β-Galactosidase-Producing Isolates in Mucoromycota: Screening, Enzyme Production, and Applications for Functional Oligosaccharide Synthesis
Abstract
1. Introduction
2. Materials and Methods
2.1. Microorganisms
2.2. Chromogenic Agar Test for β-Galactosidase Activity Screening
2.3. β-Galactosidase Production in Submerged and Solid Cultures
2.4. β-Galactosidase Activity Assay
2.5. Determination of the Protein Content
2.6. Partial Purification of β-Galactosidases
2.7. Gel Electrophoresis of Proteins
2.8. Enzymatic Synthesis Assay
2.9. HPLC-MS/MS Analysis of Carbohydrates
2.10. Growth-Promoting Activity Assay
2.11. Statistical Analysis
3. Results
3.1. Screening of β-Galactosidase Activity
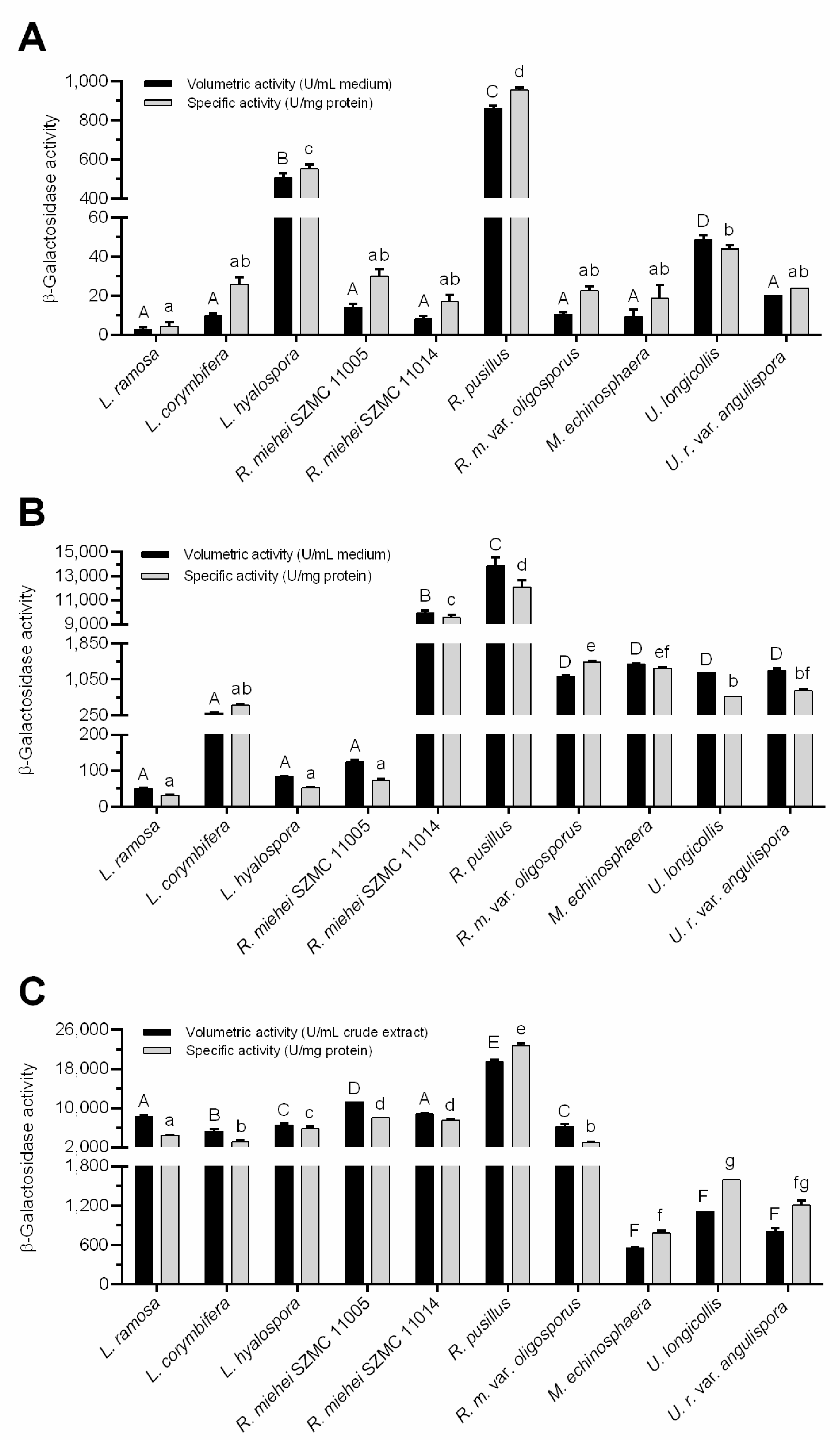
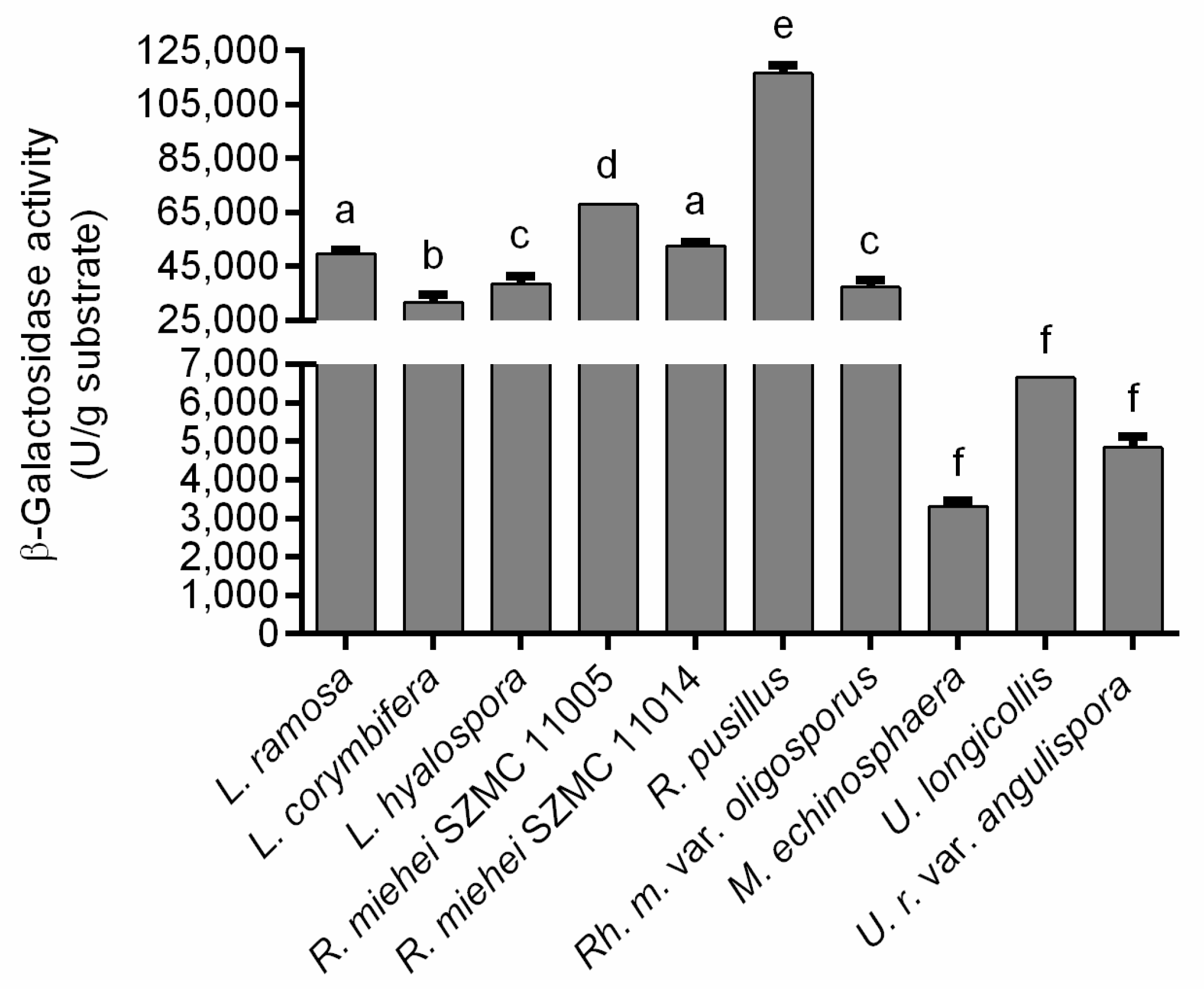
3.2. β-Galactosidase Production
3.3. Synthesis of Oligosaccharides
3.4. Growth-promoting Activity of Synthesized Oligosaccharides
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Szilagyi, A.; Ishayek, N. Lactose intolerance, dairy avoidance, and treatment options. Nutrients 2018, 10, 1994. [Google Scholar] [CrossRef]
- Saqib, S.; Akram, A.; Halim, S.A.; Tassaduq, R. Sources of β-galactosidase and its applications in food industry. 3 Biotech 2017, 7, 79. [Google Scholar] [CrossRef]
- Lu, L.; Guo, L.; Wang, K.; Liu, Y.; Xiao, M. β-Galactosidases: A great tool for synthesizing galactose-containing carbohydrates. Biotechnol. Adv. 2020, 39, 107465. [Google Scholar] [CrossRef]
- Cardelle-Cobas, A.; Corzo, N.; Olano, A.; Peláez, C.; Requena, T.; Ávila, M. Galactooligosaccharides derived from lactose and lactulose: Influence of structure on Lactobacillus, Streptococcus and Bifidobacterium growth. Int. J. Food Microbiol. 2011, 149, 81–87. [Google Scholar] [CrossRef]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, types, sources, mechanisms, and clinical applications. Foods 2019, 8, 92. [Google Scholar] [CrossRef]
- Vera, C.; Córdova, A.; Aburto, C.; Guerrero, C.; Suárez, S.; Illanes, A. Synthesis and purification of galacto-oligosaccharides: State of the art. World J. Microbiol. Biotechnol. 2016, 32, 197. [Google Scholar] [CrossRef]
- Panesar, P.S.; Kaur, R.; Singh, R.S.; Kennedy, J.F. Biocatalytic strategies in the production of galacto-oligosaccharides and its global status. Int. J. Biol. Macromol. 2018, 111, 667–679. [Google Scholar] [CrossRef]
- Torres, D.P.; Gonçalves, M.D.P.F.; Teixeira, J.A.; Rodrigues, L.R. Galacto-oligosaccharides: Production, properties, applications, and significance as prebiotics. Compr. Rev. Food Sci. Food Saf. 2010, 9, 438–454. [Google Scholar] [CrossRef]
- Anisha, G.S. β-Galactosidases. In Current Developments in Biotechnology and Bioengineering: Production, Isolation and Purification of Industrial Products, 1st ed.; Pandey, A., Negi, S., Soccol, C.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 395–421. [Google Scholar]
- Tokošová, S.; Hronská, H.; Rosenberg, M. Production of galacto-oligosaccharides by commercial preparates of fungal β-galactosidase. Acta Chim. Slovaca 2015, 8, 101–106. [Google Scholar] [CrossRef]
- Bassetto, R.Z.; Cabral, P.S.; Silveira, M.H.L.; Almeida, M.M.; Chiquetto, N.C. Optimization of β-galactosidase production from Penicillium sp. for synthesis of galactooligosaccharides. Int. Food Res. J. 2017, 24, 1793–1798. [Google Scholar]
- Silvério, S.C.; Macedo, E.A.; Teixeira, J.A.; Rodrigues, L.R. New β-galactosidase producers with potential for prebiotic synthesis. Bioresour. Technol. 2018, 250, 131–139. [Google Scholar] [CrossRef]
- Papp, T.; Nyilasi, I.; Csernetics, Á.; Nagy, G.; Takó, M.; Vágvölgyi, C. Improvement of industrially relevant biological activities in Mucoromycotina fungi. In Gene Expression Systems in Fungi: Advancements and Applications. Fungal Biology; Schmoll, M., Dattenböck, C., Eds.; Springer: Cham, Switzerland, 2016; pp. 97–118. [Google Scholar]
- Shaikh, S.A.; Khire, J.M.; Khan, M.I. Production of β-galactosidase from thermophilic fungus Rhizomucor sp. J. Ind. Microbiol. Biotechnol. 1997, 19, 239–245. [Google Scholar] [CrossRef]
- Shaikh, S.A.; Khire, J.M.; Khan, M.I. Characterization of a thermostable extracellular β-galactosidase from a thermophilic fungus Rhizomucor sp. Biochim. Biophys. Acta Gen. Subj. 1999, 1472, 314–322. [Google Scholar] [CrossRef]
- Panesar, P.S.; Kaur, R.; Singh, R.S. Isolation and screening of fungal strains for β-galactosidase production. WASET Int. J. Nutr. Food Eng. 2016, 10, 400–404. [Google Scholar]
- Kaur, R.; Panesar, P.S.; Singh, R.S. Utilization of agro-industrial residues for the production of β-galactosidase using fungal isolate under solid state fermentation conditions. Acta Aliment. 2018, 47, 162–170. [Google Scholar] [CrossRef]
- Raol, G.G.; Raol, B.V.; Prajapati, V.S.; Bhavsar, N.H. Utilization of agro-industrial waste for β-galactosidase production under solid state fermentation using halotolerant Aspergillus tubingensis GR1 isolate. 3 Biotech 2015, 5, 411–421. [Google Scholar] [CrossRef]
- Kazemi, S.; Khayati, G.; Faezi-Ghasemi, M. β-Galactosidase production by Aspergillus niger ATCC 9142 using inexpensive substrates in solid-state fermentation: Optimization by orthogonal arrays design. Iran. Biomed. J. 2016, 20, 287–294. [Google Scholar]
- Panesar, P.S.; Kaur, R.; Singla, G.; Sangwan, R.S. Bio-processing of agro-industrial wastes for production of food-grade enzymes: Progress and prospects. Appl. Food Biotechnol. 2016, 3, 208–227. [Google Scholar]
- Sharma, S.; Singh, P. Isolation and characterization of β-galactosidase enzyme producing microbe and optimization of its enzyme activity under different culture condition. Int. J. Curr. Microbiol. App. Sci. 2014, 3, 148–155. [Google Scholar]
- Vidya, B.; Palaniswamy, M.; Gopalakrishnan, V.K. Screening and optimization of β-galactosidase from fungal strains by using agro residues. World J. Pharm. Sci. 2014, 3, 1809–1821. [Google Scholar]
- Torres, J.M.O.; dela Cruz, T.E.E. Production of xylanases by mangrove fungi from the Philippines and their application in enzymatic pretreatment of recycled paper pulps. World J. Microbiol. Biotechnol. 2013, 29, 645–655. [Google Scholar] [CrossRef]
- Soren, J.P.; Halder, S.K.; Mondal, J.; Hor, P.K.; Mohapatra, P.K.D.; Mondal, K.C. A permissive approach for optimization of L-glutaminase production using wheat bran as supporting substrate and assessment of its cytotoxic potentialities. Acta Biol. Szeged. 2020, 64, 1–10. [Google Scholar] [CrossRef]
- Kotogán, A.; Zambrano, C.; Kecskeméti, A.; Varga, M.; Szekeres, A.; Papp, T.; Vágvölgyi, C.; Takó, M. An organic solvent-tolerant lipase with both hydrolytic and synthetic activities from the oleaginous fungus Mortierella echinosphaera. Int. J. Mol. Sci. 2018, 19, 1129. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Wang, P. Enzymatic synthesis of galactosyl lactic ethyl ester and its polymer for use as biomaterials. J. Biotechnol. 2007, 132, 314–317. [Google Scholar] [CrossRef]
- Ismail, S.A.; Mabrouk, S.S.; Mahoney, R.R. Purification and characterization of β-galactosidase from Mucor pusillus. J. Food Biochem. 1997, 21, 145–162. [Google Scholar] [CrossRef]
- Vishwanataha, T.; Sampath, A.; Jain, S.N.; Divyashree, B.C.; Reena, V.; Sowmya, G.; MohanKumar, B.S.; Venugopal, N.; Patil, S.J.; Siddalingeshwara, K.G. A novel approach for screening and synthesis of β-galactosidase from microbial origin. Int. J. Appl. Biotechnol. Biochem. 2012, 2, 285–290. [Google Scholar]
- Martarello, R.D.; Cunha, L.; Cardoso, S.L.; de Freitas, M.M.; Silveira, D.; Fonseca-Bazzo, Y.M.; Homem-de-Mello, M.; Filho, E.X.F.; Magalhães, P.O. Optimization and partial purification of beta-galactosidase production by Aspergillus niger isolated from Brazilian soils using soybean residue. AMB Express 2019, 9, 81. [Google Scholar] [CrossRef]
- Abd El-Salam, B.A.; Ibrahim, O.A.; Amer, A.E. Efficient enzymatic conversion of lactose in milk using fungal β-galactosidase. Biocatal. Agric. Biotechnol. 2020, 29, 101813. [Google Scholar] [CrossRef]
- De Jesus, L.F.M.C.; Guimarães, L.H.S. Production of β-galactosidase by Trichoderma sp. through solid-state fermentation targeting the recovery of galactooligosaccharides from whey cheese. J. Appl. Microbiol. 2021, 130, 865–877. [Google Scholar] [CrossRef] [PubMed]
- Vidya, B.; Palaniswamy, M.; Angayarkanni, J.; Nawaz, K.A.; Thandeeswaran, M.; Chaithanya, K.K.; Tekluu, B.; Muthusamy, K.; Gopalakrishnan, V.K. Purification and characterization of β-galactosidase from newly isolated Aspergillus terreus (KUBCF1306) and evaluating its efficacy on breast cancer cell line (MCF-7). Bioorg. Chem. 2020, 94, 103442. [Google Scholar] [CrossRef]
- Zolnere, K.; Ciprovica, I. The comparison of commercially available β-galactosidases for dairy industry: Review. In Proceedings of the 23rd Annual International Scientific Conference “Research for Rural Development 2017”, Jelgava, Latvia, 17–19 May 2017; Latvia University of Agriculture: Jelgava, Latvia, 2017; Volume 1, pp. 215–222. [Google Scholar]
- Kaur, R.; Panesar, P.S.; Singh, R.S. Utilization of whey for the production of β-galactosidase using yeast and fungal culture. WASET Int. J. Nutr. Food Eng. 2015, 9, 739–743. [Google Scholar]
- Oda, K.; Kakizono, D.; Yamada, O.; Iefuji, H.; Akita, O.; Iwashita, K. Proteomic analysis of extracellular proteins from Aspergillus oryzae grown under submerged and solid-state culture conditions. Appl. Environ. Microbiol. 2006, 72, 3448–3457. [Google Scholar] [CrossRef] [PubMed]
- Mateos Diaz, J.C.; Rodríguez, J.A.; Roussos, S.; Cordova, J.; Abousalham, A.; Carriere, F.; Baratti, J. Lipase from the thermotolerant fungus Rhizopus homothallicus is more thermostable when produced using solid state fermentation than liquid fermentation procedures. Enzym. Microb. Technol. 2006, 39, 1042–1050. [Google Scholar] [CrossRef]
- Manan, M.A.; Webb, C. Estimation of growth in solid state fermentation: A review. Malays. J. Microbiol. 2018, 14, 61–69. [Google Scholar]
- Javed, M.M.; Zahoor, S.; Shafaat, S.; Mehmooda, I.; Gul, A.; Rasheed, H.; Bukhari, A.I.; Aftab, M.N. Wheat bran as a brown gold: Nutritious value and its biotechnological applications. Afr. J. Microbiol. Res. 2012, 6, 724–733. [Google Scholar]
- Assamoi, A.A.; Bedikou, M.E.; Soro-Yao, A.A.; Niamke, L.S.; Destain, J.; Thonart, P. β-Galactosidase production by solid state fermentation of wheat bran/whole wheat without any supplement. World J. Pharm. Pharma. Sci. 2015, 4, 196–207. [Google Scholar]
- Adalberto, P.R.; Massabni, A.C.; Goulart, A.J.; Contiero, J.; Carmona, E.C.; Cardello, L.; Monti, R. Production of β-galactosidase by Trichoderma reesei FTKO-39 in wheat bran. Appl. Biochem. Biotechnol. 2006, 133, 163–170. [Google Scholar] [CrossRef]
- Saburi, W.; Ueno, H.M.; Matsui, H.; Mori, H. Acidophilic β-galactosidase from Aspergillus niger AHU7120 with lactose hydrolytic activity under simulated gastric conditions. J. Appl. Glycosci. 2014, 61, 53–57. [Google Scholar] [CrossRef]
- Patel, J.D.; Raol, B.V.; Bhavsar, N.H.; Raol, G.G. One-factor-at-a-time (OFAT) optimization of β-galactosidase production from Aspergillus flavus gr. NFCCI-2728 in submerged fermentation. J. Harmoniz. Res. Appl. Sci. 2018, 6, 227–235. [Google Scholar] [CrossRef]
- Seyis, I.; Aksoz, N. Production of lactase by Trichoderma sp. Food Technol. Biotechnol. 2004, 42, 121–124. [Google Scholar]
- Robledo, A.; Aguilar, C.N.; Belmares-Cerda, R.E.; Flores-Gallegos, A.C.; Contreras-Esquivel, J.C.; Montañez, J.C.; Mussatto, S.I. Production of thermostable xylanase by thermophilic fungal strains isolated from maize silage. CyTA J. Food 2016, 14, 302–308. [Google Scholar] [CrossRef]
- Garcia, N.F.L.; da Silva Santos, F.R.; Bocchini, D.A.; da Paz, M.F.; Fonseca, G.G.; Leite, R.S.R. Catalytic properties of cellulases and hemicellulases produced by Lichtheimia ramosa: Potential for sugarcane bagasse saccharification. Ind. Crops Prod. 2018, 122, 49–56. [Google Scholar] [CrossRef]
- De Andrade Silva, C.A.; da Silva, P.G.P.; da Silva, G.F.A.; Dantas, D.P.; Leite, R.S.R.; Fonseca, G.G. Biotransformation of fruit residues via solid state bioprocess using Lichtheimia ramosa. SN Appl. Sci. 2020, 2, 861. [Google Scholar] [CrossRef]
- Rodriguez-Colinas, B.; Poveda, A.; Jimenez-Barbero, J.; Ballesteros, A.O.; Plou, F.J. Galacto-oligosaccharide synthesis from lactose solution or skim milk using the β-galactosidase from Bacillus circulans. J. Agric. Food Chem. 2012, 60, 6391–6398. [Google Scholar] [CrossRef]
- Fischer, C.; Kleinschmidt, T. Synthesis of galactooligosaccharides in milk and whey: A review. Compr. Rev. Food Sci. Food Saf. 2018, 17, 678–697. [Google Scholar] [CrossRef]
- Hua, X.; Yang, R.; Zhang, W.; Fei, Y.; Jin, Z.; Jiang, B. Dual-enzymatic synthesis of lactulose in organic-aqueous two-phase media. Food Res. Int. 2010, 43, 716–722. [Google Scholar] [CrossRef]
- Song, Y.S.; Suh, Y.J.; Park, C.; Kim, S.W. Improvement of lactulose synthesis through optimization of reaction conditions with immobilized β-galactosidase. Korean J. Chem. Eng. 2013, 30, 160–165. [Google Scholar] [CrossRef]
- Guerrero, C.; Vera, C.; Serna, N.; Illanes, A. Immobilization of Aspergillus oryzae β-galactosidase in an agarose matrix functionalized by four different methods and application to the synthesis of lactulose. Bioresour. Technol. 2017, 232, 53–63. [Google Scholar] [CrossRef]
- Aburto, C.; Guerrero, C.; Vera, C.; Illanes, A. Improvement in the yield and selectivity of lactulose synthesis with Bacillus circulans β-galactosidase. LWT Food Sci. Technol. 2020, 118, 108746. [Google Scholar] [CrossRef]
- Tochio, T.; Kadota, Y.; Tanaka, T.; Koga, Y. 1-Kestose, the smallest fructooligosaccharide component, which efficiently stimulates Faecalibacterium prausnitzii as well as Bifidobacteria in humans. Foods 2018, 7, 140. [Google Scholar] [CrossRef]
- Lorenzoni, A.S.G.; Aydos, L.F.; Klein, M.P.; Rodrigues, R.C.; Hertz, P.F. Fructooligosaccharides synthesis by highly stable immobilized β-fructofuranosidase from Aspergillus aculeatus. Carbohydr. Polym. 2014, 103, 193–197. [Google Scholar] [CrossRef]
- Veana, F.; Flores-Gallegos, A.C.; Gonzalez-Montemayor, A.M.; Michel-Michel, M.; Lopez-Lopez, L.; Aguilar-Zarate, P.; Ascacio-Valdés, J.A.; Rodríguez-Herrera, R. Invertase: An enzyme with importance in confectionery food industry. In Enzymes in Food Technology; Kuddus, M., Ed.; Springer: Singapore, 2018; pp. 187–212. [Google Scholar]
- Cunha, J.S.; Ottoni, C.A.; Morales, S.A.V.; Silva, E.S.; Maiorano, A.E.; Perna, R.F. Synthesis and characterization of fructosyltransferase from Aspergillus oryzae IPT-301 for high fructooligosaccharides production. Braz. J. Chem. Eng. 2019, 36, 657–668. [Google Scholar] [CrossRef]
- Chaisuwan, W.; Manassa, A.; Phimolsiripol, Y.; Jantanasakulwong, K.; Chaiyaso, T.; Pathom-aree, W.; You, S.; Seesuriyachan, P. Integrated ultrasonication and microbubble-assisted enzymatic synthesis of fructooligosaccharides from brown sugar. Foods 2020, 9, 1833. [Google Scholar] [CrossRef]
- Lateef, A.; Oloke, J.K.; Kana, E.B.G.; Oyeniyi, S.O.; Onifade, O.R.; Oyeleye, A.O.; Oladosu, O.C. Rhizopus stolonifer LAU 07: A novel source of fructosyltransferase. Chem. Pap. 2008, 62, 635–638. [Google Scholar] [CrossRef]
- Orikasa, Y.; Oda, Y. Molecular characterization of β-fructofuranosidases from Rhizopus delemar and Amylomyces rouxii. Folia Microbiol. 2013, 58, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Orikasa, Y.; Oda, Y.; Ohwada, T. Identification of sucA, encoding β-fructofuranosidase, in Rhizopus microsporus. Microorganisms 2018, 6, 26. [Google Scholar] [CrossRef]
- Ojwach, J.; Kumar, A.; Mutanda, T.; Mukaratirwa, S. Fructosyltransferase and inulinase production by indigenous coprophilous fungi for the biocatalytic conversion of sucrose and inulin into oligosaccharides. Biocatal. Agric. Biotechnol. 2020, 30, 101867. [Google Scholar] [CrossRef]
- Chen, C.; Deng, J.; Lv, X.; Li, J.; Du, G.; Li, H.; Liu, L. Biocatalytic synthesis of lactosucrose using a recombinant thermostable β-fructofuranosidase from Arthrobacter sp. 10138. Bioengineered 2020, 11, 416–427. [Google Scholar] [CrossRef] [PubMed]
- Villamiel, M.; Montilla, A.; Olano, A.; Corzo, N. Production and bioactivity of oligosaccharides derived from lactose. In Food Oligosaccharides: Production, Analysis and Bioactivity; Moreno, F.J., Sanz, M.L., Eds.; Wiley-Blackwell: Chichester, UK, 2014; pp. 137–167. [Google Scholar]
- Duarte, L.S.; da Natividade Schöffer, J.; Lorenzoni, A.S.G.; Rodrigues, R.C.; Rodrigues, E.; Hertz, P.F. A new bioprocess for the production of prebiotic lactosucrose by an immobilized β-galactosidase. Process Biochem. 2017, 55, 96–103. [Google Scholar] [CrossRef]
- Liao, X.; Huang, J.; Zhou, Q.; Guo, L.; Lin, J.; You, L.; Liu, S.; Yang, J. Designing of a novel β-galactosidase for production of functional oligosaccharides. Eur. Food Res. Technol. 2017, 243, 979–986. [Google Scholar] [CrossRef]
- Cardoso, B.B.; Silvério, S.C.; Abrunhosa, L.; Teixeira, J.A.; Rodrigues, L.R. β-Galactosidase from Aspergillus lacticoffeatus: A promising biocatalyst for the synthesis of novel prebiotics. Int. J. Food Microbiol. 2017, 257, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Suyama, K.; Adachi, S.; Toba, T.; Sohma, T.; Hwang, C.J.; Itoh, T. Isoraffinose (6G-β-galactosylsucrose) synthesized by the intermolecular transgalactosylation reaction of Escherichia coli β-galactosidase. Agric. Biol. Chem. 1986, 50, 2069–2075. [Google Scholar] [CrossRef]
- Verkhnyatskaya, S.; Ferrari, M.; de Vos, P.; Walvoort, M.T.C. Shaping the infant microbiome with non-digestible carbohydrates. Front. Microbiol. 2019, 10, 343. [Google Scholar] [CrossRef] [PubMed]
- Sheikhi, A.; Shakerian, M.; Giti, H.; Baghaeifar, M.; Jafarzadeh, A.; Ghaed, V.; Heibor, M.R.; Baharifar, N.; Dadafarin, Z.; Bashirpour, G. Probiotic yogurt culture Bifidobacterium animalis subsp. lactis BB-12 and Lactobacillus acidophilus LA-5 modulate the cytokine secretion by peripheral blood mononuclear cells from patients with ulcerative colitis. Drug Res. 2016, 66, 300–305. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Szajewska, H.; Konarska, Z.; Kołodziej, M. Probiotic bacterial and fungal strains: Claims with evidence. Dig. Dis. 2016, 34, 251–259. [Google Scholar] [CrossRef]
- Hill, D.; Sugrue, I.; Tobin, C.; Hill, C.; Stanton, C.; Ross, R.P. The Lactobacillus casei group: History and health related applications. Front. Microbiol. 2018, 9, 2107. [Google Scholar] [CrossRef]
- Silva, M.P.; Rossoni, R.D.; Junqueira, J.C.; Jorge, A.O.C. Probiotics for prevention and treatment of candidiasis and other infectious diseases: Lactobacillus spp. and other potential bacterial species. In Probiotics and Prebiotics in Human Nutrition and Health; Rao, V., Rao, L.G., Eds.; IntechOpen: London, UK, 2016; pp. 241–262. [Google Scholar]
- Invernici, M.M.; Furlaneto, F.A.C.; Salvador, S.L.; Ouwehand, A.C.; Salminen, S.; Mantziari, A.; Vinderola, G.; Ervolino, E.; Santana, S.I.; Silva, P.H.F.; et al. Bifidobacterium animalis subsp. lactis HN019 presents antimicrobial potential against periodontopathogens and modulates the immunological response of oral mucosa in periodontitis patients. PLoS ONE 2020, 15, e0238425. [Google Scholar] [CrossRef]
- Pais, P.; Almeida, V.; Yılmaz, M.; Teixeira, M.C. Saccharomyces boulardii: What makes it tick as successful probiotic? J. Fungi 2020, 6, 78. [Google Scholar] [CrossRef]
- Oh, N.S.; Kim, K.; Oh, S.; Kim, Y. Enhanced production of galactooligosaccharides enriched skim milk and applied to potentially synbiotic fermented milk with Lactobacillus rhamnosus 4B15. Food Sci. Anim. Resour. 2019, 39, 725–741. [Google Scholar] [CrossRef]
- Fara, A.; Sabater, C.; Palacios, J.; Requena, T.; Montilla, A.; Zárate, G. Prebiotic galactooligosaccharides production from lactose and lactulose by Lactobacillus delbrueckii subsp. bulgaricus CRL450. Food Funct. 2020, 11, 5875–5886. [Google Scholar] [CrossRef]
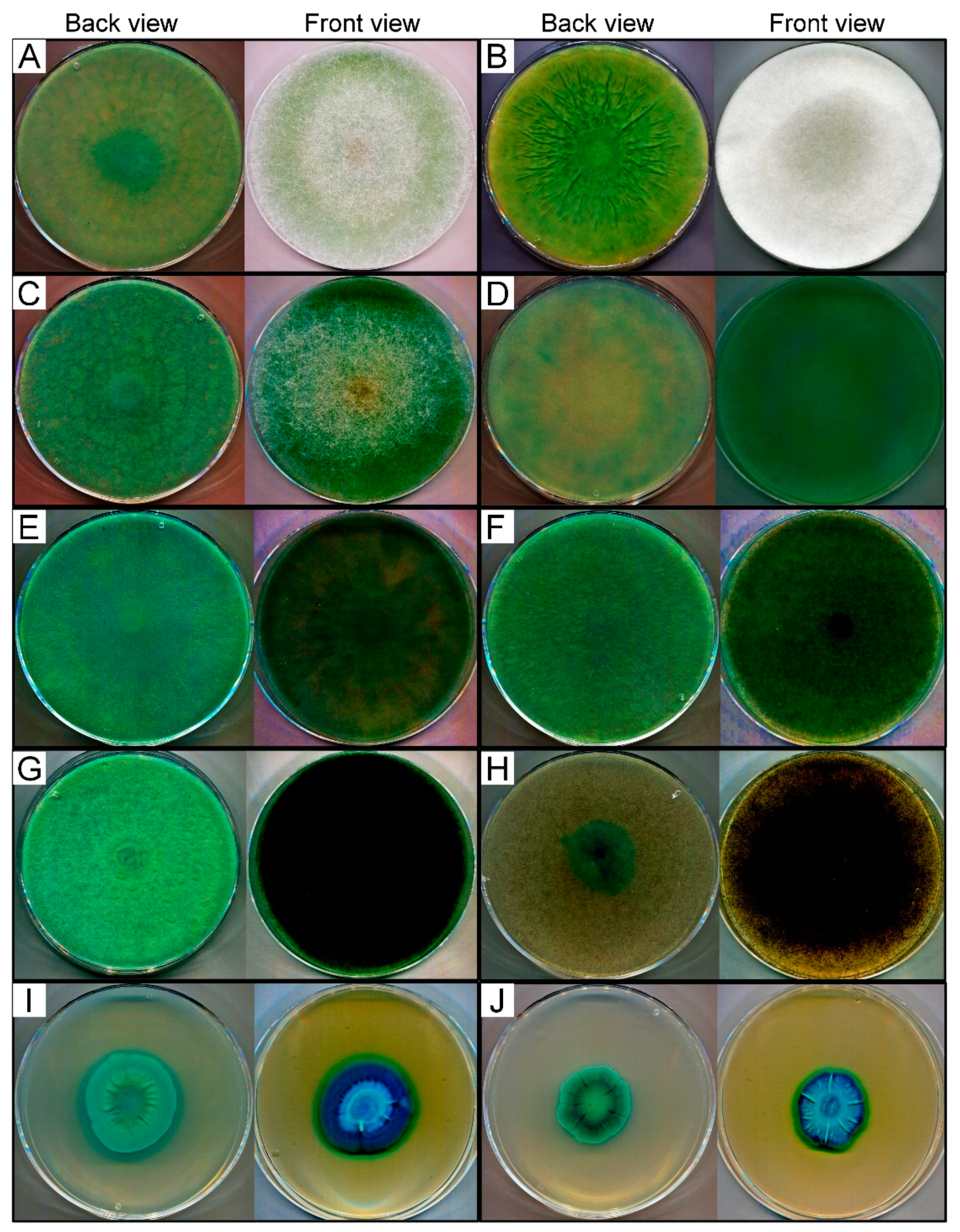


| Fungal Strains | β-Galactosidase Activity 1 | Cultivation Temperature (°C) | Source | |||||
|---|---|---|---|---|---|---|---|---|
| t(incubation)/day | ||||||||
| 1 | 2 | 4 | 6 | 8 | 10 | |||
| Lichtheimia ramosa SZMC 2 11360 | +++ | +++++ | +++++ | +++++ | +++++ | +++++ | 37 | soil/unknown |
| Lichtheimia corymbifera SZMC 11361 | ++ | ++++ | ++++ | +++++ | +++++ | +++++ | 37 | soil/Afghanistan |
| Lichtheimia hyalospora SZMC 11364 | ++ | ++++ | ++++ | +++++ | +++++ | +++++ | 37 | Manihot esculenta stem / Ghana |
| Rhizomucor miehei SZMC 11005 | ++ | +++ | ++++ | ++++ | ++++ | +++++ | 37 | peppermint compost/India |
| Rhizomucor miehei SZMC 11014 | ++ | ++++ | +++++ | +++++ | +++++ | +++++ | 37 | compost/Switzerland |
| Rhizomucor pusillus SZMC 11025 | + | +++ | +++ | ++++ | ++++ | +++++ | 37 | dead fallen leaves/California, USA |
| Rhizopus microsporus var. oligosporus SZMC 13619 | NC | + | +++ | ++++ | ++++ | +++++ | 37 | tempeh/Indonesia |
| Mortierella echinosphaera SZMC 11251 | + | + | +++ | ++++ | ++++ | +++++ | 20 | begonia/Netherlands |
| Umbelopsis longicollis SZMC 11208 | + | +++ | ++++ | +++++ | +++++ | +++++ | 25 | soil/Australia |
| Umbelopsis ramanniana var. angulispora SZMC 11234 | + | +++ | +++++ | +++++ | +++++ | +++++ | 25 | unknown/Russia |
| Initial Materials | Crude β-Galactosidase Used | Oligosaccharide Concentration (mg/L) 1 | Oligosaccharide Yield (%) 2 | |
|---|---|---|---|---|
| Trisaccharide | Tetrasaccharide | |||
| Lactose | L. ramosa | 129.5 ± 10.7 | 34.5 ± 1.1 | 0.11 |
| R. pusillus | 197.2 ± 2.5 | 60.4 ± 3.8 | 0.17 | |
| Skim milk | L. ramosa | 14.4 ± 0.5 | 1.4 ± 0.03 | 0.032 |
| R. pusillus | 20.1 ± 0.02 | 2.62 ± 0.02 | 0.047 | |
| Lactose–fructose | L. ramosa | 88.6 ± 5.9 | 18.7 ± 0.4 | 0.11 |
| R. pusillus | 187.6 ± 5.4 | 52.2 ± 3.1 | 0.24 | |
| oNPG–sucrose | L. ramosa | 856.5 ± 16.5 | 48.1 ± 2.1 | 0.91 |
| R. pusillus | 2,040.1 ± 36.8 | 196.2 ± 3.4 | 2.24 | |
| Initial Materials | L. casei | L. acidophilus | B. animalis subsp. lactis | S. boulardii |
|---|---|---|---|---|
| Lactose | ||||
| Trisaccharide | 0.991 | 0.985 | 0.979 | 0.999 |
| Tetrasaccharide | 0.998 | 0.999 | 0.999 | 0.988 |
| Skim milk | ||||
| Trisaccharide | 0.791 | 0.995 | 0.994 | 0.961 |
| Tetrasaccharide | 0.635 | 0.992 | 0.993 | 0.875 |
| Lactose–fructose | ||||
| Trisaccharide | 0.785 | 0.986 | 0.999 | 0.926 |
| Tetrasaccharide | 0.698 | 0.957 | 0.989 | 0.869 |
| Mucoromycota Fungi | Fermentation Condition | Substrate | Enzyme Activity | Reference |
|---|---|---|---|---|
| Mucor sp. | SmF 1 | lactose | 228 U/L | [12] |
| Rhizomucor sp. | SmF | lactose | 0.55 U/mL (0.21 U/mg) | [14] |
| SSF 2 | wheat bran | 5.5 U/mL (2.04 U/mg) | ||
| Rhizomucor pusillus | SmF | lactose | 2.14 IU/mL | [16] |
| Rhizomucor pusillus | SSF | wheat bran | 101.89 U/gds | [17] |
| Rhizopus sp. | SmF | lactose | less than 10 U/mL | [43] |
| Rhizopus stolonifer | SmF | lactose | 2.250 IU | [28] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Volford, B.; Varga, M.; Szekeres, A.; Kotogán, A.; Nagy, G.; Vágvölgyi, C.; Papp, T.; Takó, M. β-Galactosidase-Producing Isolates in Mucoromycota: Screening, Enzyme Production, and Applications for Functional Oligosaccharide Synthesis. J. Fungi 2021, 7, 229. https://doi.org/10.3390/jof7030229
Volford B, Varga M, Szekeres A, Kotogán A, Nagy G, Vágvölgyi C, Papp T, Takó M. β-Galactosidase-Producing Isolates in Mucoromycota: Screening, Enzyme Production, and Applications for Functional Oligosaccharide Synthesis. Journal of Fungi. 2021; 7(3):229. https://doi.org/10.3390/jof7030229
Chicago/Turabian StyleVolford, Bettina, Mónika Varga, András Szekeres, Alexandra Kotogán, Gábor Nagy, Csaba Vágvölgyi, Tamás Papp, and Miklós Takó. 2021. "β-Galactosidase-Producing Isolates in Mucoromycota: Screening, Enzyme Production, and Applications for Functional Oligosaccharide Synthesis" Journal of Fungi 7, no. 3: 229. https://doi.org/10.3390/jof7030229
APA StyleVolford, B., Varga, M., Szekeres, A., Kotogán, A., Nagy, G., Vágvölgyi, C., Papp, T., & Takó, M. (2021). β-Galactosidase-Producing Isolates in Mucoromycota: Screening, Enzyme Production, and Applications for Functional Oligosaccharide Synthesis. Journal of Fungi, 7(3), 229. https://doi.org/10.3390/jof7030229









