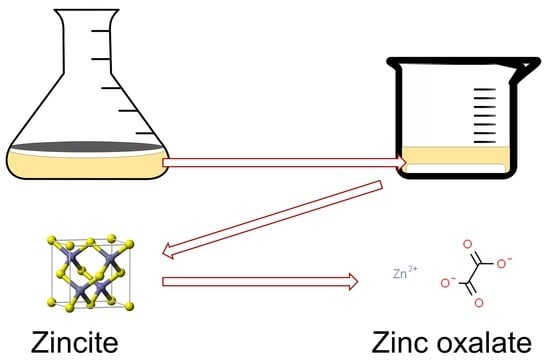
Fungus Aspergillus niger Processes Exogenous Zinc Nanoparticles into a Biogenic Oxalate Mineral
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of ZnO Nanoparticle Suspensions and ZnSO4 Solution
2.2. Cultivation of Aspergillus niger
2.3. Transformation of ZnO Nanoparticles by Extracellular Metabolites of Aspergillus niger
2.4. Characterization of ZnO Nanoparticles and Bulk ZnO
3. Results
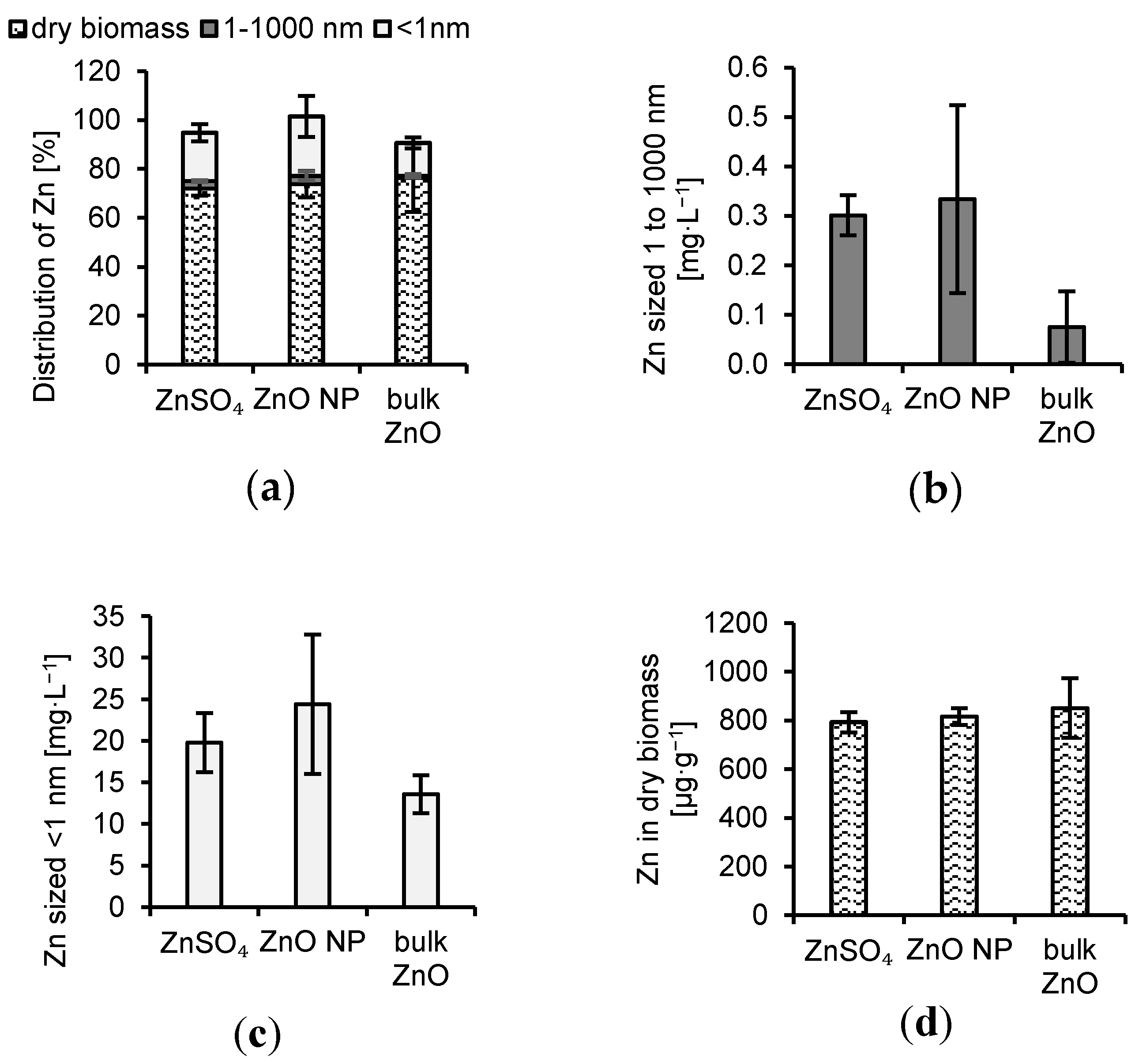
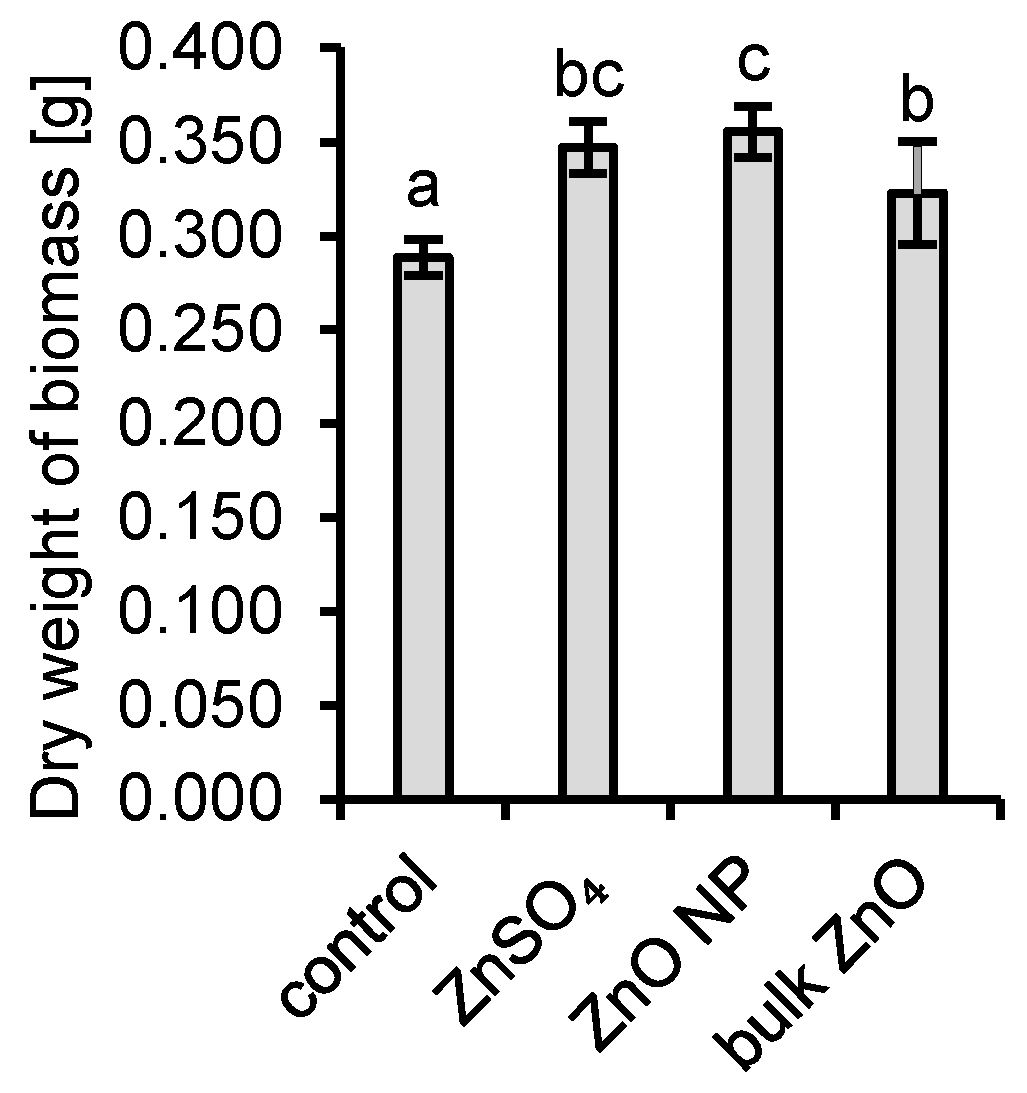
3.1. Interactions of Aspergillus niger with ZnO and Aqueous Zn
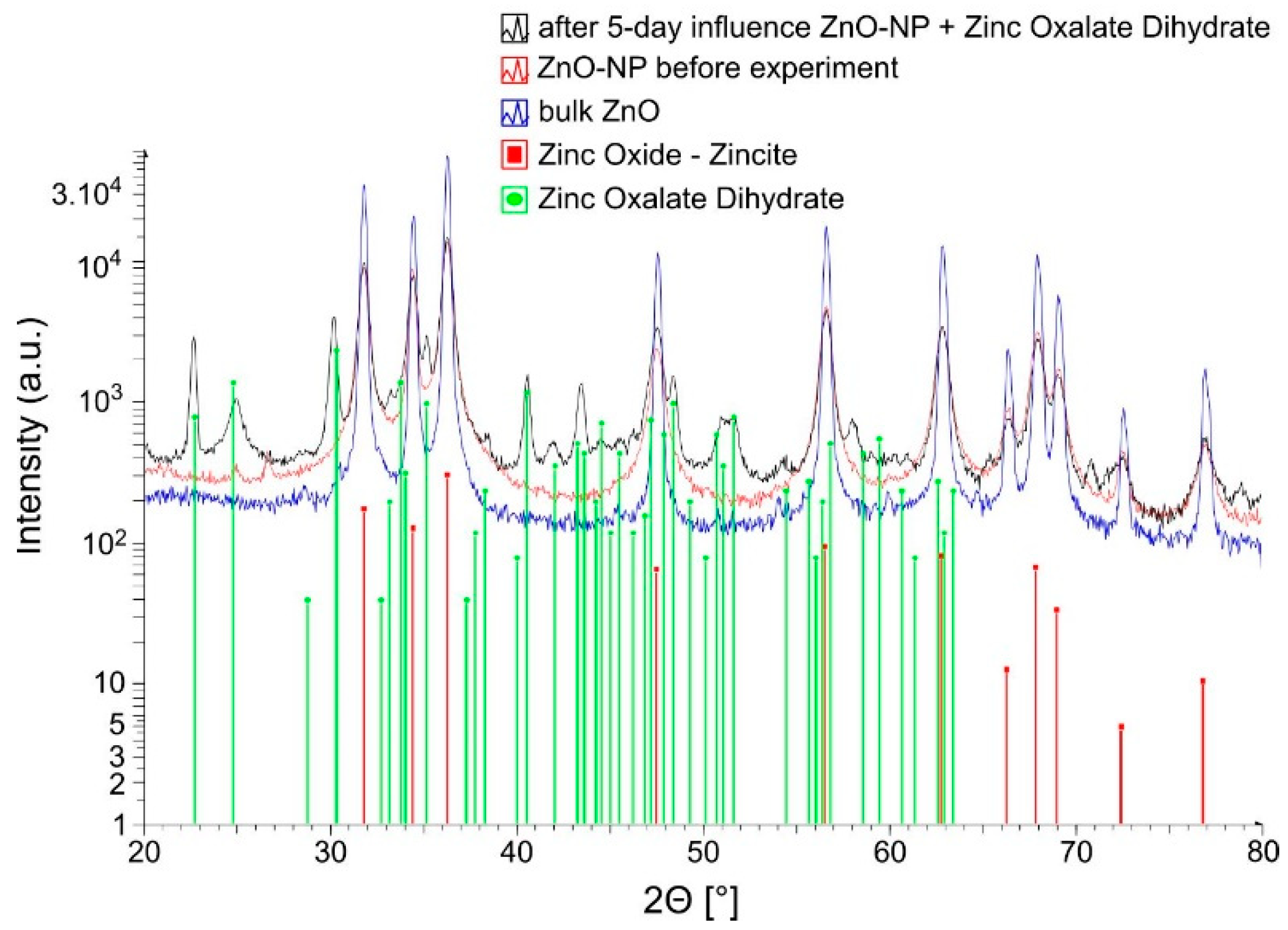
3.2. Transformation of ZnO Nanoparticles by Fungal Extracellular Metabolites
4. Discussion
4.1. Interactions of Aspergillus niger with ZnO and Aqueous Zn
4.2. Transformation of ZnO Nanoparticles by Fungal Extracellular Metabolites
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hochella, M.F.; Mogk, D.W.; Ranville, J.; Allen, I.C.; Luther, G.W.; Marr, L.C.; McGrail, B.P.; Murayama, M.; Qafoku, N.P.; Rosso, K.M.; et al. Natural, incidental, and engineered nanomaterials and their impacts on the Earth system. Science 2019, 363, eaau8299. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.Y.; Mitrano, D.M.; Bornhöft, N.A.; Scheringer, M.; Hungerbühler, K.; Nowack, B. Envisioning Nano Release Dynamics in a Changing World: Using Dynamic Probabilistic Modeling to Assess Future Environmental Emissions of Engineered Nanomaterials. Environ. Sci. Technol. 2017, 51, 2854–2863. [Google Scholar] [CrossRef] [PubMed]
- Peijnenburg, W.; Praetorius, A.; Scott-Fordsmand, J.; Cornelis, G. Fate assessment of engineered nanoparticles in solids dominated media—Current insights and the way forward. Environ. Pollut. 2016, 218, 1365–1369. [Google Scholar] [CrossRef] [PubMed]
- Šebesta, M.; Matúš, P. Separation, determination, and characterization of inorganic engineered nanoparticles in complex environmental samples. Chem. List. 2018, 112, 583–589. [Google Scholar]
- Keller, A.A.; McFerran, S.; Lazareva, A.; Suh, S. Global life cycle releases of engineered nanomaterials. J. Nanopart. Res. 2013, 15, 1–17. [Google Scholar] [CrossRef]
- MarketsandMarkets. Zinc Oxide Market by Application & by Region—Global Trends and Forecasts to 2020; MarketsandMarkets™ Research Private Ltd.: Hadapsar, India, 2015. [Google Scholar]
- Future Markets Inc. The Global Market for Zinc Oxide Nanoparticles; Future Markets Inc.: Edinburgh, UK, 2020. [Google Scholar]
- MarketsandMarkets. Zinc Oxide Market by Process (French Process, Wet Process, American Process), Grade (Standard, Treated, USP, FCC), Application (Rubber, Ceramics, Chemicals, Agriculture, Cosmetics & Personal Care, Pharmaceuticals), Region—Global Forecast to 2024; MarketsandMarkets™ Research Private Ltd.: Hadapsar, India, 2019. [Google Scholar]
- Šebesta, M.; Nemček, L.; Urík, M.; Kolenčík, M.; Bujdoš, M.; Vávra, I.; Dobročka, E.; Matúš, P. Partitioning and stability of ionic, nano- and microsized zinc in natural soil suspensions. Sci. Total Environ. 2020, 700, 134445. [Google Scholar] [CrossRef]
- Raliya, R.; Saharan, V.; Dimkpa, C.; Biswas, P. Nanofertilizer for Precision and Sustainable Agriculture: Current State and Future Perspectives. J. Agric. Food Chem. 2018, 66, 6487–6503. [Google Scholar] [CrossRef]
- Prasad, R.; Bhattacharyya, A.; Nguyen, Q.D. Nanotechnology in Sustainable Agriculture: Recent Developments, Challenges, and Perspectives. Front. Microbiol. 2017, 8, 1014. [Google Scholar] [CrossRef]
- Medina-Velo, I.A.; Barrios, A.C.; Zuverza-Mena, N.; Hernandez-Viezcas, J.A.; Chang, C.H.; Ji, Z.; Zink, J.I.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Comparison of the effects of commercial coated and uncoated ZnO nanomaterials and Zn compounds in kidney bean (Phaseolus vulgaris) plants. J. Hazard. Mater. 2017, 332, 214–222. [Google Scholar] [CrossRef]
- Raliya, R.; Tarafdar, J.C.; Biswas, P. Enhancing the Mobilization of Native Phosphorus in the Mung Bean Rhizosphere Using ZnO Nanoparticles Synthesized by Soil Fungi. J. Agric. Food Chem. 2016, 64, 3111–3118. [Google Scholar] [CrossRef]
- Liu, R.; Lal, R. Potentials of engineered nanoparticles as fertilizers for increasing agronomic productions. Sci. Total Environ. 2015, 514, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Kabata-Pendias, A.; Szteke, B. Trace Elements in Abiotic and Biotic Environments; CRC Press: Boca Raton, FL, USA, 2015; pp. 355–366. [Google Scholar]
- Kolenčík, M.; Ernst, D.; Komár, M.; Urík, M.; Šebesta, M.; Dobročka, E.; Černý, I.; Illa, R.; Kanike, R.; Qian, Y. Effect of foliar spray application of zinc oxide nanoparticles on quantitative, nutritional, and physiological parameters of foxtail millet (Setaria italica l.) under field conditions. Nanomaterials 2019, 9, 1559. [Google Scholar] [CrossRef] [PubMed]
- García-Gómez, C.; Fernández, M.D.; García, S.; Obrador, A.F.; Letón, M.; Babín, M. Soil pH effects on the toxicity of zinc oxide nanoparticles to soil microbial community. Environ. Sci. Pollut. Res. 2018, 25, 28140–28152. [Google Scholar] [CrossRef] [PubMed]
- Šebesta, M.; Kolenčík, M.; Matúš, P.; Kořenková, L. Transport and distribution of engineered nanoparticles in soils and sediments. Chem. List. 2017, 111, 322–328. [Google Scholar]
- Šebesta, M.; Kolenčík, M.; Urík, M.; Bujdoš, M.; Vávra, I.; Dobročka, E.; Smilek, J.; Kalina, M.; Diviš, P.; Pavúk, M.; et al. Increased Colloidal Stability and Decreased Solubility—Sol-Gel Synthesis of Zinc Oxide Nanoparticles with Humic Acids. J. Nanosci. Nanotechnol. 2019, 19, 3024–3030. [Google Scholar] [CrossRef]
- Erazo, A.; Mosquera, S.A.; Rodríguez-Paéz, J.E. Synthesis of ZnO nanoparticles with different morphology: Study of their antifungal effect on strains of Aspergillus niger and Botrytis cinerea. Mater. Chem. Phys. 2019, 234, 172–184. [Google Scholar] [CrossRef]
- Rajput, V.D.; Minkina, T.M.; Behal, A.; Sushkova, S.N.; Mandzhieva, S.; Singh, R.; Gorovtsov, A.; Tsitsuashvili, V.S.; Purvis, W.O.; Ghazaryan, K.A.; et al. Effects of zinc-oxide nanoparticles on soil, plants, animals and soil organisms: A review. Environ. Nanotechnol. Monit. Manag. 2018, 9, 76–84. [Google Scholar] [CrossRef]
- Nisar, P.; Ali, N.; Rahman, L.; Ali, M.; Shinwari, Z.K. Antimicrobial activities of biologically synthesized metal nanoparticles: An insight into the mechanism of action. JBIC J. Biol. Inorg. Chem. 2019, 24, 929–941. [Google Scholar] [CrossRef]
- Mohamed, A.A.; Fouda, A.; Abdel-Rahman, M.A.; Hassan, S.E.-D.; El-Gamal, M.S.; Salem, S.S.; Shaheen, T.I. Fungal strain impacts the shape, bioactivity and multifunctional properties of green synthesized zinc oxide nanoparticles. Biocatal. Agric. Biotechnol. 2019, 19, 101103. [Google Scholar] [CrossRef]
- Gadd, G.M. Metals, minerals and microbes: Geomicrobiology and bioremediation. Microbiology 2010, 156, 609–643. [Google Scholar] [CrossRef]
- Qin, W.; Wang, C.; Ma, Y.; Shen, M.; Li, J.; Jiao, K.; Tay, F.R.; Niu, L. Microbe-Mediated Extracellular and Intracellular Mineralization: Environmental, Industrial, and Biotechnological Applications. Adv. Mater. 2020, 32, 1907833. [Google Scholar] [CrossRef] [PubMed]
- Burford, E.P.; Fomina, M.; Gadd, G.M. Fungal involvement in bioweathering and biotransformation of rocks and minerals. Mineral. Mag. 2003, 67, 1127–1155. [Google Scholar] [CrossRef]
- Polák, F.; Urík, M.; Bujdoš, M.; Matúš, P. Aspergillus niger enhances oxalate production as a response to phosphate deficiency induced by aluminium(III). J. Inorg. Biochem. 2020, 204, 110961. [Google Scholar] [CrossRef] [PubMed]
- Fomina, M.; Burford, E.P.; Gadd, G.M. Fungal dissolution and transformation of minerals: Significance for nutrient and metal mobility. In Fungi in Biogeochemical Cycles; Gadd, G.M., Ed.; Cambridge University Press: Cambridge, UK, 2006; pp. 236–266. ISBN 9780511550522. [Google Scholar]
- Polák, F.; Urík, M.; Matúš, P. Low molecular weight organic acids in soil environment. Chem. List. 2019, 113, 307–314. [Google Scholar]
- Boriová, K.; Urík, M.; Bujdoš, M.; Pifková, I.; Matúš, P. Chemical mimicking of bio-assisted aluminium extraction by Aspergillus niger’s exometabolites. Environ. Pollut. 2016, 218, 281–288. [Google Scholar] [CrossRef]
- Ström, L.; Owen, A.G.; Godbold, D.L.; Jones, D.L. Organic acid behaviour in a calcareous soil implications for rhizosphere nutrient cycling. Soil Biol. Biochem. 2005, 37, 2046–2054. [Google Scholar] [CrossRef]
- Sutjaritvorakul, T.; Gadd, G.M.; Whalley, A.J.S.; Suntornvongsagul, K.; Sihanonth, P. Zinc Oxalate Crystal Formation by Aspergillus nomius. Geomicrobiol. J. 2016, 33, 289–293. [Google Scholar] [CrossRef]
- Urík, M.; Hlodák, M.; Mikušová, P.; Matúš, P. Potential of Microscopic Fungi Isolated from Mercury Contaminated Soils to Accumulate and Volatilize Mercury(II). Water Air Soil Pollut. 2014, 225, 2219. [Google Scholar] [CrossRef]
- Sádecká, J.; Polonský, J. Determination of organic acids in tobacco by capillary isotachophoresis. J. Chromatogr. A 2003, 988, 161–165. [Google Scholar] [CrossRef]
- Bačík, P.; Ozdín, D.; Miglierini, M.; Kardošová, P.; Pentrák, M.; Haloda, J. Crystallochemical effects of heat treatment on Fe-dominant tourmalines from Dolní Bory (Czech Republic) and Vlachovo (Slovakia). Phys. Chem. Miner. 2011, 38, 599–611. [Google Scholar] [CrossRef]
- Ondruška, J.; Trnovcová, V.; Štubňa, I.; Bačík, P. Depolarization currents in illite. J. Therm. Anal. Calorim. 2018, 131, 2285–2289. [Google Scholar] [CrossRef]
- Du, W.; Sun, Y.; Ji, R.; Zhu, J.; Wu, J.; Guo, H. TiO2 and ZnO nanoparticles negatively affect wheat growth and soil enzyme activities in agricultural soil. J. Environ. Monit. 2011, 13, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Kool, P.L.; Ortiz, M.D.; van Gestel, C.A.M. Chronic toxicity of ZnO nanoparticles, non-nano ZnO and ZnCl2 to Folsomia candida (Collembola) in relation to bioavailability in soil. Environ. Pollut. 2011, 159, 2713–2719. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Liang, X.; Pendlowski, H.; Hillier, S.; Suntornvongsagul, K.; Sihanonth, P.; Gadd, G.M. Fungal biotransformation of zinc silicate and sulfide mineral ores. Environ. Microbiol. 2013, 15, 2173–2186. [Google Scholar] [CrossRef] [PubMed]
- Couri, S.; Pinto, G.A.S.; de Senna, L.F.; Martelli, H.L. Influence of metal ions on pellet morphology and polygalacturonase synthesis by Aspergillus niger 3T5B8. Braz. J. Microbiol. 2003, 34, 16–21. [Google Scholar] [CrossRef]
- Tarafdar, J.C.; Agrawal, A.; Raliya, R.; Kumar, P.; Burman, U.; Kaul, R.K. ZnO nanoparticles induced synthesis of polysaccharides and phosphatases by Aspergillus fungi. Adv. Sci. Eng. Med. 2012, 4, 324–328. [Google Scholar] [CrossRef]
- Wold, W.S.M.; Suzuki, I. The citric acid fermentation by Aspergillus niger: Regulation by zinc of growth and acidogenesis. Can. J. Microbiol. 1976, 22, 1083–1092. [Google Scholar] [CrossRef]
- Sardella, D.; Gatt, R.; Valdramidis, V.P. Assessing the efficacy of zinc oxide nanoparticles against Penicillium expansum by automated turbidimetric analysis. Mycology 2018, 9, 43–48. [Google Scholar] [CrossRef]
- Sawai, J.; Yoshikawa, T. Quantitative evaluation of antifungal activity of metallic oxide powders (MgO, CaO and ZnO) by an indirect conductimetric assay. J. Appl. Microbiol. 2004, 96, 803–809. [Google Scholar] [CrossRef]
- Sayer, J.A.; Gadd, G.M. Solubilization and transformation of insoluble inorganic metal compounds to insoluble metal oxalates by Aspergillus niger. Mycol. Res. 1997, 101, 653–661. [Google Scholar] [CrossRef]
- Hulkoti, N.I.; Taranath, T.C. Biosynthesis of nanoparticles using microbes—A review. Colloids Surf. B Biointerfaces 2014, 121, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Gadd, G.M. Fungi, Rocks, and Minerals. Elements 2017, 13, 171–176. [Google Scholar] [CrossRef]
- Akhtar, N.; Mannan, M.A. Mycoremediation: Expunging environmental pollutants. Biotechnol. Rep. 2020, 26, e00452. [Google Scholar] [CrossRef] [PubMed]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šebesta, M.; Urík, M.; Bujdoš, M.; Kolenčík, M.; Vávra, I.; Dobročka, E.; Kim, H.; Matúš, P. Fungus Aspergillus niger Processes Exogenous Zinc Nanoparticles into a Biogenic Oxalate Mineral. J. Fungi 2020, 6, 210. https://doi.org/10.3390/jof6040210
Šebesta M, Urík M, Bujdoš M, Kolenčík M, Vávra I, Dobročka E, Kim H, Matúš P. Fungus Aspergillus niger Processes Exogenous Zinc Nanoparticles into a Biogenic Oxalate Mineral. Journal of Fungi. 2020; 6(4):210. https://doi.org/10.3390/jof6040210
Chicago/Turabian StyleŠebesta, Martin, Martin Urík, Marek Bujdoš, Marek Kolenčík, Ivo Vávra, Edmund Dobročka, Hyunjung Kim, and Peter Matúš. 2020. "Fungus Aspergillus niger Processes Exogenous Zinc Nanoparticles into a Biogenic Oxalate Mineral" Journal of Fungi 6, no. 4: 210. https://doi.org/10.3390/jof6040210
APA StyleŠebesta, M., Urík, M., Bujdoš, M., Kolenčík, M., Vávra, I., Dobročka, E., Kim, H., & Matúš, P. (2020). Fungus Aspergillus niger Processes Exogenous Zinc Nanoparticles into a Biogenic Oxalate Mineral. Journal of Fungi, 6(4), 210. https://doi.org/10.3390/jof6040210