Regulation of Candida albicans Hyphal Morphogenesis by Endogenous Signals
Abstract
1. Introduction
2. The Role of Hyphal Morphogenesis in the Virulence of C. albicans
3. Mechanism of Hyphal Morphogenesis
4. Induction of the Hyphal Morphogenesis of C. albicans by External Stimuli
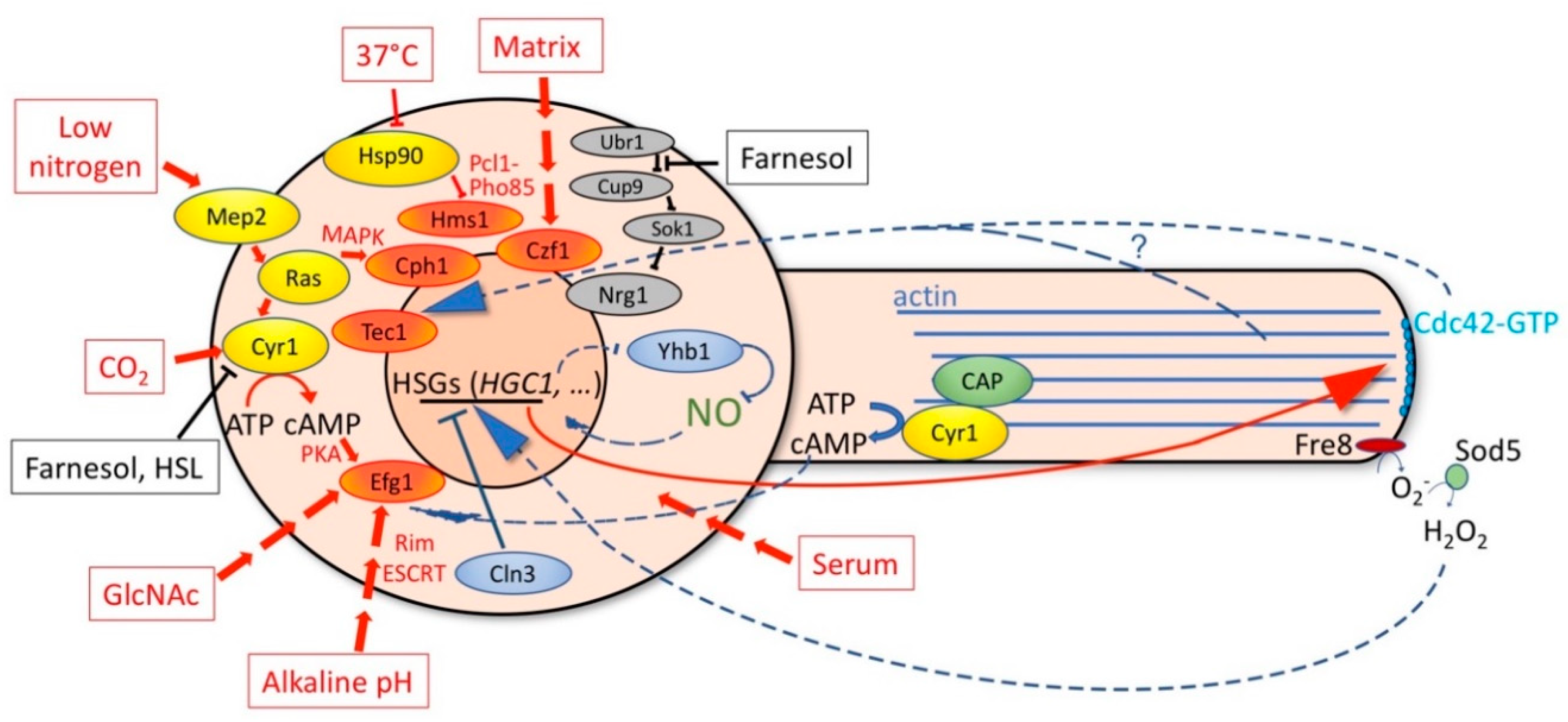
5. Regulation of Hyphal Morphogenesis and HSG Expression by Internal Signals
5.1. Interference with Yeast Proliferation Induces Hyphal Morphogenesis and HSG Expression
5.2. Actin Depolymerization Blocks Hyphal Morphogenesis and Inhibits HSG Expression
5.3. Inhibition of Endocytosis Inhibits Hyphal Morphogenesis and HSG Expression
6. Feedback Regulations in Hyphal Morphogenesis
6.1. Positive Feedback Regulations of Hyphal Transcription
6.2. Cdc42
6.3. Reactive Oxygen and Nitrogen Species
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Nash, A.K.; Auchtung, T.A.; Wong, M.C.; Smith, D.P.; Gesell, J.R.; Ross, M.C.; Stewart, C.J.; Metcalf, G.A.; Muzny, D.M.; Gibbs, R.A.; et al. The gut mycobiome of the Human Microbiome Project healthy cohort. Microbiome 2017, 5, 153. [Google Scholar] [CrossRef] [PubMed]
- Ghannoum, M.A.; Jurevic, R.J.; Mukherjee, P.K.; Cui, F.; Sikaroodi, M.; Naqvi, A.; Gillevet, P.M. Characterization of the Oral Fungal Microbiome (Mycobiome) in Healthy Individuals. PLoS Pathog. 2010, 6, e1000713. [Google Scholar] [CrossRef] [PubMed]
- Kam, A.P.; Xu, J. Diversity of commensal yeasts within and among healthy hosts. Diagn. Microbiol. Infect. Dis. 2002, 43, 19–28. [Google Scholar] [CrossRef]
- Calderone, R.A.; Clancy, C.J. Candida and Candidiasis, 2nd ed.; Calderone, R.A., Clancy, C.J., Eds.; ASM Press: Washington, DC, USA, 2012; ISBN 9781555815394. [Google Scholar]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of invasive candidiasis: A persistent public health problem. Clin. Microbiol. Rev. 2007, 20, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of invasive mycoses in North America. Crit. Rev. Microbiol. 2010, 36, 1–53. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Roussos, N.; Vardakas, K.Z. Relative frequency of albicans and the various non-albicans Candida spp among candidemia isolates from inpatients in various parts of the world: A systematic review. Int. J. Infect. Dis. 2010, 14, e954–e966. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.; Neofytos, D.; Diekema, D.; Azie, N.; Meier-Kriesche, H.U.; Quan, S.P.; Horn, D. Epidemiology and outcomes of candidemia in 3648 patients: Data from the Prospective Antifungal Therapy (PATH Alliance(R)) registry, 2004-2008. Diagn. Microbiol. Infect. Dis. 2012, 74, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Kullberg, B.J.; Arendrup, M.C. Invasive Candidiasis. N. Engl. J. Med. 2015, 373, 1445–1456. [Google Scholar] [CrossRef] [PubMed]
- Gudlaugsson, O.; Gillespie, S.; Lee, K.; Vande Berg, J.; Hu, J.; Messer, S.; Herwaldt, L.; Pfaller, M.; Diekema, D. Attributable mortality of nosocomial candidemia, revisited. Clin. Infect. Dis. 2003, 37, 1172–1177. [Google Scholar] [CrossRef] [PubMed]
- Hassan, I.; Powell, G.; Sidhu, M.; Hart, W.M.; Denning, D.W. Excess mortality, length of stay and cost attributable to Candidaemia. J. Infect. 2009, 59, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Mora-Duarte, J.; Betts, R.; Rotstein, C.; Colombo, A.L.; Thompson-Moya, L.; Smietana, J.; Lupinacci, R.; Sable, C.; Kartsonis, N.; Perfect, J. Comparison of caspofungin and amphotericin B for invasive candidiasis. N. Engl. J. Med. 2002, 347, 2020–2029. [Google Scholar] [CrossRef] [PubMed]
- Cuervo, G.; Garcia-Vidal, C.; Nucci, M.; Puchades, F.; Fernández-Ruiz, M.; Obed, M.; Manzur, A.; Gudiol, C.; Pemán, J.; Aguado, J.M.; et al. Breakthrough Candidaemia in the era of broad-spectrum antifungal therapies. Clin. Microbiol. Infect. 2016, 22, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Pappas, P.G.; Kauffman, C.A.; Andes, D.R.; Clancy, C.J.; Marr, K.A.; Ostrosky-Zeichner, L.; Reboli, A.C.; Schuster, M.G.; Vazquez, J.A.; Walsh, T.J.; et al. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2015, 62, civ933. [Google Scholar] [CrossRef] [PubMed]
- Bongomin, F.; Gago, S.; Oladele, R.; Denning, D. Global and Multi-National Prevalence of Fungal Diseases—Estimate Precision. J. Fungi 2017, 3, 57. [Google Scholar] [CrossRef] [PubMed]
- Sudbery, P.; Gow, N.; Berman, J. The distinct morphogenic states of Candida albicans. Trends Microbiol. 2004, 12, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Sharma, J.; Rosiana, S.; Razzaq, I.; Shapiro, R. Linking Cellular Morphogenesis with Antifungal Treatment and Susceptibility in Candida Pathogens. J. Fungi 2019, 5, 17. [Google Scholar] [CrossRef] [PubMed]
- Stokes, C.; Moran, G.P.; Spiering, M.J.; Cole, G.T.; Coleman, D.C.; Sullivan, D.J. Lower filamentation rates of Candida dubliniensis contribute to its lower virulence in comparison with Candida albicans. Fungal Genet. Biol. 2007, 44, 920–931. [Google Scholar] [CrossRef] [PubMed]
- Lackey, E.; Vipulanandan, G.; Childers, D.S.; Kadosh, D. Comparative Evolution of Morphological Regulatory Functions in Candida Species. Eukaryot. Cell 2013, 12, 1356–1368. [Google Scholar] [CrossRef] [PubMed]
- Yue, H.; Bing, J.; Zheng, Q.; Zhang, Y.; Hu, T.; Du, H.; Wang, H.; Huang, G. Filamentation in Candida auris, an emerging fungal pathogen of humans: Passage through the mammalian body induces a heritable phenotypic switch. Emerg. Microbes Infect. 2018, 7, 188. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.A.; Cottier, F.; Muhlschlegel, F.A. Molecular networks in the fungal pathogen Candida albicans. Adv. Appl. Microbiol. 2009, 67, 191–212. [Google Scholar] [CrossRef] [PubMed]
- Sudbery, P.E. Growth of Candida albicans hyphae. Nat. Rev. Microbiol. 2011, 9, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Richardson, M.D.; Warnock, D.W. Fungal Infection–Diagnosis and Management, 2nd ed.; Blackwell Sciences Ltd.: Oxford, UK, 1997. [Google Scholar]
- Koh, A.Y.; Kohler, J.R.; Coggshall, K.T.; Van Rooijen, N.; Pier, G.B. Mucosal damage and neutropenia are required for Candida albicans dissemination. PLoS Pathog. 2008, 4, e35. [Google Scholar] [CrossRef] [PubMed]
- Marcil, A.; Harcus, D.; Thomas, D.Y.; Whiteway, M. Candida albicans killing by RAW 264.7 mouse macrophage cells: Effects of Candida genotype, infection ratios, and gamma interferon treatment. Infect. Immun. Infect. Immun. 2002, 70, 6319–6329. [Google Scholar] [CrossRef]
- Ghosh, S.; Navarathna, D.H.M.L.P.; Roberts, D.D.; Cooper, J.T.; Atkin, A.L.; Petro, T.M.; Nickerson, K.W. Arginine-Induced germ tube formation in Candida albicans is essential for escape from murine macrophage line RAW 264.7. Infect. Immun. 2009, 77, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, C.G.; Koser, U.; Lewis, L.E.; Bain, J.M.; Mora-Montes, H.M.; Barker, R.N.; Gow, N.A.; Erwig, L.P. Contribution of Candida albicans cell wall components to recognition by and escape from murine macrophages. Infect. Immun. 2010, 78, 1650–1658. [Google Scholar] [CrossRef] [PubMed]
- Nobile, C.J.; Johnson, A.D. Candida albicans Biofilms and Human Disease. Annu. Rev. Microbiol. 2015, 69, 71–92. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Wang, Y. Hgc1, a novel hypha-specific G1 cyclin-related protein regulates Candida albicans hyphal morphogenesis. EMBO J. 2004, 23, 1845–1856. [Google Scholar] [CrossRef] [PubMed]
- Lo, H.-J.; Kohler, J.; DiDomenico, B.; Loebenberg, D.; Cacciapuoti, A.; Fink, G.R. Nonfilamentous C. albicans mutants are avirulent. Cell 1997, 90, 939–950. [Google Scholar] [CrossRef]
- Saville, S.P.; Lazzell, A.L.; Monteagudo, C.; Lopez-Ribot, J.L. Engineered control of cell morphology in vivo reveals distinct roles for yeast and filamentous forms of Candida albicans during infection. Eukaryot. Cell 2003, 2, 1053–1060. [Google Scholar] [CrossRef] [PubMed]
- Saville, S.P.; Lazzell, A.L.; Bryant, A.P.; Fretzen, A.; Monreal, A.; Solberg, E.O.; Monteagudo, C.; Lopez-Ribot, J.L.; Milne, G.T. Inhibition of filamentation can be used to treat disseminated candidiasis. Antimicrob. Agents Chemother. 2006, 50, 3312–3316. [Google Scholar] [CrossRef] [PubMed]
- Noble, S.M.; French, S.; Kohn, L.A.; Chen, V.; Johnson, A.D. Systematic screens of a Candida albicans homozygous deletion library decouple morphogenetic switching and pathogenicity. Nat. Genet. 2010, 42, 590–598. [Google Scholar] [CrossRef] [PubMed]
- O’Meara, T.R.; Veri, A.O.; Ketela, T.; Jiang, B.; Roemer, T.; Cowen, L.E. Global analysis of fungal morphology exposes mechanisms of host cell escape. Nat. Commun. 2015, 6, 6741. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, I.D.; Wilson, D.; Wachtler, B.; Brunke, S.; Naglik, J.R.; Hube, B. Candida albicans dimorphism as a therapeutic target. Expert Rev. Anti-Infect. Ther. 2012, 10, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Vila, T.; Romo, J.A.; Pierce, C.G.; McHardy, S.F.; Saville, S.P.; Lopez-Ribot, J.L. Targeting Candida albicans filamentation for antifungal drug development. Virulence 2017, 8, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Ruhnke, M. Skin and mucous membrane infections. In Candida and candidiasis; Calderone, R.A., Ed.; ASM Press: Washington, DC, USA, 2002; pp. 307–325. [Google Scholar]
- Blostein, F.; Levin-Sparenberg, E.; Wagner, J.; Foxman, B. Recurrent vulvovaginal candidiasis. Ann. Epidemiol. 2017, 27, 575–582.e3. [Google Scholar] [CrossRef] [PubMed]
- Sobel, J.D.; Muller, G.; Buckley, H.R. Critical role of germ tube formation in the pathogenesis of Candidal vaginitis. Infect. Immun. 1984, 44, 576–580. [Google Scholar] [PubMed]
- Martin, M.V.; Craig, G.T.; Lamb, D.J. An investigation of the role of true hypha production in the pathogenesis of experimental oral candidosis. Sabouraudia 1984, 22, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Hisajima, T.; Ishibashi, H.; Yamada, T.; Nishiyama, Y.; Yamaguchi, H.; Funakoshi, K.; Abe, S. Invasion process of Candida albicans to tongue surface in early stages of experimental murine oral candidiasis. Med. Mycol. 2008, 46, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Peters, B.M.; Palmer, G.E.; Nash, A.K.; Lilly, E.A.; Fidel, P.L., Jr.; Noverr, M.C. Fungal morphogenetic pathways are required for the hallmark inflammatory response during Candida albicans vaginitis. Infect. Immun. 2014, 82, 532–543. [Google Scholar] [CrossRef] [PubMed]
- Lew, D.J.; Reed, S.I. Morphogenesis in the yeast cell cycle: Regulation by Cdc28 and cyclins. J. Cell Biol. 1993, 120, 1305–1320. [Google Scholar] [CrossRef] [PubMed]
- Chiou, J.; Balasubramanian, M.K.; Lew, D.J. Cell Polarity in Yeast. Annu. Rev. Cell Dev. Biol. 2017, 33, 77–101. [Google Scholar] [CrossRef] [PubMed]
- Liu, H. Transcriptional control of dimorphism in Candida albicans. Curr. Opin. Microbiol. 2001, 4, 728–735. [Google Scholar] [CrossRef]
- Steinberg, G.; Peñalva, M.A.; Riquelme, M.; Wösten, H.A.; Harris, S.D. Cell Biology of Hyphal Growth. Microbiol. Spectr. 2017, 5, 1–34. [Google Scholar] [CrossRef]
- Riquelme, M.; Aguirre, J.; Bartnicki-García, S.; Braus, G.H.; Feldbrügge, M.; Fleig, U.; Hansberg, W.; Herrera-Estrella, A.; Kämper, J.; Kück, U.; et al. Fungal Morphogenesis, from the Polarized Growth of Hyphae to Complex Reproduction and Infection Structures. Microbiol. Mol. Biol. Rev. 2018, 82, e00068-17. [Google Scholar] [CrossRef] [PubMed]
- Bartnicki-Garcia, S.; Garduño-Rosales, M.; Delgado-Alvarez, D.L.; Mouriño-Pérez, R.R. Experimental measurement of endocytosis in fungal hyphae. Fungal Genet. Biol. 2018, 118, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Hernández-González, M.; Bravo-Plaza, I.; Pinar, M.; de Los Ríos, V.; Arst, H.N.; Peñalva, M.A. Endocytic recycling via the TGN underlies the polarized hyphal mode of life. PLoS Genet. 2018, 14, e1007291. [Google Scholar] [CrossRef] [PubMed]
- Araujo-Bazán, L.; Peñalva, M.A.; Espeso, E.A. Preferential localization of the endocytic internalization machinery to hyphal tips underlies polarization of the actin cytoskeleton in Aspergillus nidulans. Mol. Microbiol. 2008, 67, 891–905. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, K.; Kaji, H.; Nishimura, K.; Miyaji, M. The role of microfilaments and microtubules in apical growth and dimorphism of Candida albicans. J. Gen. Microbiol. 1990, 136, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Rida, P.C.G.; Nishikawa, A.; Won, G.Y.; Dean, N. Yeast-to-Hyphal Transition Triggers Formin-dependent Golgi Localization to the Growing Tip in Candida albicans. Mol. Biol. Cell 2006, 17, 4364–4378. [Google Scholar] [CrossRef] [PubMed]
- Crampin, H.; Finley, K.; Gerami-Nejad, M.; Court, H.; Gale, C.; Berman, J.; Sudbery, P. Candida albicans hyphae have a Spitzenkörper that is distinct from the polarisome found in yeast and pseudohyphae. J. Cell Sci. 2005, 118, 2935–2947. [Google Scholar] [CrossRef] [PubMed]
- Weiner, A.; Orange, F.; Lacas-Gervais, S.; Rechav, K.; Ghugtyal, V.; Bassilana, M.; Arkowitz, R.A. On-site secretory vesicle delivery drives filamentous growth in the fungal pathogen Candida albicans. Cell. Microbiol. 2018, e12963. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.A.; Sudbery, P.E. Spitzenkörper, exocyst, and polarisome components in Candida albicans hyphae show different patterns of localization and have distinct dynamic properties. Eukaryot. Cell 2010, 9, 1455–1465. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Walther, A.; Wendland, J. Ras1-induced hyphal development in Candida albicans requires the formin Bni1. Eukaryot. Cell 2005, 4, 1712–1724. [Google Scholar] [CrossRef] [PubMed]
- Li, C.R.; Wang, Y.M.; De Zheng, X.; Liang, H.Y.; Tang, J.C.W.; Wang, Y. The formin family protein CaBni1p has a role in cell polarity control during both yeast and hyphal growth in Candida albicans. J. Cell Sci. 2005, 118, 2637–2648. [Google Scholar] [CrossRef] [PubMed]
- Hazan, I.; Liu, H. Hyphal tip-associated localization of Cdc42 is F-actin dependent in Candida albicans. Eukaryot Cell 2002, 1, 856–864. [Google Scholar] [CrossRef] [PubMed]
- Ushinsky, S.C.; Harcus, D.; Ash, J.; Dignard, D.; Marcil, A.; Morchhauser, J.; Thomas, D.Y.; Whiteway, M.; Leberer, E. CDC42 is required for polarized growth in human pathogen Candida albicans. Eukaryot. Cell 2002, 1, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Arkowitz, R.A.; Bassilana, M. Regulation of hyphal morphogenesis by Ras and Rho small GTPases. Fungal Biol. Rev. 2015, 29, 7–19. [Google Scholar] [CrossRef]
- Bassilana, M.; Hopkins, J.; Arkowitz, R.A. Regulation of the Cdc42/Cdc24 GTPase module during Candida albicans hyphal growth. Eukaryot Cell 2005, 4, 588–603. [Google Scholar] [CrossRef] [PubMed]
- Court, H.; Sudbery, P. Regulation of Cdc42 GTPase Activity in the Formation of Hyphae in Candida albicans. Mol. Biol. Cell 2007, 18, 265–281. [Google Scholar] [CrossRef] [PubMed]
- Pulver, R.; Heisel, T.; Gonia, S.; Robins, R.; Norton, J.; Haynes, P.; Gale, C.A. Rsr1 focuses CDC42 activity at hyphal tips and promotes maintenance of hyphal development in Candida albicans. Eukaryot. Cell 2013, 12, 482–495. [Google Scholar] [CrossRef] [PubMed]
- Brand, A.; Vacharaksa, A.; Bendel, C.; Norton, J.; Haynes, P.; Henry-Stanley, M.; Wells, C.; Ross, K.; Gow, N.A.R.; Gale, C.A. An internal polarity landmark is important for externally induced hyphal behaviors in Candida albicans. Eukaryot. Cell 2008, 7, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Thomson, D.D.; Wehmeier, S.; Byfield, F.J.; Janmey, P.A.; Caballero-Lima, D.; Crossley, A.; Brand, A.C. Contact-induced apical asymmetry drives the thigmotropic responses of Candida albicans hyphae. Cell. Microbiol. 2015, 17, 342–354. [Google Scholar] [CrossRef] [PubMed]
- Csank, C.; Schroppel, K.; Leberer, E.; Harcus, D.; Mohamed, O.; Meloche, S.; Thomas, D.Y.; Whiteway, M. Roles of the Candida albicans mitogen-activated protein kinase homolog, Cek1p, in hyphal development and systemic candidiasis. Infect. Immun. 1998, 66, 2713–2721. [Google Scholar] [PubMed]
- Rocha, C.R.; Schroppel, K.; Harcus, D.; Marcil, A.; Dignard, D.; Taylor, B.N.; Thomas, D.Y.; Whiteway, M.; Leberer, E. Signaling through adenylyl cyclase is essential for hyphal growth and virulence in the pathogenic fungus Candida albicans. Mol. Biol. Cell 2001, 12, 3631–3643. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, M.; Castilla, R.; Bolduc, N.; Zelada, A.; Martineau, P.; Bouillon, M.; Magee, B.B.; Passeron, S.; Giasson, L.; Cantore, M.L. The two isoforms of the cAMP-dependent protein kinase catalytic subunit are involved in the control of dimorphism in the human fungal pathogen Candida albicans. Fungal Genet. Biol. 2003, 38, 133–141. [Google Scholar] [CrossRef]
- Nantel, A.; Dignard, D.; Bachewich, C.; Harcus, D.; Marcil, A.; Bouin, A.P.; Sensen, C.W.; Hogues, H.; van het Hoog, M.; Gordon, P.; et al. Transcription profiling of Candida albicans cells undergoing the yeast-to-hyphal transition. Mol. Biol. Cell 2002, 13, 3452–3465. [Google Scholar] [CrossRef] [PubMed]
- Kadosh, D.; Johnson, A.D. Induction of the Candida albicans filamentous growth program by relief of transcriptional repression: A genome-wide analysis. Mol. Biol. Cell 2005, 16, 2903–2912. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Kohler, J.; Fink, G.R. Suppression of hyphal formation in Candida albicans by mutation of a STE12 homolog. Science 1994, 266, 1723–1726. [Google Scholar] [CrossRef] [PubMed]
- Stoldt, V.R.; Sonneborn, A.; Leuker, C.E.; Ernst, J.F. Efg1p, an essential regulator of morphogenesis of the human pathogen Candida albicans, is a member of a conserved class of bHLH proteins regulating morphogenetic processes in fungi. EMBO J. 1997, 16, 1982–1991. [Google Scholar] [CrossRef] [PubMed]
- Lane, S.; Zhou, S.; Pan, T.; Dai, Q.; Liu, H. The basic helix-loop-helix transcription factor Cph2 regulates hyphal development in Candida albicans partly via TEC1. Mol Cell Biol 2001, 21, 6418–6428. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.H., Jr.; Giusani, A.D.; Chen, X.; Kumamoto, C.A. Filamentous growth of Candida albicans in response to physical environmental cues and its regulation by the unique CZF1 gene. Mol. Microbiol. 1999, 34, 651–662. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, A.; Rupp, S.; Taylor, B.N.; Rollinghoff, M.; Schroppel, K. The TEA/ATTS transcription factor CaTec1p regulates hyphal development and virulence in Candida albicans. Mol. Microbiol. 2000, 38, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Davis, D.; Edwards, J.E., Jr.; Mitchell, A.P.; Ibrahim, A.S. Candida albicans RIM101 pH response pathway is required for host-pathogen interactions. Infect Immun. 2000, 68, 5953–5959. [Google Scholar] [PubMed]
- Shapiro, R.S.; Sellam, A.; Tebbji, F.; Whiteway, M.; Nantel, A.; Cowen, L.E. Pho85, Pcl1, and Hms1 Signaling Governs Candida albicans Morphogenesis Induced by High Temperature or Hsp90 Compromise. Curr. Biol. 2012, 22, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Braun, B.R.; Johnson, A.D. Control of filament formation in Candida albicans by the transcriptional repressor TUP1. Science 1997, 277, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Murad, A.M.; Leng, P.; Straffon, M.; Wishart, J.; Macaskill, S.; MacCallum, D.; Schnell, N.; Talibi, D.; Marechal, D.; Tekaia, F.; et al. NRG1 represses yeast-hypha morphogenesis and hypha-specific gene expression in Candida albicans. EMBO J. 2001, 20, 4742–4752. [Google Scholar] [CrossRef] [PubMed]
- Cao, F.; Lane, S.; Raniga, P.P.; Lu, Y.; Zhou, Z.; Ramon, K.; Chen, J.; Liu, H. The Flo8 Transcription Factor Is Essential for Hyphal Development and Virulence in Candida albicans. Mol. Biol. Cell 2006, 17, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Guan, G.; Xie, J.; Sun, Y.; Tong, Y.; Zhang, L.; Huang, G. Roles of Candida albicans Gat2, a GATA-Type Zinc Finger Transcription Factor, in Biofilm Formation, Filamentous Growth and Virulence. PLoS ONE 2012, 7, e29707. [Google Scholar] [CrossRef] [PubMed]
- Rottmann, M.; Dieter, S.; Brunner, H.; Rupp, S. A screen in Saccharomyces cerevisiae identified CaMCM1, an essential gene in Candida albicans crucial for morphogenesis. Mol. Microbiol. 2003, 47, 943–959. [Google Scholar] [CrossRef] [PubMed]
- Bensen, E.S.; Filler, S.G.; Berman, J. A forkhead transcription factor is important for true hyphal as well as yeast morphogenesis in Candida albicans. Eukaryot. Cell 2002, 1, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, M.; Thompson, D.S.; Lazzell, A.; Carlisle, P.L.; Pierce, C.; Monteagudo, C.; Lopez-Ribot, J.L.; Kadosh, D. UME6, a novel filament-specific regulator of Candida albicans hyphal extension and virulence. Mol. Biol. Cell 2008, 19, 1354–1365. [Google Scholar] [CrossRef] [PubMed]
- Zeidler, U.; Lettner, T.; Lassnig, C.; Muller, M.; Lajko, R.; Hintner, H.; Breitenbach, M.; Bito, A. UME6 is a crucial downstream target of other transcriptional regulators of true hyphal development in Candida albicans. FEMS Yeast Res 2009, 9, 126–142. [Google Scholar] [CrossRef] [PubMed]
- Bockmuhl, D.P.; Ernst, J.F. A potential phosphorylation site for an A-type kinase in the Efg1 regulator protein contributes to hyphal morphogenesis of Candida albicans. Genetics 2001, 157, 1523–1530. [Google Scholar] [PubMed]
- Lu, Y.; Su, C.; Wang, A.; Liu, H. Hyphal development in Candida albicans requires two temporally linked changes in promoter chromatin for initiation and maintenance. PLoS Biol 2011, 9, e1001105. [Google Scholar] [CrossRef]
- Lane, S.; Birse, C.; Zhou, S.; Matson, R.; Liu, H. DNA array studies demonstrate convergent regulation of virulence factors by Cph1, Cph2, and Efg1 in Candida albicans. J. Biol. Chem. 2001, 276, 48988–48996. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Albrecht-Eckardt, D.; Brunke, S.; Hube, B.; Hünniger, K.; Kurzai, O. A Core Filamentation Response Network in Candida albicans Is Restricted to Eight Genes. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Carlisle, P.L.; Banerjee, M.; Lazzell, A.; Monteagudo, C.; Lopez-Ribot, J.L.; Kadosh, D. Expression levels of a filament-specific transcriptional regulator are sufficient to determine Candida albicans morphology and virulence. Proc. Natl. Acad. Sci. USA 2009, 106, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Biswas, K.; Morschhauser, J. The Mep2p ammonium permease controls nitrogen starvation-induced filamentous growth in Candida albicans. Mol. Microbiol. 2005, 56, 649–669. [Google Scholar] [CrossRef] [PubMed]
- Maidan, M.M.; De Rop, L.; Serneels, J.; Exler, S.; Rupp, S.; Tournu, H.; Thevelein, J.M.; Van Dijck, P. The G protein-coupled receptor Gpr1 and the Galpha protein Gpa2 act through the cAMP-protein kinase A pathway to induce morphogenesis in Candida albicans. Mol. Biol. Cell 2005, 16, 1971–1986. [Google Scholar] [CrossRef] [PubMed]
- Bockmuhl, D.P.; Krishnamurthy, S.; Gerads, M.; Sonneborn, A.; Ernst, J.F. Distinct and redundant roles of the two protein kinase A isoforms Tpk1p and Tpk2p in morphogenesis and growth of Candida albicans. Mol. Microbiol. 2001, 42, 1243–1257. [Google Scholar] [CrossRef] [PubMed]
- Kohler, J.R.; Fink, G.R. Candida albicans strains heterozygous and homozygous for mutations in mitogen-activated protein kinase signaling components have defects in hyphal development. Proc. Natl. Acad. Sci. USA 1996, 93, 13223–13228. [Google Scholar] [CrossRef] [PubMed]
- Leberer, E.; Harcus, D.; Broadbent, I.D.; Clark, K.L.; Dignard, D.; Ziegelbauer, K.; Schmidt, A.; Gow, N.A.; Brown, A.J.; Thomas, D.Y. Signal transduction through homologs of the Ste20p and Ste7p protein kinases can trigger hyphal formation in the pathogenic fungus Candida albicans. Proc. Natl. Acad. Sci. USA 1996, 93, 13217–13222. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, R.S.; Uppuluri, P.; Zaas, A.K.; Collins, C.; Senn, H.; Perfect, J.R.; Heitman, J.; Cowen, L.E. Hsp90 orchestrates temperature-dependent Candida albicans morphogenesis via Ras1-PKA signaling. Curr. Biol. 2009, 19, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Carlisle, P.L.; Kadosh, D. Candida albicans Ume6, a filament-specific transcriptional regulator, directs hyphal growth via a pathway involving Hgc1 cyclin-related protein. Eukaryot. Cell 2010, 9, 1320–1328. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.D.; Lee, R.T.; Wang, Y.M.; Lin, Q.S.; Wang, Y. Phosphorylation of Rga2, a Cdc42 GAP, by CDK/Hgc1 is crucial for Candida albicans hyphal growth. EMBO J. 2007, 26, 3760–3769. [Google Scholar] [CrossRef] [PubMed]
- Bishop, A.; Lane, R.; Beniston, R.; Chapa-y-Lazo, B.; Smythe, C.; Sudbery, P. Hyphal growth in Candida albicans requires the phosphorylation of Sec2 by the Cdc28-Ccn1/Hgc1 kinase. EMBO J. 2010, 29, 2930–2942. [Google Scholar] [CrossRef] [PubMed]
- Caballero-Lima, D.; Sudbery, P.E. In Candida albicans, phosphorylation of Exo84 by Cdk1-Hgc1 is necessary for efficient hyphal extension. Mol. Biol. Cell 2014, 25, 1097–1110. [Google Scholar] [CrossRef] [PubMed]
- Sinha, I.; Wang, Y.M.; Philp, R.; Li, C.R.; Yap, W.H.; Wang, Y. Cyclin-dependent kinases control septin phosphorylation in Candida albicans hyphal development. Dev. Cell 2007, 13, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Huang, Z.X.; Au Yong, J.Y.; Zou, H.; Zeng, G.; Gao, J.; Wang, Y.; Wong, A.H.H.; Wang, Y. CDK phosphorylates the polarisome scaffold Spa2 to maintain its localization at the site of cell growth. Mol. Microbiol. 2016, 101, 250–264. [Google Scholar] [CrossRef] [PubMed]
- Davis, D. Adaptation to environmental pH in Candida albicans and its relation to pathogenesis. Curr. Genet. 2003, 44, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, F.J.; Konopka, J.B. Identification of an N-Acetylglucosamine Transporter That Mediates Hyphal Induction in Candida albicans. Mol. Biol. Cell 2007, 18, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Basso, V.; D’Enfert, C.; Znaidi, S.; Bachellier-Bassi, S. From Genes to Networks: The Regulatory Circuitry Controlling Candida albicans Morphogenesis. In Current Topics in Microbiology and Immunology; Springer: Berlin/Heidelberg, Germany, 2018; in press. [Google Scholar]
- Albuquerque, P.; Casadevall, A. Quorum sensing in fungi a review. Med. Mycol. 2012, 50, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Hornby, J.M.; Jensen, E.C.; Lisec, A.D.; Tasto, J.J.; Jahnke, B.; Shoemaker, R.; Dussault, P.; Nickerson, K.W. Quorum sensing in the dimorphic fungus Candida albicans is mediated by farnesol. Appl. Environ. Microbiol. 2001, 67, 2982–2992. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.A.; Turner, K.J.; Chaloupka, J.; Cottier, F.; de Sordi, L.; Sanglard, D.; Levin, L.R.; Buck, J.; Mühlschlegel, F.A. The quorum-sensing molecules farnesol/homoserine lactone and dodecanol operate via distinct modes of action in Candida albicans. Eukaryot. Cell 2011, 10, 1034–1042. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Su, C.; Unoje, O.; Liu, H. Quorum sensing controls hyphal initiation in Candida albicans through Ubr1-mediated protein degradation. Proc. Natl. Acad. Sci. USA 2014, 111, 1975–1980. [Google Scholar] [CrossRef] [PubMed]
- Polke, M.; Sprenger, M.; Scherlach, K.; Albán-Proaño, M.C.; Martin, R.; Hertweck, C.; Hube, B.; Jacobsen, I.D. A functional link between hyphal maintenance and quorum sensing in Candida albicans. Mol. Microbiol. 2017, 103, 595–617. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Summers, E.; Guo, B.; Fink, G. Ras signaling is required for serum-induced hyphal differentiation in Candida albicans. J. Bacteriol. 1999, 181, 6339–6346. [Google Scholar] [PubMed]
- Leberer, E.; Harcus, D.; Dignard, D.; Johnson, L.; Ushinsky, S.; Thomas, D.Y.; Schroppel, K. Ras links cellular morphogenesis to virulence by regulation of the MAP kinase and cAMP signalling pathways in the pathogenic fungus Candida albicans. Mol. Microbiol. 2001, 42, 673–687. [Google Scholar] [CrossRef] [PubMed]
- Bar-Yosef, H.; Vivanco Gonzalez, N.; Ben-Aroya, S.; Kron, S.J.; Kornitzer, D. Chemical inhibitors of Candida albicans hyphal morphogenesis target endocytosis. Sci. Rep. 2017, 7, 5692. [Google Scholar] [CrossRef] [PubMed]
- Naseem, S.; Araya, E.; Konopka, J.B. Hyphal growth in Candida albicans does not require induction of hyphal-specific gene expression. Mol. Biol. Cell 2015, 26, 1174–1187. [Google Scholar] [CrossRef] [PubMed]
- Braun, B.R.; Head, W.S.; Wang, M.X.; Johnson, A.D. Identification and characterization of TUP1-regulated genes in Candida albicans. Genetics 2000, 156, 31–44. [Google Scholar] [PubMed]
- Weissman, Z.; Kornitzer, D. A family of Candida cell surface haem-binding proteins involved in haemin and haemoglobin-iron utilization. Mol. Microbiol. 2004, 53, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Loeb, J.D.; Kerentseva, T.A.; Pan, T.; Sepulveda-Becerra, M.; Liu, H. Saccharomyces cerevisiae G1 cyclins are differentially involved in invasive and pseudohyphal growth independent of the filamentation mitogen-activated protein kinase pathway. Genetics 1999, 153, 1535–1546. [Google Scholar] [PubMed]
- Rua, D.; Tobe, B.T.; Kron, S.J. Cell cycle control of yeast filamentous growth. Curr. Opin. Microbiol. 2001, 4, 720–727. [Google Scholar] [CrossRef]
- Berman, J. Morphogenesis and cell cycle progression in Candida albicans. Curr. Opin. Microbiol. 2006, 9, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Bachewich, C.; Thomas, D.Y.; Whiteway, M. Depletion of a polo-like kinase in Candida albicans activates cyclase-dependent hyphal-like growth. Mol. Biol. Cell 2003, 14, 2163–2180. [Google Scholar] [CrossRef] [PubMed]
- Bachewich, C.; Nantel, A.; Whiteway, M. Cell cycle arrest during S or M phase generates polarized growth via distinct signals in Candida albicans. Mol. Microbiol. 2005, 57, 942–959. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.M.; Wang, Y.M.; Zheng, X.D.; Lee, R.T.; Wang, Y. Critical role of DNA checkpoints in mediating genotoxic-stress-induced filamentous growth in Candida albicans. Mol. Biol. Cell 2007, 18, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Bachewich, C.; Whiteway, M. Cyclin Cln3p links G1 progression to hyphal and pseudohyphal development in Candida albicans. Eukaryot. Cell 2005, 4, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Chapa y Lazo, B.; Bates, S.; Sudbery, P. The G1 cyclin Cln3 regulates morphogenesis in Candida albicans. Eukaryot. Cell 2005, 4, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Willems, A.R.; Schwab, M.; Tyers, M. A hitchhiker’s guide to the cullin ubiquitin ligases: SCF and its kin. Biochim. Biophys. Acta 2004, 1695, 133–170. [Google Scholar] [CrossRef] [PubMed]
- Sela, N.; Atir-Lande, A.; Kornitzer, D. Neddylation and CAND1 independently stimulate SCF ubiquitin ligase activity in Candida albicans. Eukaryot. Cell 2012, 11, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Trunk, K.; Gendron, P.; Nantel, A.; Lemieux, S.; Roemer, T.; Raymond, M. Depletion of the cullin Cdc53p induces morphogenetic changes in Candida albicans. Eukaryot. Cell 2009, 8, 756–767. [Google Scholar] [CrossRef] [PubMed]
- Atir-Lande, A.; Gildor, T.; Kornitzer, D. Role for the SCFCDC4 ubiquitin ligase in Candida albicans morphogenesis. Mol. Biol. Cell 2005, 16, 2772–2785. [Google Scholar] [CrossRef] [PubMed]
- Mendelsohn, S.; Pinsky, M.; Weissman, Z.; Kornitzer, D. Regulation of the Candida albicans Hypha-Inducing Transcription Factor Ume6 by the CDK1 Cyclins Cln3 and Hgc1. mSphere 2017, 2, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Woolford, C.A.; Lagree, K.; Xu, W.; Aleynikov, T.; Adhikari, H.; Sanchez, H.; Cullen, P.J.; Lanni, F.; Andes, D.R.; Mitchell, A.P. Bypass of Candida albicans Filamentation/Biofilm Regulators through Diminished Expression of Protein Kinase Cak1. PLoS Genet. 2016, 12, e1006487. [Google Scholar] [CrossRef] [PubMed]
- Woolford, C.A.; Lagree, K.; Aleynikov, T.; Mitchell, A.P. Negative control of Candida albicans filamentation-associated gene expression by essential protein kinase gene KIN28. Curr. Genet. 2017, 63, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Ofir, A.; Hofmann, K.; Weindling, E.; Gildor, T.; Barker, K.S.; Rogers, P.D.; Kornitzer, D. Role of a Candida albicans Nrm1/Whi5 homologue in cell cycle gene expression and DNA replication stress response. Mol. Microbiol. 2012, 84, 778–794. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zeng, G.; Wang, Y. G1 and S phase arrest in Candida albicans induces filamentous growth via distinct mechanisms. Mol. Microbiol. 2018, 110, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Glory, A.; van Oostende, C.T.; Geitmann, A.; Bachewich, C. Depletion of the mitotic kinase Cdc5p in Candida albicans results in the formation of elongated buds that switch to the hyphal fate over time in a Ume6p and Hgc1p-dependent manner. Fungal Genet. Biol. 2017, 107, 51–66. [Google Scholar] [CrossRef] [PubMed]
- Wolyniak, M.J.; Sundstrom, P. Role of actin cytoskeletal dynamics in activation of the cyclic AMP pathway and HWP1 gene expression in Candida albicans. Eukaryot. Cell 2007, 6, 1824–1840. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Fang, H.M.; Zhu, Y.; Wang, Y. Candida albicans Cyr1, Cap1 and G-actin form a sensor/effector apparatus for activating cAMP synthesis in hyphal growth. Mol. Microbiol. 2010, 75, 579–591. [Google Scholar] [CrossRef] [PubMed]
- Douglas, L.M.; Martin, S.W.; Konopka, J.B. BAR domain proteins Rvs161 and Rvs167 contribute to Candida albicans endocytosis, morphogenesis, and virulence. Infect. Immun. 2009, 77, 4150–4160. [Google Scholar] [CrossRef] [PubMed]
- Bar-Yosef, H.; Gildor, T.; Ramírez-Zavala, B.; Schmauch, C.; Weissman, Z.; Pinsky, M.; Naddaf, R.; Morschhäuser, J.; Arkowitz, R.A.; Kornitzer, D. A Global Analysis of Kinase Function in Candida albicans Hyphal Morphogenesis Reveals a Role for the Endocytosis Regulator Akl1. Front. Cell. Infect. Microbiol. 2018, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Su, C.; Solis, N.V.; Filler, S.G.; Liu, H. Synergistic regulation of hyphal elongation by hypoxia, CO(2), and nutrient conditions controls the virulence of Candida albicans. Cell Host Microbe 2013, 14, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Braun, B.R.; Kadosh, D.; Johnson, A.D. NRG1, a repressor of filamentous growth in C. albicans, is down-regulated during filament induction. EMBO J. 2001, 20, 4753–4761. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Su, C.; Liu, H. A GATA Transcription Factor Recruits Hda1 in Response to Reduced Tor1 Signaling to Establish a Hyphal Chromatin State in Candida albicans. PLoS Pathog. 2012, 8, e1002663. [Google Scholar] [CrossRef] [PubMed]
- Bassilana, M.; Blyth, J.; Arkowitz, R.A. Cdc24, the GDP-GTP exchange factor for Cdc42, is required for invasive hyphal growth of Candida albicans. Eukaryot. Cell 2003, 2, 9–18. [Google Scholar] [CrossRef] [PubMed]
- VandenBerg, A.L.; Ibrahim, A.S.; Edwards, J.E.; Toenjes, K.A.; Johnson, D.I. Cdc42p GTPase regulates the budded-to-hyphal-form transition and expression of hypha-specific transcripts in Candida albicans. Eukaryot. Cell 2004, 3, 724–734. [Google Scholar] [CrossRef] [PubMed]
- Warris, A.; Ballou, E.R. Oxidative responses and fungal infection biology. Semin. Cell Dev. Biol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Nasution, O.; Srinivasa, K.; Kim, M.; Kim, Y.J.; Kim, W.; Jeong, W.; Choi, W. Hydrogen peroxide induces hyphal differentiation in Candida albicans. Eukaryot. Cell 2008, 7, 2008–2011. [Google Scholar] [CrossRef] [PubMed]
- Rossi, D.C.P.; Gleason, J.E.; Sanchez, H.; Schatzman, S.S.; Culbertson, E.M.; Johnson, C.J.; McNees, C.A.; Coelho, C.; Nett, J.E.; Andes, D.R.; et al. Candida albicans FRE8 encodes a member of the NADPH oxidase family that produces a burst of ROS during fungal morphogenesis. PLoS Pathog. 2017, 13, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Koch, B.; Barugahare, A.A.; Lo, T.L.; Huang, C.; Schittenhelm, R.B.; Powell, D.R.; Beilharz, T.H.; Traven, A. A Metabolic Checkpoint for the Yeast-to-Hyphae Developmental Switch Regulated by Endogenous Nitric Oxide Signaling. Cell Rep. 2018, 25, 2244–2258.e7. [Google Scholar] [CrossRef] [PubMed]
- Hromatka, B.S.; Noble, S.M.; Johnson, A.D. Transcriptional Response of Candida albicans to Nitric Oxide and the Role of the YHB1 Gene in Nitrosative Stress and Virulence. Mol. Biol. Cell 2005, 16, 4814–4826. [Google Scholar] [CrossRef] [PubMed]
- Harcus, D.; Nantel, A.; Marcil, A.; Rigby, T.; Whiteway, M. Transcription Profiling of Cyclic AMP Signaling in Candida albicans. Mol. Biol. Cell 2004, 15, 4490–4499. [Google Scholar] [CrossRef] [PubMed]
- Blank, H.M.; Callahan, M.; Pistikopoulos, I.P.E.; Polymenis, A.O.; Polymenis, M. Scaling of G1 Duration with Population Doubling Time by a Cyclin in Saccharomyces cerevisiae. Genetics 2018, 210, 895–906. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kornitzer, D. Regulation of Candida albicans Hyphal Morphogenesis by Endogenous Signals. J. Fungi 2019, 5, 21. https://doi.org/10.3390/jof5010021
Kornitzer D. Regulation of Candida albicans Hyphal Morphogenesis by Endogenous Signals. Journal of Fungi. 2019; 5(1):21. https://doi.org/10.3390/jof5010021
Chicago/Turabian StyleKornitzer, Daniel. 2019. "Regulation of Candida albicans Hyphal Morphogenesis by Endogenous Signals" Journal of Fungi 5, no. 1: 21. https://doi.org/10.3390/jof5010021
APA StyleKornitzer, D. (2019). Regulation of Candida albicans Hyphal Morphogenesis by Endogenous Signals. Journal of Fungi, 5(1), 21. https://doi.org/10.3390/jof5010021



