Recent Advances in the Use of Galleria mellonella Model to Study Immune Responses against Human Pathogens
Abstract
:1. Introduction
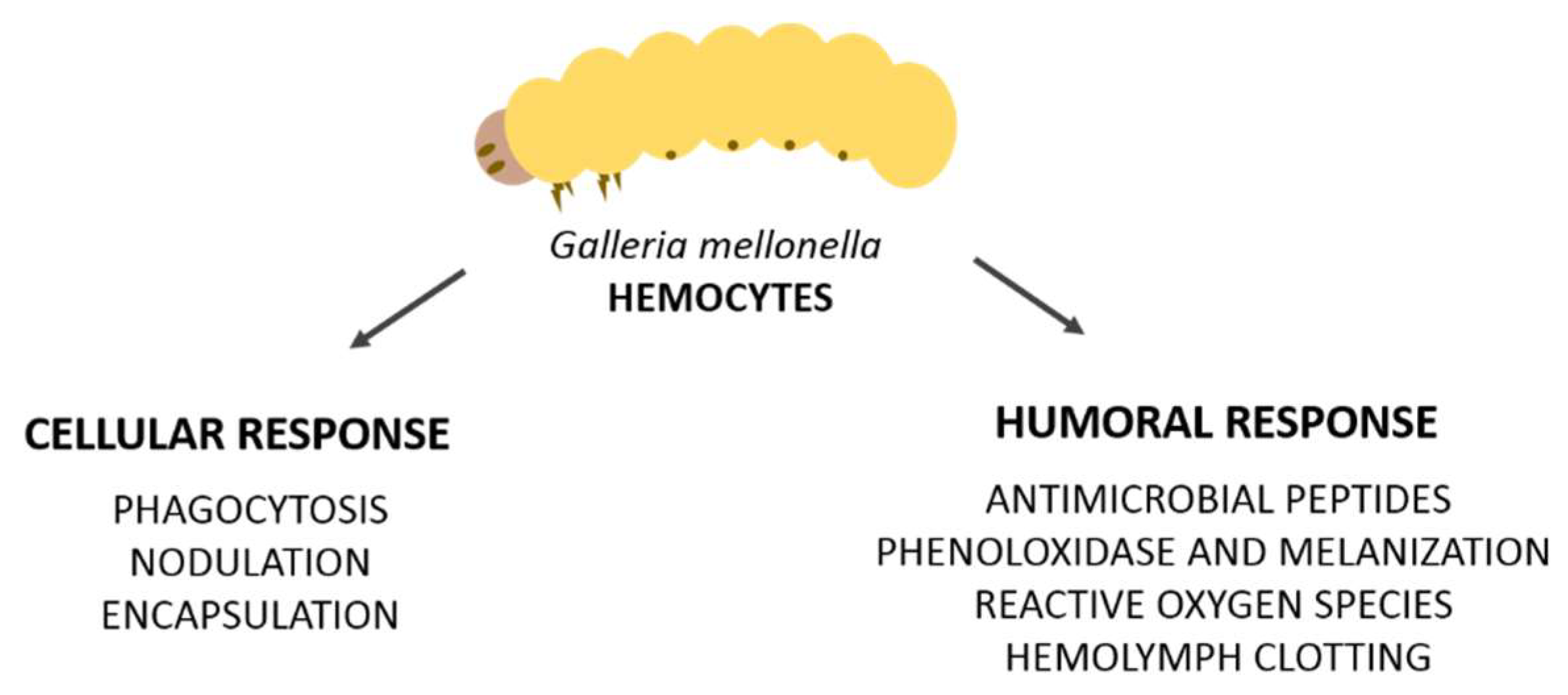
2. Cellular Immune Response
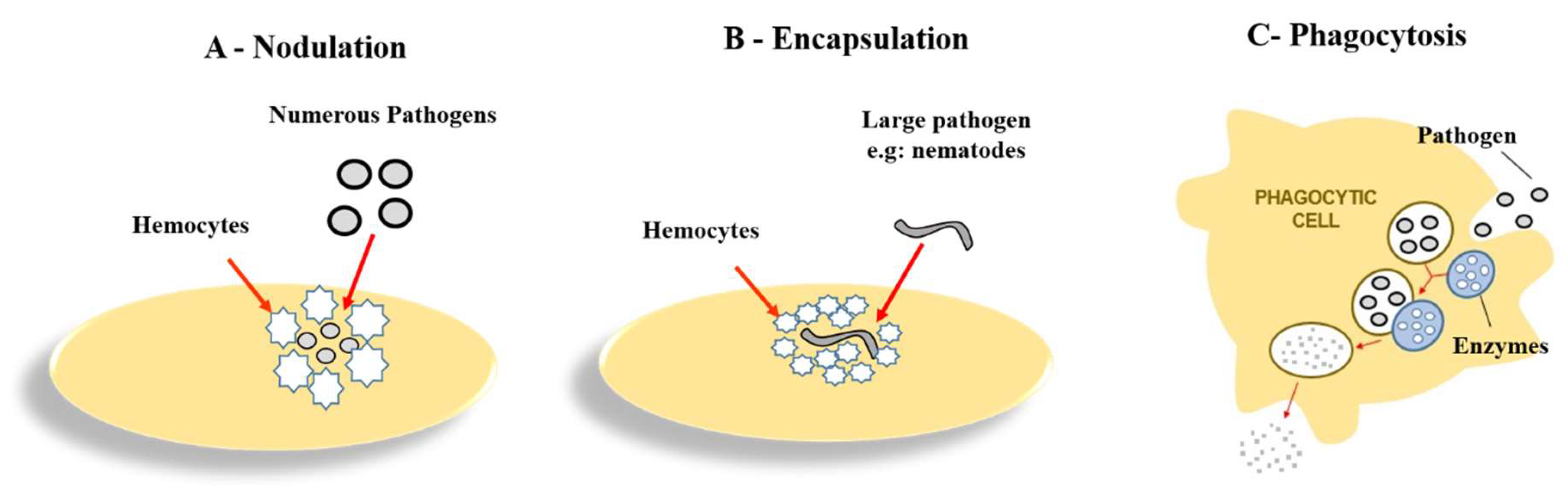
Hemocytes and Involved Reactions (Phagocytosis, Nodulation, and Encapsulation)
3. Humoral Immune Response
3.1. Antimicrobial Peptides (AMPS)
3.2. Prophenoloxidase and Melanization
3.3. Hemolymph Clotting
3.4. Reactive Oxygen Species (ROS)
3.5. Primary Immunization
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kavanagh, K.; Reeves, E.P. Exploiting the potential of insects for in vivo pathogenicity testing of microbial pathogens. FEMS Microbiol. Rev. 2004, 28, 101–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chao, C.C.; Hsu, P.C.; Jen, C.F.; Chen, I.H.; Wang, C.H.; Chan, H.C.; Tsai, P.W.; Tung, K.C.; Lan, C.Y.; Chuang, Y.J. Zebrafish as a model host for Candida albicans infection. Infect. Immun. 2010, 78, 2512–2521. [Google Scholar] [CrossRef] [PubMed]
- Hamilos, G.; Samonis, G.; Kontoyiannis, D.P. Recent Advances in the Use of Drosophila melanogaster as a Model to Study Immunopathogenesis of Medically Important Filamentous Fungi. Int. J. Microbiol. 2012, 2012, 583792. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, I.D.; Grosse, K.; Hube, B. Embryonated chicken eggs as alternative infection model for pathogenic fungi. Methods Mol. Biol. 2012, 845, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Muhammed, M.; Coleman, J.J.; Mylonakis, E. Caenorhabditis elegans: A nematode infection model for pathogenic fungi. Methods Mol. Biol. 2012, 845, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, B.B.; Mylonakis, E. Using non-mammalian hosts to study fungal virulence and host defense. Curr. Opin. Microbiol. 2006, 9, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Desalermos, A.; Fuchs, B.B.; Mylonakis, E. Selecting an invertebrate model host for the study of fungal pathogenesis. PLoS Pathog. 2012, 8, e1002451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramarao, N.; Nielsen-Leroux, C.; Lereclus, D. The insect Galleria mellonella as a powerful infection model to investigate bacterial pathogenesis. J. Vis. Exp. 2012, 70, e4392. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.J.; Loh, J.M.; Proft, T. Galleria mellonella infection models for the study of bacterial diseases and for antimicrobial drug testing. Virulence 2016, 7, 214–229. [Google Scholar] [CrossRef] [PubMed]
- Wojda, I. Immunity of the greater wax moth Galleria mellonella. Insect. Sci. 2017, 24, 342–357. [Google Scholar] [CrossRef] [PubMed]
- Binder, U.; Maurer, E.; Lass-Flörl, C. Galleria mellonella: An invertebrate model to study pathogenicity in correctly defined fungal species. Fungal Biol. 2016, 120, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Smoot, L.M.; Smoot, J.C.; Graham, M.R.; Somerville, G.A.; Sturdevant, D.E.; Migliaccio, C.A.; Sylva, G.L.; Musser, J.M. Global differential gene expression in response to growth temperature alteration in group A Streptococcus. Proc. Natl. Acad. Sci. USA 2001, 98, 10416–10421. [Google Scholar] [CrossRef] [PubMed]
- Konkel, M.E.; Tilly, K. Temperature-regulated expression of bacterial virulence genes. Microbes Infect. 2000, 2, 157–166. [Google Scholar] [CrossRef]
- Lange, A.; Beier, S.; Huson, D.H.; Parusel, R.; Iglauer, F.; Frick, J.S. Genome Sequence of Galleria mellonella (Greater Wax Moth). Genome Announc. 2018, 6. [Google Scholar] [CrossRef] [PubMed]
- Vogel, H.; Altincicek, B.; Glockner, G.; Vilcinskas, A. A comprehensive transcriptome and immune-gene repertoire of the lepidopteran model host Galleria mellonella. BMC Genom. 2011, 12, 308. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, K.; Vilcinskas, A. Development and immunity-related microRNAs of the lepidopteran model host Galleria mellonella. BMC Genom. 2014, 15, 705. [Google Scholar] [CrossRef] [PubMed]
- Loh, J.M.; Adenwalla, N.; Wiles, S.; Proft, T. Galleria mellonella larvae as an infection model for group A streptococcus. Virulence 2013, 4, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Olsen, R.J.; Watkins, M.E.; Cantu, C.C.; Beres, S.B.; Musser, J.M. Virulence of serotype M3 Group A Streptococcus strains in wax worms (Galleria mellonella larvae). Virulence 2011, 2, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Evans, B.A.; Rozen, D.E. A Streptococcus pneumoniae infection model in larvae of the wax moth Galleria mellonella. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2653–2660. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, S.L.; Leanti La Rosa, S.; Casey, P.G.; Hill, C.; Diep, D.B.; Nes, I.F.; Brede, D.A. In vivo assessment of growth and virulence gene expression during commensal and pathogenic lifestyles of luxABCDE-tagged Enterococcus faecalis strains in murine gastrointestinal and intravenous infection models. Appl. Environ. Microbiol. 2013, 79, 3986–3997. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, S.L.; Diep, D.B.; Nes, I.F.; Brede, D.A. Construction and application of a luxABCDE reporter system for real-time monitoring of Enterococcus faecalis gene expression and growth. Appl. Environ. Microbiol. 2012, 78, 7003–7011. [Google Scholar] [CrossRef] [PubMed]
- Benachour, A.; Ladjouzi, R.; Le Jeune, A.; Hébert, L.; Thorpe, S.; Courtin, P.; Chapot-Chartier, M.P.; Prajsnar, T.K.; Foster, S.J.; Mesnage, S. The lysozyme-induced peptidoglycan N-acetylglucosamine deacetylase PgdA (EF1843) is required for Enterococcus faecalis virulence. J. Bacteriol. 2012, 194, 6066–6073. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, F.; Teixeira, N.; Rigottier-Gois, L.; Marujo, P.; Nielsen-LeRoux, C.; Crespo, M.T.; Lopes, M.e.F.; Serror, P. Virulence of Enterococcus faecalis dairy strains in an insect model: The role of fsrB and gelE. Microbiology 2009, 155, 3564–3571. [Google Scholar] [CrossRef] [PubMed]
- Chibebe Junior, J.; Fuchs, B.B.; Sabino, C.P.; Junqueira, J.C.; Jorge, A.O.; Ribeiro, M.S.; Gilmore, M.S.; Rice, L.B.; Tegos, G.P.; Hamblin, M.R.; et al. Photodynamic and antibiotic therapy impair the pathogenesis of Enterococcus faecium in a whole animal insect model. PLoS ONE 2013, 8, e55926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lebreton, F.; Le Bras, F.; Reffuveille, F.; Ladjouzi, R.; Giard, J.C.; Leclercq, R.; Cattoir, V. Galleria mellonella as a model for studying Enterococcus faecium host persistence. J. Mol. Microbiol. Biotechnol. 2011, 21, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Desbois, A.P.; Coote, P.J. Wax moth larva (Galleria mellonella): An in vivo model for assessing the efficacy of antistaphylococcal agents. J. Antimicrob. Chemother. 2011, 66, 1785–1790. [Google Scholar] [CrossRef] [PubMed]
- Joyce, S.A.; Gahan, C.G. Molecular pathogenesis of Listeria monocytogenes in the alternative model host Galleria mellonella. Microbiology 2010, 156, 3456–3468. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, K.; Abu Mraheil, M.; Silva, S.; Müller, D.; Cemic, F.; Hemberger, J.; Hain, T.; Vilcinskas, A.; Chakraborty, T. Anti-Listeria activities of Galleria mellonella hemolymph proteins. Appl. Environ. Microbiol. 2011, 77, 4237–4240. [Google Scholar] [CrossRef] [PubMed]
- Andrejko, M.; Mizerska-Dudka, M. Elastase B of Pseudomonas aeruginosa stimulates the humoral immune response in the greater wax moth, Galleria mellonella. J. Invertebr. Pathol. 2011, 107, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Andrejko, M.; Mizerska-Dudka, M. Effect of Pseudomonas aeruginosa elastase B on level and activity of immune proteins/peptides of Galleria mellonella hemolymph. J. Insect Sci. 2012, 12, 88. [Google Scholar] [CrossRef] [PubMed]
- Ciesielczuk, H.; Betts, J.; Phee, L.; Doumith, M.; Hope, R.; Woodford, N.; Wareham, D.W. Comparative virulence of urinary and bloodstream isolates of extra-intestinal pathogenic Escherichia coli in a Galleria mellonella model. Virulence 2015, 6, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Williamson, D.A.; Mills, G.; Johnson, J.R.; Porter, S.; Wiles, S. In vivo correlates of molecularly inferred virulence among extraintestinal pathogenic Escherichia coli (ExPEC) in the wax moth Galleria mellonella model system. Virulence 2014, 5, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Wand, M.E.; McCowen, J.W.; Nugent, P.G.; Sutton, J.M. Complex interactions of Klebsiella pneumoniae with the host immune system in a Galleria mellonella infection model. J. Med. Microbiol. 2013, 62, 1790–1798. [Google Scholar] [CrossRef] [PubMed]
- Insua, J.L.; Llobet, E.; Moranta, D.; Pérez-Gutiérrez, C.; Tomás, A.; Garmendia, J.; Bengoechea, J.A. Modeling Klebsiella pneumoniae pathogenesis by infection of the wax moth Galleria mellonella. Infect. Immun. 2013, 81, 3552–3565. [Google Scholar] [CrossRef] [PubMed]
- Harding, C.R.; Schroeder, G.N.; Reynolds, S.; Kosta, A.; Collins, J.W.; Mousnier, A.; Frankel, G. Legionella pneumophila pathogenesis in the Galleria mellonella infection model. Infect. Immun. 2012, 80, 2780–2790. [Google Scholar] [CrossRef] [PubMed]
- Aperis, G.; Fuchs, B.B.; Anderson, C.A.; Warner, J.E.; Calderwood, S.B.; Mylonakis, E. Galleria mellonella as a model host to study infection by the Francisella tularensis live vaccine strain. Microbes Infect. 2007, 9, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, A.C.; Thompson, M.G.; Black, C.C.; Kessler, J.L.; Clark, L.P.; McQueary, C.N.; Gancz, H.Y.; Corey, B.W.; Moon, J.K.; Si, Y.; et al. AB5075, a Highly Virulent Isolate of Acinetobacter baumannii, as a Model Strain for the Evaluation of Pathogenesis and Antimicrobial Treatments. MBio 2014, 5, e01076-14. [Google Scholar] [CrossRef] [PubMed]
- Schell, M.A.; Lipscomb, L.; DeShazer, D. Comparative genomics and an insect model rapidly identify novel virulence genes of Burkholderia mallei. J. Bacteriol. 2008, 190, 2306–2313. [Google Scholar] [CrossRef] [PubMed]
- Slater, J.L.; Gregson, L.; Denning, D.W.; Warn, P.A. Pathogenicity of Aspergillus fumigatus mutants assessed in Galleria mellonella matches that in mice. Med. Mycol. 2011, 49, S107–S113. [Google Scholar] [CrossRef] [PubMed]
- Thomaz, L.; García-Rodas, R.; Guimarães, A.J.; Taborda, C.P.; Zaragoza, O.; Nosanchuk, J.D. Galleria mellonella as a model host to study Paracoccidioides lutzii and Histoplasma capsulatum. Virulence 2013, 4, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Scorzoni, L.; de Paula e Silva, A.C.; Singulani, J.e.L.; Leite, F.S.; de Oliveira, H.C.; da Silva, R.A.; Fusco-Almeida, A.M.; Mendes-Giannini, M.J. Comparison of virulence between Paracoccidioides brasiliensis and Paracoccidioides lutzii using Galleria mellonella as a host model. Virulence 2015, 6, 766–776. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.J.; Muhammed, M.; Kasperkovitz, P.V.; Vyas, J.M.; Mylonakis, E. Fusarium pathogenesis investigated using Galleria mellonella as a heterologous host. Fungal Biol. 2011, 115, 1279–1289. [Google Scholar] [CrossRef] [PubMed]
- Firacative, C.; Duan, S.; Meyer, W. Galleria mellonella model identifies highly virulent strains among all major molecular types of Cryptococcus gattii. PLoS ONE 2014, 9, e105076. [Google Scholar] [CrossRef] [PubMed]
- Bouklas, T.; Diago-Navarro, E.; Wang, X.; Fenster, M.; Fries, B.C. Characterization of the virulence of Cryptococcus neoformans strains in an insect model. Virulence 2015, 6, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Cotter, G.; Doyle, S.; Kavanagh, K. Development of an insect model for the in vivo pathogenicity testing of yeasts. FEMS Immunol. Med. Microbiol. 2000, 27, 163–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Champion, O.L.; Wagley, S.; Titball, R.W. Galleria mellonella as a model host for microbiological and toxin research. Virulence 2016, 7, 840–845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jafri, R.H.; Chaudhry, M.B. Development of Tipula iridescent virus (TIV) in Galleria mellonella larvae exposed to gamma radiation. J. Invertebr. Pathol. 1971, 18, 46–50. [Google Scholar] [CrossRef]
- Garzon, S.; Charpentier, G.; Kurstak, E. Morphogenesis of the nodamura virus in the larbae of the lepidopteran Galleria mellonella (L.). Arch. Virol. 1978, 56, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Buchatskiĭ, L.P.; Litvinov, G.S.; Lebedinets, N.N.; Filenko, O.M.; Podberezova, L.M. Effect of temperature on the infectivity of the iridovirus and densonucleosis virus of blood-sucking mosquitoes. Vopr. Virusol. 1988, 33, 603–606. [Google Scholar] [PubMed]
- Constantino, M.; Christian, P.; Marina, C.F.; Williams, T. A comparison of techniques for detecting Invertebrate iridescent virus 6. J. Virol. Methods 2001, 98, 109–118. [Google Scholar] [CrossRef]
- Büyükgüzel, E.; Tunaz, H.; Stanley, D.; Büyükgüzel, K. Eicosanoids mediate Galleria mellonella cellular immune response to viral infection. J. Insect Physiol. 2007, 53, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Özkan, S.; Coutts, R.H. Aspergillus fumigatus mycovirus causes mild hypervirulent effect on pathogenicity when tested on Galleria mellonella. Fungal Genet. Biol. 2015, 76, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Wittwer, D.; Franchini, A.; Ottaviani, E.; Wiesner, A. Presence of IL-1- and TNF-like molecules in Galleria mellonella (Lepidoptera) haemocytes and in an insect cell line Fromestigmene acraea (Lepidoptera). Cytokine 1999, 11, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Browne, N.; Heelan, M.; Kavanagh, K. An analysis of the structural and functional similarities of insect hemocytes and mammalian phagocytes. Virulence 2013, 4, 597–603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bergin, D.; Reeves, E.P.; Renwick, J.; Wientjes, F.B.; Kavanagh, K. Superoxide production in Galleria mellonella hemocytes: Identification of proteins homologous to the NADPH oxidase complex of human neutrophils. Infect. Immun. 2005, 73, 4161–4170. [Google Scholar] [CrossRef] [PubMed]
- Renwick, J.; Reeves, E.P.; Wientjes, F.B.; Kavanagh, K. Translocation of proteins homologous to human neutrophil p47phox and p67phox to the cell membrane in activated hemocytes of Galleria mellonella. Dev. Comp. Immunol. 2007, 31, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Segal, E.; Frenkel, M. Experimental in Vivo Models of Candidiasis. J. Fungi 2018, 4, 21. [Google Scholar] [CrossRef] [PubMed]
- Freires, I.A.; Sardi, J.C.; de Castro, R.D.; Rosalen, P.L. Alternative Animal and Non-Animal Models for Drug Discovery and Development: Bonus or Burden? Pharm. Res. 2017, 34, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Lavine, M.D.; Strand, M.R. Insect hemocytes and their role in immunity. Insect Biochem. Mol. Biol. 2002, 32, 1295–1309. [Google Scholar] [CrossRef]
- Griesch, J.; Wedde, M.; Vilcinskas, A. Recognition and regulation of metalloproteinase activity in the haemolymph of Galleria mellonella: A new pathway mediating induction of humoral immune responses. Insect Biochem. Mol. Biol. 2000, 30, 461–472. [Google Scholar] [CrossRef]
- Altincicek, B.; Linder, M.; Linder, D.; Preissner, K.T.; Vilcinskas, A. Microbial metalloproteinases mediate sensing of invading pathogens and activate innate immune responses in the lepidopteran model host Galleria mellonella. Infect. Immun. 2007, 75, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Gibreel, T.M.; Upton, M. Synthetic epidermicin NI01 can protect Galleria mellonella larvae from infection with Staphylococcus aureus. J. Antimicrob. Chemother. 2013, 68, 2269–2273. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Lopez, A.; Forastiero, A.; Cendejas-Bueno, E.; Gregson, L.; Mellado, E.; Howard, S.J.; Livermore, J.L.; Hope, W.W.; Cuenca-Estrella, M. An invertebrate model to evaluate virulence in Aspergillus fumigatus: The role of azole resistance. Med. Mycol. 2014, 52, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Koch, G.; Nadal-Jimenez, P.; Cool, R.H.; Quax, W.J. Assessing Pseudomonas virulence with nonmammalian host: Galleria mellonella. Methods Mol. Biol. 2014, 1149, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Gómez, A.; Corredor, M.; Benítez-Páez, A.; Peláez, C. Development of quantitative proteomics using iTRAQ based on the immunological response of Galleria mellonella larvae challenged with Fusarium oxysporum microconidia. PLoS ONE 2014, 9, e112179. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, L.E.; Rossoni, R.D.; Barbosa, J.O.; Jorge, A.O.; Junqueira, J.C.; Valera, M.C. Different extracts of Zingiber officinale decrease Enterococcus faecalis infection in Galleria mellonella. Braz. Dent. J. 2015, 26, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Junqueira, J.C. Galleria mellonella as a model host for human pathogens: Recent studies and new perspectives. Virulence 2012, 3, 474–476. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, G.; Kavanagh, K. Analysis of the early cellular and humoral responses of Galleria mellonella larvae to infection by Candida albicans. Virulence 2018, 9, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Holz, A.; Bossinger, B.; Strasser, T.; Janning, W.; Klapper, R. The two origins of hemocytes in Drosophila. Development 2003, 130, 4955–4962. [Google Scholar] [CrossRef] [PubMed]
- Boman, H.G.; Hultmark, D. Cell-free immunity in insects. Annu. Rev. Microbiol. 1987, 41, 103–126. [Google Scholar] [CrossRef] [PubMed]
- Tojo, S.; Naganuma, F.; Arakawa1, K.; Yokoo1, S. Involvement of both granular cells and plasmatocytes in phagocytic reactions in the greater wax moth, Galleria mellonella. J. Insect Physiol. 2000, 46, 1129–1135. [Google Scholar] [CrossRef]
- Arteaga Blanco, L.A.; Crispim, J.S.; Fernandes, K.M.; de Oliveira, L.L.; Pereira, M.F.; Bazzolli, D.M.S.; Martins, G.F. Differential cellular immune response of Galleria mellonella to Actinobacillus pleuropneumoniae. Cell Tissue Res. 2017, 370, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Strand, M.R. The insect cellular immune response. Insect Sci. 2008, 15, 1–14. [Google Scholar] [CrossRef]
- Strand, M.R.; Beck, M.H.; Lavine, M.D.; Clark, K.D. Microplitis demolitor bracovirus inhibits phagocytosis by hemocytes from Pseudoplusia includens. Arch. Insect Biochem. Physiol. 2006, 61, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Whitsel, B.L.; Petrucelli, L.M.; Werner, G. Symmetry and connectivity in the map of the body surface in somatosensory area II of primates. J. Neurophysiol. 1969, 32, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Pech, L.L.; Strand, M.R. Granular cells are required for encapsulation of foreign targets by insect haemocytes. J. Cell Sci. 1996, 109, 2053–2060. [Google Scholar] [PubMed]
- Strand, M.R.; Pech, L.L. Immunological basis for compatibility in parasitoid-host relationships. Annu. Rev. Entomol. 1995, 40, 31–56. [Google Scholar] [CrossRef] [PubMed]
- Grizanova, E.V.; Semenova, A.D.; Komarov, D.A.; Chertkova, E.A.; Slepneva, I.A.; Dubovskiy, I.M. Maintenance of redox balance by antioxidants in hemolymph of the greater wax moth Galleria mellonella larvae during encapsulation response. Arch. Insect Biochem. Physiol. 2018, 98, e21460. [Google Scholar] [CrossRef] [PubMed]
- Jiravanichpaisal, P.; Lee, B.L.; Söderhäll, K. Cell-mediated immunity in arthropods: Hematopoiesis, coagulation, melanization and opsonization. Immunobiology 2006, 211, 213–236. [Google Scholar] [CrossRef] [PubMed]
- Baggiolini, M.; Wymann, M.P. Turning on the respiratory burst. Trends Biochem. Sci. 1990, 15, 69–72. [Google Scholar] [CrossRef]
- Ratcliffe, N.A. Invertebrate immunity--a primer for the non-specialist. Immunol. Lett. 1985, 10, 253–270. [Google Scholar] [CrossRef]
- Gotz, P.; Weise, C.; Kopacek, P.; Losen, S.; Wiesner, A. Isolated Apolipophorin III from Galleria mellonella Stimulates the Immune Reactions of This Insect. J. Insect Physiol. 1997, 43, 383–391. [Google Scholar] [PubMed]
- Halwani, A.E.; Dunphy, G.B. Apolipophorin-III in Galleria mellonella potentiates hemolymph lytic activity. Dev. Comp. Immunol. 1999, 23, 563–570. [Google Scholar] [CrossRef]
- Whitten, M.M.; Tew, I.F.; Lee, B.L.; Ratcliffe, N.A. A novel role for an insect apolipoprotein (apolipophorin III) in beta-1,3-glucan pattern recognition and cellular encapsulation reactions. J. Immunol. 2004, 172, 2177–2185. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.Y.; Whitten, M.M.; Cho, M.Y.; Lee, K.Y.; Kim, M.S.; Ratcliffe, N.A.; Lee, B.L. Calreticulin enriched as an early-stage encapsulation protein in wax moth Galleria mellonella larvae. Dev. Comp. Immunol. 2002, 26, 335–343. [Google Scholar] [CrossRef]
- Trevijano-Contador, N.; Herrero-Fernandez, I.; Garcia-Barbazan, I.; Scorzoni, L.; Rueda, C.; Rossi, S.A.; Garcia-Rodas, R.; Zaragoza, O. Cryptococcus neoformans induces antimicrobial responses and behaves as a facultative intracellular pathogen in the non mammalian model Galleria mellonella. Virulence 2015, 6, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Gago, S.; García-Rodas, R.; Cuesta, I.; Mellado, E.; Alastruey-Izquierdo, A. Candida parapsilosis, Candida orthopsilosis, and Candida metapsilosis virulence in the non-conventional host Galleria mellonella. Virulence 2014, 5, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Mowlds, P.; Kavanagh, K. Effect of pre-incubation temperature on susceptibility of Galleria mellonella larvae to infection by Candida albicans. Mycopathologia 2008, 165, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Maurer, E.; Hortnagl, C.; Lackner, M.; Grassle, D.; Naschberger, V.; Moser, P.; Segal, E.; Semis, M.; Lass-Florl, C.; Binder, U. Galleria mellonella as a model system to study virulence potential of mucormycetes and evaluation of antifungal treatment. Med. Mycol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Maurer, E.; Browne, N.; Surlis, C.; Jukic, E.; Moser, P.; Kavanagh, K.; Lass-Florl, C.; Binder, U. Galleria mellonella as a host model to study Aspergillus terreus virulence and amphotericin B resistance. Virulence 2015, 6, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Chiers, K.; De Waele, T.; Pasmans, F.; Ducatelle, R.; Haesebrouck, F. Virulence factors of Actinobacillus pleuropneumoniae involved in colonization, persistence and induction of lesions in its porcine host. Vet. Res. 2010, 41, 65. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.F.; Rossi, C.C.; de Queiroz, M.V.; Martins, G.F.; Isaac, C.; Bosse, J.T.; Li, Y.; Wren, B.W.; Terra, V.S.; Cuccui, J.; et al. Galleria mellonella is an effective model to study Actinobacillus pleuropneumoniae infection. Microbiology 2015, 161, 387–400. [Google Scholar] [CrossRef] [PubMed]
- Senior, N.J.; Bagnall, M.C.; Champion, O.L.; Reynolds, S.E.; La Ragione, R.M.; Woodward, M.J.; Salguero, F.J.; Titball, R.W. Galleria mellonella as an infection model for Campylobacter jejuni virulence. J. Med. Microbiol. 2011, 60, 661–669. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scorzoni, L.; de Lucas, M.P.; Mesa-Arango, A.C.; Fusco-Almeida, A.M.; Lozano, E.; Cuenca-Estrella, M.; Mendes-Giannini, M.J.; Zaragoza, O. Antifungal efficacy during Candida krusei infection in non-conventional models correlates with the yeast in vitro susceptibility profile. PLoS ONE 2013, 8, e60047. [Google Scholar] [CrossRef] [PubMed]
- Ames, L.; Duxbury, S.; Pawlowska, B.; Ho, H.L.; Haynes, K.; Bates, S. Galleria mellonella as a host model to study Candida glabrata virulence and antifungal efficacy. Virulence 2017, 8, 1909–1917. [Google Scholar] [CrossRef] [PubMed]
- Mesa-Arango, A.C.; Forastiero, A.; Bernal-Martínez, L.; Cuenca-Estrella, M.; Mellado, E.; Zaragoza, O. The non-mammalian host Galleria mellonella can be used to study the virulence of the fungal pathogen Candida tropicalis and the efficacy of antifungal drugs during infection by this pathogenic yeast. Med. Mycol. 2013, 51, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rodas, R.; Casadevall, A.; Rodriguez-Tudela, J.L.; Cuenca-Estrella, M.; Zaragoza, O. Cryptococcus neoformans capsular enlargement and cellular gigantism during Galleria mellonella infection. PLoS ONE 2011, 6, e24485. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.A.; Trevijano-Contador, N.; Scorzoni, L.; Mesa-Arango, A.C.; de Oliveira, H.C.; Werther, K.; de Freitas Raso, T.; Mendes-Giannini, M.J.; Zaragoza, O.; Fusco-Almeida, A.M. Impact of Resistance to Fluconazole on Virulence and Morphological Aspects of Cryptococcus neoformans and Cryptococcus gattii Isolates. Front. Microbiol. 2016, 7, 153. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, G.; Clarke, G.; Kavanagh, K. Characterisation of the cellular and proteomic response of Galleria mellonella larvae to the development of invasive aspergillosis. BMC Microbiol. 2018, 18, 63. [Google Scholar] [CrossRef] [PubMed]
- Renwick, J.; Daly, P.; Reeves, E.P.; Kavanagh, K. Susceptibility of larvae of Galleria mellonella to infection by Aspergillus fumigatus is dependent upon stage of conidial germination. Mycopathologia 2006, 161, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Zdybicka-Barabas, A.; Sowa-Jasilek, A.; Staczek, S.; Jakubowicz, T.; Cytrynska, M. Different forms of apolipophorin III in Galleria mellonella larvae challenged with bacteria and fungi. Peptides 2015, 68, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.P.; Entwistle, F.; Coote, P.J. Effective immunosuppression with dexamethasone phosphate in the Galleria mellonella larva infection model resulting in enhanced virulence of Escherichia coli and Klebsiella pneumoniae. Med. Microbiol. Immunol. 2016, 205, 333–343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Satyavathi, V.V.; Minz, A.; Nagaraju, J. Nodulation: An unexplored cellular defense mechanism in insects. Cell Signal. 2014, 26, 1753–1763. [Google Scholar] [CrossRef] [PubMed]
- Yi, H.Y.; Chowdhury, M.; Huang, Y.D.; Yu, X.Q. Insect antimicrobial peptides and their applications. Appl. Microbiol. Biotechnol. 2014, 98, 5807–5822. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al Souhail, Q.; Hiromasa, Y.; Rahnamaeian, M.; Giraldo, M.C.; Takahashi, D.; Valent, B.; Vilcinskas, A.; Kanost, M.R. Characterization and regulation of expression of an antifungal peptide from hemolymph of an insect, Manduca sexta. Dev. Comp. Immunol. 2016, 61, 258–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bolouri Moghaddam, M.R.; Tonk, M.; Schreiber, C.; Salzig, D.; Czermak, P.; Vilcinskas, A.; Rahnamaeian, M. The potential of the Galleria mellonella innate immune system is maximized by the co-presentation of diverse antimicrobial peptides. Biol. Chem. 2016, 397, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Thevissen, K.; Warnecke, D.C.; François, I.E.; Leipelt, M.; Heinz, E.; Ott, C.; Zähringer, U.; Thomma, B.P.; Ferket, K.K.; Cammue, B.P. Defensins from insects and plants interact with fungal glucosylceramides. J. Biol. Chem. 2004, 279, 3900–3905. [Google Scholar] [CrossRef] [PubMed]
- Brogden, K.A. Antimicrobial peptides: Pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 2005, 3, 238–250. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.E.; Howard, A.; Kasprzak, A.B.; Gordon, K.H.; East, P.D. A peptidomics study reveals the impressive antimicrobial peptide arsenal of the wax moth Galleria mellonella. Insect Biochem. Mol. Biol. 2009, 39, 792–800. [Google Scholar] [CrossRef] [PubMed]
- Rahnamaeian, M. Antimicrobial peptides: Modes of mechanism, modulation of defense responses. Plant Signal. Behav. 2011, 6, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Bergin, D.; Murphy, L.; Keenan, J.; Clynes, M.; Kavanagh, K. Pre-exposure to yeast protects larvae of Galleria mellonella from a subsequent lethal infection by Candida albicans and is mediated by the increased expression of antimicrobial peptides. Microbes Infect. 2006, 8, 2105–2112. [Google Scholar] [CrossRef] [PubMed]
- Vilcinskas, A. Anti-infective therapeutics from the Lepidopteran model host Galleria mellonella. Curr. Pharm. Des. 2011, 17, 1240–1245. [Google Scholar] [CrossRef] [PubMed]
- Taszłow, P.; Vertyporokh, L.; Wojda, I. Humoral immune response of Galleria mellonella after repeated infection with Bacillus thuringiensis. J. Invertebr. Pathol. 2017, 149, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.E.; Howard, A.; Kasprzak, A.B.; Gordon, K.H.; East, P.D. The discovery and analysis of a diverged family of novel antifungal moricin-like peptides in the wax moth Galleria mellonella. Insect Biochem. Mol. Biol. 2008, 38, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Cytryńska, M.; Mak, P.; Zdybicka-Barabas, A.; Suder, P.; Jakubowicz, T. Purification and characterization of eight peptides from Galleria mellonella immune hemolymph. Peptides 2007, 28, 533–546. [Google Scholar] [CrossRef] [PubMed]
- Mak, P.; Zdybicka-Barabas, A.; Cytryńska, M. A different repertoire of Galleria mellonella antimicrobial peptides in larvae challenged with bacteria and fungi. Dev. Comp. Immunol. 2010, 34, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Zdybicka-Barabas, A.; Mak, P.; Klys, A.; Skrzypiec, K.; Mendyk, E.; Fiołka, M.J.; Cytryńska, M. Synergistic action of Galleria mellonella anionic peptide 2 and lysozyme against Gram-negative bacteria. Biochim. Biophys. Acta. 2012, 1818, 2623–2635. [Google Scholar] [CrossRef] [PubMed]
- Zdybicka-Barabas, A.; Stączek, S.; Mak, P.; Skrzypiec, K.; Mendyk, E.; Cytryńska, M. Synergistic action of Galleria mellonella apolipophorin III and lysozyme against Gram-negative bacteria. Biochim. Biophys. Acta 2013, 1828, 1449–1456. [Google Scholar] [CrossRef] [PubMed]
- Büyükgüzel, E.; Tunaz, H.; Stanley, D.; Büyükgüzel, K. The influence of chronic eicosanoid biosynthesis inhibition on life history of the greater waxmoth, Galleria mellonella and its ectoparasitoid, Bracon hebetor. J. Insect Physiol. 2011, 57, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Stanley, D.; Haas, E.; Miller, J. Eicosanoids: Exploiting Insect Immunity to Improve Biological Control Programs. Insects 2012, 3, 492–510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schuhmann, B.; Seitz, V.; Vilcinskas, A.; Podsiadlowski, L. Cloning and expression of gallerimycin, an antifungal peptide expressed in immune response of greater wax moth larvae, Galleria mellonella. Arch. Insect Biochem. Physiol. 2003, 53, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Yun, E.K.; Jang, W.S.; Kim, I.; Lee, J.H.; Park, S.Y.; Ryu, K.S.; Seo, S.J.; Kim, C.H.; Lee, I.H. Purification, cDNA cloning and expression of an insect defensin from the great wax moth, Galleria mellonella. Insect Mol. Biol. 2004, 13, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; Lee, J.H.; Kim, I.; Seo, S.J.; Son, S.M.; Lee, K.Y.; Lee, I.H. Purification and cDNA cloning of a cecropin-like peptide from the great wax moth, Galleria mellonella. Mol. Cells 2004, 17, 262–266. [Google Scholar] [PubMed]
- Wojda, I.; Jakubowicz, T. Humoral immune response upon mild heat-shock conditions in Galleria mellonella larvae. J. Insect Physiol. 2007, 53, 1134–1144. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.; Kavanagh, K. Caspofungin primes the immune response of the larvae of Galleria mellonella and induces a non-specific antimicrobial response. J. Med. Microbiol. 2011, 60, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Rossoni, R.D.; Fuchs, B.B.; de Barros, P.P.; Velloso, M.D.; Jorge, A.O.; Junqueira, J.C.; Mylonakis, E. Lactobacillus paracasei modulates the immune system of Galleria mellonella and protects against Candida albicans infection. PLoS ONE 2017, 12, e0173332. [Google Scholar] [CrossRef] [PubMed]
- Kopacek, P.; Weise, C.; Gotz, P. The prophenoloxidase from the wax moth Galleria mellonella: Purification and characterization of the proenzyme. Insect Biochem. Mol. Biol. 1995, 25, 1081–1091. [Google Scholar] [CrossRef]
- Ashida, M. Purification and characterization of pre-phenoloxidase from hemolymph of the silkworm Bombyx mori. Arch. Biochem. Biophys. 1971, 144, 749–762. [Google Scholar] [CrossRef]
- Schmit, A.R.; Ratcliffe, N.A. The encapsulation of foreign tissue implants in Galleria mellonella larvae. J. Insect Physiol. 1977, 23, 175–184. [Google Scholar] [CrossRef]
- Perdoni, F.; Falleni, M.; Tosi, D.; Cirasola, D.; Romagnoli, S.; Braidotti, P.; Clementi, E.; Bulfamante, G.; Borghi, E. A histological procedure to study fungal infection in the wax moth Galleria mellonella. Eur. J. Histochem. 2014, 58, 2428. [Google Scholar] [CrossRef] [PubMed]
- Demir, D.; Gencer, N.; Er, A. Purification and characterization of prophenoloxidase from Galleria mellonella L. Artif. Cells Blood Substit. Immobil. Biotechnol. 2012, 40, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Ashida, M. The prophenoloxidase cascade in insect immunity. Res. Immunol. 1990, 141, 908–910. [Google Scholar] [CrossRef]
- Cerenius, L.; Lee, B.L.; Soderhall, K. The proPO-system: Pros and cons for its role in invertebrate immunity. Trends Immunol. 2008, 29, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.; Zhang, Q.; Zhang, J.; Yang, B.; Wu, K.; Xie, W.; Luan, Y.X.; Ling, E. Insect prophenoloxidase: The view beyond immunity. Front. Physiol. 2014, 5, 252. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, J.A.; Reichhart, J.M.; Hetru, C. Innate immunity in higher insects. Curr. Opin. Immunol. 1996, 8, 8–13. [Google Scholar] [CrossRef]
- Park, S.Y.; Kim, C.H.; Jeong, W.H.; Lee, J.H.; Seo, S.J.; Han, Y.S.; Lee, I.H. Effects of two hemolymph proteins on humoral defense reactions in the wax moth, Galleria mellonella. Dev. Comp. Immunol. 2005, 29, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, J.P.; Kanost, M.R.; Trenczek, T. Biological mediators of insect immunity. Annu. Rev. Entomol. 1997, 42, 611–643. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, H.; Kinoshita, K.; Ashida, M. Purification of a peptidoglycan recognition protein from hemolymph of the silkworm, Bombyx mori. J. Biol. Chem. 1996, 271, 13854–13860. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, N.; Imamura, M.; Kadotani, T.; Yaoi, K.; Iwahana, H.; Sato, R. The lipopolysaccharide-binding protein participating in hemocyte nodule formation in the silkworm Bombyx mori is a novel member of the C-type lectin superfamily with two different tandem carbohydrate-recognition domains. FEBS Lett. 1999, 443, 139–143. [Google Scholar] [CrossRef]
- Yu, X.Q.; Zhu, Y.F.; Ma, C.; Fabrick, J.A.; Kanost, M.R. Pattern recognition proteins in Manduca sexta plasma. Insect Biochem. Mol. Biol. 2002, 32, 1287–1293. [Google Scholar] [CrossRef]
- Li, D.; Scherfer, C.; Korayem, A.M.; Zhao, Z.; Schmidt, O.; Theopold, U. Insect hemolymph clotting: Evidence for interaction between the coagulation system and the prophenoloxidase activating cascade. Insect Biochem. Mol. Biol. 2002, 32, 919–928. [Google Scholar] [CrossRef]
- Tang, H. Regulation and function of the melanization reaction in Drosophila. Fly 2009, 3, 105–111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karaman, M.; Alvandian, A.; Bahar, I.H. Galleria mellonella larva model in evaluating the effects of biofilm in Candida albicans. Mikrobiyol. Bul. 2017, 51, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Borghi, E.; Romagnoli, S.; Fuchs, B.B.; Cirasola, D.; Perdoni, F.; Tosi, D.; Braidotti, P.; Bulfamante, G.; Morace, G.; Mylonakis, E. Correlation between Candida albicans biofilm formation and invasion of the invertebrate host Galleria mellonella. Future Microbiol. 2014, 9, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, B.B.; O’Brien, E.; Khoury, J.B.; Mylonakis, E. Methods for using Galleria mellonella as a model host to study fungal pathogenesis. Virulence 2010, 1, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Jorjao, A.L.; de Oliveira, F.E.; Leao, M.V.P.; Jorge, A.O.C.; de Oliveira, L.D. Effect of Lactobacillus rhamnosus on the response of Galleria mellonella against Staphylococcus aureus and Escherichia coli infections. Arch. Microbiol. 2018, 200, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Alcazar-Fuoli, L.; Buitrago, M.; Gomez-Lopez, A.; Mellado, E. An alternative host model of a mixed fungal infection by azole susceptible and resistant Aspergillus spp. strains. Virulence 2015, 6, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Kloezen, W.; Parel, F.; Bruggemann, R.; Asouit, K.; Helvert-van Poppel, M.; Fahal, A.; Mouton, J.; van de Sande, W. Amphotericin B and terbinafine but not the azoles prolong survival in Galleria mellonella larvae infected with Madurella mycetomatis. Med. Mycol. 2018, 56, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Rueda, C.; Cuenca-Estrella, M.; Zaragoza, O. Paradoxical growth of Candida albicans in the presence of caspofungin is associated with multiple cell wall rearrangements and decreased virulence. Antimicrob. Agents Chemother. 2014, 58, 1071–1083. [Google Scholar] [CrossRef] [PubMed]
- Eisenman, H.C.; Duong, R.; Chan, H.; Tsue, R.; McClelland, E.E. Reduced virulence of melanized Cryptococcus neoformans in Galleria mellonella. Virulence 2014, 5, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Matzinger, P. The danger model: A renewed sense of self. Science 2002, 296, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Rowley, A.F.; Ratcliffe, N.A. The granular cells of Galleria mellonella during clotting and phagocytic reactions in vitro. Tissue Cell 1976, 8, 437–446. [Google Scholar] [CrossRef]
- Dushay, M.S. Insect hemolymph clotting. Cell. Mol. Life Sci. 2009, 66, 2643–2650. [Google Scholar] [CrossRef] [PubMed]
- Altincicek, B.; Stotzel, S.; Wygrecka, M.; Preissner, K.T.; Vilcinskas, A. Host-derived extracellular nucleic acids enhance innate immune responses, induce coagulation, and prolong survival upon infection in insects. J. Immunol. 2008, 181, 2705–2712. [Google Scholar] [CrossRef] [PubMed]
- Nagai, T.; Kawabata, S. A link between blood coagulation and prophenol oxidase activation in arthropod host defense. J. Biol. Chem. 2000, 275, 29264–29267. [Google Scholar] [CrossRef] [PubMed]
- Reeves, E.P.; Lu, H.; Jacobs, H.L.; Messina, C.G.; Bolsover, S.; Gabella, G.; Potma, E.O.; Warley, A.; Roes, J.; Segal, A.W. Killing activity of neutrophils is mediated through activation of proteases by K+ flux. Nature 2002, 416, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Scandalios, J.G. Oxidative stress: Molecular perception and transduction of signals triggering antioxidant gene defenses. Braz. J. Med. Biol. Res. 2005, 38, 995–1014. [Google Scholar] [CrossRef] [PubMed]
- Nappi, A.J.; Vass, E. Hydrogen peroxide production in immune-reactive Drosophila melanogaster. J. Parasitol. 1998, 84, 1150–1157. [Google Scholar] [CrossRef] [PubMed]
- Slepneva, I.A.; Glupov, V.V.; Sergeeva, S.V.; Khramtsov, V.V. EPR detection of reactive oxygen species in hemolymph of Galleria mellonella and Dendrolimus superans sibiricus (Lepidoptera) larvae. Biochem. Biophys. Res. Commun. 1999, 264, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, N.; Davis, A.J.; Giebultowicz, J.M. Circadian regulation of response to oxidative stress in Drosophila melanogaster. Biochem. Biophys. Res. Commun. 2008, 374, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Whitten, M.M.; Mello, C.B.; Gomes, S.A.; Nigam, Y.; Azambuja, P.; Garcia, E.S.; Ratcliffe, N.A. Role of superoxide and reactive nitrogen intermediates in Rhodnius prolixus (Reduviidae)/Trypanosoma rangeli interactions. Exp. Parasitol. 2001, 98, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Lanz-Mendoza, H.; Hernández-Martínez, S.; Ku-López, M.; Rodríguez, M.e.C.; Herrera-Ortiz, A.; Rodríguez, M.H. Superoxide anion in Anopheles albimanus hemolymph and midgut is toxic to Plasmodium berghei ookinetes. J. Parasitol. 2002, 88, 702–706. [Google Scholar] [CrossRef]
- Bánfi, B.; Clark, R.A.; Steger, K.; Krause, K.H. Two novel proteins activate superoxide generation by the NADPH oxidase NOX1. J. Biol. Chem. 2003, 278, 3510–3513. [Google Scholar] [CrossRef] [PubMed]
- Geiszt, M.; Lekstrom, K.; Witta, J.; Leto, T.L. Proteins homologous to p47phox and p67phox support superoxide production by NAD(P)H oxidase 1 in colon epithelial cells. J. Biol. Chem. 2003, 278, 20006–20012. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, N.; Hyrsl, P.; Simek, V. Nitric oxide production by hemocytes of larva and pharate prepupa of Galleria mellonella in response to bacterial lipopolysaccharide: Cytoprotective or cytotoxic? Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 2006, 142, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Nappi, A.J.; Vass, E.; Frey, F.; Carton, Y. Nitric oxide involvement in Drosophila immunity. Nitric Oxide 2000, 4, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Komarov, D.A.; Slepneva, I.A.; Glupov, V.V.; Khramtsov, V.V. Superoxide and hydrogen peroxide formation during enzymatic oxidation of DOPA by phenoloxidase. Free Radic. Res. 2005, 39, 853–858. [Google Scholar] [CrossRef] [PubMed]
- McCord, J.M.; Fridovich, I. The utility of superoxide dismutase in studying free radical reactions. I. Radicals generated by the interaction of sulfite, dimethyl sulfoxide, and oxygen. J. Biol. Chem. 1969, 244, 6056–6063. [Google Scholar] [PubMed]
- Lyakhovich, V.V.; Vavilin, V.A.; Zenkov, N.K.; Menshchikova, E.B. Active defense under oxidative stress. The antioxidant responsive element. Biochemistry (Mosc) 2006, 71, 962–974. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Pardini, R.S. Mechanisms for regulating oxygen toxicity in phytophagous insects. Free Radic. Biol. Med. 1990, 8, 401–413. [Google Scholar] [CrossRef]
- Ahmad, S. Oxidative stress from environmental pollutants. Arch. Insect Biochem. Physiol. 1995, 29, 135–157. [Google Scholar] [CrossRef] [PubMed]
- Felton, G.W.; Summers, C.B. Antioxidant systems in insects. Arch. Insect Biochem. Physiol. 1995, 29, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Champion, O.L.; Cooper, I.A.; James, S.L.; Ford, D.; Karlyshev, A.; Wren, B.W.; Duffield, M.; Oyston, P.C.; Titball, R.W. Galleria mellonella as an alternative infection model for Yersinia pseudotuberculosis. Microbiology 2009, 155, 1516–1522. [Google Scholar] [CrossRef] [PubMed]
- Dubovskiy, I.M.; Krukova, N.A.; Glupov, V.V. Phagocytic activity and encapsulation rate of Galleria mellonella larval haemocytes during bacterial infection by Bacillus thuringiensis. J. Invertebr. Pathol. 2008, 98, 360–362. [Google Scholar] [CrossRef] [PubMed]
- Dubovskiĭ, I.M.; Grizanova, E.V.; Chertkova, E.A.; Slepneva, I.A.; Komarov, D.A.; Vorontsova, I.L.; Glupov, V.V. Generation of reactive oxygen species and activity of antioxidants in larva hemolymph of Galleria mellonella (L.) (lepidoptera: Piralidae) at development of process of encapsulation. Zh. Evol. Biokhim. Fiziol. 2010, 46, 30–36. [Google Scholar] [PubMed]
- Kryukova, N.A.; Dubovskiy, I.M.; Chertkova, E.A.; Vorontsova, Y.L.; Slepneva, I.A.; Glupov, V.V. The effect of Habrobracon hebetor venom on the activity of the prophenoloxidase system, the generation of reactive oxygen species and encapsulation in the haemolymph of Galleria mellonella larvae. J. Insect Physiol. 2011, 57, 796–800. [Google Scholar] [CrossRef] [PubMed]
- Gálvez, D.; Chapuisat, M. Immune priming and pathogen resistance in ant queens. Ecol. Evol. 2014, 4, 1761–1767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melillo, D.; Marino, R.; Italiani, P.; Boraschi, D. Innate Immune Memory in Invertebrate Metazoans: A Critical Appraisal. Front. Immunol. 2018, 9, 1915. [Google Scholar] [CrossRef] [PubMed]
- Little, T.J.; Kraaijeveld, A.R. Ecological and evolutionary implications of immunological priming in invertebrates. Trends Ecol. Evol. 2004, 19, 58–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pham, L.N.; Dionne, M.S.; Shirasu-Hiza, M.; Schneider, D.S. A specific primed immune response in Drosophila is dependent on phagocytes. PLoS Pathog. 2007, 3, e26. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, S.; Wang, Z. Responses of alternative complement expression to challenge with different combinations of Vibrio anguillarum, Escherichia coli and Staphylococcus aureus: Evidence for specific immune priming in amphioxus Branchiostoma belcheri. Fish Shellfish Immunol. 2009, 26, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, J. Specific memory within innate immune systems. Trends Immunol. 2005, 26, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Zhao, Z.; Liu, C.; Qiu, L. Priming Galleria mellonella (Lepidoptera: Pyralidae) larvae with heat-killed bacterial cells induced an enhanced immune protection against Photorhabdus luminescens TT01 and the role of innate immunity in the process. J. Econ. Entomol. 2014, 107, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Mowlds, P.; Coates, C.; Renwick, J.; Kavanagh, K. Dose-dependent cellular and humoral responses in Galleria mellonella larvae following beta-glucan inoculation. Microbes Infect. 2010, 12, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Fallon, J.P.; Troy, N.; Kavanagh, K. Pre-exposure of Galleria mellonella larvae to different doses of Aspergillus fumigatus conidia causes differential activation of cellular and humoral immune responses. Virulence 2011, 2, 413–421. [Google Scholar] [CrossRef] [PubMed]
| The Most Common Types of Hemocytes | Morphology and Functions | References |
|---|---|---|
| Prohemocytes | Small circular cells with large nucleus. Prohemocytes are not present in all types of insects and are considered progenitor cells. | [54,72,73] |
| Plasmatocytes | Most common in G. mellonella, possessing lysosomal enzymes in the cytoplasm, participate directly in phagocytosis and are also capsule-forming hemocytes. | [54,72,73,74] |
| Granulocytes | Small nucleus, and granules in the cytoplasm. Participate indirectly in phagocytosis and directly in the encapsulation process. Usually the most common hemocyte type. | [71,73] |
| Spherulocytes | Present in some insects, it has spherical inclusions. They transport and secrete cuticular components (as the 66 kDa peptide). Its function in insect immunity is not well understood. | [54,72] |
| Oenotocytes | Large and spherule cells. Are non-adhesive and no phagocytic cells and containphenoloxidase (PO) cascade components. | [54,59,72,73] |
| Microorganism | Hemocyte Response after Hours (h) of Infection | Rate of Cells with Phagocytosed Pathogens (%) | Reference |
|---|---|---|---|
| Candida albicans | 2 h: Decrease 6 h Increase 3 h: Decrease | Not evaluated 4% | [68] [94] |
| Candida glabrata | 2 h: Decrease 6 h: Decrease | Not evaluated | [95] |
| Candida parapsilosis | 2 h: Increase | 5% | [87] |
| Candida orthopsilosis | 2 h: Increase | 15% | [87] |
| Candida metapsilosis | 2 h: Increase | 18% | [87] |
| Candida krusei | 3 h: Decrease | 4% | [94] |
| Candida tropicalis | 2 h: Decrease 7 h: Decrease | 2 h: 18% | [96] |
| Cryptococcus neoformans | 2 h: Increase Not evaluated | 20–30% (*) 20–40% (*) | [97] [98] |
| Cryptococcus gattii | Not evaluated | 10–40% (*) | [98] |
| Aspergillus fumigatus | 2 h: Increase 2 h: Similar to the control 4 h: Increase 24 h: Similar to the control | 34% Evaluated but not quantified | [99,100] [63] |
| Fusarium oxysporum | 1 h: Decrease | Not evaluated | [101] |
| Paracoccidioides brasiliensis | 1 h: Decrease 3h: Decrease | 5% | [41] |
| Paracoccidioides lutzii | 1 h: Decrease 3 h: Decrease | 5% | [41] |
| Escherichia coli | Not evaluated | 1 h: 25% 2 h 30% | [102] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira, T.C.; De Barros, P.P.; Fugisaki, L.R.d.O.; Rossoni, R.D.; Ribeiro, F.d.C.; De Menezes, R.T.; Junqueira, J.C.; Scorzoni, L. Recent Advances in the Use of Galleria mellonella Model to Study Immune Responses against Human Pathogens. J. Fungi 2018, 4, 128. https://doi.org/10.3390/jof4040128
Pereira TC, De Barros PP, Fugisaki LRdO, Rossoni RD, Ribeiro FdC, De Menezes RT, Junqueira JC, Scorzoni L. Recent Advances in the Use of Galleria mellonella Model to Study Immune Responses against Human Pathogens. Journal of Fungi. 2018; 4(4):128. https://doi.org/10.3390/jof4040128
Chicago/Turabian StylePereira, Thais Cristine, Patrícia Pimentel De Barros, Luciana Ruano de Oliveira Fugisaki, Rodnei Dennis Rossoni, Felipe de Camargo Ribeiro, Raquel Teles De Menezes, Juliana Campos Junqueira, and Liliana Scorzoni. 2018. "Recent Advances in the Use of Galleria mellonella Model to Study Immune Responses against Human Pathogens" Journal of Fungi 4, no. 4: 128. https://doi.org/10.3390/jof4040128






