4.1. Bacterial Community of the Rumen of Cows
Cellulolytic fermentation supplies the body of cows with substrates and ensures the digestibility of nutrients from indigestible to digested materials and transforms them into monomers with the help of microbial enzymes [
48]. In general, the experimental groups of animals showed no deviations from the norm for cellulolytic, amylolytic, transit and pathogenic bacteria, which indicated balanced feeding. The introduction of Agro-Matic protein concentrate into rations caused a decrease in the number of cellulolytic microorganisms fermenting the structural carbohydrates of the plant feed [
47,
48,
49,
50] as microbiota with antimicrobial activity, it was noted a higher content of such as
Bifidobacterium sp. and
Bacillus sp. The observed depression of fiber digestion when adding protein concentrate Agro-Matic to the ration was a consequence of the negative associative effect of feed concentrate on ruminant activity and digestion. The level of cellulolytic bacteria in the experimental groups was higher than normal; however, the total sum in the 2 AM group was lower than that in the 1 AM group of lactating cows and the control group.
The effectiveness of the use of non-protein nitrogen by the rumen microflora for their own needs and protein synthesis of the body depends primarily on the ratio of sugar and protein in the ration [
49]. It is known that if the dry matter of the animal diet contains 10–12% protein, then non-protein nitrogen is used well; if the protein is more than 14–16%, then the degree of assimilation of non-protein nitrogen decreases sharply. At the same time, for the development of microorganisms and the formation of protein, a sufficient number of easily digestible carbohydrates must be present in the feed [
50,
51].
At the beginning of the lactation period, calcium is poorly mobilized from the skeleton. As a result of intensive milk synthesis, the mobile supply of calcium is rapidly removed from the blood, disrupting regulatory functions. During this period, it is important that a sufficient level of magnesium is supplied to the ration. This element participates in the process of calcium mobilization from the cow’s backbone after calving. Magnesium deficiency makes it difficult to form a parathyroid hormone that mobilizes calcium. When analyzing the diet of cows by mineral nutrition, attention is paid to the ratio of calcium to phosphorus as part of the mineral metabolism of the body. To a greater extent, the ratio under consideration should be normalized in the diets of highly productive cows [
52].
It is known that calcium ions are necessary for three predominant types of rumen cellulolytic bacteria, such as
Fibrobacter succinogenes,
Ruminococcus flavefaciens and
Ruminococcus albus, which are present in the rumen fluid. Bacteria of the genus
Ruminococcus probably synthesize cellulosomes that require the presence of calcium ions in the structure. The function of calcium in the degradation of cellulose by
F. succinogenes is unknown, but it is also associated with the secretion or activation of cellulolytic enzymes [
53].
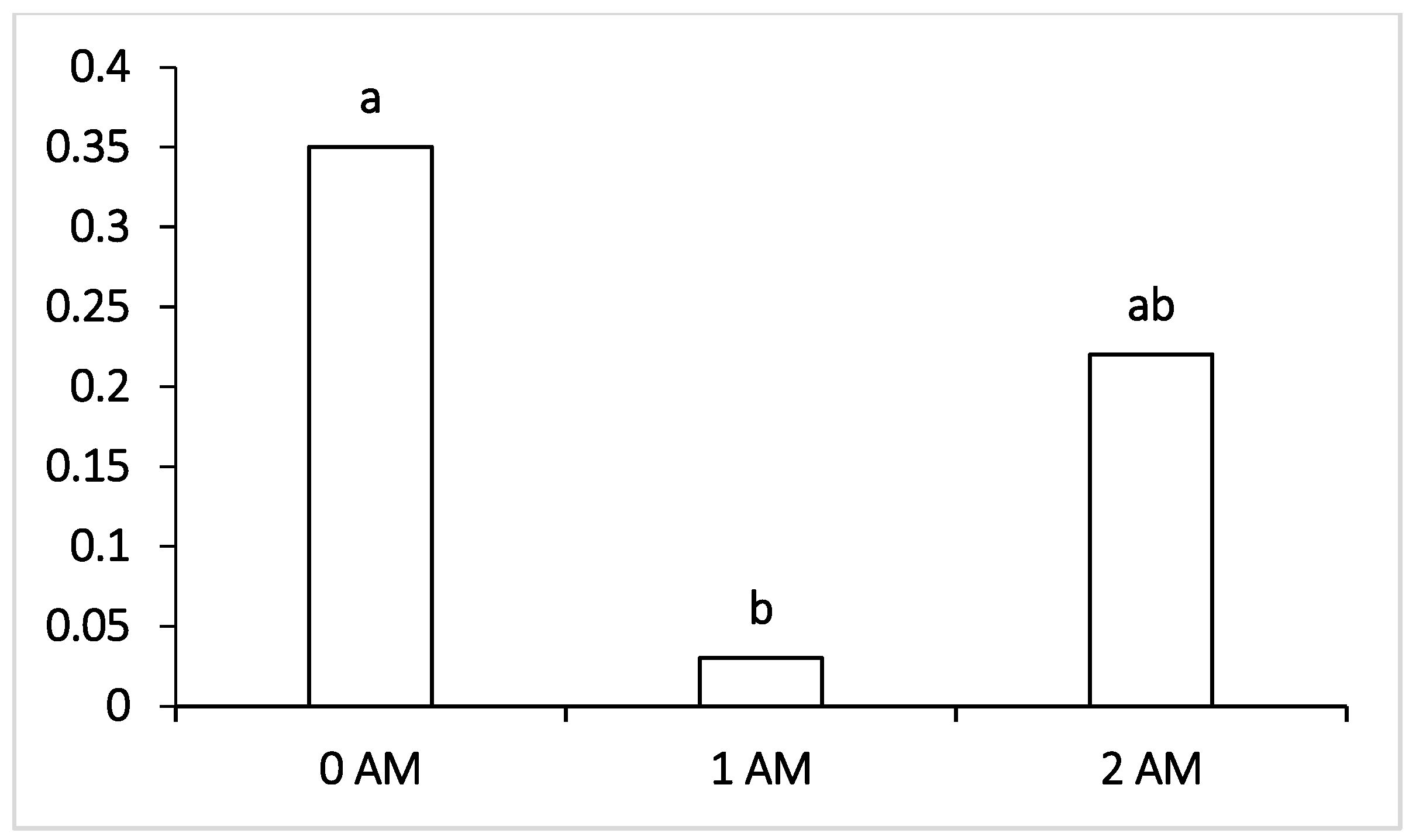
When studying the microbiota of the rumen of cows at the end of the lactation period, we found that the inclusion of Agro-Matic protein concentrate in the rations directly affected the number of cellulolytic microorganisms fermenting structural carbohydrates of plant feed. Concerning the rumen microflora at the end of lactation, the sum of cellulolytic bacteria increased in all experimental groups. The highest level of cellulolytic was observed in animals of the 1 AM group. On the other hand, the decrease in the level of cellulolytic bacteria Lachnospiraceae and Ruminococcaceae in cows of the 1 AM group was probably due to a drop in the pH of the rumen content, as the concentration of these microorganisms largely depends on the acidity of the rumen, which is highly correlated with amylolytic microorganisms, such as Bacteroides and Succinivibrio, which are formed during the fermentation of concentrated feeds with a high level of starch.
It is known that Bacteroidetes are necessary for the ecosystem of the rumen of cows since they use starch of concentrated feed as a substrate for their vital activity and the formation of byproducts: lactic, acetic, propionic, succinic and butyric acids [
52,
53,
54]. Additionally, in a study by Osborne and Dehority (1989) [
54], it was suggested that some representatives of the Bacteroidetes phylum, such as Prevotella ruminicola, may synergistically participate in the degradation of plant cell walls in order to fully use feed cellulose, hemicellulose and pectin for conversion into volatile fatty acids. On the other hand, Bacteroidetes are responsible for the breakdown of protein [
46] and starch in concentrated feed. In our study, in the experimental groups at the end of lactation period, a higher content of Bacteroidetes was observed. Therefore, the reduction in the number of this group in the control group was due to an increase in the level of available protein necessary for the growth of the number of bacteria. In this regard, it is possible to prove the increase in the intensity of protein metabolism in the rumen of the experimental lactating cows.
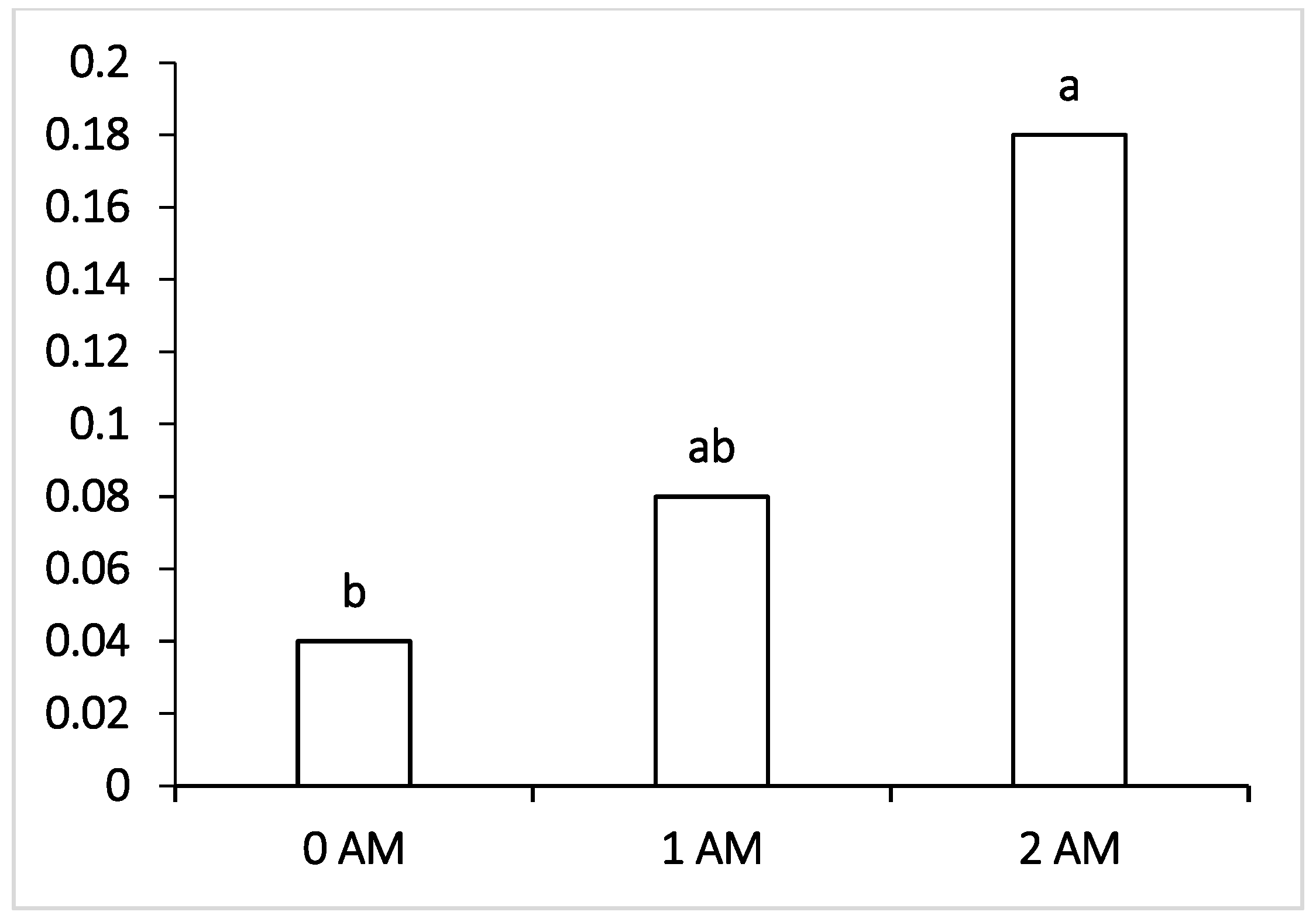
It was interesting to note that, at the beginning of lactation (120 DIM), both experimental groups (1 AM and 2 AM) revealed a lower content of Bacteroidetes compared to the control, amounting to 1.5% and 2.9%, respectively. The increase in the proportion of these bacteria noted at the end of lactation in animals of the experimental groups compared with the control group was probably due to the reorganization of the microbiome that occurred during lactation under the influence of the protein concentrate Agro-Matic. Interestingly, changes in the representation of bacteria of the order Selemonadales in the rumen of cows during the first period (120 DIM) and at the end of lactation had reverse trends of change compared with the content of Bacteroidetes. During the fermentation of starch and easily digestible carbohydrates, a high number of fermentation byproducts are formed—organic acids, among which is a high proportion of lactic acid, which is a substrate for bacteria of the Selenomonadales group. It has been reported that acid-utilizing bacteria of the genera Megasphaera, Selenomonas and Dialister are physiologically important groups for cattle, since they do not allow lactate to accumulate in the rumen, the increase in the proportion of which can lead to the decrease in pH and initiate the development of lactate acidosis [
47,
48]. At the beginning of the lactation period, the representation of these microorganisms in the rumen of cows of the experimental groups was higher compared to the control by 1.7% and 4.1%. At the end of lactation, the content of Selenomonadales was highest in cows of the control group, which is probably due to the presence of a large amount of substrate for their growth.
Obviously, the presence of lactate-producing Lactobacillus species was also associated with the number of Selenomonadales. It should be noted that Selenomonadales, like cellulolytic taxa, are sensitive to the decrease in pH, which may explain their slight decrease in the rumen with the increase in the proportion of Bacteroidetes at the end of the lactation period. There was a direct pattern of changes in the proportion of VFA-fermenting Selenomonadales compared with other microorganisms, particularly compared with the content of representatives of Clostridiaceae. It is known that many representatives of this family have proteolytic properties and participate in the fermentation of oligosaccharides, starch and sugar to form formate, acetate, lactate, propionate, butyrate and other volatile fatty acids, which provide most of the energy for the host [
49].
Generally, throughout the course of our research, there was a modulation of the microbial community associated with the increase in the representation of the proportion of bacteria with protein, cellulose and amylolytic properties in the rumen of cows by the end of lactation when feeding with a feed additive. This potentially indicates the increase in the activity of the microbiome in relation to protein, polysaccharides and easily digestible feed components, which influenced the level of dairy productivity of cows in the study which were supplemented with the protein concentrate Agro-Matic. The VFA formed during the fermentation of carbohydrates, in particular, propionic, are used in the body for milk synthesis. The increase in the content of propionate can also stimulate insulin secretion, blood flow to the udder and milk protein synthesis [
49,
50]. Acetate and butyrate in the rumen are converted by the liver into cholesterol in order to further participate in the synthesis of milk fat [
50,
51].
In addition, short-chain fatty acids produced by bacteria have a number of other important properties. For example, they are involved in the epigenomic regulation of interactions between the microbiota and the host macroorganism [
52,
53,
54,
55,
56,
57,
58]. It is also known that epigenetic modifications are able to regulate gene expression, affecting its intensity and duration, without changes in the main DNA sequence. Moreover, many VFA produced during the decomposition of feed carbohydrates have pronounced antimicrobial properties, contributing to a decrease in the representation of pathogenic microorganisms [
59,
60,
61].
The symbiotic microflora of the rumen perform a detoxification function, regulate the digestibility of nutrients, prevent the invasion of parasites and pathogens and stimulate the development of immunity. The proof of this is that, according to the latest research data by Holman and Gzyl (2019) [
62], the microflora and microfauna of the gastrointestinal tract of animals play an important role in the development and maturation of the immune system of cows [
63] and can also provide protection of the digestive system from colonization by pathogenic microorganisms. In addition, the slow formation of the microbial community of the rumen occurs due to ecological and biological factors created by the microorganism, for example, the influence of the immune system of cows, which synthesizes immune peptides, glycolyzed animal cells and nutrients supplied to bacteria from the body of the cows.
We noted that the proportion of bacteria with antagonistic properties against pathogens in the rumen of animals increased under the action of protein concentrate Agro-Matic. The patterns of increased concentrations of bacteria that stimulate the development of immunity revealed by us in the initial period of lactation indicate a higher content of
Bifidobacterium sp. and
Bacillus sp. compared to the group 1 AM. However, at the end of lactation, animals of both groups 1 AM and 2 AM showed the highest values for Bacillus and Bifidobacterium. The saccharolytic properties of Bifidobacterium and its ability to produce acetate and lactate are also known [
31,
56,
57,
58,
59].
When analyzing the representation of various opportunistic and transit microorganisms, it was found that most of them belonged to bacteria associated with the development of gastroenteritis—the families Enterobacteriaceae and Pseudomonadaceae. Interestingly, in the early period of lactation in the cows, the proportion of these microorganisms significantly decreased compared to the control cows that did not receive the protein concentrate Agro-Matic. In the late period, the proportion of these bacteria, as well as actinomycetes, increased, which may be due to their proteolytic activity [
3]. It is known that the increase in opportunistic and pathogenic microflora in the rumen community is directly related to the decrease in the level of productivity and the state of animal health. The state of the rumen microflora directly affects the development of diseases of the liver, internal organs and limbs and mastitis and causes a decrease in reproduction rates, which was confirmed by some studies [
64].
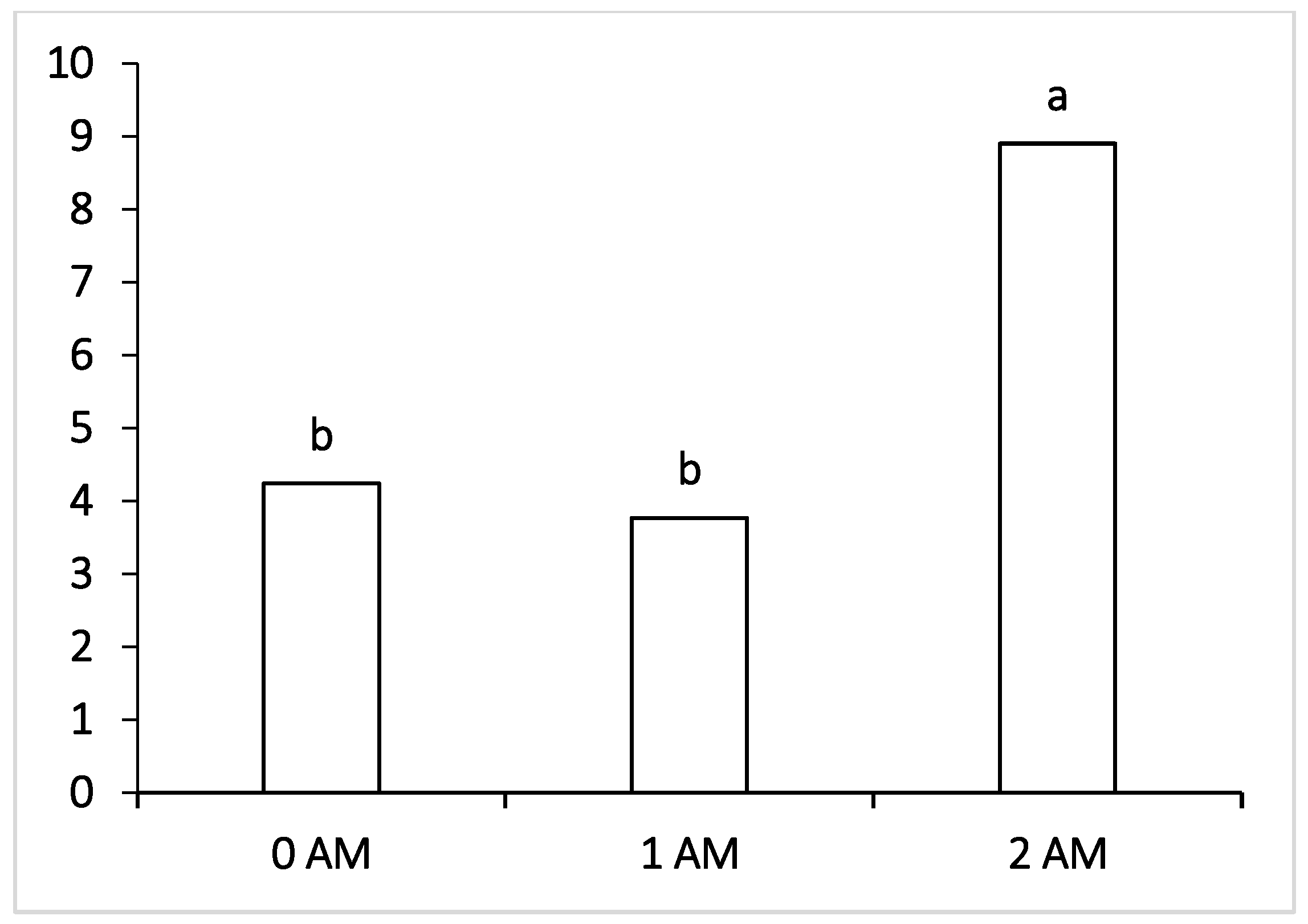
Regarding pathogenic microorganisms such as Fusobacteria, which can penetrate the blood and infect the body of cows with the development of liver abscesses and hoof lesions, their level was within the normal values in all the experimental groups of lactating cows receiving the ration with protein concentrate. The lowest content of pathogenic microorganisms, pathogens of mastitis and purulent-necrotic processes (
Staphylococcus, Peptococcus and
Fusobacterium) during the early lactation period (DIM = 120) was found in the ruminal fluid of animals of both the 1 AM and 2 AM groups that were supplemented with Agro-Matic protein concentrate as part of their diet. The number of pathogenic microorganisms—such as
Peptococcus—which cause purulent-necrotic diseases was significantly lower in animals of the 1 AM group, and Fusobacteria causing necrobacteriosis was also significantly lower in the ruminal fluid of the 2 AM group of cows. It may be interpreted that fusobacteria are capable of lactate consumption, and the decrease in their number may be correlated with the decrease in the proportion of lactate-producing Bacteroidetes, Lactobacillus and the increase in the content of other lactate-utilizing bacteria—Selenomonadales. However, at the end of lactation period, the level of Fusobacteria increased and was the highest among the compared groups in the 2 AM group versus the 0 AM group; moreover, there was an increase in pathogenic
Clostridium, which may be attributed to the fact that the advantage of protein feeds of animal origin is more associated with the participation of these groups of microorganisms. It is known that fusobacteria are characterized by high proteolytic activity [
58], which could be the reason for their increase when the protein supplement was added to the ration.
The representation of pathogenic bacteria such as Staphylococcus, Peptococcus and Campylobacteriaceae in the rumen of cows of both experimental groups at the end of the lactation period was lower compared to the control group. This indicates that the introduction of a protein feed additive had a positive effect on the composition of the rumen microbiota, contributing to a decrease in the representation of pathogenic bacteria. Similar results were obtained in the studies of Holman and Gzyl (2019) [
53], which reflects the influence of the microflora of the ruminant rumen on the development and maturation of the immune system of cows, including the protection of the digestive tract from colonization by pathogenic microorganisms.
Thus, the decrease in the abundance of representatives of the genera Campylobacter and Fusobacterium in animals in the experimental group was consistent with a decrease in the number of somatic cells in milk previously reported, since it has been proven [
65,
66,
67,
68,
69] that these microorganisms are associated with the occurrence of mastitis in cattle. The observed decrease in the number of pathogens such as Campylobacteriaceae, Staphylococcus and Peptococcus indicates the potential impact of Agro-Matic on reducing the presence of these genera, which can reduce the incidence and their spread into the environment.
Generally, molecular genetic studies conducted on lactating cows at the early lactation stage (120 DIM) and at the end of the lactation period show that the state of the rumen microflora affects not only the efficiency of feed digestion but also the immunity, health status, productivity level and the period of productive longevity of animals. In addition, the results obtained were consistent with the studies and works of Jami et al. (2014) [
3], who noted that the species composition of the rumen depends on the habitat of cows, the season of the year, the quality of feed, breed and individual characteristics of the development of the animal organism. Thus, our study showed that the composition of the bacterial community of the rumen of the studied cows was represented by a rich variety of microorganisms, the content of which was within normal values. At the same time, there were obvious shifts in the microbiome of the rumen of the cows under the influence of the protein concentrate Agro-Matic associated mainly with energy and protein metabolism.