Dermacentor marginatus and Dermacentor reticulatus, and Their Infection by SFG Rickettsiae and Francisella-Like Endosymbionts, in Mountain and Periurban Habitats of Northwestern Italy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Dermacentor spp. Ticks Collection
2.3. Tick-Borne Pathogens Detection
2.4. Statistical Analyses
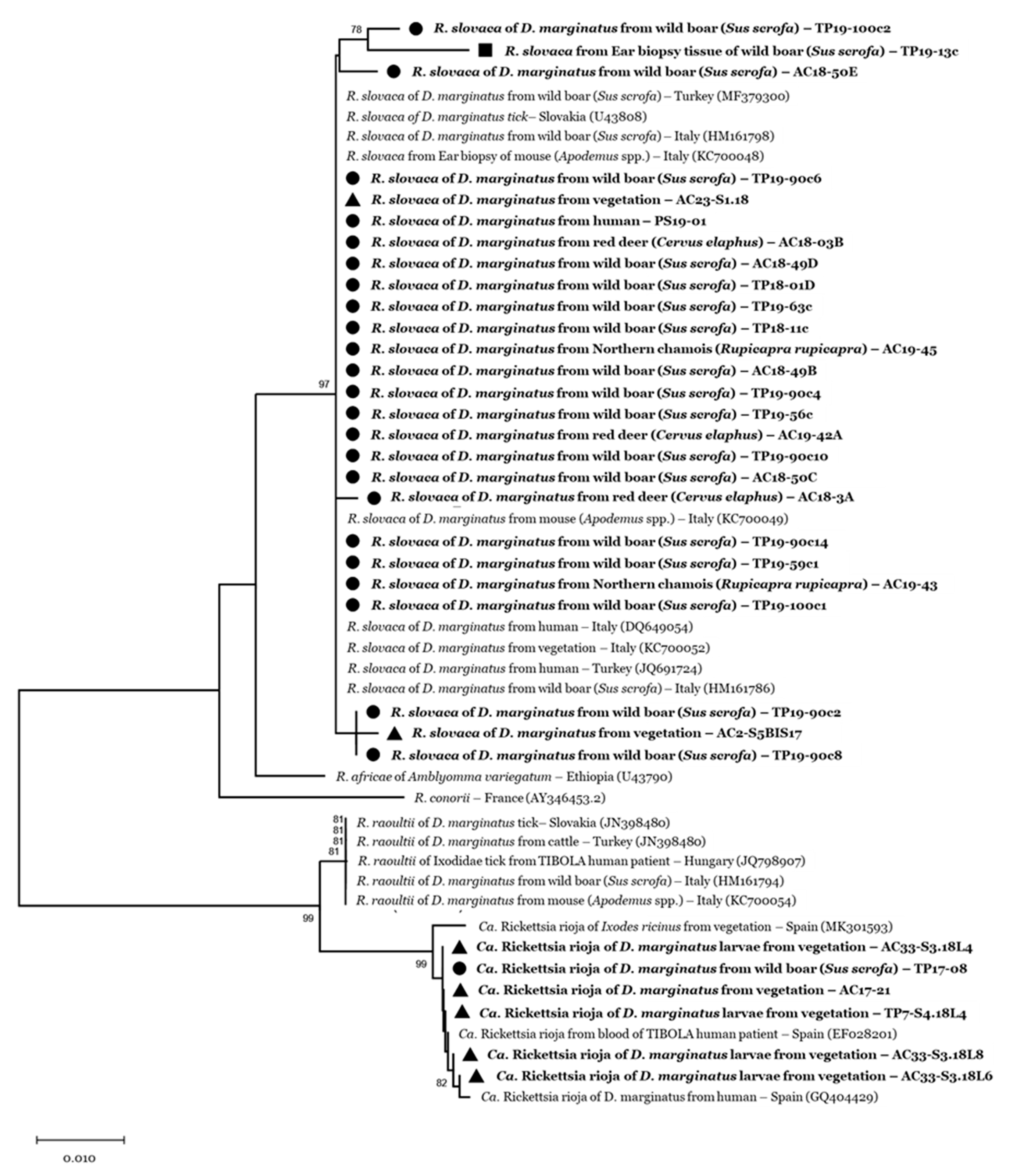
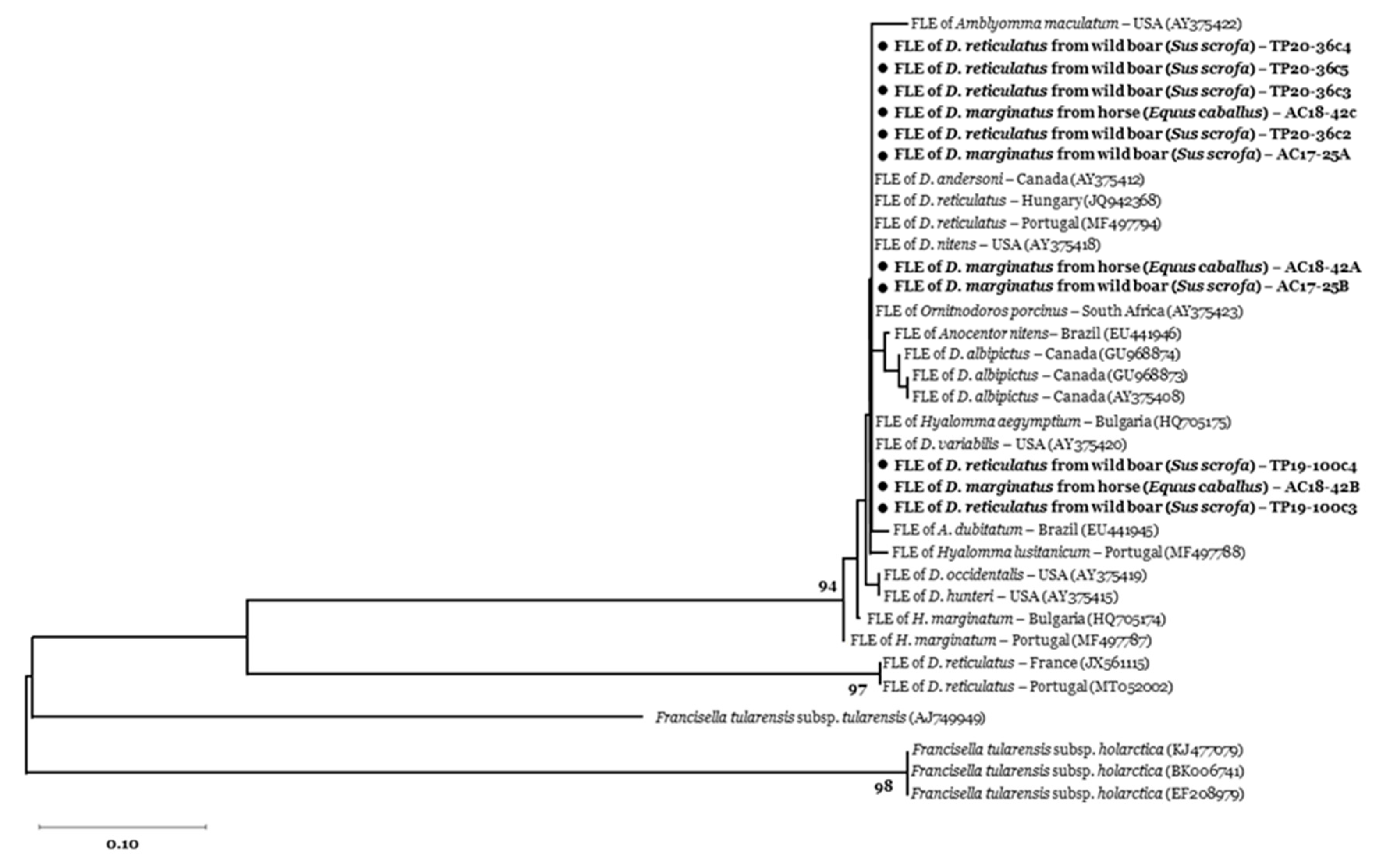
2.5. Phylogenetic Analyses
3. Results
3.1. Tick Collection
3.2. Tick-Borne Pathogen Infection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Boulanger, N.; Boyer, P.; Talagrand-Reboul, E.; Hansmann, Y. Ticks and tick-borne diseases. Med. Mal. Infect. 2019, 49, 87–97. [Google Scholar] [CrossRef]
- Medlock, J.M.; Hansford, K.M.; Bormane, A.; Derdakova, M.; Estrada-Peña, A.; George, J.C.; Golovljova, I.; Jaenson, T.G.; Jensen, J.K.; Jensen, P.M.; et al. Driving forces for changes in geographical distribution of Ixodes ricinus ticks in Europe. Parasit. Vectors. 2013, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Dautel, H.; Dippel, C.; Oehme, R.; Hartelt, K.; Schettler, E. Evidence for an increased geographical distribution of Dermacentor reticulatus in Germany and detection of Rickettsia sp. RpA4. Int. J. Med. Microbiol. 2006, 296, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Bullová, E.; Lukáň, M.; Stanko, M.; Petko, B. Spatial distribution of Dermacentor reticulatus tick in Slovakia in the beginning of the 21st century. Vet. Parasitol. 2009, 165, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Karbowiak, G. The occurrence of the Dermacentor reticulatus tick-its expansion to new areas and possible causes. Ann. Parasitol. 2014, 60, 37–47. [Google Scholar]
- Jongejan, F.; Ringenier, M.; Putting, M.; Berger, L.; Burgers, S.; Kortekaas, R.; Lenssen, J.; van Roessel, M.; Wijnveld, M.; Madder, M. Novel foci of Dermacentor reticulatus ticks infected with Babesia canis and Babesia caballi in the Netherlands and in Belgium. Parasit. Vectors. 2015, 8, 232. [Google Scholar] [CrossRef]
- Földvári, G.; Široký, P.; Szekeres, S.; Majoros, G.; Sprong, H. Dermacentor reticulatus: A vector on the rise. Parasit. Vectors. 2016, 9, 314. [Google Scholar] [CrossRef]
- Mierzejewska, E.J.; Estrada-Peña, A.; Alsarraf, M.; Kowalec, M.; Bajer, A. Mapping of Dermacentor reticulatus expansion in Poland in 2012–2014. Ticks Tick Borne Dis. 2016, 7, 94–106. [Google Scholar] [CrossRef]
- Rubel, F.; Brugger, K.; Pfeffer, M.; Chitimia-Dobler, L.; Didyk, Y.M.; Leverenz, S.; Dautel, H.; Kahl, O. Geographical distribution of Dermacentor marginatus and Dermacentor reticulatus in Europe. Ticks Tick Borne Dis. 2016, 7, 224–233. [Google Scholar] [CrossRef]
- Olivieri, E.; Zanzani, S.A.; Latrofa, M.S.; Lia, R.P.; Dantas-Torres, F.; Otranto, D.; Manfredi, M.T. The southernmost foci of Dermacentor reticulatus in Italy and associated Babesia canis infection in dogs. Parasit. Vectors. 2016, 9, 213. [Google Scholar] [CrossRef]
- Olivieri, E.; Gazzonis, A.L.; Zanzani, S.A.; Veronesi, F.; Manfredi, M.T. Seasonal dynamics of adult Dermacentor reticulatus in a peri-urban park in southern Europe. Ticks Tick Borne Dis. 2017, 8, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Maurelli, M.P.; Pepe, P.; Colombo, L.; Armstrong, R.; Battisti, E.; Morgoglione, M.E.; Counturis, D.; Rinaldi, L.; Cringoli, G.; Ferroglio, E.; et al. A national survey of Ixodidae ticks on privately owned dogs in Italy. Parasit. Vectors. 2018, 11, 420. [Google Scholar] [CrossRef]
- Cringoli, G.; Iori, A.; Rinaldi, L.; Veneziano, V.; Genchi, C. Zecche. Mappe Parassitologiche; Ronaldo Editore: Napoli, Italy, 2005; pp. 144–151. [Google Scholar]
- Estrada-Peña, A.; Jongejan, F. Ticks feeding on humans: A review of records on human-biting Ixodoidea with special reference to pathogen transmission. Exp. Appl. Acarol. 1999, 23, 685–715. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Peña, A.; Mihalca, A.D.; Petney, T.N. Ticks of Europe and North Africa: A Guide to Species Identification; Springer Nature: Cham, Switzerland, 2017; pp. 279–291. [Google Scholar] [CrossRef]
- Moraga-Fernández, A.; Ruiz-Fons, F.; Habela, M.A.; Royo-Hernández, L.; Calero-Bernal, R.; Gortazar, C.; de la Fuente, J.; Fernández de Mera, I.G. Detection of new Crimean-Congo haemorrhagic fever virus genotypes in ticks feeding on deer and wild boar, Spain. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Gargili, A.; Estrada-Peña, A.; Spengler, J.R.; Lukashev, A.; Nuttall, P.A.; Bente, D.A. The role of ticks in the maintenance and transmission of Crimean-Congo hemorrhagic fever virus: A review of published field and laboratory studies. Antiviral Res. 2017, 144, 93–119. [Google Scholar] [CrossRef] [PubMed]
- Portillo, A.; Santibáñez, S.; García-Álvarez, L.; Palomar, A.M.; Oteo, J.A. Rickettsioses in Europe. Microbes Infect. 2015, 17, 834–838. [Google Scholar] [CrossRef]
- Dubourg, G.; Socolovschi, C.; Del Giudice, P.; Fournier, P.E.; Raoult, D. Scalp eschar and neck lymphadenopathy after tick bite: An emerging syndrome with multiple causes. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 1449–1456. [Google Scholar] [CrossRef]
- Selmi, M.; Bertolotti, L.; Tomassone, L.; Mannelli, A. Rickettsia slovaca in Dermacentor marginatus and tick-borne lymphadenopathy, Tuscany, Italy. Emerg. Infect. Dis. 2008, 14, 817–820. [Google Scholar] [CrossRef]
- Ruiz-Fons, F.; Fernández de Mera, I.G.; Acevedo, P.; Höfle, U.; Vicente, J.; de la Fuente, J.; Gortazar, C. Ixodid ticks parasitizing Iberian red deer (Cervus elaphus hispanicus) and European wild boar (Sus scrofa) from Spain: Geographical and temporal distribution. Vet. Parasitol. 2006, 140, 133–142. [Google Scholar] [CrossRef]
- Chitimia-Dobler, L. Spatial distribution of Dermacentor reticulatus in Romania. Vet Parasitol. 2015, 214, 219–223. [Google Scholar] [CrossRef]
- Selmi, M.; Ballardini, M.; Salvato, L.; Ricci, E. Rickettsia spp. in Dermacentor marginatus ticks: Analysis of the host-vector-pathogen interactions in a northern Mediterranean area. Exp. Appl. Acarol. 2017, 72, 79–91. [Google Scholar] [CrossRef]
- Gomez-Barroso, D.; Vescio, M.F.; Bella, A.; Ciervo, A.; Busani, L.; Rizzo, C.; Rezza, G.; Pezzotti, P. Mediterranean spotted fever rickettsiosis in Italy, 2001–2015: Spatio-temporal distribution based on hospitalization records. Ticks Tick Borne Dis. 2019, 10, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Mancini, F.; Rezza, G.; Ciervo, A. Le rickettsiosi in Italia: Diagnosi e sorveglianza. Notiziario dell’Istituto Superiore di Sanità 2015, 28, 3–8. [Google Scholar]
- Regional Service for the Epidemiology of Infectious Diseases (SeREMI). Available online: https://www.seremi.it/ (accessed on 20 August 2020).
- Garcia-Vozmediano, A.; Krawczyk, A.I.; Sprong, H.; Rossi, L.; Ramassa, E.; Tomassone, L. Ticks climb the mountains: Ixodid tick infestation and infection by tick-borne pathogens in Western Alps. Ticks Tick Borne Dis. 2020, 11, 101489. [Google Scholar] [CrossRef] [PubMed]
- Yeh, M.T.; Bak, J.M.; Hu, R.; Nicholson, M.C.; Kelly, C.; Mather, T.N. Determining the duration of Ixodes scapularis (Acari: Ixodidae) attachment to tick-bite victims. J. Med. Entomol. 1995, 32, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Martello, E.; Mannelli, A.; Grego, E.; Ceballos, L.A.; Ragagli, C.; Stella, M.C.; Tomassone, L. Borrelia burgdorferi sensu lato and spotted fever group rickettsiae in small rodents and attached ticks in the Northern Apennines, Italy. Ticks Tick Borne Dis. 2019, 10, 862–867. [Google Scholar] [CrossRef]
- Labruna, M.B.; Whitworth, T.; Bouyer, D.H.; Mcbride, J.W.; Pinter, A.; Popov, V.; Gennari, S.M.; Walker, D.H. Rickettsia species infecting Amblyomma cooperi ticks from an area in the state of Sao Paulo, Brazil, where brazilian Spotted Fever is endemic. J. Clin. Microbiol. 2004, 42, 90–98. [Google Scholar] [CrossRef]
- Fournier, P.E.; Roux, V.; Raoult, D. Phylogenetic analysis of spotted fever group rickettsiae by study of the outer surface protein rOmpA. Int. J. Syst. Bacteriol. 1998, 48, 839–849. [Google Scholar] [CrossRef]
- Sumner, J.W.; Durden, L.A.; Goddard, J.; Stromdahl, E.Y.; Clark, K.L.; Reeves, W.K.; Paddock, C.D. Gulf coast ticks (Amblyomma maculatum) and Rickettsia parkeri, United States. Emerg. Infect. Dis. 2007, 13, 751–753. [Google Scholar] [CrossRef]
- Jensen, W.A.; Fall, M.Z.; Rooney, J.; Kordick, D.L.; Breitschwerdt, E.B. Rapid identification and differentiation of Bartonella species using a single-step PCR assay. J. Clin. Microbiol. 2000, 38, 1717–1722. [Google Scholar] [CrossRef]
- Michelet, L.; Bonnet, S.; Madani, N.; Moutailler, S. Discriminating Francisella tularensis and Francisella-like endosymbionts in Dermacentor reticulatus ticks: Evaluation of current molecular techniques. Vet. Microbiol. 2013, 163, 399–403. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: http://www.r-project.org/index.html (accessed on 5 September 2020).
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl. Acids. Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positions-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Manfredi, M.T. Dermacentor reticulatus Fabricius 1794 nel cane in Italia. Atti Soc. Ital. Sci. Vet. 1995, 49, 759–760. [Google Scholar]
- Iori, A.; Gabrielli, S.; Calderini, P.; Moretti, A.; Pietrobelli, M.; Tampieri, M.P.; Galuppi, R.; Cancrini, G. Tick reservoirs for piroplasms in central and northern Italy. Vet. Parasitol. 2010, 170, 291–296. [Google Scholar] [CrossRef]
- Acquaotta, F.; Fratianni, S.; Garzena, D. Temperature changes in the North-Western Italian Alps from 1961 to 2010. Theor. Appl. Climatol. 2015, 122, 619–634. [Google Scholar] [CrossRef]
- Selmi, M.; Tomassone, L.; Ceballos, L.A.; Crisci, A.; Ragagli, C.; Pintore, M.D.; Mignone, W.; Pautasso, A.; Ballardini, M.; Casalone, C.; et al. Analysis of the environmental and host-related factors affecting the distribution of the tick Dermacentor marginatus. Exp. Appl. Acarol. 2018, 75, 209–225. [Google Scholar] [CrossRef]
- Audino, T.; Pautasso, A.; Bellavia, V.; Carta, V.; Ferrari, A.; Verna, F.; Grattarola, C.; Iulini, B.; Pintore, M.; Bardelli, M.; et al. Ticks Infesting Humans in North-Western Italy and Associated Pathogens: A Cross-Sectional Study in a Three-Year Period (2017–2019) in North-Western Italy, PREPRINT (Version 1). 2020. [CrossRef]
- Toledo, A.; Olmeda, A.S.; Escudero, R.; Jado, I.; Valcárcel, F.; Casado-Nistal, M.A.; Rodríguez-Vargas, M.; Gil, H.; Anda, P. Tick-borne zoonotic bacteria in ticks collected from Central Spain. Am. J. Trop. Med. Hyg. 2009, 81, 67–74. [Google Scholar] [CrossRef]
- Ortuño, A.; Quesada, M.; López-Claessens, S.; Castellà, J.; Sanfeliu, I.; Antón, E.; Segura-Porta, F. The role of wild boar (Sus scrofa) in the eco-epidemiology of R. slovaca in Northeastern Spain. Vector Borne Zoonotic Dis. 2007, 7, 59–69. [Google Scholar] [CrossRef]
- Selmi, M.; Martello, E.; Bertolotti, L.; Bisanzio, D.; Tomassone, L. Rickettsia slovaca and Rickettsia raoultii in Dermacentor marginatus ticks collected on wild boars in Tuscany, Italy. J. Med. Entomol. 2009, 46, 1490–1493. [Google Scholar] [CrossRef] [PubMed]
- Špitálska, E.; Štefanidesová, K.; Kocianová, E.; Boldiš, V. Rickettsia slovaca and Rickettsia raoultii in Dermacentor marginatus and Dermacentor reticulatus ticks from Slovak Republic. Exp. Appl. Acarol. 2012, 57, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Tomassone, L.; Portillo, A.; Nováková, M.; de Sousa, R.; Oteo, J.A. Neglected aspects of tick-borne rickettsioses. Parasit. Vectors. 2018, 11, 263. [Google Scholar] [CrossRef] [PubMed]
- Parola, P.; Raoult, D. Ticks and tickborne bacterial diseases in humans: An emerging infectious threat. Clin. Infect. Dis. 2001, 32, 897–928. [Google Scholar] [CrossRef]
- Martello, E.; Selmi, M.; Ragagli, C.; Ambrogi, C.; Stella, M.C.; Mannelli, A.; Tomassone, L. Rickettsia slovaca in immature Dermacentor marginatus and tissues from Apodemus spp. in the northern Apennines, Italy. Ticks Tick Borne Dis. 2013, 4, 518–521. [Google Scholar] [CrossRef]
- Zeroual, F.; Leulmi, H.; Bitam, I.; Benakhla, A. Molecular evidence of Rickettsia slovaca in spleen of wild boars in northeastern Algeria. New Microbes New Infect. 2018, 24, 17–20. [Google Scholar] [CrossRef]
- Cicculli, V.; Oscar, M.; Casabianca, F.; Villechenaud, N.; Charrel, R.; de Lamballerie, X.; Falchi, A. Molecular detection of spotted-fever group rickettsiae in ticks collected from domestic and wild animals in Corsica, France. Pathogens 2019, 8, 138. [Google Scholar] [CrossRef]
- Ibarra, V.; Oteo, J.A.; Portillo, A.; Santibáñez, S.; Blanco, J.R.; Metola, L.; Eiros, J.M.; Pérez-Martínez, L.; Sanz, M. Rickettsia slovaca infection: DEBONEL/TIBOLA. Ann. N. Y. Acad. Sci. 2006, 1078, 206–214. [Google Scholar] [CrossRef]
- Parola, P.; Rovery, C.; Rolain, J.M.; Brouqui, P.; Davoust, B.; Raoult, D. Rickettsia slovaca and R. raoultii in Tick-borne Rickettsioses. Emerg. Infect. Dis. 2009, 15, 1105–1108. [Google Scholar] [CrossRef]
- Dutto, M.; Selmi, M. Sindrome TIBOLA in Piemonte. Decidere Med. 2012, 12, 87–88. [Google Scholar]
- Remesar, S.; Díaz, P.; Portillo, A.; Santibáñez, S.; Prieto, A.; Díaz-Cao, J.M.; López, C.M.; Panadero, R.; Fernández, G.; Díez-Baños, P.; et al. Prevalence and molecular characterization of Rickettsia spp. in questing ticks from Northwestern Spain. Exp. Appl. Acarol. 2019, 79, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Portillo, A.; Ibarra, V.; Santibáñez, S.; Pérez-Martínez, L.; Blanco, J.R.; Oteo, J.A. Genetic characterisation of ompA, ompB and gltA genes from Candidatus Rickettsia rioja. Clin. Microbiol. Infect. 2009, 15, 307–308. [Google Scholar] [CrossRef]
- Pérez-Pérez, L.; Portillo, A.; Allegue, F.; Zulaica, A.; Oteo, J.A.; Caeiro, J.L.; Fabeiro, J.M. Dermacentor-borne Necrosis Erythema and Lymphadenopathy (DEBONEL): A case associated with Rickettsia rioja. Acta Derm. Venereol. 2010, 90, 214–215. [Google Scholar] [CrossRef] [PubMed]
- Santibáñez, S.; Portillo, A.; Santibáñez, P.; Palomar, A.M.; Oteo, J.A. Usefulness of rickettsial PCR assays for the molecular diagnosis of human rickettsioses. Enferm. Infecc. Microbiol. Clin. 2013, 31, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Rar, V.A.; Fomenko, N.V.; Dobrotvorsky, A.K.; Livanova, N.N.; Rudakova, S.A.; Fedorov, E.G.; Astanin, V.B.; Morozova, O.V. Tickborne pathogen detection, Western Siberia, Russia. Emerg. Infect. Dis. 2005, 11, 1708–1715. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, S.; de la Fuente, J.; Nicollet, P.; Liu, X.; Madani, N.; Blanchard, B.; Maingourd, C.; Alongi, A.; Torina, A.; Fernández de Mera, I.G.; et al. Prevalence of tick-borne pathogens in adult Dermacentor spp. ticks from nine collection sites in France. Vector Borne Zoonotic Dis. 2013, 13, 226–236. [Google Scholar] [CrossRef]
- Satta, G.; Chisu, V.; Cabras, P.; Fois, F.; Masala, G. Pathogens and symbionts in ticks: A survey on tick species distribution and presence of tick-transmitted micro-organisms in Sardinia, Italy. J. Med. Microbiol. 2011, 60, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Chisu, V.; Foxi, C.; Mannu, R.; Satta, G.; Masala, G. A five-year survey of tick species and identification of tick-borne bacteria in Sardinia, Italy. Ticks Tick Borne Dis. 2018, 9, 678–681. [Google Scholar] [CrossRef]
- Angelakis, E.; Pulcini, C.; Waton, J.; Imbert, P.; Socolovschi, C.; Edouard, S.; Dellamoniaca, P.; Raoult, D. Scalp eschar and neck lymphadenopathy caused by Bartonella henselae after tick bite. Clin. Infect. Dis. 2010, 50, 549–551. [Google Scholar] [CrossRef]
- Cotté, V.; Bonnet, S.; Le Rhun, D.; Le Naour, E.; Chauvin, A.; Boulouis, H.J.; Lecuelle, B.; Lilin, T.; Vayssier-Taussat, M. Transmission of Bartonella henselae by Ixodes ricinus. Emerg. Infect. Dis. 2008, 14, 1074–1080. [Google Scholar] [CrossRef]
- Reis, C.; Cote, M.; Le Rhun, D.; Lecuelle, B.; Levin, M.L.; Vayssier-Taussat, M.; Bonnet, S.I. Vector competence of the tick Ixodes ricinus for transmission of Bartonella birtlesii. PLoS Negl. Trop. Dis. 2011, 5, e1186. [Google Scholar] [CrossRef]
- Sprong, H.; Fonville, M.; Docters van Leeuwen, A.; Devillers, E.; Ibañez-Justicia, A.; Stroo, A.; Hansford, K.; Cull, B.; Medlock, J.; Heyman, P.; et al. Detection of pathogens in Dermacentor reticulatus in northwestern Europe: Evaluation of a high-throughput array. Heliyon 2019, 5, e01270. [Google Scholar] [CrossRef] [PubMed]
- Michelet, L.; Delannoy, S.; Devillers, E.; Umhang, G.; Aspan, A.; Juremalm, M.; Chirico, J.; van der Wal, F.J.; Sprong, H.; Boye Pihl, T.P.; et al. High-throughput screening of tick-borne pathogens in Europe. Front. Cell. Infect. Microbiol. 2014, 4, 103. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, I.N.; Mitkova, N.; Reye, A.L.; Hubschen, J.M.; Vatcheva-Dobrevska, R.S.; Dobreva, E.G.; Kantardjiev, T.V.; Muller, C.P. Detection of new Francisella-like tick endosymbionts in Hyalomma spp. and Rhipicephalus spp. (Acari: Ixodidae) from Bulgaria. Appl. Environ. Microbiol. 2011, 77, 5562–5565. [Google Scholar] [CrossRef] [PubMed]
- Sréter-Lancz, Z.; Széll, Z.; Sréter, T.; Márialigeti, K. Detection of a novel Francisella in Dermacentor reticulatus: A need for careful evaluation of PCR-based identification of Francisella tularensis in Eurasian ticks. Vector Borne Zoonotic Dis. 2009, 9, 123–126. [Google Scholar] [CrossRef]
- de Carvalho, I.L.; Santos, N.; Soares, T.; Ze-Ze, L.; Nuncio, M.S. Francisella-like endosymbiont in Dermacentor reticulatus collected in Portugal. Vector Borne Zoonotic Dis. 2011, 11, 185–188. [Google Scholar] [CrossRef]
- Cerutti, F.; Modesto, P.; Rizzo, F.; Cravero, A.; Jurman, I.; Costa, S.; Giammarino, M.; Mandola, M.L.; Goria, M.; Radovic, S.; et al. The microbiota of hematophagous ectoparasites collected from migratory birds. PLoS ONE 2018, 13, e0202270. [Google Scholar] [CrossRef]
- Gerhart, J.G.; Dutcher, H.A.; Brenner, A.E.; Moses, A.S.; Grubhoffer, L.; Raghavan, R. Multiple Acquisitions of Pathogen-Derived Francisella Endosymbionts in Soft Ticks. Genome Biol. Evol. 2018, 10, 607–615. [Google Scholar] [CrossRef]
- Sperling, J.; MacDonald, Z.; Normandeau, J.; Merrill, E.; Sperling, F.; Magor, K. Within-population diversity of bacterial microbiomes in winter ticks (Dermacentor albipictus). Ticks Tick Borne Dis. 2020, 11, 101535. [Google Scholar] [CrossRef]
- Scoles, G.A. Phylogenetic Analysis of the Francisella-like Endosymbionts of Dermacentor ticks. J. Med. Entomol. 2004, 41, 277–286. [Google Scholar] [CrossRef]
| Location | Source | Tick Species | N Ticks Tested | % SFG Rickettsiae Infection [95% CI] | |
|---|---|---|---|---|---|
| R. slovaca | Ca. R. rioja | ||||
| Mountain area | Vegetation | D. marginatus | 23 | 27.3 [10.7–50.2] | 4.5 [0.1–22.8] |
| Chamois | 2 | 50 [1.3–98.7] | 0 | ||
| Red deer | 9 | 77.8 [40.0–97.2] | 0 | ||
| Roe deer | 1 | 100 [2.5–100] | 0 | ||
| Wild boar | 14 | 35.7 [12.8–64.9] | 7.1 [0.2–33.9] | ||
| Cattle | 4 | 50.0 [6.8–93.2] | 0 | ||
| Dog | 3 | 33.3 [0.8–90.6] | 0 | ||
| Horse | 3 | 0 | 0 | ||
| Human | 2 | 100 [15.8–100.0] | 0 | ||
| nd | 1 | 100 [2.5–100] | 0 | ||
| Periurban area | Vegetation | D. marginatus | 19 | 10.5 [1.3–33.1] | 5.3 [0.1–26.0] |
| Wild boar | D. marginatus | 40 | 52.6 [35.8–69.0] | 5.3 [0.6–17.7] * | |
| D. reticulatus | 9 | 33.3 [7.5–70.1] | 0 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Vozmediano, A.; Giglio, G.; Ramassa, E.; Nobili, F.; Rossi, L.; Tomassone, L. Dermacentor marginatus and Dermacentor reticulatus, and Their Infection by SFG Rickettsiae and Francisella-Like Endosymbionts, in Mountain and Periurban Habitats of Northwestern Italy. Vet. Sci. 2020, 7, 157. https://doi.org/10.3390/vetsci7040157
Garcia-Vozmediano A, Giglio G, Ramassa E, Nobili F, Rossi L, Tomassone L. Dermacentor marginatus and Dermacentor reticulatus, and Their Infection by SFG Rickettsiae and Francisella-Like Endosymbionts, in Mountain and Periurban Habitats of Northwestern Italy. Veterinary Sciences. 2020; 7(4):157. https://doi.org/10.3390/vetsci7040157
Chicago/Turabian StyleGarcia-Vozmediano, Aitor, Giorgia Giglio, Elisa Ramassa, Fabrizio Nobili, Luca Rossi, and Laura Tomassone. 2020. "Dermacentor marginatus and Dermacentor reticulatus, and Their Infection by SFG Rickettsiae and Francisella-Like Endosymbionts, in Mountain and Periurban Habitats of Northwestern Italy" Veterinary Sciences 7, no. 4: 157. https://doi.org/10.3390/vetsci7040157
APA StyleGarcia-Vozmediano, A., Giglio, G., Ramassa, E., Nobili, F., Rossi, L., & Tomassone, L. (2020). Dermacentor marginatus and Dermacentor reticulatus, and Their Infection by SFG Rickettsiae and Francisella-Like Endosymbionts, in Mountain and Periurban Habitats of Northwestern Italy. Veterinary Sciences, 7(4), 157. https://doi.org/10.3390/vetsci7040157





