A Serosurvey of Multiple Pathogens in American Black Bears (Ursus americanus) in Pennsylvania, USA Indicates a Lack of Association with Sarcoptic Mange
Abstract
1. Introduction
2. Materials and Methods
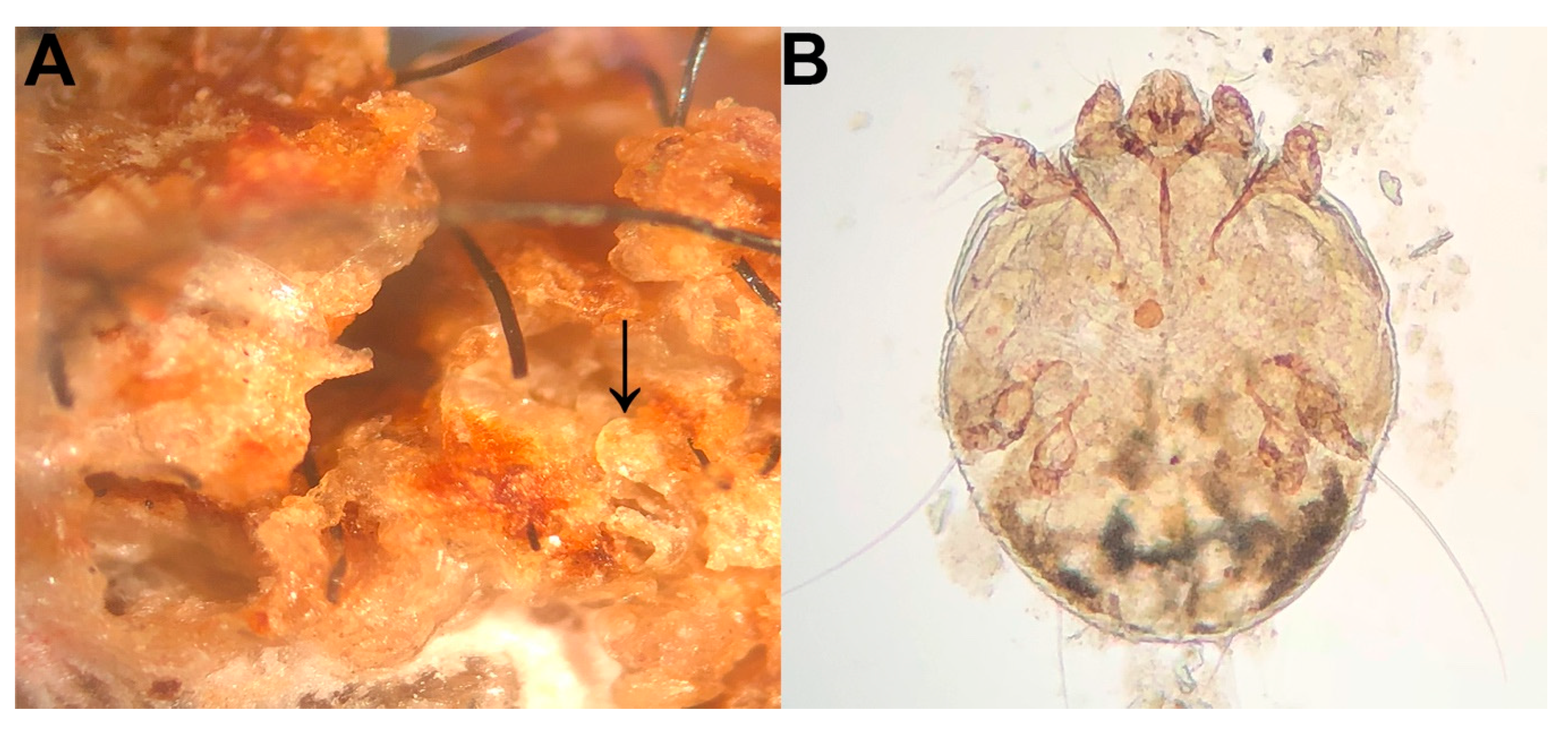
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fisher, M.C.; Henk, D.A.; Briggs, C.J.; Brownstein, J.S.; Madoff, L.C.; McCraw, S.L.; Gurr, S.J. Emerging fungal threats to animal, plant and ecosystem health. Nature 2012, 484, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.S.; Farnsworth, M.L.; Malmberg, J.L. Diseases at the livestock-wildlife interface: Status, challenges, and opportunities in the United States. Prev. Vet. Med. 2013, 110, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Gortázar, C.; Diez-Delgado, I.; Barasona, J.A.; Vicente, J.; De La Fuente, J.; Boadella, M. The wild side of disease control at the wildlife-livestock-human interface: A review. Front. Vet. Sci. 2014, 1, 27. [Google Scholar] [CrossRef] [PubMed]
- Viana, M.; Cleaveland, S.; Matthiopoulos, J.; Halliday, J.; Packer, C.; Craft, M.E.; Hampson, K.; Czupryna, A.; Dobson, A.P.; Dubovi, E.J. Dynamics of a morbillivirus at the domestic-wildlife interface: Canine distemper virus in domestic dogs and lions. Proc. Natl. Acad. Sci. USA 2015, 112, 1464–1469. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeybard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef] [PubMed]
- Hristienko, H.; MacDonald, J.E., Jr. Going into the 21st century: A perspective on trends and controversies in the management of the American black bear. Ursus. 2007, 18, 72–88. [Google Scholar] [CrossRef]
- Hassell, J.M.; Begon, M.; Ward, M.J.; Fevre, E.M. Urbanization and disease emergence: Dynamics at the wildlife-livestock-human interface. Trends Ecol. Evol. 2017, 32, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Arlian, L.G.; Morgan, M.S. A review of Sarcoptes scabiei: Past, present, and future. Parasit Vectors 2017, 297. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.J.; Huffam, S.E.; Walton, S.F.; Currie, B.J. Crusted scabies: Clinical and immunological findings in seventy-eight patients and a review of the literature. J. Infect 2005, 50, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Pence, D.B.; Ueckermann, E. Sarcoptic mange in wildlife. Rev. Sci. Tech. OIE 2002, 21, 385–398. [Google Scholar] [CrossRef]
- Riley, S.P.D.; Bromley, C.; Poppenga, R.H.; Uzal, F.A.; Whited, L.; Sauvajot, R.M. Anticoagulant exposure and notoedric mange in bobcats and mountain lions in urban southern California. J. Wildl. Manage 2007, 71, 1874–1884. [Google Scholar] [CrossRef]
- Camkerten, I.; Sahin, T.; Borazan, G.; Gokcen, A.; Erel, O.; Das, A. Evaluation of blood oxidant/antioxidant balance in dogs with sarcoptic mange. Vet. Parasitol. 2009, 161, 106–109. [Google Scholar] [CrossRef] [PubMed]
- Oleaga, A.; Vicente, J.; Ferroglio, E.; Pegoraro de Macedo, M.R.; Casais, R.; del Cerro, A.; Espi, A.; Garcia, E.J.; Gortazar, C. Concomitance and interactions of pathogens in the Iberian wolf (Canis lupus). Res. Vet. Sci. 2015, 101, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Singla, L.D.; Singla, N.; Parshad, V.R. Development of concurrent infection of notoedric mange in rabbits infected with Trypanosoma evansi. Scan J. Lab. Anim. Sci. 2015, 41, 1–6. [Google Scholar]
- Zhao, J.; Shi, N.; Sun, Y.; Martella, V.; Nikolin, V.; Zhu, C.; Zhang, H.; Hu, B.; Bai, X.; Yan, X. Pathogenesis of canine distemper virus in experimentally infected raccoon dogs, foxes, and minks. Antivir. Res. 2015, 122, 1–11. [Google Scholar] [CrossRef]
- Walton, S.F.; Pizzutto, S.; Slender, A.; Viberg, L.; Holt, D.; Hales, B.J.; Kemp, D.J.; Currie, B.J.; Rolland, J.M.; O’Hehir, R. Increased allergic immune response to Sarcoptes scabiei antigens in crusted versus orginary scabies. Clin. Vaccine Immunol. 2010, 17, 1428–1438. [Google Scholar] [CrossRef]
- Niedringhaus, K.D.; Brown, J.D.; Ternent, M.; Childress, W.; Gettings, J.R.; Yabsley, M.J. The emergence and expansion of sarcoptic mange in American black bears (Ursus americanus) in the United States. Vet. Parasitol. Reg. Stud. Rep. 2019, 100303. [Google Scholar] [CrossRef]
- Peltier, S.K.; Brown, J.D.; Ternent, M.; Niedringhaus, K.D.; Schuler, K.; Bunting, E.M.; Kirchgessner, M.; Yabsley, M.J. Genetic characterization of Sarcoptes scabiei from black bears (Ursus americanus) and other hosts in the eastern United States. J. Parasitol. 2017, 103, 593–597. [Google Scholar] [CrossRef]
- Peltier, S.K.; Brown, J.D.; Ternent, M.; Fenton, H.; Niedringhaus, K.D.; Yabsley, M.J. Assays for the detection and identification of the causative agent of mange in free-ranging black bears (Ursus americanus). J. Wildl. Dis. 2018, 54, 471–479. [Google Scholar] [CrossRef]
- Cook, W.J.; Pelton, M.R. Selected infectious and parasitic diseases of black bears in the Great Smoky Mountains National Park. In Proceedings of the Fourth Eastern Workshop on Black Bear Management and Research, Greenville, ME, USA, 3–6 April 1978; Hugie, R.D., Ed.; pp. 120–124. [Google Scholar]
- Crum, J.M.; Nettles, V.F.; Davidson, W.R. Studies on the endoparasites of the black bear (Ursus americanus) in the southeastern United States. J. Wildl. Dis. 1978, 14, 178–186. [Google Scholar] [CrossRef]
- Dies, K.H. Helminths recovered from black bears in the Peace River region of northwestern Alberta. J. Wildl. Dis. 1979, 15, 49–50. [Google Scholar] [CrossRef] [PubMed]
- Clover, J.R.; Hofstra, T.D.; Kuluris, B.G.; Schroeder, M.A.T.; Nelson, B.C.; Barnes, A.M.; Botzler, R.G. Serologic evidence of Yersinia pestis infection in small mammals and bears from a temperate rainforest of north coastal California. J. Wildl. Dis. 1989, 25, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Farajollahi, A.; Panella, N.A.; Carr, P.; Crans, W.; Burguess, K.; Komar, N. Serologic evidence of West Nile virus infection in black bears (Ursus americanus) from New Jersey. J. Wildl. Dis. 2003, 39, 894–896. [Google Scholar] [CrossRef] [PubMed]
- Yabsley, M.J.; Nims, T.N.; Savage, M.J.Y.; Durden, L.A. Ticks and tick-borne pathogens and putative symbionts of black bears (Ursus americanus floridanus) from Georgia and Florida. J. Parasitol. 2009, 95, 1125–1128. [Google Scholar] [CrossRef] [PubMed]
- Bourne, D.C.; Cracknell, J.M.; Bacon, H.J. Veterinary issues related to bears (Ursidae). Int. Zoo Yearb. 2010, 44, 16–32. [Google Scholar] [CrossRef]
- Leydet, B.F., Jr.; Liang, F.T. Detection of human bacterial pathogens in ticks collected from Louisiana black bears (Ursus americanus luteolus). Ticks Tick Borne. Dis. 2013, 4, 191–196. [Google Scholar] [PubMed]
- Stephenson, N.; Higley, J.M.; Sajecki, J.L.; Chomel, B.B.; Brown, R.N.; Foley, J.E. Demographic characteristics and infectious diseases of a population of American black bears in Humboldt County, California. Vector. Borne. Zoonotic Dis. 2015, 15, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Westmoreland, L.S.; Stoskopf, M.K.; Maggi, R.G. Prevalence of Anaplasma phagocytophilum in North Carolina Eastern black bears (Ursus americanus). J. Wildl. Dis. 2016, 52, 968–970. [Google Scholar] [CrossRef] [PubMed]
- Keel, M.K.; Terio, K.A.; McAloose, D. Canidae: Ursidae and Ailuridae. In Pathology of Wildlife and Zoo, Animals; Terio, K.A., McAloose, D., St. Leger, J., Eds.; Elsevier: San Diego, CA, USA, 2018; pp. 229–261. [Google Scholar]
- Cottrell, W.O.; Keel, M.K.; Brooks, J.W.; Mead, D.G.; Phillips, J.E. First report of clinical disease associated with canine distemper virus infection in a wild black bear (Ursus americana). J. Wildl. Dis. 2013, 49, 1024–1027. [Google Scholar] [CrossRef]
- Huffman, J.E.; Roscoe, D.E. Case report: Fatal disseminated toxoplasmosis in a black bear cub. J. Penn. Acad. Sci. 2014, 88, 101–106. [Google Scholar]
- Knowles, S.; Bodenstein, B.L.; Hamon, T.; Saxton, M.W.; Hall, J.S. Infectious canine hepatitis in a brown bear (Ursus arctos horribilis) from Alaska, USA. J. Wildl. Dis. 2018, 54, 642–645. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.S. Canine distemper. In Infectious Diseases of Wild Animals; Williams, E.S., Barker, I.K., Eds.; Blackwell Publishing: Ames, IA, USA, 2001; pp. 50–59. [Google Scholar]
- Appel, M.; Bister, S.I.; Menegus, M.; Albert, D.A.; Carmichael, L.E. Pathogenicity of low-virulence strains of two canine adenovirus types. Am. J. Vet. Res. 1973, 34, 543–550. [Google Scholar] [PubMed]
- Carmichael, L.E.; Joubert, J.C.; Pollock, R.V. Hemagglutination by canine parvovirus: Serologic studies and diagnostic applications. Am. J. Vet. Res. 1980, 41, 784–791. [Google Scholar] [PubMed]
- Appel, M.; Robson, D.S. A microneutralization test for canine distemper virus. Am. J. Vet. Res. 1973, 34, 1459–1463. [Google Scholar] [PubMed]
- Dubey, J.P.; Desmonts, G. Serological responses of equids fed Toxoplasma gondii oocysts. Equine Vet. J. 1987, 19, 337–339. [Google Scholar] [CrossRef] [PubMed]
- Dubey, J.P.; Brown, J.; Ternent, M.; Verma, S.K.; Hill, D.E.; Cerqueira-Cezar, C.K.; Kwok, O.C.H.; Calero-Bernal, R.; Humpreys, J.G. Seroepidemiologic study on the prevalence of Toxoplasma gondii and Trichinella spp. infections in black bears (Ursus americanus) in Pennsylvania, USA. Vet. Parasitol. 2016, 229, 76–80. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria. Available online: https://www.R-project.org/ (accessed on 20 September 2018).
- Chomel, B.B.; Kasten, R.W.; Chappuis, G.; Soulier, M.; Kirkuchi, Y. Serological survey of selected canine viral pathogens and zoonoses in grizzly bears (Ursus arctos horribilis) and black bears (Ursus americanus) from Alaska. Rev. Sci. Tech. Int. Epiz. 1998, 17, 756–766. [Google Scholar] [CrossRef]
- Mortenson, J.A. Serologic survey of infectious disease agents in black bears (Ursus americanus) of California, Oregon, and Washington. Master Thesis, Oregon State University, Corvalis, OR, USA, 18 November 1998. [Google Scholar]
- Dunbar, M.R.; Cunningham, M.W.; Roof, J.C. Seroprevalence of select disease agents from free-ranging black bears in Florida. J. Wildl. Dis. 1998, 34, 612–619. [Google Scholar] [CrossRef][Green Version]
- Philippa, J.D.; Leighton, F.A.; Daoust, P.Y.; Nielsen, O.; Pagliarulo, M.; Schwantje, H.; Shury, T.; Van Herwijnen, R.; Martina, B.E.; Kuiken, T.; et al. Antibodies to selected pathogens in free-ranging terrestrial carnivores and marine mammals in Canada. Vet. Record 2004, 155, 135–140. [Google Scholar] [CrossRef]
- Bronson, E.; Spiker, H.; Driscoll, C.P. Serosurvey for selected pathogens in free-ranging American black bears (Ursus americanus) in Maryland, USA. J. Wildl. Dis. 2014, 50, 829–836. [Google Scholar] [CrossRef]
- Johnson, D.; Larter, N.C.; Elkin, B.; Allaire, D.G. An Opportunistic Parasitological and Serological Examination of Nuisance Black Bears in the Dehcho Region of the Northwest Territories; Department of Environment and Natural Resources, Government of the Northwest Territories: Northwest Territories, Canada, 2013; pp. 1–70.
- Foreyt, W.J.; Evermann, J.F.; Hickman, J. Serologic survey for adenovirus infection in wild bears in Washington. J. Wildl. Manage. 1986, 50, 273–274. [Google Scholar] [CrossRef]
- Ruppanner, R.; Jessup, D.A.; Ohishi, I.; Behymer, D.E.; Franti, C.E. Serologic survey for certain zoonotic diseases in black bears in California. J. Am. Vet. Med. Assoc. 1982, 181, 1288–1291. [Google Scholar] [PubMed]
- Kinyon, H.R. Hematology, Serologic prevalence of Toxoplasma gondii, and antibiotic sensitivity of fecal Escherichia coli from New Jersey black bears (Ursus americanus). East Stroudsburg University, East Stroudsburg, PA, USA, 2004. [Google Scholar]
- Quinn, P.J.; Ramsden, R.O.; Johnston, D.H. Toxoplasmosis: A serological survey in Ontario wildlife. J. Wildl. Dis. 1976, 12, 504–510. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Binninger, C.E.; Beecham, J.J.; Thomas, L.A.; Winward, L.D. A serologic survey for selected infectious diseases of black bears in Idaho. J. Wildl. Dis. 1980, 16, 423–430. [Google Scholar] [CrossRef]
- Tizard, I.R.; Billett, J.D.B.; Ramsden, R.O. The prevalence of antibodies against Toxoplasma gondii in some Ontario mammals. J. Wildl. Dis. 1976, 12, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Zarnke, R.L.; Dubey, J.P.; Kwok, O.C.; Ver Hoef, J.M. Serologic survey for Toxoplasma gondii in selected wildlife species from Alaska. J. Wildl. Dis. 2000, 36, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Chomel, B.B.; Zarnke, R.L.; Kasten, R.W.; Kass, P.H.; Mendes, E. Serologic survey of Toxoplasma gondii in grizzly bears (Ursus arctos) and black bears (Ursus americanus), from Alaska, 1988 to 1991. J. Wildl. Dis. 1995, 31, 472–479. [Google Scholar] [CrossRef][Green Version]
- Briscoe, N.; Humphreys, J.G.; Dubey, J.P. Prevalence of Toxoplasma gondii infections in Pennsylvania black bears (Ursus americanus). J. Wildl. Dis. 1993, 29, 599–601. [Google Scholar] [CrossRef]
- Dubey, J.P.; Humphreys, J.G.; Thulliez, P. Prevalence of viable Toxoplasma gondii tissue cysts and antibodies to T. gondii by various serologic tests in black bears (Ursus americanus) from Pennsylvania. J. Parasitol. 1995, 81, 109–112. [Google Scholar] [CrossRef]
- Nutter, F.B.; Levine, J.F.; Stoskopf, M.K.; Gamble, H.R.; Dubey, J.P. Seroprevalence of Toxoplasma gondii and Trichinella spiralis in North Carolina black bears (Ursus americanus). J. Parasitol. 1998, 84, 1048–1050. [Google Scholar] [CrossRef]
- Chambers, D.L.; Ulrey, W.A.; Guthrie, J.M.; Kwok, O.C.; Cox, J.J.; Maeher, D.S.; Dubey, J.P. Seroprevalence of Toxoplasma gondii from free-ranging black bears (Ursus americanus) from Florida. J. Parasitol. 2012, 98, 674–675. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.J.; Murphy, S.M.; Augustine, B.C.; Guthrie, J.M.; Hast, J.T.; Maehr, S.C.; McDermott, J. Seroprevalence of Toxoplasma gondii in American black bears (Ursus americanus) of the Central Appalachians, USA. J. Wildl. Dis. 2017, 53, 671–673. [Google Scholar] [CrossRef] [PubMed]
- Mortenson, J.A.; Kent, M.L.; Fowler, D.R.; Chomel, B.B.; Immell, D.A. Trichinella surveillance in black bears (Ursus americanus) from Oregon, USA. J. Wildl. Dis. 2014, 50, 133–135. [Google Scholar] [CrossRef] [PubMed]
- Astorga, F.; Carver, S.; Almberg, E.S.; Sousa, G.R.; Wingfield, K.; Niedringhaus, K.D.; Van Wick, P.; Rossi, L.; Xie, Y.; Cross, P. International meeting on sarcoptic mange in wildlife, June 2018, Blacksburg, Virginia, USA. Parasit Vectors 2018, 11, 449. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.D. (Pennsylvania State University, State College, PA, USA). Personal Communication, 2019.
- Pursell, A.R.; Stuart, B.P.; Styer, E.; Case, J.L. Isolation of an adenovirus from black bear cubs. J. Wildl. Dis. 1983, 19, 269–271. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Collins, J.E.; Leslie, P.; Johnson, D.; Nelson, D.; Peden, W.; Boswell, R.; Draayer, H. Epizootic of adenovirus infection in American black bears. J. Am. Vet. Med. Assoc. 1984, 185, 1430–1432. [Google Scholar] [PubMed]
- Whetstone, C.A.; Draayer, H.; Collins, J.E. Characterization of canine adenovirus type 1 isolated from American black bears. Am. J. Vet. Res. 1988, 49, 778–780. [Google Scholar] [PubMed]
- Zarnke, R.E.; Evans, M.B. Serologic survey for infectious canine hepatitis virus in grizzly bears (Ursus arctos) from Alaska, 1973 to 1987. J. Wildl. Dis. 1989, 25, 568–573. [Google Scholar] [CrossRef]
- Ramey, A.M.; Cleveland, C.A.; Hilderbrand, G.V.; Joly, K.; Gustine, D.D.; Mangipane, B.; Leacock, W.B.; Crupi, A.P.; Hill, D.E.; Dubey, J.P. Exposure of Alaska brown bears (Ursus arctos) to bacterial, viral, and parasitic agents varies spatiotemporally and may be influenced by age. J. Wildl. Dis. 2019. [Google Scholar] [CrossRef]
- Barker, I.K.; Parrish, C.R. Parvovirus Infections. In Infectious Diseases of Wild Animals; Williams, E.S., Barker, I.K., Eds.; Blackwell Publishing: Ames, IA, USA, 2001; pp. 131–146. [Google Scholar]
- Dick, T.A.; Pozio, E. Trichinella spp. and trichinellosis. In Parasitic Diseases of Wild Mammals; Samuel, W.M., Pybus, M.J., Kocan, A.A., Eds.; Blackwell Publishing: Ames, IA, USA, 2001; pp. 380–396. [Google Scholar]
- Ternent, M.A. (Pennsylvania Game Commission, Harrisburg, PA, USA). Personal Communication.
- Chakrabarti, A.; Chatterjee, A.; Chakrabarti, K.; Sengupta, D.N. Human scabies from contact with water buffaloes infested with Sarcoptes scabiei var bubalis. Ann. Trop Med. Parasitol. 1981, 75, 353–357. [Google Scholar] [CrossRef]
- Chakrabarti, A.; Dutta, A.K.; Mandal, S.B.; Chakraborty, A.K.; Sengupta, D.N. Transmission of scabies from goats to human subjects. Indian J. Dermatol. 1981, 25, 7–13. [Google Scholar] [PubMed]
- Mitra, M.; Mahanta, S.K.; Sen, S.; Ghosh, C.; Hati, A.K. Transmission of Sarcoptes scabiei from animal to man and its control. J. Indian Med. Assoc. 1995, 93, 142–143. [Google Scholar] [PubMed]
- Heukelbach, J.; Feldmeier, H. Scabies. Lancet 2006, 367, 1767–1774. [Google Scholar] [CrossRef]
- Dubey, J.P. Sources of Toxoplasma gondii infection in pregnancy. Until rates of congenital toxoplasmosis fall, control measures are essential. BMJ 2000, 321, 127–128. [Google Scholar] [CrossRef] [PubMed]
- Tenter, A.M.; Heckeroth, A.R.; Weiss, L.M. Toxoplasma gondii: From animals to humans. Int. J. Parasitol. 2000, 30, 1217–1258. [Google Scholar] [CrossRef]
- Wilson, N.O.; Hall, R.L.; Montgomery, S.P.; Jones, J.L. Trichinellosis surveillance—United States, 2008–2012. MMWR Morb. Mortal. Wkly. Rep. 2015, 64, 1–8. [Google Scholar]
- Clausen, M.R.; Meyer, C.N.; Krantz, T.; Moser, C.; Gomme, G.; Kayser, L.; Albrectsen, J.; Kapel, C.M.; Bygbjerg, I.C. Trichinella infection and clinical disease. QJM-Int. J. Med. 1996, 89, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Hill, D.E.; Dubey, J.P. Toxoplasma gondii prevalence in farm animals in the United States. Int. J. Parasitol. 2013, 43, 107–113. [Google Scholar] [CrossRef] [PubMed]

| Pathogen | Mange No. Pos/Tested (%) | Non-Mange No. Pos/Tested (%) | Total No. Pos/Tested (%) | X2 † | p |
|---|---|---|---|---|---|
| CDV | 2/31 (6.5) | 15/209 (7.2) | 17/240 (7.1) | 0.0216 | 0.883 |
| CPV | 9/46 (19.6) | 6/48 (12.5) | 15/94 (16.0) | 0.8743 | 0.350 |
| CAV | 4/46 (8.7) | 2/41 (4.9) | 6/87 (6.9) | 0.4920 | 0.483 |
| T. gondii | 22/32 (68.8) | 172/267 (66.4) | 194/299 (64.9) | 0.2352 | 0.628 |
| Trichinella sp. | 0/32 (0) | 7/188 (3.7) | 7/220 (3.2) | 1.2 | 0.267 |
| Years Sampled | Number Positive/No. Tested (%) | Location | Reference |
|---|---|---|---|
| CDV | |||
| N/A | 0/47 (0) | Great Smoky Mt. NP, USA | [20] |
| 1988–1991 | 0/76 (0%) | Alaska, USA | [41] |
| 1993–1997 | 8/165 (4.8%) | Northwestern states, USA | [42] |
| 1993–1995 | 5/66 (8%) | Florida, USA | [43] |
| 1994–2001 | 1/38 (3%) | Banff NP and BC, Canada | [44] |
| 1999–2011 | 25/82 (30.5%) | Maryland, USA | [45] |
| 2001–2003 | 24/157 (15.3%) | California, USA | [28] |
| 2002–2010 | 2/6 (33%) | Northwest Territories, Canada | [46] |
| 2014–2016 | 17/240 (7.1%) | Pennsylvania, USA | Current Study |
| CPV | |||
| 1988–1991 | 0/76 (0%) | Alaska, USA | [41] |
| 1993–1995 | 10/62 (16%) | Florida, USA | [43] |
| 1999–2011 | 10/82 (12.2%) | Maryland, USA | [45] |
| 2001–2003 | 1/157 (0.6%) | California, USA | [28] |
| 2002–2010 | 0/14 (0%) | Northwest Territories, Canada | [46] |
| 2014–2016 | 15/94 (16%) | Pennsylvania, USA | Current Study |
| CAV | |||
| 1984 | 1/33 (3%) | Washington, USA | [47] |
| 1988–1991 | 3/76 (4%) | Alaska, USA | [41] |
| 1993–1995 | 4/66 (6%) | Florida, USA | [43] |
| 1993–1997 | 3/165 (1.8%) | Northwestern States, USA | [42] |
| 1994–2001 | 8/38 (8%) | Alberta /British Columbia, Canada | [44] |
| 1999–2011 | 7/82 (8.5%) | Maryland, USA | [45] |
| 2014–2016 | 6/87 (6.9%) | Pennsylvania, USA | Current Study |
| T. gondii | |||
| N/A | 40/149 (27%) | California, USA | [48] |
| N/A | 132/328 (40.2%) | New Jersey, USA | [49] |
| N/A | 1/3 (33%) | Ontario, Canada | [50] |
| 1971–1977 | 23/303 (8%) | Idaho, USA | [51] |
| 1971–1974 | 7/16 (43.8%) | Ontario, Canada | [52] |
| 1976–1996 | 62/143 (43%) | Alaska, USA | [53] |
| 1988–1991 | 6/40 (15%) | Alaska, USA | [54] |
| 1989–1992 | 532/665 (80%) | Pennsylvania, USA | [55] |
| 1993 | 22/28 (78.6%) | Pennsylvania, USA | [56] |
| 1993–1995 | 37/66 (56%) | Florida, USA | [43] |
| 1993–1997 | 89/198 (45%) | Northwestern States, USA | [42] |
| 1994–2001 | 5/38 (13%) | Banff NP and BC, Canada | [44] |
| 1996–1997 | 120/143 (84%) | North Carolina, USA | [57] |
| 1999–2011 | 70/82 (85.4%) | Maryland, USA | [45] |
| 2001–2003 | 67/239 (28%) | California, USA | [28] |
| 2002–2010 | 2/16 (12.5%) | Northwest Territories, Canada | [46] |
| 2004–2006 | 13/29 (44.8%) | Florida, USA | [58] |
| 2012–2013 | 33/53 (62%) | Central Appalachia, USA | [59] |
| 2014–2016 | 194/299 (64.9%) | Pennsylvania, USA | Current Study |
| Trichinella spp. | |||
| N/A | 18/141(13%) | California, USA | [48] |
| 1971–1977 | 16/122 (13%) | Idaho, USA | [51] |
| 1988–1991 | 11/76 (14.5%) | Alaska, USA | [41] |
| 1993–1997 | 2/103 (1.9%) | Oregon, USA | [60] |
| 1996–1997 | 0/79 (0%) | North Carolina, USA | [57] |
| 2001–2003 | 6/80 (7.5%) | California, USA | [28] |
| 2014–2016 | 7/220 (3.2%) | Pennsylvania, USA | Current Study |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niedringhaus, K.D.; Brown, J.D.; Ternent, M.A.; Cleveland, C.A.; Yabsley, M.J. A Serosurvey of Multiple Pathogens in American Black Bears (Ursus americanus) in Pennsylvania, USA Indicates a Lack of Association with Sarcoptic Mange. Vet. Sci. 2019, 6, 75. https://doi.org/10.3390/vetsci6040075
Niedringhaus KD, Brown JD, Ternent MA, Cleveland CA, Yabsley MJ. A Serosurvey of Multiple Pathogens in American Black Bears (Ursus americanus) in Pennsylvania, USA Indicates a Lack of Association with Sarcoptic Mange. Veterinary Sciences. 2019; 6(4):75. https://doi.org/10.3390/vetsci6040075
Chicago/Turabian StyleNiedringhaus, Kevin D., Justin D. Brown, Mark A. Ternent, Christopher A. Cleveland, and Michael J. Yabsley. 2019. "A Serosurvey of Multiple Pathogens in American Black Bears (Ursus americanus) in Pennsylvania, USA Indicates a Lack of Association with Sarcoptic Mange" Veterinary Sciences 6, no. 4: 75. https://doi.org/10.3390/vetsci6040075
APA StyleNiedringhaus, K. D., Brown, J. D., Ternent, M. A., Cleveland, C. A., & Yabsley, M. J. (2019). A Serosurvey of Multiple Pathogens in American Black Bears (Ursus americanus) in Pennsylvania, USA Indicates a Lack of Association with Sarcoptic Mange. Veterinary Sciences, 6(4), 75. https://doi.org/10.3390/vetsci6040075






