Study on NGF and VEGF during the Equine Perinatal Period—Part 1: Healthy Foals Born from Normal Pregnancy and Parturition
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Population
2.2. Clinical Data and Sample Collection
2.3. Measurement of NGF and VEGF by ELISA
2.4. Measurement of Thyroid Hormones
2.5. Placental Gene Expression
2.6. Statistical Analysis
3. Results
3.1. Population Characterization
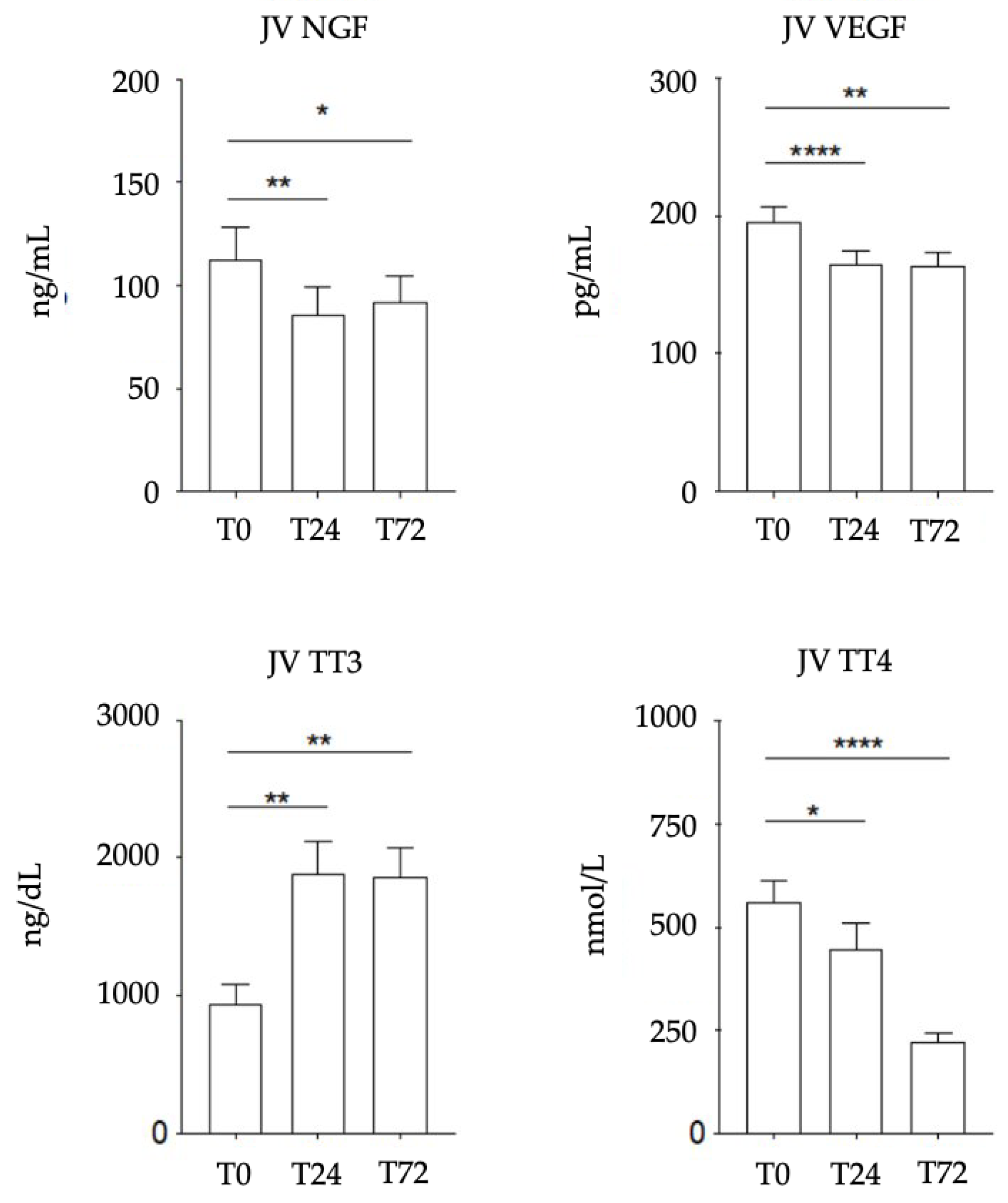
3.2. Biomarkers (NGF, VEGF, TT3 and TT4) in Biological Fluids
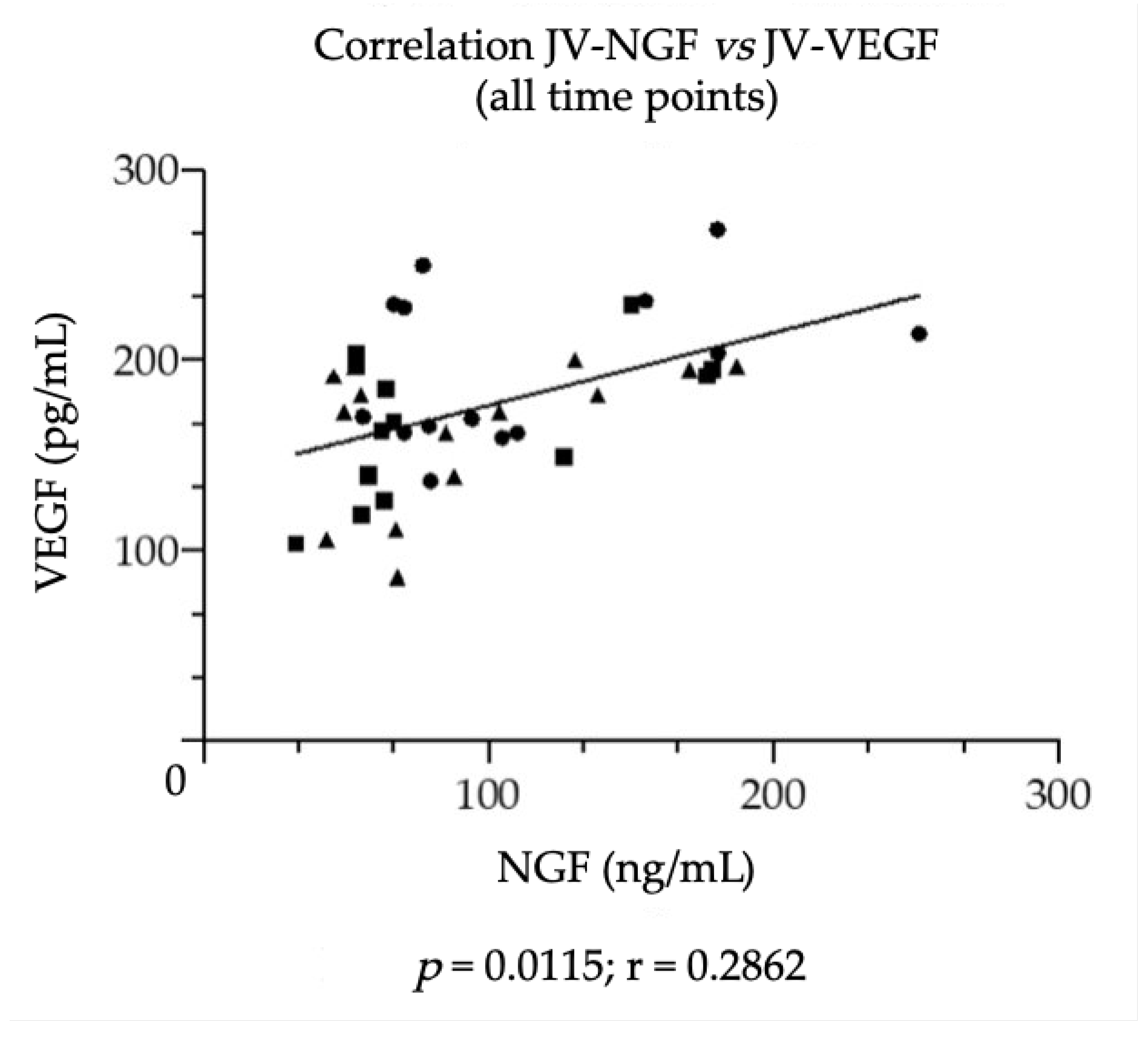
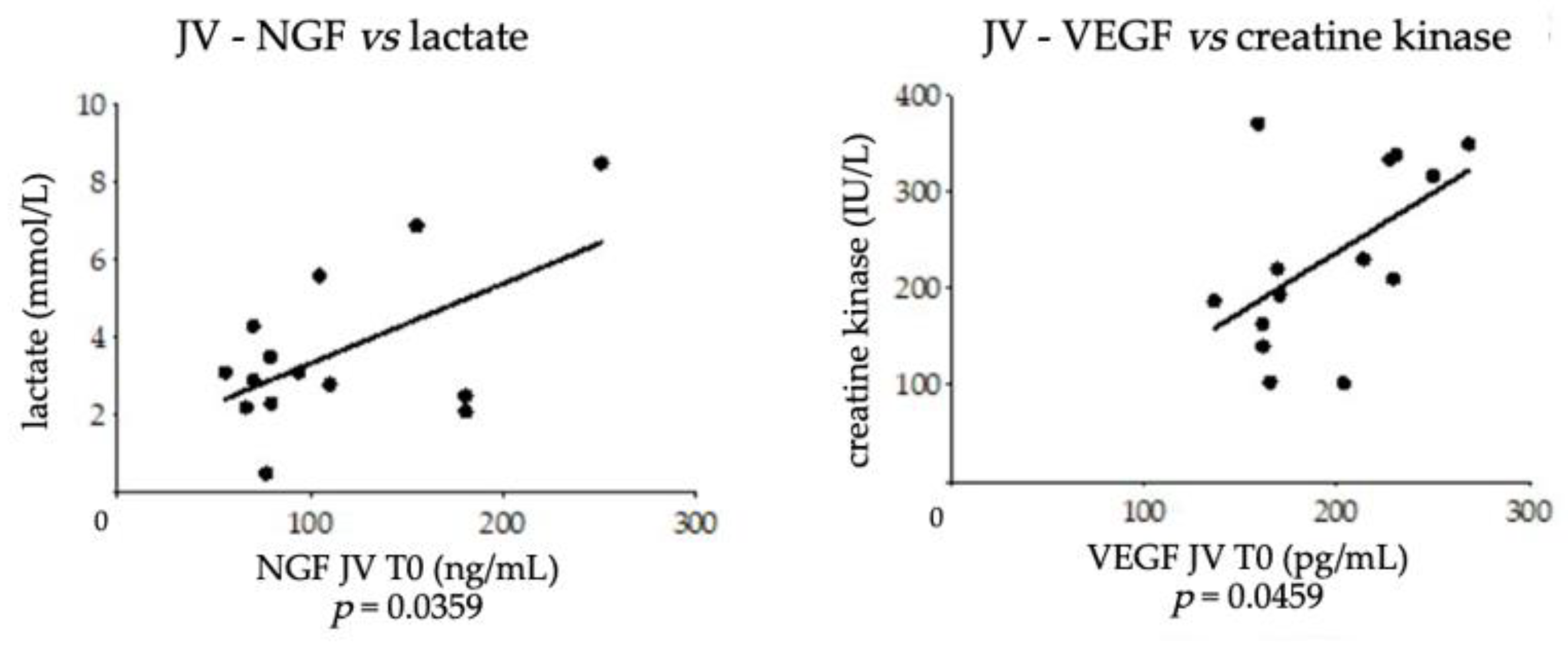
3.3. Equine NGF/VEGF and Clinical Data
3.4. Equine NGF, VEGF, BDNF and Their Receptors Gene Expression in the Placenta
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tucker, K.L.; Meyer, M.; Barde, Y. Neurotrophins are required for nerve growth during development. Nat. Neurosci. 2001, 4, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Chao, M.V. Trophic factors: An evolutionary cul-de-sac or door into higher neuronal function? J. Neurosci. Res. 2000, 59, 353–355. [Google Scholar] [CrossRef]
- Lu, B.; Figurov, A. Role of Neurotrophins in Synapse Development and Plasticity. Rev. Neurosci. 1997, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Takei, N.; Nawa, H. Roles of neurotrophins on synaptic development and functions in the central nervous system. Hum. Cell 1998, 11, 157–165. [Google Scholar] [PubMed]
- Frade, J.M.; Rodríguez-Tébar, A.; Barde, Y.-A. Induction of cell death by endogenous nerve growth factor through its p75 receptor. Nature 1996, 383, 166–168. [Google Scholar] [CrossRef] [PubMed]
- Stadelmann, C.; Kerschensteiner, M.; Misgeld, T.; Brück, W.; Hohlfeld, R.; Lassmann, H. BDNF and gp145trkB in multiple sclerosis brain lesions: Neuroprotective interactions between immune and neuronal cells? Brain 2002, 125, 75–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skaper, S.D. Nerve growth factor: A neuroimmune crosstalk mediator for all seasons. Immunology 2017, 151, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Calzà, L.; Giardino, L.; Aloe, L. NGF content and expression in the rat pituitary gland and regulation by thyroid hormone. Mol. Brain Res. 1997, 51, 60–68. [Google Scholar] [CrossRef]
- Gostynska, N.; Pannella, M.; Rocco, M.L.; Giardino, L.; Aloe, L.; Calza, L. The pleiotropic molecule NGF regulates the in vitro properties of fibroblasts, keratinocytes, and endothelial cells: Implications for wound healing. Am. J. Physiol. Cell Physiol. 2020, 318, C360–C371. [Google Scholar] [CrossRef]
- Pius-Sadowska, E.; Machaliński, B. Pleiotropic activity of nerve growth factor in regulating cardiac functions and counteracting pathogenesis. ESC Heart Fail. 2021, 8, 974–987. [Google Scholar] [CrossRef]
- Amagai, Y.; Sato, H.; Ishizaka, S.; Matsuda, K.; Aurich, C.; Tanaka, A.; Matsuda, H. Cloning and Expression of Equine β-Nerve Growth Factor. J. Equine Vet. Sci. 2016, 45, 28–31. [Google Scholar] [CrossRef]
- Charnock-Jones, D.; Kaufmann, P.; Mayhew, T. Aspects of Human Fetoplacental Vasculogenesis and Angiogenesis. I. Molecular Regulation. Placenta 2004, 25, 103–113. [Google Scholar] [CrossRef]
- Allen, W.; Gower, S.; Wilsher, S. Immunohistochemical Localization of Vascular Endothelial Growth Factor (VEGF) and its Two Receptors (Flt-I and KDR) in the Endometrium and Placenta of the Mare During the Oestrous Cycle and Pregnancy. Reprod. Domest. Anim. 2007, 42, 516–526. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, F.; Nagai, N.; Tang, Z.; Zhang, S.; Scotney, P.; Lennartsson, J.; Zhu, C.; Qu, Y.; Fang, C.; et al. VEGF-B inhibits apoptosis via VEGFR-1–mediated suppression of the expression of BH3-only protein genes in mice and rats. J. Clin. Investig. 2008, 118, 913–923. [Google Scholar] [CrossRef] [Green Version]
- Jin, K.; Zhu, Y.; Sun, Y.; Mao, X.O.; Xie, L.; Greenberg, D.A. Vascular endothelial growth factor (VEGF) stimulates neurogenesis in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 11946–11950. [Google Scholar] [CrossRef] [Green Version]
- Sawicka-Gutaj, N.; Zawalna, N.; Gut, P.; Ruchała, M. Relationship between thyroid hormones and central nervous system metabolism in physiological and pathological conditions. Pharmacol. Rep. 2022, 1–12. [Google Scholar] [CrossRef]
- Calza, L.; Fernandez, M.; Giuliani, A.; Aloe, L.; Giardino, L. Thyroid hormone activates oligodendrocyte precursors and increases a myelin-forming protein and NGF content in the spinal cord during experimental allergic encephalomyelitis. Proc. Natl. Acad. Sci. USA 2002, 99, 3258–3263. [Google Scholar] [CrossRef] [Green Version]
- Santschi, E.M.; Vaala, W.E. Identification of the high-risk pregnancy. In Equine Reproduction; McKinnon, A.O., Squires, E.L., Vaala, W.E., Varner, D.D., Eds.; Wiley-Blackwell: Oxford, UK, 2011; pp. 5–15. [Google Scholar]
- Lanci, A.; Mariella, J.; Ellero, N.; Faoro, A.; Peric, T.; Prandi, A.; Freccero, F.; Castagnetti, C. Hair Cortisol and DHEA-S in Foals and Mares as a Retrospective Picture of Feto-Maternal Relationship under Physiological and Pathological Conditions. Animals 2022, 12, 1266. [Google Scholar] [CrossRef]
- Frazer, G.S.; Perkins, N.R.; Embertson, R.M. Normal parturition and evaluation of the mare in dystocia. Equine Vet. Educ. 1999, 11, 41–46. [Google Scholar] [CrossRef]
- Vaala, W.E.; Sertich, P.L. Management Strategies for Mares at Risk for Periparturient Complications. Vet. Clin. N. Am. Equine Pract. 1994, 10, 237–265. [Google Scholar] [CrossRef]
- Ellero, N.; Lanci, A.; Ferlizza, E.; Andreani, G.; Mariella, J.; Isani, G.; Castagnetti, C. Activities of matrix metalloproteinase-2 and -9 in amniotic fluid at parturition in mares with normal and high-risk pregnancy. Theriogenology 2021, 172, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Vaala, W.E.; House, J.K.; Madigan, J.E. Initial management and physical examination of the neonate. In Large Animal Internal Medicine; Mosby: St. Louis, MO, USA, 2002; pp. 277–293. [Google Scholar]
- Bianco, C.; Pirrone, A.; Boldini, S.; Sarli, G.; Castagnetti, C. Histomorphometric parameters and fractal complexity of the equine placenta from healthy and sick foals. Theriogenology 2014, 82, 1106–1112.e1. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Rodriguez, M.F.; Cymbaluk, N.; Epp, T.; Laarveld, B.; Thrasher, M.; Card, C. A Field Study of Serum, Colostrum, Milk Iodine, and Thyroid Hormone Concentrations in Postpartum Draft Mares and Foals. J. Equine Vet. Sci. 2020, 90, 103018. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Rodriguez, M.F.; Cymbaluk, N.; Epp, T.; Laarveld, B.; Recalde, E.C.S.; Simko, E.; Card, C. Effects of the glucosinolate sinigrin in combination with a noniodine supplemented diet on serum iodine and thyroid hormone concentrations in nonpregnant mares. J. Equine Vet. Sci. 2020, 91, 103110. [Google Scholar] [CrossRef]
- Harvey, J.W. Normal hematologic values. In Equine Clinical Neonatology; Koterba, A.M., Drummond, W.H., Kosch, P.C., Eds.; Lea and Febiger: Philadelphia, PA, USA, 1990; pp. 561–570. [Google Scholar]
- Bauer, J.E.; Harvey, J.W.; Asquith, R.L.; McNulty, P.K.; Kivipelto, J. Clinical chemistry reference values of foals during the first year of life. Equine Vet. J. 1984, 16, 361–363. [Google Scholar] [CrossRef]
- Stoneham, S.J.; Palmer, L.; Cash, R.; Rossdale, P.D. Measurement of serum amyloid A in the neonatal foal using a latex agglutination immunoturbidimetric assay: Determination of the normal range, variation with age and response to disease. Equine Vet. J. 2001, 33, 599–603. [Google Scholar] [CrossRef]
- Pirrone, A.; Mariella, J.; Gentilini, F.; Castagnetti, C. Amniotic fluid and blood lactate concentrations in mares and foals in the early postpartum period. Theriogenology 2012, 78, 1182–1189. [Google Scholar] [CrossRef]
- Malamitsi-Puchner, A.; Nikolaou, K.E.; Economou, E.; Boutsikou, M.; Boutsikou, T.; Kyriakakou, M.; Puchner, K.-P.; Hassiakos, D. Intrauterine growth restriction and circulating neurotrophin levels at term. Early Hum. Dev. 2007, 83, 465–469. [Google Scholar] [CrossRef]
- Hogan, K.A.; Ambler, C.A.; Chapman, D.; Bautch, V.L. The neural tube patterns vessels developmentally using the VEGF signaling pathway. Development 2004, 131, 1503–1513. [Google Scholar] [CrossRef] [Green Version]
- Schwarz, Q.; Gu, C.; Fujisawa, H.; Sabelko, K.; Gertsenstein, M.; Nagy, A.; Taniguchi, M.; Kolodkin, A.L.; Ginty, D.D.; Shima, D.T.; et al. Vascular endothelial growth factor controls neuronal migration and cooperates with Sema3A to pattern distinct compartments of the facial nerve. Genes Dev. 2004, 18, 2822–2834. [Google Scholar] [CrossRef] [Green Version]
- Aisa, M.C.; Barbati, A.; Cappuccini, B.; De Rosa, F.; Gerli, S.; Clerici, G.; Kaptilnyy, V.A.; Ishenko, A.I.; Di Renzo, G.C. Urinary Nerve Growth Factor in full-term, preterm and intra uterine growth restriction neonates: Association with brain growth at 30–40 days of postnatal period and with neuro-development outcome at two years. A pilot study. Neurosci. Lett. 2021, 741, 135459. [Google Scholar] [CrossRef]
- Williams, M.A.; Schmidt, A.R.; Carleton, C.L.; Darien, B.J.; Goyert, G.L.; Sokol, R.J.; Derksen, F.J. Amniotic fluid analysis for ante-partum foetal assessment in the horse. Equine Vet. J. 1992, 24, 236–238. [Google Scholar] [CrossRef]
- Kodomari, I.; Wada, E.; Nakamura, S.; Wada, K. Maternal supply of BDNF to mouse fetal brain through the placenta. Neurochem. Int. 2009, 54, 95–98. [Google Scholar] [CrossRef]
- Marx, C.E.; Vance, B.J.; Jarskog, L.; Chescheir, N.C.; Gilmore, J.H. Nerve growth factor, brain-derived neurotrophic factor, and neurotrophin-3 levels in human amniotic fluid. Am. J. Obstet. Gynecol. 1999, 181, 1225–1230. [Google Scholar] [CrossRef]
- Dhobale, M.; Mehendale, S.; Pisal, H.; Nimbargi, V.; Joshi, S. Reduced maternal and cord nerve growth factor levels in preterm deliveries. Int. J. Dev. Neurosci. 2012, 30, 99–103. [Google Scholar] [CrossRef]
- Sabour, S. Prediction of preterm delivery using levels of VEGF and leptin in amniotic fluid from the second trimester: Prediction rules. Arch. Gynecol. Obstet. 2015, 291, 719. [Google Scholar] [CrossRef] [Green Version]
- De Backer, D. Lactic acidosis. Intensive Care Med. 2003, 29, 699–702. [Google Scholar] [CrossRef]
- Chiba, A.; Aoki, T.; Itoh, M.; Yamagishi, N.; Shibano, K. Hematological and Blood Biochemical Characteristics of Newborn Heavy Draft Foals After Dystocia. J. Equine Vet. Sci. 2017, 50, 69–75. [Google Scholar] [CrossRef]
- Pirrone, A.; Panzani, S.; Govoni, N.; Castagnetti, C.; Veronesi, M.C. Thyroid hormone concentrations in foals affected by perinatal asphyxia syndrome. Theriogenology 2013, 80, 624–629. [Google Scholar] [CrossRef]
- Walker, P.; Weil, N.L.; Weichsel, M.E., Jr.; Fischer, D.A. Effect of thyroxine on nerve growth factor concentration in neonatal mouse brain. Life Sci. 1981, 28, 1777–1787. [Google Scholar] [CrossRef]
- Figueiredo, B.; Almazan, G.; Ma, Y.; Tetzlaff, W.; Miller, F.; Cuello, A. Gene expression in the developing cerebellum during perinatal hypo- and hyperthyroidism. Mol. Brain Res. 1993, 17, 258–268. [Google Scholar] [CrossRef]
- Lanci, A.; Ingrà, L.; Dondi, F.; Tomasello, F.; Teti, G.; Mariella, J.; Falconi, M.; Castagnetti, C. Morphological study of equine amniotic compartment. Theriogenology 2022, 177, 165–171. [Google Scholar] [CrossRef]
- Fujita, K.; Tatsumi, K.; Kondoh, E.; Chigusa, Y.; Mogami, H.; Fujii, T.; Yura, S.; Kakui, K.; Konishi, I. Differential expression and the anti-apoptotic effect of human placental neurotrophins and their receptors. Placenta 2011, 32, 737–744. [Google Scholar] [CrossRef] [Green Version]
- Toti, P.; Ciarmela, P.; Florio, P.; Volpi, N.; Occhini, R.; Petraglia, F. Human placenta and fetal membranes express nerve growth factor mRNA and protein. J. Endocrinol. Investig. 2006, 29, 337–341. [Google Scholar] [CrossRef]
- Frank, P.; Barrientos, G.; Tirado-González, I.; Cohen, M.; Moschansky, P.; Peters, E.M.; Klapp, B.F.; Rose, M.; Tometten, M.; Blois, S.M. Balanced levels of nerve growth factor are required for normal pregnancy progression. Reproduction 2014, 148, 179–189. [Google Scholar] [CrossRef]
- Zacchigna, S.; Lambrechts, D.; Carmeliet, P. Neurovascular signalling defects in neurodegeneration. Nat. Rev. Neurosci. 2008, 9, 169–181. [Google Scholar] [CrossRef]
- Wessels, J.M.; Wu, L.; Leyland, N.A.; Wang, H.; Foster, W.G. The Brain-Uterus Connection: Brain Derived Neurotrophic Factor (BDNF) and Its Receptor (Ntrk2) Are Conserved in the Mammalian Uterus. PLoS ONE 2014, 9, e94036. [Google Scholar]
- Cassens, C.; Kleene, R.; Xiao, M.-F.; Friedrich, C.; Dityateva, G.; Schafer-Nielsen, C.; Schachner, M. Binding of the Receptor Tyrosine Kinase TrkB to the Neural Cell Adhesion Molecule (NCAM) Regulates Phosphorylation of NCAM and NCAM-dependent Neurite Outgrowth. J. Biol. Chem. 2010, 285, 28959–28967. [Google Scholar] [CrossRef] [Green Version]
- Nakamura, K.; Martin, K.C.; Jackson, J.K.; Beppu, K.; Woo, C.-W.; Thiele, C.J. Brain-Derived Neurotrophic Factor Activation of TrkB Induces Vascular Endothelial Growth Factor Expression via Hypoxia-Inducible Factor-1α in Neuroblastoma Cells. Cancer Res. 2006, 66, 4249–4255. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.H.; Paden, A.J.; Johnson, E.M. Mixed-Lineage Kinase Inhibitors Require the Activation of Trk Receptors to Maintain Long-Term Neuronal Trophism and Survival. J. Pharmacol. Exp. Ther. 2005, 312, 1007–1019. [Google Scholar] [CrossRef] [Green Version]
- Tervonen, T.A.; Ajamian, F.; De Wit, J.; Verhaagen, J.; Castrén, E.; Castrén, M. Overexpression of a truncated TrkB isoform increases the proliferation of neural progenitors. Eur. J. Neurosci. 2006, 24, 1277–1285. [Google Scholar] [CrossRef] [PubMed]
- Garcés, M.F.; Sanchez, E.; Torres-Sierra, A.L.; Ruíz-Parra, A.I.; Angel-Müller, E.; Alzate, J.P.; Sánchez, A.Y.; Gomez, M.A.; Romero, X.C.; Castañeda, Z.E.; et al. Brain-derived neurotrophic factor is expressed in rat and human placenta and its serum levels are similarly regulated throughout pregnancy in both species. Clin. Endocrinol. 2014, 81, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Dini, P.; Carossino, M.; Loynachan, A.T.; Ali, H.E.-S.; Wolfsdorf, K.E.; Scoggin, K.E.; Daels, P.; Ball, B.A. Equine hydrallantois is associated with impaired angiogenesis in the placenta. Placenta 2020, 93, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Daneshmand, S.S.; Cheung, C.Y.; Brace, R.A. Regulation of amniotic fluid volume by intramembranous absorption in sheep: Role of passive permeability and vascular endothelial growth factor. Am. J. Obstet. Gynecol. 2003, 188, 786–793. [Google Scholar] [CrossRef]
| Genes | Primer Sequences (5′–3′) |
|---|---|
| NGF Nerve growth factor | Forward: GGGCCCATTAACGGCTTTTC Reverse: CATTGCTCTCTGTGTGGGGT |
| P75NTR p75 Neurotrophin receptor | Forward: GAGCCAACCAGACTGTGTGT Reverse: GGTAGTAGCCATAGGCGCAG |
| TRKA Tropomyosin receptor kinase A | Forward: GGAGCTGAGAAACCTCACCAT Reverse: GCACAGGAACAGTCCAGAGG |
| VEGF Vascular endothelial growth factor | Forward: AACGACGAGGGCCTAGAGT Reverse: CAAGGCCCACAGGGATTTTCT |
| KDR Kinase insert domain receptor | Forward: GATGACAACCAGACGGACAGT Reverse: TTTTGCTGGGCATCAGTCCA |
| FLT1 Fms-related receptor tyrosine kinase 1 | Forward: CTGGCATCCCTGTAACCACA Reverse: AGGGTGCTAGCCGTCTTATTC |
| BDNF Brain-derived neurotrophic factor | Forward: CATGTCTATGAGGGTCCGGC Reverse: CATGTCCACTGCCGTCTTCT |
| TRKB Tropomyosin receptor kinase B | Forward: CGGGAACACCTCTCGGTCTA Reverse: CTGGACCAACACCTTGTCTTGA |
| Mare | Foal | ||||||
|---|---|---|---|---|---|---|---|
| Age | Parity | Gestational Length | Stage II Labor Length | Sex | Weight | Apgar Score | Placenta/Foal Weight Ratio |
| (Years) | (Days) | (min) | (kg) | (0–10) | (%) | ||
| 10.4 ± 5.1 | 3.8 ± 3.4 | 339.6 ± 9.2 | 11.9 ± 5.4 | Males n = 5 | 48.2 ± 5.7 | 9.4 ± 0.6 | 10.5 ± 1.4 |
| (5–20) | (1–12) | (325–355) | (6–25) | Females n = 9 | (38–55) | (8–10) | (7.8–12.2) |
| Amniotic Fluid | Umbilical Cord Vein | Mare’s JV | Foal’s JV | |||
|---|---|---|---|---|---|---|
| TP | T0 | T24 | T72 | |||
| NGF (ng/mL) | 146.8 ± 43.6 | 134.2 ± 46.8 | 101.9 ± 67.8 | 112.7 ± 57.5 | 85.9 ± 49.6 | 91.9 ± 47.6 |
| (95.4–233.0) | (63.4–214.5) | (38.3–208.4) | (56.1–251.2) | (32.6–178.8) | (43.2–187.3) | |
| (n = 14) | (n = 13) | (n = 8) | (n = 14) | (n = 14) | (n = 14) | |
| VEGF (pg/mL) | 268.1 ± 17.63 | 192.6 ± 32.0 | 134.0 ± 26.3 | 196.3 ± 40.7 | 164.9 ± 36.9 | 163.9 ± 38.2 |
| (239.6–294.2) | (131.3–231.1) | (80.8–162.9) | (136.4–268.6) | (103.5–229.4) | (85.8–200.3) | |
| (n = 12) | (n = 13) | (n = 8) | (n = 14) | (n = 14) | (n = 14) | |
| TT3 (ng/dL) | NA | 409.2 ± 102.8 | 59.8 ± 15.5 | 936.2 ± 550.1 | 1888.1 ± 861.3 | 1858.6 ± 755.7 |
| (252.0–584.0) | (40.0–87.8) | (362.0–1828.0) | (552.0–3528.0) | (412.0–3128.0) | ||
| (n = 12) | (n = 8) | (n = 14) | (n = 14) | (n = 13) | ||
| TT4 (nmol/L) | NA | 585.9 ± 280.2 | 15.5 ± 4.4 | 563.6 ± 180.2 | 446.8 ± 230.8 | 223.1 ± 71.5 |
| (336.0–1180.0) | (12.9–20.6) | (292.0–848.0) | (198.0–951.0) | (97.7–363.0) | ||
| (n = 12) | (n = 8) | (n = 14) | (n = 14) | (n = 13) | ||
| Ligand | Receptors | Ligand | Receptors | Ligand | Receptor | |||
|---|---|---|---|---|---|---|---|---|
| NGF | p75NTR | TRKA | VEGF | KDR | FLT1 | BDNF | TRKB | |
| Chorion | exp | exp | exp | exp | exp | exp | exp | ne |
| Allantois | exp | exp | ne | exp | exp | exp | exp | ne |
| Amnion | ne | ne | ne | exp | exp | exp | exp | ne |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ellero, N.; Lanci, A.; Baldassarro, V.A.; Alastra, G.; Mariella, J.; Cescatti, M.; Giardino, L.; Castagnetti, C. Study on NGF and VEGF during the Equine Perinatal Period—Part 1: Healthy Foals Born from Normal Pregnancy and Parturition. Vet. Sci. 2022, 9, 451. https://doi.org/10.3390/vetsci9090451
Ellero N, Lanci A, Baldassarro VA, Alastra G, Mariella J, Cescatti M, Giardino L, Castagnetti C. Study on NGF and VEGF during the Equine Perinatal Period—Part 1: Healthy Foals Born from Normal Pregnancy and Parturition. Veterinary Sciences. 2022; 9(9):451. https://doi.org/10.3390/vetsci9090451
Chicago/Turabian StyleEllero, Nicola, Aliai Lanci, Vito Antonio Baldassarro, Giuseppe Alastra, Jole Mariella, Maura Cescatti, Luciana Giardino, and Carolina Castagnetti. 2022. "Study on NGF and VEGF during the Equine Perinatal Period—Part 1: Healthy Foals Born from Normal Pregnancy and Parturition" Veterinary Sciences 9, no. 9: 451. https://doi.org/10.3390/vetsci9090451
APA StyleEllero, N., Lanci, A., Baldassarro, V. A., Alastra, G., Mariella, J., Cescatti, M., Giardino, L., & Castagnetti, C. (2022). Study on NGF and VEGF during the Equine Perinatal Period—Part 1: Healthy Foals Born from Normal Pregnancy and Parturition. Veterinary Sciences, 9(9), 451. https://doi.org/10.3390/vetsci9090451







