The Conserved MAP Kinase MpkB Regulates Development and Sporulation without Affecting Aflatoxin Biosynthesis in Aspergillus flavus
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Growth Conditions
2.2. Construction of Deletion and Overexpression Mutants
2.3. Conidial and Sclerotial Production Analyses
2.4. Microscopy
2.5. Nucleic Acid Extraction and Northern Blot Analysis
2.6. Aflatoxin Extraction and Analysis via Thin-Layer Chromatography (TLC)
3. Results
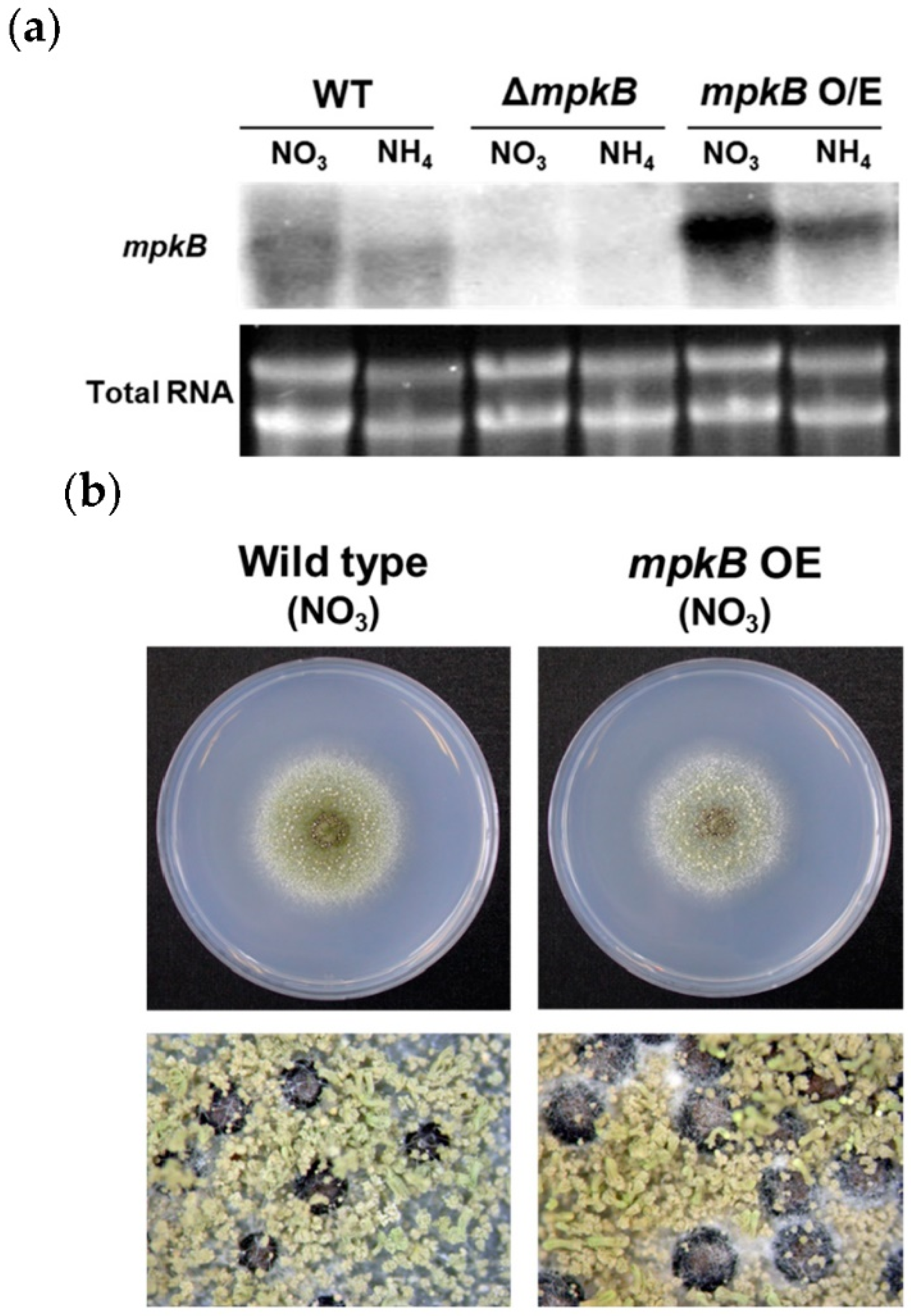
3.1. Generation of A. flavus mpkB Deletion Strains
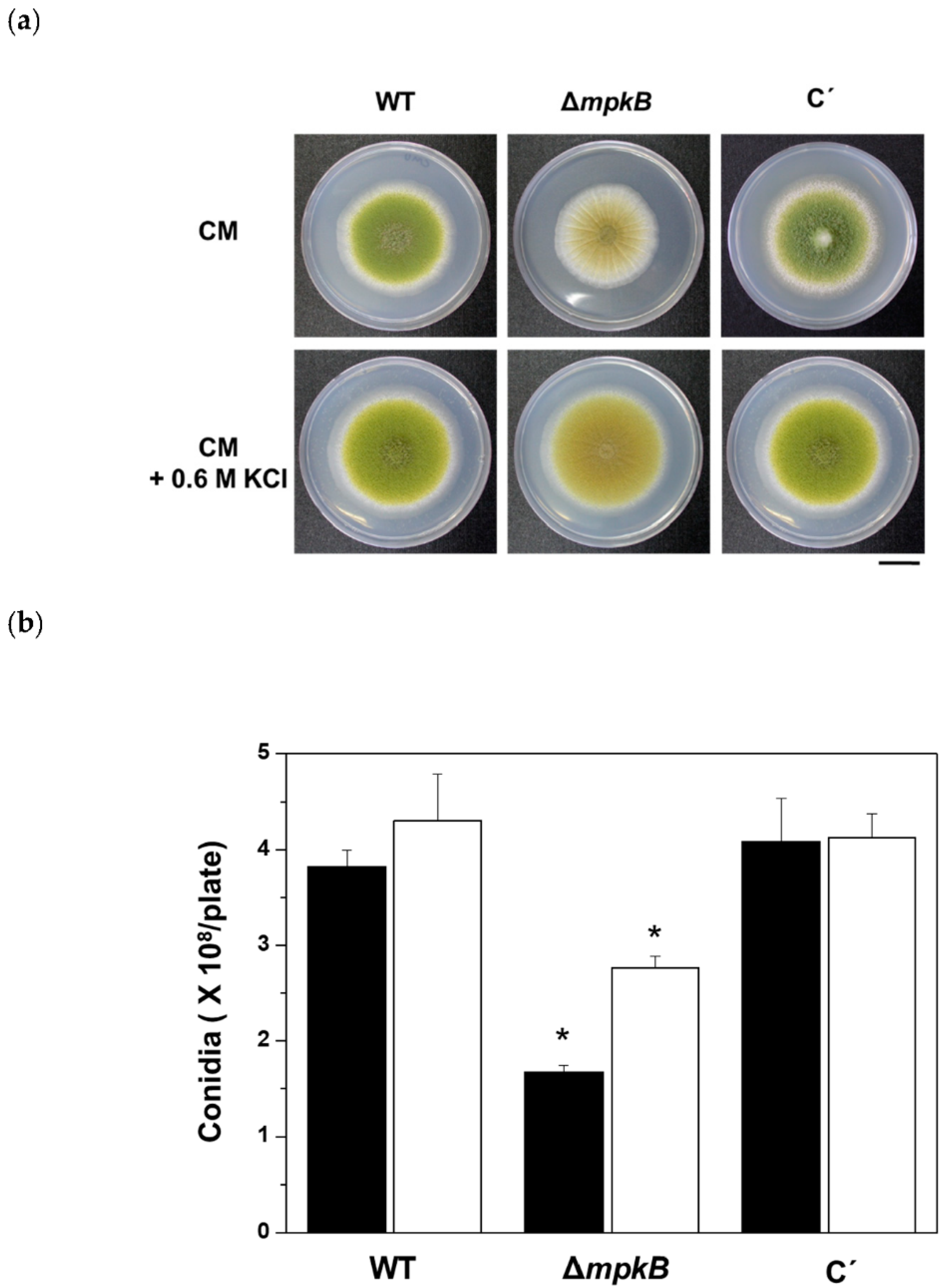
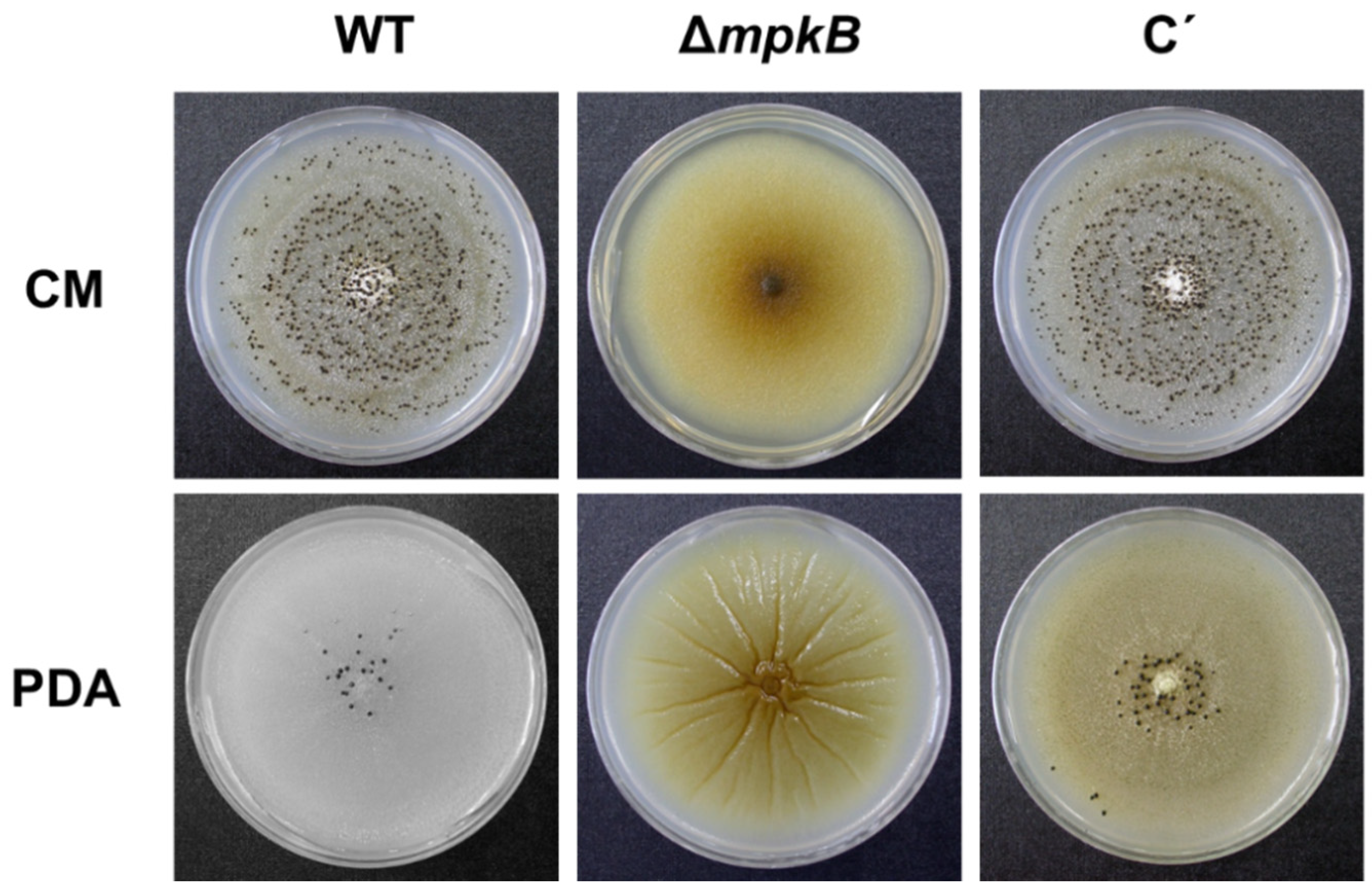
3.2. Deletion of mpkB Affects Conidiation and Sclerotia Production But Not Growth
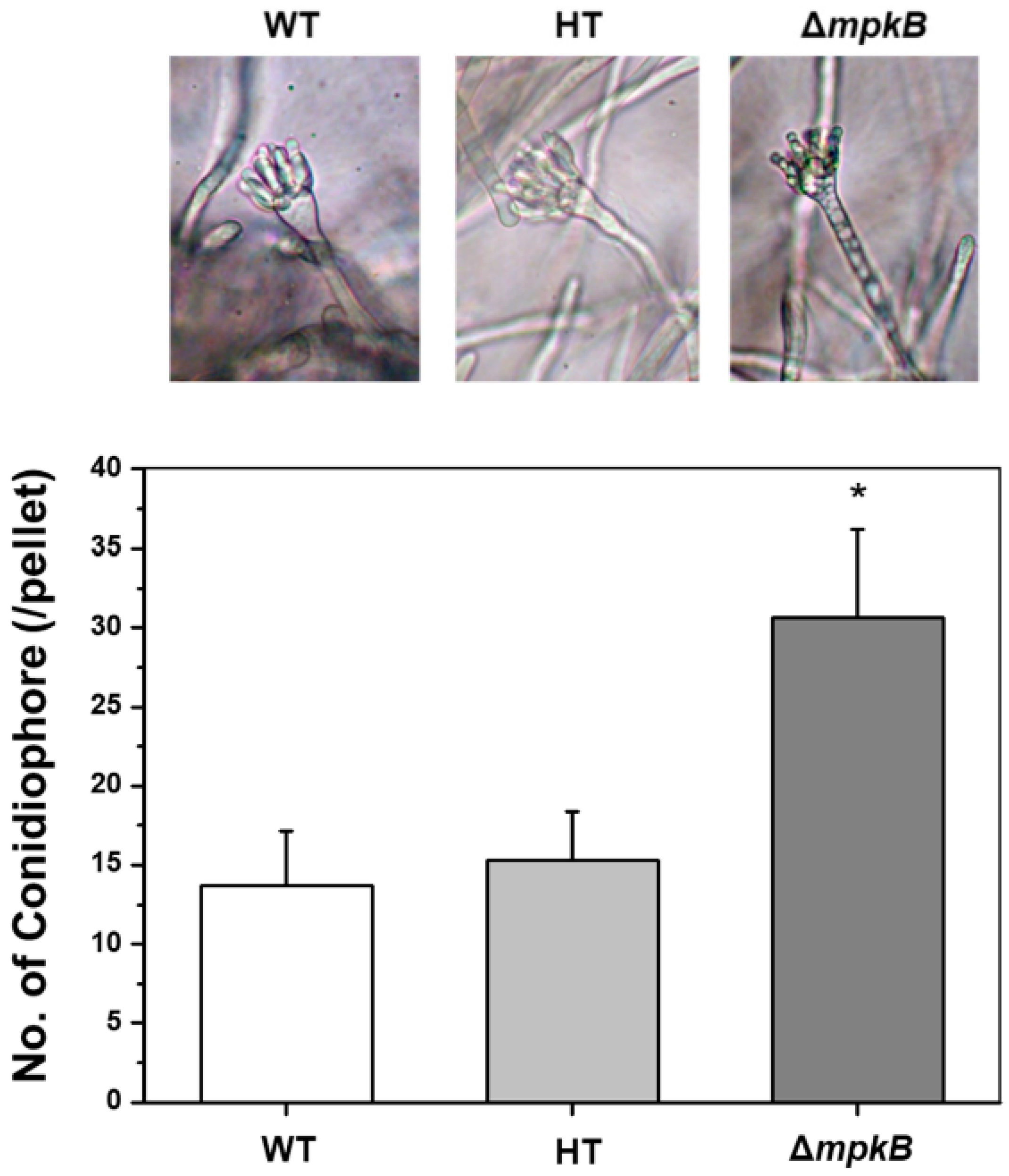
3.3. Deletion of mpkB Caused Markedly Aberrant Conidiophore Morphology
3.4. Overexpression of mpkB Does Not Affect Growth and Development
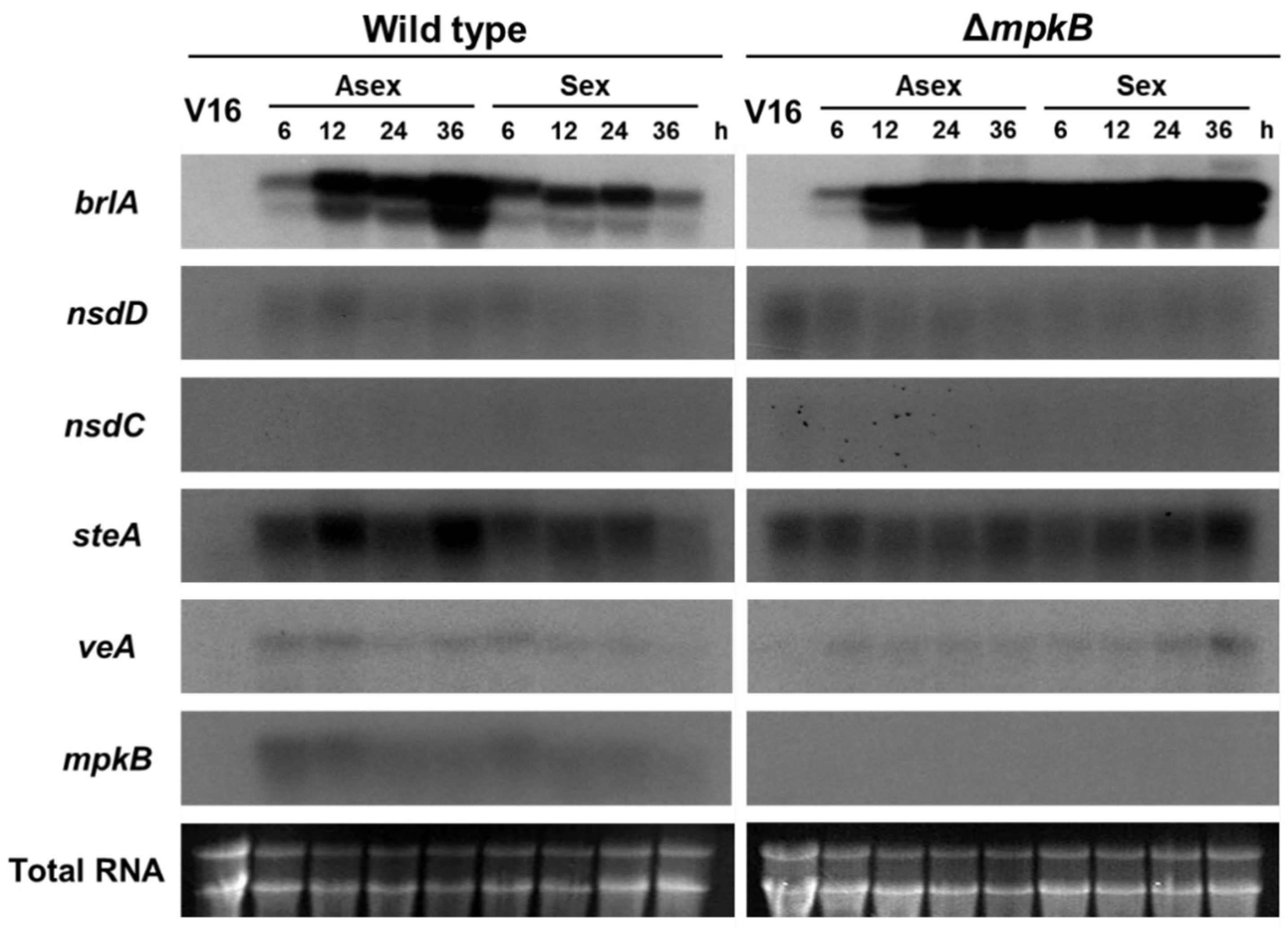
3.5. MpkB Regulates brlA Expression during the Developmental Stage
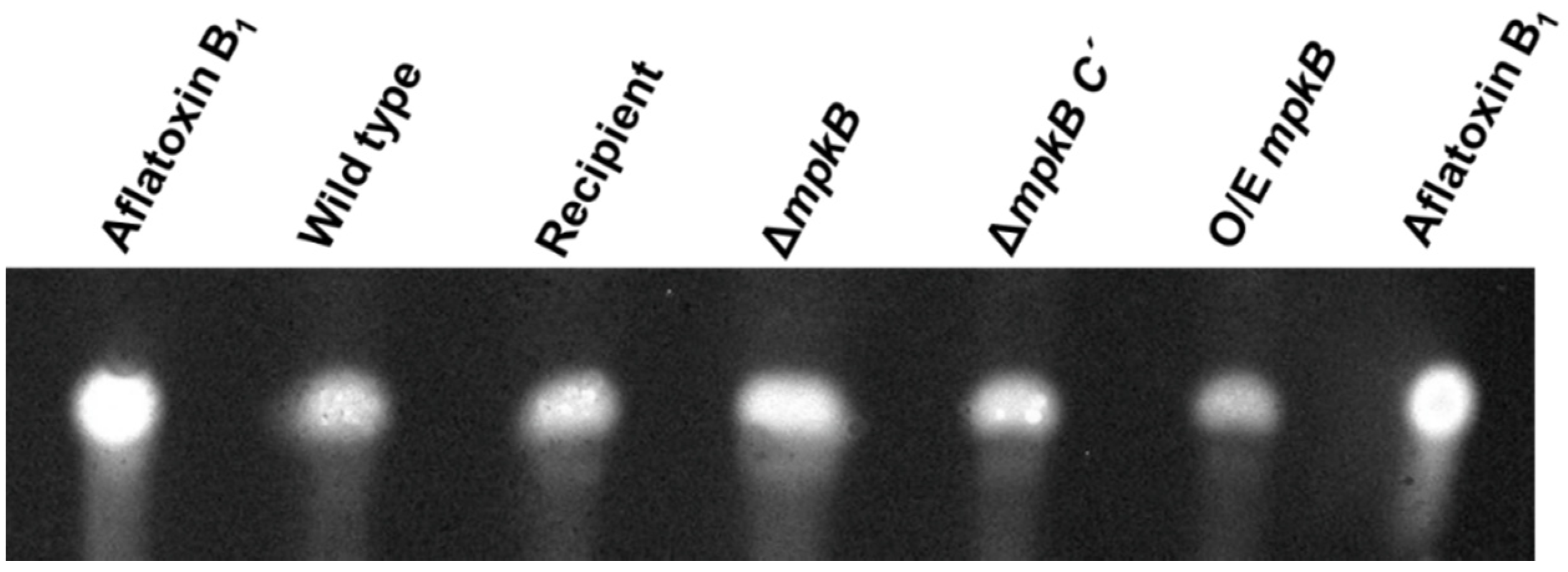
3.6. MpkB Is Not Required for Aflatoxin Production in A. flavus
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Houbraken, J.; de Vries, R.P.; Samson, R.A. Modern taxonomy of biotechnologically important Aspergillus and Penicillium species. Adv. Appl. Microbiol. 2014, 86, 199–249. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-S.; Jun, S.-C.; Han, K.-H.; Hong, S.-B.; Yu, J.-H. Diversity, application, and synthetic biology of industrially important Aspergillus fungi. Adv. Appl. Microbiol. 2017, 100, 161–202. [Google Scholar] [CrossRef] [PubMed]
- Bayman, P.; Cotty, P.J. Genetic diversity in Aspergillus flavus: Association with aflatoxin production and morphology. Can. J. Bot. 1993, 71, 23–31. [Google Scholar] [CrossRef]
- Peterson, S.W. Phylogenic analysis of Aspergillus species using DNA sequences from four loci. Mycologia 2008, 100, 205–226. [Google Scholar] [CrossRef]
- Campbell, K.W.; White, D.G. Evaluation of corn genotypes for resistance to Aspergillus ear rot, kernel infection, and aflatoxin production. Plant Dis. 1995, 79, 1039–1045. [Google Scholar] [CrossRef]
- Michailide, T.J.; Thomidis, T. First report of Aspergillus flavus causing fruit rots of peaches in Greece. Plant Pathol. 2007, 56, 352. [Google Scholar] [CrossRef]
- Amaike, S.; Keller, N.P. Aspergillus flavus. Annu. Rev. Phytopathol. 2011, 49, 107–133. [Google Scholar] [CrossRef]
- Kensler, T.W.; Roebuck, B.D.; Wogan, G.N.; Groopman, J.D. Aflatoxin: A 50-year odyssey of mechanistic and translational toxicology. Toxicol. Sci. 2011, 120, S28–S48. [Google Scholar] [CrossRef]
- Klich, M.A. Aspergillus flavus: The major producer of aflatoxin. Mol. Plant Pathol. 2007, 8, 713–722. [Google Scholar] [CrossRef]
- Turner, P.C.; Moore, S.E.; Hall, A.J.; Prentice, A.M.; Wild, C.P. Modification of immune function through exposure to dietary aflatoxin in Gambian children. Environ. Health Perspect. 2003, 111, 217–220. [Google Scholar] [CrossRef]
- Hedayati, M.T.; Pasqualotto, A.C.; Warn, P.A.; Bowyer, P.; Denning, D.W. Aspergillus flavus: Human pathogen, allergen and mycotoxin producer. Microbiology 2007, 153, 1677–1692. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, S.; Manavathu, E.K.; Chandrasekar, P.H. Aspergillus flavus: An emerging non-fumigatus Aspergillus species of significance. Mycoses 2009, 52, 206–222. [Google Scholar] [CrossRef] [PubMed]
- Diener, U.L.; Cole, R.J.; Sanders, T.H.; Payne, G.A.; Lee, L.S.; Klich, M.A. Epidemiology of aflatoxin formation by Aspergillus flavus. Annu. Rev. Phytopathol. 1987, 25, 249–270. [Google Scholar] [CrossRef]
- Ramirez-Prado, J.H.; Moore, G.G.; Horn, B.W.; Carbone, I. Characterization and population analysis for the mating-type genes in Aspergillus flavus and Aspergillus parasiticus. Fungal Genet. Biol. 2008, 45, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Horn, B.W.; Moore, G.G.; Carbone, I. Sexual reproduction in Aspergillus flavus. Mycologia 2009, 101, 423–429. [Google Scholar] [CrossRef]
- Kim, H.-S.; Han, K.-Y.; Kim, K.-J.; Han, D.-M.; Jahng, K.-Y.; Chae, K.-S. The veA gene activates sexual development in Aspergillus nidulans. Fungal Genet. Biol. 2002, 37, 72–80. [Google Scholar] [CrossRef]
- Mooney, J.L.; Hassett, D.E.; Yager, L.N. Genetic analysis of suppressors of the veA1 mutation in Aspergillus nidulans. Genetics 1990, 126, 869–874. [Google Scholar]
- Bok, J.W.; Keller, N.P. LaeA, a regulator of secondary metabolism in Aspergillus spp. Eukaryot. Cell 2004, 3, 31–37. [Google Scholar] [CrossRef]
- Bayram, O.; Krappmann, S.; Ni, M.; Bok, J.W.; Helmstaedt, K.; Valerius, O.; Braus-Stromeyer, S.; Kwon, N.-J.; Keller, N.P.; Yu, J.-H.; et al. VelB/VeA/LaeA complex coordinates light signal with fungal development and secondary metabolism. Science 2008, 320, 1504–1506. [Google Scholar] [CrossRef]
- Ni, M.; Yu, J.-H. A novel regulator couples sporogenesis and trehalose biogenesis in Aspergillus nidulans. PLoS ONE 2007, 2, e970. [Google Scholar] [CrossRef]
- Amaike, S.; Keller, N.P. Distinct role for VeA and LaeA in development and pathogenesis of Aspergillus flavus. Eukaryot. Cell 2009, 8, 1051–1060. [Google Scholar] [CrossRef] [PubMed]
- Kale, S.P.; Milde, L.; Trapp, M.K.; Frisvad, J.C.; Keller, N.P.; Bok, J.W. Requirement of LaeA for secondary metabolism and sclerotial production in Aspergillus flavus. Fungal Genet. Biol. 2008, 45, 1422–1429. [Google Scholar] [CrossRef] [PubMed]
- Calvo, A.M.; Bok, J.; Brooks, W.; Keller, N.P. veA is required for toxin and sclerotial production in Aspergillus parasiticus. Appl. Environ. Microbiol. 2004, 70, 4733–4739. [Google Scholar] [CrossRef] [PubMed]
- Duran, R.M.; Cary, J.W.; Calvo, A.M. Production of cyclopiazonic acid, aflatrem, and aflatoxin by Aspergillus flavus is regulated by veA, a gene necessary for sclerotial formation. Appl. Microbiol. Biotechnol. 2007, 73, 1158–1168. [Google Scholar] [CrossRef]
- Ben-Ami, R.; Lewis, R.E.; Leventakos, K.; Konoyiannis, D.P. Aspergillus fumigatus inhibits angiogenesis through the production of gliotoxin and other secondary metabolites. Blood 2009, 114, 5393–5399. [Google Scholar] [CrossRef]
- Sugui, J.A.; Pargo, J.; Chang, Y.C.; Mȕllbacher, A.; Zarember, K.A.; Galvez, E.M.; Bringster, L.; Zerfas, P.; Gallin, J.I.; Simon, M.M.; et al. Role of laeA in the regulation of alb1, gliP, conidial morphology and virulence in Aspergillus fumigatus. Eukaryot. Cell. 2007, 6, 1552–1561. [Google Scholar] [CrossRef]
- Han, K.-H.; Han, K.-Y.; Yu, J.-H.; Chae, K.-S.; Jahng, K.-Y.; Han, D.-M. The nsdD gene encodes a putative GATA-type transcription factor necessary for sexual development of Aspergillus nidulans. Mol. Microbiol. 2001, 41, 299–309. [Google Scholar] [CrossRef]
- Kim, H.-R.; Chae, K.-S.; Han, K.-H.; Han, D.-M. The nsdC gene encoding a putative C2H2-type transcription factor is a key activator of sexual development in Aspergillus nidulans. Genetics 2009, 182, 771–783. [Google Scholar] [CrossRef]
- Cary, J.W.; Harris-Coward, P.Y.; Ehrlich, K.C.; Mack, B.M.; Kale, S.P.; Larey, C.; Calvo, A.M. NasC and NsdD affect Aspergillus flavus morphogenesis and aflatoxin production. Eukaryot. Cell 2012, 11, 1104–1111. [Google Scholar] [CrossRef]
- Dyer, P.S.; O’Gorman, C.M. Sexual development and cryptic sexuality in fungi: Insights from Aspergillus species. FEMS Microbiol. Rev. 2012, 36, 165–192. [Google Scholar] [CrossRef]
- Elion, E.A.; Satterberg, B.; Kranz, J.E. FUS3 phosphorylates multiple components of the mating signal transduction cascade: Evidence for STE12 and FAR1. Mol. Biol. Cell 1993, 4, 495–510. [Google Scholar] [CrossRef] [PubMed]
- Peter, M.; Gartner, A.; Horecka, J.; Ammerer, G.; Herskowitz, I. FAR1 links the signal transduction pathway to the cell cycle machinery in yeast. Cell 1993, 73, 747–760. [Google Scholar] [CrossRef]
- Pruyne, D.; Bretscher, A. Polarization of cell growth in yeast. I. Establishment and maintenance of polarity states. J. Cell Sci. 2000, 113, 365–375. [Google Scholar] [PubMed]
- Atoui, A.; Bao, D.; Kaur, N.; Grayburn, W.S.; Calvo, A.M. Aspergillus nidulans natural product biosynthesis is regulated by MpkB, a putative pheromone response mitogen-activated protein kinase. Appl. Environ. Microbiol. 2008, 74, 3596–3600. [Google Scholar] [CrossRef]
- Jun, S.-C.; Lee, S.-J.; Park, H.-J.; Kang, J.-Y.; Leem, Y.-E.; Yang, T.-H.; Chang, M.-H.; Kim, J.-M.; Jang, S.-H.; Kim, H.-K.; et al. The MpkB MAP kinase plays a role in post-karyogamy processes as well as in hyphal anastomosis during sexual development in Aspergillus nidulans. J. Microbiol. 2011, 49, 418–430. [Google Scholar] [CrossRef]
- Paoletti, M.; Seymour, F.A.; Alcocer, M.J.; Kaur, N.; Calvo, A.M.; Archer, D.D.; Dyer, P.S. Mating type and the genetic basis of self-fertility in the model fungus Aspergillus nidulans. Curr. Biol. 2007, 17, 1384–1389. [Google Scholar] [CrossRef]
- Bayram, O.; Bayram, O.S.; Ahmed, Y.L.; Maruyama, J.; Valerius, O.; Rizzoli, S.O.; Ficner, R.; Irniger, S.; Braus, G.H. The Aspergillus nidulans MAPK module AnSte11-Ste50-Ste7-Fus3 controls development and secondary metabolism. PLoS Genet. 2012, 8, e1002816. [Google Scholar] [CrossRef]
- Frawley, D.; Karahoda, B.; Bayram, O.S.; Bayram, O. The HamE scaffold positively regulates MpkB phosphorylation to promote development and secondary metabolism in Aspergillus nidulans. Sci. Rep. 2018, 8, 16588. [Google Scholar] [CrossRef]
- Frawley, D.; Greco, C.; Oakley, B.; Alhussain, M.M.; Fleming, A.B.; Keller, N.P.; Bayram, O. The tetrameric pheromone module SteC-MkkB-MpkB-SteD regulates asexual sporulation, sclerotia formation and aflatoxin production in Aspergillus flavus. Cell. Microbiol. 2020, 22, e13192. [Google Scholar] [CrossRef]
- He, Z.M.; Price, M.S.; Obrian, G.R.; Georgianna, D.R.; Payne, G.A. Improved protocols for functional analysis in the pathogenic fungus Aspergillus flavus. BMC Microbiol. 2007, 7, 104. [Google Scholar] [CrossRef]
- Pontecorvo, G.; Roper, J.A.; Hemmons, L.M.; MacDonald, K.D.; Bufton, A.W.J. The genetics of Aspergillus nidulans. Adv. Genet. 1953, 5, 141–238. [Google Scholar] [CrossRef]
- Käfer, E. Meiotic and mitotic recombination in Aspergillus and its chromosomal aberrations. Adv. Genet. 1977, 19, 33–131. [Google Scholar] [CrossRef] [PubMed]
- Han, K.-H.; Lee, D.-B.; Kim, J.-H.; Kim, M.-S.; Han, K.-Y.; Kim, W.-S.; Park, Y.-S.; Kim, H.-B.; Han, D.-M. Environmental factors affecting development of Aspergillus nidulans. J. Microbiol. 2003, 41, 34–40. [Google Scholar]
- Yu, J.-H.; Hamari, Z.; Han, K.-H.; Seo, J.-A.; Reyes-Dominquez, Y.; Scazzocchio, C. Double-joint PCR: A PCR-base molecular tool for gene manipulations in filamentous fungi. Fungal Genet. Biol. 2004, 41, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-H.; Butchko, R.A.; Fernandes, M.; Keller, N.P.; Leonard, T.J.; Adams, T.H. Conservation of structure and function of the aflatoxin regulatory gene aflR from Aspergillus nidulans and A. flavus. Curr. Genet. 1996, 29, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory Manual, 3rd ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2001; p. 2344. [Google Scholar]
- Horn, B.W.; Sorensen, R.B.; Lamb, M.C.; Sobolev, V.S.; Olarte, R.A.; Worthington, C.J.; Carbone, I. Sexual reproduction in Aspergillus flavus sclerotia naturally produced in corn. Phytopathology 2014, 104, 75–85. [Google Scholar] [CrossRef]
- Zhao, P.B.; Ren, A.Z.; Li, D.C. The FUS3/KSS1-type MAP Kinase gene FPK1 is involved in hyphal growth, conidiation and plant infection of Fusarium proliferatum. J. Mol. Microbiol. Biotechnol. 2011, 21, 110–119. [Google Scholar] [CrossRef]
- Jenczmionka, N.J.; Maier, F.J.; Lösch, A.P.; Schäfer, W. Mating, conidiation and pathogenicity of Fusarium graminearum, the main causal agent of the head-blight disease of wheat, are regulated by the MAP kinase gpmk1. Curr. Genet. 2003, 43, 87–95. [Google Scholar] [CrossRef]
- Schamber, A.; Leroch, M.; Diwo, J.; Mendgen, K.; Hahn, M. The role of mitogen-activated protein (MAP) kinase signalling components and the Ste12 transcription factor in germination and pathogenicity of Botrytis cinerea. Mol. Plant Pathol. 2010, 11, 105–119. [Google Scholar] [CrossRef]
- Lin, C.H.; Yang, S.L.; Wang, N.Y.; Chung, K.R. The FUS3 MAPK signaling pathway of the citrus pathogen Alternaria alternata functions independently or cooperatively with the fungal redox-responsive AP1 regulator for diverse developmental, physiological and pathogenic processes. Fungal Genet. Biol. 2010, 47, 381–391. [Google Scholar] [CrossRef]
- Lev, S.; Sharon, A.; Hadar, R.; Ma, H.; Horwitz, B.A. A mitogen-activated protein kinase of the corn leaf pathogen Cochliobolus heterostrophus is involved in conidiation, appressorium formation, and pathogenicity: Diverse roles for mitogen-activated protein kinase homologs in foliar pathogens. Proc. Natl. Acad. Sci. USA 1999, 96, 13542–13547. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.Y.; Chun, J.; Jun, S.-C.; Han, D.-M.; Chae, K.-S.; Jahng, K.Y. The MpkB MAP kinase plays a role in autolysis and conidiation of Aspergillus nidulans. Fungal Genet. Biol. 2013, 61, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Roca, M.G.; Read, N.D.; Glass, N.L. Role of a mitogen-activated protein kinase pathway during conidial germination and hyphal fusion in Neurospora crassa. Eukaryot. Cell. 2004, 3, 348–358. [Google Scholar] [CrossRef] [PubMed]
- Rispail, N.; Di Pietro, A. Fusarium oxysporum Ste12 controls invasive growth and virulence downstream of the Fmk1 MAPK cascade. Mol. Plant Microbe Interact. 2009, 22, 830–839. [Google Scholar] [CrossRef]
- Chen, C.; Harel, A.; Gorovoits, R.; Yarden, O.; Dickman, M.B. MAPK regulation of sclerotial development in Sclerotinia sclerotiorum is linked with pH and cAMP sensing. Mol. Plant Microbe Interact. 2004, 17, 404–413. [Google Scholar] [CrossRef]
- Horowitz, B.S.; Zarnowski, R.; Sharpee, W.C.; Keller, N.P. Morphological transitions governed by density dependence and lipoxygenase activity in Aspergillus flavus. Appl. Environ. Microbiol. 2008, 74, 5674–5685. [Google Scholar] [CrossRef]
- Chang, P.-K.; Scharfenstein, L.L.; Li, P.; Ehrlich, K.C. Aspergillus flavus VelB acts distinctly from VeA in conidiation and may coordinate with FluG to modulate sclerotial production. Fungal Genet. Biol. 2013, 58–59, 71–79. [Google Scholar] [CrossRef]
- Shwab, E.K.; Keller, N.P. Regulation of secondary metabolite production in filamentous ascomycetes. Mycol. Res. 2008, 112, 225–230. [Google Scholar] [CrossRef]
- Chang, P.-K.; Scharfenstein, L.L.; Mack, B.; Ehrlich, K.C. Deletion of the Aspergillus flavus orthologue of A. nidulans fluG reduces conidiation and promotes production of sclerotia but does not abolish aflatoxin biosynthesis. Appl. Environ. Microbiol. 2012, 78, 7557–7563. [Google Scholar] [CrossRef]
- Horn, B.W.; Greene, R.L.; Sobolev, V.S.; Dorner, J.W.; Powell, J.H.; Layton, R.C. Association of morphology and mycotoxin production with vegetative compatibility groups in Aspergillus flavus, A. parasiticus, and A. tamarii. Mycologia 1996, 88, 574–587. [Google Scholar] [CrossRef]
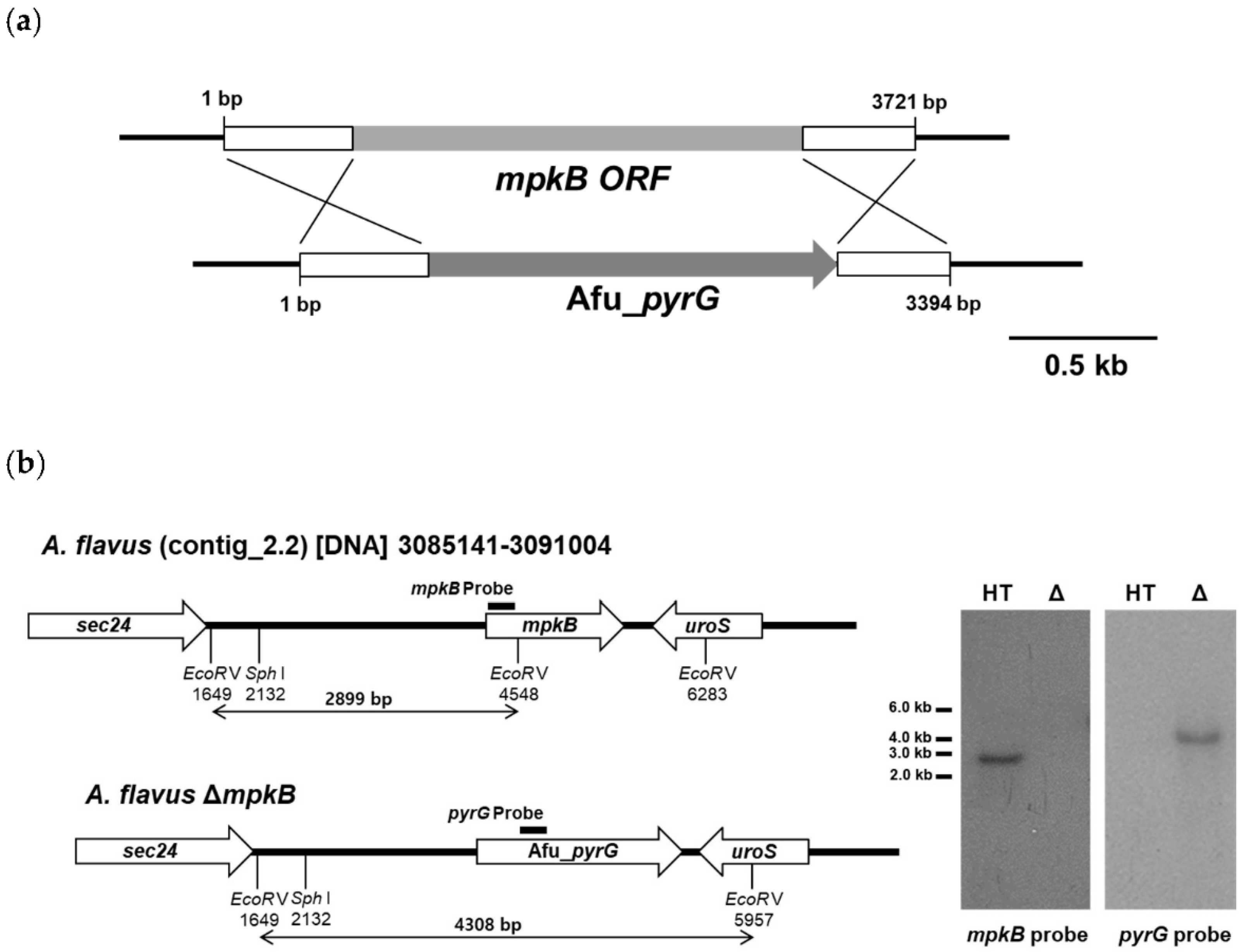

| Strain | Genotype | Use | Reference |
|---|---|---|---|
| NRRL3357 | Wild type | Wild-type strain | [40] |
| NRRL3357-5 | pyrG− | Recipient strain | [40] |
| SCWS1.02 | pyrG−, Afu_pyrG | pyrG complementation strain | This study |
| SCWS1.11 | pyrG−, ΔmpkB:Afu_pyrG | mpkB deletion strain | This study |
| SCWS1.20 | ΔmpkB::Afu_pyrG; mpkB, ptrA | mpkB complementation strain | This study |
| SCWS1.35 | pyrG−; niiA(p)::mpkB, Afu_pyrG | mpkB overexpression strain | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jun, S.-C.; Kim, J.-H.; Han, K.-H. The Conserved MAP Kinase MpkB Regulates Development and Sporulation without Affecting Aflatoxin Biosynthesis in Aspergillus flavus. J. Fungi 2020, 6, 289. https://doi.org/10.3390/jof6040289
Jun S-C, Kim J-H, Han K-H. The Conserved MAP Kinase MpkB Regulates Development and Sporulation without Affecting Aflatoxin Biosynthesis in Aspergillus flavus. Journal of Fungi. 2020; 6(4):289. https://doi.org/10.3390/jof6040289
Chicago/Turabian StyleJun, Sang-Cheol, Jong-Hwa Kim, and Kap-Hoon Han. 2020. "The Conserved MAP Kinase MpkB Regulates Development and Sporulation without Affecting Aflatoxin Biosynthesis in Aspergillus flavus" Journal of Fungi 6, no. 4: 289. https://doi.org/10.3390/jof6040289
APA StyleJun, S.-C., Kim, J.-H., & Han, K.-H. (2020). The Conserved MAP Kinase MpkB Regulates Development and Sporulation without Affecting Aflatoxin Biosynthesis in Aspergillus flavus. Journal of Fungi, 6(4), 289. https://doi.org/10.3390/jof6040289






