Candida spp./Bacteria Mixed Biofilms
Abstract
1. An Overview of Single and Polymicrobial Biofilms Involving Candida spp. and Bacterial Species
2. Epidemiology of Candida spp./Bacteria Single and Mixed Biofilms
2.1. Epidemiology of Candida spp. Single Biofilms
2.2. Epidemiology of Bacterial Single Biofilms
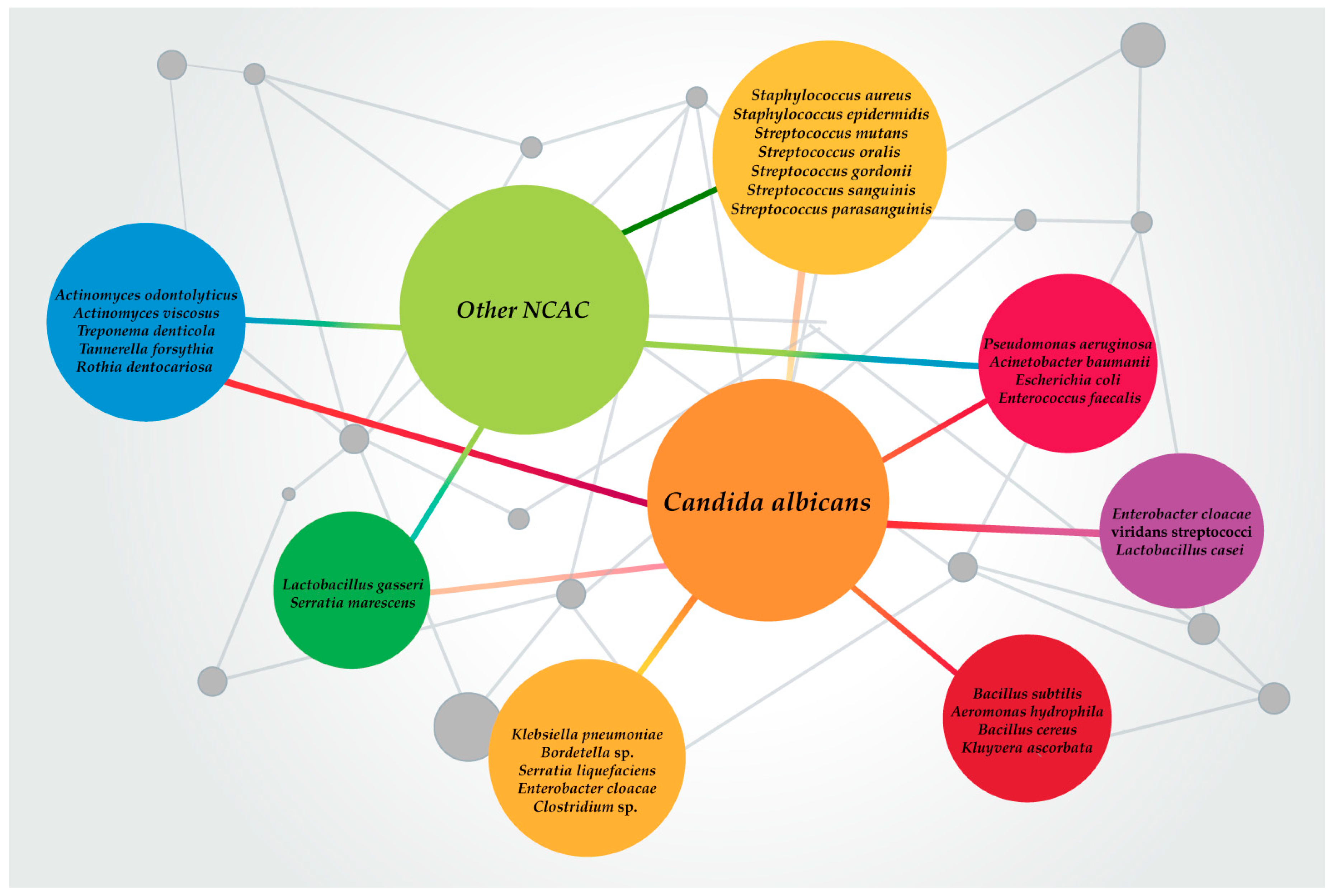
2.3. Epidemiology of Candida spp. and Bacteria Mixed Biofilms
3. Candida/Bacteria Mixed Biofilms: Characterization and the Problematic of the Biofilms’ Drug Resistance
3.1. Mixed Candida spp./Bacteria Biofilms: Features, Pathogenicity, and Virulence
3.2. Mixed Candida spp./Bacteria Biofilms vs. Oral Biofilms Features, Pathogenicity, and Virulence
4. Management of Candida spp./Bacterial Biofilms: Is this the Impossible Mission?
4.1. Oral Disease Management
4.2. Innovative Treatments of Other Diseases
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Donlan, R.M. Biofilms: Microbial life on surfaces. Emerg. Infect. Dis. 2002, 8, 881–890. [Google Scholar] [CrossRef]
- Serra, D.O.; Klauck, G.; Hengge, R. Vertical stratification of matrix production is essential for physical integrity and architecture of macrocolony biofilms of Escherichia coli. Environ. Microbiol. 2015, 17, 5073–5088. [Google Scholar] [CrossRef]
- Rodrigues, C.; Silva, S.; Henriques, M. Candida glabrata: A review of its features and resistance. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 673–688. [Google Scholar] [CrossRef]
- Rodrigues, M.E.; Silva, S.; Azeredo, J.; Henriques, M. Novel strategies to fight Candida species infection. Crit. Rev. Microbiol. 2014, 42, 594–606. [Google Scholar]
- Davies, D. Understanding biofilm resistance to antibacterial agents. Nat. Rev. Drug Discov. 2003, 2, 114–122. [Google Scholar] [CrossRef]
- dos Santos, A.L.S.; Galdino, A.C.M.; de Mello, T.P.; de Ramos, L.S.; Branquinha, M.H.; Bolognese, A.M.; Columbano Neto, J.; Roudbary, M. What are the advantages of living in a community? A microbial biofilm perspective! Mem. Inst. Oswaldo Cruz 2018, 113, e180212. [Google Scholar] [CrossRef]
- Wu, H.; Moser, C.; Wang, H.Z.; Høiby, N.; Song, Z.J. Strategies for combating bacterial biofilm infections. Int. J. Oral Sci. 2015, 7, 1–7. [Google Scholar] [CrossRef]
- Cavalheiro, M.; Teixeira, M.C. Candida Biofilms: Threats, Challenges, and Promising Strategies. Front. Med. 2018, 5, 28. [Google Scholar] [CrossRef]
- Armbruster, C.R.; Parsek, M.R. New insight into the early stages of biofilm formation. Proc. Natl. Acad. Sci. USA 2018, 115, 4317–4319. [Google Scholar] [CrossRef]
- Bjarnsholt, T. The role of bacterial biofilms in chronic infections. Supplementum. APMIS 2013, 136. [Google Scholar] [CrossRef]
- Mulani, M.S.; Kamble, E.E.; Kumkar, S.N.; Tawre, M.S.; Pardesi, K.R. Emerging strategies to combat ESKAPE pathogens in the era of antimicrobial resistance: A review. Front. Microbiol. 2019, 10, 539. [Google Scholar] [CrossRef]
- Percival, S.L.; Suleman, L.; Vuotto, C.; Donelli, G. Healthcare-Associated infections, medical devices and biofilms: Risk, tolerance and control. J. Med. Microbiol. 2015, 64, 323–334. [Google Scholar] [CrossRef]
- Lewis, K. Riddle of Biofilm Resistance. Antimicrob. Agents Chemother. 2001, 45, 999–1007. [Google Scholar] [CrossRef]
- Hancock, R.E.W. Rethinking the Antibiotic Discovery Paradigm. EBioMedicine 2015, 2, 629–630. [Google Scholar] [CrossRef][Green Version]
- Donlan, R.M.; Costerton, J.W. Biofilms: Survival mechanisms of clinically relevant microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar] [CrossRef]
- Ribeiro, S.M.; Felício, M.R.; Boas, E.V.; Gonçalves, S.; Costa, F.F.; Samy, R.P.; Santos, N.C.; Franco, O.L. New frontiers for anti-biofilm drug development. Pharmacol. Ther. 2016, 160, 133–144. [Google Scholar] [CrossRef]
- Harriott, M.M.; Noverr, M.C. Importance of Candida-bacterial polymicrobial biofilms in disease. Trends Microbiol. 2011, 19, 557–563. [Google Scholar] [CrossRef]
- Mahajan, A.; Singh, B.; Kashyap, D.; Kumar, A.; Mahajan, P. Interspecies communication and periodontal disease. Sci. World J. 2013, 2013, 765434. [Google Scholar] [CrossRef]
- Pathirana, R.U.; McCall, A.D.; Norris, H.L.; Edgerton, M. Filamentous non-albicans Candida species adhere to Candida albicans and benefit from dual biofilm growth. Front. Microbiol. 2019, 10, 1188. [Google Scholar] [CrossRef]
- Srinivasan, A.; Torres, N.S.; Leung, K.P.; Lopez-Ribot, J.L.; Ramasubramanian, A.K. nBioChip, a Lab-on-a-Chip Platform of Mono- and Polymicrobial Biofilms for High-Throughput Downstream Applications. mSphere 2017, 2, e00247-17. [Google Scholar] [CrossRef]
- Shirtliff, M.E.; Peters, B.M.; Jabra-Rizk, M.A. Cross-kingdom interactions: Candida albicans and bacteria. FEMS Microbiol. Lett. 2009, 299, 1–8. [Google Scholar] [CrossRef]
- Thein, Z.M.; Seneviratne, C.J.; Samaranayake, Y.H.; Samaranayake, L.P. Community lifestyle of Candida in mixed biofilms: A mini review. Mycoses 2009, 52, 467–475. [Google Scholar] [CrossRef]
- Hawser, S.P.; Douglas, L.J. Biofilm formation by Candida species on the surface of catheter materials in vitro. Infect. Immun. 1994, 62, 915–921. [Google Scholar]
- Kojic, E.M.; Darouiche, R.O. Candida infections of medical devices. Clin. Microbiol. Rev. 2004, 17, 255–267. [Google Scholar] [CrossRef]
- Viudes, A.; Pemán, J.; Cantón, E.; Úbeda, P.; López-Ribot, J.L.; Gobernado, M. Candidemia at a tertiary-care hospital: Epidemiology, treatment, clinical outcome and risk factors for death. Eur. J. Clin. Microbiol. Infect. Dis. 2002, 21, 767–774. [Google Scholar] [CrossRef]
- Finkel, J.S.; Mitchell, A.P. Genetic control of Candida albicans biofilm development. Nat. Rev. Microbiol. 2011, 9, 109. [Google Scholar] [CrossRef]
- Sardi, J.C.O.; Scorzoni, L.; Bernardi, T.; Fusco-Almeida, A.M.; Mendes Giannini, M.J.S.; Bernardi, T.; Scorzoni, L.; Fusco-Almeida, A.M.; Sardi, J.C.O.; Scorzoni, L.; et al. Candida species: Current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. J. Med. Microbiol. 2013, 62, 10–24. [Google Scholar] [CrossRef]
- Byers, M.; Chapman, S.; Feldman, S.; Parent, A. Fluconazole pharmacokinetics in the cerebrospinal fluid of a child with Candida tropicalis meningitis. Pediatr. Infect. Dis. J. 1992, 11, 895–896. [Google Scholar]
- Didone, L.; Oga, D.; Krysan, D.J. A novel assay of biofilm antifungal activity reveals that amphotericin B and caspofungin lyse Candida albicans cells in biofilms. Yeast 2011, 28, 561–568. [Google Scholar] [CrossRef]
- Nett, J.; Lincoln, L.; Marchillo, K.; Massey, R.; Holoyda, K.; Hoff, B.; VanHandel, M.; Andes, D. Putative role of beta-1,3 glucans in Candida albicans biofilm resistance. Antimicrob. Agents Chemother. 2007, 51, 510–520. [Google Scholar] [CrossRef]
- Ozkan, S.; Kaynak, F.; Kalkanci, A.; Abbasoglu, U.; Kustimur, S. Slime production and proteinase activity of Candida species isolated from blood samples and the comparison of these activities with minimum inhibitory concentration values of antifungal agents. Mem. Inst. Oswaldo Cruz 2005, 100, 319–324. [Google Scholar] [CrossRef]
- Dongari-Bagtzoglou, A.; Dwivedi, P.; Ioannidou, E.; Shaqman, M.; Hull, D.; Burleson, J. Oral Candida infection and colonization in solid organ transplant recipients. Oral Microbiol. Immunol. 2009, 24, 249–254. [Google Scholar] [CrossRef]
- Almirante, B.; Rodríguez, D.; Park, B.J.; Cuenca-Estrella, M.; Planes, A.M.; Almela, M.; Mensa, J.; Sanchez, F.; Ayats, J.; Gimenez, M.; et al. Epidemiology and Predictors of Mortality in Cases of Candida Bloodstream Infection: Results from Population-Based Surveillance, Barcelona, Spain, from 2002 to 2003 Epidemiology and Predictors of Mortality in Cases of Candida Bloodstream Infection: Re. J. Clin. Microbiol. 2005, 43, 1829–1835. [Google Scholar] [CrossRef]
- Lai, C.C.; Wang, C.Y.; Liu, W.L.; Huang, Y.T.; Hsueh, P.R. Time to positivity of blood cultures of different Candida species causing fungaemia. J. Med. Microbiol. 2012, 61, 701–704. [Google Scholar] [CrossRef]
- Tortorano, A.M.; Kibbler, C.; Peman, J.; Bernhardt, H.; Klingspor, L.; Grillot, R. Candidaemia in Europe: Epidemiology and resistance. Int. J. Antimicrob. Agents 2006, 27, 359–366. [Google Scholar] [CrossRef]
- Colombo, A.L.; Nucci, M.; Park, B.J.; Nouér, S.A.; Arthington-Skaggs, B.; da Matta, D.A.; Warnock, D.; Morgan, J.; Brazilian Network Candidemia Study. Epidemiology of candidemia in Brazil: A nationwide sentinel surveillance of candidemia in eleven medical centers. J. Clin. Microbiol. 2006, 44, 2816–2823. [Google Scholar] [CrossRef]
- Nucci, M.; Queiroz-Telles, F.; Tobón, A.M.; Restrepo, A.; Colombo, A.L. Epidemiology of Opportunistic Fungal Infections in Latin America. Clin. Infect. Dis. 2010, 51, 561–570. [Google Scholar] [CrossRef]
- Horn, D.L.; Neofytos, D.; Anaissie, E.J.; Fishman, J.A.; Steinbach, W.J.; Olyaei, A.J.; Marr, K.A.; Pfaller, M.A.; Chang, C.; Webster, K.M. Epidemiology and Outcomes of Candidemia in 2019 Patients: Data from the Prospective Antifungal Therapy Alliance Registry. Clin. Infect. Dis. 2009, 48, 1695–1703. [Google Scholar] [CrossRef]
- Cantón, E.; Pemán, J.; Quindós, G.; Eraso, E.; Miranda-Zapico, I.; Álvarez, M.; Merino, P.; Campos-Herrero, I.; Marco, F.; De La Pedrosa, E.G.G.; et al. Prospective multicenter study of the epidemiology, molecular identification, and antifungal susceptibility of Candida parapsilosis, Candida orthopsilosis, and Candida metapsilosis isolated from patients with candidemia. Antimicrob. Agents Chemother. 2011, 55, 5590–5596. [Google Scholar] [CrossRef]
- Colombo, A.L.; Guimarães, T.; Silva, L.R.B.F.; de Monfardini, L.P.A.; Cunha, A.K.B.; Rady, P.; Alves, T.; Rosas, R.C. Prospective Observational Study of Candidemia in São Paulo, Brazil: Incidence Rate, Epidemiology, and Predictors of Mortality. Infect. Control Hosp. Epidemiol. 2007, 28, 570–576. [Google Scholar] [CrossRef]
- Tintelnot, K.; Haase, G.; Seibold, M.; Bergmann, F.; Staemmler, M.; Franz, T.; Naumann, D. Evaluation of phenotypic markers for selection and identification of Candida dubliniensis. J. Clin. Microbiol. 2000, 38, 1599–1608. [Google Scholar]
- Lasker, B.A.; Elie, C.M.; Lott, T.J.; Espinel-Ingroff, A.; Gallagher, L.; Kuykendall, R.J.; Kellum, M.E.; Pruitt, W.R.; Warnock, D.W.; Rimland, D.; et al. Molecular epidemiology of Candida albicans strains isolated from the oropharynx of HIV-positive patients at successive clinic visits. Med. Mycol. 2001, 39, 341–352. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Messer, S.A.; Hollis, R.J.; Boyken, L.; Tendolkar, S.; Kroeger, J.; Diekema, D.J. Variation in susceptibility of bloodstream isolates of Candida glabrata to fluconazole according to patient age and geographic location in the United States in 2001 to 2007. J. Clin. Microbiol. 2009, 47, 3185–3190. [Google Scholar] [CrossRef]
- Pires-Gonçalves, R.H.; Miranda, E.T.; Baeza, L.C.; Matsumoto, M.T.; Zaia, J.E.; Mendes-Giannini, M.J.S. Genetic relatedness of commensal strains of Candida albicans carried in the oral cavity of patients’ dental prosthesis users in Brazil. Mycopathologia 2007, 164, 255–263. [Google Scholar] [CrossRef]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. 2018, 81, 7–11. [Google Scholar] [CrossRef]
- Ciofu, O.; Rojo-Molinero, E.; Macià, M.D.; Oliver, A. Antibiotic treatment of biofilm infections. APMIS 2017, 125, 304–319. [Google Scholar] [CrossRef]
- Rodrigues, C.F.; Rodrigues, M.; Henriques, M. Candida sp. Infections in Patients with Diabetes Mellitus. J. Clin. Med. 2019, 8, 76. [Google Scholar] [CrossRef]
- Vickery, K.; Hu, H.; Jacombs, A.S.; Bradshaw, D.A.; Deva, A.K. A review of bacterial biofilms and their role in device-associated infection. Healthc. Infect. 2013, 18, 61–66. [Google Scholar] [CrossRef]
- Vuotto, C.; Donelli, G. Anaerobes in biofilm-based healthcare-associated infections. Adv. Exp. Med. Biol. 2015, 830, 97–112. [Google Scholar]
- Japanese Society of Chemotherapy Committee on guidelines for treatment of anaerobic infections; Japanese Association for Anaerobic Infections Research. Chapter 1-1. Anaerobic infections (General): Epidemiology of anaerobic infections. J. Infect. Chemother. 2011, 17, 4–12. [Google Scholar] [CrossRef][Green Version]
- Watanabe, T.; Maruyama, F.; Nozawa, T.; Aoki, A.; Okano, S.; Shibata, Y.; Oshima, K.; Kurokawa, K.; Hattori, M.; Nakagawa, I.; et al. Complete genome sequence of the bacterium Porphyromonas gingivalis TDC60, which causes periodontal disease. J. Bacteriol. 2011, 193, 4259–4260. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Overman, P.R. Biofilm: A new view of plaque. J. Contemp. Dent. Pract. 2000, 1, 13–20. [Google Scholar] [CrossRef]
- Chihara, S.; Segreti, J. Osteomyelitis. Dis. Mon. 2010, 56, 6–31. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Fuchs, B.; Wang, Y.; Chen, W.; Yuen, G.; Chen, R.; Jayamani, E.; Anastassopoulou, C.; Pukkila-Worley, R.; Coleman, J.; et al. The role of Candida albicans SPT20 in filamentation, biofilm formation and pathogenesis. PLoS ONE 2014, 9, e94468. [Google Scholar] [CrossRef] [PubMed]
- Campoccia, D.; Montanaro, L.; Arciola, C.R. A review of the clinical implications of anti-infective biomaterials andinfection-resistant surfaces. Biomaterials 2013, 34, 8018–8029. [Google Scholar] [CrossRef] [PubMed]
- Arciola, C.R.; Campoccia, D.; Speziale, P.; Montanaro, L.; Costerton, J.W. Biofilm formation in Staphylococcus implant infections. A review of molecular mechanisms and implications for biofilm-resistant materials. Biomaterials 2012, 33, 5967–5982. [Google Scholar] [CrossRef]
- Treatment of Infections Associated with Surgical Implants. PubMed-NCBI. Available online: https://www.ncbi.nlm.nih.gov/pubmed/15070792 (accessed on 8 November 2019).
- Arciola, C.R.; An, Y.H.; Campoccia, D.; Donati, M.E.; Montanaro, L. Etiology of implant orthopedic infections: A survey on 1027 clinical isolates. Int. J. Artif. Organs 2005, 28, 1091–1100. [Google Scholar] [CrossRef]
- Von Eiff, C.; Arciola, C.R.; Montanaro, L.; Becker, K.; Campoccia, D. Emerging staphylococcus species as new pathogens in implant infections. Int. J. Artif. Organs 2006, 29, 360–367. [Google Scholar] [CrossRef]
- Campoccia, D.; Montanaro, L.; Visai, L.; Corazzari, T.; Poggio, C.; Pegreffi, F.; Maso, A.; Pirini, V.; Ravaioli, S.; Cangini, I.; et al. Characterization of 26 Staphylococcus warneri isolates from orthopedic infections. Int. J. Artif. Organs 2010, 33, 575–581. [Google Scholar] [CrossRef]
- Campoccia, D.; Baldassarri, L.; Pirini, V.; Ravaioli, S.; Montanaro, L.; Arciola, C.R. Molecular epidemiology of Staphylococcus aureus from implant orthopaedic infections: Ribotypes, agr polymorphism, leukocidal toxins and antibiotic resistance. Biomaterials 2008, 29, 4108–4116. [Google Scholar] [CrossRef]
- Arciola, C.R.; Campoccia, D.; Gamberini, S.; Donati, M.E.; Pirini, V.; Visai, L.; Speziale, P.; Montanaro, L. Antibiotic resistance in exopolysaccharide-forming Staphylococcus epidermidis clinical isolates from orthopaedic implant infections. Biomaterials 2005, 26, 6530–6535. [Google Scholar] [CrossRef] [PubMed]
- Campoccia, D.; Montanaro, L.; Von Eiff, C.; Pirini, V.; Ravaioli, S.; Becker, K.; Arciola, C.R. Cluster analysis of ribotyping profiles of Staphylococcus epidermidis isolates recovered from foreign body-associated orthopedic infections. J. Biomed. Mater. Res. Part A 2009, 88, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Hwang, G.; Liu, Y.; Kim, D.; Li, Y.; Krysan, D.J.; Koo, H. Candida albicans mannans mediate Streptococcus mutans exoenzyme GtfB binding to modulate cross-kingdom biofilm development in vivo. PLoS Pathog. 2017, 13, e1006407. [Google Scholar] [CrossRef] [PubMed]
- Arciola, C.R.; Baldassarri, L.; Campoccia, D.; Creti, R.; Pirini, V.; Huebner, J.; Montanaro, L. Strong biofilm production, antibiotic multi-resistance and high gelE expression in epidemic clones of Enterococcus faecalis from orthopaedic implant infections. Biomaterials 2008, 29, 580–586. [Google Scholar] [CrossRef]
- Montanaro, L.; Poggi, A.; Visai, L.; Ravaioli, S.; Campoccia, D.; Speziale, P.; Arciola, C.R. Extracellular DNA in biofilms. Int. J. Artif. Organs 2011, 34, 824–831. [Google Scholar] [CrossRef]
- Frank, K.L.; Patel, R. Poly-N-acetylglucosamine is not a major component of the extracellular matrix in biofilms formed by icaADBC-positive Staphylococcus lugdunensis isolates. Infect. Immun. 2007, 75, 4728–4742. [Google Scholar] [CrossRef]
- Fernandez Sampedro, M.; Piper, K.E.; McDowell, A.; Patrick, S.; Mandrekar, J.N.; Rouse, M.S.; Steckelberg, J.M.; Patel, R. Species of Propionibacterium and Propionibacterium acnes phylotypes associated with orthopedic implants. Diagn. Microbiol. Infect. Dis. 2009, 64, 138–145. [Google Scholar] [CrossRef]
- Mond, H.G.; Proclemer, A. The 11th world survey of cardiac pacing and implantable cardioverter-defibrillators: Calendar year 2009—A world society of Arrhythmia’s project. PACE Pacing Clin. Electrophysiol. 2011, 34, 1013–1027. [Google Scholar] [CrossRef]
- Arnold, C.J.; Chu, V.H. Cardiovascular Implantable Electronic Device Infections. Infect. Dis. Clin. N. Am. 2018, 32, 811–825. [Google Scholar] [CrossRef]
- Brabham, W.W.; Wray, D.W.; Gold, M.R. Microbiologic characteristics and antimicrobial susceptibility of pacemaker/ICD infections: A moving target! J. Cardiovasc. Electrophysiol. 2012, 23, 382–383. [Google Scholar] [CrossRef]
- Karchmer, A.W.; Longworth, D.L. Infections of intracardiac devices. Infect. Dis. Clin. N. Am. 2002, 16, 477–505. [Google Scholar] [CrossRef]
- Jan, E.; Camou, F.; Texier-Maugein, J.; Whinnett, Z.; Caubet, O.; Ploux, S.; Pellegrin, J.-L.; Ritter, P.; Metayer, P.L.; Roudaut, R.; et al. Microbiologic Characteristics and In Vitro Susceptibility to Antimicrobials in a Large Population of Patients with Cardiovascular Implantable Electronic Device Infection. J. Cardiovasc. Electrophysiol. 2012, 23, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Maki, D.G. The prevention and management of device-related infection in infusion therapy. J. Med. 1980, 11, 239–253. [Google Scholar] [PubMed]
- Stickler, D.J. Bacterial biofilms and the encrustation of urethral catheters. Biofouling 1996, 9, 293–305. [Google Scholar] [CrossRef]
- Kirmusaoğlu, S.; Yurdugül, S.; Metin, A.; Vehid, S. The effect of urinary catheters on microbial biofilms and catheter associated urinary tract infections. Urol. J. 2017, 14, 3028–3034. [Google Scholar]
- Morales, D.K.; Hogan, D.A. Candida albicans interactions with bacteria in the context of human health and disease. PLoS Pathog. 2010, 6, e1000886. [Google Scholar] [CrossRef]
- Baena-Monroy, T.; Moreno-Maldonado, V.; Franco-Martínez, F.; Aldape-Barrios, B.; Quindós, G.; Sánchez-Vargas, L.O. Candida albicans, Staphylococcus aureus and Streptococcus mutans colonization in patients wearing dental prosthesis. Med. Oral Patol. Oral Cir. Bucal 2005, 10, E27–E39. [Google Scholar]
- Lebeaux, D.; Ghigo, J.M.; Beloin, C. Biofilm-Related Infections: Bridging the Gap between Clinical Management and Fundamental Aspects of Recalcitrance toward Antibiotics. Microb. Mol. Biol. Rev. 2014, 78, 3–9. [Google Scholar] [CrossRef]
- Klotz, S.A.; Chasin, B.S.; Powell, B.; Gaur, N.K.; Lipke, P.N. Polymicrobial bloodstream infections involving Candida species: Analysis of patients and review of the literature. Diagn. Microbiol. Infect. Dis. 2007, 59, 401–406. [Google Scholar] [CrossRef]
- Pammi, M.; Zhong, D.; Johnson, Y.; Revell, P.; Versalovic, J. Polymicrobial bloodstream infections in the neonatal intensive care unit are associated with increased mortality: A case-control study. BMC Infect. Dis. 2014, 14, 390. [Google Scholar] [CrossRef]
- Klotz, S.A.; Gaur, N.K.; De Armond, R.; Sheppard, D.; Khardori, N.; Edwards, J.E.; Lipke, P.N.; El-Azizi, M. Candida albicans Als proteins mediate aggregation with bacteria and yeasts. Med. Mycol. 2007, 45, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Marks, D.J.B.; Hyams, C.; Koo, C.Y.; Pavlou, M.; Robbins, J.; Koo, C.S.; Rodger, G.; Huggett, J.F.; Yap, J.; Macrae, M.B.; et al. Clinical features, microbiology and surgical outcomes of infective endocarditis: A 13-year study from a UK tertiary cardiothoracic referral centre. QJM 2015, 108, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Thuny, F.; Grisoli, D.; Collart, F.; Habib, G.; Raoult, D. Management of infective endocarditis: Challenges and perspectives. Lancet 2012, 379, 965–975. [Google Scholar] [CrossRef]
- Allison, D.L.; Scheres, N.; Willems, H.M.E.; Bode, C.S.; Krom, B.P.; Shirtliff, M.E. The host immune system facilitates disseminated Staphylococcus aureus disease due to phagocytic attraction to Candida albicans during coinfection: A case of bait and switch. Infect. Immun. 2019, 87, e00137-19. [Google Scholar] [CrossRef]
- Schlecht, L.M.; Peters, B.M.; Krom, B.P.; Freiberg, J.A.; Hänsch, G.M.; Filler, S.G.; Jabra-Rizk, M.A.; Shirtliff, M.E. Systemic Staphylococcus aureus infection mediated by Candida albicans hyphal invasion of mucosal tissue. Microbiology 2015, 161, 168–181. [Google Scholar] [CrossRef]
- Ovchinnikova, E.S.; Krom, B.P.; Busscher, H.J.; Van Der Mei, H.C. Evaluation of adhesion forces of Staphylococcus aureus along the length of Candida albicans hyphae. BMC Microbiol. 2012, 12, 281. [Google Scholar] [CrossRef]
- Sousa, C.; Botelho, C.; Rodrigues, D.; Azeredo, J.; Oliveira, R. Infective endocarditis in intravenous drug abusers: An update. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2905–2910. [Google Scholar] [CrossRef]
- Ochieng, W.; Wanzala, P.; Bii, C.; Oishi, J.; Ichimura, H.; Lihana, R.; Mpoke, S.; Mwaniki, D.; Okoth, F.A. Tuberculosis and oral Candida species surveillance in HIV infected individuals in Northern Kenya, and the implications on tuberculin skin test screening for DOPT-P. E. Afr. Med. J. 2005, 82, 609. [Google Scholar] [CrossRef]
- Morales, D.K.; Jacobs, N.J.; Rajamani, S.; Krishnamurthy, M.; Cubillos-Ruiz, J.R.; Hogan, D.A. Antifungal mechanisms by which a novel Pseudomonas aeruginosa phenazine toxin kills Candida albicans in biofilms. Mol. Microbiol. 2010, 78, 1379–1392. [Google Scholar] [CrossRef]
- Douglas, L.J. Medical importance of biofilms in Candida infections. Rev. Iberoam. Micol. 2002, 19, 139–143. [Google Scholar]
- Metwalli, K.H.; Khan, S.A.; Krom, B.P.; Jabra-Rizk, M.A. Streptococcus mutans, Candida albicans, and the Human Mouth: A Sticky Situation. PLoS Pathog. 2013, 9, e1003616. [Google Scholar] [CrossRef] [PubMed]
- Stiefel, U.; Donskey, C.J. The Role of the Intestinal Tract as a Source for Transmission of Nosocomial Pathogens. Curr. Infect. Dis. Rep. 2004, 6, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Stuart, C.H.; Schwartz, S.A.; Beeson, T.J.; Owatz, C.B. Enterococcus faecalis: Its Role in Root Canal Treatment Failure and Current Concepts in Retreatment. J. Endod. 2006, 32, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Hermann, C.; Munzel, U.; Rüchel, R. Bacterial flora accompanying Candida yeasts in clinical specimens. Mycoses 1999, 42, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, E.J.C. Intra-Abdominal Anaerobic Infections: Bacteriology and Therapeutic Potential of Newer Antimicrobial Carbapenem, Fluoroquinolone, and Desfluoroquinolone Therapeutic Agents. Clin. Infect. Dis. 2002, 35, S106–S111. [Google Scholar] [CrossRef] [PubMed]
- Edey, M.; Hawley, C.M.; McDonald, S.P.; Brown, F.G.; Rosman, J.B.; Wiggins, K.J.; Bannister, K.M.; Johnson, D.W. Enterococcal peritonitis in Australian peritoneal dialysis patients: Predictors, treatment and outcomes in 116 cases. Nephrol. Dial. Transplant. 2010, 25, 1272–1278. [Google Scholar] [CrossRef] [PubMed]
- Barraclough, K.; Hawley, C.M.; McDonald, S.P.; Brown, F.G.; Rosman, J.B.; Wiggins, K.J.; Bannister, K.M.; Johnson, D.W. Polymicrobial Peritonitis in Peritoneal Dialysis Patients in Australia: Predictors, Treatment, and Outcomes. Am. J. Kidney Dis. 2010, 55, 121–131. [Google Scholar] [CrossRef]
- Dupont, H.; Paugam-Burtz, C.; Muller-Serieys, C.; Fierobe, L.; Chosidow, D.; Marmuse, J.P.; Mantz, J.; Desmonts, J.M.; Solomkin, J.S. Predictive factors of mortality due to polymicrobial peritonitis with Candida isolation in peritoneal fluid in critically III patients. Arch. Surg. 2002, 137, 1341–1346. [Google Scholar] [CrossRef]
- Orsi, C.F.; Sabia, C.; Ardizzoni, A.; Colombari, B.; Neglia, R.G.; Peppoloni, S.; Morace, G.; Blasi, E. Inhibitory effects of different lactobacilli on Candida albicans hyphal formation and biofilm development. J. Biol. Regul. Homeost. Agents 2014, 28, 743–752. [Google Scholar]
- Benadé, E.; Stone, W.; Mouton, M.; Postma, F.; Wilsenach, J.; Botha, A. Binary Interactions of Antagonistic Bacteria with Candida albicans Under Aerobic and Anaerobic Conditions. Microb. Ecol. 2016, 71, 645–659. [Google Scholar] [CrossRef]
- Tan, Y.; Leonhard, M.; Schneider-Stickler, B. Evaluation of culture conditions for mixed biofilm formation with clinically isolated non-albicans Candida species and Staphylococcus epidermidis on silicone. Microb. Pathog. 2017, 112, 215–220. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Junior, N.M.; Mendoza Marin, D.O.; Leite, A.R.P.; Pero, A.C.; Klein, M.I.; Compagnoni, M.A. Influence of the use of complete denture adhesives on microbial adhesion and biofilm formation by single- and mixed-species. PLoS ONE 2018, 13, e0203951. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.F.; Boas, D.; Haynes, K.; Henriques, M. The MNN2 Gene Knockout Modulates the Antifungal Resistance of Biofilms of Candida glabrata. Biomolecules 2018, 8, 130. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.F.; Henriques, M. Portrait of Matrix Gene Expression in Candida glabrata Biofilms with Stress Induced by Different Drugs. Genes 2018, 9, 205. [Google Scholar] [CrossRef] [PubMed]
- Kong, E.F.; Tsui, C.; Kucharkov, S.; Andes, D.; Van Dijck, P.; Jabra-Rizk, M.A. Commensal protection of Staphylococcus aureus against antimicrobials by Candida albicans biofilm matrix. MBio 2016, 7, e01365-16. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, X.; Huang, Y.; Zhao, G.; Lei, Y.; Ye, L.; Huang, Q.; Duan, W. Study on the Structure of Candida albicans–Staphylococcus epidermidis Mixed Species Biofilm on Polyvinyl Chloride Biomaterial. Cell Biochem. Biophys. 2015, 73, 461–468. [Google Scholar] [CrossRef]
- Bertolini, M.M.; Xu, H.; Sobue, T.; Nobile, C.J.; Del Bel Cury, A.A.; Dongari-Bagtzoglou, A. Candida-streptococcal mucosal biofilms display distinct structural and virulence characteristics depending on growth conditions and hyphal morphotypes. Mol. Oral Microbiol. 2015, 30, 307–322. [Google Scholar] [CrossRef]
- Seghir, A.; Boucherit-Otmani, Z.; Boucherit, K.; Sari-Belkharroubi, L.; Anselme-Bertrand, I. Évaluation du potentiel de formation de biofilms mixtes entre Candida albicans et quelques espèces bactériennes isolées de cathéters vasculaires périphériques au CHU de Tlemcen. Première étude en Algérie. J. Mycol. Med. 2015, 25, 123–129. [Google Scholar] [CrossRef]
- Camarillo-Márquez, O.; Córdova-Alcántara, I.M.; Hernández-Rodríguez, C.H.; García-Pérez, B.E.; Martínez-Rivera, M.A.; Rodríguez-Tovar, A.V. Antagonistic Interaction of Staphylococcus aureus Toward Candida glabrata During in vitro Biofilm Formation Is Caused by an Apoptotic Mechanism. Front. Microbiol. 2018, 9, 2031. [Google Scholar] [CrossRef]
- Martins, C.H.G.; Pires, R.H.; Cunha, A.O.; Pereira, C.A.M.; de Lacorte Singulani, J.; Abrão, F.; de Moraes, T.; Mendes-Giannini, M.J.S. Candida/Candida biofilms. First description of dual-species Candida albicans/C. rugosa biofilm. Fungal Biol. 2016, 120, 530–537. [Google Scholar] [CrossRef]
- Staib, F.; Grosse, G.; Mishra, S.K. Staphylococcus aureus and Candida albicans infection (animal experiments). Zentralbl. Bakteriol. Orig. A 1976, 234, 450–461. [Google Scholar] [PubMed]
- Carlson, E. Synergistic effect of Candida albicans and Staphylococcus aureus on mouse mortality. Infect. Immun. 1982, 38, 921–924. [Google Scholar] [PubMed]
- Peters, B.M.; Noverr, M.C. Candida albicans-Staphylococcus aureus Polymicrobial Peritonitis Modulates Host Innate Immunity. Infect. Immun. 2013, 81, 2178–2189. [Google Scholar] [CrossRef] [PubMed]
- Zago, C.E.; Silva, S.; Sanitá, P.V.; Barbugli, P.A.; Dias, C.M.I.; Lordello, V.B.; Vergani, C.E. Dynamics of biofilm formation and the Interaction between Candida albicans and methicillin-susceptible (MSSA) and-resistant Staphylococcus aureus (MRSA). PLoS ONE 2015, 10, e0123206. [Google Scholar] [CrossRef] [PubMed]
- Dutton, L.C.; Jenkinson, H.F.; Lamont, R.J.; Nobbs, A.H. Role of Candida albicans secreted aspartyl protease Sap9 in interkingdom biofilm formation. Pathog. Dis. 2016, 74, ftw005. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, Y.; Huang, Y.; Zhou, Y.; Zhao, G.; Ye, L.; Lei, Y.; Tang, Q. Function of intercellular adhesion a, fibrinogen binding protein, and accumulation-associated protein genes in formation of Staphylococcus epidermidis-Candida albicans mixed species biofilms. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2015, 29, 63–68. [Google Scholar] [PubMed]
- Krause, J.; Geginat, G.; Tammer, I. Prostaglandin E2 from Candida albicans Stimulates the Growth of Staphylococcus aureus in Mixed Biofilms. PLoS ONE 2015, 10, e0135404. [Google Scholar] [CrossRef]
- Kong, E.F.; Tsui, C.; Kucharíková, S.; Van Dijck, P.; Jabra-Rizk, M.A. Modulation of Staphylococcus aureus Response to Antimicrobials by the Candida albicans Quorum Sensing Molecule Farnesol. Antimicrob. Agents Chemother. 2017, 61, e01573-17. [Google Scholar] [CrossRef]
- de Carvalho Dias, K.; Barbugli, P.A.; de Patto, F.; Lordello, V.B.; de Aquino Penteado, L.; Medeiros, A.I.; Vergani, C.E. Soluble factors from biofilm of Candida albicans and Staphylococcus aureus promote cell death and inflammatory response. BMC Microbiol. 2017, 17, 146. [Google Scholar] [CrossRef]
- Haiko, J.; Saeedi, B.; Bagger, G.; Karpati, F.; Özenci, V. Coexistence of Candida species and bacteria in patients with cystic fibrosis. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 1071–1077. [Google Scholar] [CrossRef]
- Uppuluri, P.; Lin, L.; Alqarihi, A.; Luo, G.; Youssef, E.G.; Alkhazraji, S.; Yount, N.Y.; Ibrahim, B.A.; Bolaris, M.A.; Edwards, J.E.; et al. The Hyr1 protein from the fungus Candida albicans is a cross kingdom immunotherapeutic target for Acinetobacter bacterial infection. PLoS Pathog. 2018, 14, e1007056. [Google Scholar] [CrossRef] [PubMed]
- Charlet, R.; Bortolus, C.; Barbet, M.; Sendid, B.; Jawhara, S. A decrease in anaerobic bacteria promotes Candida glabrata overgrowth while β-glucan treatment restores the gut microbiota and attenuates colitis. Gut Pathog. 2018, 10, 50. [Google Scholar] [CrossRef] [PubMed]
- Valentine, M.; Benadé, E.; Mouton, M.; Khan, W.; Botha, A. Binary interactions between the yeast Candida albicans and two gut-associated Bacteroides species. Microb. Pathog. 2019, 135, 103619. [Google Scholar] [CrossRef] [PubMed]
- Hoarau, G.; Mukherjee, P.K.; Gower-Rousseau, C.; Hager, C.; Chandra, J.; Retuerto, M.A.; Neut, C.; Vermeire, S.; Clemente, J.; Colombel, J.F.; et al. Bacteriome and Mycobiome Interactions Underscore Microbial Dysbiosis in Familial Crohn’s Disease. MBio 2016, 7, e01250-16. [Google Scholar] [CrossRef] [PubMed]
- Junka, A.; Szymczyk, P.; Ziółkowski, G.; Karuga-Kuzniewska, E.; Smutnicka, D.; Bil-Lula, I.; Bartoszewicz, M.; Mahabady, S.; Sedghizadeh, P.P. Bad to the Bone: On In Vitro and Ex Vivo Microbial Biofilm Ability to Directly Destroy Colonized Bone Surfaces without Participation of Host Immunity or Osteoclastogenesis. PLoS ONE 2017, 12, e0169565. [Google Scholar] [CrossRef] [PubMed]
- Deusenbery, C.B.; Kalan, L.; Meisel, J.S.; Gardner, S.E.; Grice, E.; Spiller, K.L. Human macrophage response to microbial supernatants from diabetic foot ulcers. Wound Repair Regen. 2019. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.A.; Tamer, T.M.; Rageh, A.A.; Abou-Zeid, A.M.; Abd El-Zaher, E.H.F.; Kenawy, E.-R. Insight into multidrug-resistant microorganisms from microbial infected diabetic foot ulcers. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 1261–1270. [Google Scholar] [CrossRef] [PubMed]
- Meto, A.; Colombari, B.; Sala, A.; Pericolini, E.; Meto, A.; Peppoloni, S.; Blasi, E. Antimicrobial and antibiofilm efficacy of a copper/calcium hydroxide-based endodontic paste against Staphylococcus aureus, Pseudomonas aeruginosa and Candida albicans. Dent. Mater. J. 2019, 38, 591–603. [Google Scholar] [CrossRef]
- Perinelli, D.R.; Petrelli, D.; Vitali, L.A.; Bonacucina, G.; Cespi, M.; Vllasaliu, D.; Giorgioni, G.; Palmieri, G.F. Quaternary Ammonium Leucine-Based Surfactants: The Effect of a Benzyl Group on Physicochemical Properties and Antimicrobial Activity. Pharmaceutics 2019, 11, 287. [Google Scholar] [CrossRef]
- Pourhajibagher, M.; Ghorbanzadeh, R.; Bahador, A. Antimicrobial properties of acrylic resins doped with Undaria pinnatifida exposed to light-emitting diode: In silico and in vitro assessments on multispecies biofilm-producing microbiota. Photodiagnosis Photodyn. Ther. 2019, 27, 210–215. [Google Scholar] [CrossRef]
- Ardizzoni, A.; Pericolini, E.; Paulone, S.; Orsi, C.F.; Castagnoli, A.; Oliva, I.; Strozzi, E.; Blasi, E. In vitro effects of commercial mouthwashes on several virulence traits of Candida albicans, viridans streptococci and Enterococcus faecalis colonizing the oral cavity. PLoS ONE 2018, 13, e0207262. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Leonhard, M.; Moser, D.; Ma, S.; Schneider-Stickler, B. Antibiofilm efficacy of curcumin in combination with 2-aminobenzimidazole against single- and mixed-species biofilms of Candida albicans and Staphylococcus aureus. Colloids Surfaces B Biointerfaces 2019, 174, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Bucci, A.R.; Marcelino, L.; Mendes, R.K.; Etchegaray, A. The antimicrobial and antiadhesion activities of micellar solutions of surfactin, {CTAB} and {CPCl} with terpinen-4-ol: Applications to control oral pathogens. World J. Microbiol. Biotechnol. 2018, 34, 86. [Google Scholar] [CrossRef] [PubMed]
- Medvedec Mikić, I.; Cigić, L.; Kero, D.; Kalibović Govorko, D.; Prpić Mehičić, G.; Tambić Andrašević, A.; Simeon, P. Antimicrobial effectiveness of polyhexamethylene biguanide on Enterococcus faecalis, Staphylococcus epidermidis and Candida albicans. Med. Glas. 2018, 15, 132–138. [Google Scholar]
- Kim, D.; Liu, Y.; Benhamou, R.I.; Sanchez, H.; Simón-Soro, Á.; Li, Y.; Hwang, G.; Fridman, M.; Andes, D.R.; Koo, H. Bacterial-derived exopolysaccharides enhance antifungal drug tolerance in a cross-kingdom oral biofilm. ISME J. 2018, 12, 1427–1442. [Google Scholar] [CrossRef] [PubMed]
- do Rosário Palma, A.L.; de Paula-Ramos, L.; Domingues, N.; Back-Brito, G.N.; de Oliveira, L.D.; Pereira, C.A.; Jorge, A.O.C. Biofilms of Candida albicans and Streptococcus sanguinis and their susceptibility to antimicrobial effects of photodynamic inactivation. Photodiagn. Photodyn. Ther. 2018, 24, 95–101. [Google Scholar] [CrossRef]
- Diogo, P.; Fernandes, C.; Caramelo, F.; Mota, M.; Miranda, I.M.; Faustino, M.A.F.; Neves, M.G.P.M.S.; Uliana, M.P.; de Oliveira, K.T.; Santos, J.M.; et al. Antimicrobial Photodynamic Therapy against Endodontic Enterococcus faecalis and Candida albicans Mono and Mixed Biofilms in the Presence of Photosensitizers: A Comparative Study with Classical Endodontic Irrigants. Front. Microbiol. 2017, 8, 498. [Google Scholar] [CrossRef]
- Yassin, S.A.; German, M.J.; Rolland, S.L.; Rickard, A.H.; Jakubovics, N.S. Inhibition of multispecies biofilms by a fluoride-releasing dental prosthesis copolymer. J. Dent. 2016, 48, 62–70. [Google Scholar] [CrossRef]
- Shehatou, C.; Logunov, S.L.; Dunman, P.M.; Haidaris, C.G.; Klubben, W.S. Characterizing the Antimicrobial Properties of 405 nm Light and the Corning Light-Diffusing Fiber Delivery System. Lasers Surg. Med. 2019, 51, lsm.23132. [Google Scholar] [CrossRef]
- Bucki, R.; Niemirowicz-Laskowska, K.; Deptuła, P.; Wilczewska, A.Z.; Misiak, P.; Durnaś, B.; Fiedoruk, K.; Piktel, E.; Mystkowska, J.; Janmey, P.A. Susceptibility of microbial cells to the modified PIP2-binding sequence of gelsolin anchored on the surface of magnetic nanoparticles. J. Nanobiotechnol. 2019, 17, 81. [Google Scholar] [CrossRef]
- Scala, A.; Piperno, A.; Hada, A.; Astilean, S.; Vulpoi, A.; Ginestra, G.; Marino, A.; Nostro, A.; Zammuto, V.; Gugliandolo, C. Marine Bacterial Exopolymers-Mediated Green Synthesis of Noble Metal Nanoparticles with Antimicrobial Properties. Polymers 2019, 11, 1157. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, Y.; Ishikawa, T.; Takahashi, C.; Masuda, M. A Short Peptide Derived from the ZorO Toxin Functions as an Effective Antimicrobial. Toxins 2019, 11, 392. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.O.; Graham, C.E.; Cruz, M.R.; Singh, K.V.; Murray, B.E.; Lorenz, M.C.; Garsin, D.A. Antifungal Activity of the Enterococcus faecalis Peptide EntV Requires Protease Cleavage and Disulfide Bond Formation. MBio 2019, 10, e01334-19. [Google Scholar] [CrossRef] [PubMed]
- Feldman, M.; Shenderovich, J.; Lavy, E.; Friedman, M.; Steinberg, D. A Sustained-Release Membrane of Thiazolidinedione-8: Effect on Formation of a Candida/Bacteria Mixed Biofilm on Hydroxyapatite in a Continuous Flow Model. BioMed Res. Int. 2017, 2017, 3510124. [Google Scholar] [CrossRef] [PubMed]
- Raorane, C.J.; Lee, J.-H.; Kim, Y.-G.; Rajasekharan, S.K.; García-Contreras, R.; Lee, J. Antibiofilm and Antivirulence Efficacies of Flavonoids and Curcumin Against Acinetobacter baumannii. Front. Microbiol. 2019, 10, 990. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Zou, L.; Wu, J.; Liu, H.; Luo, T.; Zhou, X.; Li, W.; Ren, B. Voriconazole inhibits cross-kingdom interactions between Candida albicans and Actinomyces viscosus through the ergosterol pathway. Int. J. Antimicrob. Agents 2019, 53, 805–813. [Google Scholar] [CrossRef]
- Jafri, H.; Khan, M.S.A.; Ahmad, I. In vitro efficacy of eugenol in inhibiting single and mixed-biofilms of drug-resistant strains of Candida albicans and Streptococcus mutans. Phytomedicine 2019, 54, 206–213. [Google Scholar] [CrossRef]
- de Alteriis, E.; Lombardi, L.; Falanga, A.; Napolano, M.; Galdiero, S.; Siciliano, A.; Carotenuto, R.; Guida, M.; Galdiero, E. Polymicrobial antibiofilm activity of the membranotropic peptide gH625 and its analogue. Microb. Pathog. 2018, 125, 189–195. [Google Scholar] [CrossRef]
- Elshinawy, M.I.; Al-Madboly, L.A.; Ghoneim, W.M.; El-Deeb, N.M. Synergistic Effect of Newly Introduced Root Canal Medicaments-Ozonated Olive Oil and Chitosan Nanoparticles, Against Persistent Endodontic Pathogens. Front. Microbiol. 2018, 9, 1371. [Google Scholar] [CrossRef]
- Rogiers, O.; Holtappels, M.; Siala, W.; Lamkanfi, M.; Van Bambeke, F.; Lagrou, K.; Van Dijck, P.; Kucharíková, S. Anidulafungin increases the antibacterial activity of tigecycline in polymicrobial Candida albicans/Staphylococcus aureus biofilms on intraperitoneally implanted foreign bodies. J. Antimicrob. Chemother. 2018, 73, 2806–2814. [Google Scholar] [CrossRef]
- Waite, R.D.; Stewart, J.E.; Stephen, A.S.; Allaker, R.P. Activity of a nitric oxide-generating wound treatment system against wound pathogen biofilms. Int. J. Antimicrob. Agents 2018, 52, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Scaffaro, R.; Lopresti, F.; D’Arrigo, M.; Marino, A.; Nostro, A. Efficacy of poly(lactic acid)/carvacrol electrospun membranes against Staphylococcus aureus and Candida albicans in single and mixed cultures. Appl. Microbiol. Biotechnol. 2018, 102, 4171–4181. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, D.R.; Arias, L.S.; Fernandes, R.A.; Straioto, F.G.; Barros Barbosa, D.; Pessan, J.P.; Delbem, A.C.B. Role of tyrosol on Candida albicans, Candida glabrata and Streptococcus mutans biofilms developed on different surfaces. Am. J. Dent. 2017, 30, 35–39. [Google Scholar] [PubMed]
- Arias, L.S.; Delbem, A.C.B.; Fernandes, R.A.; Barbosa, D.B.; Monteiro, D.R. Activity of tyrosol against single and mixed-species oral biofilms. J. Appl. Microbiol. 2016, 120, 1240–1249. [Google Scholar] [CrossRef] [PubMed]
- Hassan Abdel-Rhman, S.; Mostafa El-Mahdy, A.; El-Mowafy, M. Effect of Tyrosol and Farnesol on Virulence and Antibiotic Resistance of Clinical Isolates of Pseudomonas aeruginosa. BioMed Res. Int. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.A.; Monteiro, D.R.; Arias, L.S.; Fernandes, G.L.; Delbem, A.C.B.; Barbosa, D.B. Biofilm formation by Candida albicans and Streptococcus mutans in the presence of farnesol: A quantitative evaluation. Biofouling 2016, 32, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Leonhard, M.; Moser, D.; Ma, S.; Schneider-Stickler, B. Long-term antibiofilm activity of carboxymethyl chitosan on mixed biofilm on silicone. Laryngoscope 2016, 126, E404–E408. [Google Scholar] [CrossRef]
- Tan, Y.; Leonhard, M.; Moser, D.; Ma, S.; Schneider-Stickler, B. Inhibition of mixed fungal and bacterial biofilms on silicone by carboxymethyl chitosan. Colloids Surfaces B Biointerfaces 2016, 148, 193–199. [Google Scholar] [CrossRef]
- Tan, Y.; Leonhard, M.; Ma, S.; Moser, D.; Schneider-Stickler, B. Efficacy of carboxymethyl chitosan against Candida tropicalis and Staphylococcus epidermidis monomicrobial and polymicrobial biofilms. Int. J. Biol. Macromol. 2018, 110, 150–156. [Google Scholar] [CrossRef]
- Tan, Y.; Leonhard, M.; Moser, D.; Schneider-Stickler, B. Inhibition activity of Lactobacilli supernatant against fungal-bacterial multispecies biofilms on silicone. Microb. Pathog. 2017, 113, 197–201. [Google Scholar] [CrossRef]
- Budzyńska, A.; Różalska, S.; Sadowska, B.; Różalska, B. Candida albicans/Staphylococcus aureus Dual-Species Biofilm as a Target for the Combination of Essential Oils and Fluconazole or Mupirocin. Mycopathologia 2017, 182, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Pekmezovic, M.; Aleksic, I.; Barac, A.; Arsic-Arsenijevic, V.; Vasiljevic, B.; Nikodinovic-Runic, J.; Senerovic, L. Prevention of polymicrobial biofilms composed of Pseudomonas aeruginosa and pathogenic fungi by essential oils from selected Citrus species. Pathog. Dis. 2016, 74, ftw102. [Google Scholar] [CrossRef] [PubMed]
- Soliman, S.S.M.; Semreen, M.H.; El-Keblawy, A.A.; Abdullah, A.; Uppuluri, P.; Ibrahim, A.S. Assessment of herbal drugs for promising anti-Candida activity. BMC Complement. Altern. Med. 2017, 17, 257. [Google Scholar] [CrossRef] [PubMed]
- Peeters, E.; Hooyberghs, G.; Robijns, S.; Waldrant, K.; De Weerdt, A.; Delattin, N.; Liebens, V.; Kucharíková, S.; Tournu, H.; Verstraeten, N.; et al. Modulation of the Substitution Pattern of 5-Aryl-2-Aminoimidazoles Allows Fine-Tuning of Their Antibiofilm Activity Spectrum and Toxicity. Antimicrob. Agents Chemother. 2016, 60, 6483–6497. [Google Scholar] [CrossRef]
- Lown, L.; Peters, B.M.; Walraven, C.J.; Noverr, M.C.; Lee, S.A. An Optimized Lock Solution Containing Micafungin, Ethanol and Doxycycline Inhibits Candida albicans and Mixed C. albicans—Staphylococcus aureus Biofilms. PLoS ONE 2016, 11, e0159225. [Google Scholar] [CrossRef]
- Qu, Y.; Locock, K.; Verma-Gaur, J.; Hay, I.D.; Meagher, L.; Traven, A. Searching for new strategies against polymicrobial biofilm infections: Guanylated polymethacrylates kill mixed fungal/bacterial biofilms. J. Antimicrob. Chemother. 2016, 71, 413–421. [Google Scholar] [CrossRef]
- Ariani, N.; Visser, A.; Teulings, M.R.I.M.; Dijk, M.; Rahardjo, T.B.W.; Vissink, A.; van der Mei, H.C. Efficacy of cleansing agents in killing microorganisms in mixed species biofilms present on silicone facial prostheses—An in vitro study. Clin. Oral Investig. 2015, 19, 2285–2293. [Google Scholar] [CrossRef]
- Ganske, K.; Wiegand, C.; Hipler, U.-C.; Heinze, T. Synthesis of Novel Cellulose Carbamates Possessing Terminal Amino Groups and Their Bioactivity. Macromol. Biosci. 2016, 16, 451–461. [Google Scholar] [CrossRef]
- Zielińska, S.; Wójciak-Kosior, M.; Dziągwa-Becker, M.; Gleńsk, M.; Sowa, I.; Fijałkowski, K.; Rurańska-Smutnicka, D.; Matkowski, A.; Junka, A. The Activity of Isoquinoline Alkaloids and Extracts from Chelidonium majus against Pathogenic Bacteria and Candida sp. Toxins 2019, 11, 406. [Google Scholar] [CrossRef]
- Olejar, K.J.; Ricci, A.; Swift, S.; Zujovic, Z.; Gordon, K.C.; Fedrizzi, B.; Versari, A.; Kilmartin, P.A. Characterization of an Antioxidant and Antimicrobial Extract from Cool Climate, White Grape Marc. Antioxidants 2019, 8, 232. [Google Scholar] [CrossRef]
- Rodrigues, C.; Silva, S.; Azeredo, J.; Henriques, M. Detection and Quantification of Fluconazole Within Candida glabrata Biofilms. Mycopathologia 2015, 179, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Kagan, S.; Jabbour, A.; Sionov, E.; Alquntar, A.A.; Steinberg, D.; Srebnik, M.; Nir-Paz, R.; Weiss, A.; Polacheck, I. Anti-Candida albicans biofilm effect of novel heterocyclic compounds. J. Antimicrob. Chemother. 2014, 69, 416–427. [Google Scholar] [CrossRef] [PubMed]
- Feldman, M.; Al-Quntar, A.; Polacheck, I.; Friedman, M.; Steinberg, D. Therapeutic Potential of Thiazolidinedione-8 as an Antibiofilm Agent against Candida albicans. PLoS ONE 2014, 9, e93225. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Leonhard, M.; Moser, D.; Ma, S.; Schneider-Stickler, B. Inhibitory effect of probiotic lactobacilli supernatants on single and mixed non- albicans Candida species biofilm. Arch. Oral Biol. 2018, 85, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Rocha, G.R.; Salamanca, E.J.F.; de Barros, A.L.; Lobo, C.I.V.; Klein, M.I. Effect of tt-farnesol and myricetin on in vitro biofilm formed by Streptococcus mutans and Candida albicans. BMC Complementary Altern. Med. 2018, 18, 61. [Google Scholar] [CrossRef]
- Černáková, L.; Jordao, L.; Bujdáková, H. Impact of Farnesol and Corsodyl ® on Candida albicans Forming Dual Biofilm With Streptococcus mutans. Oral Dis. 2018, 24, 6–9. [Google Scholar]
- Cateau, E.; Rodier, M.-H.; Imbert, C. In vitro efficacies of caspofungin or micafungin catheter lock solutions on Candida albicans biofilm growth. J. Antimicrob. Chemother. 2008, 62, 153–155. [Google Scholar] [CrossRef]
- Visek, J.; Ryskova, L.; Safranek, R.; Lasticova, M.; Blaha, V. In vitro comparison of efficacy of catheter locks in the treatment of catheter related blood stream infection. Clin. Nutr. ESPEN 2019, 30, 107–112. [Google Scholar] [CrossRef]
- Chandra, J.; Long, L.; Isham, N.; Mukherjee, P.K.; DiSciullo, G.; Appelt, K.; Ghannoum, M.A. In Vitro and In Vivo Activity of a Novel Catheter Lock Solution against Bacterial and Fungal Biofilms. Antimicrob. Agents Chemother. 2018, 62, e00722-18. [Google Scholar] [CrossRef]
| Mixed Candida spp./Bacteria Biofilm | Therapy | Activities | Reference(s) |
|---|---|---|---|
| Candida albicans, Staphylococcus aureus, Pseudomonas aeruginosa | Cu/CaOH2-based endodontic paste | Antimicrobial Antibiofilm | [129] |
| Candida albicans, Staphylococcus aureus, Enterococcus spp., Escherichia coli, Pseudomonas aeruginosa | Quaternary ammonium amphiphiles (derivatives of leucine esters: C10, C12 and C14) | Antimicrobial | [130] |
| Streptococcus mutans, Streptococcus sanguinis, Lactobacillus acidophilus, Candida albicans | Acrylic resin containing U. pinnatifida, ensuing photo-activation using LED | Antimicrobial | [131] |
| Streptococcus mutans, viridans streptococci, Streptococcus salivarius, Candida albicans | Alcohol-free commercial mouthwashes with chlorhexidine digluconate, fluoride and cetylpyridinium chloride | Antimicrobial Antibiofilm | [132] |
| Candida albicans, Staphylococcus aureus | Curcumin and 2-aminobenzimidazole | Antimicrobial Antibiofilm | [133] |
| Streptococcus mutans, Candida albicans | Micellar solutions of surfactants (cetylpyridinium chloride and cetyltrimethylammonium bromide and sufactin) and terpinen-4-ol (TP) (a plant natural product) was studied. | Antimicrobial | [134] |
| Enterococcus faecalis, Candida albicans and Streptococcus epidermidis | 0.2% polyhexamethilene biguanide (PHMB) | Antimicrobial Antibiofilm | [135] |
| Candida albicans, Streptococcus mutans | Association of topical antifungal fluconazole and povidone iodine | Antimicrobial Antibiofilm | [136] |
| Candida albicans and Streptococcus sanguinis | Photodynamic inactivation (PDI) | Antimicrobial Antibiofilm | [137] |
| Enterococcus faecalis and Candida albicans | Photodynamic therapy (aPDT) with the Zn(II)chlorin e6 methyl ester (Zn(II)e6Me) activated by red light | Antimicrobial Antibiofilm | [138] |
| Candida albicans, Lactobacillus casei, and Streptococcus mutans | Fluoride-releasing copolymer, constituted by methyl methacrylate (MMA) and 2-hydroxyethyl methacrylate (HEMA) with polymethyl methacrylate (PMMA) | Antimicrobial Antibiofilm | [139] |
| ESKAPE and Staphylococcus epidermidis, Streptococcus pyogenes, Candida albicans, Escherichia coli | Corning® light-diffusing fiber (LDF) | Antimicrobial | [140] |
| Staphylococcus aureus MRSA (Xen 30), Pseudomonas aeruginosa (Xen 5) and Candida spp. | Magnetic nanoparticles and PBP10 (peptide) | Antimicrobial | [141] |
| Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans | Marine bacterial exopolymers-Mediated green synthesis of noble metal nanoparticles | Antimicrobial | [142] |
| Staphylococcus aureus, Bacillus subtilis, and Candida albicans | Peptide derived from the ZorO E. coli toxin | Antimicrobial | [143] |
| Gram-positive bacteria and Candida albicans | EntV (bacteriocin) | Antibiofilm | [144] |
| Candida albicans and Streptococcus mutans | Derivative thiazolidinedione-8 (S-8), in solution or incorporated into a sustained-release membrane (SRM-S-8) | Antimicrobial Antibiofilm | [145] |
| Candida albicans and Acinetobacter baumannii | Fisetin, phloretin and curcumin (flavonoids) | Antibiofilm Antivirulence | [146] |
| Candida albicans and Actinomyces viscosus | Voriconazole | Inhibition of cross-kingdom interactions | [147] |
| Candida albicans and Streptococcus mutans | Eugenol | Antibiofilm | [148] |
| Candida tropicalis-Serratia marcescens, and Candida tropicalis-Staphylococcus aureus | gH625-GCGKKKK (derivative of the membranotropic peptide gH625) | Antiadhesion Antibiofilm | [149] |
| Enterococcus faecalis, Streptococcus mutans, and Candida albicans | Chitosan (Ch-NPs), silver Nanoparticles (Ag-NPs), ozonated olive oil (O3-oil), single or combined | Antiadhesion Antibiofilm | [150] |
| Candida albicans-Staphylococcus aureus | Anidulafungin | Rise of the antibacterial activity of tigecycline, (synergistic effect) Reduction of S. aureus poly-β-(1,6)-N-acetylglucosamine | [151] |
| Staphylococcus aureus, Pseudomonas aeruginosa, Acinetobacter baumannii, Escherichia coli, and Candida spp. | Two-layer nitric oxide-generating system (NOx) | Antimicrobial | [152] |
| Staphylococcus aureus and Candida albicans | Electrospun membranes of poly(lactic acid) and carvacrol | Antimicrobial Antibiofilm | [153] |
| Candida spp. and Streptococcus mutans | Tyrosol | Reduction of the metabolic activity | [154] |
| Candida albicans (ATCC 10231), Candida glabrata (ATCC 90030) and Streptococcus mutans (ATCC 25175) | Tyrosol | Antibiofilm | [155] |
| Pseudomonas aeruginosa-Candida albicans | Tyrosol and tyrosol + farnesol | Tyrosol: blockage of the production of hemolysin and protease in P. aeruginosa Farnesol: slight blockage of the production of hemolysin in P. aeruginosa | [156] |
| Candida albicans and Streptococcus mutans | Farnesol | Antibiofilm | [157] |
| Candida albicans, Candida tropicalis, Lactobacillus gasseri, Streptococcus salivarius, Rothia dentocariosa, and Staphylococcus epidermidis | Carboxymethyl chitosan | Antibiofilm Antiadhesion Inhibition of Candida spp. yeast-to-hyphal transition | [158,159,160] |
| Several fungal–bacterial multispecies | Lactobacilli supernatant | Antibiofilm Antiadhesion Antimicrobial Inhibition of Candida spp. yeast-to-hyphal transition Reduction of the metabolic activity | [161] |
| Pseudomonas aeruginosa, Candida albicans, Staphylococcus aureus | Combination geranium, citronella and clove (essential oils) and fluconazole or mupirocin. | Inhibition of fungal growth Antimicrobial Disturbance of quorum sensing | [162] |
| Pseudomonas aeruginosa, and Candida albicans | Pompia and grapefruit essential oils | Antimicrobial Antibiofilm | [163] |
| Candida albicans, Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Acinetobacter baumannii, and Klebsiella pneumoniae | Portulaca oleracea (Baq’lah), Lawsania inermis (Henna) ethanol extracts | Antimicrobial | [164] |
| Gram-positive and Candida albicans | N1- and 2N-substituted 5-aryl-2-aminoimidazoles | Antiadhesion Antimicrobial | [165] |
| Candida albicans, Staphylococcus aureus | lock solution with micafungin, ethanol and doxycycline | Moderatly antibacterial Antibiofilm | [166] |
| Candida albicans, Staphylococcus aureus | Guanylated polymethacrylates with or without drug combinations | Antimicrobial Antibiofilm | [167] |
| Staphylococcus epidermidis (MFP5-5), Staphylococcus xylosus (MFP28-3), Candida albicans (MFP8), Candida parapsilosis (MFP16-2), Candida famata (MFP29-1) | Antibacterial soap, essential-oil-containing mouth rinse, ethanol 27%, chlorhexidine mouth rinse, and buttermilk | Antimicrobial | [168] |
| Candida albicans, Staphylococcus aureus, Klebsiella pneumoniae | Novel cellulose carbamates (e.g., ω-aminoethylcellulose carbamate) with or without p-amino-benzylamine | Antimicrobial | [169] |
| Candida albicans, Staphylococcus aureus, Pseudomonas aeruginosa | Extracts of Chelidonium majus (alkaloid: chelerythrine and chelidonine) single or in combination | Antimicrobial Antibiofilm | [170] |
| Staphylococcus aureus (6538), Escherichia coli (25922), Candida albicans | Phenolic compounds from winery waste (monomeric and tannin polyphenols) | Antimicrobial | [171] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigues, M.E.; Gomes, F.; Rodrigues, C.F. Candida spp./Bacteria Mixed Biofilms. J. Fungi 2020, 6, 5. https://doi.org/10.3390/jof6010005
Rodrigues ME, Gomes F, Rodrigues CF. Candida spp./Bacteria Mixed Biofilms. Journal of Fungi. 2020; 6(1):5. https://doi.org/10.3390/jof6010005
Chicago/Turabian StyleRodrigues, Maria Elisa, Fernanda Gomes, and Célia F. Rodrigues. 2020. "Candida spp./Bacteria Mixed Biofilms" Journal of Fungi 6, no. 1: 5. https://doi.org/10.3390/jof6010005
APA StyleRodrigues, M. E., Gomes, F., & Rodrigues, C. F. (2020). Candida spp./Bacteria Mixed Biofilms. Journal of Fungi, 6(1), 5. https://doi.org/10.3390/jof6010005






