Bilateral Renal Large B Cell Lymphoma in a Dog: A Case Report and Review of the Literature
Abstract
1. Introduction
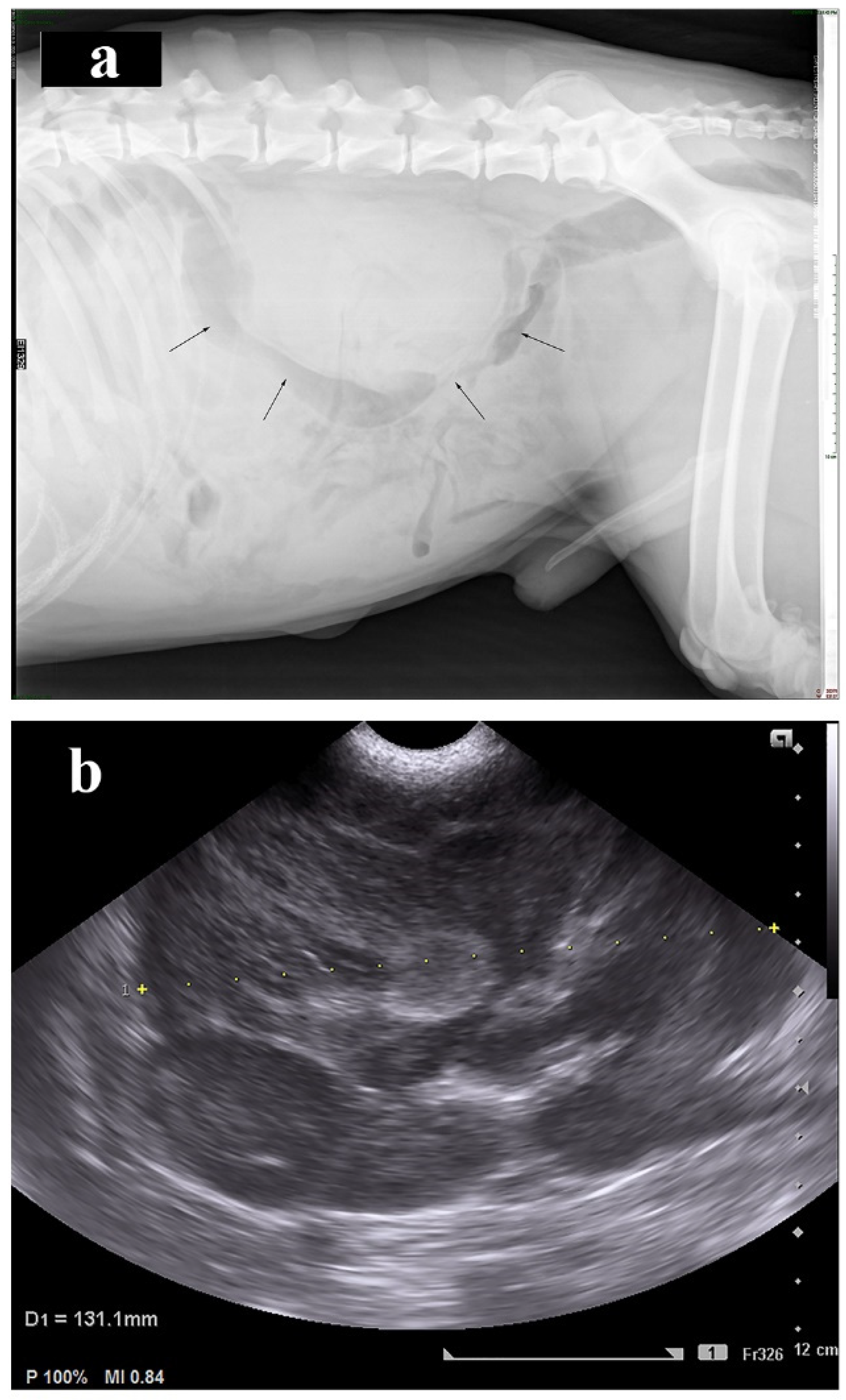
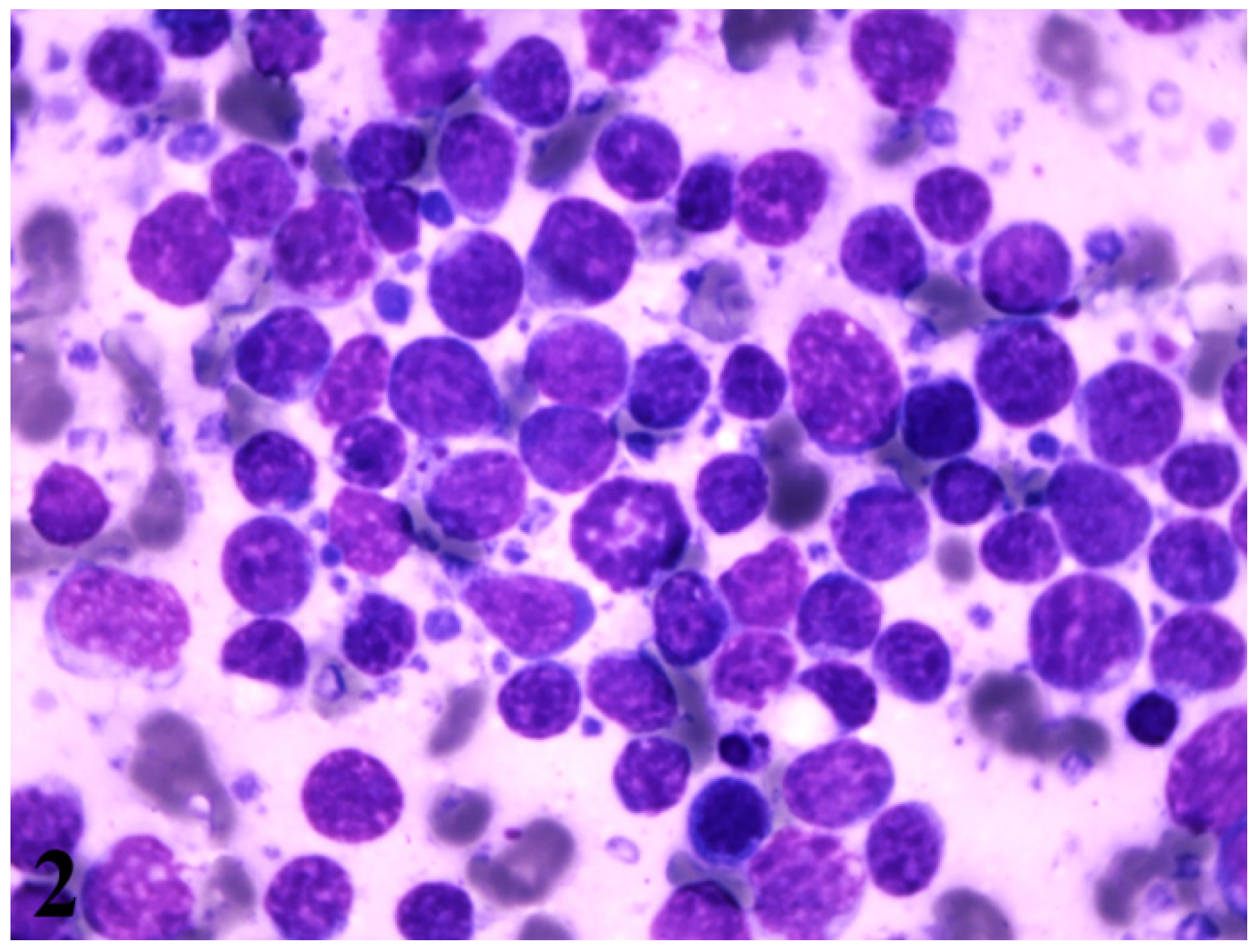
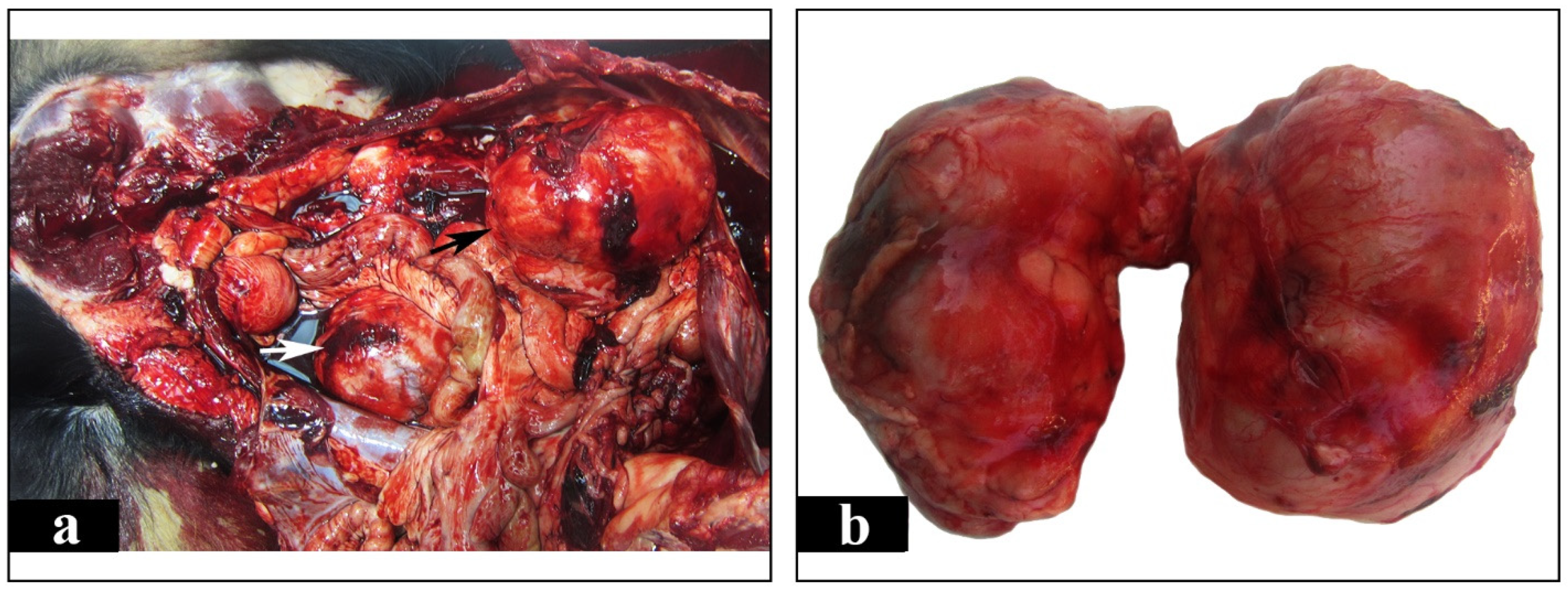
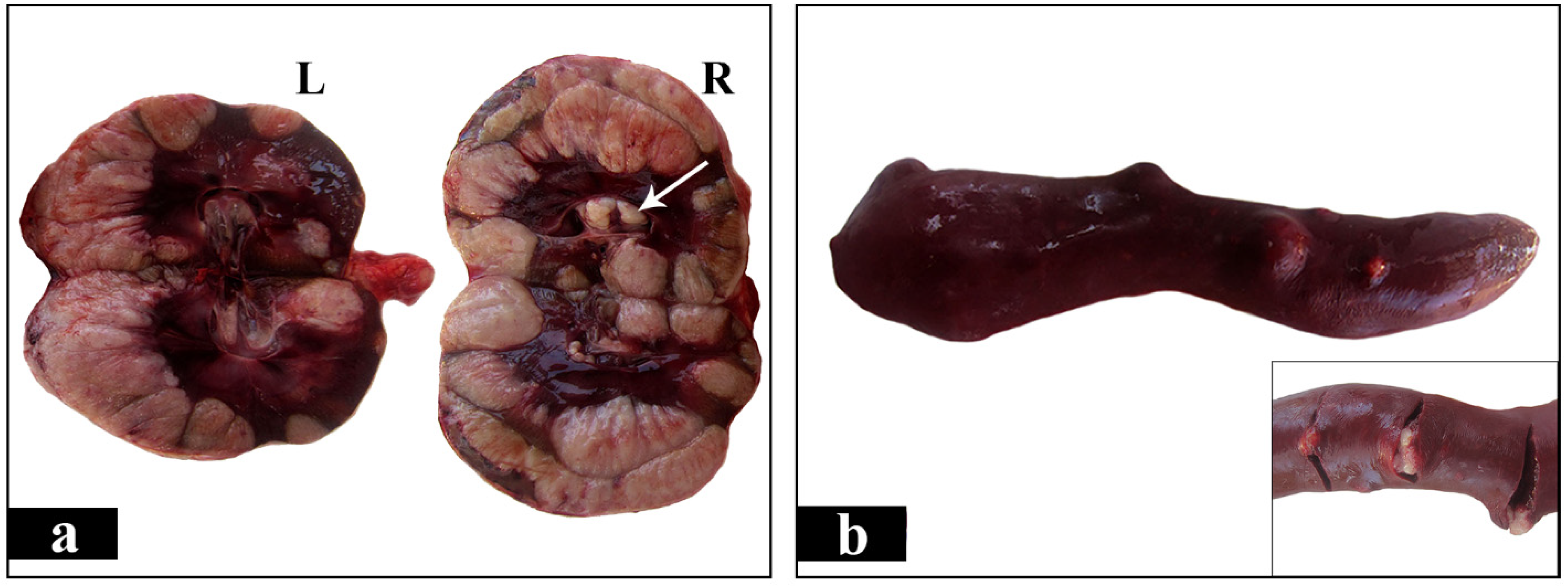
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Baskin, G.B.; De Paoli, A. Primary renal neoplasms of the dog. Vet. Pathol. 1977, 14, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Valli, V.E.; Bienzle, D.; Meuten, D.J.; Linder, K.E. Tumors of the Hemolymphatic System. In Tumors in Domestic Animals, 5th ed.; Meuten, D.J., Ed.; John Wiley & Sons: Ames, IA, USA, 2017; pp. 203–321. [Google Scholar]
- Cienava, E.A.; Barnhart, K.F.; Brown, R.; Mansell, J.; Dunstan, R.; Credille, K. Morphologic, immunohistochemical, and molecular characterization of hepatosplenic T-cell lymphoma in a dog. Vet. Clin. Pathol. 2004, 33, 105–110. [Google Scholar] [CrossRef]
- Dank, G.; Rassnick, K.M.; Kristal, O.; Rodriguez, C.O.; Clifford, C.A.; Ward, R.; Mallett, C.L.; Gieger, T.; Segev, G. Clinical characteristics, treatment, and outcome of dogs with presumed primary hepatic lymphoma: 18 cases (1992–2008). J. Am. Vet. Med. Assoc. 2011, 239, 966–971. [Google Scholar] [CrossRef] [PubMed]
- Kaldrymidou, E.; Papaioannou, N.; Poutahidis, T.; Karayannopoulou, M.; Gruys, E.; Toliou, T.; Tsangaris, T. Malignant lymphoma in nasal cavity and paranasal sinuses of a dog. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2000, 47, 457–462. [Google Scholar] [CrossRef]
- Keller, S.M.; Vernau, W.; Hodges, J.; Kass, P.H.; Vilches-Moure, J.G.; McElliot, V.; Moore, P.F. Hepatosplenic and hepatocytotropic T-cell lymphoma: Two distinct types of T-cell lymphoma in dogs. Vet. Pathol. 2013, 50, 281–290. [Google Scholar] [CrossRef]
- Long, S.N.; Johnston, P.E.; Anderson, T.J. Primary T-cell lymphoma of the central nervous system in a dog. J. Am. Vet. Med. Assoc. 2001, 218, 719–722. [Google Scholar] [CrossRef]
- Maiolino, P.; Devico, G. Primary Epitheliotropic T-cell Lymphoma of the Urinary Bladder in a Dog. Vet. Pathol. 2000, 37, 184–186. [Google Scholar] [CrossRef] [PubMed]
- Thuilliez, C.; Watrelot-Virieux, D.; Chanut, F.; Fournel-Fleury, C.; Ponce, F.; Marchal, T. Presumed primary muscular lymphoma in a dog. J. Vet. Diagn. Investig. 2008, 20, 824–826. [Google Scholar] [CrossRef]
- Takeuchi, Y.; Fujino, Y.; Goto-Koshino, Y.; Ohno, K.; Uchida, K.; Nakayama, H.; Tsujimoto, H. Long Term Survival of Primary Skeletal Muscle Lymphoma in a Miniature Dachshund. J. Vet. Med. Sci. 2010, 72, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Bryan, J.N.; Henry, C.J.; Turnquist, S.E.; Tyler, J.W.; Liptak, J.M.; Rizzo, S.A.; Sfiligoi, G.; Steinberg, S.J.; Smith, A.N.; Jackson, T. Primary renal neoplasia of dogs. J. Vet. Intern. Med. 2006, 20, 1155–1160. [Google Scholar] [CrossRef]
- Batchelor, D.J.; Bright, S.R.; Ibarrola, P.; Tzannes, S.; Blackwood, L. Long-term survival after combination chemotherapy for bilateral renal malignant lymphoma in a dog. N. Z. Vet. J. 2006, 54, 147–150. [Google Scholar] [CrossRef]
- Breshears, M.A.; Meinkoth, J.H.; Stern, A.W.; Buoncompagni, S.; Thomason, J.D. Pathology in practice. Renal lymphoma. J. Am. Vet. Med. Assoc. 2011, 238, 167–169. [Google Scholar] [CrossRef]
- Cook, S.M.; Lothrop, C.D.J. Serum erythropoietin concentrations measured by radioimmunoassay in normal, polycythemic, and anemic dogs and cats. J. Vet. Intern. Med. 1994, 8, 18–25. [Google Scholar] [CrossRef]
- Cotchin, E. Further observations on neoplasms in dogs, with particular reference to site of origin and malignancy. Part II: Male genital, skeletal, lymphatic and other systems. Br. Vet. J. 1954, 110, 274–286. [Google Scholar] [CrossRef]
- Durno, A.S.; Webb, J.A.; Gauthier, M.J.; Bienzle, D. Polycythemia and inappropriate erythropoietin concentrations in two dogs with renal T-cell lymphoma. J. Am. Anim. Hosp. Assoc. 2011, 47, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Froment, R.; Gara-Boivin, C. Bilateral renal T-cell lymphoma with hepatic infiltration and secondary polycythemia in a dog: Utility of cytology slides. Can. Vet. J. 2015, 56, 1287–1291. [Google Scholar]
- Klein, M.K.; Cockerell, G.L.; Harris, C.K.; Withrow, S.J.; Lulich, J.P.; Ogilvie, G.K.; Norris, A.M.; Harvey, H.J.; Richardson, R.F.; Fowler, J.D.; et al. Canine primary renal neoplasms: A retrospective review of 54 cases. J. Am. Anim. Hosp. Assoc. 1988, 24, 443–452. [Google Scholar]
- Lane, E.P.; Lobetti, R.G. Renal T-cell lymphoma with cerebral metastasis in a dog with chronic canine ehrlichiosis. J. S. Afr. Vet. Assoc. 2002, 73, 83–85. [Google Scholar] [CrossRef][Green Version]
- Lascelles, B.D.; Monnet, E.; Liptak, J.; Johnson, J.; Dernell, W.S. Surgical treatment of right-sided renal lymphoma with invasion of the caudal vena cava. J. Small Anim. Pract. 2003, 44, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Nelson, R.W.; Hager, D.; Zanjani, E.D. Renal lymphosarcoma with inappropriate erythropoietin production in a dog. J. Am. Vet. Med. Assoc. 1983, 182, 1396–1397. [Google Scholar]
- Osborne, C.A.; Johnson, K.H.; Kurtz, H.J.; Hanlon, G.F. Renal lymphoma in the dog and cat. J. Am. Vet. Med. Assoc. 1971, 158, 2058–2070. [Google Scholar]
- Snead, E.C. A case of bilateral renal lymphosarcoma with secondary polycythaemia and paraneoplastic syndromes of hypoglycaemia and uveitis in an English springer spaniel. Vet. Comp. Oncol. 2005, 3, 139–144. [Google Scholar] [CrossRef]
- Walter, P.A.; Feeney, D.A.; Johnston, G.R.; O’Leary, T.P. Ultrasonographic evaluation of renal parenchymal diseases in dogs: 32 cases. J. Am. Vet. Med. Assoc. 1986, 191, 999–1007. [Google Scholar]
- Zhao, D.; Yamaguchi, R.; Tateyama, S.; Yamazaki, Y.; Ogawa, H. Bilateral renal lymphosarcoma in a dog. J. Vet. Med. Sci. 1993, 55, 657–659. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zandvliet, M. Canine lymphoma: A review. Vet. Q. 2016, 36, 76–104. [Google Scholar] [CrossRef]
- Chen, X.; Hu, D.; Fang, L.; Chen, Y.; Che, X.; Tao, J.; Weng, G.; Ye, X. Primary renal lymphoma: A case report and literature review. Oncol. Lett. 2016, 12, 4001–4008. [Google Scholar] [CrossRef] [PubMed]
- Comazzi, S.; Marelli, S.; Cozzi, M.; Rizzi, R.; Finotello, R.; Henriques, J.; Pastor, J.; Ponce, F.; Rohrer-Bley, C.; Rütgen, B.C.; et al. Breed-associated risks for developing canine lymphoma differ among countries: An European canine lymphoma network study. BMC Vet. Res. 2018, 14, 232. [Google Scholar] [CrossRef]
- Grindem, C.B.; Breitschwerdt, E.B.; Corbett, W.T.; Page, R.L.; Jans, H.E. Thrombocytopenia Associated with Neoplasia in Dogs. J. Vet. Intern. Med. 1994, 8, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Kooistra, H.S. Diseases and Disorders. In Clinical Veterinary Advisor: Dogs and Cats, 3rd ed.; Cote, E., Ed.; Elsevier: St Louis, MI, USA, 2017; pp. 4968–4979. [Google Scholar]
- Nelson, R.W. Endocrine, Metabolic, and Lipid Disorders. In Small Animal Clinical Diagnosis by Laboratory Methods, 5th ed.; Willard, M.D., Tvedten, H., Eds.; Elsevier: St Louis, MI, USA, 2012; pp. 156–190. [Google Scholar]
- Weiss, D.J.; Tvedten, H. Erythrocyte Disorders. In Small Animal Clinical Diagnosis by Laboratory Methods, 5th ed.; Willard, M.D., Tvedten, H., Eds.; Elsevier: St Louis, MI, USA, 2012; pp. 38–62. [Google Scholar]
- Meuten, D.J.; Meuten, T.L.K. Tumors of the Urinary System. In Tumors in Domestic Animals, 5th ed.; Meuten, D.J., Ed.; John Wiley & Sons: Ames, IA, USA, 2017; pp. 632–688. [Google Scholar]
- Rissman, C.M.; Dagrosa, L.M.; Pettus, J.R.; Dillon, J.L.; Sverrisson, E.F. Primary renal lymphoma: An unusual finding following radical nephrectomy. Clin. Nephrol. Case Stud. 2017, 5, 1–4. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ingle, S.B.; Ingle, C.R.H. Primary splenic lymphoma: Current diagnostic trends. World J. Clin. Cases 2016, 4, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, T.; Coombes, B.; Brasfield, R.D. Primary malignant neoplasms of the spleen. Surg. Gynecol. Obstet. 1965, 120, 947–960. [Google Scholar]
- Skarin, A.T.; Davey, F.R.; Moloney, W.C. Lymphosarcoma of the spleen. Arch. Intern. Med. 1971, 127, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, B.B.; Osborne, B.M.; Butler, J.J. Primary splenic presentation of malignant lymphoma and related disorders—A study of 49 cases. Cancer 1984, 54, 1606–1619. [Google Scholar] [CrossRef]
- Kehoe, J.; Straus, D.J. Primary lymphoma of the spleen: Clinical features and outcome after splenectomy. Cancer 1988, 62, 1434–1438. [Google Scholar] [CrossRef]

| Breed | Age | Sex | Laboratory Tests | Imaging | FNA | Necropsy | Histopathology | IHC/Cell Type |
|---|---|---|---|---|---|---|---|---|
| Scottish Terrier | 5 | ♂ | NA | NA | NA | (+) | NA | NA |
| Basenji | 2 | ♀ | (+) | (+) | NA | (+) | (+) | NA |
| Collie | 5.5 | ♂ | NA | NA | NA | (+) | (+) | NA |
| Labrador | 9 | ♂ | (+) | (+) | NA | NA | (+) | NA |
| Unknown | NA | NA | NA | (+) | NA | NA | (+) | NA |
| Medium or large breed | NA | NA | (+) | (+) | NA | (+) | (+) | NA |
| ShibaInu | 3 | ♀ | (+) | (+) | NA | (+) | (+) | NA |
| Doberman | 5 | ♀ | (+) | NA | NA | NA | (+) | NA |
| Staffordshire | 5 | ♀ | (+) | NA | NA | (+) | (+) | (+)/T |
| Basset Hound | 8 | ♂ | (+) | (+) | NA | NA | (+) | (+)/T |
| Springer Spaniel | 8 | ♂ | (+) | (+) | (+) | (+) | (+) | NA |
| Flat coated Retriever | 6 | ♂ | (+) | (+) | (+) | NA | NA | NA |
| Mixed breed | 2 | ♂ | (+) | (+) | NA | (+) | (+) | (+)/T |
| Cocker Spaniel | 3 | ♂ | (+) | (+) | NA | (+) | (+) | (+)/T |
| Border Collie | 9 | ♀ | (+) | (+) | NA | (+) | (+) | (+)/T |
| Bernese mountain | 8 | ♂ | (+) | (+) | (+) | NA | NA | (+)/T |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Apostolopoulou, E.P.; Vlemmas, I.; Pardali, D.; Adamama-Moraitou, K.K.; Poutahidis, T.; Papadopoulou, P.L.; Brellou, G.D. Bilateral Renal Large B Cell Lymphoma in a Dog: A Case Report and Review of the Literature. Vet. Sci. 2021, 8, 258. https://doi.org/10.3390/vetsci8110258
Apostolopoulou EP, Vlemmas I, Pardali D, Adamama-Moraitou KK, Poutahidis T, Papadopoulou PL, Brellou GD. Bilateral Renal Large B Cell Lymphoma in a Dog: A Case Report and Review of the Literature. Veterinary Sciences. 2021; 8(11):258. https://doi.org/10.3390/vetsci8110258
Chicago/Turabian StyleApostolopoulou, Emmanouela P., Ioannis Vlemmas, Dimitra Pardali, Katerina K. Adamama-Moraitou, Theofilos Poutahidis, Paraskevi L. Papadopoulou, and Georgia D. Brellou. 2021. "Bilateral Renal Large B Cell Lymphoma in a Dog: A Case Report and Review of the Literature" Veterinary Sciences 8, no. 11: 258. https://doi.org/10.3390/vetsci8110258
APA StyleApostolopoulou, E. P., Vlemmas, I., Pardali, D., Adamama-Moraitou, K. K., Poutahidis, T., Papadopoulou, P. L., & Brellou, G. D. (2021). Bilateral Renal Large B Cell Lymphoma in a Dog: A Case Report and Review of the Literature. Veterinary Sciences, 8(11), 258. https://doi.org/10.3390/vetsci8110258






