Analysis of Microbial Community Structure and Cultivation Performance Assessment in Tremella fuciformis Liquid Inoculum
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Strains and Culture Conditions
2.3. The Method of T. fuciformis Liquefied Spawn Preparation
2.4. Physiological Parameter Assay of T. fuciformis Liquefied Spawn
2.5. Microbial Community Structure Analysis
2.6. Preparation of T. fuciformis Liquid Spawn
2.7. Cultivation Tests
2.7.1. Cultivation Test of Liquefied Spawn
2.7.2. Cultivation Test of Liquid Spawn
2.8. Statistical Analysis
3. Results and Discussion
3.1. Preparation of T. fuciformis Liquefied Spawn
3.2. Quality Dynamics of T. fuciformis Liquefied Spawn During the Cultivation Process
3.3. Comparative Analysis of Microbial Community Shifts During Liquefied Spawn Browning
3.4. Cultivation Performance of T. fuciformis Liquefied Spawn
3.5. Preparation of T. fuciformis Liquid Spawn and Physiological Parameter Assay
3.6. Cultivation Test of T. fuciformis Liquid Spawn
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Deng, Y.; Guo, L.; Lin, L.; Li, Y.; Zhang, J.; Zhang, Y.; Yuan, B.; Ke, L.; Xie, B.; Ming, R. Meiosis in an asymmetric dikaryotic genome of Tremella fuciformis Tr01 facilitates new chromosome formation. Genome Biol. 2023, 24, 280. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhan, G.; Tu, M.; Wang, Y.; Cao, J.; Sun, S. A chromosome-scale genome and proteome draft of Tremella fuciformis. Int. J. Biol. Macromol. 2023, 247, 125749. [Google Scholar] [CrossRef]
- Li, Y.; Li, J.; Fan, L. Effects of combined drying methods on physicochemical and rheological properties of instant Tremella fuciformis soup. Food Chem. 2022, 396, 133644. [Google Scholar] [CrossRef]
- Xu, Y.; Xie, L.; Zhang, Z.; Zhang, W.; Tang, J.; He, X.; Zhou, J.; Peng, W. Tremella fuciformis Polysaccharides Inhibited Colonic Inflammation in Dextran Sulfate Sodium-Treated Mice via Foxp3+ T Cells, Gut Microbiota, and Bacterial Metabolites. Front. Immunol. 2021, 12, 648162. [Google Scholar] [CrossRef]
- Zhu, H.; Tian, B.; Liu, W.; Zhang, S.; Cao, C.; Zhang, Y.; Zou, W. A three-stage culture process for improved exopolysaccharide production by Tremella fuciformis. Bioresour. Technol. 2012, 116, 526–528. [Google Scholar] [CrossRef]
- Xie, L.; Yang, K.; Liang, Y.; Zhu, Z.; Yuan, Z.; Du, Z. Tremella fuciformis polysaccharides alleviate induced atopic dermatitis in mice by regulating immune response and gut microbiota. Front. Pharmacol. 2022, 13, 944801. [Google Scholar] [CrossRef]
- Wu, Y.J.; Wei, Z.X.; Zhang, F.M.; Linhardt, R.J.; Sun, P.L.; Zhang, A.Q. Structure, bioactivities and applications of the polysaccharides from Tremella fuciformis mushroom: A review. Int. J. Biol. Macromol. 2019, 121, 1005–1010. [Google Scholar] [CrossRef]
- Lo, H.C.; Wasser, S.P. Medicinal mushrooms for glycemic control in diabetes mellitus: History, current status, future perspectives, and unsolved problems (review). Int. J. Med. Mushrooms 2011, 13, 401–426. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.L.; Chen, A.W. Mixed-Culture Cultivation of Edible and Medicinal Mushroom Tremella fudformis Berk. (Phragmobasidiomycetes) on Synthetic Logs. Int. J. Med. Mushrooms 2002, 4, 6. [Google Scholar] [CrossRef]
- Tong, Q.; Yang, L.; Zhang, J.; Zhang, Y.; Jiang, Y.; Liu, X.; Deng, Y. Comprehensive investigations of 2-phenylethanol production by the filamentous fungus Annulohypoxylon stygium. Appl. Microbiol. Biotechnol. 2024, 108, 374. [Google Scholar] [CrossRef] [PubMed]
- Wingfield, B.D.; Bills, G.F.; Dong, Y.; Huang, W.; Nel, W.J.; Swalarsk-Parry, B.S.; Vaghefi, N.; Wilken, P.M.; An, Z.; de Beer, Z.W.; et al. IMA Genome-F 9: Draft genome sequence of Annulohypoxylon stygium, Aspergillus mulundensis, Berkeleyomyces basicola (syn. Thielaviopsis basicola), Ceratocystis smalleyi, two Cercospora beticola strains, Coleophoma cylindrospora, Fusarium fracticaudum, Phialophora cf. hyalina, and Morchella septimelata. IMA Fungus 2018, 9, 199–223. [Google Scholar]
- Deng, Y.; Hsiang, T.; Li, S.; Lin, L.; Wang, Q.; Chen, Q.; Xie, B.; Ming, R. Comparison of the Mitochondrial Genome Sequences of Six Annulohypoxylon stygium Isolates Suggests Short Fragment Insertions as a Potential Factor Leading to Larger Genomic Size. Front. Microbiol. 2018, 9, 2079. [Google Scholar] [CrossRef] [PubMed]
- Berovic, M.; Zhong, J.J. Advances in Pilot-Scale Stirred Bioreactors in Solid-State and Submerged Cultivations of Medicinal Mushrooms. Adv. Biochem. Eng. Biotechnol. 2023, 184, 163–185. [Google Scholar]
- Ma, L.; Lin, Y.Q.; Yang, C.; Ying, Z.H.; Jiang, X.L. Production of liquid spawn of an edible mushroom, Sparassis latifolia by submerged fermentation and mycelial growth on pine wood sawdust. Sci. Hortic. 2016, 209, 22–30. [Google Scholar] [CrossRef]
- Pérez, G.; Lopez-Moya, F.; Chuina, E.; Ibañez-Vea, M.; Garde, E.; López-Llorca, L.V.; Pisabarro, A.G.; Ramírez, L. Strain Degeneration in Pleurotus ostreatus: A Genotype Dependent Oxidative Stress Process Which Triggers Oxidative Stress, Cellular Detoxifying and Cell Wall Reshaping Genes. J. Fungi 2021, 7, 862. [Google Scholar] [CrossRef]
- Friel, M.; Mcloughlin, A.J. Production of a liquid inoculum/spawn of Agaricus bisporus. Biotechnol. Lett. 2000, 22, 351–354. [Google Scholar] [CrossRef]
- Zhang, W.R.; Liu, S.R.; Kuang, Y.B.; Zheng, S.Z. Development of a Novel Spawn (Block Spawn) of an Edible Mushroom, Pleurotus ostreatus, in Liquid Culture and its Cultivation Evaluation. Mycobiology 2019, 47, 97–104. [Google Scholar] [CrossRef]
- Kawai, G.; Kobayashi, H.; Fukushima, Y.; Ohsaki, K. Effect of liquid mycelial culture used as a spawn on sawdust cultivation of shiitake (Lentinula edodes). Mycoscience 1996, 37, 201–207. [Google Scholar] [CrossRef]
- Zhang, R.Y.; Hu, D.D.; Ma, X.T.; Li, S.G.; Gu, J.G.; Hu, Q.X. Adopting stick spawn reduced the spawn running time and improved mushroom yield and biological efficiency of Pleurotus eryngii. Sci. Hortic. 2014, 175, 156–159. [Google Scholar] [CrossRef]
- Guo, J.; Liu, Y.F.; Tang, C.H.; Zhang, J.S.; Feng, J. The key technologies of Ganoderma lucidum liquid spawn preparation and scale expansion. World J. Microbiol. Biotechnol. 2023, 39, 138. [Google Scholar] [CrossRef]
- Kim, J.M.; Ra, K.S.; Noh, D.O.; Suh, H.J. Optimization of submerged culture conditions for the production of angiotensin converting enzyme inhibitor from Flammulina velutipes. J. Ind. Microbiol. Biotechnol. 2002, 29, 292–295. [Google Scholar] [CrossRef]
- Ren, H.H.; Zhang, Y.J.; Wang, Y.J.; Xiao, S.S.; Liu, S.Y.; Sun, B.Y.; Li, S.M.; Li, M.; Wen, Z.Q.; Li, X. Advancing Industrial Production of White Grifola frondosa: Liquid Inoculum Culture Parameter Optimization and Molecular Insights into Fruiting Body Development. Horticulturae 2025, 11, 1151. [Google Scholar] [CrossRef]
- Zhu, H.; Sun, X.; Liu, D.; Zheng, L.; Chen, L.; Ma, A. An Improved Total RNA Extraction Method for White Jelly Mushroom Tremella fuciformis Rich in Polysaccharides. Mycobiology 2017, 45, 434–437. [Google Scholar] [CrossRef]
- Zhu, H.; Yuan, Y.; Liu, J.; Zheng, L.; Chen, L.; Ma, A. Comparing the sugar profiles and primary structures of alkali-extracted water-soluble polysaccharides in cell wall between the yeast and mycelial phases from Tremella fuciformis. J. Microbiol. 2016, 54, 381–386. [Google Scholar] [CrossRef]
- Tang, Q.Q.; Zou, D.R.; Shi, G.P. Contrast Test on Pleurotus eryngii Cultivation of Liquefied Solid Spawn and Solid Spawn. Edible Fungi China 2018, 37, 16–45. [Google Scholar]
- Lombardi, V.; Trande, M.; Back, M.; Patwardhan, S.V.; Benedetti, A. Facile Cellulase Immobilisation on Bioinspired Silica. Nanomaterials 2022, 12, 626. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Y.; Cao, L.; Huang, Z.; Zhou, Y.; Fan, R.; Li, C. Preparation and Characterization of Intracellular and Exopolysaccharides during Cycle Cultivation of Spirulina platensis. Foods 2023, 12, 1067. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Mo, C.; Meng, X.; Wei, X.; Ma, A. Production, physico-chemical properties and antioxidant activity of melanin from Annulohypoxylon stygium (Lév.). Nat. Prod. Res. 2025, 39, 3021–3030. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, J.; Zhou, Y.; Liu, K.; Lu, Y.; Zhu, L.; Chen, X. Microbial community structure and interactions between Aspergillus oryzae and bacteria in traditional solid-state fermentation of Jiangqu. Food Microbiol. 2023, 116, 104346. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Li, D.; Ai, M.; Tang, Q.; Huang, J.; Ding, X.; Wu, C.; Zhou, R. Correlation between volatile profiles and microbial communities: A metabonomic approach to study Jiang-flavor liquor Daqu. Food Res. Int. 2019, 121, 422–432. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, W.R.; Kuang, Y.B. Production of stalk spawn of an edible mushroom (Pleurotus ostreatus) in liquid culture as a suitable substitute for stick spawn in mushroom cultivation. Sci. Hortic. 2018, 240, 572–577. [Google Scholar] [CrossRef]
- Abdullah, N.; Ismail, R.; Johari, N.M.K.; Annuar, M.S.M. Production of liquid spawn of an edible grey oyster mushroom, Pleurotus pulmonarius (Fr.) Quél by submerged fermentation and sporophore yield on rubber wood sawdust. Sci. Hortic. 2013, 161, 65–69. [Google Scholar] [CrossRef]
- Wang, L.Y.; Cheong, K.L.; Wu, D.T.; Meng, L.Z.; Zhao, J.; Li, S.P. Fermentation optimization for the production of bioactive polysaccharides from Cordyceps sinensis fungus UM01. Int. J. Biol. Macromol. 2015, 79, 180–185. [Google Scholar] [CrossRef]
- Avila, I.A.F.; da Silva Alves, L.; Zied, D.C. Bioconversion of rice straw by Lentinula edodes under different spawn formulations. Braz. J. Microbiol. 2023, 54, 3137–3146. [Google Scholar] [CrossRef]
- Lu, F.; Wang, Z.; Zhao, W.; Chu, J.; Zhuang, Y. A simple novel approach for real-time monitoring of sodium gluconate production by on-line physiological parameters in batch fermentation by Aspergillus niger. Bioresour. Technol. 2016, 202, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Zalai, D.; Hevér, H.; Lovász, K.; Molnár, D.; Wechselberger, P.; Hofer, A.; Párta, L.; Putics, Á.; Herwig, C. A control strategy to investigate the relationship between specific productivity and high-mannose glycoforms in CHO cells. Appl. Microbiol. Biotechnol. 2016, 100, 7011–7024. [Google Scholar] [CrossRef]
- Gao, J.; Wenderoth, M.; Doppler, M.; Schuhmacher, R.; Marko, D.; Fischer, R. Fungal Melanin Biosynthesis Pathway as Source for Fungal Toxins. mBio 2022, 13, e0021922. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, X.; Mao, Y.; Blaschek, H.P. Genome-wide dynamic transcriptional profiling in Clostridium beijerinckii NCIMB 8052 using single-nucleotide resolution RNA-Seq. BMC Genom. 2012, 13, 102. [Google Scholar] [CrossRef]
- Lobeda, K.; Jin, Q.; Wu, J.; Zhang, W.; Huang, H. Lactic acid production from food waste hydrolysate by Lactobacillus pentosus: Focus on nitrogen supplementation, initial sugar concentration, pH, and fed-batch fermentation. J. Food Sci. 2022, 87, 3071–3083. [Google Scholar] [CrossRef]
- Liu, D.; Pujiana, D.; Wang, Y.; Zhang, Z.; Zheng, L.; Chen, L.; Ma, A. Comparative transcriptomic analysis identified differentially expressed genes and pathways involved in the interaction between Tremella fuciformis and Annulohypoxylon stygium. Anton. Leeuw. Int. J. G. 2019, 112, 1675–1689. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Li, F.; Xu, X.; Liu, Y.; Kong, X.; Chen, J.; Liu, T.; Chen, L. Study on the community structure and function of symbiotic bacteria from different growth and developmental stages of Hypsizygus marmoreus. BMC Microbiol. 2020, 20, 311. [Google Scholar] [CrossRef]
- Xu, J.; Zou, Y.; Guo, L.; Lin, J.; Jiang, Z.; Zheng, Q. Rheological and microstructural properties of polysaccharide obtained from the gelatinous Tremella fuciformis fungus. Int. J. Biol. Macromol. 2023, 228, 153–164. [Google Scholar] [CrossRef]
- Liu, D.; Sun, X.; Yan, B.; Ma, A. Alternative oxidase is involved in oxidative stress resistance and melanin synthesis in Annulohypoxylon stygium, a companion fungus of Tremella fuciformis. Anton. Leeuw. Int. J. G. 2022, 115, 365–374. [Google Scholar] [CrossRef]
- Ahsan, T.; Mahnoor; Alharbi, S.A.; Alshiekheid, M.A.; Sabour, A.A.A.; Khan, I.; Ansari, M.J. Genome Mining and Antagonism of Stenotrophomonas geniculata MK-1, Against Peanut Foliage Fungus Diseases. J. Basic. Microbiol. 2025, 65, e70016. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Hu, J.; Chi, J.; Li, Y.; Yang, B.; Hu, W.; Chen, F.; Xu, C.; Chai, L.; Bao, Y. Label-Free Proteomics Reveals the Molecular Mechanism of Subculture Induced Strain Degeneration and Discovery of Indicative Index for Degeneration in Pleurotus ostreatus. Molecules 2020, 25, 4920. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Dong, Q.; Zhang, S.; Xie, L.; Zhang, Q.; Shu, X.; Zhou, J.; Peng, W. Lignocellulose adaptation drives polysaccharide biosynthesis in Tremella fuciformis: Metabolomic and proteomic insights into CAZyme regulation. Front. Fungal. Biol. 2025, 6, 1617458. [Google Scholar] [CrossRef] [PubMed]
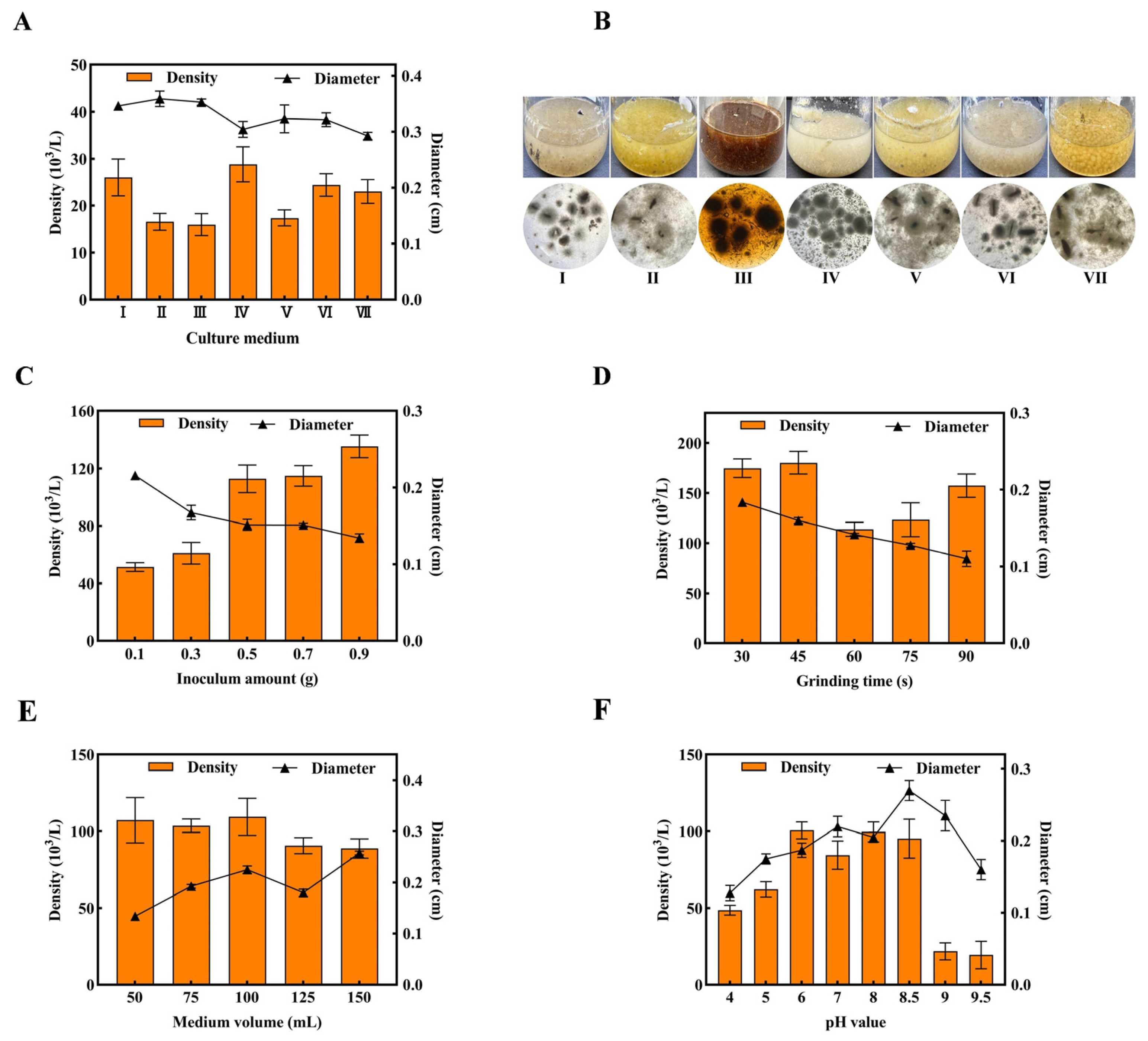





| No. | Formulation (g/L) |
|---|---|
| I | Potato 200, KH2PO4 3, Glucose 20, MgSO4 1.5, Thiamine HCl 0.01; |
| II | Potato 200, Glucose 20, Peptone 5, KH2PO4 2, Mg SO4 1, Thiamine HCl 0.01; |
| III | Potato 200, Sawdust 50, Glucose 20, Peptone 6, KH2PO4 0.5, Thiamine HCl 0.01; |
| IV | Corn flour 10, Soybean flour 7, Glucose 20, Yeast extract 1, Peptone 1, KH2PO4 0.5, MgSO4 0.5; |
| V | Potato 200, Glucose 20, KH2PO4 3, Peptone 3, K2HPO4 3, MgSO4 1, Thiamine HCl 0.01, Corn flour 1; |
| VI | Potato 200, Yeast extract 3, Glucose 20, MgSO4 1, KH2PO4 1; |
| VII | Glucose 30, KH2PO4 0.5, Peptone 8.3, MgSO4·7H2O 3, Thiamine HCl 0.01. |
| Batch | Yield (g) | Fruiting Ratio (%) | Diameter (mm) | Height (mm) |
|---|---|---|---|---|
| X1 | 549.8 ± 11.0 a | 96 | 129.0 ± 7.9 a | 65.9 ± 2.4 a |
| X2 | 489.2 ± 11.9 d | 98 | 116.9 ± 14.6 c | 60.7 ± 3.1 c |
| X3 | 510.6 ± 16.2 c | 98 | 118.6 ± 11.8 c | 62.1 ± 3.9 bc |
| X4 | 537.9 ± 5.6 b | 98 | 122.5 ± 9.6 b | 64.3 ± 4.3 ab |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, H.; Xiong, Q.; Huang, W.; Dai, X.; Yang, Y.; Huang, W.; Lai, S.; Sun, S.; Zhang, L. Analysis of Microbial Community Structure and Cultivation Performance Assessment in Tremella fuciformis Liquid Inoculum. J. Fungi 2025, 11, 825. https://doi.org/10.3390/jof11120825
Lin H, Xiong Q, Huang W, Dai X, Yang Y, Huang W, Lai S, Sun S, Zhang L. Analysis of Microbial Community Structure and Cultivation Performance Assessment in Tremella fuciformis Liquid Inoculum. Journal of Fungi. 2025; 11(12):825. https://doi.org/10.3390/jof11120825
Chicago/Turabian StyleLin, Hui, Qi Xiong, Wenxuan Huang, Xinghua Dai, Yingxi Yang, Wenlin Huang, Shufang Lai, Shujing Sun, and Liaoyuan Zhang. 2025. "Analysis of Microbial Community Structure and Cultivation Performance Assessment in Tremella fuciformis Liquid Inoculum" Journal of Fungi 11, no. 12: 825. https://doi.org/10.3390/jof11120825
APA StyleLin, H., Xiong, Q., Huang, W., Dai, X., Yang, Y., Huang, W., Lai, S., Sun, S., & Zhang, L. (2025). Analysis of Microbial Community Structure and Cultivation Performance Assessment in Tremella fuciformis Liquid Inoculum. Journal of Fungi, 11(12), 825. https://doi.org/10.3390/jof11120825






