Oral Candidosis: Pathophysiology and Best Practice for Diagnosis, Classification, and Successful Management
Abstract
:1. Introduction
2. Interplay of Host Defenses and Candida Virulence Factors in Oral Candidosis
3. Iron Deficiency and Immunosuppression in Oral Candidosis
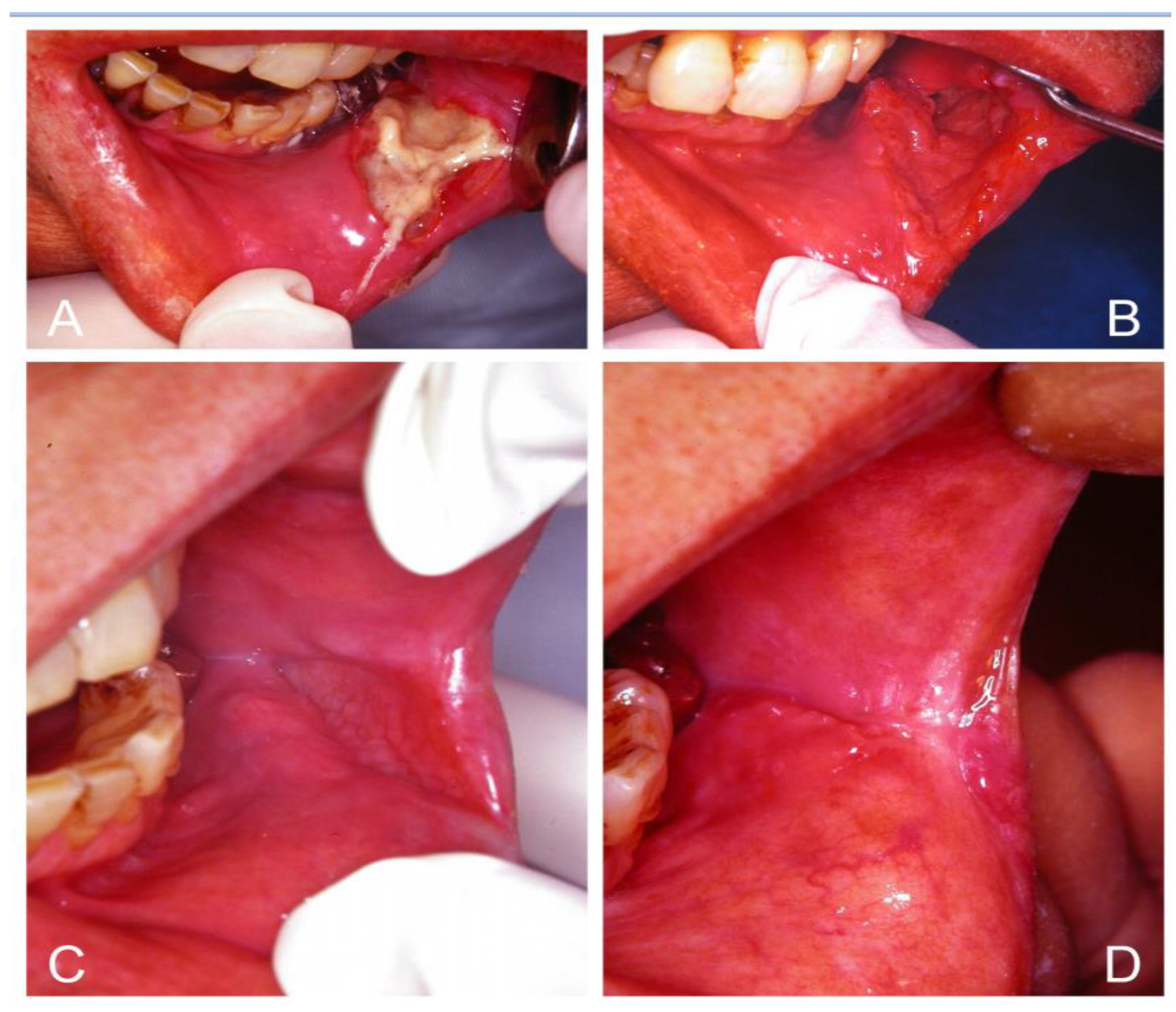
4. Diagnosis of Oral Candidosis
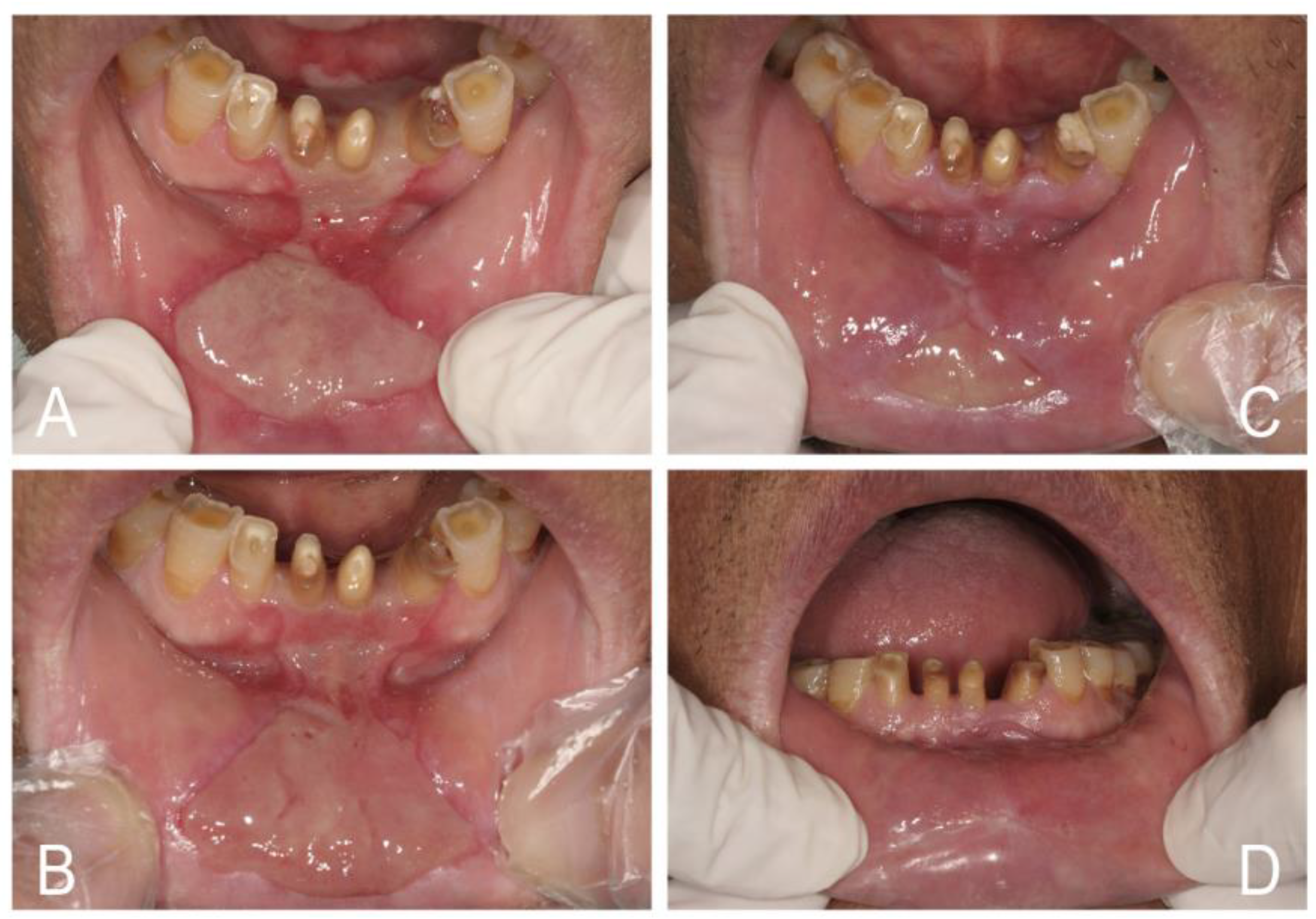
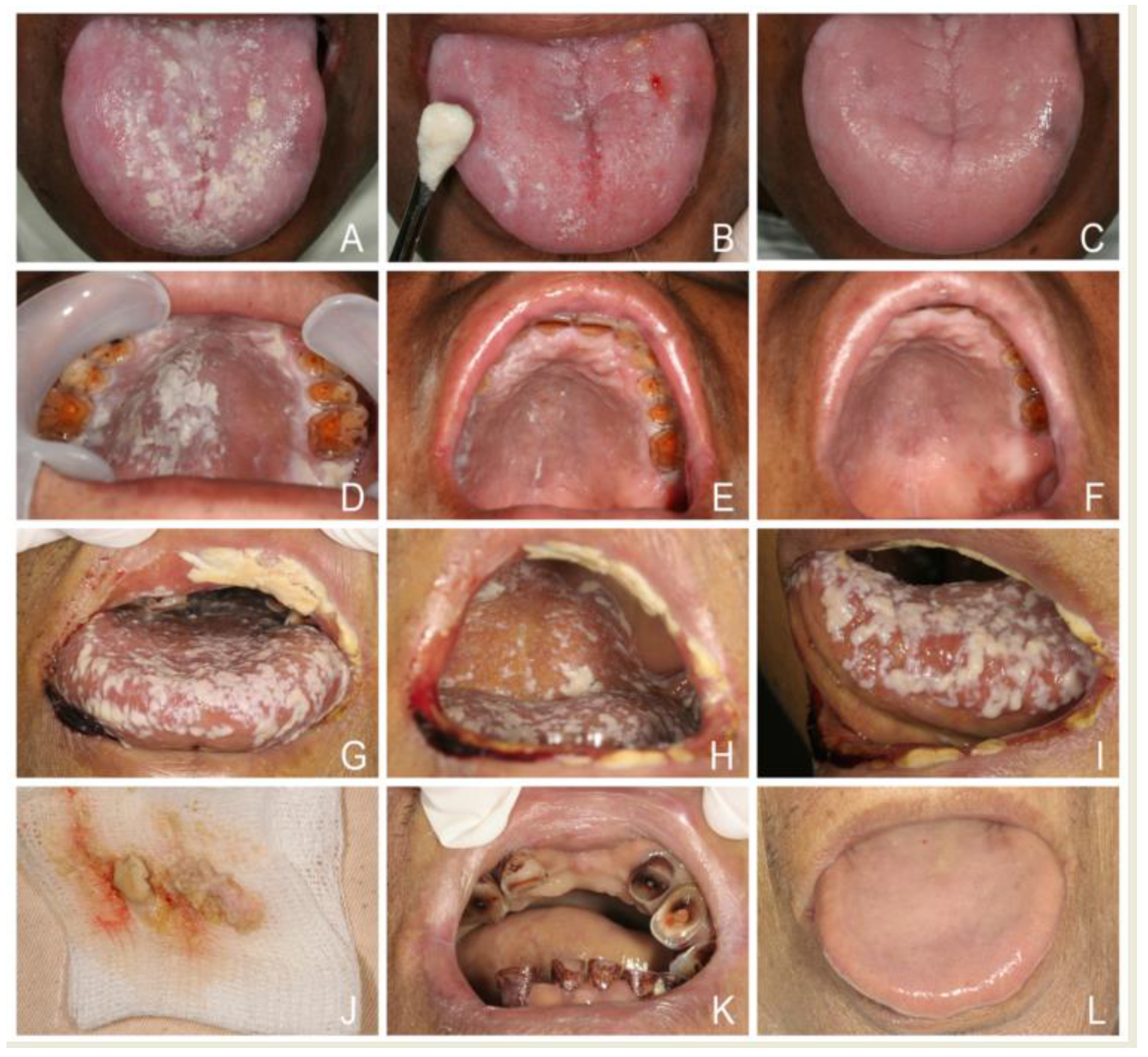
5. Classification of Oral Candidosis
6. Management for Oral Candidosis Patients
7. Conclusions and Important Suggestions for Management of Oral Candidosis
- (1)
- Maintenance of good oral and denture hygiene is crucial. It is important to remove dentures overnight, use denture cleanser, or make a new denture if an ill-fitting denture with stomatitis exists.
- (2)
- Rinsing the mouth after use of an inhaled steroid is helpful to prevent oral candidosis.
- (3)
- Glucose promotes yeast growth, and a high-carbohydrate diet enhances its adherence to oral epithelial cells. Limiting their consumption is helpful in the control of oral Candida colonization and infection.
- (4)
- Removal of heavy candidal plaques or biofilm from oral lesions by mechanical means can improve antifungal action and speed healing.
- (5)
- Nystatin tablets are significantly superior to nystatin oral suspension in treating oral candidosis. Swallowing nystatin tablets rather than sucking or dissolving them in the mouth is ineffective to treat oral candidosis.
- (6)
- The duration of antifungal treatment should be sufficient or prolonged for at least four weeks to achieve a more permanent mycological cure.
- (7)
- Early fluconazole monotherapy or fluconazole combined with nystatin is helpful to treat oropharyngeal candidosis, suppurative candodosis, or Candida-related chronic oral ulcers.
- (8)
- Attempts to increase CD4 count in patients with HIV/AIDS or thymoma are helpful to treat oral candidosis. HAART can reduce recurrent oropharyngeal candidosis.
- (9)
- Underlying predisposing factors should be identified and treated simultaneously as well as monitored regularly.
- (10)
- The final eradication of oral candidosis is by host defense system.
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vila, T.; Sultan, A.S.; Montelongo-Jauregui, D.; Jabra-Rizk, M.A. Oral candidiasis: A disease of opportunity. J. Fungi 2020, 6, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Darwazeh, A.M.G.; Darwazeh, T.A. What makes oral candidiasis recurrent infection? A clinical view. J. Mycol. 2014, 2014, 1–5. [Google Scholar] [CrossRef]
- Schsuer, F.; Hanschke, R. Taxonomy and ecology of the genus Candida. Mycoses 1999, 42, 12–21. [Google Scholar] [CrossRef]
- Akpan, A.; Morgan, R. Oral candidiasis. Postgrad. Med. J. 2002, 78, 455–459. [Google Scholar] [CrossRef]
- Samaranayake, L.P.; MacFarlane, T.W. (Eds.) Oral Candidosis; Butterworth: London, UK, 1990; pp. 1–259. [Google Scholar]
- Ruhnke, M. Epidemiology of Candida albicans infections and role of non-Candida albicans yeasts. Curr. Drug Targets 2006, 7, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Supriya, H.; Harishchandra, R.; Suhasini, P.D.; Rajalekshmi, V. Pathogenic mechanism of Candida albicans in oral mucosa—A review. Int. J. Health Sci. Res. 2016, 1, 489–497. [Google Scholar]
- Rao, P.N.; Rao, K.N. Study of the normal conjunctival flora (bacterial and fungal) and its relations to external ocular infections. Indian I. Ophthalmol. 1972, 20, 164–170. [Google Scholar]
- Sthapit, P.R.; Tuladhar, N.R. Conjunctival flora of normal human eye. JSM Ophthalmol. 2014, 2, 1021. [Google Scholar]
- Alrayyes, S.F.; Alruwaili, H.M.; Taher, I.A.; Elrahawy, K.M.; Almaeen, A.H.; Ashekhi, A.O.; Alam, M.K. Oral candidal carriage and associated risk indicators among adults in Sakaka, Saudi Arabia. BMC Oral Health 2019, 22, 86. [Google Scholar] [CrossRef] [Green Version]
- Zomorodian, K.; Kavoosi, F.; Pishdad, G.R.; Mehriar, P.; Ebrahimi, A.; Bandegani, A.; Pakshir, K. Prevalence of oral Candida colonization in patients with diabetes mellitus. J. Mycol. Med. 2016, 26, 103–110. [Google Scholar] [CrossRef]
- Vidya, K.M.; Rao, U.K.; Nittayananta, W.; Liu, H.; Owotade, F.J. Oral mycoses and other oppontunistic infectionsin HIV: Therapy and emerging problems-a workshop report. Oral Dis. 2016, 22, 158–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schelenz, S.; Abdallah, S.; Gray, G.; Stubbings, H.; Gow, I.; Baker, P. Epidemiology of oral yeast colonization and infection in patients with hematological malignancies, head neck and solid tumors. J. Oral Pathol. Med. 2011, 40, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Rennie, J.S.; MacDonald, D.G.; Dagg, J.H. Iron and the oral epithelium: A review. J. R. Soc. Med. 1984, 77, 602–607. [Google Scholar] [CrossRef] [Green Version]
- Guinea, J. Global trends in the distribution of Candida species causing candidemia. Clin. Microbiol. Infect. 2014, 20, 5–10. [Google Scholar] [CrossRef] [Green Version]
- Kaur, R.; Dhakad, M.S.; Goyal, R.; Kumar, R. Emergence of non-albicans Candida species and antifungal resistance in intensive care unit patients. Asian Pac. J. Trop. Biomed. 2016, 6, 455–460. [Google Scholar] [CrossRef] [Green Version]
- Quindós, G. Epidemiology of candidemia and invasive candidiasis. A changing face. Rev. Iberoam Micol. 2014, 31, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.; Majumdar, B.; Sarode, S.C.; Sarode, G.S.; Awan, K.H. Oropharyngeal candidosis in HIV-infected patients—An update. Front. Microbiol. 2018, 9, 980. [Google Scholar] [CrossRef] [Green Version]
- Thompson, G.R., III; Patel, P.K.; Kirkpatrick, W.R.; Westbrook, S.D.; Berg, D.; Erlandsen, J.; Redding, S.W.; Patterson, T.F. Oropharyngeal candidiasis in the era of antiretroviral therapy. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 109, 488–495. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.L.; Lo, H.J. Mechanisms of antifungal agent resistance. J. Microbiol. Immunol. Infect. 2001, 34, 79–86. [Google Scholar]
- Mishra, N.N.; Prasad, T.; Sharma, N.; Payasi, A.; Prasad, R.; Gupta, D.K.; Singh, R. Pathogenicity and drug resistance in Candida albicans and other yeast species. Acta Microbiol. Immunol. Hung. 2007, 54, 201–235. [Google Scholar] [CrossRef]
- Tobgi, R.S.; Samaranayake, L.P.; MacFariane, T.W. In vitro susceptibility of Candida species to lysozyme. Oral Microbiol. Immunol. 1988, 3, 36–39. [Google Scholar] [CrossRef]
- Sawasdipuksa, N.; Lei, Z.; Sumner, L.W.; Niyomploy, P.; Sangvanich, P. A lysozyme with antifungal activity from Pithecellobium dulce seeds. Food Technol. Biotechnol. 2011, 49, 489–494. [Google Scholar]
- Galvez-Iriqui, A.C.; Plascencia-Jatomea, M. Lysozymes: Characteristics, mechanism of action and technological applications on the control of pathogenic microrganisms. Mex. J. Phytopathol. 2020, 38, 360–383. [Google Scholar] [CrossRef]
- Redding, S.W.; Dahiya, C.; Kirkpatrick, W.R.; Coco, B.J.; Patterson, T.F.; Fothergill, A.W.; Rinaldi, M.G.; Thomas, C.R., Jr. Candida glabrata is an emerging cause of oropharyngeal candidiasis in patients receiving radiation for head and neck cancer. Oral Surg. Oral Med. Oral Pathol. Oral Radio. Endod. 2004, 97, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Van Wyk, C.; Steenkamp, C. Host factors affecting oral candidiasis. South. Afr. J. Epidemiol. Infect. 2011, 26, 18–21. [Google Scholar] [CrossRef] [Green Version]
- Mayer, F.L.; Wilson, D.; Hube, B. Candida albicans pathogenicity mechanism. Virulence 2013, 4, 119–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, S.Y. Perception of iron deficiency from oral mucosa alterations that show a high prevalence of Candida infection. J. Formos. Med. Assoc. 2016, 115, 619–627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lai, W.H.; Lu, S.Y.; Eng, H.L. Levamisole aid in the treatment of refractory oral candidosis in two cases of myasthenia gravis. Chang. Gung Med. J. 2002, 25, 606–612. [Google Scholar] [PubMed]
- Lu, S.Y.; Wu, H.C. Initial diagnosis of anemia from sore mouth and improved classification of anemias by MCV and RDW in 30 patients. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2004, 98, 679–685. [Google Scholar] [CrossRef]
- Lu, S.Y.; Eng, H.L. Diagnosis of secondary syphilis-related oral ulcers. Chang. Gung Med. J. 2002, 25, 683–688. [Google Scholar]
- Chiang, C.P.; Chang, J.Y.F.; Wang, Y.P.; Wu, Y.H.; Lu, S.Y.; Sun, A. Oral lichen planus—Differential diagnoses, serum autoantibodies, hematinic deficiencies, and management. J. Formos. Med. Assoc. 2018, 117, 756–765. [Google Scholar] [CrossRef]
- Jabra-Rizk, M.A.; Kong, E.; Tsui, C.; Nguyen, M.; Clancy, C.; Fidel, P.; Noverr, M. Candida albicans pathogenesis: Fitting within the host-microbe damage response framework. Infect. Immun. 2016, 84, 2724–2739. [Google Scholar] [CrossRef] [Green Version]
- Höfs, S.; Mogavero, S.; Hube, B. Interaction of Candida albicans with host cells: Virulence factors, host defense, escape strategies and the microbiota. J. Microbiol. 2016, 53, 149–169. [Google Scholar] [CrossRef] [PubMed]
- Schaller, M.; Borelli, C.; Korting, H.C.; Hube, B. Hydrolytic enzymes as virulence factors of Candida Albicans. Mycoses 2005, 48, 365–377. [Google Scholar] [CrossRef] [PubMed]
- Verstrepen, K.J.; Klis, F.M. Flocculation, adhesion and biofilm formation in yeasts. Mol. Microbiol. 2006, 60, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Tenovuo, J.; Moldoveanu, Z.; Mestecky, J.; Pruitt, K.M.; Rahemtulla, B.M. Interaction of specific and innate factors of immunity: IgA enhances the antimicrobial effect of the lactoperoxidase system against Streptococcus mutans. J. Immunol. 1982, 128, 726–731. [Google Scholar]
- De Groot, P.W.; Bader, O.; de Boer, A.D.; Weig, M.; Chauhan, N. Adhesins in human fungal pathogens: Glue with plenty of stick. Eukaryot. Cell. 2013, 12, 470–481. [Google Scholar] [CrossRef] [Green Version]
- Desai, J.V. Candida albicans hyphae: From growth initiation to invasion. J. Fungi 2018, 4, 10. [Google Scholar] [CrossRef] [Green Version]
- Phan, Q.T.; Myers, C.L.; Fu, F.; Sheppard, D.C.; Yeaman, M.R.; Welch, W.H.; Ibrahim, A.S.; Edwards, J.E., Jr.; Filler, S.G. Als3 is a Candida albicans invasin that binds to cadherins and induces endocytosis by host cells. PLoS Biol. 2007, 5, e64. [Google Scholar] [CrossRef] [Green Version]
- Swidergall, M.; Filler, S.G. Oropharyngeal candidiasis: Fungal invasion and epithelial cell responses. PLoS Pathog. 2017, 13, e1006056. [Google Scholar] [CrossRef]
- Soll, D.R. High-frequency switching in Candida albicans. Clin. Microbiol. Rev. 1992, 5, 183–203. [Google Scholar] [CrossRef] [PubMed]
- Cannon, R.D.; Chaffin, W.L. Colonization is a crucial factor in oral candidiasis. J. Dent. Educ. 2001, 65, 785–787. [Google Scholar] [CrossRef]
- Molero, G.; Díez-Orejas, R.; Navarro García, F.; Monteoliva, L.; Pla, J.; Gil, C. Candida albicans: Genetics, dimorphism and pathogenicity. Int. Microbiol. 1998, 1, 95–106. [Google Scholar]
- Zhu, W.; Filler, S.G. Interactions of Candida albicans with epithelial cells. Cell Microbiol. 2010, 12, 273–282. [Google Scholar] [CrossRef] [Green Version]
- Williams, D.W.; Jordan, R.P.C.; Wei, X.Q.; Alves, C.; Wise, M.P.; Wilson, M.J.; Lewis, M. Interactions of Candida albicans with host epithelial surfaces. J. Oral Microbiol. 2013, 5, 3402. [Google Scholar] [CrossRef]
- Silverman, R.J.; Nobbs, A.H.; Vickerman, M.M.; Barbour, M.E.; Jenkinson, H.F. Interaction of Candida albicans cell wall Als3 protein with Streptococcus gordonii AspB adhesin promotes development of mixed-species communities. Infect. Immun. 2010, 78, 4644–4652. [Google Scholar] [CrossRef] [Green Version]
- Wall, G.; Montelongo-Jauregui, D.; Vidal Bonifacio, B.; Lopez-Ribot, J.L.; Uppuluri, P. Candida albicans biofilm growth and dispersal: Contributions to pathogenesis. Curr. Opin. Microbiol. 2019, 11, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Swidergall, M.; Khalaji, M.; Solis, N.V.; Moyes, D.L.; Drummond, R.A.; Hube, B.; Lionakis, M.S.; Murdoch, C.; Filler, S.G.; Naglik, J.R. Candidalysin is required for neutrophil recruitment and virulence during systemic Candida albicans infection. J. Infect. Dis. 2019, 220, 1477–1488. [Google Scholar] [CrossRef]
- Verma, A.H.; Richardson, J.P.; Zhou, C.; Coleman, B.M.; Moyes, D.L.; Ho, J.; Huppler, A.R.; Ramani, K.; McGeachy, M.J.; Mufazalov, I.A.; et al. Oral epithelial cells orchestrate innate type 17 responses to Candida albicans through the virulence factor candidalysin. Sci. Immunol. 2017, 2, eaam8834. [Google Scholar] [CrossRef] [Green Version]
- Santana, I.L.; Goncalves, L.M.; Vasconcellos, A.A.; da Silva, W.J.; Cury, J.A.; Cury, A.A.D.B. Dietary carbohydrates modulate Candida albicans biofilm development on the denture surface. PLoS ONE 2013, 8, e64645. [Google Scholar] [CrossRef] [Green Version]
- Mandal, S.M.; Mahata, D.; Migliolo, L.; Parekh, A.; Addy, P.S.; Mandal, M.; Basak, A. Glucose directly promotes antifungal resistance in the fungal pathogen, Candida spp. J. Biol. Chem. 2014, 289, 25469–25473. [Google Scholar] [CrossRef] [Green Version]
- Taff, H.T.; Mitchell, K.F.; Edward, J.A.; Andes, D.R. Mechanisms of Candida biofilm drug resistance. Future Microbiol. 2013, 8, 1325–1337. [Google Scholar] [CrossRef] [Green Version]
- Odds, F.C.; Abbot, A.B. A simple system for the presumptive identification of Candida albicans and differentiation of strains with the species. Sabouraudia 1980, 18, 301–318. [Google Scholar] [CrossRef]
- Samaranayake, L.P.; MacFarlane, T.W. Factors affecting the in vitro adherence of the fungal oral pathogen Candida albicans to epithelial cells of human origin. Arch. Oral Biol. 1982, 27, 869–873. [Google Scholar] [CrossRef]
- Abu-Elteen, K.H. The influence of dietary carbohydrate on in vitro adherenc of four Candida species to human buccal epithelial cells. Micro. Ecol. Health Dis. 2005, 17, 156–162. [Google Scholar]
- Samaranayake, L.P. Nutritional factors and oral candidosis. J. Oral Pathol. 1986, 15, 61–65. [Google Scholar] [CrossRef]
- WHO/UNICEF/UNU. Iron Deficiency Anemia Assessment, Prevention, and Control: A Guide for Programme Managers; World health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Higgs, J.M.; Wells, R.S. Chronic muco-cutaneous candidiasis: New approaches to treatment. Br. J. Dermatol. 1973, 89, 179–190. [Google Scholar] [CrossRef]
- Feller, L.; Khammissa, R.A.; Chandran, R.; Altini, M.; Lemmer, J. Oral candidosis in relation to oral immunity. J. Oral Pathol. Med. 2014, 43, 563–569. [Google Scholar] [CrossRef] [Green Version]
- Sun, A.; Lin, H.P.; Wang, Y.P.; Chiang, C.P. Significant association of deficiency of hemoglobin, iron and vitamin B12, high homocysteine level, and gastric parietal cell antibody positivity with atrophic glossitis. J. Oral Pathol. Med. 2012, 41, 500–504. [Google Scholar] [CrossRef] [PubMed]
- Short, M.W.; Domagalski, J.E. Iron deficiency anemia: Evaluation and management. Am. Fam. Physician 2013, 87, 98–104. [Google Scholar] [PubMed]
- Bermejo, F.; García-López, S. A guide to diagnosis of iron deficiency and iron deficiency anemia in digestive diseases. World J. Gastroenterol. 2009, 15, 4638–4643. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.H.; Chau, W.K.; Hsu, H.C.; Gau, J.P.; You, J.Y.; Chen, C.C. Predictive risk factors and prevalence of malignancy in patients with iron deficiency anemia in Taiwan. Am. J. Hematol. 2005, 78, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Rennie, J.S.; Hutcheon, A.W.; MacFarlane, T.W.; MacDonald, D.G. The role of iron deficiency in experimentally induced oral candidosis in the rat. J. Med. Microbiol. 1983, 16, 363–369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fletcher, J.; Mather, J.; Lewis, M.J.; Whiting, G. Mouth lesions in iron-deficient anemia: Relationship to Candida albicans in saliva and to impairment of lymphocyte transformation. J. Infec. Dis. 1975, 131, 44–45. [Google Scholar] [CrossRef]
- Murphy, N.C.; Bissada, N.F. Iron deficiency: An overlooked predisposing factor in angular cheilitis. J. Am. Dent. Assoc. 1979, 99, 640–641. [Google Scholar] [CrossRef]
- Rennie, J.S.; MacDonald, D.G.; Dauglas, T.A. Haemoglobin, serum iron and transferrin values in adult male Syrian hamsters (Mesocricetus auratus). Lab. Anim. 1981, 15, 35–36. [Google Scholar] [CrossRef]
- Miller, J.L. Iron deficiency anemia: A common and curable disease. Cold Spring Harb. Perspect. Med. 2013, 3, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, V.; Choudhry, V.P. Iron deficiency and infection. Indian J. Pediatr. 2010, 77, 789–793. [Google Scholar] [CrossRef]
- Naderi, N.; Etaati, Z.; Joibari, M.R.; Sobhani, S.A.; Tashnizi, S.H. Immune deviation in recurrent vulvovaginal candidiasis: Correlation with iron deficiency anemia. Iran. J. Immunol. 2013, 10, 118–126. [Google Scholar]
- Beard, J.L. Iron biology in immune function, muscle metabolism and neuronal functioning. J. Nutr. 2001, 131, 568S–569S. [Google Scholar] [CrossRef]
- Wharton, B.A. Iron deficiency in children: Detection and prevention. Br. J. Haematol. 1999, 106, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Joynson, D.H.M.; Walker, D.M.; Jacobs, A.; Dolby, A.E. Defects of cell mediated immunity in patients with iron deficient anemia. Lancet 1972, 2, 1058–1059. [Google Scholar] [CrossRef]
- Farthing, M.J.G. Iron and immunity. Acta. Paediatr. Scand. Suppl. 1989, 361, 44–52. [Google Scholar] [CrossRef]
- Killip, S.; Bennett, J.M.; Chambers, M.D. Iron deficiency anemia. Am. Fam. Physician 2007, 75, 671–678. [Google Scholar]
- Garry, P.J.; Goodwin, J.S.; Hunt, W.C. Iron status and anemia in the elderly: New findings and a review of previous studies. J. Am. Geriatr. Soc. 1983, 31, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Rennie, J.S.; MacDonald, G.; Daggs, J.H. Quantitative analysis of human buccal epithelium in iron deficiency anemia. J. Oral Pathol. 1982, 11, 39–46. [Google Scholar] [CrossRef]
- Pappas, P.G.; Rex, J.H.; Sobel, J.D.; Filler, S.G.; Dismukes, W.E.; Walsh, T.J.; Edwards, J.E. Infectious Diseases Society of America: Guidelines for Treatment of Candidiasis. Clin. Infect. Dis. 2004, 38, 161–189. [Google Scholar] [CrossRef] [Green Version]
- Bandar, I.N.; Widodo, D.; Djauzi, S.; Muthalib, A.; Soegondo, S.; Wahyuningsih, R. Correlation between CD4 count and intensity of Candida colonization in the oropharynx of HIV-infected/ AIDS patient. Acta Med. Indones. 2006, 38, 119–125. [Google Scholar]
- Romanelli, F.; Matheny, S.C. HIV infection: The role of primary care. Am. Fam. Physician 2009, 80, 946–952. [Google Scholar]
- Pappas, P.G.; Kauffman, C.A.; Andes, D.; Benjamin, D.K., Jr.; Calandra, T.F.; Edwards, J.E., Jr.; Filler, S.G.; Fisher, J.F.; Kullberg, B.J.; Ostrosky-Zeichner, L.; et al. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2009, 48, 503–535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dongari-Bagtzoglou, A.; Fidel, P.L. The host cytokine responses and protective immunity in oropharyngeal candidiasis. J. Dent. Res. 2005, 84, 966–977. [Google Scholar] [CrossRef] [PubMed]
- Conti, H.R.; Gaffen, S.L. Host responses to Candida albicans: Th17 cells and mucosal candidiasis. Microb. Infect. 2010, 12, 518–527. [Google Scholar] [CrossRef] [Green Version]
- Lehner, T.; Wilton, J.M.A.; Ivanyi, L. Immunodeficiences in chronic muco-cutaneous candidosis. Immunology 1972, 22, 755–787. [Google Scholar]
- Hiller, J.; Hagl, B.; Effner, R.; Puel, A.; Schaller, M.; Mascher, B.; Eyerich, S.; Eyerich, K.; Jansson, A.F.; Ring, J.; et al. STAT1 gain-of-function and dominant negative STAT3 mutations impair IL-17 and IL-22 immunity associated with CMC. J. Investig. Dermatol. 2018, 138, 711–714. [Google Scholar] [CrossRef] [Green Version]
- Terai, H.; Shimahara, M. Usefulness of culture test and direct examination for the diagnosis of oral atrophic candidiasis. Int. J. Dermatol. 2009, 48, 371–373. [Google Scholar] [CrossRef]
- Coronado-Castellote, L.; Jiménez-Soriano, Y. Clinical and microbiological diagnosis of oral candidiasis. J. Clin. Exp. Dent. 2013, 5, e279–e286. [Google Scholar] [CrossRef] [Green Version]
- Ramani, R.; Gromadzki, S.; Pincus, D.H.; Salkin, I.F.; Chaturvedi, V. Efficacy of API 20C and ID 32C systems for identification of common and rare clinical yeast isolates. J. Clin. Microbiol. 1998, 36, 3396–3398. [Google Scholar] [CrossRef] [Green Version]
- Singhal, N.; Kumar, M.; Kanaujia, P.K.; Virdi, J.S. MALDI-TOF mass spectrometry: An emerging technology for microbial identification and diagnosis. Front. Microbiol. 2015, 6, 791. [Google Scholar] [CrossRef] [Green Version]
- Bal, A.M.; McGill, M. Rapid species identification of Candida directly from blood culture broths by Sepsityper-MALDI-TOF mass spectrometry: Impact on antifungal therapy. J. R. Coll Physicians Ediinb. 2018, 48, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Bessman, J.D.; Gilmer, P.R., Jr.; Gardner, F.H. Improved classification of anemias by MCV and RDW. Am. J. Clin. Pathol. 1983, 80, 322–326. [Google Scholar] [CrossRef]
- Bertero, M.T.; Caligaris-Cappio, F. Anemia of chronic disorders in systemic autoimmune diseases. Haematology 1997, 82, 375–381. [Google Scholar]
- Wu, Y.C.; Wang, Y.P.; Chang, J.Y.F.; Cheng, S.J.; Chen, H.M.; Sun, A. Oral manifestations and blood profile in patients with iron deficiency anemia. J. Formos. Med. Assoc. 2014, 113, 83–87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tyldesley, W.R. Stomatitis and recurrent oral ulcerations: Is a full blood screen necessary? Br. J. Oral Surg. 1983, 21, 27–30. [Google Scholar] [CrossRef]
- Marti, H.R.; Fischer, S.; Killer, D.; Burgi, W. Can automated hematology analysers discriminate thalassemia from iron deficiency? Acta. Haemat. 1987, 78, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.P.; Chang, J.Y.F.; Wu, Y.C.; Cheng, S.J.; Chen, H.M.; Sun, A. Oral manifestaions and blood profile in patients with thalassemia trait. J. Formos. Med. Assoc. 2013, 112, 761–765. [Google Scholar] [CrossRef] [Green Version]
- Lynch, D.P. Oral candidiasis: History, classification, and clinical presentation. Oral Surg. Oral Med. Oral Pathol. 1994, 78, 189–193. [Google Scholar] [CrossRef]
- Holmstrup, P.; Axéll, T. Classification and clinical manifestations of oral yeast infections. Acta. Odontol. Scand. 1990, 48, 57–59. [Google Scholar] [CrossRef]
- Sitheeque, M.A.M.; Samranayake, L.P. Chronic hyperplastic candidosis/ candidiasis (Candidal Leukoplakia). Crit. Rev. Oral Biol. Med. 2003, 14, 253–267. [Google Scholar] [CrossRef]
- Odds, F.C. Chronic mucocutaneous candidosis. In Candida and Candidosis; Bailliere Tindall: London, UK, 1988; pp. 143–152. [Google Scholar]
- Terai, H.; Shimahara, M. Atrophic tongue associated with Candida. J. Oral Pathol. Med. 2005, 34, 397–400. [Google Scholar] [CrossRef]
- Cooke, B.E.D. Median rhomboid glossitis: Candidosis and not a developmental anomaly. Br. J. Dermato. 1975, 93, 399–405. [Google Scholar] [CrossRef]
- Van der Waal, I.; Beemster, G.; van der Kwast, W.A.M. Median rhomboid glossitis caused by Candida? Oral Surg. Oral Med. Oral pathol. 1979, 47, 31–35. [Google Scholar] [CrossRef]
- Gendreau, L.; Loewy, Z.G. Epidemiology and etiology of denture stomatitis. J. Prostho. 2011, 20, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Cawson, R.A. Induction of epithelial hyperplasia by Candida albicans. Bri. J. Derma. 1973, 89, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, R.L. The etiology of inflammatory papillary hyperplasia. J. Prosth. Dent. 1975, 34, 254–261. [Google Scholar] [CrossRef]
- Salehi, M.; Ahmadikia, K.; Mahmoudi, S.; Kalantari, S.; Jamalimoghadamsiahkali, S.; Izadi, A.; Kord, M.; Manshadi, S.A.D.; Seifi, A.; Ghiasvand, F.; et al. Oropharyngeal candidiasis in hospitalized COVID-19 patients from Iran: Species identification and antifungal susceptibility pattern. Mycoses 2020, 63, 771–778. [Google Scholar] [CrossRef]
- Shay, K.; Truhlar, M.R.; Renner, R.P. Oropharyngeal candidosis in the older patient. J. Am. Geriatr. Soc. 1997, 45, 863–870. [Google Scholar] [CrossRef] [Green Version]
- Pinel, B.; Cassou-Mounat, T.; Bensadoun, R.J. Oropharyngeal candidiasis and radiotherapy. Cancer Radiother. 2012, 16, 222–229. [Google Scholar] [CrossRef]
- Gallo, W.J.; Shapiro, D.N.; Moss, M. Suppurative candidiasis: Review of the literature and report of case. J. Am. Dent. Assoc. 1976, 92, 936–939. [Google Scholar] [CrossRef]
- McManners, J.; Samaranayake, L.P. Suppurative oral candidosis-review of the literature and report of a case. Int. J. Oral Maxillofac. Surg. 1990, 19, 257–259. [Google Scholar] [CrossRef]
- Bouquet, J.E.; Fenton, S.J. Juvenile juxtavermilion candidiasis: Yet another form of an old disease? J. Am. Dent. Assoc. 1988, 116, 187–192. [Google Scholar] [CrossRef]
- Lee, S.Y.; Choi, J.Y.; Kim, J.W.; Yu, D.S.; Lee, Y.B. A case of cheilocandidiasis. Ann. Dermatol. 2019, 31, 22–23. [Google Scholar] [CrossRef]
- Williams, D.; Lewis, M. Pathogenesis and treatment of oral candidosis. J. Oral Microbiol. 2011, 3, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Xin, L.; Chen, Z.; Zhi-min, Y.; Hong, H. Efficacy of nystatin for the treatmemt of oral candidiasis: A systemic review and meta-analysis. Drug Des. Dev. Ther. 2016, 10, 1161–1171. [Google Scholar]
- Barkvoll, P.; Attramadal, A. Effect of nystatin and chlorhexidine digluconate on Candida albicans. Oral Surg. Oral Med. Oral Pathol. 1989, 67, 279–281. [Google Scholar] [CrossRef]
- Teva Pharmaceuticals USA Inc. Ketoconazole Tablets Prescribing Information; Teva Pharmaceuticals USA Inc.: North Wales, PA, USA, 2015; p. 387. [Google Scholar]
- Janssen Pharmaceuticals, Inc. Nizoral (Ketoconazole) Tablets Prescribing Information; Janssen Pharmaceuticals, Inc.: Titusville, NJ, USA, 2013. [Google Scholar]
- Brammer, K.W.; Farrow, P.R.; Faulkner, J.K. Clinical pharmacology—Pharmacokinetics and tissue/fluid penetration of fluconazole in humans. Rev. Infect. Dis. 1990, 12, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Shiba, K.; Saito, A.; Miyahara, T. Safety and pharmacokinetics of single oral and intravenous doses of fluconazole in healthy subjects. Clin. Pharmacol. Ther. 1990, 12, 206–215. [Google Scholar]
- Troke, P.F.; Andrews, R.J.; Pye, G.W.; Richardson, K. Fluconazole and other azoles: Translation of in vitro activity to in vivo and clinical efficacy. Rev. Infect. Dis. 1990, 12, 276–280. [Google Scholar] [CrossRef]
- Golin, R.A.; Ascher, D.; Waecker, N.; Arnold, J.; Moorefield, E. Comparison of fluconazole and nystatin oral suspensions for treatment of oral candidiasis in infants. Pediatric Infec. Dis. J. 2002, 21, 1165–1167. [Google Scholar]
- Allen, U. Antifungal agents for the treatment of systemic fungal infections in children. Can. J. Infec. Dis. Med. Microbiol. 2010, 21, e116–e121. [Google Scholar] [CrossRef]
- Benitez, L.L.; Carver, P.L. Adverse effects associated with long-term administration of azole antifungal agents. Drugs 2019, 79, 833–853. [Google Scholar] [CrossRef] [PubMed]
| Systemic Host Factors | Local Host Factors |
|---|---|
| Altered physiological status | Mucosal barrier alterations |
| Infancy/old age | Exogenous epithelial change |
| Altered hormonal status | Trauma |
| Diabetes | Loss of occlusion |
| Hypothyroidism/Hypoparathyroidism | Maceration |
| Cushing’s syndrome | Endogenous epithelial changes |
| Altered hematinic or nutritional status | Atrophy |
| Iron deficiency | Hyperplasia |
| Hypovitaminosis, Vit B12, folic acid | Dysplasia/Oral cancer |
| Malnutrition | Saliva quantitative changes |
| Altered immune status | Xerostomia |
| Defects in cell-mediated immunity | Sjogren’s syndrome |
| Reduced numbers of phagocytes | Radiotherapy/Cytotoxic therapy |
| Lymphopenia or leukopenia | Saliva qualitative changes |
| Decreased CD4 count | pH/glucose concentration |
| Due to infective states/ HIV | Poor oral or denture hygiene |
| Blood dyscrasias/ malignancies | High carbohydrate diet |
| Immunosuppressant/ chemotherapy | Heavy smoking/ Betel nut chewing |
| Broad spectrum antibiotics | Inhaled steroid |
| Virulence Factors | Effects |
|---|---|
| Adherence | Promote retention in the mouth |
| Expression of cell surface adhesins | Specific adhesion |
| Cell surface hydrophobicity | Nonspecific adhesion |
| Invasion and destruction of host tissue | Enhance pathogenicity |
| Hyphae development/thigmotropism | Promote invasion/active penetration |
| Secret hydrolytic enzymes | Cytotoxicity to oral epithelium |
| Secret acidic metabolites | Degrade basement membrane/matrix |
| Endocytosis | Passive penetration of epithelium |
| Evasion of host defense | Reduce phagocytosis/help retention |
| Phenotypic switching | Antigenic modification |
| Proteolytic degrading immune factors | Destroy sIgA/ antimicrobial peptides |
| Binding of complement | Antigenic masking |
| Synergism with bacteria | Promote mixed-species retention |
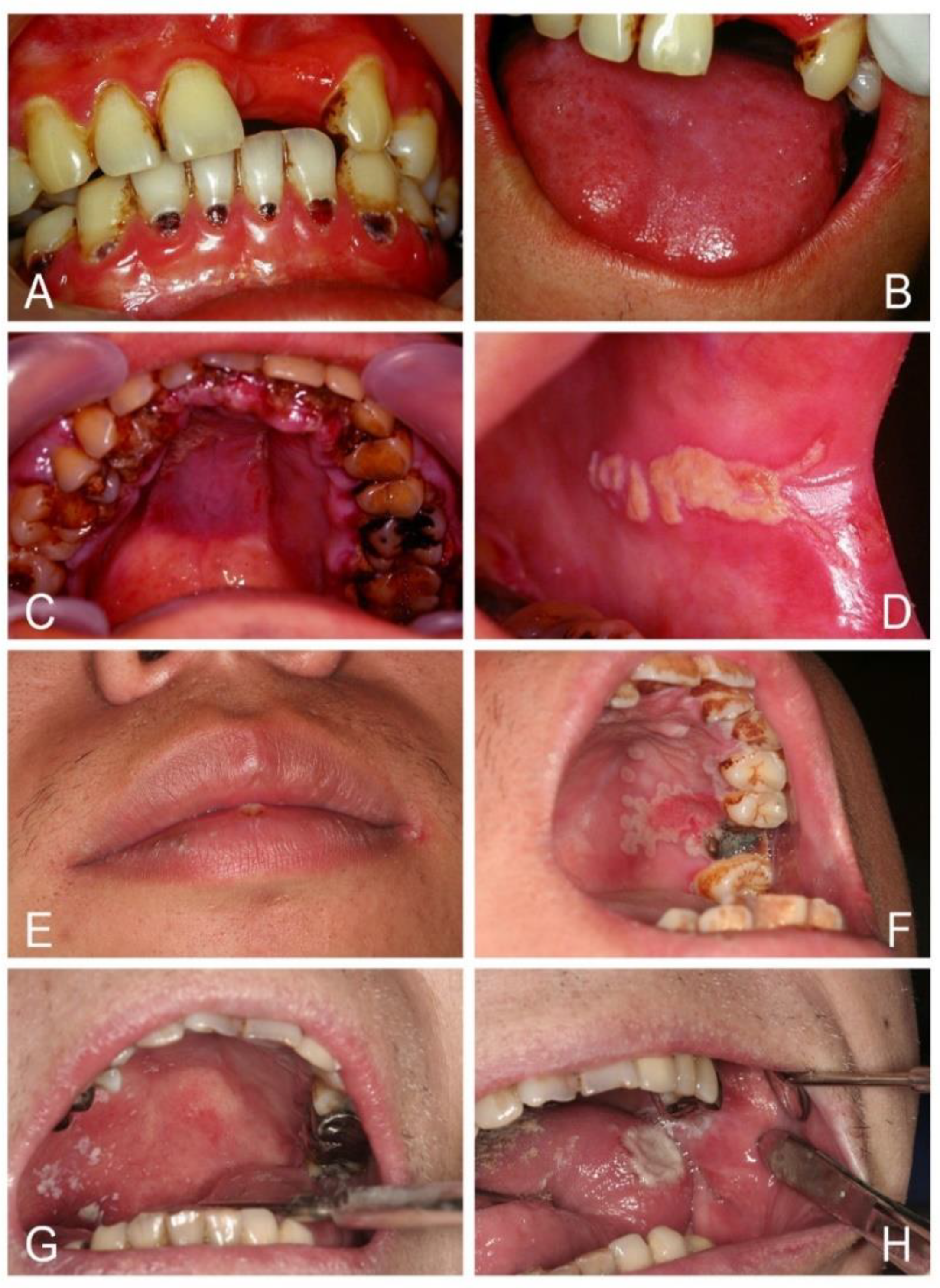
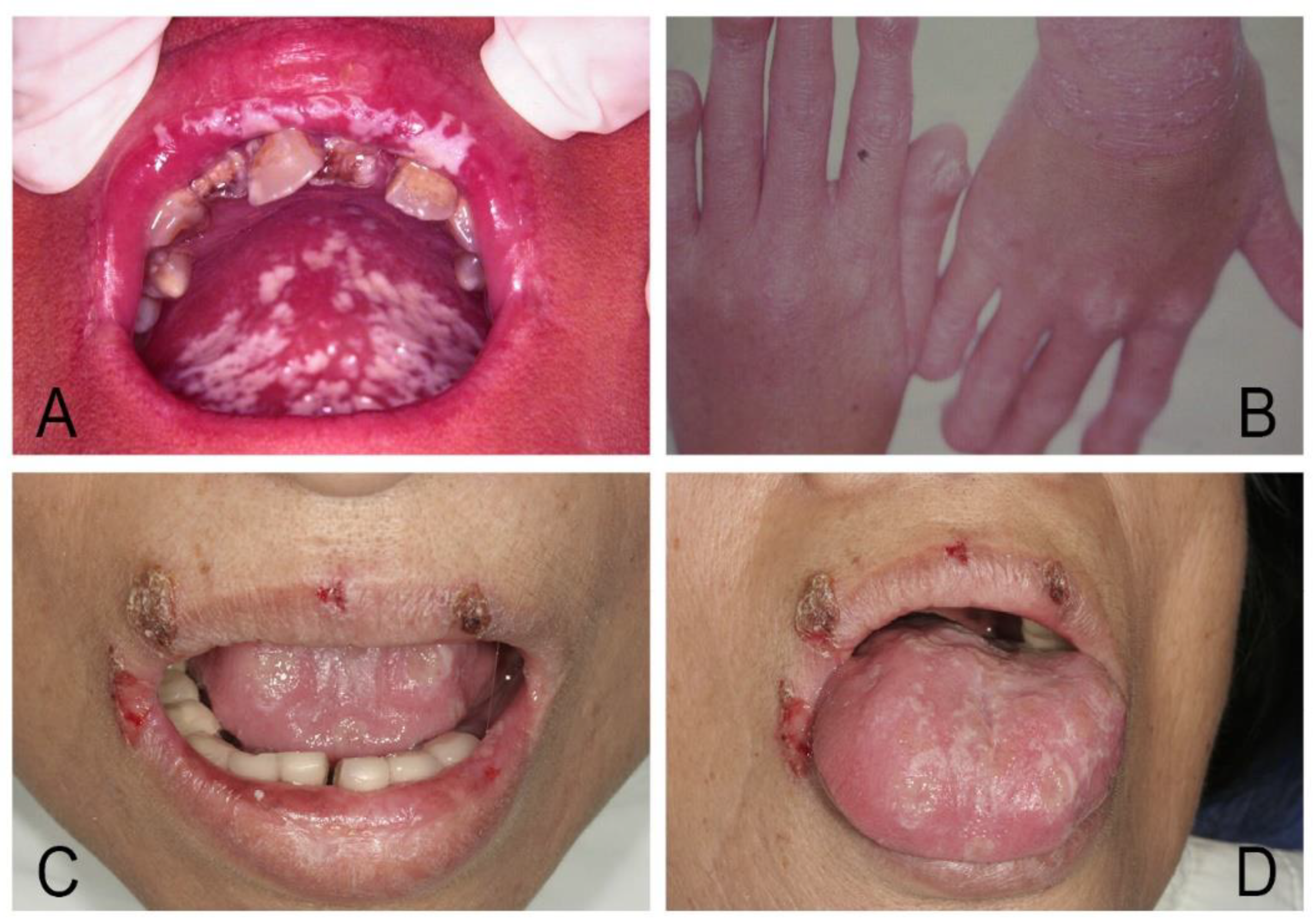
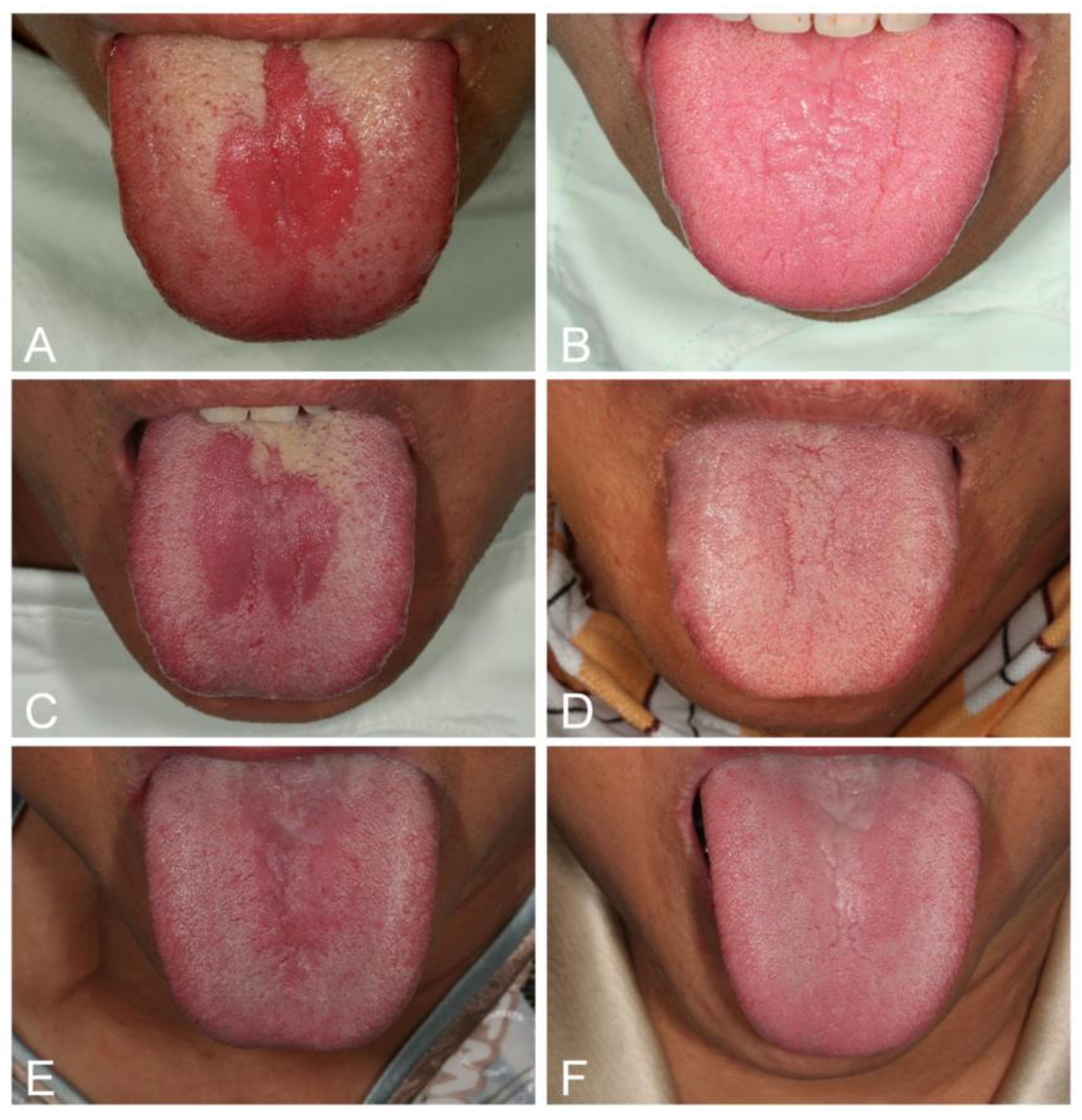
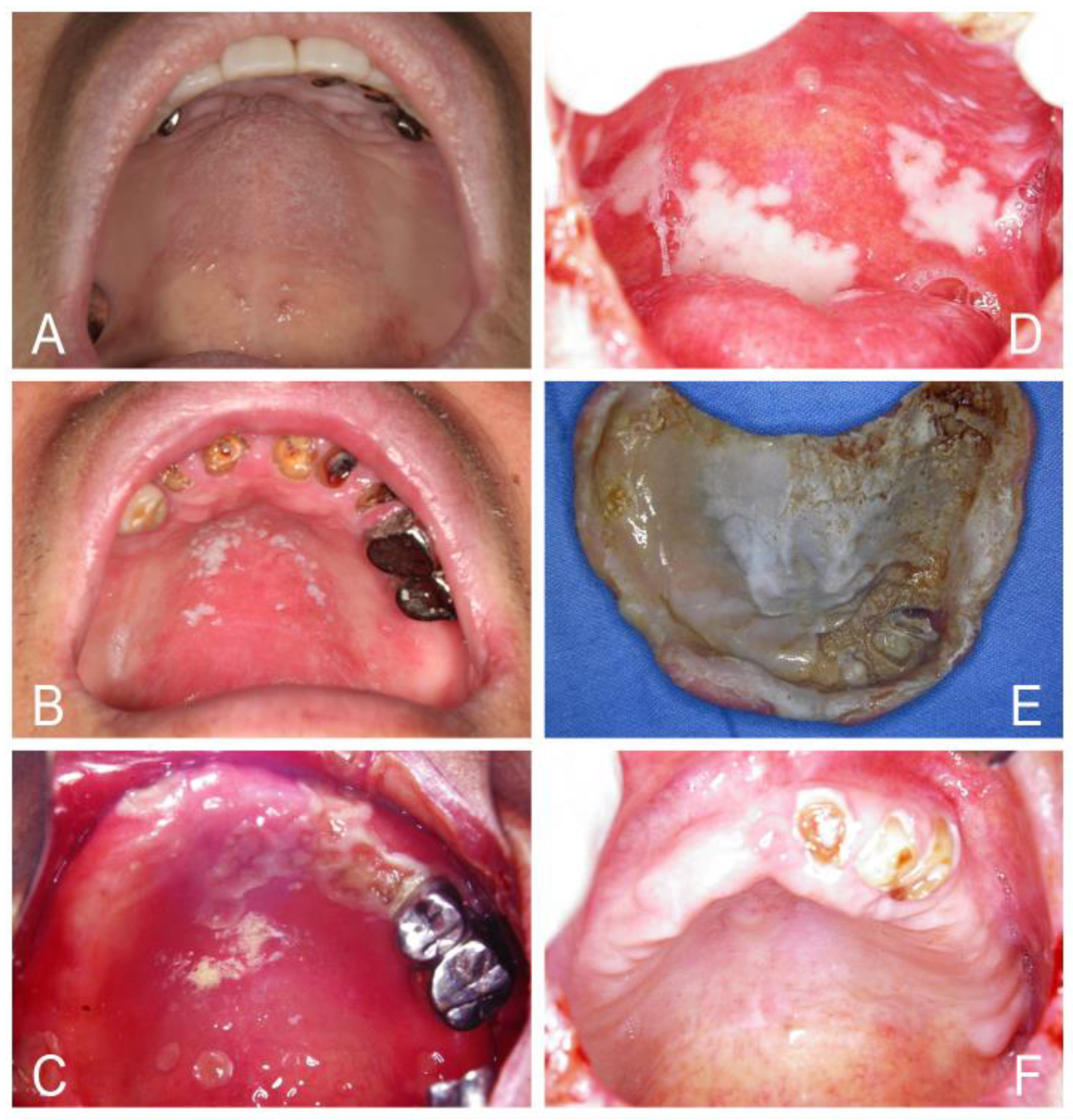
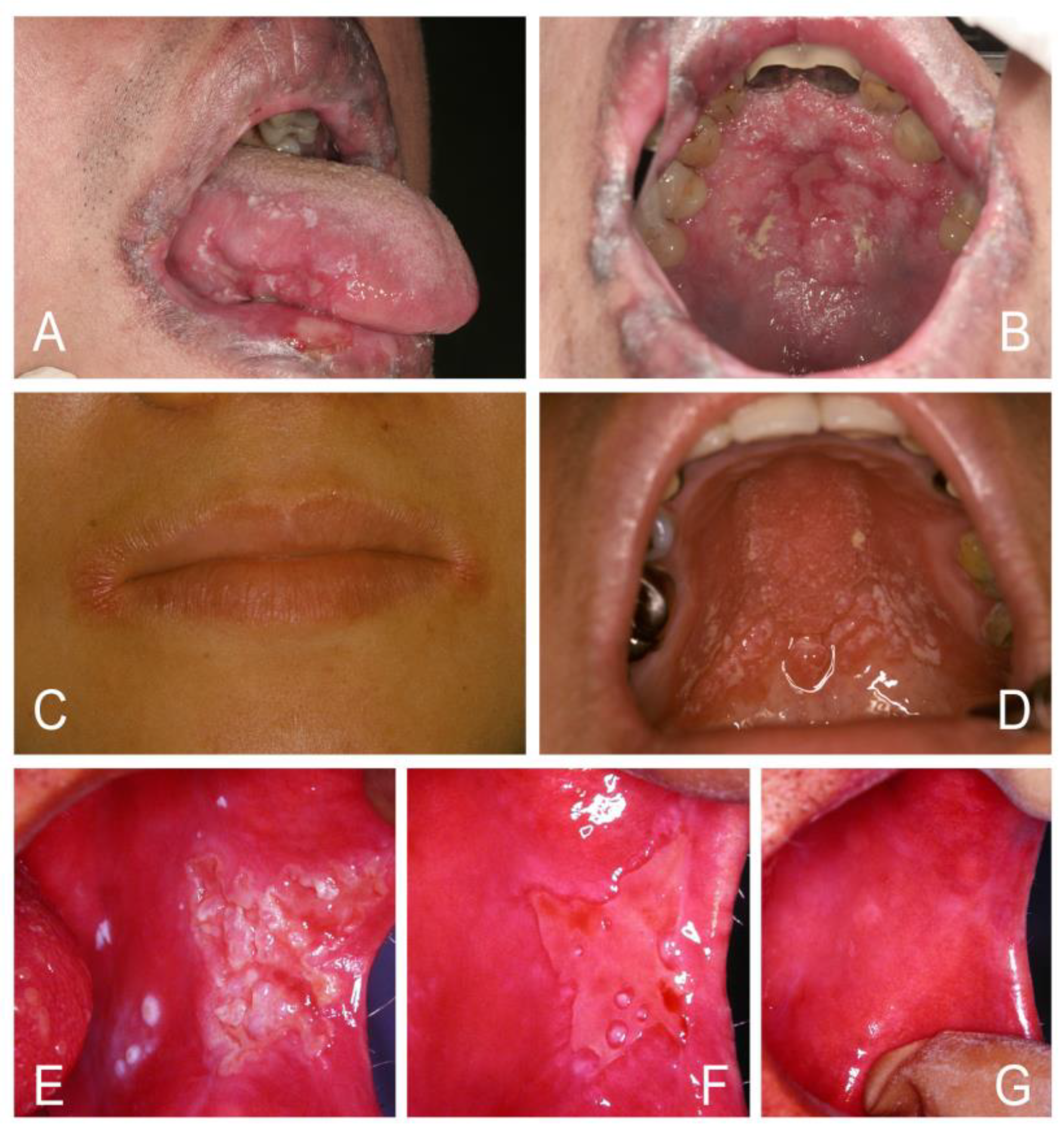
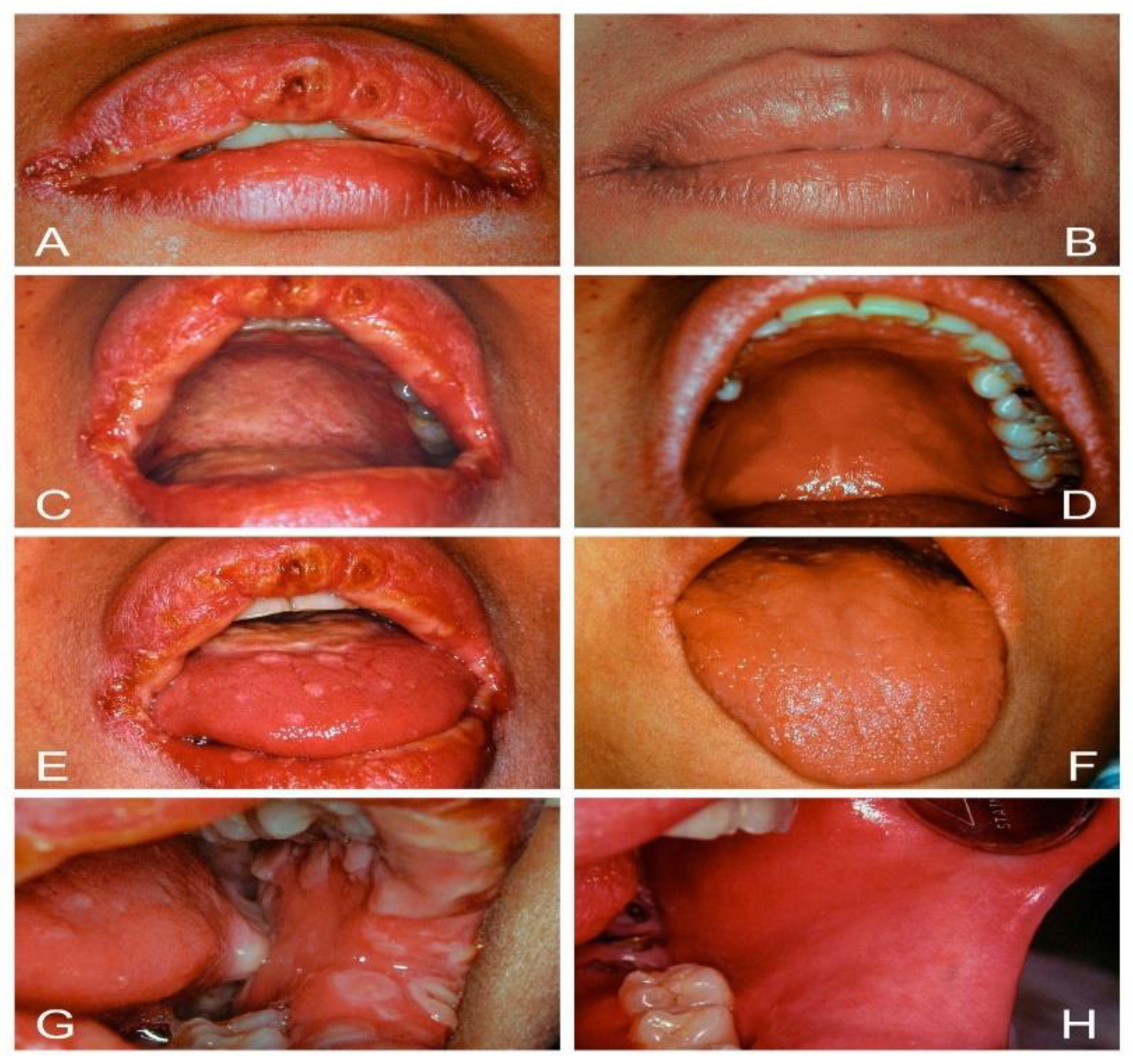
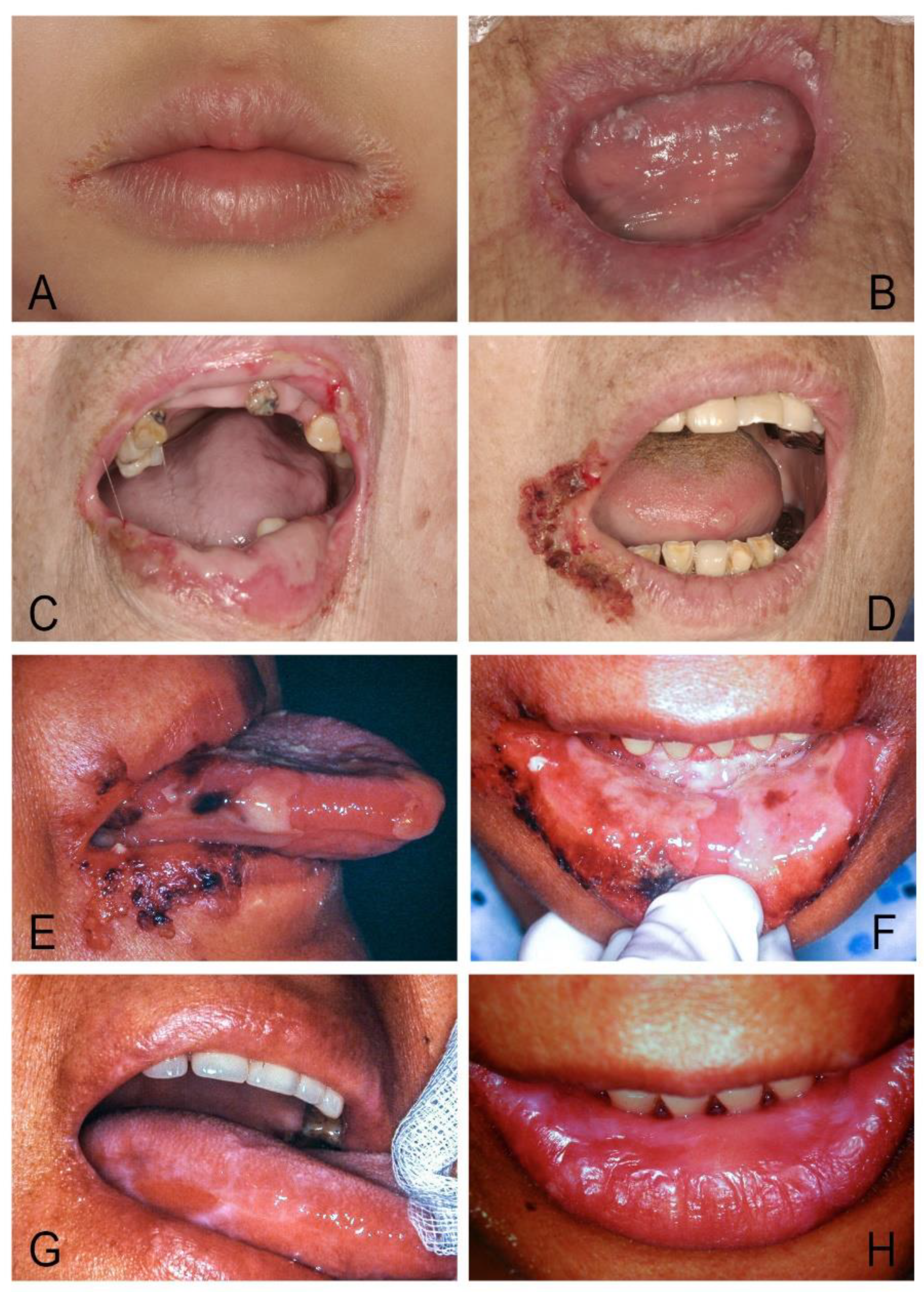
| Acute/Chronic Candidosis | Hyperplastic Candidosis |
| Pseudomembranous candidosis | Plaque-like |
| Erythematous candidosis | Nodular-like |
| Candida-associated lesions | Granular or papillary-like * |
| Denture stomatitis | Keratinized primary lesions superinfected with Candida |
| Localized simple inflammation | Leukoplakia |
| Diffuse erythematous type | Oral lichen planus |
| Granular type (inflammatory papillary hyperplasia) * | Lupus erythematosus |
| Median rhomboid glossitis (MRG) | Chronic mucocutaneous candidosis (CMC) |
| Angular cheilitis | Congenital/ Familial CMC, diffuse type |
| Cheilocandidosis | Acquired CMC, focal, or diffuse type |
| Juxtavermillion candidosis | Endocrinopathy associated |
| Oropharyngeal candidosis | Hypothyroidism, hypoparathyroidism, |
| Dangerous sign of immunosuppression | MG-thymoma, Addison’s disease, etc. |
| Suppurative oral candidosis | Immunosuppression associated |
| Focal necrotizing ulcerative mucositis or osteomyelitis | Diabetes, iron deficiency, HIV/AIDS, neutropenia, etc. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, S.-Y. Oral Candidosis: Pathophysiology and Best Practice for Diagnosis, Classification, and Successful Management. J. Fungi 2021, 7, 555. https://doi.org/10.3390/jof7070555
Lu S-Y. Oral Candidosis: Pathophysiology and Best Practice for Diagnosis, Classification, and Successful Management. Journal of Fungi. 2021; 7(7):555. https://doi.org/10.3390/jof7070555
Chicago/Turabian StyleLu, Shin-Yu. 2021. "Oral Candidosis: Pathophysiology and Best Practice for Diagnosis, Classification, and Successful Management" Journal of Fungi 7, no. 7: 555. https://doi.org/10.3390/jof7070555
APA StyleLu, S.-Y. (2021). Oral Candidosis: Pathophysiology and Best Practice for Diagnosis, Classification, and Successful Management. Journal of Fungi, 7(7), 555. https://doi.org/10.3390/jof7070555





