Development of an Improved Carotenoid Extraction Method to Characterize the Carotenoid Composition under Oxidative Stress and Cold Temperature in the Rock Inhabiting Fungus Knufia petricola A95
Abstract
1. Introduction
2. Material and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Diaz-Sanchez, V.; Estrada, A.F.; Trautmann, D.; Limon, M.C.; Al-Babili, S.; Avalos, J. Analysis of al-2 mutations in Neurospora. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Hausmann, A.; Sandmann, G. A single five-step desaturase is involved in the carotenoid biosynthesis pathway to β-carotene and torulene in Neurospora crassa. Fungal Genet. Biol. 2000, 30, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Tafer, H.; Lopandic, K.; Blasi, B.; Poyntner, C.; Sterflinger, K. Draft genome sequence of Exophiala mesophila, a black yeast with high bioremediation potential. Genome Announc. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Sandmann, G. Carotenoid biosynthesis in microorganisms and plants. Eur. J. Biochem. 1994, 223, 7–24. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, G.A.; Hearst, J.E. Carotenoids 2: Genetics and molecular biology of carotenoid pigment biosynthesis. FASEB J. 1996, 10, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Avalos, J.; Carmen Limon, M. Biological roles of fungal carotenoids. Curr. Genet. 2014, 61, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Frengova, G.I.; Beshkova, D.M. Carotenoids from Rhodotorula and Phaffia: Yeasts of biotechnological importance. J. Ind. Microbiol. Biotechnol. 2009, 36, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Mata-Gomez, L.C.; Montanez, J.C.; Mendez-Zavala, A.; Aguilar, C.N. Biotechnological production of carotenoids by yeasts: An overview. Microb. Cell Fact. 2014, 13, 12. [Google Scholar] [CrossRef] [PubMed]
- Moline, M.; Libkind, D.; van Broock, M. Production of torularhodin, torulene, and β-carotene by Rhodotorula yeasts. Methods Mol. Biol. 2012, 898, 275–283. [Google Scholar] [PubMed]
- Amaretti, A.; Simone, M.; Quartieri, A.; Masino, F.; Raimondi, S.; Leonardi, A.; Rossi, M. Isolation of carotenoid-producing yeasts from an alpine glacier. Chem. Eng. Trans. 2014, 38, 217–222. [Google Scholar]
- Arcangeli, C.; Cannistraro, S. In situ raman microspectroscopic identification and localization of carotenoids: Approach to monitoring of UV-B irradiation stress on antarctic fungus. Biopolymers 2000, 57, 179–186. [Google Scholar] [CrossRef]
- Libkind, D.; Moline, M.; Sampaio, J.P.; van Broock, M. Yeasts from high-altitude lakes: Influence of UV radiation. FEMS Microbiol. Ecol. 2009, 69, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Moline, M.; Libkind, D.; Dieguez Mdel, C.; van Broock, M. Photoprotective role of carotenoids in yeasts: Response to UV-B of pigmented and naturally-occurring albino strains. J. Photochem. Photobiol. B 2009, 95, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, M.M.; Moreno, L.F.; Stielow, B.J.; Muszewska, A.; Hainaut, M.; Gonzaga, L.; Abouelleil, A.; Patane, J.S.; Priest, M.; Souza, R.; et al. Exploring the genomic diversity of black yeasts and relatives (Chaetothyriales, Ascomycota). Stud. Mycol. 2017, 86, 1–28. [Google Scholar] [CrossRef] [PubMed]
- De Hoog, G.S.; Vicente, V.A.; Gorbushina, A.A. The bright future of darkness—the rising power of black fungi: Black yeasts, microcolonial fungi, and their relatives. Mycopathologia 2013, 175, 365–368. [Google Scholar] [CrossRef] [PubMed]
- Gunde-Cimerman, N.; Grube, M.; de Hoog, G.S. The emerging potential of melanized fungi: Black yeast between beauty and the beast. Fungal Biol. 2011, 115, 935–936. [Google Scholar] [CrossRef] [PubMed]
- Gorbushina, A.A.; Broughton, W.J. Microbiology of the atmosphere-rock interface: How biological interactions and physical stresses modulate a sophisticated microbial ecosystem. Annu. Rev. Microbiol. 2009, 63, 431–450. [Google Scholar] [CrossRef] [PubMed]
- Sterflinger, K. Temperature and NaCl-tolerance of rock-inhabiting meristematic fungi. Antonie Van Leeuwenhoek 1998, 74, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Kogej, T.; Stein, M.; Volkmann, M.; Gorbushina, A.A.; Galinski, E.A.; Gunde-Cimerman, N. Osmotic adaptation of the halophilic fungus hortaea werneckii: Role of osmolytes and melanization. Microbiol-Sgm 2007, 153, 4261–4273. [Google Scholar] [CrossRef] [PubMed]
- Kogej, T.; Wheeler, M.H.; Rizner, T.L.; Gunde-Cimerman, N. Evidence for 1,8-dihydroxynaphthalene melanin in three halophilic black yeasts grown under saline and non-saline conditions. FEMS Microbiol. Lett. 2004, 232, 203–209. [Google Scholar] [CrossRef]
- Volkmann, M.; Gorbushina, A.A. A broadly applicable method for extraction and characterization of mycosporines and mycosporine-like amino acids of terrestrial, marine and freshwater origin. FEMS Microbiol. Lett. 2006, 255, 286–295. [Google Scholar] [CrossRef] [PubMed]
- Gorbushina, A.A.; Kotlova, E.R.; Sherstneva, O.A. Cellular responses of microcolonial rock fungi to long-term desiccation and subsequent rehydration. Stud. Mycol. 2008, 61, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Wollenzien, U.; de Hoog, G.S.; Krumbein, W.; Uijthof, J.M. Sarcinomyces petricola, a new microcolonial fungus from marble in the mediterranean basin. Antonie Van Leeuwenhoek 1997, 71, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Gueidan, C.; Villasenor, C.R.; de Hoog, G.S.; Gorbushina, A.A.; Untereiner, W.A.; Lutzoni, F. A rock-inhabiting ancestor for mutualistic and pathogen-rich fungal lineages. Stud. Mycol. 2008, 61, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Nai, C.; Wong, H.Y.; Pannenbecker, A.; Broughton, W.J.; Benoit, I.; de Vries, R.P.; Gueidan, C.; Gorbushina, A.A. Nutritional physiology of a rock-inhabiting, model microcolonial fungus from an ancestral lineage of the Chaetothyriales (Ascomycetes). Fungal Genet. Biol. 2013, 56, 54–66. [Google Scholar] [CrossRef] [PubMed]
- Sandmann, G. Carotenoids of biotechnological importance. Adv. Biochem. Eng. Biotechnol. 2015, 148, 449–467. [Google Scholar] [PubMed]
- Weber, R.W.; Anke, H.; Davoli, P. Simple method for the extraction and reversed-phase high-performance liquid chromatographic analysis of carotenoid pigments from red yeasts (Basidiomycota, fungi). J. Chromatogr. A 2007, 1145, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Bona-Lovasz, J.; Bona, A.; Ederer, M.; Sawodny, O.; Ghosh, R. A rapid method for the extraction and analysis of carotenoids and other hydrophobic substances suitable for systems biology studies with photosynthetic bacteria. Metabolites 2013, 3, 912–930. [Google Scholar] [CrossRef] [PubMed]
- Zang, L.Y.; Sommerburg, O.; VanKuijk, F.J.G.M. Absorbance changes of carotenoids in different solvents. Free Radic. Biol. Med. 1997, 23, 1086–1089. [Google Scholar] [CrossRef]
- Takaichi, S. Characterization of carotenes in a combination of a C (18) HPLC column with isocratic elution and absorption spectra with a photodiode-array detector. Photosynthesis Res. 2000, 65, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Geciova, J.; Bury, D.; Jelen, P. Methods for disruption of microbial cells for potential use in the dairy industry—A review. Int. Dairy J. 2002, 12, 541–553. [Google Scholar] [CrossRef]
- Gu, Z.; Deming, C.; Yongbin, H.; Zhigang, C.; Feirong, G. Optimization of carotenoids extraction from Rhodobacter sphaeroides. LWT-Food Sci. Technol. 2008, 41, 1082–1088. [Google Scholar] [CrossRef]
- Papaioannou, E.; Roukas, T.; Liakopoulou-Kyriakides, M. Effect of biomass pre-treatment and solvent extraction on β-carotene and lycopene recovery from Blakeslea trispora cells. Prep. Biochem. Biotechnol. 2008, 38, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Craft, N.E.; Soares, J.H. Relative solubility, stability, and absorptivity of lutein and β-carotene in organic-solvents. J. Agric. Food Chem. 1992, 40, 431–434. [Google Scholar] [CrossRef]
- Su, Q.; Rowley, K.G.; Balazs, N.D.H. Carotenoids: Separation methods applicable to biological samples. J. Chromatogr. B 2002, 781, 393–418. [Google Scholar] [CrossRef]
- Sperstad, S.; Lutnaes, B.F.; Stormo, S.K.; Liaaen-Jensen, S.; Landfald, B. Torularhodin and torulene are the major contributors to the carotenoid pool of marine Rhodosporidium babjevae (golubev). J. Ind. Microbiol. Biotechnol. 2006, 33, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Moline, M.; Flores, M.R.; Libkind, D.; Dieguez Mdel, C.; Farias, M.E.; van Broock, M. Photoprotection by carotenoid pigments in the yeast rhodotorula mucilaginosa: The role of torularhodin. Photochem. Photobiol. Sci. 2010, 9, 1145–1151. [Google Scholar] [CrossRef] [PubMed]
- Iigusa, H.; Yoshida, Y.; Hasunuma, K. Oxygen and hydrogen peroxide enhance light-induced carotenoid synthesis in Neurospora crassa. FEBS Lett. 2005, 579, 4012–4016. [Google Scholar] [CrossRef] [PubMed]
- Seiffert, F.; Bandow, N.; Bouchez, J.; von Blanckenburg, F.; Gorbushina, A.A. Microbial colonization of bare rocks: Laboratory biofilm enhances mineral weathering. Procedia Earth Planet. Sci. 2014, 10, 123–129. [Google Scholar] [CrossRef]
- Britton, G.; Liaaen-Jensen, S.; Pfander, H. Carotenoids Handbook; Birkhäuser Verlag: Basel, Switzerland, 2004; ISBN 3764361808. [Google Scholar]
- Foppen, F.H. Tables for the identification of carotenoid pigments. Chromatogr. Rev. 1971, 14, 133–298. [Google Scholar] [CrossRef]
- Choi, S.K.; Kim, J.H.; Park, Y.S.; Kim, Y.J.; Chang, H.I. An efficient method for the extraction of astaxanthin from the red yeast Xanthophyllomyces dendrorhous. J. Microbiol. Biotechnol. 2007, 17, 847–852. [Google Scholar] [PubMed]
- Sedmak, J.J.; Weerasinghe, D.; Jolly, S. Extraction and quantitation of astaxanthin from Phaffia rhodozyma. Biotechnol. Technol. 1990, 4, 107–112. [Google Scholar] [CrossRef]
- Libkind, D.; Perez, P.; Sommaruga, R.; Dieguez, M.D.; Ferraro, M.; Brizzio, S.; Zagarese, H.; van Broock, M. Constitutive and UV-inducible synthesis of photoprotective compounds (carotenoids and mycosporines) by freshwater yeasts. Photochem. Photobiol. Sci. 2004, 3, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Gmoser, R.; Ferreira, J.A.; Lennartsson, P.R.; Taherzadeh, M.J. Filamentous ascomycetes fungi as a source of natural pigments. Fungal Biol. Biotechnol. 2017, 4, 4. [Google Scholar] [CrossRef] [PubMed]
- Zoz, L.; Carvalho, J.C.; Soccol, V.T.; Casagrande, T.C.; Cardoso, L. Torularhodin and torulene: Bioproduction, properties and prospective applications in food and cosmetics—A review. Braz. Arch. Biol. Technol. 2015, 58, 278–288. [Google Scholar] [CrossRef]
- Buzzini, P.; Innocenti, M.; Turchetti, B.; Libkind, D.; van Broock, M.; Mulinacci, N. Carotenoid profiles of yeasts belonging to the genera Rhodotorula, Rhodosporidium, Sporobolomyces, and Sporidiobolus. Can. J. Microbiol. 2007, 53, 1024–1031. [Google Scholar] [CrossRef] [PubMed]
- Avalos, J.; Prado-Cabrero, A.; Estrada, A.F. Neurosporaxanthin production by Neurospora and Fusarium. Methods Mol. Biol. 2012, 898, 263–274. [Google Scholar] [PubMed]
- Albrecht, M.; Takaichi, S.; Steiger, S.; Wang, Z.-Y.; Sandmann, G. Novel hydroxycarotenoids with improved antioxidative properties produced by gene combination in Escherichia coli. Nat. Biotechnol. 2000, 18, 843. [Google Scholar] [CrossRef] [PubMed]
- Olives Barba, A.I.; Camara Hurtado, M.; Sanchez Mata, M.C.; Fernadez Ruiz, V.; Lopez Saenz de Tejada, M. Application of a UV–vis detection-HPLC method for a rapid determination of lycopene and β-carotene in vegetables. Food Chem. 2006, 95, 328–336. [Google Scholar] [CrossRef]
- Busch, M.; Seuter, A.; Hain, R. Functional analysis of the early steps of carotenoid biosynthesis in tobacco. Plant Physiol. 2012, 128, 439–453. [Google Scholar] [CrossRef] [PubMed]
- Schnitzler, N.; Peltroche-Llacsahuanga, H.; Bestier, N.; Zundorf, J.; Lutticken, R.; Haase, G. Effect of melanin and carotenoids of Exophiala (Wangiella) dermatitidis on phagocytosis, oxidative burst, and killing by human neutrophils. Infect. Immun. 1999, 67, 94–101. [Google Scholar] [PubMed]
- Zhang, Z.; Zhang, X.; Tan, T. Lipid and carotenoid production by Rhodotorula glutinis under irradiation/high-temperature and dark/low-temperature cultivation. Bioresour. Technol. 2014, 157, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Arrach, N.; Schmidhauser, T.J.; Avalos, J. Mutants of the carotene cyclase domain of al-2 from Neurospora crassa. Mol. Genet. Genom. 2002, 266, 914–921. [Google Scholar]
- Du, C.; Li, Y.; Guo, Y.; Han, M.; Zhang, W.; Qian, H. The suppression of torulene and torularhodin treatment on the growth of PC-3 xenograft prostate tumors. Biochem. Biophys. Res. Commun. 2016, 469, 1146–1152. [Google Scholar] [CrossRef] [PubMed]
- Sakaki, H.; Nochide, H.; Komemushi, S.; Miki, W. Effect of active oxygen species on the productivity of torularhodin by Rhodotorula glutinis No. 21. J. Biosci. Bioeng. 2002, 93, 338–340. [Google Scholar] [CrossRef]
- Nanou, K.; Roukas, T.; Papadakis, E.; Kotzekidou, P. Carotene production from waste cooking oil by Blakeslea trispora in a bubble column reactor: The role of oxidative stress. Eng. Life Sci. 2017, 17, 775–780. [Google Scholar] [CrossRef]
- Li, C.; Zhang, N.; Li, B.; Xu, Q.; Song, J.; Wei, N.; Wang, W.; Zou, H. Increased torulene accumulation in red yeast Sporidiobolus pararoseus NGR as stress response to high salt conditions. Food Chem. 2017, 237, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Geis, P.A.; Szaniszlo, P.J. Carotenoid-pigments of the dematiaceous fungus Wangiella-dermatitidis. Mycologia 1984, 76, 268–273. [Google Scholar] [CrossRef]
- Madhour, A.; Anke, H.; Mucci, A.; Davoli, P.; Weber, R.W.S. Biosynthesis of the xanthophyll plectaniaxanthin as a stress response in the red yeast Dioszegia (Tremellales, Heterobasidiomycetes, fungi). Phytochemistry 2005, 66, 2617–2626. [Google Scholar] [CrossRef] [PubMed]

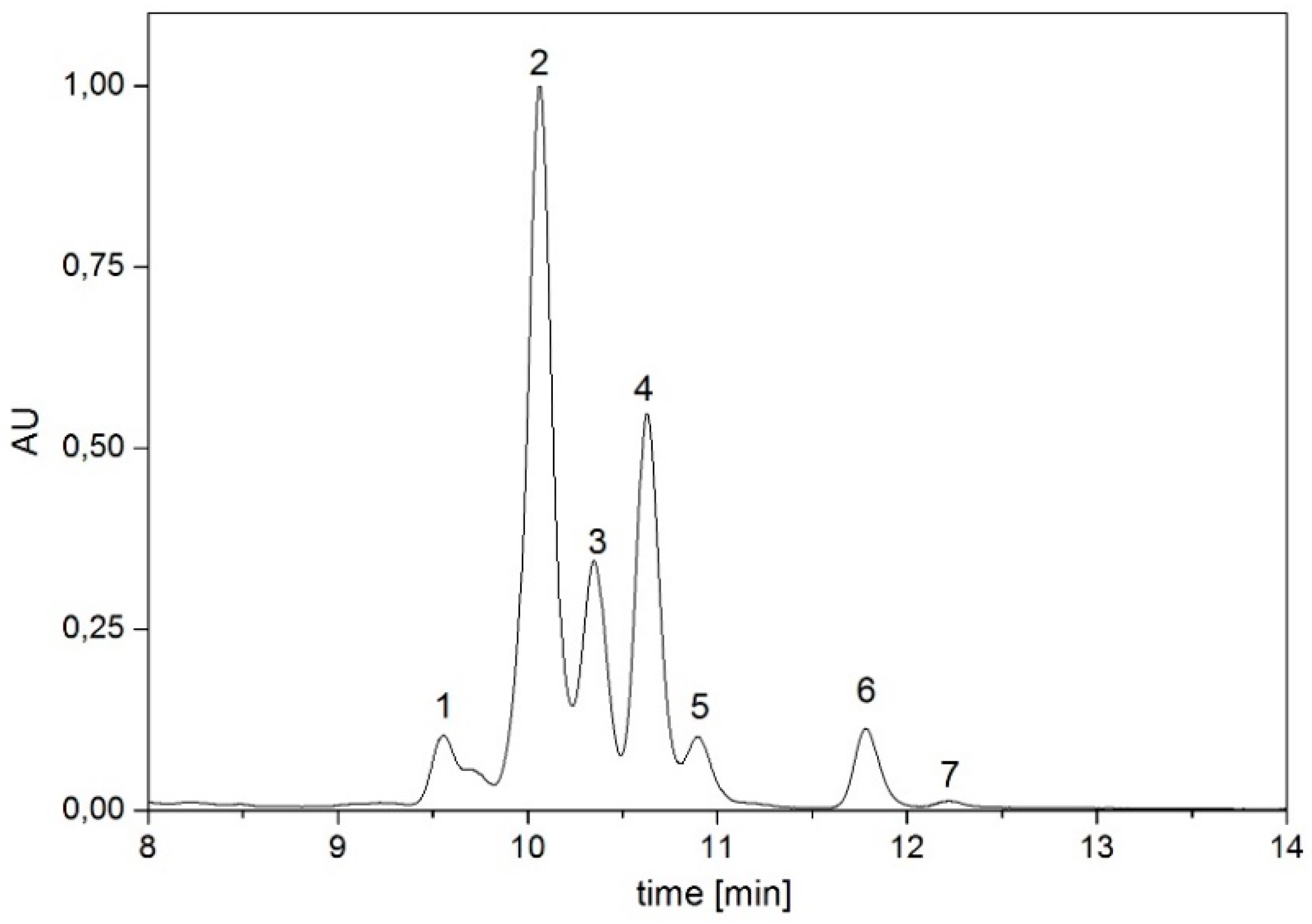
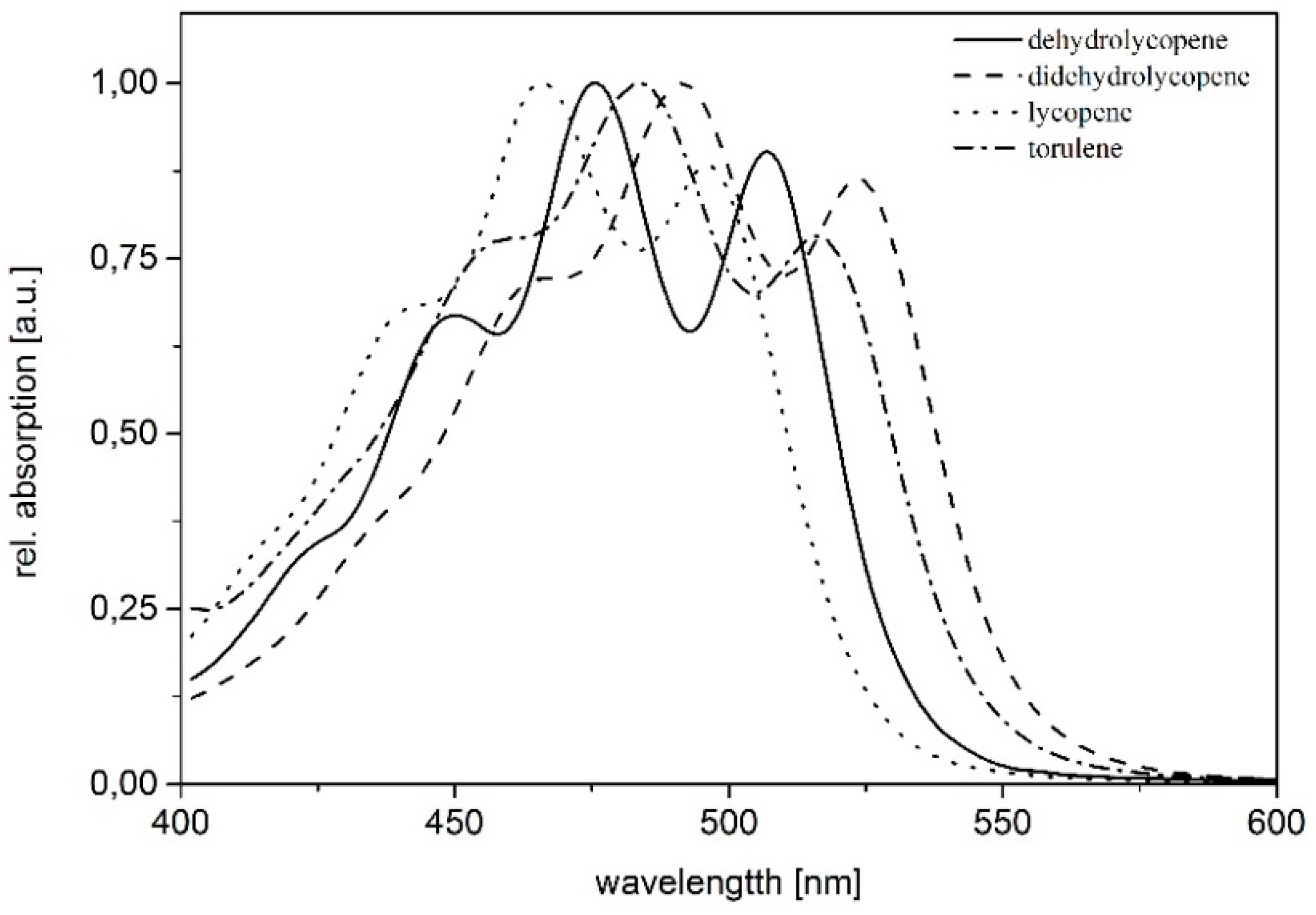
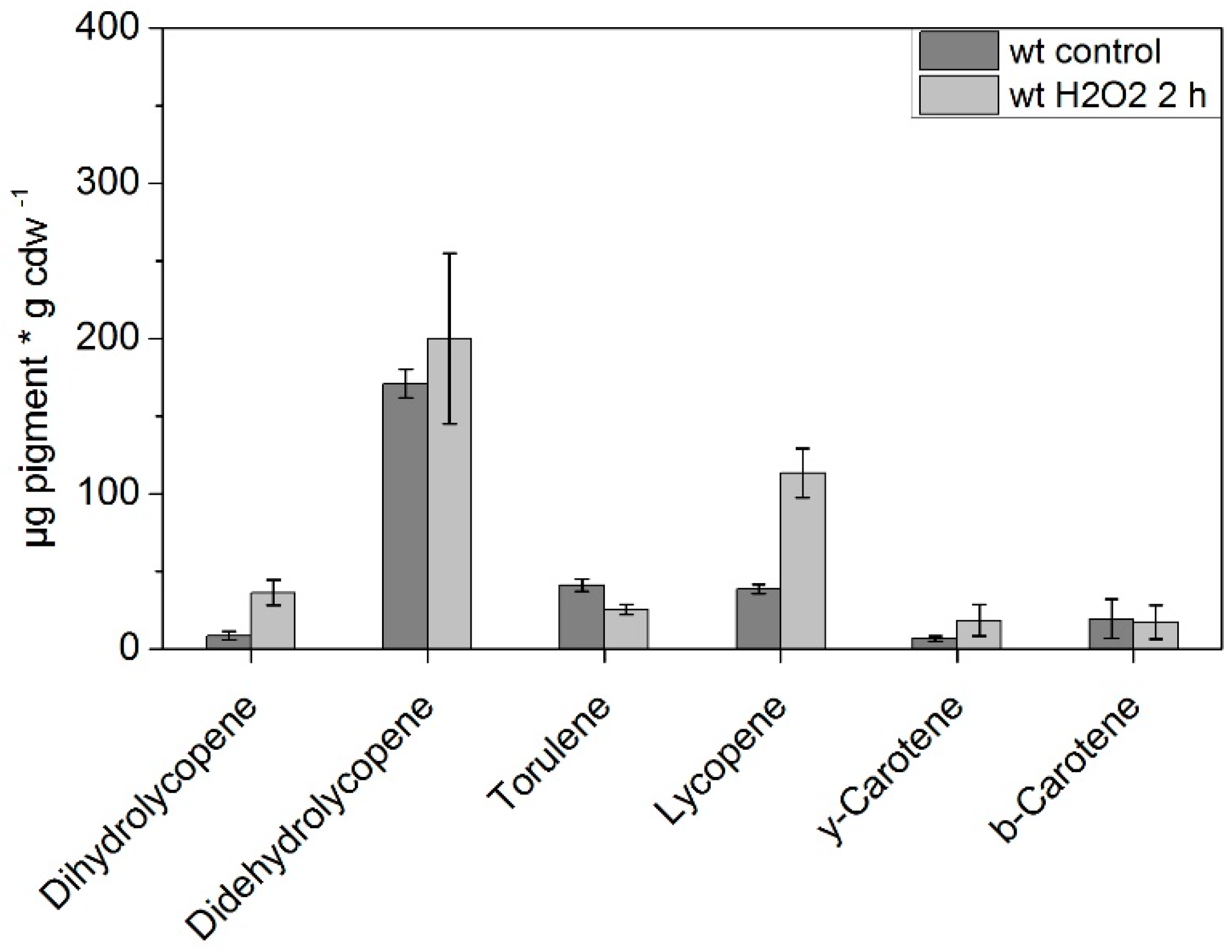
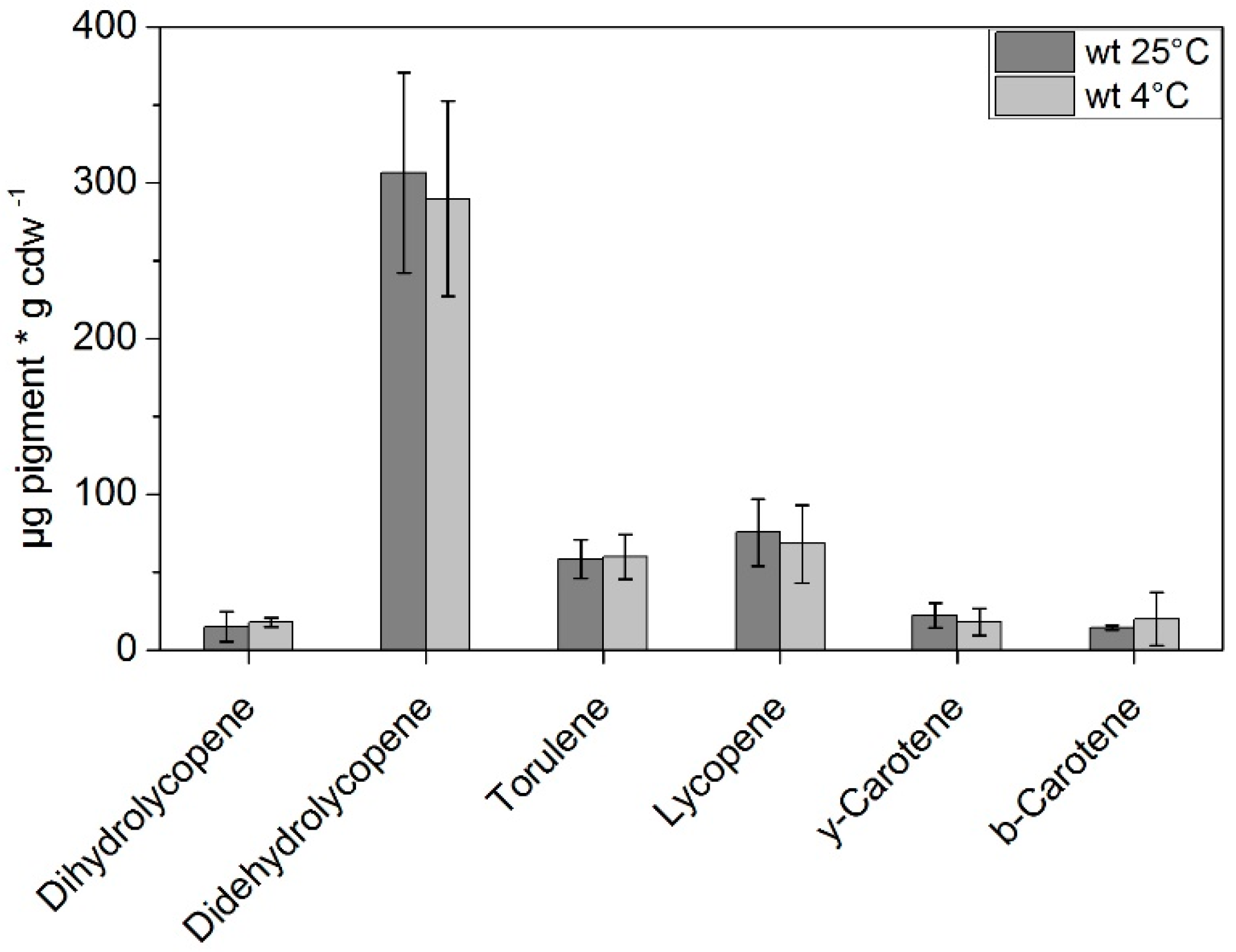
| Time [min] | Eluent A | Eluent B |
|---|---|---|
| 0 | 60% | 40% |
| 7 | 50% | 50% |
| 17 | 40% | 60% |
| 21 | 30% | 70% |
| 28.5 | 20% | 80% |
| 29.5 | 10% | 90% |
| 30.5 to 42 min | 60% | 40% |
| Pigment | Absorption Maxima [nm] | Concentration [µg/g dry weight] |
|---|---|---|
| dihydrolycopene | 448, 476, 507 | 30.74 |
| didehydrolycopene | 463, 492, 523 | 383.12 |
| torulene | 454, 484, 515 | 85.39 |
| lycopene | 440, 465, 496 | 100.30 |
| γ-carotene | 460, 489 | 23.88 |
| β-carotene | 457, 483 | 7.53 |
| torularhodin | 469, 497, 525 | 5.84 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flieger, K.; Knabe, N.; Toepel, J. Development of an Improved Carotenoid Extraction Method to Characterize the Carotenoid Composition under Oxidative Stress and Cold Temperature in the Rock Inhabiting Fungus Knufia petricola A95. J. Fungi 2018, 4, 124. https://doi.org/10.3390/jof4040124
Flieger K, Knabe N, Toepel J. Development of an Improved Carotenoid Extraction Method to Characterize the Carotenoid Composition under Oxidative Stress and Cold Temperature in the Rock Inhabiting Fungus Knufia petricola A95. Journal of Fungi. 2018; 4(4):124. https://doi.org/10.3390/jof4040124
Chicago/Turabian StyleFlieger, Kerstin, Nicole Knabe, and Jörg Toepel. 2018. "Development of an Improved Carotenoid Extraction Method to Characterize the Carotenoid Composition under Oxidative Stress and Cold Temperature in the Rock Inhabiting Fungus Knufia petricola A95" Journal of Fungi 4, no. 4: 124. https://doi.org/10.3390/jof4040124
APA StyleFlieger, K., Knabe, N., & Toepel, J. (2018). Development of an Improved Carotenoid Extraction Method to Characterize the Carotenoid Composition under Oxidative Stress and Cold Temperature in the Rock Inhabiting Fungus Knufia petricola A95. Journal of Fungi, 4(4), 124. https://doi.org/10.3390/jof4040124




