Production of a β-Glucosidase-Rich Cocktail from Talaromyces amestolkiae Using Raw Glycerol: Its Role for Lignocellulose Waste Valorization
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals, Microorganisms, Culture Media, and Crude Enzyme Extract Production
2.2. Determination of Biomass, Proteins, Enzyme Activity, and Glycerol Consumption
2.3. Proteomic Analysis of T. amestolkiae Secretomes
2.4. Saccharification of Pretreated Wheat Straw
- -
- 2 U of β-glucosidase activity from Celluclast 1.5 L (Novozymes, Copenhagen, Denmark). This is a commercial cocktail, rich in cellobiohydrolase and endoglucanase activities, and was used for comparison purposes;
- -
- 1 U of β-glucosidase activity from Celluclast 1.5 L + 1 U of β-glucosidase activity from T. amestolkiae enzymatic crudes obtained in this work, using 1% glycerol as the carbon source.
3. Results
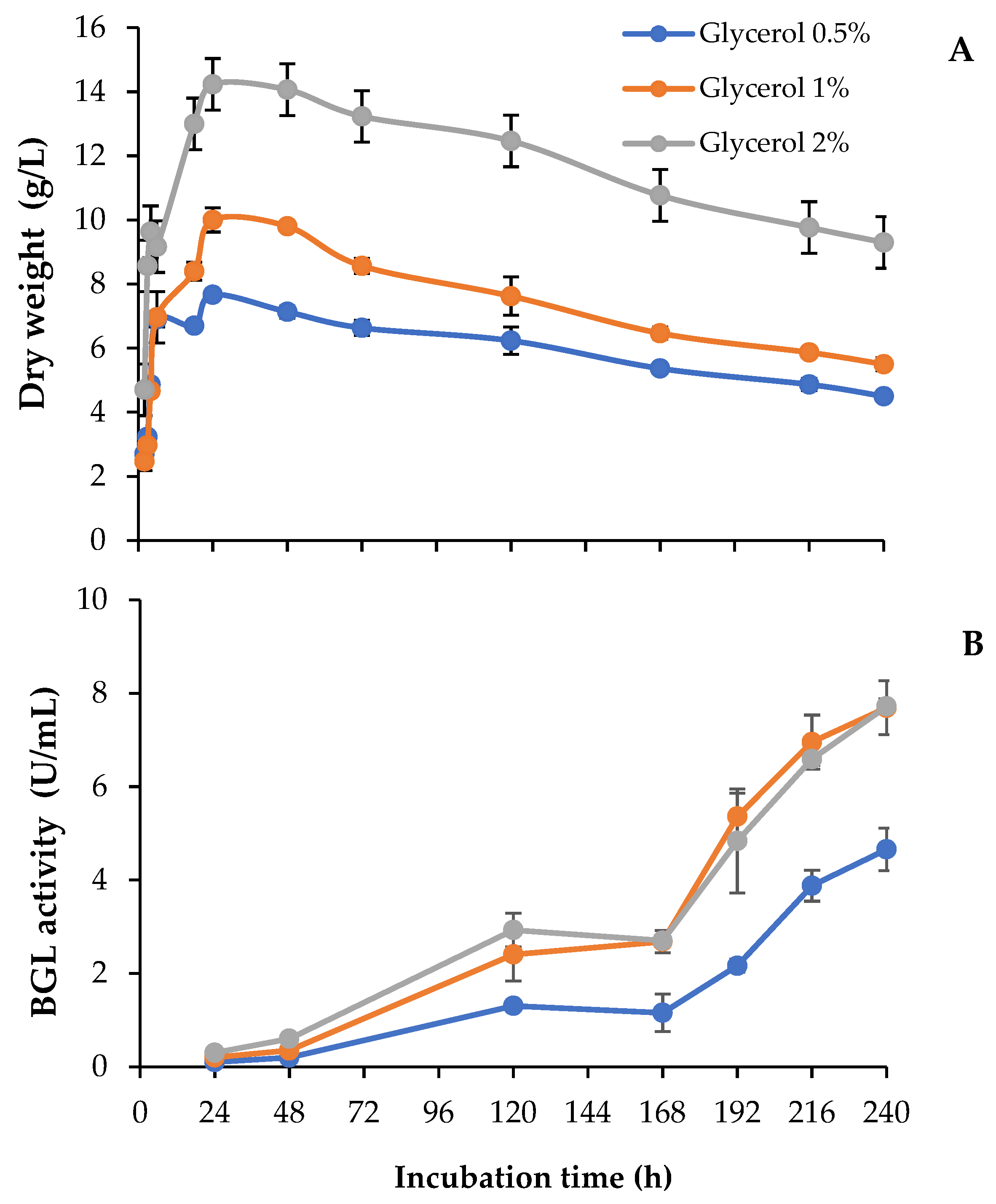
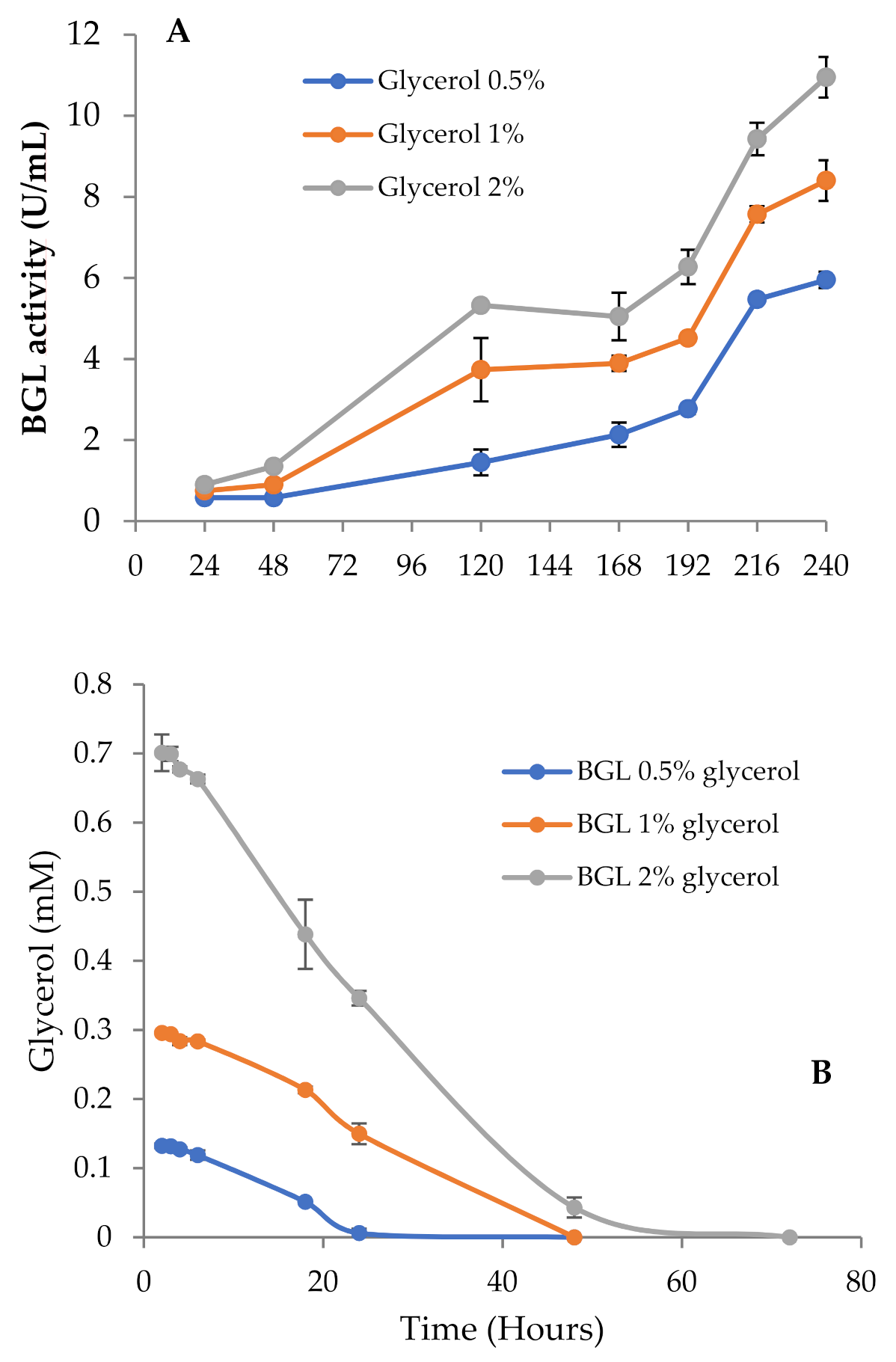
3.1. BGL Production and Growth of T. amestolkiae in Media with Raw Glycerol
3.2. Fungal Secretome Analysis
3.3. Wheat Straw Saccharification
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giusti, L. A review of waste management practices and their impact on human health. WASTE Manag. 2009, 29, 2227–2239. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, M.R.; Kugelmeier, C.L.; Pinheiro, R.S.; Batalha, M.O.; da Silva César, A. Glycerol from biodiesel production: Technological paths for sustainability. Renew. Sustain. Energy Rev. 2018, 88, 109–122. [Google Scholar] [CrossRef]
- Lapinskiene, A.; Martinkus, P.; Rebzdaite, V. Eco-toxicological studies of diesel and biodiesel fuels in aerated soil. Environ. Pollut. 2006, 142, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Boehme, L.; Lam, H.; Zhang, Z. Pichia pastoris fermentation for phytase production using crude glycerol from biodiesel production as the sole carbon source. Biochem. Eng. J. 2009, 43, 157–162. [Google Scholar] [CrossRef]
- Luo, X.; Ge, X.; Cui, S.; Li, Y. Value-added processing of crude glycerol into chemicals and polymers. Bioresour. Technol. 2016, 215, 144–154. [Google Scholar] [CrossRef]
- Magdouli, S.; Guedri, T.; Tarek, R.; Brar, S.K.; Blais, J.F. Valorization of raw glycerol and crustacean waste into value added products by Yarrowia lipolytica. Bioresour. Technol. 2017, 243, 57–68. [Google Scholar] [CrossRef]
- Yang, F.; Hanna, M.A.; Sun, R. Value added use crude glycerol-a byproduct of biodiesel production. Biotechnol. Biofuels 2012, 5, 1–10. [Google Scholar]
- Guo, H.; Chang, Y.; Lee, D.J. Enzymatic saccharification of lignocellulosic biorefinery: Research focuses. Bioresour. Technol. 2018, 252, 198–215. [Google Scholar] [CrossRef]
- de Eugenio, L.; Méndez-Líter, J.; Nieto-Domínguez, M.; Alonso, L.; Gil-Muñoz, J.; Barriuso, J.; Prieto, A.; Martínez, M. Differential β-glucosidase expression as a function of carbon source availability in Talaromyces amestolkiae: A genomic and proteomic approach. Biotechnol. Biofuels 2017, 10, 1–14. [Google Scholar] [CrossRef]
- Méndez-Líter, J.A.; Gil-Muñoz, J.; Nieto-Domínguez, M.; Barriuso, J.; De Eugenio, L.I.; Martínez, M.J. A novel, highly efficient β-glucosidase with a cellulose-binding domain: Characterization and properties of native and recombinant proteins. Biotechnol. Biofuels 2017, 10, 1–15. [Google Scholar] [CrossRef]
- Méndez-Líter, J.A.; de Eugenio, L.I.; Prieto, A.; Martínez, M.J. The β-glucosidase secreted by Talaromyces amestolkiae under carbon starvation: A versatile catalyst for biofuel production from plant and algal biomass. Biotechnol. Biofuels 2018, 11, 1–14. [Google Scholar] [CrossRef]
- de Sousa, M.H.; da Silva, A.S.F.; Correia, R.C.; Leite, N.P.; Bueno, C.E.G.; dos Santos Pinheiro, R.L.; de Santana, J.S.; da Silva, J.L.; Sales, A.T.; de Souza, C.C.; et al. Valorizing municipal organic waste to produce biodiesel, biogas, organic fertilizer, and value-added chemicals: An integrated biorefinery approach. Biomass Convers. Biorefinery 2021. [Google Scholar] [CrossRef]
- Mandels, M.; Weber, J. The production of cellulases. In Cellulases and Their Applications; American Chemistry Society: Washington, DC, USA, 1969; pp. 391–414. ISBN 0-8412-0095-5. [Google Scholar]
- Kall, L.; Krogh, A.; Sonnhammer, E.L.L. A combined transmembrane topology and signal peptide prediction method. J. Mol. Biol. 2004, 338, 1027–1036. [Google Scholar] [CrossRef]
- Liu, H.B.; Sadygov, R.G.; Yates, J.R. A model for random sampling and estimation of relative protein abundance in shotgun proteomics. Anal. Chem. 2004, 76, 4193–4201. [Google Scholar] [CrossRef]
- Schulze, W.X.; Usadel, B. Quantitation in Mass-Spectrometry-Based Proteomics. Annu. Rev. Plant Biol. 2010, 61, 491–516. [Google Scholar] [CrossRef]
- Mardones, W.; Di Genova, A.; Cortés, M.P.; Travisany, D.; Maass, A.; Eyzaguirre, J. The genome sequence of the soft-rot fungus Penicillium purpurogenum reveals a high gene dosage for lignocellulolytic enzymes. Mycology 2018, 9, 59–69. [Google Scholar] [CrossRef]
- Notararigo, S.; Nacher-Vazquez, M.; Ibarburu, I.; Laura Werning, M.; de Palencia, P.; Teresa Duenas, M.; Aznar, R.; Lopez, P.; Prieto, A. Comparative analysis of production and purification of homo- and hetero-polysaccharides produced by lactic acid bacteria. Carbohydr. Polym. 2013, 93, 57–64. [Google Scholar] [CrossRef]
- Schneider, W.D.H.; Goncalves, T.A.; Uchima, C.A.; Couger, M.B.; Prade, R.; Squina, F.M.; Pinheiro Dillon, A.J.; Camassola, M. Penicillium echinulatum secretome analysis reveals the fungi potential for degradation of lignocellulosic biomass. Biotechnol. Biofuels 2016, 9. [Google Scholar] [CrossRef]
- Abdella, A.; Mazeed, T.E.-S.; El-Baz, A.F.; Yang, S.-T. Production of beta-glucosidase from wheat bran and glycerol by Aspergillus niger in stirred tank and rotating fibrous bed bioreactors. Process Biochem. 2016, 51, 1331–1337. [Google Scholar] [CrossRef]
- Ramani, G.; Meera, B.; Vanitha, C.; Rao, M.; Gunasekaran, P. Production, Purification, and Characterization of a beta-Glucosidase of Penicillium funiculosum NCL1. Appl. Biochem. Biotechnol. 2012, 167, 959–972. [Google Scholar] [CrossRef]
- Vaishnav, N.; Singh, A.; Adsul, M.; Dixit, P.; Sandhu, S.K.; Mathur, A.; Puri, S.K.; Singhania, R.R. Penicillium: The next emerging champion for cellulase production. Bioresour. Technol. Rep. 2018, 2, 131–140. [Google Scholar] [CrossRef]
- Decker, C.H.; Visser, J.; Schreier, P. beta-glucosidases from five black Aspergillus species: Study of their physico-chemical and biocatalytic properties. J. Agicult. Food Chem. 2000, 48, 4929–4936. [Google Scholar] [CrossRef] [PubMed]
- Riou, C.; Salmon, J.M.; Vallier, M.J.; Gunata, Z.; Barre, P. Purification, characterization, and substrate specificity of a novel highly glucose-tolerant beta-glucosidase from Aspergillus oryzae. Appl. Environ. Microbiol. 1998, 64, 3607–3614. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, H.; Olsson, L. Production of cellulases by Penicillium brasilianum IBT 20888 - Effect of substrate on hydrolytic performance. Enzyme Microb. Technol. 2006, 38, 381–390. [Google Scholar] [CrossRef]
- Sun, X.; Liu, Z.; Zheng, K.; Song, X.; Qu, Y. The composition of basal and induced cellulase system in Penicillium decumbens under induction or repression conditions. Enzym. Microb. Technol. 2008, 42, 560–567. [Google Scholar] [CrossRef]
- Chaabouni, S.E.; Hadjtaieb, N.; Mosrati, R.; Ellouz, R. Preliminary assessment of Penicillium occitanis cellulase: A futher useful system. Enzyme Microb. Technol. 1994, 16, 538–542. [Google Scholar] [CrossRef]
- Solov’eva, I.V.; Okunev, O.N.; Vel’kov, V.V.; Koshelev, A.V.; Bubnova, T.V.; Kondrat’eva, E.G.; Skomarovskii, A.A.; Sinitsyn, A.P. The selection and properties of Penicillium verruculosum mutants with enhanced production of cellulases and xylanases. Microbioloy 2005, 74, 141–146. [Google Scholar] [CrossRef]
- Nitsche, B.M.; Jorgensen, T.R.; Akeroyd, M.; Meyer, V.; Ram, A.F.J. The carbon starvation response of Aspergillus niger during submerged cultivation: Insights from the transcriptome and secretome. BMC Genom. 2012, 13. [Google Scholar] [CrossRef]
- van Munster, J.M.; Daly, P.; Delmas, S.; Pullan, S.T.; Blythe, M.J.; Malla, S.; Kokolski, M.; Noltorp, E.C.M.; Wennberg, K.; Fetherston, R.; et al. The role of carbon starvation in the induction of enzymes that degrade plant-derived carbohydrates in Aspergillus niger. Fungal Genet. Biol. 2014, 72, 34–47. [Google Scholar] [CrossRef]
- Prieto, A.; Bernabe, M.; Leal, J.A. Isolation, purification and chemical characterization of alkali-extractable polysacharides from the cell-walls of Talaromyces species. Mycol. Res. 1995, 99, 69–75. [Google Scholar] [CrossRef]
- Méndez-Líter, J.A.; Nieto-Domínguez, M.; Fernández De Toro, B.; González Santana, A.; Prieto, A.; Asensio, J.L.; Cañada, F.J.; De Eugenio, L.I.; Martínez, M.J. A glucotolerant β-glucosidase from the fungus Talaromyces amestolkiae and its conversion into a glycosynthase for glycosylation of phenolic compounds. Microb. Cell Fact. 2020, 19, 1–13. [Google Scholar] [CrossRef]
- Iskalieva, A.; Yimmou, B.M.; Gogate, P.R.; Horvath, M.; Horvath, P.G.; Csoka, L. Cavitation assisted delignification of wheat straw: A review. Ultrason. Sonochem. 2012, 19, 984–993. [Google Scholar] [CrossRef]
- Pan, X.J.; Xie, D.; Gilkes, N.; Gregg, D.J.; Saddler, J.N. Strategies to enhance the enzymatic hydrolysis of pretreated softwood with high residual lignin content. Appl. Biochem. Biotechnol. 2005, 121, 1069–1079. [Google Scholar] [CrossRef]
- Galbe, M.; Wallberg, O. Pretreatment for biorefineries: A review of common methods for efficient utilisation of lignocellulosic materials. Biotechnol. Biofuels 2019, 12, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Aparicio, M.; Ballesteros, I.; Gonzalez, A.; Oliva, J.; Ballesteros, M.; Negro, M. Effect of inhibitors released during steam-explosion pretreatment of barley straw on enzymatic hydrolysis. Appl. Biochem. Biotechnol. 2006, 129, 278–288. [Google Scholar] [CrossRef]
- Kumar, R.; Wyman, C.E. Effects of cellulase and xylanase enzymes on the deconstruction of solids from pretreatment of poplar by leading technologies. Biotechnol. Prog. 2009, 25, 302–314. [Google Scholar] [CrossRef]
- Moreno, A.D.; Ibarra, D.; Alvira, P.; Tomás-Pejó, E.; Ballesteros, M. A review of biological delignification and detoxification methods for lignocellulosic bioethanol production. Crit. Rev. Biotechnol. 2015, 35, 342–354. [Google Scholar] [CrossRef]
- Gao, D.; Haarmeyer, C.; Balan, V.; Whitehead, T.A.; Dale, B.E.; Chundawat, S.P.S. Lignin triggers irreversible cellulase loss during pretreated lignocellulosic biomass saccharification. Biotechnol. Biofuels 2014, 7. [Google Scholar] [CrossRef]
- Yarbrough, J.M.; Mittal, A.; Mansfield, E.; Taylor II, L.E.; Hobdey, S.E.; Sammond, D.W.; Bomble, Y.J.; Crowley, M.F.; Decker, S.R.; Himmel, M.E.; et al. New perspective on glycoside hydrolase binding to lignin from pretreated corn stover. Biotechnol. Biofuels 2015, 8. [Google Scholar] [CrossRef]
- Lima, M.A.; Oliveira-Neto, M.; Kadowaki, M.A.S.; Rosseto, F.R.; Prates, E.T.; Squina, F.M.; Leme, A.F.P.; Skaf, M.S.; Polikarpov, I. Aspergillus niger beta-Glucosidase Has a cellulase-like tadpole molecular shape: Insights into gycoside hydrolase famyly 3 (GH3) beta-glucosidase structure and function. J. Biol. Chem. 2013, 288, 32991–33005. [Google Scholar] [CrossRef]


| % PSM | ||
|---|---|---|
| Glycerol | Glucose | |
| A—RNA processing and modification | 0.63 | 0.22 |
| C—Energy production and conversion | 2.72 | 4.90 |
| E—Amino acid metabolism and transport | 13.11 | 10.38 |
| F—Nucleotide metabolism and transport | 0.74 | 1.42 |
| G—Carbohydrate metabolism and transport | 55.01 | 65.16 |
| I—Lipid metabolism | 0.09 | 0.17 |
| M—Cell wall/membrane/envelop biogenesis | 0.77 | 3.61 |
| O—Post-translational modification. protein turnover. chaperone functions | 1.96 | 1.71 |
| Q—Secondary structure | 2.55 | 0.91 |
| R—General functional prediction only | 4.81 | 1.89 |
| S—Function unknown | 5.79 | 3.71 |
| T—Signal transduction | 4.13 | 4.86 |
| % PSM | ||
|---|---|---|
| GH Family | Glycerol | Glucose |
| GH2 | 3.2 | 1.7 |
| GH3 | 16.6 | 16.3 |
| GH13 | 3.0 | 4.3 |
| GH15 | 10.1 | 28.4 |
| GH18 | 2.8 | 0.6 |
| GH20 | 3.7 | 4.8 |
| GH27 | 2.1 | 1.7 |
| GH31 | 8.6 | 11.8 |
| GH35 | 2.7 | 1.8 |
| GH55 | 8.1 | 3.9 |
| GH71 | 4.0 | 0.1 |
| GH72 | 3.2 | 1.3 |
| GH92 | 5.3 | 1.1 |
| GH127 | 3.6 | 3.2 |
| Accession ID | % PSM (Average) | Predicted Protein Function | Cazyme Family | Mw (kDa) |
|---|---|---|---|---|
| g377 (BGL-3) | 7.09 | beta-glucosidase | GH3 | 88.7 |
| g3995 | 6.47 | Glutaminase | - | 76.4 |
| g8295 | 3.59 | alpha-glucosidase | GH31 | 98.6 |
| g2158 | 3.28 | Glucoamylase | GH15 | 65.2 |
| g9324 | 3.18 | Exo-beta-1,3-glucanase | GH55 | 84.3 |
| g2140 | 2.50 | Glucoamylase | GH15 | 67.7 |
| g5915 | 2.23 | non-reducing end β-L-arabinofuranosidase | GH127 | 68.8 |
| g4076 | 2.00 | hexosaminidase | GH20 | 67.9 |
| g216 | 1.77 | neutral/alkaline nonlysosomal ceramidase | - | 160.0 |
| g9148 | 1.58 | catalase | - | 79.1 |
| Accession ID | % PSM (Average) | Cazyme Family | Mw (kDa) |
|---|---|---|---|
| g377 (BGL-3) | 7.09 | GH3 | 88.7 |
| g9150 | 1.54 | GH3 | 86.5 |
| g8384 | 0.85 | GH1 | 68.1 |
| g6857 | 0.79 | GH3 | 109 |
| g3139 | 0.30 | GH3 | 93.6 |
| g6753 | 0.09 | GH3 | 81.8 |
| SE | AcSE | AP | |
|---|---|---|---|
| Cellulose | 49.0% | 43.6% | 71.8% |
| Hemicellulose | 15.4% | 17.1% | 24.1% |
| Lignin | 35.6% | 39.3% | 4.1% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Méndez-Líter, J.A.; de Eugenio, L.I.; Hakalin, N.L.S.; Prieto, A.; Martínez, M.J. Production of a β-Glucosidase-Rich Cocktail from Talaromyces amestolkiae Using Raw Glycerol: Its Role for Lignocellulose Waste Valorization. J. Fungi 2021, 7, 363. https://doi.org/10.3390/jof7050363
Méndez-Líter JA, de Eugenio LI, Hakalin NLS, Prieto A, Martínez MJ. Production of a β-Glucosidase-Rich Cocktail from Talaromyces amestolkiae Using Raw Glycerol: Its Role for Lignocellulose Waste Valorization. Journal of Fungi. 2021; 7(5):363. https://doi.org/10.3390/jof7050363
Chicago/Turabian StyleMéndez-Líter, Juan A., Laura I. de Eugenio, Neumara L. S. Hakalin, Alicia Prieto, and María Jesús Martínez. 2021. "Production of a β-Glucosidase-Rich Cocktail from Talaromyces amestolkiae Using Raw Glycerol: Its Role for Lignocellulose Waste Valorization" Journal of Fungi 7, no. 5: 363. https://doi.org/10.3390/jof7050363
APA StyleMéndez-Líter, J. A., de Eugenio, L. I., Hakalin, N. L. S., Prieto, A., & Martínez, M. J. (2021). Production of a β-Glucosidase-Rich Cocktail from Talaromyces amestolkiae Using Raw Glycerol: Its Role for Lignocellulose Waste Valorization. Journal of Fungi, 7(5), 363. https://doi.org/10.3390/jof7050363






