Longitudinal Study of Mycobacterium avium Subsp. paratuberculosis Antibody Kinetics in Dairy Cattle Using Sera and Milk throughout the Lactation Period
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statement
2.2. Selection of Animals
2.3. Sampling and MAP Antibody Testing
2.4. Statistical Analysis
3. Results
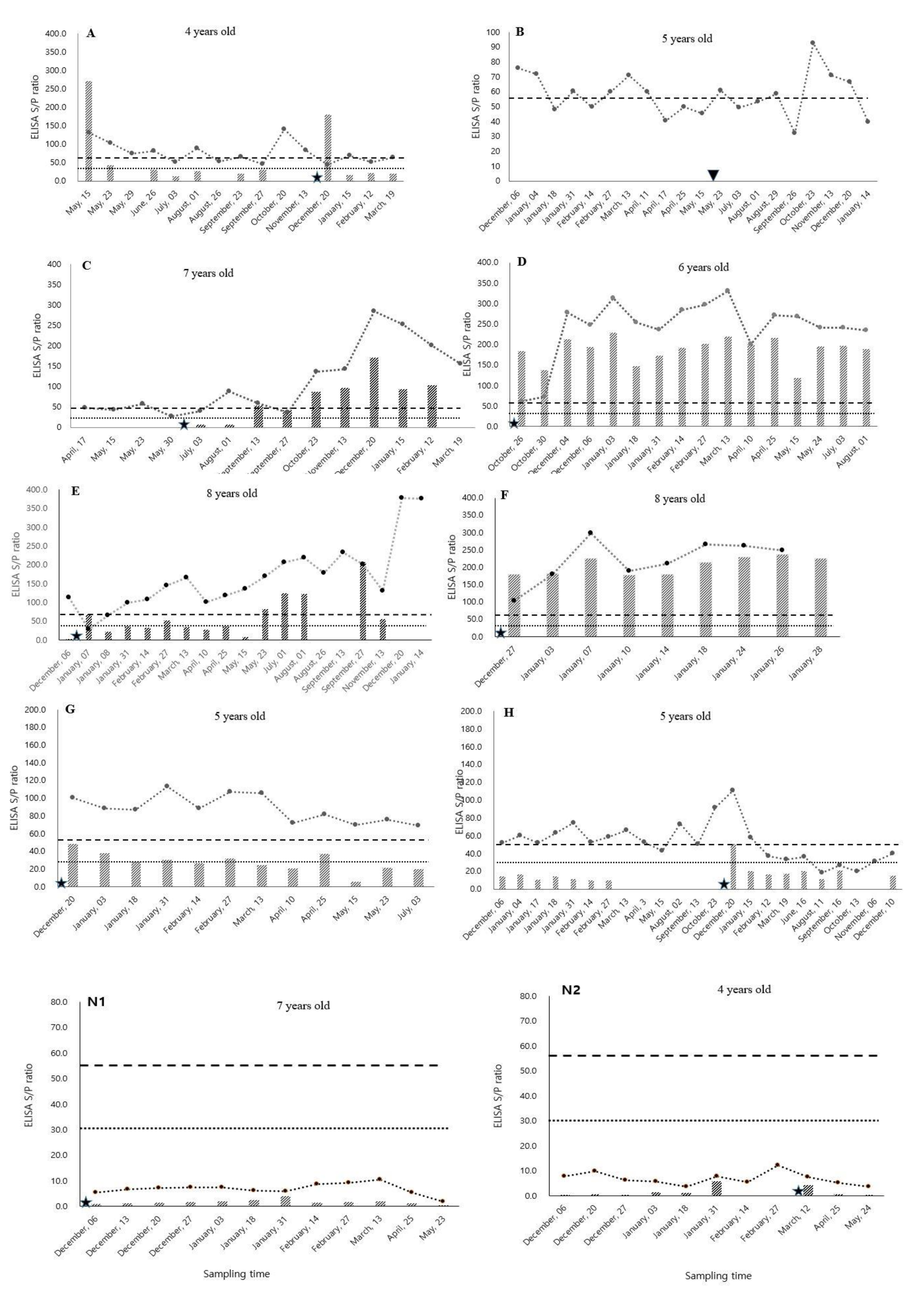
3.1. MAP Antibody Fluctuation
3.2. Dynamics of MAP Antibody Changes After Parturition
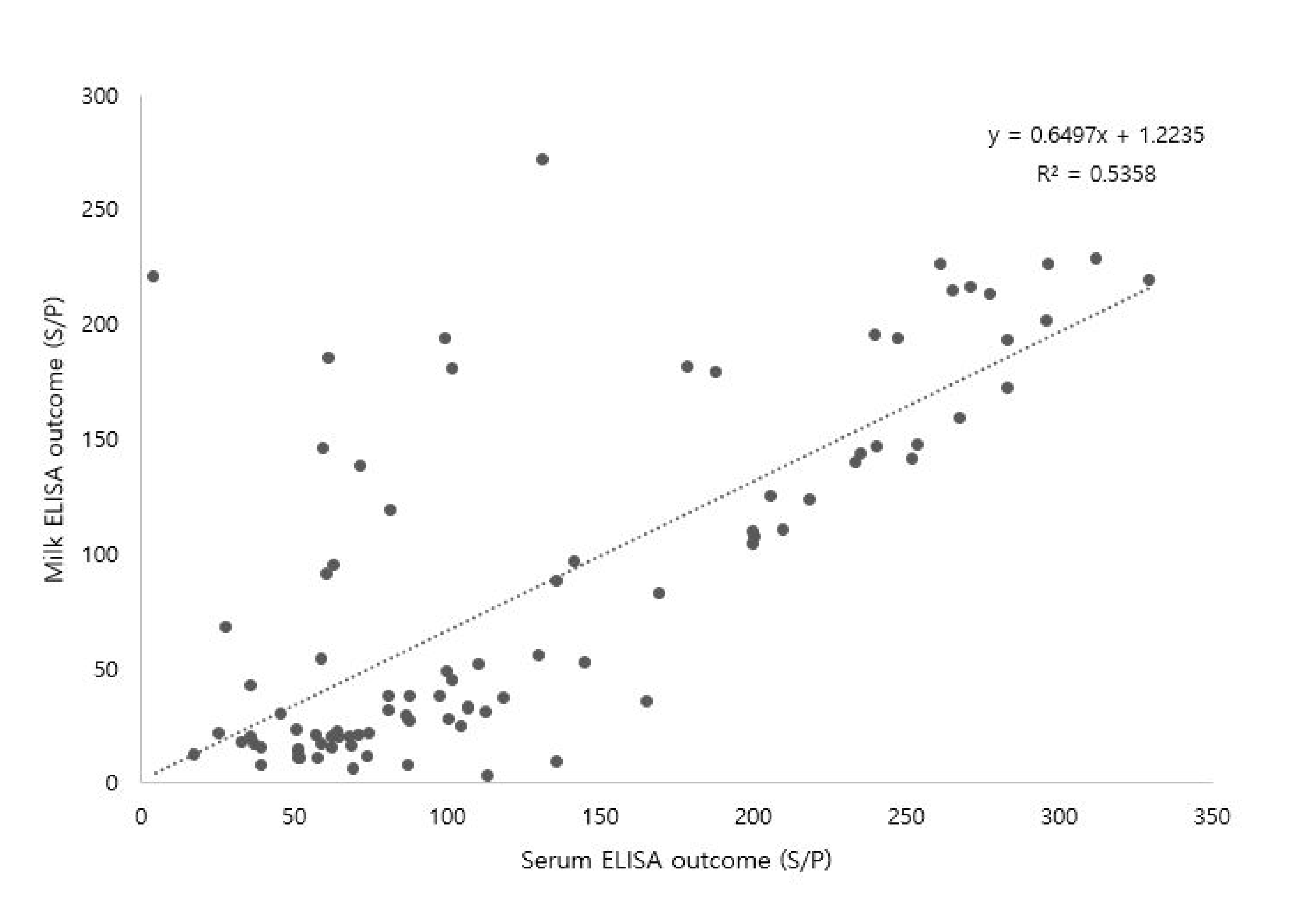
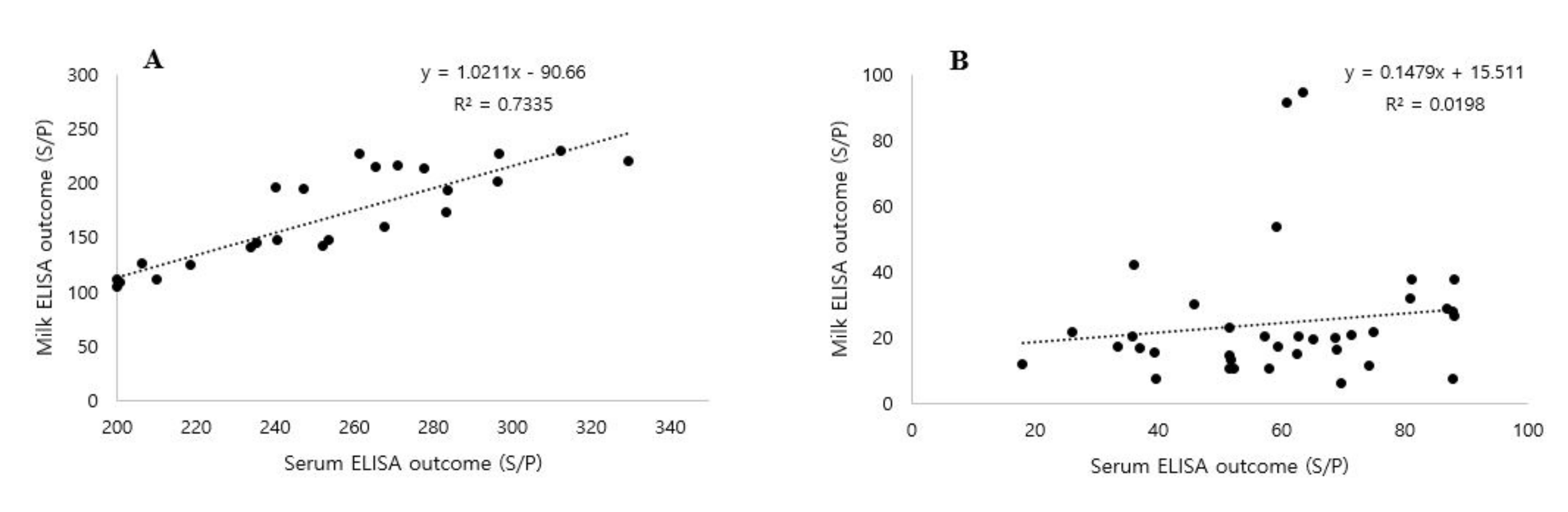
3.3. Correlation between Serum and Milk MAP Antibodies
4. Discussion
4.1. Fluctuation of MAP Antibody Titers
4.2. Increasing Trend of MAP Antibody Titers after Parturition
4.3. Steady or Decreasing Trend of MAP Antibody after Parturition
4.4. Effect of Age on MAP Antibody Titer
4.5. Correlation between Serum and Milk MAP Antibodies
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tiwari, A.; VanLeeuwen, J.A.; McKenna, S.L.B.; Keefe, G.P.; Barkema, H.W. Johne’s disease in Canada Part I: Clinical symptoms, pathophysiology, diagnosis, and prevalence in dairy herds. Can. Vet. J. 2006, 47, 874–882. [Google Scholar] [PubMed]
- Kim, J.; Ahn, J.; Woo, S.; Jo, D.; Jo, Y.; Park, J.; Yoon, Y.; Chang, G. A survey of paratuberculosis by immunological methods in dairy and Korean native cattle. Korean J. Vet. Res. 1994, 34, 93–97. [Google Scholar]
- Lee, K.W.; Jung, B.Y. Seroprevalence of Mycobacterium avium subspecies paratuberculosis in cattle in Korea. Vet. Rec. 2009, 165, 661–662. [Google Scholar] [CrossRef] [PubMed]
- Wan-Choul, K.; Hyoung-seok, Y.; Ko, J.A.; Lee, S.L.; Son, W. Prevalence of Jhone’s disease of Korean native cattle in Jeju province. Korean J. Vet. Serv. 2015, 38, 221–225. [Google Scholar]
- VanLeeuwen, J.A.; Keefe, G.P.; Tremblay, R.; Power, C.; Wichtel, J.J. Seroprevalence of infection with Mycobacterium avium subspecies paratuberculosis, bovine leukemia virus, and bovine viral diarrhea virus in maritime Canada dairy cattle. Can. Vet. J. 2001, 42, 193–198. [Google Scholar] [PubMed]
- Ott, S.L.; Wells, S.J.; Wagner, B.A. Herd-level economic losses associated with Johne’s disease on US dairy operations. Prev. Vet. Med. 1999, 40, 179–192. [Google Scholar] [CrossRef]
- Chiodini, R.J. Immunology: Resistance to Paratuberculosis. Vet. Clin. N. Am. Food Anim. Pract. 1996, 12, 313–343. [Google Scholar] [CrossRef]
- Nielsen, S.S.; Toft, N. A review of prevalences of paratuberculosis in farmed animals in Europe. Prev. Vet. Med. 2009, 88, 1–14. [Google Scholar] [CrossRef]
- Wolf, R. Control of Mycobacterium avium subspecies paratuberculosis on Western Canadian dairy farms: Prevalence, diagnostics and risk factors. Ph.D. Thesis, University of Calgary, Calgary, AB, Canada, 8 January 2015. [Google Scholar]
- Clark, D.L.; Koziczkowski, J.J.; Radcliff, R.P.; Carlson, R.A.; Ellingson, J.L.E. Detection of Mycobacterium avium subspecies paratuberculosis: Comparing fecal culture versus serum enzyme-linked immunosorbent assay and direct fecal polymerase chain reaction. J. Dairy Sci. 2008, 91, 2620–2627. [Google Scholar] [CrossRef]
- Nielsen, S.S. Transitions in diagnostic tests used for detection of Mycobacterium avium subsp. paratuberculosis infections in cattle. Vet. Microbiol. 2008, 132, 274–282. [Google Scholar]
- Moksony, F. Small Is Beautiful: The Use and Interpretation of R2 in Social Research. Szociologiai Szemle 1990, 130–138. [Google Scholar]
- Moore, D.S.; Notz, W.I.; Fligner, M.A. The Basic Practice of Statistics, Macmillan Higher Education, 8th ed.; WH Freeman Publisher: New York, NY, USA, 2015. [Google Scholar]
- Zikmud, W.G. Buisiness Research Methods, 5th ed.; Fort Worth Publisher: Dryden, ON, Canada, 1997; pp. 65–71. [Google Scholar]
- Sweeney, R.W.; Whitlock, R.H.; Rosenberger, A.E. Mycobacterium paratuberculosis cultured from milk and supramammary lymph nodes of infected asymptomatic cows. J. Clin. Microbiol. 1992, 30, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Bannantine, J.P.; Bayles, D.O.; Waters, W.R.; Palmer, M.V.; Stabel, J.R.; Paustian, M.L. Early antibody response against Mycobacterium avium subspecies paratuberculosis antigens in subclinical cattle. Proteome Sci. 2008, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Jacob, S.K.; Ramnath, V.; Philomina, P.T.; Raghunandhanan, K.V.; Kannan, A. Assessment of physiological stress in periparturient cows and neonatal calves. Indian J. Physiol. Pharmacol. 2001, 45, 233–238. [Google Scholar]
- Nielsen, S.S.; Enevoldsen, C.; Gröhn, Y.T. The Mycobacterium avium subsp. paratuberculosis ELISA response by parity and stage of lactation. Prev. Vet. Med. 2002, 54, 1–10. [Google Scholar]
- Fernández, B.; Gilardoni, L.R.; Jolly, A.; Colavecchia, S.B.; Paolicchi, F.A.; Mundo, S.L. Detection of bovine IgG isotypes in a PPA-ELISA for johne disease diagnosis in infected herds. Vet. Med. Int. 2012, 1–6. [Google Scholar] [CrossRef]
- Lombard, J.E.; Byrem, T.M.; Wagner, B.A.; McCluskey, B.J. Comparison of milk and serum enzyme–linked immunosorbent assays for diagnosis of Mycobacterium avium subspecies paratuberculosis infection in dairy cattle. J. Vet. Diagnostic Investig. 2006, 18, 448–458. [Google Scholar] [CrossRef]
- Koets, A.P.; Eda, S.; Sreevatsan, S. The within host dynamics of Mycobacterium avium ssp. paratuberculosis infection in cattle: Where time and place matter. Vet. Res. 2015, 46, 1–17. [Google Scholar]
- Eisenberg, S.W.F.; Veldman, E.; Rutten, V.P.M.G.; Koets, A.P. A longitudinal study of factors influencing the result of a Mycobacterium avium ssp. paratuberculosis antibody ELISA in milk of dairy cows. J. Dairy Sci. 2015, 98, 2345–2355. [Google Scholar] [PubMed]
- Gilardoni, L.R.; Fernández, B.; Morsella, C.; Mendez, L.; Jar, A.M.; Paolicchi, F.A.; Mundo, S.L. Mycobacterium paratuberculosis detection in cow’s milk in Argentina by immunomagnetic separation-PCR. Braz. J. Microbiol. 2016, 47, 506–512. [Google Scholar] [CrossRef]
- Nielsen, S.S.; Toft, N. Effect of days in milk and milk yield on testing positive in milk antibody ELISA to Mycobacterium avium subsp. paratuberculosis in dairy cattle. Vet. Immunol. Immunopathol. 2012, 149, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Beaver, A.; Cazer, C.; Ruegg, P.; Gröhn, Y.; Schukken, Y. Implications of PCR and ELISA results on the routes of bulk-tank contamination with Mycobacterium avium ssp. paratuberculosis. J. Dairy Sci. 2016, 99, 1391–1405. [Google Scholar] [CrossRef] [PubMed]
- Laurin, E.L.; Sanchez, J.; Chaffer, M.; McKenna, S.L.; Keefe, G.P. Assessment of the relative sensitivity of milk ELISA for detection of Mycobacterium avium ssp. paratuberculosis infectious dairy cows. J. Dairy Sci. 2017, 100, 598–607. [Google Scholar]
- Sweeney, R.W. Pathogenesis of paratuberculosis. Vet. Clin. Food Anim. Pract. 2011, 27, 537–546. [Google Scholar] [CrossRef]
- Li, L.; Wagner, B.; Freer, H.; Schilling, M.; Bannantine, J.P.; Campo, J.J.; Katani, R.; Grohn, Y.T.; Radzio-Basu, J.; Kapur, V. Early detection of Mycobacterium avium subsp. paratuberculosis infection in cattle with multiplex-bead based immunoassays. PLoS ONE 2017, 12, 1–16. [Google Scholar]
- Bates, A.; O’Brien, R.; Liggett, S.; Griffin, F. The effect of sub-clinical infection with Mycobacterium avium subsp. paratuberculosis on milk production in a New Zealand dairy herd. BMC Vet Res. 2018, 14, 1–11. [Google Scholar]
- Hardin, L.E.; Thorne, J.G. Comparison of milk with serum ELISA for the detection of paratuberculosis in dairy cows. J. Am. Vet. Med. Assoc. 1996, 209, 120–122. [Google Scholar]
- Cazer, C.L.; Mitchell, R.M.; Cicconi-Hogan, K.M.; Gamroth, M.; Richert, R.M.; Ruegg, P.L.; Schukken, Y.H. Associations between Mycobacterium avium subsp. paratuberculosis antibodies in bulk tank milk, season of sampling and protocols for managing infected cows. BMC Vet. Res. 2013, 9, 234. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faruk, M.S.A.; Jung, Y.-h.; Hur, T.-y.; Lee, S.-s.; Cho, Y.-i. Longitudinal Study of Mycobacterium avium Subsp. paratuberculosis Antibody Kinetics in Dairy Cattle Using Sera and Milk throughout the Lactation Period. Vet. Sci. 2020, 7, 81. https://doi.org/10.3390/vetsci7030081
Faruk MSA, Jung Y-h, Hur T-y, Lee S-s, Cho Y-i. Longitudinal Study of Mycobacterium avium Subsp. paratuberculosis Antibody Kinetics in Dairy Cattle Using Sera and Milk throughout the Lactation Period. Veterinary Sciences. 2020; 7(3):81. https://doi.org/10.3390/vetsci7030081
Chicago/Turabian StyleFaruk, Md. Shohel Al, Young-hoon Jung, Tai-young Hur, Sang-suk Lee, and Yong-il Cho. 2020. "Longitudinal Study of Mycobacterium avium Subsp. paratuberculosis Antibody Kinetics in Dairy Cattle Using Sera and Milk throughout the Lactation Period" Veterinary Sciences 7, no. 3: 81. https://doi.org/10.3390/vetsci7030081
APA StyleFaruk, M. S. A., Jung, Y.-h., Hur, T.-y., Lee, S.-s., & Cho, Y.-i. (2020). Longitudinal Study of Mycobacterium avium Subsp. paratuberculosis Antibody Kinetics in Dairy Cattle Using Sera and Milk throughout the Lactation Period. Veterinary Sciences, 7(3), 81. https://doi.org/10.3390/vetsci7030081




