Zinc-Based Nanomaterials for Diagnosis and Management of Plant Diseases: Ecological Safety and Future Prospects
Abstract
1. Introduction
1.1. Use of Zinc Element as a Pesticide
1.2. Status of Use of Nanomaterials in Plant Pathology
2. Nanomaterials: Can Nanosizing Matter Alter Its Properties?
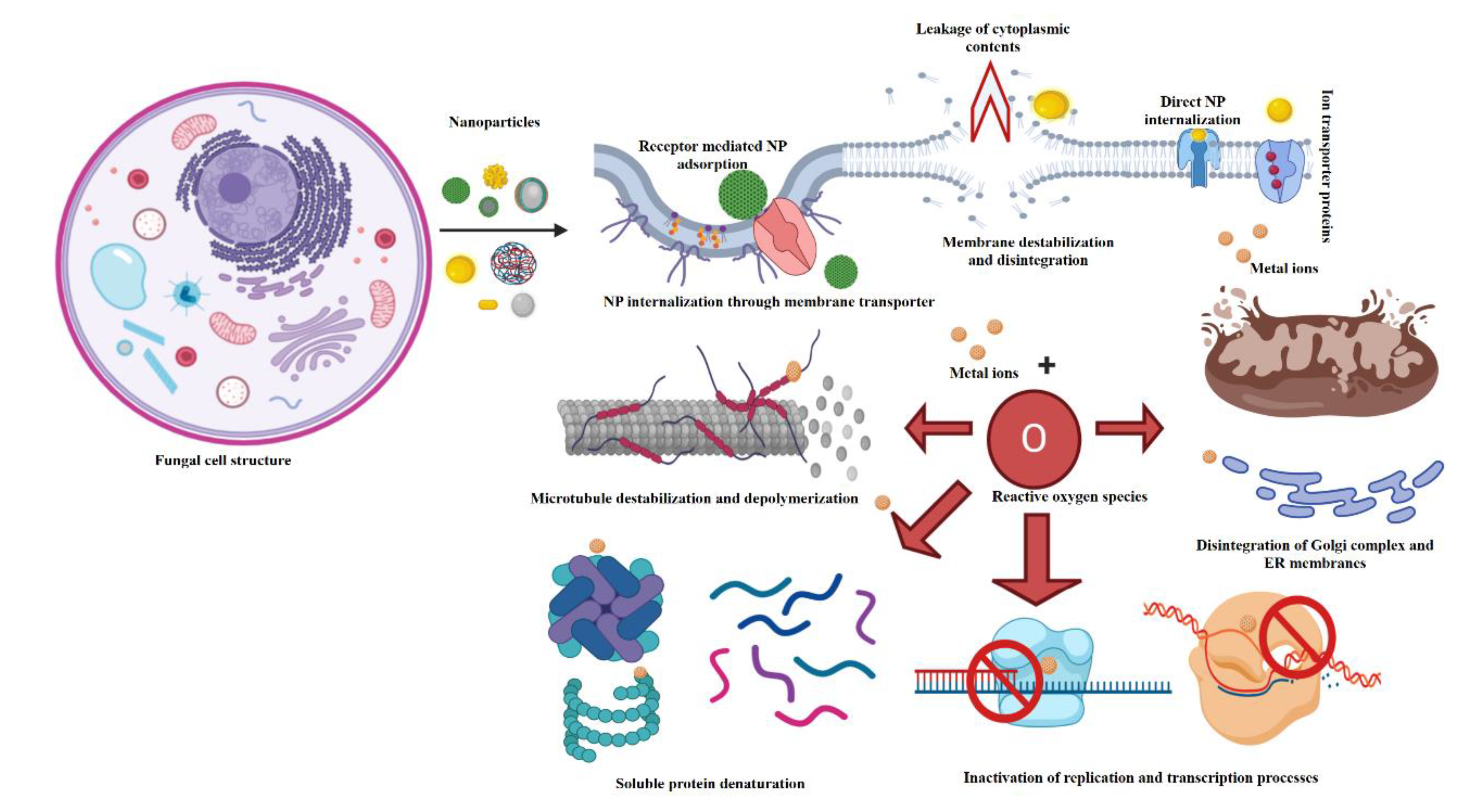
2.1. Mechanism of Antimicrobial Activity
2.1.1. Metal/Metal Oxides, Metalloid, and Nonmetal Nanomaterials
Mechanism of Antibacterial Activity of Nanomaterials
Mechanism of Antimycotic Activity of Nanomaterials
3. Zinc Nanomaterials and Their Use for Curbing Plant Disease-Causing Pathogens
3.1. Antibacterial and Mollicute Controlling Potential
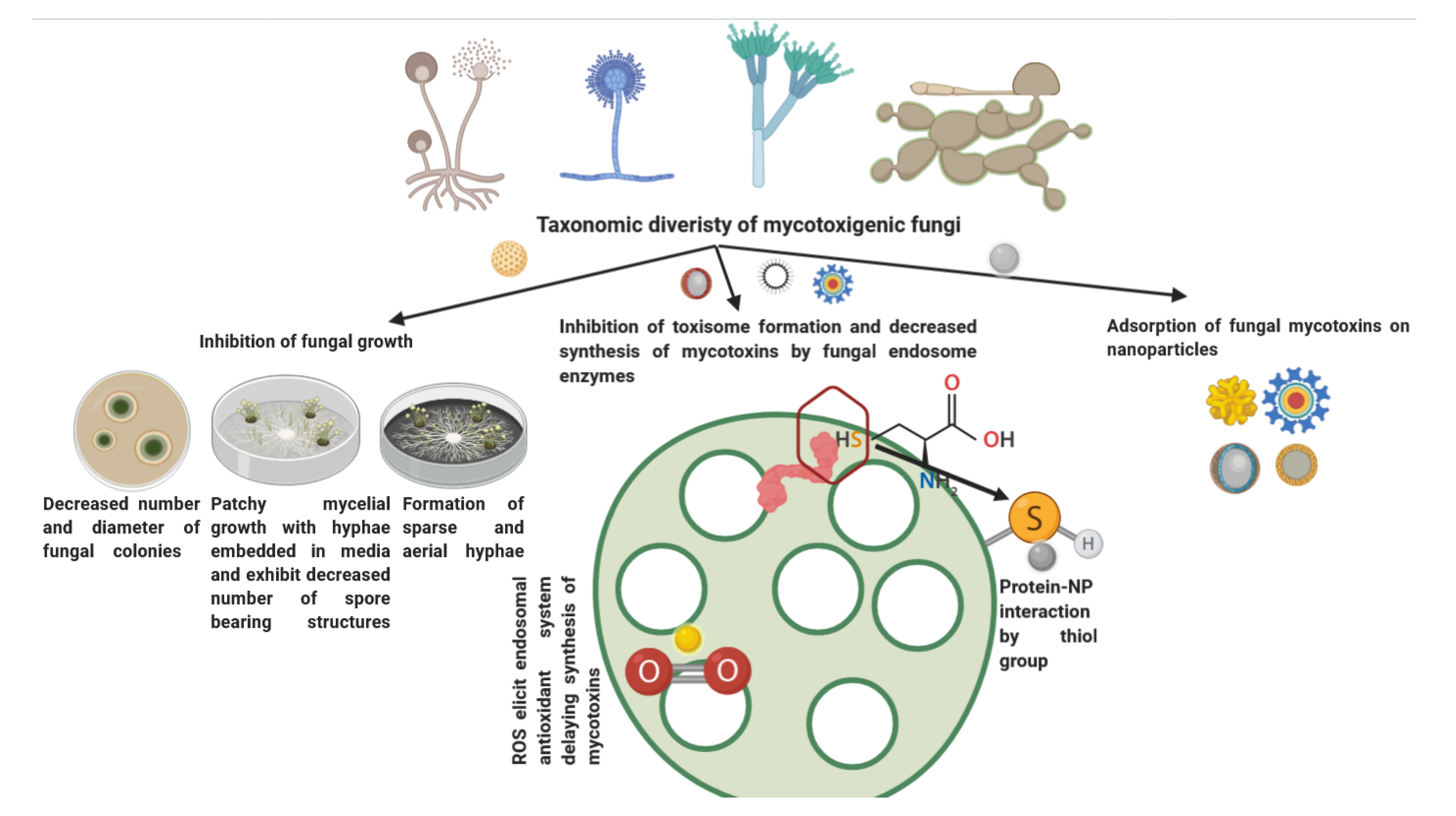
3.2. Antimycotic and Mycotoxin Neutralizing/Inhibiting Activity
3.2.1. Mechanism of Antimycotic Activity
3.2.2. Mycotoxin Neutralizing/Inhibiting Activity
3.2.3. Zinc Nanomaterials for Curbing Plant Viruses/Viroid Diseases
4. Zinc Nanoformulations: In Planta Studies and Crop Plant Responses to Pathogen Attacks
5. Zinc-Derived Nanomaterials for the Development of Tools/Devices for Plant Disease Diagnosis
6. Potential Application of Zn-Based Nanomaterials and Future Use
6.1. Ecosafety Issues of Nanozinc-Derived Products and Devices
6.2. Improved Nanozinc Formulations: The Scar and Sanctity of Stability and Biosafety
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Savary, S.; Willocquet, L.; Pethybridge, S.J.; Esker, P.; McRoberts, N.; Nelson, A. The global burden of pathogens and pests on major food crops. Nat. Ecol. Evol. 2019, 3, 430–439. [Google Scholar] [CrossRef]
- FAO. The State of Food and Agriculture 2019. In Moving Forward on Food Loss and Waste Reduction; FAO: Rome, Italy, 2019. [Google Scholar]
- Rajasekaran, P.; Kannan, H.; Das, S.; Young, M.; Santra, S. Comparative analysis of copper and zinc-based agrichemical biocide products: Materials characteristics, phytotoxicity and in vitro antimicrobial efficacy. AIMS Environ. Sci. 2016, 3, 439–455. [Google Scholar] [CrossRef]
- U.S. EPA/OPPTS (Environmental Protection Agency- Office of Prevention, Pesticides and Toxic Substances). Reregistration Eligibility Decision (R.E.D.) Facts-Zinc Salts. EPA 738-R-98-007, 1992, Washington, DC, U.S. Available online: https://archive.epa.gov/pesticides/reregistration/web/pdf/zinc_salt.pdf (accessed on 12 October 2020).
- Curry, A.S.; Price, D.E.; Tryhorn, F.G. Absorption of zinc phosphide particles. Nature 1959, 184, 642–643. [Google Scholar] [CrossRef]
- Hood, G.A. Zinc Phosphide-A new look at an old rodenticide for field rodents. In Proceedings of the 5th Vertebrate Pest Conference, Fresno, CA, USA, 7–9 March 1972; Volume 5, pp. 85–92. [Google Scholar]
- US-EPA. Zinc oxide: Exemption from the requirement of a tolerance. Fed. Regist. 2018, 83, 42783–42787. Available online: https://www.govinfo.gov/content/pkg/FR-2018-08-24/pdf/2018-18402.pdf (accessed on 12 October 2020).
- Almoudi, M.M.; Hussein, A.S.; Abu Hassan, M.I.; Mohamad Zain, N. A systematic review on antibacterial activity of zinc against Streptococcus mutans. Saudi Dent. J. 2018, 30, 283–291. [Google Scholar] [CrossRef]
- Burgess, J.; Prince, R.H. Zinc: Inorganic & Coordination Chemistry. In Encyclopedia of Inorganic Chemistry, 1st ed.; King, R.B., Ed.; John Wiley & Sons, Ltd., Wiley: Hoboken, NJ, USA, 2006; pp. 1–26. [Google Scholar]
- Dos Santos, R.A.A.; D’Addazio, V.; Silva, J.V.G.; Falqueto, A.R.; Barreto da Silva, M.; Schmildt, E.R.; Fernandes, A.A. Antifungal Activity of Copper, Zinc and Potassium Compounds on Mycelial Growth and Conidial Germination of Fusarium solani f. sp. piperis. Microbiol. Res. J. Int. 2019, 29, 1–11. [Google Scholar] [CrossRef]
- Goodwin, F.E. Zinc Compounds. In Kirk-Othmer Encycl. Chem. Technol.; Kroschwitz, J., Howe-Grant, M., Eds.; John Wiley & Sons, Inc.: New York, NY, USA, 1998; pp. 840–853. [Google Scholar]
- Qureshi, S.A.; Shafeeq, A.; Ijaz, A.; Butt, M.M. Development of algae guard façade paint with statistical modeling under natural phenomena. Coatings 2018, 8, 440. [Google Scholar] [CrossRef]
- Gupta, S.; Sharma, D.; Gupta, M. Climate change impact on plant diseases: Opinion, trends and mitigation strategies. In Microbes for Climate Resilient Agriculture; Kashyap, P.L., Srivastava, A.K., Tiwari, S.P., Kumar, S., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018; pp. 41–56. [Google Scholar]
- Luck, J.; Asaduzzaman, M.; Banerjee, S.; Bhattacharya, I.; Coughlan, K.; Debnath, G.; De Boer, D.; Dutta, S.; Forbes, G.; Griffiths, W.; et al. The Effects of Climate Change on Pests and Diseases of Major Food Crops in the Asia Pacific Region; Final Report for APN Project (Project Reference: ARCP2010-05CMY-Luck); Asia Pacific Network for Global Change Research: Kobe, Japan, 2010. [Google Scholar]
- Lurwanu, Y.; Wang, Y.P.; Abdul, W.; Zhan, J.; Yang, L.N. Temperature-mediated plasticity regulates the adaptation of Phytophthora infestans to azoxystrobin fungicide. Sustainability 2020, 12, 1188. [Google Scholar] [CrossRef]
- Ishii, H. Impact of fungicide resistance in plant pathogens on crop disease control and agricultural environment. Jpn. Agric. Res. Q. 2006, 40, 205–211. [Google Scholar] [CrossRef]
- Kalia, A.; Manchanda, P.; Bhardwaj, S.; Singh, G. Biosynthesized silver nanoparticles from aqueous extracts of sweet lime fruit and callus tissues possess variable antioxidant and antimicrobial potentials. Inorg. Nano-Metal Chem. 2020, 50, 1053–1062. [Google Scholar] [CrossRef]
- Kaur, G.; Kalia, A.; Sodhi, H.S. Size controlled, time-efficient biosynthesis of silver nanoparticles from Pleurotus florida using ultra-violet, visible range, and microwave radiations. Inorg. Nano-Metal Chem. 2020, 50, 35–41. [Google Scholar] [CrossRef]
- Munir, M.U.; Ihsan, A.; Javed, I.; Ansari, M.T.; Bajwa, S.Z.; Bukhari, S.N.A.; Ahmed, A.; Malik, M.Z.; Khan, W.S. Controllably biodegradable hydroxyapatite nanostructures for cefazolin delivery against antibacterial resistance. ACS Omega 2019, 4, 7524–7532. [Google Scholar] [CrossRef]
- Kalainila, P.; Ravindran, R.S.E.; Rohit, R.; Renganathan, S. Anti-bacterial effect of biosynthesized silver nanoparticles using Kigelia africana. J. Nanosci. Nanoengn. 2015, 1, 225–232. [Google Scholar]
- Jangra, S.L.; Stalin, K.; Dilbaghi, N.; Kumar, S.; Tawale, J.; Singh, S.P.; Pasricha, R. Antimicrobial activity of zirconia (ZrO2) nanoparticles and zirconium complexes. J. Nanosci. Nanotechnol. 2012, 12, 7105–7112. [Google Scholar] [CrossRef]
- Satyavani, K.; Gurudeeban, S.; Ramanathan, T.; Balasubramanian, T. Biomedical potential of silver nanoparticles synthesized from calli cells of Citrullus colocynthis (L.) Schrad. J. Nanobiotechnol. 2011, 9, 43. [Google Scholar] [CrossRef]
- Huang, F.; Long, Y.; Liang, Q.; Purushotham, B.; Swamy, M.K.; Duan, Y. Safed Musli (Chlorophytum borivilianum L.) Callus-Mediated Biosynthesis of Silver Nanoparticles and Evaluation of their Antimicrobial Activity and Cytotoxicity against Human Colon Cancer Cells. J. Nanomater. 2019, 1–8. [Google Scholar] [CrossRef]
- Azizi, S.; Mohamad, R.; Shahri, M.M.; McPhee, D.J. Green microwave-assisted combustion synthesis of zinc oxide nanoparticles with Citrullus colocynthis (L.) schrad: Characterization and biomedical applications. Molecules 2017, 22, 301. [Google Scholar] [CrossRef]
- Sánchez-López, E.; Gomes, D.; Esteruelas, G.; Bonilla, L.; Lopez-Machado, A.L.; Galindo, R.; Cano, A.; Espina, M.; Ettcheto, M.; Camins, A.; et al. Metal-based nanoparticles as antimicrobial agents: An overview. Nanomaterials 2020, 10, 292. [Google Scholar] [CrossRef]
- Singh, J.; Vishwakarma, K.; Ramawat, N.; Rai, P.; Singh, V.K.; Mishra, R.K.; Kumar, V.; Tripathi, D.K.; Sharma, S. Nanomaterials and microbes’ interactions: A contemporary overview. 3 Biotech 2019, 9, 68. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M. Antibacterial activity of nanomaterials. Nanomaterials 2018, 8, 359. [Google Scholar] [CrossRef]
- Mostafa, M.; Almoammar, H.; Abd-Elsalam, K.A. Zinc-based nanostructures in plant protection applications. In Nanobiotechnology Applications in Plant Protection, Nanotechnology in the Life Sciences; Abd-Elsalam, K.A., Prasad, R., Eds.; Springer Nature Switzerland AG: Cham, Switzerland, 2019; pp. 49–83. ISBN 9783030132965. [Google Scholar]
- Vollath, D. Nanomaterials: An Introduction to Synthesis, Properties, and Applications, 2nd ed.; Wiley-VCH, Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; ISBN 9780470927076. [Google Scholar]
- Siegel, R. Nanostructured materials. In Advanced Topics in Materials Science and Engineering; Morán-López, J.L., Sanchez, J.M., Eds.; Springer: Boston, MA, USA; New York, NY, USA, 1993; pp. 273–288. ISBN 9788578110796. [Google Scholar]
- Murr, L.E. Handbook of materials structures, properties, processing and performance. In Handbook of Materials Structures, Properties, Processing and Performance; Murr, L., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 719–746. ISBN 9783319018157. [Google Scholar]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Mourdikoudis, S.; Pallares, R.M.; Thanh, N.T.K. Characterization techniques for nanoparticles: Comparison and complementarity upon studying nanoparticle properties. Nanoscale 2018, 10, 12871–12934. [Google Scholar] [CrossRef]
- Roduner, E. Physics and Chemistry of Nanostructures: Why nano is different. Encycl. Life Support Syst. 2009, 1–35. [Google Scholar]
- Roduner, E. Size matters: Why nanomaterials are different. Chem. Soc. Rev. 2006, 35, 583–592. [Google Scholar] [CrossRef] [PubMed]
- de Voorde, M.; Tulinski, M.; Jurczyk, M. Engineered nanomaterials: A discussion of the major categories of nanomaterials. In Metrology and Standardization for Nanotechnology: Protocols and Industrial Innovations; Mansfield, E., Kaiser, M., Fujita, D., de Voorde, M., Eds.; Wiley-VCH, Verlag GmbH & Co. KgaA: Weinheim, Germany, 2017; pp. 49–73. [Google Scholar]
- Christian, P. Nanomaterials: Properties, Preparation and Applications. In Environmental and Human Health Impacts of Nanotechnology; Lead, J.R., Smith, E., Eds.; Wiley-Blackwell Publishing Ltd.: Chichester, UK, 2009; pp. 31–77. ISBN 978-1-405-17634-7. [Google Scholar]
- Sun, C.Q. Size dependence of nanostructures: Impact of bond order deficiency. Prog. Solid State Chem. 2007, 35, 1–159. [Google Scholar] [CrossRef]
- Andrievskii, R.A. Size-dependent effects in properties of nanostructured materials. Rev. Adv. Mater. Sci. 2009, 21, 107–133. [Google Scholar]
- Kalia, A.; Kaur, J.; Kaur, A.; Singh, N. Antimycotic activity of biogenically synthesised metal and metal oxide nanoparticles against plant pathogenic fungus Fusarium moniliforme (F. fujikuroi). Indian J. Exp. Biol. 2020, 58, 263–270. [Google Scholar]
- Khan, M.; Shaik, M.R.; Khan, S.T.; Adil, S.F.; Kuniyil, M.; Khan, M.; Al-Warthan, A.A.; Siddiqui, M.R.H.; Nawaz Tahir, M. Enhanced Antimicrobial Activity of Biofunctionalized Zirconia Nanoparticles. ACS Omega 2020, 5, 1987–1996. [Google Scholar] [CrossRef]
- Van Der Wal, A.; Norde, W.; Zehnder, A.J.B.; Lyklema, J. Determination of the total charge in the cell walls of Gram-positive bacteria. Colloids Surf. B Biointerfaces 1997, 9, 81–100. [Google Scholar] [CrossRef]
- Chen, M.; Zeng, G.; Xu, P.; Lai, C.; Tang, L. How Do Enzymes ‘Meet’ Nanoparticles and Nanomaterials? Trends Biochem. Sci. 2017, 42, 914–930. [Google Scholar] [CrossRef]
- Kaur, M.; Kalia, A. Role of salt precursors for the synthesis of zinc oxide nanoparticles and in imparting variable antimicrobial activity. J. Appl. Nat. Sci. 2016, 8, 1039–1048. [Google Scholar] [CrossRef]
- Cai, L.; Liu, C.; Fan, G.; Liu, C.; Sun, X. Preventing viral disease by ZnONPs through directly deactivating TMV and activating plant immunity in Nicotiana benthamiana. Environ. Sci. Nano 2019, 6, 3653–3669. [Google Scholar] [CrossRef]
- Alum, A.; Rashid, A.; Mobasher, B.; Abbaszadegan, M. Cement-based biocide coatings for controlling algal growth in water distribution canals. Cem. Concr. Compos. 2008, 30, 839–847. [Google Scholar] [CrossRef]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Antimicrobial activity of the metals and metal oxide nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef]
- Raghupathi, K.R.; Koodali, R.T.; Manna, A.C. Size-dependent bacterial growth inhibition and mechanism of antibacterial activity of zinc oxide nanoparticles. Langmuir 2011, 27, 4020–4028. [Google Scholar] [CrossRef]
- El-Sayed, A.S.A.; Ali, D.M.I. Biosynthesis and comparative bactericidal activity of silver nanoparticles synthesized by Aspergillus flavus and Penicillium crustosum against the multidrug-resistant bacteria. J. Microbiol. Biotechnol. 2018. [Google Scholar] [CrossRef]
- Jamdagni, P.; Rana, J.S.; Khatri, P.; Nehra, K. Comparative account of antifungal activity of green and chemically synthesized Zinc Oxide nanoparticles in combination with agricultural fungicides. Int. J. Nano Dimens. 2018, 9, 198–208. [Google Scholar]
- Auyeung, A.; Casillas-Santana, M.Á.; Martínez-Castañón, G.A.; Slavin, Y.N.; Zhao, W.; Asnis, J.; Häfeli, U.O.; Bach, H. Effective control of molds using a combination of nanoparticles. PLoS ONE 2017, 12, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Lamsal, K.; Kim, S.W.; Jung, J.H.; Kim, Y.S.; Kim, K.S.; Lee, Y.S. Inhibition effects of silver nanoparticles against powdery mildews on cucumber and pumpkin. Mycobiology 2011, 39, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-J.; Kim, S.H.; Kim, H.J.; Choi, S.-H. A new composition of nanosized silica-silver for control of various plant diseases. Plant Pathol. J. 2006, 22, 295–302. [Google Scholar] [CrossRef]
- Kim, H.; Kang, H.; Chu, G.; Byun, H. Antifungal effectiveness of nanosilver colloid against rose powdery mildew in greenhouses. Solid State Phenom. 2008, 135, 15–18. [Google Scholar] [CrossRef]
- Li, J.; Sang, H.; Guo, H.; Popko, J.T.; He, L.; White, J.C.; Parkash Dhankher, O.; Jung, G.; Xing, B. Antifungal mechanisms of ZnO and Ag nanoparticles to Sclerotinia homoeocarpa. Nanotechnology 2017, 28, 155101. [Google Scholar] [CrossRef] [PubMed]
- De La Rosa-García, S.C.; Martínez-Torres, P.; Gómez-Cornelio, S.; Corral-Aguado, M.A.; Quintana, P.; Gómez-Ortíz, N.M. Antifungal activity of ZnO and MgO nanomaterials and their mixtures against colletotrichum gloeosporioides strains from tropical fruit. J. Nanomater. 2018, 2018. [Google Scholar] [CrossRef]
- Karimiyan, A.; Najafzadeh, H.; Ghorbanpour, M.; Hekmati-Moghaddam, S.H. Antifungal Effect of Magnesium Oxide, Zinc Oxide, Silicon Oxide and Copper Oxide Nanoparticles Against Candida albicans. Zahedan J. Res. Med. Sci. 2015, 17, 2–4. [Google Scholar] [CrossRef]
- Roy, A.; Gauri, S.S.; Bhattacharya, M.; Bhattacharya, J. Antimicrobial activity of CaO nanoparticles. J. Biomed. Nanotechnol. 2013, 9, 1570–1578. [Google Scholar] [CrossRef]
- Malandrakis, A.A.; Kavroulakis, N.; Chrysikopoulos, C.V. Use of copper, silver and zinc nanoparticles against foliar and soil-borne plant pathogens. Sci. Total Environ. 2019, 670, 292–299. [Google Scholar] [CrossRef]
- Choudhary, M.A.; Manan, R.; Aslam Mirza, M.; Rashid Khan, H.; Qayyum, S.; Ahmed, Z. Biogenic Synthesis of Copper oxide and Zinc oxide Nanoparticles and their Application as Antifungal Agents. Int. J. Mater. Sci. Eng. 2018, 4, 1–6. [Google Scholar] [CrossRef]
- Hao, Y.; Cao, X.; Ma, C.; Zhang, Z.; Zhao, N.; Ali, A.; Hou, T.; Xiang, Z.; Zhuang, J.; Wu, S.; et al. Potential Applications and Antifungal Activities of Engineered Nanomaterials against Gray Mold Disease Agent Botrytis cinerea on Rose Petals. Front. Plant Sci. 2017, 8, 1–9. [Google Scholar] [CrossRef]
- Al-Dhabaan, F.A.; Shoala, T.; Ali, A.A.; Alaa, M.; Abd-Elsalam, K.; Abd-Elsalam, K. Chemically-produced copper, zinc nanoparticles and chitosan-bimetallic nanocomposites and their antifungal activity against three phytopathogenic fungi. Int. J. Agric. Technol. 2017, 13, 753–769. [Google Scholar]
- Vahedi, M.; Hosseini-Jazani, N.; Yousefi, S.; Ghahremani, M. Evaluation of anti-bacterial effects of nickel nanoparticles on biofilm production by Staphylococcus epidermidis. Iran. J. Microbiol. 2017, 9, 160–168. [Google Scholar]
- Srihasam, S.; Thyagarajan, K.; Korivi, M.; Lebaka, V.R.; Mallem, S.P.R. Phytogenic generation of NiO nanoparticles using stevia leaf extract and evaluation of their in-vitro antioxidant and antimicrobial properties. Biomolecules 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Bogdan, J.; Zarzyńska, J.; Pławińska-Czarnak, J. Comparison of Infectious Agents Susceptibility to Photocatalytic Effects of Nanosized Titanium and Zinc Oxides: A Practical Approach. Nanoscale Res. Lett. 2015, 10. [Google Scholar] [CrossRef]
- Yehia, R.; Ahmed, O.F. In vitro study of the antifungal efficacy of zinc oxide nanoparticles against Fusarium oxysporum and Penicilium expansum. Afr. J. Microbiol. Res. 2013, 7, 1917–1923. [Google Scholar] [CrossRef]
- Elsharkawy, M.; Derbalah, A.; Hamza, A.; El-Shaer, A. Zinc oxide nanostructures as a control strategy of bacterial speck of tomato caused by Pseudomonas syringae in Egypt. Environ. Sci. Pollut. Res. 2018, 27, 19049–19057. [Google Scholar] [CrossRef] [PubMed]
- Arciniegas-Grijalba, P.A.; Patiño-Portela, M.C.; Mosquera-Sánchez, L.P.; Guerrero-Vargas, J.A.; Rodríguez-Páez, J.E. ZnO nanoparticles (ZnO-NPs) and their antifungal activity against coffee fungus Erythricium salmonicolor. Appl. Nanosci. 2017, 7, 225–241. [Google Scholar] [CrossRef]
- Rana, P.; Abdullah, M.; Hameed, H.Q.; Hasan, A.A. The effect of Olea europea L. leaves extract and ZrO2 nanoparticles on Acinetobacter baumannii. J. Pharm. Sci. Res. 2019, 11, 2019. [Google Scholar]
- Joshi, S.M.; De Britto, S.; Jogaiah, S.; Ito, S.I. Mycogenic selenium nanoparticles as potential new generation broad spectrum antifungal molecules. Biomolecules 2019, 9, 419. [Google Scholar] [CrossRef]
- Srivastava, N.; Mukhopadhyay, M. Green synthesis and structural characterization of selenium nanoparticles and assessment of their antimicrobial property. Bioprocess Biosyst. Eng. 2015, 38. [Google Scholar] [CrossRef]
- Khiralla, G.M.; El-Deeb, B.A. Antimicrobial and antibiofilm effects of selenium nanoparticles on some foodborne pathogens. LWT Food Sci. Technol. 2015, 63, 1001–1007. [Google Scholar] [CrossRef]
- Shakibaie, M.; Forootanfar, H.; Golkari, Y.; Mohammadi-Khorsand, T.; Shakibaie, M.R. Anti-biofilm activity of biogenic selenium nanoparticles and selenium dioxide against clinical isolates of Staphylococcus aureus, Pseudomonas aeruginosa, and Proteus mirabilis. J. Trace Elem. Med. Biol. 2015, 29, 235–241. [Google Scholar] [CrossRef]
- Abo Elsoud, M.M.; Al-Hagar, O.E.A.; Abdelkhalek, E.S.; Sidkey, N.M. Synthesis and investigations on tellurium myconanoparticles. Biotechnol. Rep. 2018, 18, e00247. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.D.; Cruz, D.M.; Roy, A.K.; Webster, T.J. Synthesis and characterization of PVP-coated tellurium nanorods and their antibacterial and anticancer properties. J. Nanopart. Res. 2018, 20, 254. [Google Scholar] [CrossRef]
- Siddiqi, K.S.; ur Rahman, A.; Tajuddin; Husen, A. Properties of Zinc Oxide Nanoparticles and Their Activity Against Microbes. Nanoscale Res. Lett. 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Jaffri, S.B.; Ahmad, K.S. Foliar-mediated Ag:ZnO nanophotocatalysts: Green synthesis, characterization, pollutants degradation, and in vitro biocidal activity. Green Process. Synth. 2019, 8, 172–182. [Google Scholar] [CrossRef]
- Mohamed, M.A.; Abd-Elsalam, K.A. Nanoantimicrobials for Plant Pathogens Control: Potential Applications and Mechanistic Aspects. In Nanobiotechnology Applications in Plant Protection-Nanotechnology in the Life Sciences; Abd-Elsalam, K.A., Prasad, R., Eds.; Springer International Publishing AG: Cham, Switzerland, 2018; pp. 111–135. ISBN 978-3-319-91160-1. [Google Scholar]
- Agarwal, H.; Menon, S.; Venkat Kumar, S.; Rajeshkumar, S. Mechanistic study on antibacterial action of zinc oxide nanoparticles synthesized using green route. Chem. Biol. Interact. 2018, 286, 60–70. [Google Scholar] [CrossRef]
- Al-Shabib, N.A.; Husain, F.M.; Ahmed, F.; Khan, R.A.; Ahmad, I.; Alsharaeh, E.; Khan, M.S.; Hussain, A.; Rehman, M.T.; Yusuf, M.; et al. Biogenic synthesis of Zinc oxide nanostructures from Nigella sativa seed: Prospective role as food packaging material inhibiting broad-spectrum quorum sensing and biofilm. Sci. Rep. 2016, 6, 1–16. [Google Scholar] [CrossRef]
- Ogunyemi, S.O.; Abdallah, Y.; Zhang, M.; Fouad, H.; Hong, X.; Ibrahim, E.; Masum, M.M.I.; Hossain, A.; Mo, J.; Li, B. Green synthesis of zinc oxide nanoparticles using different plant extracts and their antibacterial activity against Xanthomonas oryzae pv. oryzae. Artif. Cells Nanomed. Biotechnol. 2019, 47, 341–352. [Google Scholar] [CrossRef]
- Galié, S.; García-Gutiérrez, C.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Biofilms in the food industry: Health aspects and control methods. Front. Microbiol. 2018, 9, 1–18. [Google Scholar] [CrossRef]
- Kim, S.W.; Kim, K.S.; Lamsal, K.; Kim, Y.J.; Kim, S.B.; Jung, M.; Sim, S.J.; Kim, H.S.; Chang, S.J.; Kim, J.K.; et al. An in vitro study of the antifungal effect of silver nanoparticles on oak wilt pathogen Raffaelea sp. J. Microbiol. Biotechnol. 2009, 19, 760–764. [Google Scholar] [CrossRef]
- Lamsal, K.; Kim, S.W.; Jung, J.H.; Kim, Y.S.; Kim, K.S.; Lee, Y.S. Application of silver nanoparticles for the control of Colletotrichum species in vitro and pepper anthracnose disease in field. Mycobiology 2011, 39, 194–199. [Google Scholar] [CrossRef]
- Elmer, W.; White, J.C. The Future of Nanotechnology in Plant Pathology. Annu. Rev. Phytopathol. 2018, 56, 111–133. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Park, H.H.; Kim, S.Y.; Kim, S.J.; Woo, K.; Ko, G.P. Antiviral properties of silver nanoparticles on a magnetic hybrid colloid. Appl. Environ. Microbiol. 2014, 80, 2343–2350. [Google Scholar] [CrossRef] [PubMed]
- Galdiero, S.; Falanga, A.; Vitiello, M.; Cantisani, M.; Marra, V.; Galdiero, M. Silver nanoparticles as potential antiviral agents. Molecules 2011, 16, 8894–8918. [Google Scholar] [CrossRef]
- Zeedan, G.S.G.; Abd El-Razik, K.A.; Allam, A.M.; Abdalhamed, A.M.; Abou Zeina, H.A. Evaluations of potential antiviral effects of green zinc oxide and silver nanoparticles against bovine herpesvirus-1. Adv. Anim. Vet. Sci. 2020, 8, 433–443. [Google Scholar] [CrossRef]
- Bekele, A.Z.; Gokulan, K.; Williams, K.M.; Khare, S. Dose and Size-Dependent Antiviral Effects of Silver Nanoparticles on Feline Calicivirus, a Human Norovirus Surrogate. Foodborne Pathog. Dis. 2016, 13, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Shionoiri, N.; Sato, T.; Fujimori, Y.; Nakayama, T.; Nemoto, M.; Matsunaga, T.; Tanaka, T. Investigation of the antiviral properties of copper iodide nanoparticles against feline calicivirus. J. Biosci. Bioeng. 2012, 113, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Kerry, R.G.; Malik, S.; Redda, Y.T.; Sahoo, S.; Patra, J.K.; Majhi, S. Nano-based approach to combat emerging viral (NIPAH virus) infection. Nanomed. Nanotechnol. Biol. Med. 2019, 18, 196–220. [Google Scholar] [CrossRef]
- Ben Salem, A.N.; Zyed, R.; Lassoued, M.A.; Nidhal, S.; Sfar, S.; Mahjoub, A. Plant-derived nanoparticles enhance antiviral activity against coxsakievirus B3 by acting on virus particles and vero cells. Dig. J. Nanomater. Biostructs 2012, 7, 737–744. [Google Scholar]
- Rai, M.; Deshmukh, S.D.; Ingle, A.P.; Gupta, I.R.; Galdiero, M.; Galdiero, S. Metal nanoparticles: The protective nanoshield against virus infection. Crit. Rev. Microbiol. 2016, 42, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Singh, L.; Kruger, H.G.; Maguire, G.E.M.; Govender, T.; Parboosing, R. The role of nanotechnology in the treatment of viral infections. Ther. Adv. Infect. Dis. 2017, 4, 105–131. [Google Scholar] [CrossRef] [PubMed]
- Milovanovic, M.; Arsenijevic, A.; Milovanovic, J.; Kanjevac, T.; Arsenijevic, N. Nanoparticles in Antiviral Therapy. Antimicrob. Nanoarchit. Synth. Appl. 2017, 383–410. [Google Scholar] [CrossRef]
- Nikaeen, G.; Abbaszadeh, S.; Yousefinejad, S. Application of nanomaterials in treatment, anti-infection and detection of coronaviruses. Nanomedicine 2020. [Google Scholar] [CrossRef]
- Itani, R.; Tobaiqy, M.; Al Faraj, A. Optimizing use of theranostic nanoparticles as a life-saving strategy for treating COVID-19 patients. Theranostics 2020, 10, 5932–5942. [Google Scholar] [CrossRef]
- Haggag, E.G.; Elshamy, A.M.; Rabeh, M.A.; Gabr, N.M.; Salem, M.; Youssif, K.A.; Samir, A.; Bin Muhsinah, A.; Alsayari, A.; Abdelmohsen, U.R. Antiviral potential of green synthesized silver nanoparticles of Lampranthus coccineus and Malephora lutea. Int. J. Nanomed. 2019, 14, 6217–6229. [Google Scholar] [CrossRef] [PubMed]
- Meléndez-Villanueva, M.A.; Morán-Santibañez, K.; Martínez-Sanmiguel, J.J.; Rangel-López, R.; Garza-Navarro, M.A.; Rodríguez-Padilla, C.; Zarate-Triviño, D.G.; Trejo-Ávila, L.M. Virucidal activity of gold nanoparticles synthesized by green chemistry using garlic extract. Viruses 2019, 11, 1111. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Sahoo, G.; Pandey, K.; Nayak, M.K.; Topno, R.; Rabidas, V.; Das, P. Virostatic potential of zinc oxide (ZnO) nanoparticles on capsid protein of cytoplasmic side of chikungunya virus. Int. J. Infect. Dis. 2018, 73, 368. [Google Scholar] [CrossRef]
- Abdul, W.; Muhammad, A.; Atta Ullah, K.; Asmat, A.; Abdul, B. Role of nanotechnology in diagnosing and treating COVID-19 during the Pandemi. Int. J. Clin. Virol. 2020, 4, 65–70. [Google Scholar] [CrossRef]
- El-Dougdoug, N.K.; Bondok, A.M.; El-Dougdoug, K.A. Evaluation of Silver Nanoparticles as Antiviral Agent Against ToMV and PVY in Tomato Plants. Middle East J. Appl. Sci. 2018, 8, 100–111. [Google Scholar]
- Shafie, R.M.; Salama, A.M.; Farroh, K.Y. Silver nanoparticles activity against Tomato spotted wilt virus. Middle East J. Appl. Sci. 2018, 7, 1251–1267. [Google Scholar]
- Elbeshehy, E.K.F.; Elazzazy, A.M.; Aggelis, G. Silver nanoparticles synthesis mediated by new isolates of Bacillus spp., nanoparticle characterization and their activity against Bean Yellow Mosaic Virus and human pathogens. Front. Microbiol. 2015, 6, 1–13. [Google Scholar] [CrossRef]
- Elazzazy, A.M.; Elbeshehy, E.K.F.; Betiha, M.A. In vitro assessment of activity of graphene silver composite sheets against multidrug-resistant bacteria and Tomato Bushy Stunt Virus. Trop. J. Pharm. Res. 2017, 16, 2705–2711. [Google Scholar] [CrossRef]
- Cai, L.; Cai, L.; Jia, H.; Liu, C.; Wang, D.; Sun, X. Foliar exposure of Fe3O4 nanoparticles on Nicotiana benthamiana: Evidence for nanoparticles uptake, plant growth promoter and defense response elicitor against plant virus. J. Hazard. Mater. 2020, 393, 122415. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Yuan, W.; Ma, C.; White, J.C.; Zhang, Z.; Adeel, M.; Zhou, T.; Rui, Y.; Xing, B. Engineered nanomaterials suppress Turnip mosaic virus infection in tobacco (Nicotiana benthamiana). Environ. Sci. Nano 2018, 5, 1685–1693. [Google Scholar] [CrossRef]
- Hanley, C.; Layne, J.; Punnoose, A.; Reddy, K.M.; Coombs, I.; Coombs, A.; Feris, K.; Wingett, D. Preferential killing of cancer cells and activated human T cells using ZnO nanoparticles. Nanotechnology 2008, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Premanathan, M.; Karthikeyan, K.; Jeyasubramanian, K.; Manivannan, G. Selective toxicity of ZnO nanoparticles toward Gram-positive bacteria and cancer cells by apoptosis through lipid peroxidation. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.M.; Feris, K.; Bell, J.; Wingett, D.G.; Hanley, C.; Punnoose, A. Selective toxicity of zinc oxide nanoparticles to prokaryotic and eukaryotic systems. Appl. Phys. Lett. 2007, 90, 1–8. [Google Scholar] [CrossRef]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on zinc oxide nanoparticles: Antibacterial activity and toxicity mechanism. Nano-Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef]
- Tiwari, V.; Mishra, N.; Gadani, K.; Solanki, P.S.; Shah, N.A.; Tiwari, M. Mechanism of anti-bacterial activity of zinc oxide nanoparticle against Carbapenem-Resistant Acinetobacter baumannii. Front. Microbiol. 2018, 9, 1–10. [Google Scholar] [CrossRef]
- Sun, T.; Hao, H.; Hao, W.T.; Yi, S.M.; Li, X.P.; Li, J.R. Preparation and antibacterial properties of titanium-doped ZnO from different zinc salts. Nanoscale Res. Lett. 2014, 9, 1–11. [Google Scholar] [CrossRef]
- Jiang, J.; Pi, J.; Cai, J. The Advancing of Zinc Oxide Nanoparticles for Biomedical Applications. Bioinorg. Chem. Appl. 2018, 2018. [Google Scholar] [CrossRef]
- Pinto, R.M.; Lopes-De-Campos, D.; Martins, M.C.L.; Van Dijck, P.; Nunes, C.; Reis, S. Impact of nanosystems in Staphylococcus aureus biofilms treatment. FEMS Microbiol. Rev. 2019, 43, 622–641. [Google Scholar] [CrossRef] [PubMed]
- Graham, J.H.; Johnson, E.G.; Myers, M.E.; Young, M.; Rajasekaran, P.; Das, S.; Santra, S. Potential of Nano-Formulated Zinc Oxide for Control of Citrus Canker on Grapefruit Trees. Plant Dis. 2016, 100, 2442–2447. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, R.; Duman, K.; Jones, J.B.; Paret, M.L. Bactericidal Activity of Copper-Zinc Hybrid Nanoparticles on Copper-Tolerant Xanthomonas perforans. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, Z.A.; Khan, A.; Khan, M.R.; Abd-Allah, E.F. Effects of zinc oxide nanoparticles (ZnO NPs) and some plant pathogens on the growth and nodulation of lentil (Lens culinaris medik.). Acta Phytopathol. Entomol. Hungarica 2018, 53, 195–212. [Google Scholar] [CrossRef]
- Khan, M.; Siddiqui, Z.A. Zinc oxide nanoparticles for the management of Ralstonia solanacearum, Phomopsis vexans and Meloidogyne incognita incited disease complex of eggplant. Indian Phytopathol. 2018, 71, 355–364. [Google Scholar] [CrossRef]
- Alves, M.M.; Bouchami, O.; Tavares, A.; Córdoba, L.; Santos, C.F.; Miragaia, M.; De Fátima Montemor, M. New Insights into Antibiofilm Effect of a Nanosized ZnO Coating against the Pathogenic Methicillin Resistant Staphylococcus aureus. ACS Appl. Mater. Interfaces 2017, 9, 28157–28167. [Google Scholar] [CrossRef]
- Fontecha-Umaña, F.; Ríos-Castillo, A.G.; Ripolles-Avila, C.; Rodríguez-Jerez, J.J. Antimicrobial activity and prevention of bacterial biofilm formation of silver and zinc oxide nanoparticle-containing polyester surfaces at various concentrations for use. Foods 2020, 9, 442. [Google Scholar] [CrossRef]
- Jindal, S.; Anand, S.; Huang, K.; Goddard, J.; Metzger, L.; Amamcharla, J. Evaluation of modified stainless steel surfaces targeted to reduce biofilm formation by common milk spore formers. J. Dairy Sci. 2016, 99, 9502–9513. [Google Scholar] [CrossRef]
- Espitia, P.J.P.; Otoni, C.G.; Soares, N.F.F. Zinc Oxide Nanoparticles for Food Packaging Applications; Elsevier Inc.: Amsterdam, The Netherlands, 2016; ISBN 9780128007235. [Google Scholar]
- Kaur, M.; Kalia, A.; Thakur, A. Effect of biodegradable chitosan–rice-starch nanocomposite films on post-harvest quality of stored peach fruit. Starch/Staerke 2017, 69, 1–12. [Google Scholar] [CrossRef]
- Naskar, A.; Khan, H.; Sarkar, R.; Kumar, S.; Halder, D.; Jana, S. Anti-biofilm activity and food packaging application of room temperature solution process-based polyethylene glycol capped Ag-ZnO-graphene nanocomposite. Mater. Sci. Eng. C 2018, 91, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Gundersen, D.E.; Lee, I.M.; Rehner, S.A.; Davis, R.E.; Kingsbury, D.T. Phylogeny of mycoplasmalike organisms (phytoplasmas): A basis for their classification. J. Bacteriol. 1994, 176, 5244–5254. [Google Scholar] [CrossRef] [PubMed]
- Bové, J.M.; Garnier, M. Walled and wall-less eubacteria from plants: Sieve-tube-restricted plant pathogens. Plant Cell. Tissue Organ Cult. 1998, 52, 7–16. [Google Scholar] [CrossRef]
- Lee, I.; Davis, R.E.; Dawn, E. Phytoplasma: Phytopathogenic Mollicutes. Annu. Rev. Microbiol. 2000, 54, 221–255. [Google Scholar] [CrossRef] [PubMed]
- Jurga, M.; Zwolińska, A. Phytoplasmas in Poaceae species: A threat to the most important cereal crops in Europe. J. Plant Pathol. 2020, 102, 287–297. [Google Scholar] [CrossRef]
- Rao, G.P.; Madhupriya; Thorat, V.; Manimekalai, R.; Tiwari, A.K.; Yadav, A. A century progress of research on phytoplasma diseases in India. Phytopathog. Mollicutes 2017, 7, 1. [Google Scholar] [CrossRef]
- Kumari, S.; Nagendran, K.; Rai, A.B.; Singh, B.; Rao, G.P.; Bertaccini, A. Global status of phytoplasma diseases in vegetable crops. Front. Microbiol. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Namba, S. Molecular and biological properties of phytoplasmas. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2019, 95, 401–418. [Google Scholar] [CrossRef]
- Fletcher, J.; Wayadande, A.; Melcher, U.; Ye, F. The phytopathogenic mollicute-insect vector interface: A closer look. Phytopathology 1998, 88, 1351–1358. [Google Scholar] [CrossRef]
- Bendix, C.; Lewis, J.D. The enemy within: Phloem-limited pathogens. Mol. Plant Pathol. 2018, 19, 238–254. [Google Scholar] [CrossRef]
- Cagliari, D.; Dias, N.P.; Galdeano, D.M.; dos Santos, E.Á.; Smagghe, G.; Zotti, M.J. Management of pest insects and plant diseases by non-transformative RNAi. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Liu, S.; Jaouannet, M.; Dempsey, D.A.; Imani, J.; Coustau, C.; Kogel, K.H. RNA-based technologies for insect control in plant production. Biotechnol. Adv. 2020, 39, 107463. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Powell, C.A.; Duan, Y.; Shatters, R.; Zhang, M. Antimicrobial nanoemulsion formulation with improved penetration of foliar spray through citrus leaf cuticles to control citrus huanglongbing. PLoS ONE 2015, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Zhong, Y.; Powell, C.A.; Doud, M.S.; Duan, Y.; Huang, Y.; Zhang, M. Antimicrobial Compounds Effective against Candidatus Liberibacter asiaticus Discovered via Graft-based Assay in Citrus. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Gabiel, D.W.; Zhang, S. Use of Aldehydes Formulated with Nanoparticles and/or Nanoemulsions to Enhance Disease Resistance of Plants to Liberibacters. US Patent (US20170006863), 12 January 2017. [Google Scholar]
- Ghosh, D.K.; Kokane, S.; Kumar, P.; Ozcan, A.; Warghane, A.; Motghare, M.; Santra, S.; Sharma, A.K. Antimicrobial nano-zinc oxide-2S albumin protein formulation significantly inhibits growth of “Candidatus Liberibacter asiaticus” in planta. PLoS ONE 2018, 13, 1–20. [Google Scholar] [CrossRef]
- Wagner, G.; Korenkov, V.; Judy, J.D.; Bertsch, P.M. Nanoparticles composed of Zn and ZnO inhibit Peronospora tabacina spore germination in vitro and P. tabacina infectivity on tobacco leaves. Nanomaterials 2016, 6, 50. [Google Scholar] [CrossRef]
- Zabrieski, Z.; Morrell, E.; Hortin, J.; Dimkpa, C.; McLean, J.; Britt, D.; Anderson, A. Pesticidal activity of metal oxide nanoparticles on plant pathogenic isolates of Pythium. Ecotoxicology 2015, 24, 1305–1314. [Google Scholar] [CrossRef] [PubMed]
- Patra, P.; Mitra, S.; Debnath, N.; Goswami, A. Biochemical-, biophysical-, and microarray-based antifungal evaluation of the buffer-mediated synthesized nano zinc oxide: An in vivo and in vitro toxicity study. Langmuir 2012, 28, 16966–16978. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Liu, Y.; Mustapha, A.; Lin, M. Antifungal activity of zinc oxide nanoparticles against Botrytis cinerea and Penicillium expansum. Microbiol. Res. 2011, 166, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.; Zayton, M.A.; El-feky, S.A. Role of green synthesized ZnO nanoparticles as antifungal against post-harvest gray and black mold of sweet bell. J. Biotechnol. Bioeng. 2019, 3, 8–15. [Google Scholar]
- Dimkpa, C.O.; McLean, J.E.; Britt, D.W.; Anderson, A.J. Antifungal activity of ZnO nanoparticles and their interactive effect with a biocontrol bacterium on growth antagonism of the plant pathogen Fusarium graminearum. BioMetals 2013, 26, 913–924. [Google Scholar] [CrossRef]
- El-argawy, E.; Rahhal, M.M.H.; Elshabrawy, E.M.; Eltahan, R.M. Efficacy of some nanoparticles to control damping-off and root rot of sugar beet in El-Behiera Governorate. Asian J. Plant Pathol. 2016, 11, 35–47. [Google Scholar] [CrossRef]
- Sardella, D.; Gatt, R.; Valdramidis, V.P. Assessing the efficacy of zinc oxide nanoparticles against Penicillium expansum by automated turbidimetric analysis. Mycology 2018, 9, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Shoeb, M.; Singh, B.R.; Khan, J.A.; Khan, W.; Singh, B.N.; Singh, H.B.; Naqvi, A.H. ROS-dependent anticandidal activity of zinc oxide nanoparticles synthesized by using egg albumen as a biotemplate. Adv. Nat. Sci. Nanosci. Nanotechnol. 2013, 4. [Google Scholar] [CrossRef]
- Horky, P.; Skalickova, S.; Baholet, D.; Skladanka, J. Nanoparticles as a solution for eliminating the risk of mycotoxins. Nanomaterials 2018, 8, 727. [Google Scholar] [CrossRef]
- Gacem, M.A.; Gacem, H.; Telli, A.; Ould El Hadj Khelil, A. Mycotoxins: Decontamination and nanocontrol methods. In Nanomycotoxicology: Treating Mycotoxins in the Nano Way; Rai, M., Abd-Elsalam, K.A., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2020; pp. 189–216. ISBN 9780128179987. [Google Scholar]
- Tsang, C.C.; Tang, J.Y.M.; Lau, S.K.P.; Woo, P.C.Y. Taxonomy and evolution of Aspergillus, Penicillium and Talaromyces in the omics era—Past, present and future. Comput. Struct. Biotechnol. J. 2018, 16, 197–210. [Google Scholar] [CrossRef] [PubMed]
- El-banna, A.A.; Pitt, J.I.; Leistner, L. Production of mycotoxins by Penicillium species. Syst. Appl. Microbiol. 1987, 10, 42–46. [Google Scholar] [CrossRef]
- Frisvad, J.C. A critical review of producers of small lactone mycotoxins: Patulin, penicillic acid and moniliformin. World Mycotoxin J. 2018, 11, 73–100. [Google Scholar] [CrossRef]
- Jimenez-Garcia, S.N.; Garcia-Mier, L.; Garcia-Trejo, J.F.; Ramirez-Gomez, X.S.; Guevara-Gonzalez, R.G.; Feregrino-Perez, A.A. Fusarium mycotoxins and metabolites that modulate their production. In Fusarium—Plant Diseases, Pathogen Diversity, Genetic Diversity, Resistance and Molecular Markers; InTech: London, UK, 2018. [Google Scholar] [CrossRef]
- Hulvová, H.; Galuszka, P.; Frébortová, J.; Frébort, I. Parasitic fungus Claviceps as a source for biotechnological production of ergot alkaloids. Biotechnol. Adv. 2013, 31, 79–89. [Google Scholar] [CrossRef]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef]
- Schardl, C.L. Introduction to the toxins special issue on ergot alkaloids. Toxins 2015, 7, 4232–4237. [Google Scholar] [CrossRef]
- Ostry, V. Alternaria mycotoxins: An overview of chemical characterization, producers, toxicity, analysis and occurrence in foodstuffs. World Mycotoxin J. 2008, 1, 175–188. [Google Scholar] [CrossRef]
- Jesmin, R.; Chanda, A. Restricting mycotoxins without killing the producers: A new paradigm in nano-fungal interactions. Appl. Microbiol. Biotechnol. 2020, 104, 2803–2813. [Google Scholar] [CrossRef] [PubMed]
- Kaur, A.; Saini, S.S. Nanoadsorbents for the preconcentration of some toxic substances: A minireview. Int. Lett. Chem. Phys. Astron. 2013, 21, 22–35. [Google Scholar] [CrossRef][Green Version]
- Zahoor, M.; Ali Khan, F. Adsorption of aflatoxin B1 on magnetic carbon nanocomposites prepared from bagasse. Arab. J. Chem. 2018, 11, 729–738. [Google Scholar] [CrossRef]
- Moghaddam, S.H.M.; Jebali, A.; Daliri, K. The use of MgO-SiO2 nanocomposite for adsorption of aflatoxin in wheat flour samples. In Proceedings of the NanoCon 2010, Olomouc, Czech Republic, 12–14 October 2010; pp. 10–15. [Google Scholar]
- Daković, A.; Tomašević-Čanović, M.; Dondur, V.; Rottinghaus, G.E.; Medaković, V.; Zarić, S. Adsorption of mycotoxins by organozeolites. Colloids Surf. B Biointerfaces 2005, 46, 20–25. [Google Scholar] [CrossRef]
- Kovač, T.; Borišev, I.; Crevar, B.; Čačić Kenjerić, F.; Kovač, M.; Strelec, I.; Ezekiel, C.N.; Sulyok, M.; Krska, R.; Šarkanj, B. Fullerol C60(OH)24 nanoparticles modulate aflatoxin B1 biosynthesis in Aspergillus flavus. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef]
- Asghar, M.A.; Zahir, E.; Shahid, S.M.; Khan, M.N.; Asghar, M.A.; Iqbal, J.; Walker, G. Iron, copper and silver nanoparticles: Green synthesis using green and black tea leaves extracts and evaluation of antibacterial, antifungal and aflatoxin B1 adsorption activity. LWT Food Sci. Technol. 2018, 90, 98–107. [Google Scholar] [CrossRef]
- Hernández-Meléndez, D.; Salas-Téllez, E.; Zavala-Franco, A.; Téllez, G.; Méndez-Albores, A.; Vázquez-Durán, A. Inhibitory effect of flower-shaped zinc oxide nanostructures on the growth and aflatoxin production of a highly toxigenic strain of Aspergillus flavus Link. Materials 2018, 11, 1265. [Google Scholar] [CrossRef]
- Mitra, C.; Gummadidala, P.M.; Merrifield, R.; Omebeyinje, M.H.; Jesmin, R.; Lead, J.R.; Chanda, A. Size and coating of engineered silver nanoparticles determine their ability to growth-independently inhibit aflatoxin biosynthesis in Aspergillus parasiticus. Appl. Microbiol. Biotechnol. 2019, 103, 4623–4632. [Google Scholar] [CrossRef]
- Mitra, C.; Gummadidala, P.M.; Afshinnia, K.; Merrifield, R.C.; Baalousha, M.; Lead, J.R.; Chanda, A. Citrate-Coated Silver Nanoparticles Growth-Independently Inhibit Aflatoxin Synthesis in Aspergillus parasiticus. Environ. Sci. Technol. 2017, 51, 8085–8093. [Google Scholar] [CrossRef]
- Lakshmeesha, T.R.; Kalagatur, N.K.; Mudili, V.; Mohan, C.D.; Rangappa, S.; Prasad, B.D.; Ashwini, B.S.; Hashem, A.; Alqarawi, A.A.; Malik, J.A.; et al. Biofabrication of zinc oxide nanoparticles with Syzygium aromaticum flower buds extract and finding its novel application in controlling the growth and mycotoxins of Fusarium graminearum. Front. Microbiol. 2019, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mohd Yusof, H.; Mohamad, R.; Zaidan, U.H.; Abdul Rahman, N.A. Microbial synthesis of zinc oxide nanoparticles and their potential application as an antimicrobial agent and a feed supplement in animal industry: A review. J. Anim. Sci. Biotechnol. 2019, 10, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Jabar, A.K.; Aldhahi, H.H.K.; Salim, H.A. Effect of manufactured iron oxides in control of tomato yellow leaf curl virus (TYLCV). Plant Arch. 2020, 20, 2131–2134. [Google Scholar]
- Savi, G.D.; Bortoluzzi, A.J.; Scussel, V.M. Antifungal properties of Zinc-compounds against toxigenic fungi and mycotoxin. Int. J. Food Sci. Technol. 2013, 48, 1834–1840. [Google Scholar] [CrossRef]
- Anderson, A.J.; McLean, J.E.; Jacobson, A.R.; Britt, D.W. CuO and ZnO nanoparticles modify interkingdom cell signaling processes relevant to crop production. J. Agric. Food Chem. 2018, 66, 6513–6524. [Google Scholar] [CrossRef]
- Liang, Y.; Duan, Y.; Fan, C.; Dong, H.; Yang, J.; Tang, J.; Tang, G.; Wang, W.; Jiang, N.; Cao, Y. Preparation of kasugamycin conjugation based on ZnO quantum dots for improving its effective utilization. Chem. Eng. J. 2019, 361, 671–679. [Google Scholar] [CrossRef]
- Young, M.; Ozcan, A.; Myers, M.E.; Johnson, E.G.; Graham, J.H.; Santra, S. Multimodal generally recognized as safe ZnO/Nanocopper composite: A novel antimicrobial material for the management of citrus phytopathogens. J. Agric. Food Chem. 2018, 66, 6604–6608. [Google Scholar] [CrossRef]
- Lahuf, A.A.; Kareem, A.A.; Al-Sweedi, T.M.; Alfarttoosi, H.A. Evaluation the potential of indigenous biocontrol agent Trichoderma harzianum and its interactive effect with nanosized ZnO particles against the sunflower damping-off pathogen, Rhizoctonia solani. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2019; Volume 365. [Google Scholar] [CrossRef]
- Li, Z.; Yu, T.; Paul, R.; Fan, J.; Yang, Y.; Wei, Q. Agricultural nanodiagnostics for plant diseases: Recent advances and challenges. Nanoscale Adv. 2020. [Google Scholar] [CrossRef]
- Khiyami, M.A.; Almoammar, H.; Awad, Y.M.; Alghuthaymi, M.A. Plant pathogen nanodiagnostic techniques: Forthcoming changes? Biotechnol. Biotechnol. Equip. 2014, 28, 775–785. [Google Scholar] [CrossRef]
- Giraldo, J.P.; Wu, H.; Newkirk, G.M.; Kruss, S. Nanobiotechnology approaches for engineering smart plant sensors. Nat. Nanotechnol. 2019, 14, 541–553. [Google Scholar] [CrossRef]
- Kumar, V.; Arora, K. Trends in nano-inspired biosensors for plants. Mater. Sci. Energy Technol. 2020, 3, 255–273. [Google Scholar] [CrossRef]
- Ansari, A.A.; Kaushik, A.; Solanki, P.R.; Malhotra, B.D. Nanostructured zinc oxide platform for mycotoxin detection. Bioelectrochemistry 2010, 77, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Martynenko, I.V.; Litvin, A.P.; Purcell-Milton, F.; Baranov, A.V.; Fedorov, A.V.; Gun’Ko, Y.K. Application of semiconductor quantum dots in bioimaging and biosensing. J. Mater. Chem. B 2017, 5, 6701–6727. [Google Scholar] [CrossRef]
- Willard, D.M.; Carillo, L.L.; Jung, J.; van Orden, A. CdSe-ZnS Quantum Dots as Resonance Energy Transfer Donors in a Model Protein-Protein Binding Assay. Nano Lett. 2001, 1, 469–474. [Google Scholar] [CrossRef]
- Chen, F.; Gerion, D. Fluorescent CdSe/ZnS nanocrystal-peptide conjugates for long-term, nontoxic imaging and nuclear targeting in living cells. Nano Lett. 2004, 4, 1827–1832. [Google Scholar] [CrossRef]
- Hong, S.; Lee, C. The current status and future outlook of quantum dot-based biosensors for plant virus detection. Plant Pathol. J. 2018, 34, 85–92. [Google Scholar] [CrossRef]
- Medintz, I.L.; Sapsford, K.E.; Konnert, J.H.; Chatterji, A.; Lin, T.; Johnson, J.E.; Mattoussi, H. Decoration of discretely immobilized cowpea mosaic virus with luminescent quantum dots. Langmuir 2005, 21, 5501–5510. [Google Scholar] [CrossRef]
- Moulick, A.; Milosavljevic, V.; Vlachova, J.; Podgajny, R.; Hynek, D.; Kopel, P.; Adam, V. Using CdTe/ZnSe core/shell quantum dots to detect DNA and damage to DNA. Int. J. Nanomed. 2017, 12, 1277–1291. [Google Scholar] [CrossRef]
- Rispail, N.; De Matteis, L.; Santos, R.; Miguel, A.S.; Custardoy, L.; Testillano, P.S.; Risueño, M.C.; Pérez-De-Luque, A.; Maycock, C.; Fevereiro, P.; et al. Quantum dot and superparamagnetic nanoparticle interaction with pathogenic fungi: Internalization and toxicity profile. ACS Appl. Mater. Interfaces 2014, 6, 9100–9110. [Google Scholar] [CrossRef]
- Tereshchenko, A.; Fedorenko, V.; Smyntyna, V.; Konup, I.; Konup, A.; Eriksson, M.; Yakimova, R.; Ramanavicius, A.; Balme, S.; Bechelany, M. ZnO films formed by atomic layer deposition as an optical biosensor platform for the detection of Grapevine virus A-type proteins. Biosens. Bioelectron. 2017, 92, 763–769. [Google Scholar] [CrossRef]
- Al-Fandi, M.G.; Alshraiedeh, N.H.; Oweis, R.J.; Hayajneh, R.H.; Alhamdan, I.R.; Alabed, R.A.; Al-Rawi, O.F. Direct electrochemical bacterial sensor using ZnO nanorods disposable electrode. Sens. Rev. 2018, 38, 326–334. [Google Scholar] [CrossRef]
- Tahir, M.A.; Hameed, S.; Munawar, A.; Amin, I.; Mansoor, S.; Khan, W.S.; Bajwa, S.Z. Investigating the potential of multiwalled carbon nanotubes based zinc nanocomposite as a recognition interface towards plant pathogen detection. J. Virol. Methods 2017, 249, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Siddiquee, S.; Rovina, K.; Yusof, N.A.; Rodrigues, K.F.; Suryani, S. Nanoparticle-enhanced electrochemical biosensor with DNA immobilization and hybridization of Trichoderma harzianum gene. Sens. Bio-Sens. Res. 2014, 2, 16–22. [Google Scholar] [CrossRef]
- Kalia, A.; Kaur, H. Agri-Applications of Nano-Scale Micronutrients: Prospects for Plant Growth Promotion; Raliya, R., Ed.; CRC Press: Boca Raton, FL, USA, 2019; ISBN 9781315123950. [Google Scholar]
- Bala, R.; Kalia, A.; Dhaliwal, S.S. Evaluation of Efficacy of ZnO Nanoparticles as Remedial Zinc Nanofertilizer for Rice. J. Soil Sci. Plant Nutr. 2019, 19, 379–389. [Google Scholar] [CrossRef]
- Kalia, A.; Sharma, S.P.; Kaur, H.; Kaur, H. Novel nanocomposite-based controlled-release fertilizer and pesticide formulations: Prospects and challenges. In Multifunctional Hybrid Nanomaterials for Sustainable Agri-food and Ecosystem; Abd-Elsalam, K.A., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2020; pp. 99–134. [Google Scholar]
- Kalia, A.; Sharma, S.P.; Kaur, H. Nanoscale Fertilizers: Harnessing Boons for Enhanced Nutrient Use Efficiency and Crop Productivity. In Nanotechnology in the Life Sciences; Abd-Elsalam, K.A., Prasad, R., Eds.; Springer International Publishing: Cham, Switzerland, 2019; ISBN 978-3-030-13295-8. [Google Scholar]
- Kairyte, K.; Kadys, A.; Luksiene, Z. Antibacterial and antifungal activity of photoactivated ZnO nanoparticles in suspension. J. Photochem. Photobiol. B Biol. 2013, 128, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Li, J.; Le, T. Zinc Oxide Nanoparticle as a Novel Class of Antifungal Agents: Current Advances and Future Perspectives. J. Agric. Food Chem. 2018, 66, 11209–11220. [Google Scholar] [CrossRef] [PubMed]
- Jameel, M.; Shoeb, M.; Khan, M.T.; Ullah, R.; Mobin, M.; Farooqi, M.K.; Adnan, S.M. Enhanced Insecticidal Activity of Thiamethoxam by Zinc Oxide Nanoparticles: A Novel Nanotechnology Approach for Pest Control. ACS Omega 2019. [Google Scholar] [CrossRef]
- Medina-Pérez, G.; Fernández-Luqueño, F.; Vazquez-Nuñez, E.; López-Valdez, F.; Prieto-Mendez, J.; Madariaga-Navarrete, A.; Miranda-Arámbula, M. Remediating Polluted Soils Using Nanotechnologies: Environmental Benefits and Risks. Polish J. Environ. Stud. 2019, 28, 1013–1030. [Google Scholar] [CrossRef]
- Das, S.; Chakraborty, J.; Chatterjee, S.; Kumar, H. Prospects of biosynthesized nanomaterials for the remediation of organic and inorganic environmental contaminants. Environ. Sci. Nano 2018, 5, 2784–2808. [Google Scholar] [CrossRef]
- Guerra, F.; Attia, M.; Whitehead, D.; Alexis, F. Nanotechnology for Environmental Remediation: Materials and Applications. Molecules 2018, 23, 1760. [Google Scholar] [CrossRef]
- Fu, L.; Wang, Z.; Dhankher, O.P.; Xing, B. Nanotechnology as a new sustainable approach for controlling crop diseases and increasing agricultural production. J. Exp. Bot. 2020, 71, 507–519. [Google Scholar] [PubMed]
- Kookana, R.S.; Boxall, A.B.A.; Reeves, P.T.; Ashauer, R.; Beulke, S.; Chaudhry, Q.; Cornelis, G.; Fernandes, T.F.; Gan, J.; Kah, M.; et al. Nanopesticides: Guiding principles for regulatory evaluation of environmental risks. J. Agric. Food Chem. 2014, 62, 4227–4240. [Google Scholar] [CrossRef] [PubMed]
- Walker, G.W.; Kookana, R.S.; Smith, N.E.; Kah, M.; Doolette, C.L.; Reeves, P.T.; Lovell, W.; Anderson, D.J.; Turney, T.W.; Navarro, D.A. Ecological risk assessment of nano-enabled pesticides: A perspective on problem formulation. J. Agric. Food Chem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kah, M.; Tufenkji, N.; White, J.C. Nano-enabled strategies to enhance crop nutrition and protection. Nat. Nanotechnol. 2019, 14, 532–540. [Google Scholar] [CrossRef]
- Beegam, A.; Prasad, P.; Jose, J.; Oliveira, M.; Costa, F.G.; Soares, A.M.V.M.; Gonçalves, P.P.; Trindade, T.; Kalarikkal, N.; Thomas, S.; et al. Environmental Fate of Zinc Oxide Nanoparticles: Risks and Benefits. In Toxicology-New Aspects to This Scientific Conundrum; Soloneski, S., Larramendy, M.L., Eds.; IntechOpen: London, UK, 2016. [Google Scholar] [CrossRef]
- Charitidis, C.A.; Georgiou, P.; Koklioti, M.A.; Trompeta, A.F.; Markakis, V. Manufacturing nanomaterials: From research to industry. Manuf. Rev. 2014, 1, 11. [Google Scholar] [CrossRef]
- Kalia, A.; Singh, S. Myco-decontamination of azo dyes: Nano-augmentation technologies. 3 Biotech 2020, 10, 384. [Google Scholar] [CrossRef]
- Kaur, P.; Taggar, M.S.; Kalia, A. Characterization of magnetic nanoparticle–immobilized cellulases for enzymatic saccharification of rice straw. Biomass Convers. Biorefinery 2020. [Google Scholar] [CrossRef]
- Huang, Y.; Ding, L.; Li, C.; Wu, M.; Wang, M.; Yao, C.; Yin, X.; Zhang, J.; Liu, J.; Zhang, Y.; et al. Safety Issue of Changed Nanotoxicity of Zinc Oxide Nanoparticles in the Multicomponent System. Part. Part. Syst. Charact. 2019, 36, 1–14. [Google Scholar] [CrossRef]
- Nel, A.; Grainger, D.; Alvarez, P.J.; Badesha, S.; Castranova, V.; Ferrari, M.; Godwin, H.; Grodzinski, P.; Morris, J.; Savage, N.; et al. Nanotechnology Environmental, Health, and Safety Issues. In Nanotechnology Research Directions for Societal Needs in 2020; Roco, M.C., Hersam, M.C., Mirkin, C.A., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 159–220. ISBN 9789400711686. [Google Scholar]
- Ali, A.; Phull, A.R.; Zia, M. Elemental zinc to zinc nanoparticles: Is ZnO NPs crucial for life? Synthesis, toxicological, and environmental concerns. Nanotechnol. Rev. 2018, 7, 413–441. [Google Scholar] [CrossRef]
- Paul, S.K.; Dutta, H.; Sarkar, S.; Sethi, L.N.; Ghosh, S.K. Nanosized Zinc Oxide: Super-functionalities, present scenario of application, safety issues, and future prospects in food processing and allied industries. Food Rev. Int. 2019, 35, 505–535. [Google Scholar] [CrossRef]
- Naveed Ul Haq, A.; Nadhman, A.; Ullah, I.; Mustafa, G.; Yasinzai, M.; Khan, I. Synthesis Approaches of Zinc Oxide Nanoparticles: The Dilemma of Ecotoxicity. J. Nanomater. 2017, 2017, 1–14. [Google Scholar] [CrossRef]
- Haque, J.; Bellah, M.; Hassan, R.; Rahman, S. Synthesis of ZnO nanoparticles by two different methods and comparison of their structural, antibacterial, photocatalytic and optical properties. Nano Express 2020, 1, 010007. [Google Scholar] [CrossRef]
- Marsalek, R. Particle size and Zeta Potential of ZnO. Procedia-Soc. Behav. Sci. 2014, 9, 13–17. [Google Scholar] [CrossRef]
- Chai, M.H.H.; Amir, N.; Yahya, N.; Saaid, I.M. Characterization and Colloidal Stability of Surface Modified Zinc Oxide Nanoparticle Characterization and Colloidal Stability of Surface Modified Zinc Oxide Nanoparticle. IOP Conf. Ser. J. Phys. Conf. Ser. 2018, 1123. [Google Scholar] [CrossRef]
- Zhulina, E.B.; Borisov, O.V.; Priamitsyn, V.A. Theory of steric stabilization of colloid dispersions by grafted polymers. J. Colloid Interface Sci. 1990, 137, 495–511. [Google Scholar] [CrossRef]
- Fiedot, M.; Rac, O.; Suchorska-Woźniak, P.; Karbownik, I.; Teterycz, H. Polymer-surfactant interactions and their influence on zinc oxide nanoparticles morphology. In Manufacturing Nanostructures; Ahmad, W., Ali, N., Eds.; One Central Press: Manchester, UK, 2014; pp. 108–128. [Google Scholar]
- Meibner, T.; Oelschlagel, K.; Potthoff, A. Implications of the stability behavior of zinc oxide nanoparticles for toxicological studies. Int. Nano Lett. 2014, 4, 116. [Google Scholar] [CrossRef]
- Hsiao, I.; Huang, Y. Effects of various physicochemical characteristics on the toxicities of ZnO and TiO2 nanoparticles toward human lung epithelial cells. Sci. Total Environ. 2011, 409, 1219–1228. [Google Scholar] [CrossRef]

| Type of Zn-Nanomaterial Used | Zn-Nanomaterial Characterization | Working Concentration | Study Conditions (Exposure Technique) | Zn-Nanomaterial Application Method | Pathogen Inoculation Technique | Pathogen Studied | Impact | References |
|---|---|---|---|---|---|---|---|---|
| Bacterial pathogens | ||||||||
| Zinkicide SG4, Zinkicide SG6 | 2-D nanoplate-like structure (dimensions: 0.2–0.5 mm, thickness: ~10.0 nm) nanoparticulate (size: 4–6 nm) | 2000 to 1.96 mg/mL | In vitro assay (broth microdilution technique) | Addition in broth at different working concentrations | Broth inoculation | X. alfalfae subsp. citrumelonis | Two-fold and 7/8-fold lower MIC for Zinkicide SG4 and SG6, respectively | [116] |
| ZnO NPs | Commercial formulation (size <100 nm) | 0.1 mg mL−1 | In planta assay | Foliar spray of ZnO NPs suspension (10 mL per lentil plant) under pot culture conditions | Nutrient broth culture (10 mL of 1.2 × 105 CFU mL−1) added around the seedling | Xanthomonas axonopodis pv. phaseoli | Reduction in disease severity on pathogen challenge | [118] |
| Zinkicide SG4, Zinkicide SG6 | 2-D nanoplate-like structure (dimensions: 0.2–0.5 mm, thickness: ~10.0 nm) nanoparticulate (size: 4–6 nm) | Zn (30% w/v) | In planta assay | -Foliar spray of Zn formulation (10 mL per grapefruit seedling) using air-brush in greenhouse assay -Foliar spray of Zn formulations (3.0 L per grapefruit tree) with a handgun sprayer | Broth culture (104 CFU mL−1) in PBS injection-infiltrated in midrib of leaf 3 each site at both surfaces | Xanthomonas citri subsp. citri | -Reduction in citrus canker disease -Effective disease control comparable or better than Cu2O/Cu2O-ZnO bactericides (no phytotoxicity) | [116] |
| ZnONPs | TEM: 41–51 nm | 4, 8, and 16 μg mL−1 | In vitro assay | Variable concentrations of ZnO NPs (10 μL each) dropped on 1-day old bacterial lawn culture | Lawn growth obtained by spread plating of (100 μL, 108 cfu mL−1) broth culture followed by incubation for 24 h | Xanthomonas oryzae pv. oryzae (strain GZ 0003) | Effective antimicrobial agent for bacterial leaf blight of rice | [81] |
| Cu-Zn hybrid NPs | TEM: 40–100 nm | 1000, 500, 200, and 100 µg mL−1 | In vitro assay | NP formulations added to broth at different concentrations | Broth culture (20 µL, 105 CFU mL−1) | Xanthomonas perforans (Cu-tolerant GEV485) | Complete inhibition of growth till 24 h of incubation | [117] |
| Cu-Zn hybrid NPs | TEM: 40–100 nm | 500, 200, 100, and 50 µg mL−1 | In planta assay | Foliar spray on 4-week old seedlings of tomato variety FL 47 under growth chamber conditions | Pathogen inoculation-foliar spray | Xanthomonas perforans (Cu-tolerant GEV485) | Statistically highest decrease in disease symptoms at 500 µg/mL | [117] |
| Fungal pathogens | ||||||||
| ZnO NPs | Commercial formulation (< 50 nm particles size) | 0, 1, 10, 100, 500, and 1000 μg/mL | In vitro assay (poison food technique) | Supplementation of PDA with different working concentrations | Mycelial plug (5 mm) cut from master culture PDA plate (4-day old growth from edge) | Alternaria alternata | -Mean inhibition rate (EC50) range 235 and 848 μg/mL -higher efficacy compared to ZnSO4 | [59] |
| ZnO NPs/CS-Zn-CuNPs | DLS: 1.5–20 nm TEM: 6–21 nm | 0, 30, 60, and 90 µg mL−1 | In vitro assay (poison food technique) | Addition various working concentrations of prepared nanomaterials in PDA media | Mycelial plug (5 mm) cut from edge of 1-week old fungal growth on PDA media | Alternaria alternata, B. cinerea, R. solani | -Highest mycelial inhibition by chitosan mixed Zn-Cu nanocomposite | [62] |
| 3D flower-shaped nanostructured ZnO | FE-SEM: 700–800 nm XRD: crystallite size—42.0 ± 0.8 nm | 0.3125–5.0 mM | In vitro assay (broth culture experiment) | Supplementation of broth with different concentrations of Zn nanomaterial | Aqueous conidial suspension (125 µL, 4 × 106 spores mL−1) added to Sabouraud dextrose broth (100 mL) | Aspergillus flavus Link (UNIGRAS-1231) | -For 1.25–5.0 mM concentrations -78.0% decrease in mycelial growth -99.7% decrease in aflatoxin synthesis | [167] |
| Metallic (Au/Ag) and ZnO NPs | Commercial formulation DLS: 7 and 477 nm, respectively | 50:10 μg/mL | In vitro assay (A. broth microtiter plate test, B. Kirby-Bauer disk diffusion technique) | A. NP suspension (20 μL in 75 μL SDB) B. NP impregnated on sterilized filter paper disks (6 mm diameter) | A. Spore suspension (5 μL, 1 × 105 spores/well) B. Spread plating of spore suspension | Aspergillus flavus (NRRL 3518)/A. fumigatus (ATCC 1022) | -combination of mix metallic NPs and ZnO-NPs effectively inhibited the fungal growth | [51] |
| ZNPs | DLS: 30–40 nm TEM: 15–20 nm (average particle size) | 50, 100, 250, and 500 ppm | In vitro assay (poison food technique) | Different ZnO NPs concentrations mixed in sterilized PDA media | Fungal spore suspension (3 µL, ~104 mL−1) spot plated in center of PDA media plate | Aspergillus niger | -dose-dependent decrease in radial growth diameter | [143] |
| ZnO NPs | Commercial formulation (TEM: 70 ± 15 nm) | 0, 3, 6, and 12 mM L−1 | In vitro assay (poison food technique) | ZnO NPs mixed in different concentrations in PDA media | Aqueous spore suspension (~104 mL−1) | Aspergillus niger (MTCC-10180), Fusarium oxysporum (NCIM-1043, NCIM-1072) | -Significant inhibition in hyphal growth at concentration of 3 mM L−1 | [144] |
| ZnO NPs | Leaf extract of derived NPs | 200, 300 and 400 µg mL−1 | In vitro assay (poison food technique) | Supplementation of PDA with different working concentrations of NPs | Fungal disc (5 mm diameter) from 5-day old culture growth | Alternaria alternata, Botrytis cinerea | -Concentration-dependent decrease in fungal growth | [145] |
| A. ZnO NPs, B. ZnO:MgO NPs C. ZnO:Mg(OH)2 composite | A. TEM: 22–37 nm B. TEM: 23–30 nm C. TEM: 23–49 nm | Serial dilution ranging from 5 to 0.002 mg mL−1 | In vitro assay (broth microdilution and agar-media based poison food technique) | DMSO dissolved NPs were diluted with PDB in a geometric progression | Aqueous spore suspension (1 × 106 conidia mL−1) added in PDB | Colletotrichum gloeosporioides | -ZnO NPs alone exhibited highest inhibition of the hyphal growth -Addition of MgO diminished the antifungal potential of ZnO NPs | [56] |
| ZnO NPs | TEM: 20 nm (spherical), 37 nm (acicular) | 3, 6, 9, and 12 mM L-1 | In vitro assay (poison food technique) | Supplementation of PDA with different working concentrations of NPs | Mycelial plug (1.5 cm diameter) from 16-day old fungal culture | Erythricium salmonicolor | -substantial mycelial growth inhibition at 6 mmol L−1 | [68] |
| ZnO NPs | Commercial formulation (size <100 nm) | 0, 100, 250, and 500 mg [Zn] L−1 | In vitro assay (poison food technique) | Different concentrations of ZnO NPs supplemented in mung bean agar media | Mycelial plugs (~0.5 × 1.0 cm) cut from the margins of the 5-day old fungal growth | Fusarium graminearum | -dose-dependent inhibition of fungal growth | [146] |
| ZnO NPs | TEM: 30–40 nm SEM: triangular- to hexagonal-shaped particles XRD: crystallite size—35.69 nm | 25, 50, 75, 100, 125, and 140 µg mL−1 | In vitro assay (broth culture experiment) | Different concentrations of ZnO NPs supplemented in Czapek Dox broth | Spore suspension (10 µL, 106 spores mL−1 in peptone water + 0.01% Tween 80) in Czapek Dox broth (100 mL) | Fusarium graminearum | In dose-dependent manner -ROS accumulation in treated mycelial -reduction in deoxynivalenol and zearalenone production | [170] |
| ZnO NPs | TEM: spherical-shaped 30 nm size NPs XRD: wurtzite crystal nature | 10, 25, 50, and 100 mM | In vitro assay (poison food technique) | -Variable concentrations added to PDA -Highest Zn-compounds concentration added to PDA | Mycelial disc (6 mm) obtained from 7-day-old fungal cultures from edge | Fusarium graminearum, Aspergillus flavus, Penicillium citrinum | -concentration-dependent decrease in hyphal growth -significant decrease in deoxynivalenol and aflatoxin B1 only by ZnO NPs compared to control | [173] |
| ZnO NPs | DLS: 111.53 ± 1.3 nm TEM: < 100 nm ζ-potential: −15.89 mV | 100–800 ppm | In vitro assay (poison food technique) | -Different concentrations of ZnO NPs added to Czapek Dox agar | Mycelial disc (5 mm diameter) was cut from 5-day old culture | Fusarium moniliforme | -Less hyphal growth inhibition due larger sized particles | [40] |
| ZnO NPs | Commercial formulation (size: 70 ± 15 nm) | 0, 2, 4, 6, 8, and 12 mg L−1 | In vitro assay (poison food technique) | Different concentrations of ZnO NPs with autoclaved PD agar medium | Fungal mycelia plug (1 cm diameter) taken from the edge of one-week old culture | Fusarium oxysporum | -19.3–77.5% hyphal growth inhibition corresponding to for 2–12 mg L−1 ZnO NP concentration | [66] |
| ZnO NPs | Commercial formulation (spherical-shaped 20–30 ± 10 nm NPs) | 25, 50, and 100 ppm | In vitro assay (poison food technique) | Working concentrations of ZnO NPs derived from 1000 ppm stock solution added to sterilized PDA medium | Fungal disc (0.5 cm diameter) obtained from 7-old culture | Fusarium oxysporum f. sp. betae | -49.3% inhibition of radial hyphal growth at 100 ppm | [147] |
| ZnO NPs | Commercial formulation (size: <50 nm) | 0–15 mM equivalent to 0–1221 ppm | In vitro assay (automated turbidimetric assay) | ZnO NPs suspension-soaked filter papers | Spore suspension (1.73 × 103 conidia mL−1) were serially diluted | Penicillium expansum | -MIC: 9.8 mM (798 ppm) and NIC: 1.8 mM (147 ppm) | [148] |
| A. Zn NPs B. ZnO NPs | A. TEM: mean diameter 264 nm; hydrodynamic diameter: 615.8 nm; ζ-potential: −1.6 ± 3.7 B. TEM: mean particle diameter 19.3 nm; hydrodynamic diameter: 453.3; ζ-potential: 23.3 ± 5.0 | 0–65 mg L−1 | In vitro spore germination and infectivity tests | Different concentrations of nano-Zn formulations incubated with fungal spore suspension | Spore suspension (106 spores mL−1) mixed with DI | Peronospora tabacina | -Inhibition of spore germination frequency spore by Zn NPs, ZnO NPs, and ZnCl2 (<10 mg L−1) -Significantly higher inhibition by ZnO NPs compared to bulk ZnO -Reduction in leaf infection in tobacco leaf assay | [141] |
| ZnO and CuO NPs | Commercial formulation | 50, 100, 250, and 500 mg L−1 | In vitro assay (poison food technique) | Different concentrations of NPs amended in autoclaved PDA media | Fungal growth plug (0.5 cm2) placed in center of PDA media | Pythium ultimum, Pythium aphanidermatum | -Inhibition of growth at low concentrations -morphological changes in the hyphae | [142] |
| Viral pathogens | ||||||||
| ZnO NPs | TEM: 18 nm spherical-shaped particles | A. 100 μg mL−1 B. 100 μg mL−1 (5 mL NP solution foliar spray for 3, 7, and 12 days) | A. In vitro assay B. In planta assay (Nicotiana benthamiana) | A. ZnO NP suspension mixed with purified TMV particles B. Foliar spray of NPs suspensions | A. Purified TMV particles mixed with NPs B. Inoculation by rubbing infected leaves onto the oldest leaf | Tobacco mosaic virus | A. aggregation or breakage of tobacco mosaic virus particles B. marked suppression (35.33%) of TMV invasion in the inoculated leaves | [45] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalia, A.; Abd-Elsalam, K.A.; Kuca, K. Zinc-Based Nanomaterials for Diagnosis and Management of Plant Diseases: Ecological Safety and Future Prospects. J. Fungi 2020, 6, 222. https://doi.org/10.3390/jof6040222
Kalia A, Abd-Elsalam KA, Kuca K. Zinc-Based Nanomaterials for Diagnosis and Management of Plant Diseases: Ecological Safety and Future Prospects. Journal of Fungi. 2020; 6(4):222. https://doi.org/10.3390/jof6040222
Chicago/Turabian StyleKalia, Anu, Kamel A. Abd-Elsalam, and Kamil Kuca. 2020. "Zinc-Based Nanomaterials for Diagnosis and Management of Plant Diseases: Ecological Safety and Future Prospects" Journal of Fungi 6, no. 4: 222. https://doi.org/10.3390/jof6040222
APA StyleKalia, A., Abd-Elsalam, K. A., & Kuca, K. (2020). Zinc-Based Nanomaterials for Diagnosis and Management of Plant Diseases: Ecological Safety and Future Prospects. Journal of Fungi, 6(4), 222. https://doi.org/10.3390/jof6040222







