Estimating the Postmortem Interval of Wild Boar Carcasses
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial 1 (Domestic Pig vs. Wild Boar)
2.2. Trial 2 (Piglets in Sun, Shade, Wallow)
2.3. Trial 3 (Adult Wild Boar in Sun vs. Shade)
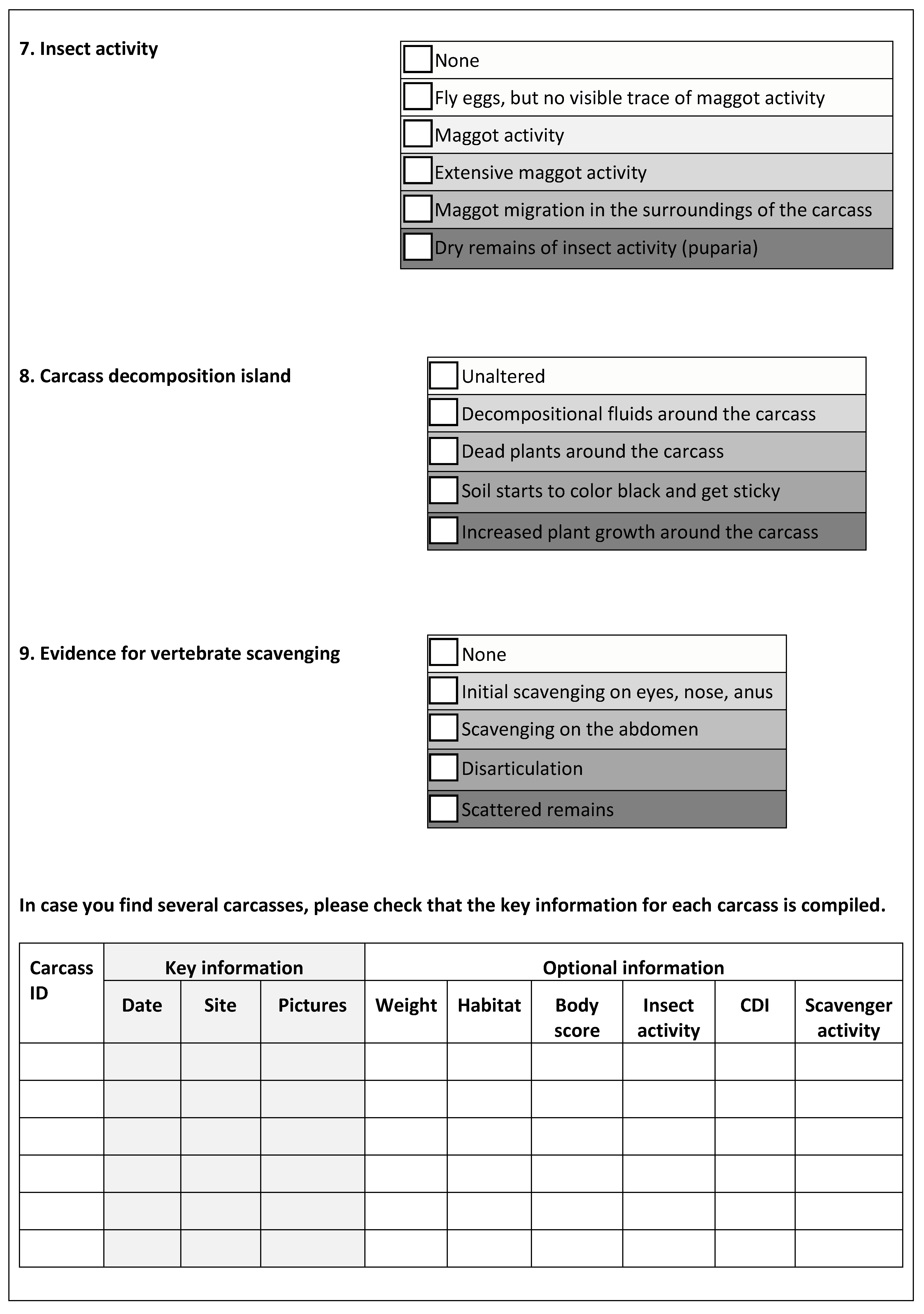
2.4. Scoring Decomposition
3. Results
3.1. Trial 1 (Domestic Pig vs. Wild Boar)
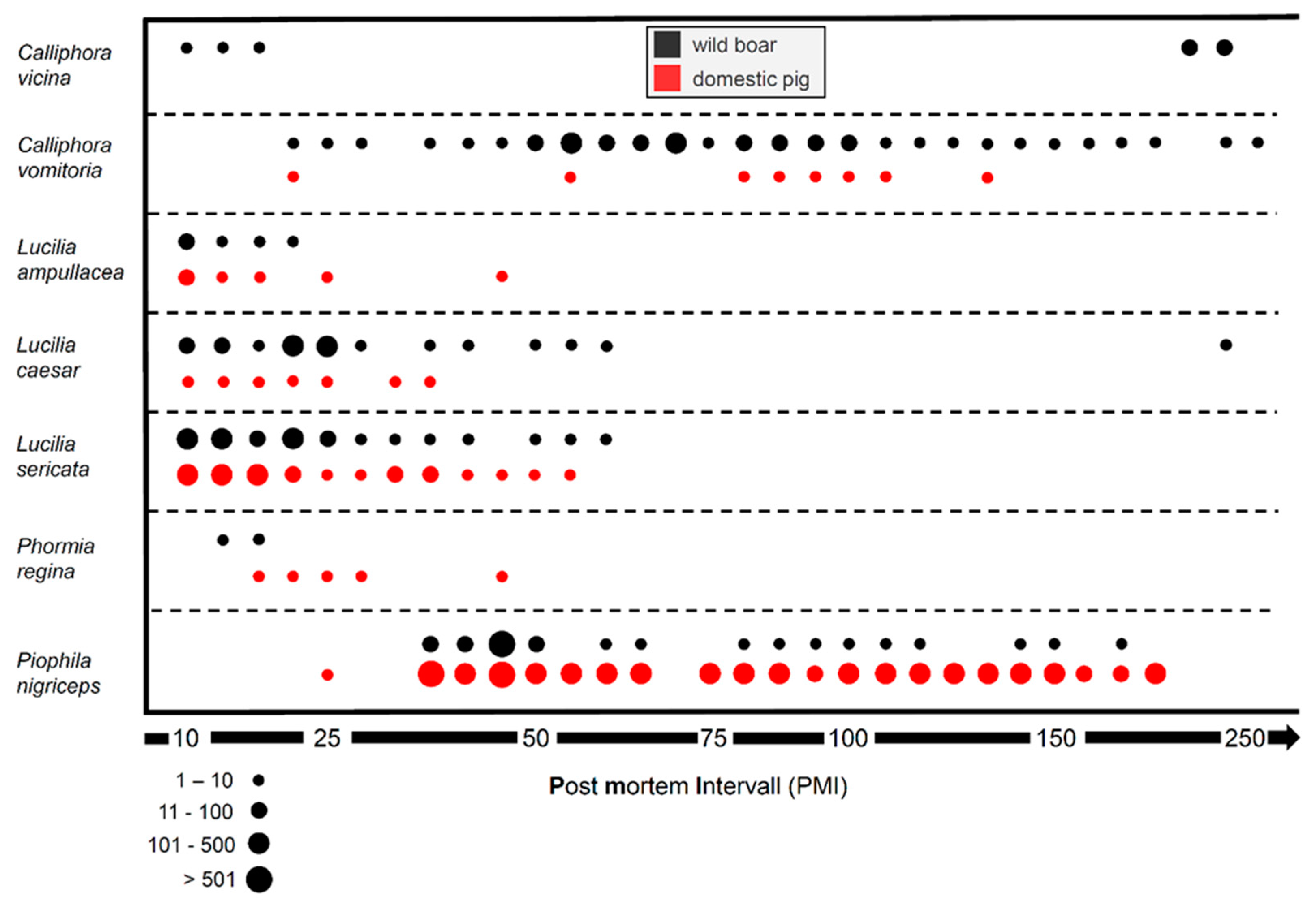
3.1.1. Entomology: Species Richness and Diversity
3.1.2. Entomology: Succession
3.2. Trial 2 (Piglets in Sun, Shade, Water)
3.3. Trial 3 (Adult Wild Boar in Sun vs. Shade)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Matuszewski, S.; Hall, M.J.R.; Moreau, G.; Schoenly, K.G.; Tarone, A.M.; Villet, M.H. Pigs vs. people: The use of pigs as analogues for humans in forensic entomology and taphonomy research. Int. J. Legal Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Probst, C.; Globig, A.; Knoll, B.; Conraths, F.J.; Depner, K. Behaviour of free ranging wild boar towards their dead fellows: Potential implications for the transmission of African swine fever. R. Soc. Open Sci. 2017, 4, 170054. [Google Scholar] [CrossRef] [PubMed]
- Probst, C.; Gethmann, J.; Amler, S.; Globig, A.; Knoll, B.; Conraths, F.J. Welche Rolle spielen Wildschweinkadaver in der Epidemiologie der ASP? In Proceedings of the Tagung der DVG-Fachgruppe Tierseuchen, Bundesinstitut für Risikobewertung, Berlin, Germany, 28 May 2019. [Google Scholar]
- Brooks, J.W. Postmortem Changes in Animal Carcasses and Estimation of the Postmortem Interval. Vet. Pathol. 2016, 53, 929–940. [Google Scholar] [CrossRef] [PubMed]
- Vass, A.A.; Bass, W.M.; Wolt, J.D.; Foss, J.E.; Ammons, J.T. Time since death determinations of human cadavers using soil solution. J. Forensic Sci. 1992, 37, 1236–1253. [Google Scholar] [CrossRef] [PubMed]
- Amendt, J.; Richards, C.S.; Campobasso, C.P.; Zehner, R.; Hall, M.J. Forensic entomology: Applications and limitations. Forensic Sci. Med. Pathol. 2011, 7, 379–392. [Google Scholar] [CrossRef]
- Von Hoermann, C.; Jauch, D.; Kubotsch, C.; Reichel-Jung, K.; Steiger, S.; Ayasse, M. Effects of abiotic environmental factors and land use on the diversity of carrion-visiting silphid beetles (Coleoptera: Silphidae): A large scale carrion study. PLoS ONE 2018, 13, e0196839. [Google Scholar] [CrossRef]
- Anton, E.; Niederegger, S.; Beutel, R.G. Beetles and flies collected on pig carrion in an experimental setting in Thuringia and their forensic implications. Med. Vet. Entomol. 2011, 25, 353–364. [Google Scholar] [CrossRef]
- Grassberger, M.; Frank, C. Initial study of arthropod succession on pig carrion in a central European urban habitat. J. Med. Entomol. 2004, 41, 511–523. [Google Scholar] [CrossRef]
- Matuszewski, S.; Bajerlein, D.; Konwerski, S.; Szpila, K. An initial study of insect succession and carrion decomposition in various forest habitats of Central Europe. Forensic Sci. Int. 2008, 180, 61–69. [Google Scholar] [CrossRef]
- Matuszewski, S.; Bajerlein, D.; Konwerski, S.; Szpila, K. Insect succession and carrion decomposition in selected forests of Central Europe. Part 1: Pattern and rate of decomposition. Forensic Sci. Int. 2010, 194, 85–93. [Google Scholar] [CrossRef]
- Carter, D.O. Forensic taphonomy: Processes Associated with Cadaver Decomposition in Soil. Ph.D. Thesis, James Cook University, Townsville, Australia, 2005. Available online: http://eprints.jcu.edu.au/1292 (accessed on 1 October 2019).
- Carter, D.O.; Yellowlees, D.; Tibbett, M. Cadaver decomposition in terrestrial ecosystems. Naturwissenschaften 2007, 94, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Benninger, L.A.; Carter, D.O.; Forbes, S.L. The biochemical alteration of soil beneath a decomposing carcass. Forensic Sci. Int. 2008, 180, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, D.W.; Wiltshire, P.E.J.; Turner, B.D. Microbial characteristics of soils from graves: An investigation at the interface of soil microbiology and forensic science. Appl. Soil. Ecol. 2000, 14, 283–288. [Google Scholar] [CrossRef]
- Pechal, J.L.; Crippen, T.L.; Benbow, M.E.; Tarone, A.M.; Dowd, S.; Tomberlin, J.K. The potential use of bacterial community succession in forensics as described by high throughput metagenomic sequencing. Int. J. Legal Med. 2014, 128, 193–205. [Google Scholar] [CrossRef]
- Larizza, M. Physical and Chemical Analysis of Pig Carcass Decomposition in a Fine Sand. Master’s Thesis, University of Ontario Institute of Technology, Oshawa, ON, Canada, 2010. [Google Scholar]
- Szelecz, I.; Koenig, I.; Seppey, C.V.W.; Le Bayon, R.C.; Mitchell, E.A.D. Soil chemistry changes beneath decomposing cadavers over a one-year period. Forensic Sci. Int. 2018, 286, 155–165. [Google Scholar] [CrossRef]
- Henssge, C.; Madea, B. Estimation of the time since death. Forensic Sci. Int. 2007, 165, 182–184. [Google Scholar] [CrossRef]
- Payne, J.A. A Summer Carrion Study of the Baby Pig Sus Scrofa Linnaeus. Ecology 1965, 46, 592–602. [Google Scholar] [CrossRef]
- Moffatt, C.; Simmons, T.; Lynch-Aird, J. An improved equation for TBS and ADD: Establishing a reliable postmortem Interval framework for casework and experimental studies. J. Forensic Sci. 2016, 61 (Suppl. 1), S201–S207. [Google Scholar] [CrossRef]
- Gelderman, H.T.; Boer, L.; Naujocks, T.; ACM, I.J.; Duijst, W. The development of a post-mortem interval estimation for human remains found on land in the Netherlands. Int. J. Legal Med. 2018, 132, 863–873. [Google Scholar] [CrossRef]
- Amendt, J.; Campobasso, C.P.; Gaudry, E.; Reiter, C.; LeBlanc, H.N.; Hall, M.J. Best practice in forensic entomology—standards and guidelines. Int. J. Legal Med. 2007, 121, 90–104. [Google Scholar] [CrossRef]
- Moreau, G.; Michaud, J.P.; Schoenly, K. Experimental Design, Inferential Statistics and Computer Modeling. Comput. Sci. 2015, 205–230. [Google Scholar] [CrossRef]
- Galloway, A.; Birkby, W.H.; Jones, A.M.; Henry, T.E.; Parks, B.O. Decay rates of human remains in an arid environment. J. Forensic Sci. 1989, 34, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Megyesi, M.S.; Nawrocki, S.P.; Haskell, N.H. Using accumulated degree-days to estimate the postmortem interval from decomposed human remains. J. Forensic Sci. 2005, 50, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Keough, N.; Myburgh, J.; Steyn, M. Scoring of Decomposition: A Proposed Amendment to the Method When Using a Pig Model for Human Studies. J. Forensic Sci. 2017, 62, 986–993. [Google Scholar] [CrossRef]
- Matuszewski, S.; Konwerski, S.; Frątczak, K.; Szafałowicz, M. Effect of body mass and clothing on decomposition of pig carcasses. Int. J. Legal Med. 2014, 128, 1039–1048. [Google Scholar] [CrossRef]
- Sutherland, A.; Myburgh, J.; Steyn, M.; Becker, P.J. The effect of body size on the rate of decomposition in a temperate region of South Africa. Forensic Sci. Int. 2013, 231, 257–262. [Google Scholar] [CrossRef]
- Campobasso, C.P.; Di Vella, G.; Introna, F. Factors affecting decomposition and Diptera colonization. Forensic Sci. Int. 2001, 120, 18–27. [Google Scholar] [CrossRef]
- Roberts, L.G.; Dabbs, G.R. A Taphonomic Study Exploring the Differences in Decomposition Rate and Manner between Frozen and Never Frozen Domestic Pigs (Sus scrofa). J. Forensic Sci. 2015, 60, 588–594. [Google Scholar] [CrossRef]
- Villet, M.; Richards, C.; Midgley, J. Contemporary Precision, Bias and Accuracy of Minimum Post-Mortem Intervals Estimated Using Development of Carrion-Feeding Insects. In Current Concepts in Forensic Entomology; Amendt, J., Campobasso, C., Goff, M., Grassberger, M., Eds.; Springer: Berlin, Germany, 2010; pp. 109–137. [Google Scholar] [CrossRef]
- Willey, P.; Snyder, L.M. Canid modification of human remains: Implications for time-since-death estimations. J. Forensic Sci. 1989, 34, 894–901. [Google Scholar] [CrossRef]
- Lutz, L.; Verhoff, M.A.; Amendt, J. Environmental factors influencing flight activity of forensically important female blow flies in Central Europe. Int. J. Legal Med. 2019, 133, 1267–1278. [Google Scholar] [CrossRef]
- Carter, D.O.; Yellowlees, D.; Tibbett, M. Moisture can be the dominant environmental parameter governing cadaver decomposition in soil. Forensic Sci. Int. 2010, 200, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Tumer, A.R.; Karacaoglu, E.; Namli, A.; Keten, A.; Farasat, S.; Akcan, R.; Sert, O.; Odabaşi, A.B. Effects of different types of soil on decomposition: An experimental study. Legal Med. 2013, 15, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Archer, M.S. Annual variation in arrival and departure times of carrion insects at carcasses: Implications for succession studies in forensic entomology. Aust. J. Zool. 2003, 51, 569–576. [Google Scholar] [CrossRef]
- Turner, A.K. The Use of Time and Energy by Aerial Feeding Birds. Ph.D. Thesis, Faculty of Natural Sciences Legacy Departments, University of Stirling, Stirling, Scotland, 1981. [Google Scholar]
- Møller, A.P. Long-term trends in wind speed, insect abundance and ecology of an insectivorous bird. Ecosphere 2013, 4, 1–11. [Google Scholar] [CrossRef]
- Heinzel, H.-G.; Böhm, H. The wind-orientation of walking carrion beetles. J. Comp. Physiol. 1989, 164, 775–786. [Google Scholar] [CrossRef]
- Hallmann, C.A.; Sorg, M.; Jongejans, E.; Siepel, H.; Hofland, N.; Schwan, H.; Stenmans, W.; Müller, A.; Sumser, H.; Hörren, T.; et al. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 2017, 12, e0185809. [Google Scholar] [CrossRef]
- Digby, P.S.B. Flight activity in the Blowfly, Calliphora Erythrocephala, in relation to wind speed, with special reference to adaptation. J. Exp. Biol. 1958, 35, 776–795. [Google Scholar]
- Żmijewski, T.; Korzeniowski, W. Tissue composition of wild boars carcasses. Electron. J. Pol. Agric. Univ. 2001, 4, 1–7. [Google Scholar]
- Marsico, G.; Rasulo, A.; Dimatteo, S.; Tarricone, S.; Pinto, F.; Ragni, M. Pig, F1 (wild boar x pig) and wild boar meat quality. In Proceedings of the Scientific Association of Animal Production (ASPA), Alghero, Italy, 29 May–1 June 2007; pp. 701–703. [Google Scholar]
- Zhang, S.H.; Shen, L.Y.; Luo, J.; Wu, Z.H.; Jiang, Y.Z.; Tang, G.Q.; Li, M.Z.; Bai, L.; Li, X.W.; Zhu, L. Analysis of carcass and meat quality traits and nutritional values of hybrid wild boars under different crossing systems. Genet. Mol. Res. 2015, 14, 2608–2616. [Google Scholar] [CrossRef]
- Sales, J.; Kotrba, R. Meat from wild boar (Sus scrofa L.): A review. Meat Sci. 2013, 94, 187–201. [Google Scholar] [CrossRef]
- Ubelaker, D.; Zarenko, K. Adipocere: What Is Known after over Two Centuries of Research. Forensic Sci. Int. 2011, 208, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Morelle, K.; Jezek, M.; Licoppe, A.; Podgorski, T. Deathbed choice by ASF-infected wild boar can help find carcasses. Transbound. Emerg. Dis. 2019, 66, 1821–1826. [Google Scholar] [CrossRef] [PubMed]
- Van Daalen, M.A.; de Kat, D.S.; Oude Grotebevelsborg, B.F.; De Leeuwe, R.; Warnaar, J.; Oostra, R.J.; MDuijst-Heesters, W.L. An Aquatic Decomposition Scoring Method to Potentially Predict the Postmortem Submersion Interval of Bodies Recovered from the North Sea. J. Forensic Sci. 2017, 62, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Ayers, L. Differential Decomposition in Terrestrial, Freshwater, and Saltwater Environments: A Pilot Study; Texas State University: San Marcos, TX, USA, 2010. [Google Scholar]
- Rattenbury, A.E. Chapter 2—Forensic Taphonomy. In Forensic Ecogenomics; Ralebitso Senior, T.K., Ed.; Academic Press: London, UK, 2018; pp. 37–59. [Google Scholar] [CrossRef]
- Simmons, T.; Cross, P.A.; Adlam, R.E.; Moffatt, C. The Influence of Insects on Decomposition Rate in Buried and Surface Remains. J. Forensic Sci. 2010, 55, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Gaudry, E. The Insects Colonisation of Buried Remains. In Current Concepts in Forensic Entomology; Amendt, J., Campobasso, C., Goff, M., Grassberger, M., Eds.; Springer: Berlin, Germany, 2010; pp. 273–311. [Google Scholar] [CrossRef]
- VanLaerhoven, S.L.; Anderson, G.S. Insect succession on buried carrion in two biogeoclimatic zones of British Columbia. J. Forensic Sci. 1999, 44, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Marais-Werner, A.; Myburgh, J.; Becker, P.J.; Steyn, M. A comparison between decomposition rates of buried and surface remains in a temperate region of South Africa. Int. J. Legal Med. 2017, 132, 301–309. [Google Scholar] [CrossRef]
- Merck, M.; Miller, D.M. Postmortem Changes and the Postmortem Interval. In Veterinary Forensics: Animal Cruelty Investigations; Merck, M.D., Ed.; Wiley Online Library: Hoboken, NJ, USA, 2012; pp. 255–271. [Google Scholar] [CrossRef]
- Nawrocka, M.; Fratczak, K.; Matuszewski, S. Inter-Rater Reliability of Total Body Score-A Scale for Quantification of Corpse Decomposition. J. Forensic Sci. 2016, 61, 798–802. [Google Scholar] [CrossRef]
- Lashley, M.A.; Jordan, H.R.; Tomberlin, J.K.; Barton, B.T. Indirect effects of larval dispersal following mass mortality events. Ecology 2018, 99, 491–493. [Google Scholar] [CrossRef]
- Probst, C.; Gethmann, J.; Amler, S.; Globig, A.; Knoll, B.; Conraths, F.J. The potential role of scavengers in spreading African swine fever among wild boar. Sci. Rep. 2019, 9, 11450. [Google Scholar] [CrossRef]
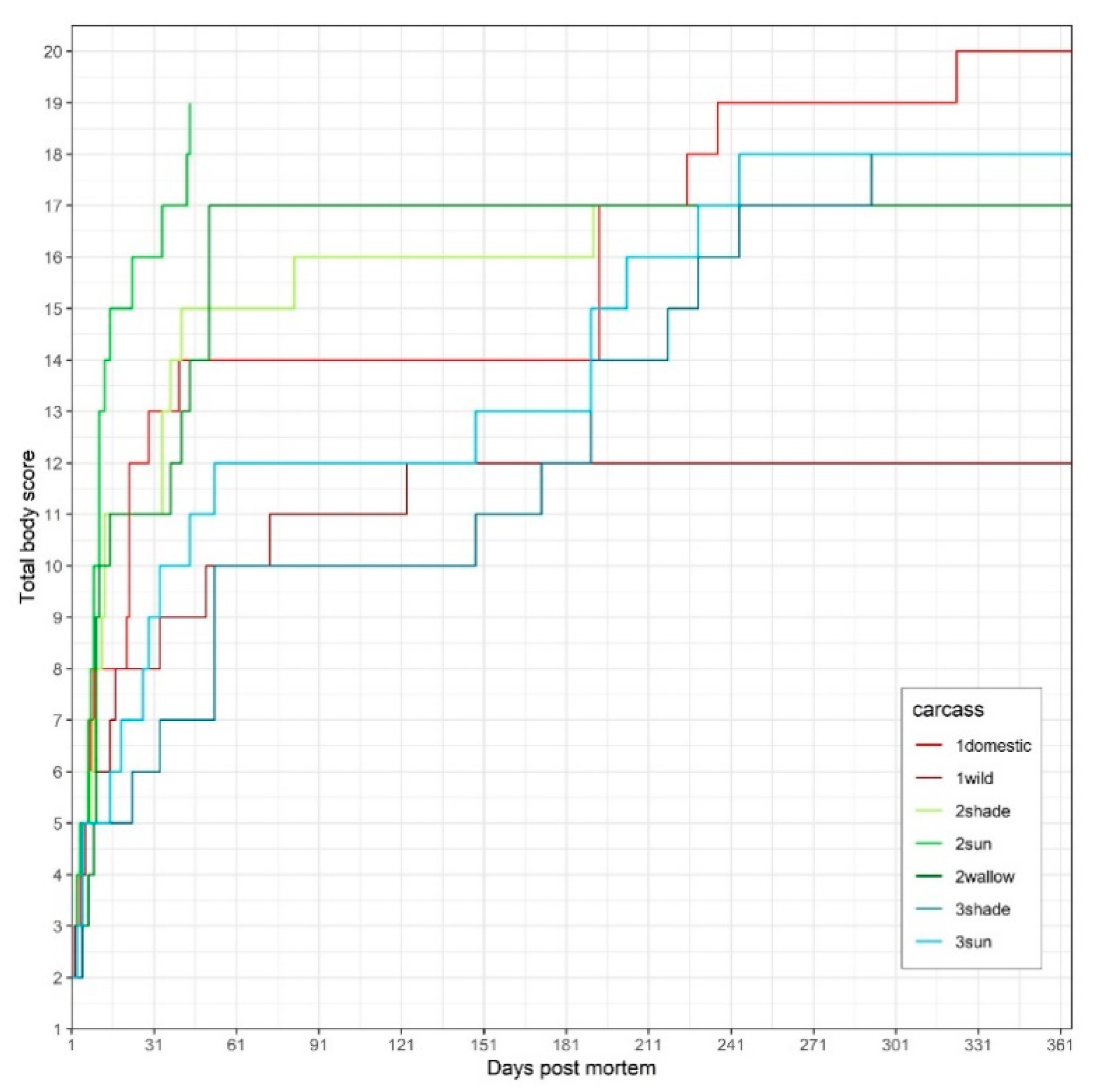










| Carcass | Stage | Days Post-Mortem | Points Total | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fresh | Early | Advanced | Skeletonization | ||||||||||
| Points | 1 | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 1 | 2 | 3 | ||
| 1domestic | PBSH | 2 | 8 | 22 | 21 | 193 | 5 | ||||||
| PBST | 1 | 4 | 5 | 8 | 9 | 22 | 29 | 40 | 193 | 225 | 323 | 11 | |
| PBSL | 1 | 22 | 193 | 236 | 4 | ||||||||
| 1wild | PBSH | 2 | 8 | 15 | 433 | 708 | 5 | ||||||
| PBST | 1 | 5 | 6 | 17 | 33 | 50 | 123 | 637 | 8 | ||||
| PBSL | 1 | 73 | 433 | 708 | 4 | ||||||||
| 2sun | PBSH | 3 | 7 | 9 | 11 | 34 | 5 | ||||||
| PBST | 1 | 3 | 4 | 7 | 8 | 9 | 11 | 15 | 23 | 44 | 10 | ||
| PBSL | 1 | 11 | 13 | 43 | 4 | ||||||||
| 2shade | PBSH | 3 | 9 | 13 | 34 | 413 | 5 | ||||||
| PBST | 1 | 3 | 5 | 8 | 10 | 12 | 34 | 37 | 41 | 191 | 10 | ||
| PBSL | 1 | 13 | 82 | 413 | 4 | ||||||||
| 2wallow | PBSH | 7 | 3 | 11 | 37 | 41 | 5 | ||||||
| PBST | 1 | 9 | n.o. | 11 | 13 | 10 | n.o. | 51 | 8 | ||||
| PBSL | 1 | 15 | 44 | 51 | 4 | ||||||||
| 3sun | PBSH | 5 | 27 | 33 | 53 | 190 | 5 | ||||||
| PBST | 1 | 5 | 3 | 15 | 19 | 44 | 190 | 203 | 244 | 9 | |||
| PBSL | 1 | 29 | 148 | 229 | 4 | ||||||||
| 3shade | PBSH | 5 | 33 | 53 | 190 | 244 | 5 | ||||||
| PBST | 1 | 5 | 5 | 23 | 190 | 53 | 190 | 218 | 292 | 9 | |||
| PBSL | 1 | 173 | 148 | 229 | 4 | ||||||||
| Order | Family | Species | 1wild | 1domestic |
|---|---|---|---|---|
| Diptera | Calliphoridae | Calliphora vicina | x | |
| Calliphora vomitoria | x | x | ||
| Lucilia ampullacea | x | x | ||
| Lucilia caesar | x | x | ||
| Lucilia sericata | x | x | ||
| Phormia regina | x | x | ||
| Muscidae | Hydrotaea ignava | x | x | |
| Muscina prolapse | x | x | ||
| Ophyra capensis | x | |||
| Piophilidae | Piophila nigriceps | x | x | |
| Coleoptera | Cleridae | Necrobia ruficollis | x | |
| Necrobia rufipes | x | |||
| Dermestidae | Dermestidae sp. | x | ||
| Histeridae | Histeridae sp. | x | ||
| Nitidulidae | Omosita colon | x | ||
| Staphylinidae | Aleochara sp. | x | ||
| Creophilus maxillosus | x | x | ||
| Omalium sp. | x | |||
| Philonthus sp. | x | x | ||
| Silphidae | Necrodes littoralis | x | x |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Probst, C.; Gethmann, J.; Amendt, J.; Lutz, L.; Teifke, J.P.; Conraths, F.J. Estimating the Postmortem Interval of Wild Boar Carcasses. Vet. Sci. 2020, 7, 6. https://doi.org/10.3390/vetsci7010006
Probst C, Gethmann J, Amendt J, Lutz L, Teifke JP, Conraths FJ. Estimating the Postmortem Interval of Wild Boar Carcasses. Veterinary Sciences. 2020; 7(1):6. https://doi.org/10.3390/vetsci7010006
Chicago/Turabian StyleProbst, Carolina, Jörn Gethmann, Jens Amendt, Lena Lutz, Jens Peter Teifke, and Franz J. Conraths. 2020. "Estimating the Postmortem Interval of Wild Boar Carcasses" Veterinary Sciences 7, no. 1: 6. https://doi.org/10.3390/vetsci7010006
APA StyleProbst, C., Gethmann, J., Amendt, J., Lutz, L., Teifke, J. P., & Conraths, F. J. (2020). Estimating the Postmortem Interval of Wild Boar Carcasses. Veterinary Sciences, 7(1), 6. https://doi.org/10.3390/vetsci7010006






