; Bugaret, Y.; Liminana, J.M.; Mayet, V.; Lecomte, P. First report of black foot disease associated with Cylindrocarpon sp. in Lebanon. J. Plant Pathol. 2009, 91, 231–240. [Google Scholar]
- Agustí-Brisach, C.; Cabral, A.; González-Domínguez, E.; Pérez-Sierra, A.; León, M.; Abad-Campos, P.; García-Jiménez, J.; Oliveira, H.; Armengol, J. Characterization of Cylindrodendrum, Dactylonectria and Ilyonectria isolates associated with loquat decline in Spain, with description of Cylindrodendrum alicantinum sp. nov. Eur. J. Plant Pathol. 2016, 145, 103–118. [Google Scholar] [CrossRef]
- Guarnaccia, V.; Groenewald, J.Z.; Woodhall, J.; Armengol, J.; Cinelli, T.; Eichmeier, A.; Ezra, D.; Fontaine, F.; Gramaje, D.; Gutierrez-Aguirregabiria, A.; et al. Diaporthe diversity and pathogenicity revealed from a broad survey of grapevine diseases in Europe. Persoonia 2018, 40, 135–153. [Google Scholar] [CrossRef] [PubMed]
- Luongo, L.; Santori, A.; Riccioni, L.; Belisario, A. Phomopsis sp. associated with post-harvest fruit rot of kiwifruit in Italy. J. Plant Pathol. 2011, 93, 205–209. [Google Scholar]
- Udayanga, D.; Castlebury, L.A.; Rossman, A.Y.; Chukeatirote, E.; Hyde, K.D. Insights into the genus Diaporthe: Phylogenetic species delimitation in the D. eres species complex. Fungal Divers. 2014, 67, 203–229. [Google Scholar] [CrossRef]
- Udayanga, D.; Castlebury, L.A.; Rossman, A.Y.; Hyde, K.D. Species limits in Diaporthe: Molecular re-assessment of D. citri, D. cytosporella, D. foeniculina and D. rudis. Persoonia 2014, 32, 83–101. [Google Scholar] [CrossRef]
- Lawrence, D.P.; Travadon, R.; Baumgartner, K. Diversity of Diaporthe species associated with wood cankers of fruit and nut crops in northern California. Mycologia 2015, 107, 926–940. [Google Scholar] [CrossRef] [PubMed]
- Scheper, R.W.A.; Crane, D.C.; Whisson, D.L.; Scott, E.S. The Diaporthe teleomorph of Phomopsis Taxon 1 on grapevine. Mycol. Res. 2000, 104, 226–231. [Google Scholar] [CrossRef]
- Gomes, R.R.; Glienke, C.; Videira, S.I.; Lombard, L.; Groenewald, J.Z.; Crous, P.W. Diaporthe: A genus of endophytic, saprobic and plant pathogenic fungi. Persoonia 2013, 31, 1–41. [Google Scholar] [CrossRef]
- Huang, F.; Udayanga, D.; Wang, X.; Hou, X.; Mei, X.; Fu, Y.; Hyde, K.D.; Li, H. Endophytic Diaporthe associated with Citrus: A phylogenetic reassessment with seven new species from China. Fungal Biol. 2015, 119, 331–347. [Google Scholar] [CrossRef]
- Kanematsu, S.; Minaka, N.; Kobayashi, T.; Kudo, A.; Ohtsu, Y. Molecular Phylogenetic Analysis of Ribosomal DNA Internal Transcribed Spacer Regions and Comparison of Fertility in Phomopsis Isolates from Fruit Trees. J. Gen. Plant Pathol. 2000, 66, 191–201. [Google Scholar] [CrossRef]
- Mostert, L.; Crous, P.W.; Kang, J.-C.; Alan, J.L.P. Species of Phomopsis and a Libertella sp. Occurring on Grapevines with Specific Reference to South Africa: Morphological, Cultural, Molecular and Pathological Characterization. Mycologia 2001, 93, 146–167. [Google Scholar] [CrossRef]
- van Niekerk, J.M.; Groenewald, J.Z.; Farr, D.F.; Fourie, P.H.; Halleer, F.; Crous, P.W. Reassessment of Phomopsis species on grapevines. Australas. Plant Pathol. 2005, 34, 27–39. [Google Scholar] [CrossRef]
- Bobev, S. Reference Guide for the Diseases of Cultivated Plants. Available online: http://nt.ars-grin.gov/fungaldatabases (accessed on 29 November 2021).
- Santos, J.M.; Correia, V.G.; Phillips, A.J. Primers for mating-type diagnosis in Diaporthe and Phomopsis: Their use in teleomorph induction in vitro and biological species definition. Fungal Biol. 2010, 114, 255–270. [Google Scholar] [CrossRef]
- Greuter, W.; Poelt, J.; Raimondo, F.M. A checklist of Sicillian fungi. Bocconea 1991, 2, 222. [Google Scholar]
- Van Niekerk, J.M.; Crous, P.W.; Groenewald, J.Z.; Fourie, P.H.; Halleen, F. DNA phylogeny, morphology and pathogenicity of Botryosphaeria species on grapevines. Mycologia 2004, 96, 781–798. [Google Scholar] [CrossRef] [PubMed]
- Whitelaw-Weckert, M.; Rahman, L.; Appleby, L.M.; Hall, A.; Clark, A.C.; Waite, H.; Hardie, W.J. Co-infection by Botryosphaeriaceae and Ilyonectria spp. fungi during propagation causes decline of young grafted grapevines. Plant Pathol. 2013, 62, 1226–1237. [Google Scholar] [CrossRef]
- Savas, N.; Akgul, D.S.; Albaz, E.A. First report of Ilyonectria liriodendra associated with black foot disease of grapevine in Turkey. Plant Dis. 2015, 99, 156. [Google Scholar] [CrossRef]
- Parkinson, L.E.; Shivas, R.G.; Dann, E.K. Pathogenicity of Nectriaceous Fungi on Avocado in Australia. Phytopathology 2017, 107, 1479–1485. [Google Scholar] [CrossRef] [PubMed]
- Crous, P.W.; Slippers, B.; Wingfield, M.J.; Rheeder, J.; Marasas, W.F.; Philips, A.J.; Alves, A.; Burgess, T.; Barber, P.; Groenewald, J.Z. Phylogenetic lineages in the Botryosphaeriaceae. Stud. Mycol. 2006, 55, 235–253. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, M. Primer catalogo de enfermedades de plantas Mexicanas. Fitofilo 1976, 71, 1–169. [Google Scholar]
- Cho, W.; Shin, H.D. List of Plant Diseases in Korea, 4th ed.; Korean Society of Plant Pathology: Suwon, Korea, 2004. [Google Scholar]
- Phillips, A.J.L.; Fonseca, F.; Povoa, V.; Castilho, R.; Nolasco, G. A reassessement of the anamorphic fungus Fusicoccum luteum and description of its teleomorph Botryosphaeria lutea sp. nov. Sydowia 2002, 54, 59–77. [Google Scholar]
- Phillips, A.; Alves, A.; Correia, A.; Luque, J. Two new species of Botryosphaeria with brown, 1-septate ascospores and Dothiorella anamorphs. Mycologia 2005, 97, 513–529. [Google Scholar] [CrossRef]
- Luque, J.; Martos, S.; Phillips, A.J.L. Botryosphaeria viticola sp. nov. on grapevines: A new species with a Dothiorella anamorph. Mycologia 2005, 97, 1111–1121. [Google Scholar] [CrossRef]
- Úrbez-Torres, J.R.; Leavitt, G.M.; Voegel, T.M.; Gubler, W.D. Identification and distribution of Botryosphaeria spp. associated with grapevine cankers in California. Plant Dis. 2006, 90, 1490–1503. [Google Scholar] [CrossRef]
- Cunnington, J.H.; Priest, M.J.; Powney, R.A.; Cother, N.J. Diversity of Botryosphaeria species on horticultural plants in Victoria and New South Wales. Australas 2007, 36, 157–159. [Google Scholar]
- Baskarathevan, J.; Jaspers, M.V.; Jones, E.E.; Ridgway, H.J. Distribution of Botryosphaeria species causing grapevine dieback and decline in New Zealand vineyards. N. Z. Plant Prot. 2008, 61, 392. [Google Scholar] [CrossRef]
- Sakalidis, M.L.; Slippers, B.; Wingfield, B.D.; St, J.; Hardy, G.E.; Burgess, T.I. The challenge of understanding the origin, pathways and extent of fungal invasions: Global populations of the Neofusicoccum parvum—N. ribis species complex. Divers. Distrib. 2013, 19, 873–883. [Google Scholar] [CrossRef]
- Wu, D.D.; Fu, G.; Hu, F.Y.; Ye, Y.F.; Mou, H.F.; Qin, L.L.; Jiang, N. First report of Neofusicoccum parvum causing panicle blight and leaf spot on Vitis heyneana in China. Plant Dis. 2015, 99, 417. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Pando, M.; Somers, A.; Green, C.D.; Priest, M.; Sriskanthades, M. Fungi associated with dieback of Semillon grapevines in the Hunter Valley of New South Wales. Australas. Plant Pathol. 2001, 30, 59–63. [Google Scholar] [CrossRef]
- Mostert, L.; Halleen, F.; Fourie, P.; Crous, P.W. A review of Phaeoacremonium species involved in Petri disease and esca of grapevines. Phytopathol. Mediterr. 2006, 45, 12–29. [Google Scholar]
- Baloyi, M.A.; Halleen, F.; Mostert, L.; Eskalen, A. First report of Togninia minima perithecia on esca- and Petri-diseased grapevines in South Africa. Plant Dis. 2013, 97, 1247. [Google Scholar] [CrossRef]
- Dupont, J.; Magnin, S.; Cesari, C.; Gatica, M. ITS and β-tubulin markers help delineate Phaeoacremonium species, and the occurrence of P. parasiticum in grapevine disease in Argentina. Mycol. Res. 2002, 106, 1143–1150. [Google Scholar] [CrossRef]
- Berraf-Tebbal, A.; Bouznad, Z.; Santos, J.M.; Coelho, M.A.; Peros, J.-P.; Phillips, A.J.L. Phaeoacremonium species associated with Eutypa dieback and esca of grapevines in Algeria. Phytopathol. Mediterr. 2011, 50, S86–S97. [Google Scholar]
- Gramaje, D.; Armengol, J.; Mohammadi, H.; Banihashemi, Z.; Mostert, L. Novel Phaeoacremonium species associated with Petri disease and esca of grapevine in Iran and Spain. Mycologia 2009, 101, 920–929. [Google Scholar] [CrossRef]
- White, C.-L.; Halleen, F.; Fischer, M.; Mostert, L. Characterisation of the fungi associated with esca diseased grapevines in South Africa. Phytopathol. Mediterr. 2011, 50, 204–223. [Google Scholar]
- Larignon, P.; Dubos, B. Fungi associated with esca disease in grapevine. Eur. J. Plant Pathol. 1997, 103, 147–157. [Google Scholar] [CrossRef]
- Dupont, J.; Laloui, W.; Roquebert, M.F. Partial ribosomal DNA sequences show an important divergence between Phaeoacremonium species isolated from Vitis vinifera. Mycol. Res. 1998, 102, 631–637. [Google Scholar] [CrossRef]
- Scheck, H.; Vasquez, S.J.; Fogle, D.; Gubler, W.D. Grape growers report losses to black-foot and grapevine decline. Calif. Agric. 1998, 52, 19–23. [Google Scholar] [CrossRef][Green Version]
- Chicau, G.; Aboim-Inglez, M.; Cabral, S.; Cabral, J.P.S. Phaeoacremonium chlamydosporum and Phaeoacremonium angustius associated with esca and grapevine decline in Vinho Verde grapevines in northwest Portugal. Phytopathol. Mediterr. 2000, 39, 80–86. [Google Scholar]
- Pascoe, I.; Cottral, E. Developments in grapevine trunk diseases research in Australia. Phytopathol. Mediterr. 2000, 39, 68–75. [Google Scholar]
- Auger, J.; Esterio, M.; Ricke, G.; Perez, I. Black dead arm and basal canker of Vitis vinifera cv. red globe cause by Botryosphaeria obtuse in Chile. Plant Dis. 2004, 88, 1286. [Google Scholar] [CrossRef]
- Lardner, R.; Stummer, B.E.; Sosnowski, M.R.; Scott, E.S. Molecular identification and detection of Eutypa lata in grapevine. Mycol. Res. 2005, 109, 799–808. [Google Scholar] [CrossRef]
- Santos, C.; Fragoeiro, S.; Valentim, H.; Phillips, A. Phenotypic characterisation of Phaeoacremonium and Phaeomoniella strains isolated from grapevines: Enzyme production and virulence of extra-cellular filtrate on grapevine calluses. Sci. Hortic. 2006, 107, 123–130. [Google Scholar] [CrossRef]
- NagRaj, T.R. Coelomycetous Anamorphs with Appendage-Bearing Conidia; Mycologue Publications: Waterloo, ON, Canada, 1993. [Google Scholar]
- Arzanlou, M.; Narmani, A.; Moshari, S.; Khodaei, S.; Babai-Ahari, A. Truncatella angustata associated with grapevine trunk disease in northern Iran. Arch. Phytopathol. Plant Prot. 2013, 46, 1168–1181. [Google Scholar] [CrossRef]
- Gramaje, D.; Di Marco, S. Identifying practices likely to have impacts on grapevine trunk disease infections: A European nursery survey. Phytopathol. Mediterr. 2015, 54, 313–324. [Google Scholar]
- Agustí-Brisach, C.; Gramaje, D.; Garcia Jiménez, J.; Armengol Fortí, J. Detection of black-foot and Petri disease pathogens in soils of grapevine nurseries and vineyards using bait plants. Plant Soil 2013, 364, 5–13. [Google Scholar] [CrossRef]
- Birebent, M. Grafting rootstocks for sustainable vines. Old techniques for the viticulture of the future? In Proceedings of the Conference La Vigne au 2ème Siècle—Retour vers le Futur, Piolenc, France, 5 February 2015. [Google Scholar]
- Lecomte, P.; Darrieutort, G.; . Liminana, J.-M.; Comont, G.; Muruamendiarsaz, A.; Legorburu, F.-J.; Choueiri, E.; Jreijiri, F.; El Amil, R.; Fermaud, M. New Insights into Esca of Grapevine: The Development of Foliar Symptoms and Their Association with Xylem Discoloration. Plant Dis. 2012, 96, 924–934. [Google Scholar] [CrossRef]
- Hofstetter, V.; Buyck, B.; Croll, D.; Viret, O.; Couloux, A.; Gindro, K. What if esca disease of grapevine were not a fungal disease? Fungal Divers. 2012, 54, 51–67. [Google Scholar] [CrossRef]
- Frías-Lopez, J.; Zerkle, A.L.; Bonheyo, G.T.; Fouke, B.W. Partitioning of bacterial communities between seawater and healthy, black band diseased, and dead coral surfaces. Appl. Environ. Microbiol. 2002, 68, 2214–2228. [Google Scholar] [CrossRef]
- Chacón-Vozmediano, J.L.; Gramaje, D.; León, M.; Armengol, J.; Moral, J.; Izquierdo-Cañas, P.M.; Martínez-Gascueña, J. Cultivar Susceptibility to Natural Infections Caused by Fungal Grapevine Trunk Pathogens in La Mancha Designation of Origin (Spain). Plants 2021, 10, 1171. [Google Scholar] [CrossRef] [PubMed]
- Toledo-Hernández, C.; Zuluaga-Montero, A.; Bones-González, A.; Rodríguez, J.A.; Sabat, A.; Bayman, P. Fungi in healthy and diseased sea fans (Gorgonia ventalina): Is Aspergillus sydowii always the pathogen? Coral Reefs 2008, 27, 707–714. [Google Scholar] [CrossRef]
- González, V.; Tello, M.L. The endophytic mycota associated with Vitis vinifera in central Spain. Fungal Divers. 2011, 47, 29–42. [Google Scholar] [CrossRef]
- Kulišová, M.; Vrublevskaya, M.; Lovecká, P.; Vrchotová, B.; Stránská, M.; Kolařík, M.; Kolouchová, I. Fungal Endophytes of Vitis vinifera—Plant Growth Promotion Factors. Agriculture 2021, 11, 1250. [Google Scholar] [CrossRef]
- Shi, J.; Zeng, Q.; Liu, Y.; Pan, Z. Alternaria sp. MG1, a resveratrol-producing fungus: Isolation, identification, and optimal cultivation conditions for resveratrol production. Appl. Microbiol. Biotechnol. 2012, 95, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Maharachchikumbura, S.; Larigon, P.; Hyde, K.D.; Al-Sadi, A.M.; Liu, Z. Characteriztion of Neopestalotiopsis, Pestalotiopsis and Truncatella species associated with grapevine trunk disease in France. Phytopathol. Mediterr. 2016, 55, 380–390. [Google Scholar]
- Fotios, B.; Sotirios, V.; Elena, P.; Anastasios, S.; Stefanos, T.; Danae, G.; Georgia, T.; Aliki, T.; Epaminondas, P.; Emmanuel, M.; et al. Grapevine wood microbiome analysis identifies key fungal pathogens and potential interactions with the bacterial community implicated in grapevine trunk disease appearance. Environ. Microbiomes 2021, 16, 23. [Google Scholar] [CrossRef] [PubMed]
| Rootstock | 41B | R-110 | RU-140 | SO4 | Grand Total |
|---|---|---|---|---|---|
| Red Varieties | 60 | 48 | 42 | 150 | |
| Autumn Royal | 6 | 6 | |||
| Cabernet Sauvignon | 12 | 6 | 12 | 30 | |
| Caladoc | 6 | 6 | |||
| Carignan | 6 | 6 | |||
| Garnacha tinta | 24 | 18 | 42 | ||
| Merlot | 6 | 6 | 12 | ||
| Pinot Noir | 6 | 6 | |||
| Sumoll | 6 | 6 | |||
| Syrah | 12 | 6 | 6 | 24 | |
| Tempranillo | 6 | 6 | |||
| Xarel.lo vermell | 6 | 6 | |||
| White Varieties | 36 | 84 | 54 | 66 | 240 |
| Chardonnay | 6 | 12 | 6 | 24 | |
| Macabeu | 18 | 36 | 12 | 24 | 90 |
| Malvasia | 6 | 6 | |||
| Parellada | 6 | 12 | 12 | 30 | |
| Xarel.lo | 12 | 30 | 18 | 30 | 90 |
| Grand Total | 36 | 144 | 102 | 108 | 390 |
| Factor | Graft Unions (r2) | Root Collars (r2) |
|---|---|---|
| Nursery | 0.064 *** | 0.011 n.s. |
| Variety | 0.019 * | - |
| Rootstock | - | 0.101 *** |
| Storage | 0.008 n.s. | 0.013 n.s. |
| A. Total Community | |||
| Factor | Graftunions(r2) | Root collars (r2) | Total (r2) |
| Tissue type | - | - | 0.560 *** |
| Nursery | 0.363 *** | 0.159 *** | 0.057 *** |
| Variety | 0.174 *** | 0.136 *** | 0.045 *** |
| Color | 0.038 *** | 0.030 *** | 0.012 ** |
| Rootstock | 0.047 *** | 0.022 * | 0.004 n.s. |
| Storage | 0.436 n.s. | 0.701 n.s. | 0.760 n.s. |
| B. GTD Community | |||
| Factor | Graft unions (r2) | Root collars (r2) | Total (r2) |
| Tissue type | - | - | 0.107 *** |
| Nursery | 0.240 *** | 0.208 *** | 0.179 *** |
| Variety | 0.115 *** | 0.095 *** | 0.072 *** |
| Color | 0.024 *** | 0.014 *** | 0.016 *** |
| Rootstock | 0.045 *** | 0.005 n.s. | 0.014 ** |
| Storage | 0.003 n.s. | 0.049 *** | 0.016 *** |
| Graft Union | Nursery (r2) | Storage (r2) | ||
|---|---|---|---|---|
| Variety | Total | GTDs | Total | GTDs |
| Cabernet (3) | 0.490 *** | 0.266 ** | 0.185** | 0.010 n.s. |
| Chardonnay (4) | 0.649 *** | 0.346 * | 0.137 n.s. | 0.064 n.s. |
| Garnacha tinta (5) | 0.457 *** | 0.220 * | 0.117 ** | 0.037 n.s. |
| Macabeu (6) | 0.599 *** | 0.448 *** | 0.003 n.s. | 0.000 n.s. |
| Xarel·lo (5) | 0.430 *** | 0.303 *** | 0.087 ** | 0.038 * |
| Root collar | ||||
| Rootstock | Total | GTDs | Total | GTDs |
| R-110 (6) | 0.184 *** | 0.211 *** | 0.051 ** | 0.052 ** |
| SO4 (6) | 0.290 *** | 0.329 *** | 0.069 ** | 0.020 n.s. |
| RU-140 (5) | 0.247 *** | 0.143 ** | 0.090 *** | 0.099 *** |
| 41B (3) | 0.443 *** | 0.315 ** | 0.156 * | 0.219 ** |
| Graft union | Variety (r2) | Storage (r2) | Rootstock (r2) | |||
|---|---|---|---|---|---|---|
| Nursery | Total | GTDs | Total | GTDs | Total | GTDs |
| I (10) | 0.337 *** | 0.266 ** | 0.153 *** | 0.021 n.s. | - | - |
| II (7) | 0.211 * | 0.270 ** | 0.123 *** | 0.005 n.s. | - | - |
| III (7) | 0.173 ** | 0.051 n.s. | 0.160 *** | 0.048 * | - | - |
| IV (4) | 0.259 ** | 0.181 * | 0.301 *** | 0.106 ** | - | - |
| V (5) | 0.149 * | 0.138 * | 0.179 *** | 0.000 n.s. | - | - |
| VI (3) | 0.303 ** | 0.338 ** | 0.448 *** | 0.316 *** | - | - |
| Root collar | ||||||
| Nursery | Total | GTDs | Total | GTDs | Total | GTDs |
| I (10) | - | - | 0.028 n.s. | 0.143 *** | 0.126 ** | 0.053 n.s. |
| II (7) | - | - | 0.198 *** | 0.005 n.s. | 0.080 n.s. | 0.049 n.s. |
| III (7) | - | - | 0.033 * | 0.424 *** | 0.108 ** | 0.031 n.s. |
| IV (4) | - | - | 0.279 *** | 0.038 n.s. | 0.060 n.s. | 0.038 n.s. |
| V (5) | - | - | 0.104 ** | 0.002 n.s. | 0.110 * | 0.066 n.s. |
| VI (3) | - | - | 0.269 *** | 0.133 n.s. | 0.242 * | 0.145 n.s. |
| Factor | Graft Union (X2) | Root Collar (X2) |
|---|---|---|
| Nursery | 29.608 *** | 24.954 *** |
| Variety | 28.641 * | - |
| Rootstock | - | 2.419 n.s. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lade, S.B.; Štraus, D.; Oliva, J. Variation in Fungal Community in Grapevine (Vitis vinifera) Nursery Stock Depends on Nursery, Variety and Rootstock. J. Fungi 2022, 8, 47. https://doi.org/10.3390/jof8010047
Lade SB, Štraus D, Oliva J. Variation in Fungal Community in Grapevine (Vitis vinifera) Nursery Stock Depends on Nursery, Variety and Rootstock. Journal of Fungi. 2022; 8(1):47. https://doi.org/10.3390/jof8010047
Chicago/Turabian StyleLade, Sarah B., Dora Štraus, and Jonàs Oliva. 2022. "Variation in Fungal Community in Grapevine (Vitis vinifera) Nursery Stock Depends on Nursery, Variety and Rootstock" Journal of Fungi 8, no. 1: 47. https://doi.org/10.3390/jof8010047
APA StyleLade, S. B., Štraus, D., & Oliva, J. (2022). Variation in Fungal Community in Grapevine (Vitis vinifera) Nursery Stock Depends on Nursery, Variety and Rootstock. Journal of Fungi, 8(1), 47. https://doi.org/10.3390/jof8010047






