Bee Health and Productivity in Apis mellifera, a Consequence of Multiple Factors
Abstract
1. Introduction
2. Materials and Methods
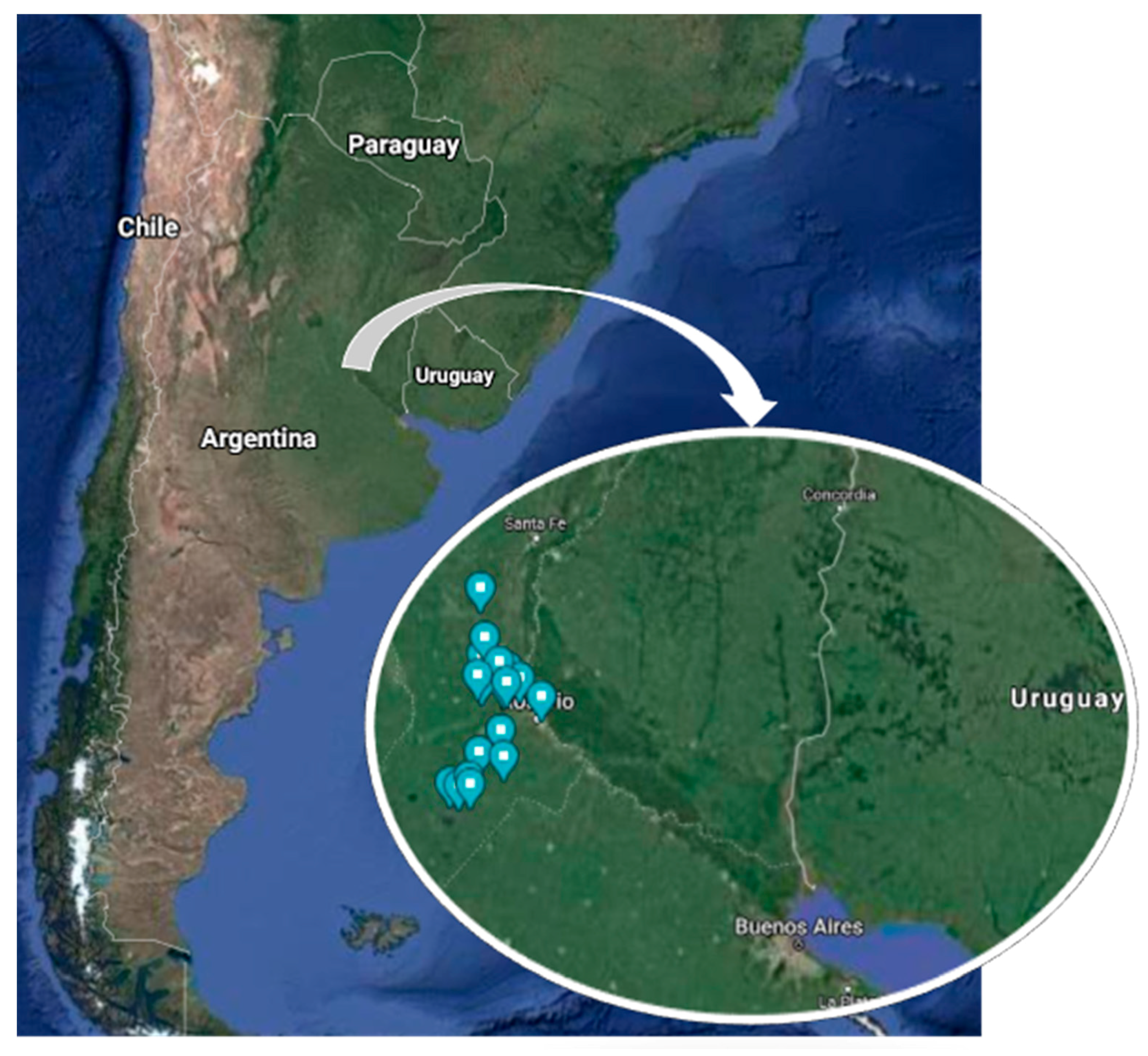
2.1. Experimental Design and Bee Colonies Selection
2.2. Methodology for Collecting Field Data
2.3. Infestation Rate (IR%) by Varroa sp. Mite
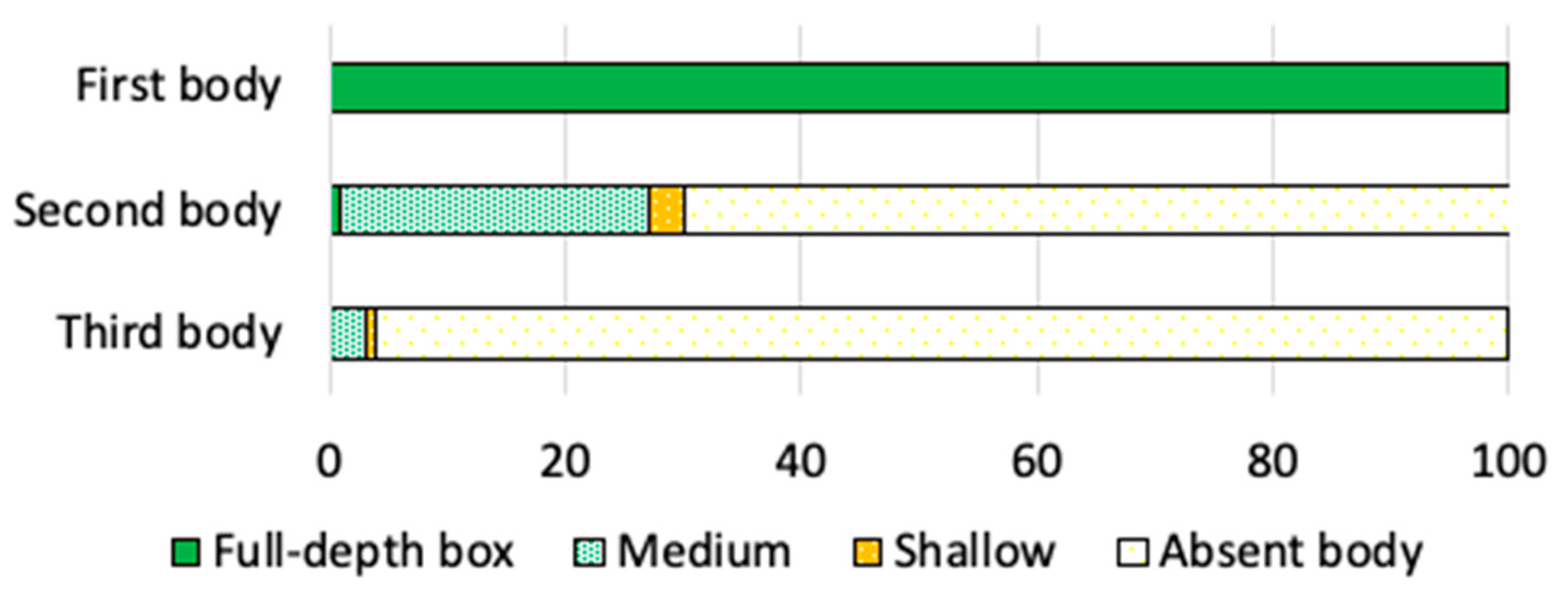
2.4. Honeybee Colony Strength
2.5. Statistical Analysis
3. Results
3.1. Field Information
3.1.1. General Characterisation of the Participants
3.1.2. Characterization of Zootechnical and Animal Health Management
3.1.3. Perception of Sanitary Gaps by Beekeepers
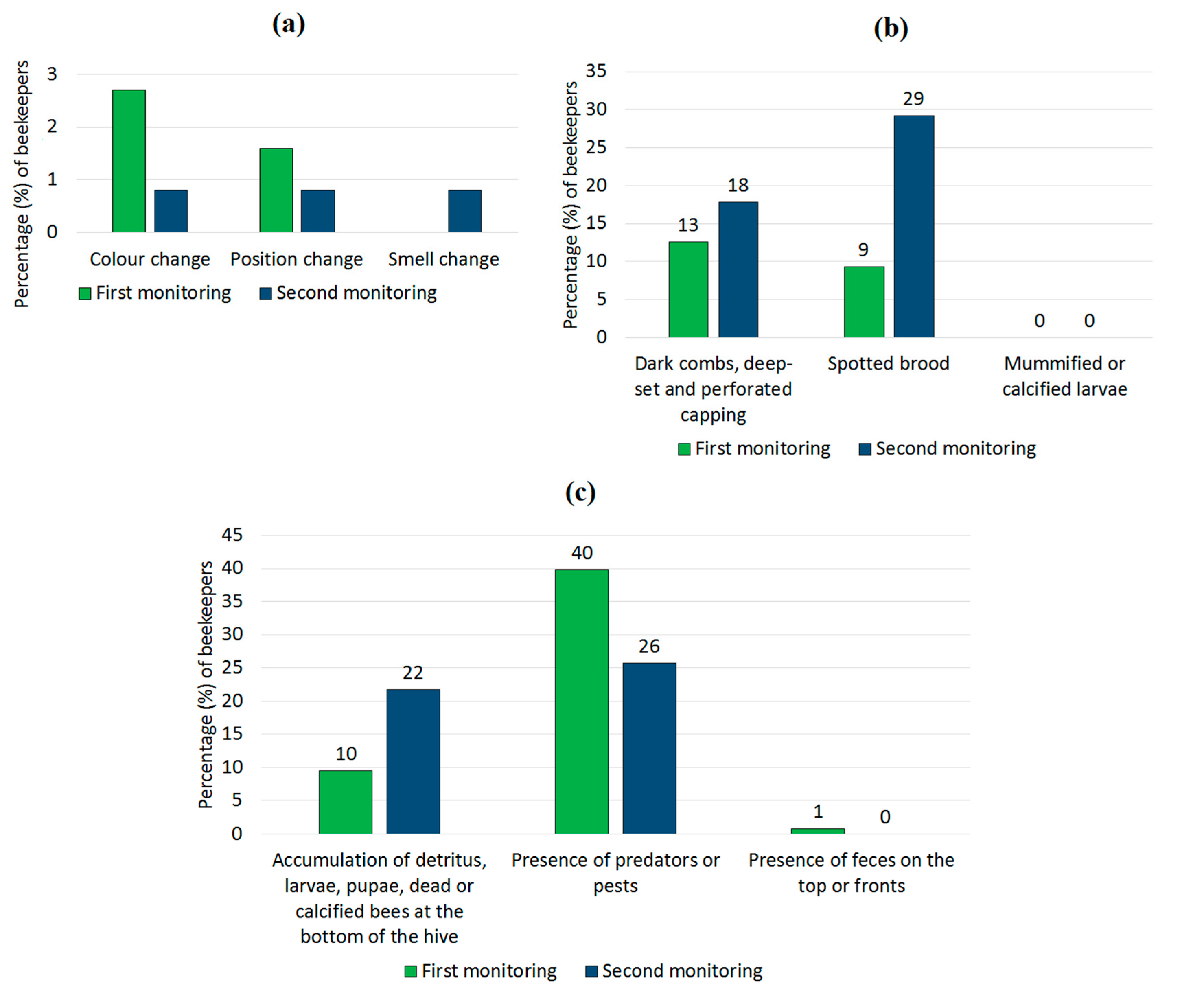
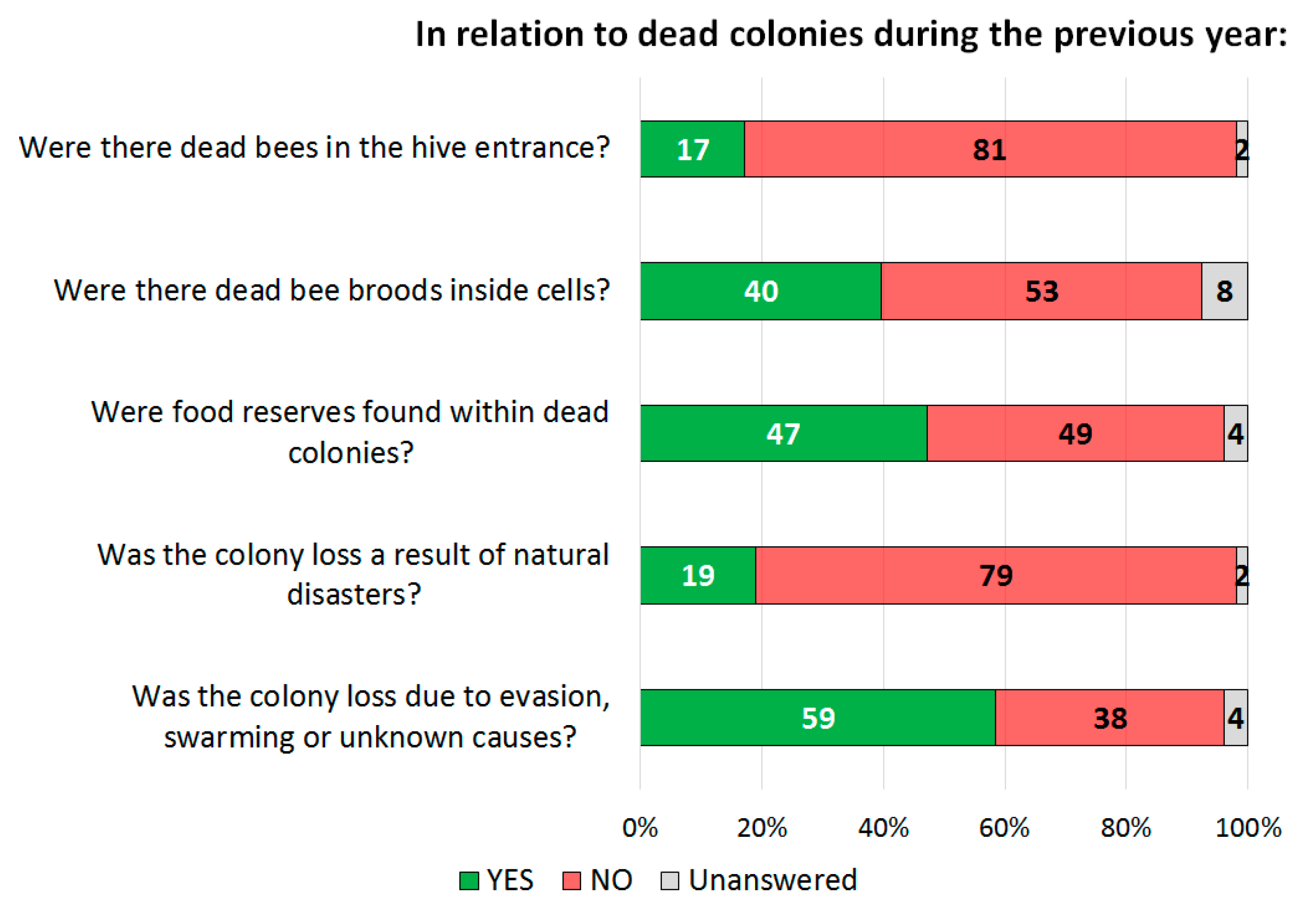
3.1.4. Clinical Observations Related to Diseases
3.2. Infestation Rate (IR%) by Varroa sp. Mite
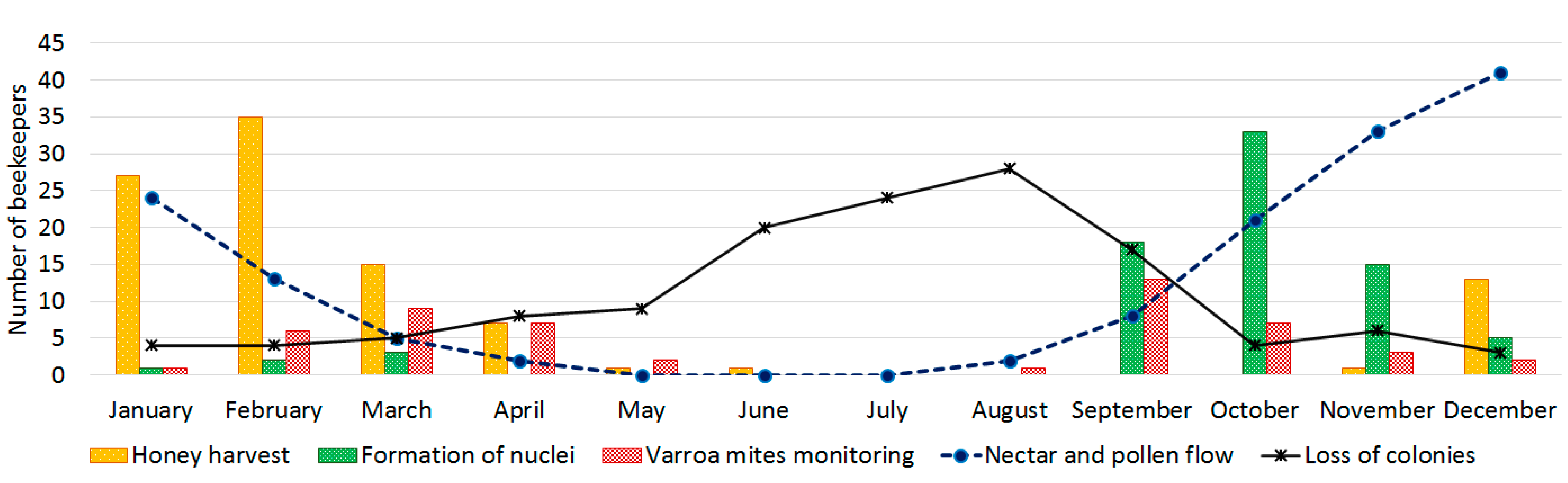
3.3. Honeybee Colony Strength and Weather Conditions
3.4. Productivity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ollerton, J. Pollinator diversity: Distribution, ecological function, and conservation. Annu. Rev. Ecol. Evol. Syst. 2017, 48, 353–376. [Google Scholar] [CrossRef]
- Bayer Bee Care Center. The Importance of Insect Pollinators for Agriculture. 2018. Available online: https://www.cropscience.bayer.com/sites/cropscience/files/inline-files/BEEINFOrmed_7_The-Importance-of-Insect-Pollinatorsjlouz8q1.pdf (accessed on 12 April 2021).
- Isaacs, R.; Williams, N.; Ellis, J.; Pitts-Singer, T.L.; Bommarco, R.; Vaughan, M. Integrated Crop Pollination: Combining strategies to ensure stable and sustainable yields of pollination-dependent crops. Basic Appl. Ecol. 2017, 22, 44–60. [Google Scholar] [CrossRef]
- Klatt, B.K.; Holzschuh, A.; Westphal, C.; Clough, Y.; Smit, I.; Pawelzik, E.; Tscharntke, T. Bee pollination improves crop quality, shelf life and commercial value. P. R. Soc. B 2014, 281, 20132440. [Google Scholar] [CrossRef]
- Pirk, C.W.W.; Crewe, R.M.; Moritz, R.F.A. Risks and benefits of the biological interface between managed and wild bee pollinators. Funct. Ecol. 2017, 31, 47–55. [Google Scholar] [CrossRef]
- Steinhauer, N.; vanEngelsdorp, D.; Saegerman, C. Priorizing changes in management practices associated with reduced winter honey bee colony losses for US beekeepers. Sci. Total Environ. 2021, 753, 141629. [Google Scholar] [CrossRef]
- Kulhanek, K.; Steinhauer, N.; Wilkes, J.; Wilson, M.; Spivak, M.; Sagili, R.R.; Tarpy, D.R.; McDermott, E.; Garavito, A.; Rennich, K.; et al. Survey-derived best management practices for backyard beekeepers improve colony health and reduce mortality. PLoS ONE 2021, 16, e0245490. [Google Scholar] [CrossRef] [PubMed]
- Wakgari, M.; Yigezu, G. Honeybee keeping constrains and future prospects. Cogent Food Agric. 2021, 7, 1872192. [Google Scholar] [CrossRef]
- OIE. Código Sanitario para los Animales Terrestres. Título 7. Capítulo 7.1. Introducción a Las Recomendaciones Para el Bienestar de Los Animales. 2019. Available online: https://www.oie.int/fileadmin/Home/esp/Health_standards/tahc/current/chapitre_aw_introduction.pdf (accessed on 15 February 2021).
- Verde, M.; Demedio, J.; Gómez, T. Apicultura, salud y Producción. Guía técnica para el Apicultor. 1a Ed. Instituto de Medicina Veterinaria, Ministerio de Agricultura: Cuba. 2013. Available online: https://eac.unr.edu.ar/wp-content/uploads/archivos/Apicultores%20y%20salud%2013-11-2013%20digital_compressed.pdf (accessed on 15 February 2021).
- Dolezal, A.G.; St Clair, A.L.; Zhang, G.; Toth, A.L.; O’Neal, M.E. Native habitat mitigates feast–famine conditions faced by honey bees in an agricultural landscape. Proc. Natl. Acad. Sci. USA 2019, 116, 25147–25155. [Google Scholar] [CrossRef] [PubMed]
- St. Clair, A.L.; Zhang, G.; Dolezal, A.G.; O’Neal, M.E.; Toth, A.L. Diversified farming in a monoculture landscape: Effects on honey bee health and wild bee communities. Environ. Entom. 2020, 49, 753–764. [Google Scholar] [CrossRef] [PubMed]
- Neov, B.; Georgieva, A.; Radoslavov, G.; Hristov, P. Biotic and abiotic stressors associated with colonies mortalities of managed honey bee (Apis mellifera). Diversity 2019, 11, 237. [Google Scholar] [CrossRef]
- Cantalapiedra, J.J.; Puerta, J.L.; Yllera, M.d.M.; Blanco, I.; Fernández, M.E. Bienestar Animal. Salud y Enfermedad en Relación con el Comportamiento. Xunta de Galicia: España. 2017. Available online: https://www.researchgate.net/publication/334612499 (accessed on 25 February 2021).
- Braga, A.R.; Gomes, D.G.; Rogers, R.; Hassler, E.E.; Freitas, B.M.; Cazier, J.A. A method for mining combined data from in-hive sensors, weather and apiary inspections to forecast the health status of honey bee colonies. Comput. Electron. Agric. 2020, 169, 105161. [Google Scholar] [CrossRef]
- Maggi, M.; Antúnez, K.; Invernizzi, C.; Aldea, P.; Vargas, M.; Negri, P.; Brasesco, C.; De Jong, D.; Message, D.; Teixeira, E.W.; et al. Honey bee health in South America. Apidologie 2016, 47, 835–854. [Google Scholar] [CrossRef]
- Medina-Flores, C.A.; Guzmán-Novoa, E.; Aréchiga-Flores, C.; Aguilera-Soto, J.; Gutiérrez-Piña, F. Efecto del nivel de infestación de Varroa destructor sobre la producción de miel de colonias de Apis mellifera en el altiplano semiárido de México. Rev. Mex. Cienc. Pecu. 2011, 2, 313–317. [Google Scholar]
- Nisbet, C.; Güler, A.; Biyik, S. Effects of different environmental conditions on the cognitive function of honeybee (Apis mellifera L.) and mineral content of honey. Ankara Üniv. Vet. Fak. Derg. 2019, 66, 95–101. [Google Scholar]
- Trade Map. Available online: https://www.trademap.org/Country_SelProductCountry.aspx?nvpm=1%7c032%7c%7c%7c%7c040900%7c%7c%7c6%7c1%7c1%7c2%7c1%7c1%7c2%7c1%7c1%7c1 (accessed on 15 February 2021).
- Carrasco, A.E.; Sánchez, N.E.; Tamagno, L.E. Modelo Agrícola E Impacto Socio-Ambiental en la Argentina: Monocultivo y Agronegocios. AUGM-Comité de Medio Ambiente. Serie Monográfica Sociedad y Ambiente: Reflexiones para una nueva América Latina (1st ed.) AUGM-Comité de Medio Ambiente: Argentina. 2012. Available online: http://sedici.unlp.edu.ar/bitstream/handle/10915/24722/Documento_completo__.pdf?sequence=3&isAllowed=y (accessed on 25 February 2021).
- Aizen, M.A.; Garibaldi, L.A.; Dondo, M. Soybean Expansion and Agriculture Diversity in Argentina. Ecol. Austral. 2009, 19, 45–54. Available online: https://bibliotecadigital.exactas.uba.ar/download/ecologiaaustral/ecologiaaustral_v019_n01_p045.pdf (accessed on 25 February 2021).
- Manuel-Navarrete, D.; Gallopín, G.; Blanco, M.; Díaz-Zorita, M.; Ferraro, D.; Herzer, H.; Laterra, P.; Morello, J.; Murmis, M.R.; Pengue, W.; et al. Análisis sistémico de la Agriculturización en la Pampa Húmeda Argentina y Sus Consecuencias en Regiones Extrapampeanas: Sostenibilidad, Brechas de Conocimiento e Integración de Políticas. División de Desarrollo Sostenible y Asentamientos Humanos. CEPAL. ONU: Publicación de las Naciones Unidas. 2005. Available online: https://repositorio.cepal.org/bitstream/handle/11362/5656/1/S051013_es.pdf (accessed on 25 February 2021).
- Requier, F.; Antúnez, K.; Morales, C.L.; Aldea Sánches, P.; Castilhos, D.; Garrido, P.M.; Giacobino, A.; Reynaldi, F.J.; Rosso Londoño, J.M.; Santos, E.; et al. Trends in beekeeping and honey bee colony losses in Latin America. J. Apicult. Res. 2018, 57, 657–662. [Google Scholar] [CrossRef]
- Lende, S.G. Soybean model in Argentina (1996-2014), an example of accumulation by dispossession. Mercator 2015, 14, 7–25. [Google Scholar] [CrossRef][Green Version]
- IPEC. Comercio Exterior—Exportaciones con Origen en la Provincia de Santa Fe—2018 al 2020. Available online: http://www.estadisticasantafe.gob.ar/contenido/exportaciones-con-origen-en-la-provincia-de-santa-fe/ (accessed on 25 February 2021).
- Giacobino, A.; Bulacio Cagnolo, N.; Merke, J.; Orellano, E.; Bertozzi, E.; Masciangelo, G.; Pietronave, H.; Salto, C.; Signorini, M. Risk factors associated with the presence of Varroa destructor in honey bee colonies from east-central Argentina. Prev. Vet. Med. 2014, 115, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Giacobino, A.; Pacini, A.; Molineri, A.; Rodríguez, G.; Crisanti, P.; Bulacio Cagnolo, N.; Merke, J.; Orellano, E.; Bertozzi, E.; Pietronave, H.; et al. Potential associations between the mite Varroa destructor and other stressors in honeybee colonies (Apis mellifera L.) in temperate and subtropical climate from Argentina. Prev. Vet. Med. 2018, 159, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Donkersley, P.; Elsner-Adams, E.; Maderson, S. A One-Health model for reversing honeybee (Apis mellifera L.) decline. Vet. Sci. 2020, 7, 119. [Google Scholar] [CrossRef]
- Castignani, H. Zonas Agroeconómicas Homogéneas Santa Fe. Estudios Socioeconómicos de la Sustentabilidad de los Sistemas de Producción y Recursos Naturales. INTA, EEA: Rafaela, Santa Fe. 2011. Available online: https://inta.gob.ar/sites/default/files/script-tmp-inta-_zah_santa_fe.pdf (accessed on 19 February 2021).
- Dietemann, V.; Ellis, J.D.; Neumann, P. The COLOSS BEEBOOK Volume II, Standard methods for Apis mellifera pest and pathogen research: Introduction. J. Apicult. Res. 2013, 52, 1–4. [Google Scholar] [CrossRef]
- Delaplane, K.S.; van der Steen, J.; Guzman-Novoa, E. Standard methods for estimating strength parameters of Apis mellifera colonies. J. Apicult. Res. 2013, 52, 1–12. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. B Met. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Pacheco, P. Soybean and Oil Palm Expansion in South America: A Review of Main Trends and Implications. Working Paper 90. CIFOR: Bogor. Indonesia. 2012. Available online: https://www.cifor.org/publications/pdf_files/WPapers/WP90Pacheco.pdf (accessed on 25 February 2021).
- De Groot, G.S.; Aizen, M.A.; Sáez, A.; Morales, C.L. Large-scale monoculture reduces honey yield: The case of soybean expansion in Argentina. Agric. Ecosyst. Environ. 2021, 306, 107203. [Google Scholar] [CrossRef]
- Winfree, R.; Aguilar, R.; Vasquez, D.P.; LeBunn, G.; Aizen, M.A. A meta-analysis of bees’ responses to anthropogenic disturbance. Ecology 2009, 90, 2068–2076. [Google Scholar] [CrossRef]
- Pérez Casar, L. Conservar la Biodiversidad, Clave Para Garantizar la Seguridad Alimentaria. RIA 2020, 45, 328–331. Available online: http://ria.inta.gob.ar/sites/default/files/actualidadimasd/conservar_la_biodiversidad_clave_para_garantizar_la_seguridad_alimentaria_ria3_dic_2019.pdf (accessed on 15 April 2021).
- Salizzi, E. Reestructuración Económica y Transformaciones Económicas en el Agro Pampeano: La Expansión del Cultivo de la Soja y Sus Efectos Sobre la Apicultura Bonaerense en los Inicios del Siglo XXI. Estud. Soc. 2014, 16. Available online: http://www.scielo.org.ar/scielo.php?script=sci_arttext&pid=S1853-43922014000200002&lng=es&nrm=iso&tlng=es (accessed on 15 April 2021).
- Spivak, M.; Browning, Z.; Goblirsch, M.; Lee, K.; Otto, C.; Smart, M.; Wu-Smart, J. Why does Bee Health Matter? The Science Surrounding Honeybee Health Concerns and What We Can Do about It. CAST Commentary. 2017. Available online: https://www.cast-science.org/wp-content/uploads/2018/12/QTA20171_Bee_Health_565CB839D149E.pdf (accessed on 25 February 2021).
- Potts, S.G.; Imperatriz-Fonseca, V.; Ngo, H.T.; Aizen, M.A.; Biesmeijer, J.C.; Breeze, T.D.; Dicks, L.V.; Garibaldi, L.A.; Hill, R.; Sttele, J.; et al. Safeguarding pollinators and their values to human well-being. Nature 2016, 540, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Ricigliano, V.A.; Mott, B.M.; Floyd, A.S.; Copeland, D.C.; Carroll, M.J.; Anderson, K.E. Honey bees overwintering in a southern climate: Longitudinal effects of nutrition and queen age on colony-level molecular physiology and performance. Sci. Rep. 2018, 8, 10475. [Google Scholar] [CrossRef] [PubMed]
- Naug, D. Nutritional stress due to habitat loss may explain recent honeybee colony collapses. Biol. Conserv. 2009, 142, 2369–2372. [Google Scholar] [CrossRef]
- Branchiccela, B.; Castelli, L.; Corona, M.; Díaz-Cetti, S.; Invernizzi, C.; Martínez de la Escalera, G.; Mendoza, Y.; Santos, E.; Silva, C.; Zunino, P.; et al. Impact of nutritional stress on the honeybee colony health. Sci. Rep. 2019, 9, 10156. [Google Scholar] [CrossRef]
- Corby-Harris, V.; Bowsher, J.H.; Carr-Markell, M.; Carroll, M.J.; Centrella, M.; Cook, S.C.; Couvillon, M.; DeGrandi-Hoffman, G.; Dolezal, A.; Jones, J.C.; et al. Emerging themes from the ESA symposium entitled “Pollinator nutrition: Lessons from bees at individual to landscape levels”. BEE World 2019, 96, 3–9. [Google Scholar] [CrossRef]
- Dolezal, A.G.; Toth, A.L. Feedbacks between nutrition and disease in honey bee health. Curr. Opin. Insect Sci. 2018, 26, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Farrar, C.L. The Influence of Colony Populations on Honey Production. J. Agric. Res. 1937, 54, 945–954. Available online: https://naldc.nal.usda.gov/download/IND43969007/PDF (accessed on 25 February 2021).
- Rosenkranz, P.; Aumeier, P.; Ziegelmann, B. Biology and control of Varroa destructor. J. Invertebr. Pathol. 2010, 103, S96–S119. [Google Scholar] [CrossRef] [PubMed]
- Dynes, T.L.; Berry, J.A.; Delaplane, K.S.; de Roode, J.C.; Brosi, B.J. Assessing virulence of Varroa destructor mites from different honey bee management regimes. Apidologie 2020, 51, 276–289. [Google Scholar] [CrossRef] [PubMed]
- Gregorc, A.; Alburaki, M.; Sampson, B.; Knight, P.R.; Adamczyk, J. Toxicity of selected acaricides to honey bees (Apis mellifera) and varroa (Varroa destructor Anderson and Trueman) and their use in controlling varroa within honey bee colonies. Insects 2018, 9, 55. [Google Scholar] [CrossRef]
- Higes, M.; Martín-Hernández, R.; Hernández-Rodríguez, C.S.; González-Cabrera, J. Assessing the resistance to acaricides in Varroa destructor from several Spanish locations. Parasitol. Res. 2020, 119, 3595–3601. [Google Scholar] [CrossRef]
- DeGrandi-Hoffman, G.; Ahumada, F.; Zazueta, V.; Chambers, M.; Hidalgo, G.; DeJong, E. Population growth of Varroa destructor (Acari: Varroidae) in honey bee colonies is affected by the number of foragers with mites. Exp. Appl. Acarol. 2016, 69, 21–34. [Google Scholar] [CrossRef]
- Harun, A.; Zaaba, S.K.; Kamarudin, L.M.; Zakaria, A.; Mohamed Farook, R.S.; Ndzi, D.L.; Shakaff, A.Y.M. Stingless bee colony health sensing through integrated wireless system. J. Teknol. 2015, 77, 85–90. [Google Scholar]
- Barrucand, M.G.; Rusticucci, M.M. Climatología de Temperaturas Extremas en la Argentina. Variabilidad Temporal y Regional. 2001. Available online: https://www.researchgate.net/publication/228916672_Climatologia_de_temperaturas_extremas_en_la_Argentina_Variabilidad_temporal_y_regional (accessed on 15 April 2021).
- Gregorc, A. Monitoring of Honey Bee Colony Losses: A Special Issue. Diversity 2020, 12, 403. [Google Scholar] [CrossRef]
| Variable | Percentage of Beekeepers (%) | Observations | |
|---|---|---|---|
| Yes | No | ||
| Does the beekeeper produce honey? | 100.0 | 0.0 | |
| Pollination | 9.4 | 90.6 | |
| Colony migration (transhumance) | 5.7 | 94.3 | |
| Is beekeeping the main source of income? | 17.0 | 83.0 | |
| Near crops | 62.3 | 37.7 | Distance: 0.1–100 m: 60.3% 100.1–500 m: 24.6% >1000 m: 5.7% |
| Training activities | 77.4 | 22.6 | Annually: 39.6% Biannual: 18.9% More than two years: 18.9% |
| Records of their productive activities | 37.7 | 62.3 | |
| Change of bee queens | 45.3 | 54.7 | Annually: 13.2% Biannual: 9.4% More than two years: 22.7% |
| Creation of nuclei | 77.0 | 23.0 | |
| Food supplements | 94.4 | 5.6 | Energy food: 77.4% Protein food: 0.0% Both: 17.0% |
| Does the beekeeper suspect a pest or disease in his apiary? | 78.4 | 21.6 | Which one? Varroa sp. 77.4% Foulbrood 13.2% Nosema spp. 3.8% Ants: 1.9% |
| Does the beekeeper monitor Varroa sp.? | 52.8 | 47.2 | |
| Who monitors or diagnoses diseases or pests? | - | - | Beekeeper: 92.5% A specialist: 3.8% Both: 1.9% None: 1.9% |
| Does the beekeeper confirm suspicions of other diseases by sending a sample to a laboratory? | 11.3 | 88.7 | |
| Beekeeping material disinfection | 54.7 | 45.3 | Flaming: 32.1% Boiling water: 20.8% Caustic soda: 5.7% Others: 22.6% |
| Are beekeeping materials stored in a dedicated storage space? | 60.4 | 39.6 | |
| Does the beekeeper have a plant for honey extraction? | 54.7 | 45.3 | Single plant: 26.4% Shared plant: 71.7% |
| Presence of other apiaries in the vicinity | 79.2 | 20.8 | Distance: <1 km: 26.5% 1.0–2.0 km: 39.6% >2.0 km: 13.1% None: 20.8% |
| Monitoring | Location (Department) | Mean | SD | Maximum |
|---|---|---|---|---|
| 1st | Casilda | 1.38 | 3.08 | 15.22 |
| Constitucion | 3.36 | 5.54 | 29.64 | |
| Iriondo | 4.67 | 7.30 | 32.29 | |
| Rosario | 5.20 | 9.00 | 41.62 | |
| San Jeronimo | 3.77 | 7.91 | 34.40 | |
| San Lorenzo | 2.39 | 4.99 | 22.26 | |
| Total 1st monitoring | 3.38 | 6.40 | 41.62 | |
| 2nd | Casilda | 0.09 | 0.24 | 0.97 |
| Constitucion | 0.80 | 2.21 | 10.86 | |
| Iriondo | 2.58 | 3.16 | 12.18 | |
| Rosario | 1.65 | 2.78 | 11.36 | |
| San Jeronimo | 0.56 | 1.85 | 9.14 | |
| San Lorenzo | 0.68 | 1.53 | 7.25 | |
| Total 2nd monitoring | 0.97 | 2.18 | 12.18 | |
| Total | Total | 2.24 | 5.03 | 41.62 |
| Monitoring | Found Frames | Comb Sides with: | Frame Heads with Bees (*) | Bees Entering the hive/min (*) | Temperature (°C) (*) | %RH (%) (*) | Wind Speed (km/h) (*) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adult Bees | Capped Brood (*) | Open Brood (*) | Honey (*) | Pollen (*) | ||||||||
| 1st | Mean | 9 | 5.92 | 0.47 | 0.19 | 6.38 | 0.76 | 3.07 | 9.85 | 22.35 | 62.10 | 2.57 |
| SD | 1 | 2.80 | 0.80 | 0.30 | 3.70 | 0.94 | 2.39 | 11.97 | 3.52 | 18.19 | 4.14 | |
| Min. | 8 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 13.00 | 25.00 | 0.00 | |
| Max. | 10 | 15.50 | 5.00 | 1.75 | 16.50 | 6.25 | 9.00 | 63.00 | 30.80 | 99.00 | 20.00 | |
| 2nd | Mean | 9 | 5.90 | 4.00 | 1.93 | 2.76 | 1.07 | 2.42 | 33.27 | 28.44 | 33.27 | 2.93 |
| SD | 1 | 3.12 | 2.59 | 1.27 | 2.61 | 0.89 | 2.39 | 26.50 | 5.40 | 10.80 | 3.65 | |
| Min. | 5 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 17.30 | 17.00 | 0.00 | |
| Max. | 10 | 15.25 | 12.50 | 5.75 | 12.00 | 4.50 | 9.00 | 99.00 | 46.00 | 68.00 | 17.00 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olate-Olave, V.R.; Verde, M.; Vallejos, L.; Perez Raymonda, L.; Cortese, M.C.; Doorn, M. Bee Health and Productivity in Apis mellifera, a Consequence of Multiple Factors. Vet. Sci. 2021, 8, 76. https://doi.org/10.3390/vetsci8050076
Olate-Olave VR, Verde M, Vallejos L, Perez Raymonda L, Cortese MC, Doorn M. Bee Health and Productivity in Apis mellifera, a Consequence of Multiple Factors. Veterinary Sciences. 2021; 8(5):76. https://doi.org/10.3390/vetsci8050076
Chicago/Turabian StyleOlate-Olave, Verónica Rachel, Mayda Verde, Leslie Vallejos, Leonel Perez Raymonda, Maria Carla Cortese, and Marnix Doorn. 2021. "Bee Health and Productivity in Apis mellifera, a Consequence of Multiple Factors" Veterinary Sciences 8, no. 5: 76. https://doi.org/10.3390/vetsci8050076
APA StyleOlate-Olave, V. R., Verde, M., Vallejos, L., Perez Raymonda, L., Cortese, M. C., & Doorn, M. (2021). Bee Health and Productivity in Apis mellifera, a Consequence of Multiple Factors. Veterinary Sciences, 8(5), 76. https://doi.org/10.3390/vetsci8050076






