The “Little Iron Waltz”: The Ternary Response of Paracoccidioides spp. to Iron Deprivation
Abstract
1. Introduction
2. “Time Signature”: The Transcriptional Reprogramming of Paracoccidioides spp. Facing Iron Deprivation
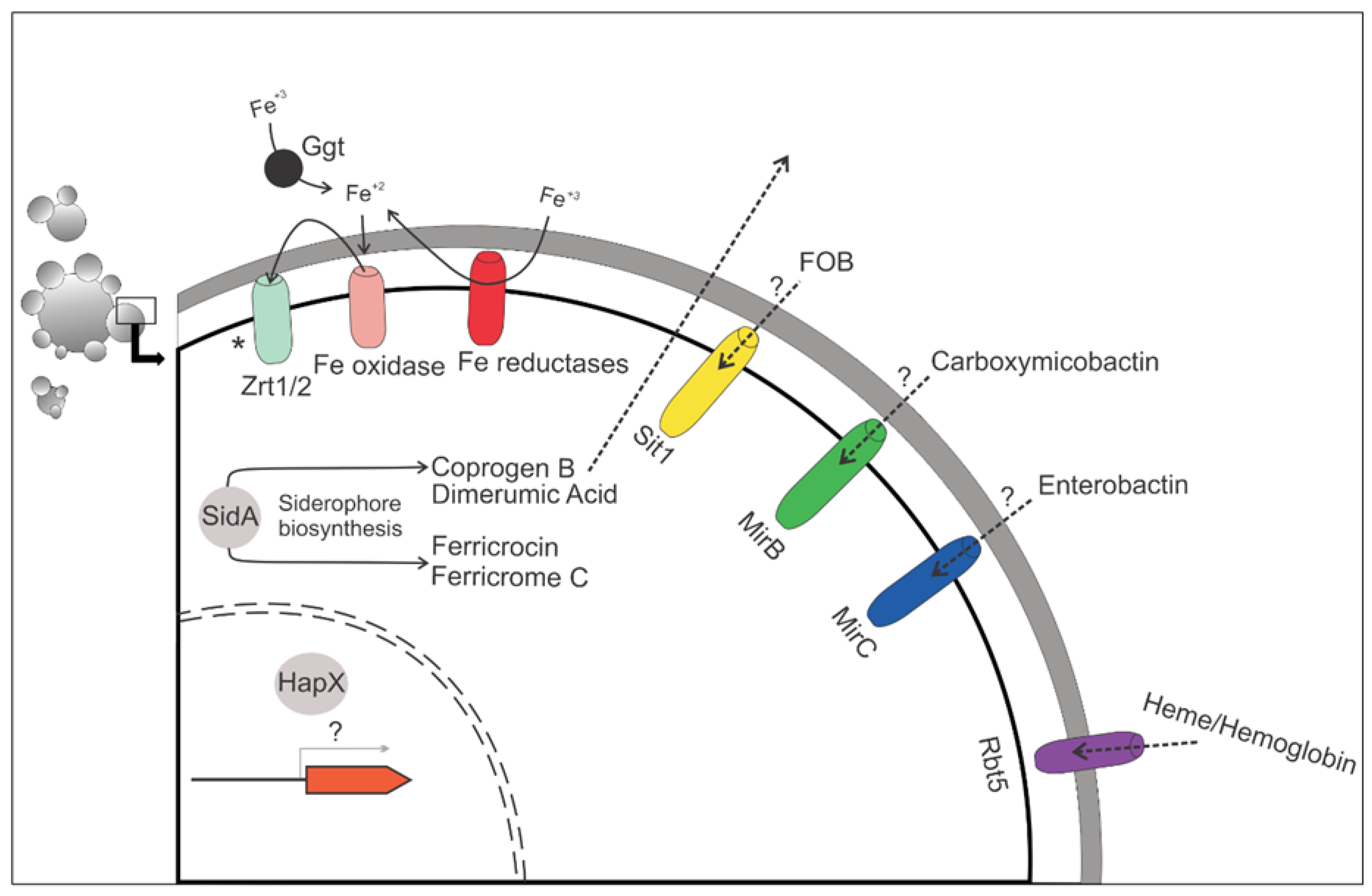
3. “The First Beat”: The Non-Classical Reductive Pathway of Iron Assimilation in Paracoccidioides spp.
4. “The Second Beat”: Exploring the Host’s Iron-Containing Proteins
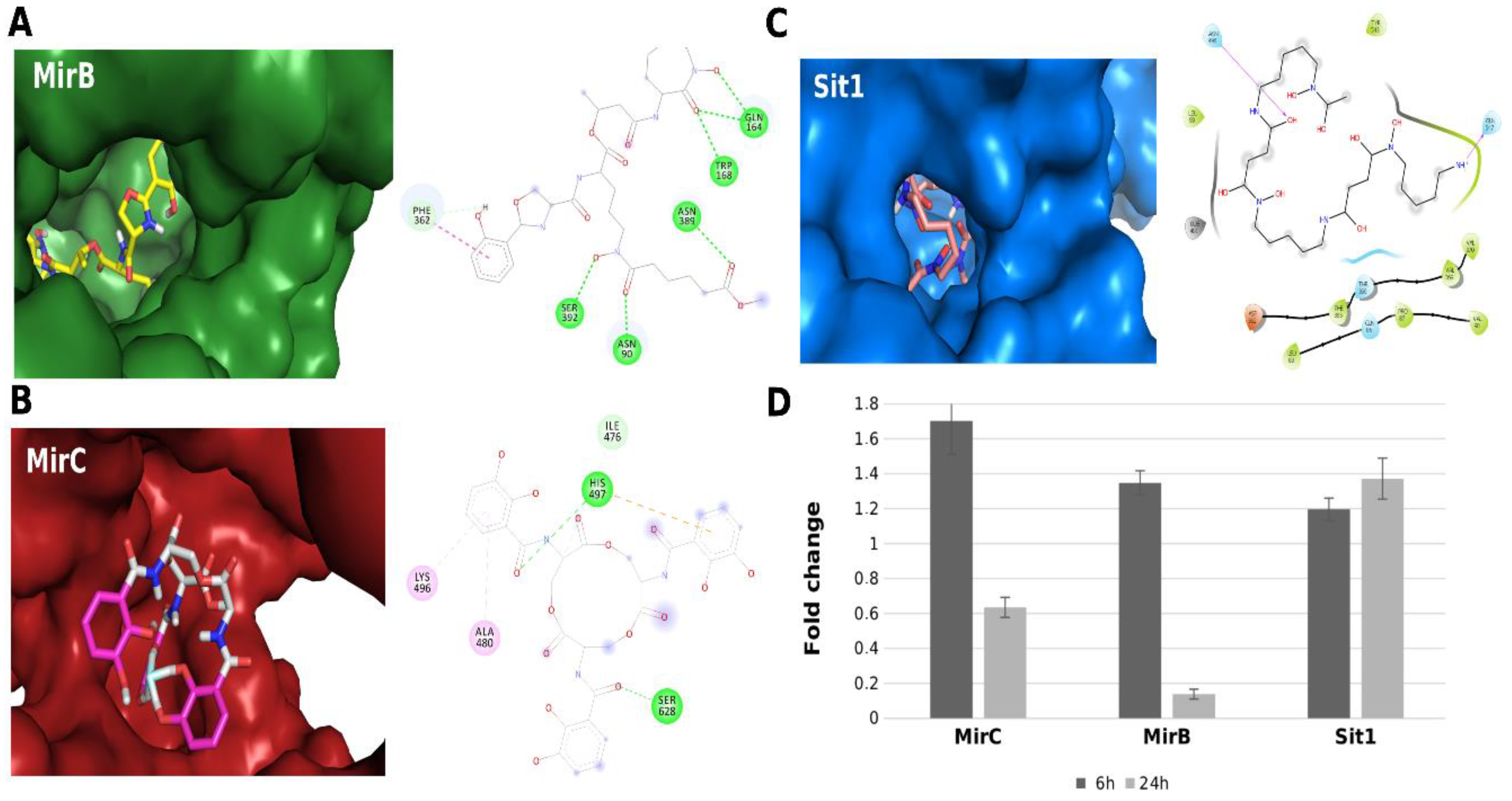
5. “The Third Beat”: Biosynthesis and Uptake of Siderophores by Paracoccidioides spp.
6. “The Addition of Chialteras”: Recent Findings and Future Perspectives on the Response of Paracoccidioides spp. to Fe Deprivation
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Abraham, S.N.; St. John, A.L. Mast cell-orchestrated immunity to pathogens. Nat. Rev. Immunol. 2010, 10, 440–452. [Google Scholar] [CrossRef] [PubMed]
- Audia, J.P.; Webb, C.C.; Foster, J.W. Breaking through the acid barrier: An orchestrated response to proton stress by enteric bacteria. Int. J. Med. Microbiol. 2001, 291, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Lemon, B.; Tjian, R. Orchestrated response: A symphony of transcription factors for gene control. Genes Dev. 2000, 14, 2551–2569. [Google Scholar] [CrossRef] [PubMed]
- Asehnoune, K.; Villadangos, J.; Hotchkiss, R.S. Understanding host–pathogen interaction. Intensive Care Med. 2016, 42, 2084–2086. [Google Scholar] [CrossRef]
- Casadevall, A.; Pirofski, L.A. Host-pathogen interactions: Basic concepts of microbial commensalism, colonization, infection, and disease. Infect. Immun. 2000, 68, 6511–6518. [Google Scholar] [CrossRef]
- Weinberg, E.D. Nutritional Immunity. J. Am. Mecical Assoc. 1975, 231, 39–41. [Google Scholar] [CrossRef]
- Ganz, T. Iron and infection. Int. J. Hematol. 2018, 107, 7–15. [Google Scholar] [CrossRef]
- Cassat, J.E.; Skaar, E.P. Iron in Infection and Immunity. Cell Host Microbe 2013, 13, 509–519. [Google Scholar] [CrossRef]
- Caza, M.; Kronstad, J.W. Shared and distinct mechanisms of iron acquisition by bacterial and fungal pathogens of humans. Front. Celullar Infect. Microbiol. 2013, 3, 1–23. [Google Scholar] [CrossRef]
- Dlouhy, A.C.; Outten, C.E. The Iron Methallome in Eukaryotic Organisms. Met. Ions Life Sci. 2013, 12, 241–278. [Google Scholar] [CrossRef]
- Aisen, P.; Enns, C.; Wessling-Resnick, M. Chemistry and biology of eukaryotic iron metabolism. Int. J. Biochem. Cell Biol. 2001, 33, 940–959. [Google Scholar] [CrossRef]
- Papanikolaou, G.; Pantopoulos, K. Iron metabolism and toxicity. Toxicol. Appl. Pharmacol. 2005, 202, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Tandara, L.; Salamunic, I. Iron metabolism: Current facts and future directions. Biochem. Medica 2012, 22, 311–328. [Google Scholar] [CrossRef] [PubMed]
- Malavia, D.; Crawford, A.; Wilson, D. Nutritional Immunity and Fungal Pathogenesis: The Struggle for Micronutrients at the Host–Pathogen Interface, 1st ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; Volume 70. [Google Scholar]
- Huynh, C.; Yuan, X.; Miguel, D.C.; Renberg, R.L.; Protchenko, O.; Philpott, C.C.; Hamza, I.; Andrews, N.W. Heme uptake by Leishmania amazonensis is mediated by the transmembrane protein LHR1. PLoS Pathog. 2012, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Bairwa, G.; Hee Jung, W.; Kronstad, J.W. Iron acquisition in fungal pathogens of humans. Metallomics 2017, 9, 215–227. [Google Scholar] [CrossRef]
- Haas, H. Fungal siderophore metabolism with a focus on Aspergillus fumigatus. Nat. Prod. Rep. 2014, 31, 1266–1276. [Google Scholar] [CrossRef]
- Shikanai-Yasuda, M.A.; Telles Filho, F.D.Q.; Mendes, R.P.; Colombo, A.L.; Moretti, M.L.; Kono, A.; Tresoldi, A.T.; Wanke, B.; Carvalho, C.R.; Benard, G.; et al. Consenso em paracoccidioidomicose. Rev. Soc. Bras. Med. Trop. 2006, 39, 297–310. [Google Scholar] [CrossRef]
- Brummer, E.; Castaneda, E.; Restrepo, A. Paracoccidioidomycosis: An update. Clin. Microbiol. Rev. 1993, 6, 89–117. [Google Scholar] [CrossRef]
- Shikanai-Yasuda, M.A.; Mendes, R.P.; Colombo, A.L.; Queiroz-telles, F.D.; Satie, A.; Kono, G.; Paniago, A.M.M.; Nathan, A.; Carlos, A.; Bagagli, E. Brazilian guidelines for the clinical management of paracoccidioidomycosis. Rev. Soc. Bras. Med. Trop. 2017. [Google Scholar] [CrossRef]
- Turissini, D.A.; Gomez, O.M.; Teixeira, M.M.; Mcewen, J.G.; Matute, D.R. Species boundaries in the human pathogen Paracoccidioides. Fungal Genet. Biol. 2017, 106, 9–25. [Google Scholar] [CrossRef]
- Restrepo, A.; Jiménez, B.E. Growth of Paracoccidioides brasiliensis yeast phase in a chemically defined culture medium. J. Clin. Microbiol. 1980, 12, 279–281. [Google Scholar] [CrossRef] [PubMed]
- Cano, L.E.; Gomez, B.; Brummer, E.; Restrepo, A.; Stevens, D.A. Inhibitory effect of deferoxamine or macrophage activation on transformation of Paracoccidioides brasiliensis conidia ingested by macrophages: Reversal by holotransferrin. Infect. Immun. 1994, 62, 1494–1496. [Google Scholar] [CrossRef] [PubMed]
- Dias-Melicio, L.A.; Calvi, S.A.; Peraçoli, M.T.S.; Soares, Â.M.V.D.C. Inhibitory effect of deferoxamine on Paracoccidioides brasiliensis survival in human monocytes: Reversal by holotransferrin not by apotransferrin. Rev. Inst. Med. Trop. Sao Paulo 2005, 47, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Parente, A.F.A.; Bailão, A.M.; Borges, C.L.; Parente, J.A.; Magalhães, A.D.; Ricart, C.A.O.; Soares, C.M.A. Proteomic analysis reveals that iron availability alters the metabolic status of the pathogenic fungus Paracoccidioides brasiliensis. PLoS ONE 2011, 6, e22810. [Google Scholar] [CrossRef] [PubMed]
- Bailão, E.F.L.C.; Lima, P.d.S.; Silva-Bailão, M.G.; Bailão, A.M.; Fernandes, G.d.R.; Kosman, D.J.; Soares, C.M.d.A. Paracoccidioides spp. ferrous and ferric iron assimilation pathways. Front. Microbiol. 2015, 6, 1–12. [Google Scholar] [CrossRef]
- Bailão, E.F.L.C.; Parente, J.A.; Pigosso, L.L.; de Castro, K.P.; Fonseca, F.L.; Silva-Bailão, M.G.; Báo, S.N.; Bailão, A.M.; Rodrigues, M.L.; Hernandez, O.; et al. Hemoglobin Uptake by Paracoccidioides spp. Is Receptor-Mediated. PLoS Negl. Trop. Dis. 2014, 8, e2856. [Google Scholar] [CrossRef]
- Kuznets, G.; Vigonsky, E.; Weissman, Z.; Lalli, D.; Gildor, T.; Kauffman, S.J.; Turano, P.; Becker, J.; Lewinson, O.; Kornitzer, D. A Relay Network of Extracellular Heme-Binding Proteins Drives C. albicans Iron Acquisition from Hemoglobin. PLoS Pathog. 2014, 10, e1004407. [Google Scholar] [CrossRef]
- Baek, Y.U.; Li, M.; Davis, D.A. Candida albicans ferric reductases are differentially regulated in response to distinct forms of iron limitation by the Rim101 and CBF transcription factors. Eukaryot. Cell 2008, 7, 1168–1179. [Google Scholar] [CrossRef]
- Liang, Y.; Gui, L.; Wei, D.S.; Zheng, W.; Xing, L.J.; Li, M.C. Candida albicans ferric reductase FRP1 is regulated by direct interaction with Rim101p transcription factor. FEMS Yeast Res. 2009, 9, 270–277. [Google Scholar] [CrossRef]
- Kronstad, J.W.; Hu, G.; Jung, W.H. An encapsulation of iron homeostasis and virulence in Cryptococcus neoformans. Trends Microbiol. 2013, 21, 457–465. [Google Scholar] [CrossRef]
- Lee, H.; Chang, Y.C.; Varma, A.; Kwon-Chung, K.J. Regulatory diversity of TUP1 in Cryptococcus neoformans. Eukaryot. Cell 2009, 8, 1901–1908. [Google Scholar] [CrossRef] [PubMed]
- Weissman, Z.; Kornitzer, D. A family of Candida cell surface haem-binding proteins involved in haemin and haemoglobin-iron utilization. Mol. Microbiol. 2004, 53, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Braun, B.R.; Head, W.S.; Wang, M.X.; Johnson, A.D. Identification and characterization of TUP1-regulated genes in Candida albicans. Genetics 2000, 156, 31–44. [Google Scholar] [PubMed]
- Schrettl, M.; Kim, H.S.; Eisendle, M.; Kragl, C.; Nierman, W.C.; Heinekamp, T.; Werner, E.R.; Jacobsen, I.; Illmer, P.; Yi, H.; et al. SreA-mediated iron regulation in Aspergillus fumigatus. Mol. Microbiol. 2008, 70, 27–43. [Google Scholar] [CrossRef] [PubMed]
- Hortschansky, P.; Eisendle, M.; Al-Abdallah, Q.; Schmidt, A.D.; Bergmann, S.; Thön, M.; Kniemeyer, O.; Abt, B.; Seeber, B.; Werner, E.R.; et al. Interaction of HapX with the CCAAT-binding complex—A novel mechanism of gene regulation by iron. EMBO J. 2007, 26, 3157–3168. [Google Scholar] [CrossRef]
- Schrettl, M.; Beckmann, N.; Varga, J.; Heinekamp, T.; Jacobsen, I.D.; Jöchl, C.; Moussa, T.A.; Wang, S.; Gsaller, F.; Blatzer, M.; et al. HapX-Mediated adaption to iron starvation is crucial for virulence of Aspergillus fumigatus. PLoS Pathog. 2010, 6, e1001124. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Ma, Z.; Zhong, G.; Sheppard, D.C.; Lu, L.; Zhang, S. The mitochondrial thiamine pyrophosphate transporter TptA promotes adaptation to low iron conditions and virulence in fungal pathogen Aspergillus fumigatus. Virulence 2019, 10, 234–247. [Google Scholar] [CrossRef]
- Lima, P.d.S.; Chung, D.; Bailão, A.M.; Cramer, R.A.; Soares, C.M.d.A. Characterization of the Paracoccidioides Hypoxia Response Reveals New Insights into Pathogenesis Mechanisms of This Important Human Pathogenic Fungus. PLoS Negl. Trop. Dis. 2015, 9, e0004282. [Google Scholar] [CrossRef]
- Almeida, R.S.; Wilson, D.; Hube, B. Candida albicans iron acquisition within the host. FEMS Yeast Res. 2009, 9, 1000–1012. [Google Scholar] [CrossRef]
- Han, K.; Do, E.; Jung, W.H. A human fungal pathogen Cryptococcus neoformans expresses three distinct iron permease homologs. J. Microbiol. Biotechnol. 2012, 22, 1644–1652. [Google Scholar] [CrossRef]
- Zarnowski, R.; Woods, J.P. Glutathione-dependent extracellular ferric reductase activities in dimorphic zoopathogenic fungi. Microbiology 2005, 151, 2233–2240. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zarnowski, R.; Cooper, K.G.; Brunold, L.S.; Calaycay, J.; Woods, J.P. Histoplasma capsulatum secreted γ-glutamyltransferase reduces iron by generating an efficient ferric reductant. Mol. Microbiol. 2008, 70, 352–368. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.G.; Schrank, A.; Bailão, E.F.L.C.; Bailão, A.M.; Borges, C.L.; Staats, C.C.; Parente, J.A.; Pereira, M.; Salem-Izacc, S.M.; Mendes-Giannini, M.J.S.; et al. The homeostasis of iron, copper, and zinc in Paracoccidioides brasiliensis, Cryptococcus neoformans var. Grubii, and Cryptococcus gattii: A comparative analysis. Front. Microbiol. 2011, 2, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Kosman, D.J.; Bailão, E.F.L.C.; Silva-Bailão, M.G.; Soares, C.M.d.A. 59Fe Uptake Assays in Paracoccidioides Species. Bio-Protocol 2016, 6, e1930. [Google Scholar] [CrossRef]
- Bailão, E.F.L.C.; Parente, A.F.A.; Parente, J.A.; Silva-Bailão, M.G.; de Castro, K.P.; Kmetzsch, L.; Staats, C.C.; Schrank, A.; Vainstein, M.H.; Borges, C.L.; et al. Metal Acquisition and Homeostasis in Fungi. Curr. Fungal Infect. Rep. 2012, 6, 257–266. [Google Scholar] [CrossRef]
- Núñez, G.; Sakamoto, K.; Soares, M.P. Innate Nutritional Immunity. J. Immunol. 2018, 201, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Almeida, R.S.; Brunke, S.; Albrecht, A.; Thewes, S.; Laue, M.; Edwards, J.E.; Filler, S.G.; Hube, B. The hyphal-associated adhesin and invasin Als3 of Candida albicans mediates iron acquisition from host ferritin. PLoS Pathog. 2008, 4, e1000217. [Google Scholar] [CrossRef]
- Okamoto-Shibayama, K.; Kikuchi, Y.; Kokubu, E.; Sato, Y.; Ishihara, K. Csa2, a member of the Rbt5 protein family, is involved in the utilization of iron from human hemoglobin during Candida albicans hyphal growth. FEMS Yeast Res. 2014, 14, 674–677. [Google Scholar] [CrossRef]
- Pinsky, M.; Roy, U.; Moshe, S.; Weissman, Z.; Kornitzer, D. Human Serum Albumin Facilitates Heme-Iron Utilization by Fungi. MBio 2020, 11, 1–14. [Google Scholar] [CrossRef]
- Nasser, L.; Weissman, Z.; Pinsky, M.; Amartely, H.; Dvir, H.; Kornitzer, D. Structural basis of haem-iron acquisition by fungal pathogens. Nat. Microbiol. 2016, 1, 16156. [Google Scholar] [CrossRef]
- Ding, C.; Vidanes, G.M.; Maguire, S.L.; Guida, A.; Synnott, J.M.; Andes, D.R.; Butler, G. Conserved and divergent roles of Bcr1 and CFEM proteins in Candida parapsilosis and Candida albicans. PLoS ONE 2011, 6, e28151. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, V.K.; Suneetha, K.J.; Kaur, R. A systematic analysis reveals an essential role for high-affinity iron uptake system, haemolysin and CFEM domain-containing protein in iron homoeostasis and virulence in Candida glabrata. Biochem. J. 2014, 463, 103–114. [Google Scholar] [CrossRef]
- Weissman, Z.; Shemer, R.; Conibear, E.; Kornitzer, D. An endocytic mechanism for haemoglobin-iron acquisition in Candida albicans. Mol. Microbiol. 2008, 69, 201–217. [Google Scholar] [CrossRef] [PubMed]
- Cadieux, B.; Lian, T.; Hu, G.; Wang, J.; Biondo, C.; Teti, G.; Liu, V.; Murphy, M.E.P.; Creagh, A.L.; Kronstad, J.W. The mannoprotein cig1 supports iron acquisition from heme and virulence in the pathogenic fungus Cryptococcus neoformans. J. Infect. Dis. 2013, 207, 1339–1347. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Caza, M.; Cadieux, B.; Chan, V.; Liu, V.; Kronstad, J. Cryptococcus neoformans requires the ESCRT protein Vps23 for iron acquisition from heme, for capsule formation, and for virulence. Infect. Immun. 2013, 81, 292–302. [Google Scholar] [CrossRef]
- Guanggan, H.; Caza, M.; Cadieux, B.; Bakkeren, E.; Do, E.; Jung, W.H.; Kronstad, J.W. The ESCRT machinery influences haem uptake and capsule elaboration in Cryptococcus neoformans. Mol. Microbiol. 2015, 96, 973–992. [Google Scholar] [CrossRef]
- Saha, M.; Sarkar, S.; Sarkar, B.; Sharma, B.K.; Bhattacharjee, S.; Tribedi, P. Microbial siderophores and their potential applications: A review. Environ. Sci. Pollut. Res. 2016, 23, 3984–3999. [Google Scholar] [CrossRef]
- Neilands, J.B. Siderophores. Arq. Biochem. Biophys. 1993, 302, 1–3. [Google Scholar] [CrossRef]
- Nevitt, T. War-Fe-re: Iron at the core of fungal virulence and host immunity. Biometals 2011, 24, 547–558. [Google Scholar] [CrossRef]
- Miethke, M.; Marahiel, M.A. Siderophore-Based Iron Acquisition and Pathogen Control. Microbiol. Mol. Biol. Rev. 2007, 71, 413–451. [Google Scholar] [CrossRef]
- Sah, S.; Singh, R. Siderophore: Structural And Functional Characterisation—A Comprehensive Review. Agriculture 2015, 61, 97–114. [Google Scholar] [CrossRef]
- Wells, R.M.; Jones, C.M.; Xi, Z.; Speer, A.; Danilchanka, O.; Doornbos, K.S.; Sun, P.; Wu, F.; Tian, C.; Niederweis, M. Discovery of a Siderophore Export System Essential for Virulence of Mycobacterium tuberculosis. PLoS Pathog. 2013, 9, e1003120. [Google Scholar] [CrossRef] [PubMed]
- Raymond, K.N.; Dertz, E.A.; Kim, S.S. Enterobactin: An archetype for microbial iron transport. Proc. Natl. Acad. Sci. USA 2003, 100, 3584–3588. [Google Scholar] [CrossRef]
- Khan, A.; Singh, P.; Srivastava, A. Synthesis, nature and utility of universal iron chelator—Siderophore: A review. Microbiol. Res. 2018, 212–213, 103–111. [Google Scholar] [CrossRef]
- Raymond-Bouchard, I.; Carroll, C.S.; Nesbitt, J.R.; Henry, K.A.; Pinto, L.J.; Moinzadeh, M.; Scott, J.K.; Moore, M.M. Structural requirements for the activity of the MirB ferrisiderophore transporter of Aspergillus fumigatus. Eukaryot. Cell 2012, 11, 1333–1344. [Google Scholar] [CrossRef] [PubMed]
- Silva-Bailão, M.G.; Bailão, E.F.L.C.; Lechner, B.E.; Gauthier, G.M.; Lindner, H.; Bailão, A.M.; Haas, H.; Soares, C.M.d.A. Hydroxamate production as a high affinity iron acquisition mechanism in Paracoccidioides Spp. PLoS ONE 2014, 9, e105805. [Google Scholar] [CrossRef]
- Silva, M.G.; de Curcio, J.S.; Silva-Bailão, M.G.; Lima, R.M.; Tomazett, M.V.; de Souza, A.F.; Cruz-Leite, V.R.M.; Sbaraini, N.; Bailão, A.M.; Rodrigues, F.; et al. Molecular characterization of siderophore biosynthesis in Paracoccidioides brasiliensis. IMA Fungus 2020, 11, 11. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, Y. I-TASSER server: New development for protein structure and function predictions. Nucleic Acids Res. 2015, 43, W174–W181. [Google Scholar] [CrossRef]
- Moynié, L.; Milenkovic, S.; Mislin, G.L.A.; Gasser, V.; Malloci, G.; Baco, E.; McCaughan, R.P.; Page, M.G.P.; Schalk, I.J.; Ceccarelli, M.; et al. The complex of ferric-enterobactin with its transporter from Pseudomonas aeruginosa suggests a two-site model. Nat. Commun. 2019, 10, 3673. [Google Scholar] [CrossRef]
- Podkowa, K.J.; Briere, L.A.K.; Heinrichs, D.E.; Shilton, B.H. Crystal and solution structure analysis of FhuD2 from Staphylococcus aureus in multiple Unliganded conformations and bound to ferrioxamine-B. Biochemistry 2014, 53, 2017–2031. [Google Scholar] [CrossRef]
- Agoro, R.; Mura, C. Iron supplementation therapy, a friend and foe of mycobacterial infections? Pharmaceuticals 2019, 12, 75. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Bookout, A.L.; Cummins, C.L.; Kramer, M.F.; Pesola, J.M.; Mangelsdorf, D.J. High-Throughput Real-Time Quantitative Reverse Transcription PCR. Curr. Protoc. Mol. Biol. 2006, 73, 15.8.1–15.8.28. [Google Scholar]
- Juan, N.R.; Daniela, S.M.; Tania, V.L.; Sergio, E.G.; Alejandrina, R.F.; Rito, Z.L. Paracoccidioidomicosis y TBC-MR en portador de VIH/VHC. Rev. Chil. Infectol. 2010, 27, 551–555. [Google Scholar] [CrossRef][Green Version]
- Torres-Pereira, C.; Giovanini, A.F.; Stramandinoli, R.T.; Amenabar, J.M.; Piazzetta, C.M. Oral paracoccidioidomycosis and pulmonary tuberculosis co-infection: Relevance of oral biopsy in establishing the diagnosis and therapeutic approach. Int. J. Infect. Dis. 2007, 13, 112–114. [Google Scholar] [CrossRef] [PubMed]
| Species a | Gene ID b | Product Description c | CFEM (E-Value) d | GPI Modification Site Prediction? e | SignalP f | SecretomeP g |
|---|---|---|---|---|---|---|
| P. lutzii | PAAG_04763 | Hypothetical protein | 2.0 × 10−8 | None | Yes | - |
| PAAG_11627 | Hypothetical protein | 2.8 × 10−13 | None | Yes | - | |
| PAAG_05158 | Rbt5 # | 3.3 × 10−15 | Yes | Yes | - | |
| PAAG_02225 | Csa1 # | 5.2 × 10−12 | None | Yes | - | |
| PAAG_00918 | Hypothetical protein | 5.5 × 10−11 | None | Yes | - | |
| P. brasiliensis | PADG_11659 | Hypothetical protein | 1.5 × 10−11 | None | - | - |
| PADG_05363 | Csa1 # | 1.5 × 10−11 | None | Yes | - | |
| PADG_02506 | Hypothetical protein | 1.7 × 10−8 | None | Yes | - | |
| PADG_03909 | Hypothetical protein | 2.1 × 10−8 | Yes | Yes | - | |
| PADG_06374 | Hypothetical protein | 3.6 × 10−13 | None | - | Yes | |
| PADG_05000 | Rbt5 # | 4.5 × 10−15 | Yes | Yes | - | |
| P. americana | PABG_12009 | Hypothetical protein | 1.6 × 10−11 | None | Yes | - |
| PABG_00115 | Hypothetical protein | 1.7 × 10−8 | None | Yes | - | |
| PABG_01323 | Hypothetical protein | 2.4 × 10−8 | Yes | Yes | - | |
| PABG_04599 | Rbt51 # | 4.5 × 10−15 | Yes | Yes | - |
| Species | Described Mechanism | Proteins Involved |
|---|---|---|
| Paracoccidioides spp. | Non-classical RIA a,b | Ferric reductases, ferroxidase, GGT, Zrt1/2 |
| Use of host Fe-proteins | Rbt5 (heme/hemoglobin uptake) | |
| Biosynthesis and uptake of siderophores | SidA, Sit1, MirB, MirC | |
| Candida albicans | RIA | Ferric reductases, ferroxidases, ferric permeases |
| Use of host Fe-proteins | Rbt5, Pga7, Csa2 (heme/hemoglobin uptake); Als3 (iron uptake from ferritin) | |
| Uptake of siderophores | Sit1 | |
| Cryptococcus neoformans | RIA | Ferric reductases, ferroxidases, ferric permeases |
| Use of host Fe-proteins | Cig1 (heme/hemoglobin uptake) | |
| Uptake of siderophores | Sit1 | |
| Aspergillus spp. | Biosynthesis and uptake of siderophores | SidA, MirA, MirB, MirC |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Souza, A.F.; de Paula, M.S.; Lima, R.M.; Silva, M.G.; de Curcio, J.S.; Pereira, M.; de Almeida Soares, C.M. The “Little Iron Waltz”: The Ternary Response of Paracoccidioides spp. to Iron Deprivation. J. Fungi 2020, 6, 221. https://doi.org/10.3390/jof6040221
de Souza AF, de Paula MS, Lima RM, Silva MG, de Curcio JS, Pereira M, de Almeida Soares CM. The “Little Iron Waltz”: The Ternary Response of Paracoccidioides spp. to Iron Deprivation. Journal of Fungi. 2020; 6(4):221. https://doi.org/10.3390/jof6040221
Chicago/Turabian Stylede Souza, Aparecido Ferreira, Marcella Silva de Paula, Raisa Melo Lima, Marielle Garcia Silva, Juliana Santana de Curcio, Maristela Pereira, and Célia Maria de Almeida Soares. 2020. "The “Little Iron Waltz”: The Ternary Response of Paracoccidioides spp. to Iron Deprivation" Journal of Fungi 6, no. 4: 221. https://doi.org/10.3390/jof6040221
APA Stylede Souza, A. F., de Paula, M. S., Lima, R. M., Silva, M. G., de Curcio, J. S., Pereira, M., & de Almeida Soares, C. M. (2020). The “Little Iron Waltz”: The Ternary Response of Paracoccidioides spp. to Iron Deprivation. Journal of Fungi, 6(4), 221. https://doi.org/10.3390/jof6040221





