Cytokine Expression in Canine Lymphoma, Osteosarcoma, Mammary Gland Tumour and Melanoma: Comparative Aspects
Abstract
1. Introduction
2. Role of Cytokines in Cancer Development
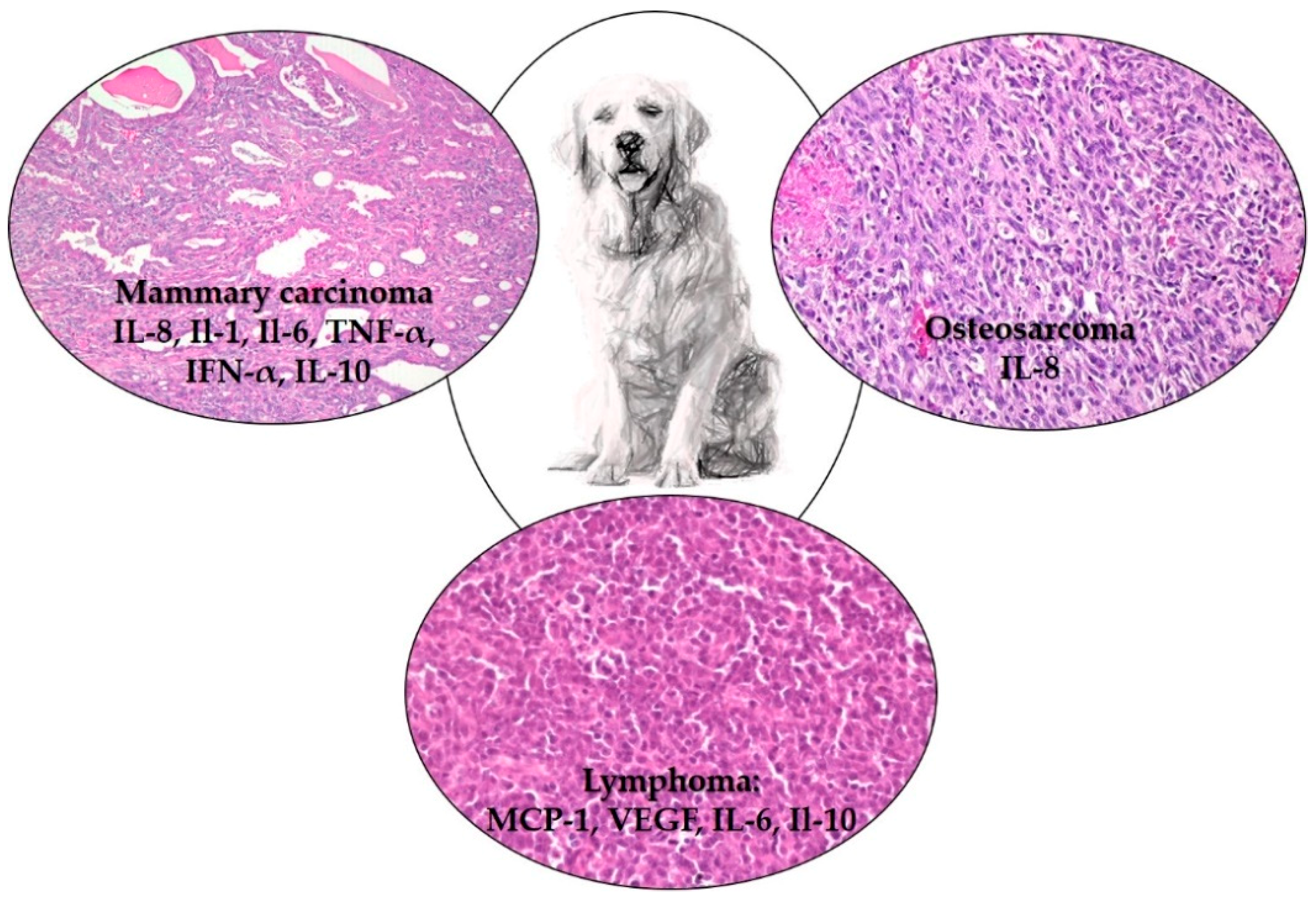
3. Cytokines and Lymphoma
4. Cytokines and Osteosarcoma
5. Cytokine and Mammary Gland Tumours
6. Cytokines and Melanoma
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| (CMT) | Canine mammary tumours |
| (CXCL9) | Chemokine Ligand 9 |
| CX3CL1 | chemokine (C-X3-C motif) ligand 1 |
| FK | fractalkine |
| (HGF) | Hepatocyte Growth Factor |
| IFN | interferon |
| IFN-α/β | interferon alpha/beta |
| IFN-λ | interferon gamma receptor |
| IL-1 | interleukin-1 |
| IL-2 | interleukin-2 |
| IL-6 | interleukin-6 |
| IL-8 | interleukin-8 |
| IL-10 | interleukin-10 |
| IL-12 | interleukin-12 |
| IL-13 | interleukin-13 |
| IL-15 | interleukin-15 |
| IL-17 | interleukin-17 |
| IL-34 | interleukin-34 |
| (IP-10) | Interferon gamma-induced protein 10 |
| IL-12p40 | (the p40 subunit of IL-12) |
| (LPDs) | Lymphoproliferative disorders |
| (Mip-1α/β) | Macrophage inflammatory protein-1α/β |
| M-CSF | macrophage-colony stimulating factor |
| CSF-1 | colony stimulating factor |
| (MCP-1) | Macrophage Chemoattractant Protein-1 |
| (NHL) | non-Hodgkin lymphoma |
| OSA | osteosarcoma |
| PEG | polyethylene glycol |
| TGFβ | transforming growth factor beta |
| (TNF-α/β) | Tumor Necrosis Factor-α/β |
| (LTBP4) | TGFβ and latent TGFβ binding protein 4 |
| (LY2109761) | TGFβRI/II inhibitor |
| (VEGF) | Vascular endothelial growth factor |
References
- Kent, M.S.; Burton, J.H.; Dank, G.; Bannasch, D.L.; Rebhun, R.B. Association of cancer-related mortality, age and gonadectomy in golden retriever dogs at a veterinary academic center (1989–2016). PLoS ONE 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Gardner, H.L.; Fenger, J.M.; London, C.A. Dogs as a Model for Cancer. Annu. Rev. Anim. Biosci. 2016, 4. [Google Scholar] [CrossRef]
- Abdelmegeed, S.M.; Mohammed, S. Canine mammary tumors as a model for human disease (Review). Oncol. Lett. 2018, 15, 8195–8205. [Google Scholar] [CrossRef]
- Hernandez, B.; Adissu, H.A.; Wei, B.R.; Michael, H.T.; Merlino, G.; Mark Simpson, R. Naturally occurring canine melanoma as a predictive comparative oncology model for human mucosal and other triple wild-type melanomas. IJMS 2018, 19, 394. [Google Scholar] [CrossRef]
- Mcinnes, I.B. Cytokines. In Kelley Firestein’s Textbook of Rheumatolology, 10th ed.; Firestein, G.S., Budd, R.C., Gabriel, S.E., McInnes, I.B., O’Dell, J.R., Eds.; Elsevier: St. Louis, MO, USA, 2017; Volume 2, pp. 396–407. [Google Scholar]
- Deans, C.; Wigmore, S.J. Systemic inflammation, cachexia and prognosis in patients with cancer. Curr. Opin. Clin. Nutr. Metab. Care 2005, 8, 265–269. [Google Scholar] [CrossRef]
- Salvatore, V.; Teti, G.; Focaroli, S.; Mazzotti, M.C.; Mazzotti, A.; Falconi, M. The tumor microenvironment promotes cancer progression and cell migration. Oncotarget 2017, 8, 9608–9616. [Google Scholar] [CrossRef]
- Derin, D.; Soydinc, H.O.; Guney, N.; Tas, F.; Camlica, H.; Duranyildiz, D.; Yasasever, V.; Topuz, E. Serum IL-8 and IL-12 levels in breast cancer. Med. Oncol. 2007, 24, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Machado, V.S.; Crivellenti, L.Z.; Bottari, N.B.; Tonin, A.A.; Pelinson, L.P.; Borin-Crivellenti, S.; Santana, A.E.; Torbitz, V.D.; Moresco, R.N.; Duarte, T.; et al. Oxidative stress and inflammatory response biomarkers in dogs with mammary carcinoma. Pathol. Res. Pract. 2015, 211. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Yu, C.H.; Yhee, J.Y.; Im, K.S.; Sur, J.H. Lymphocyte infiltration, expression of interleukin (IL) -1, IL-6 and expression of mutated breast cancer susceptibility gene-1 correlate with malignancy of canine mammary tumours. J. Comp. Pathol. 2010, 142, 177–186. [Google Scholar] [CrossRef]
- Lopes-Júnior, L.C.; da Silveira, D.S.C.; Vulczak1, A.; dos Santos, J.C.; Veronez, L.C.; Fisch, A.; Flória-Santos, M.; de Lima, R.A.G.; da-Silva, G.P. Emerging Cytokine Networks in Osteosarcoma. Cancer Cell Microenviron. 2017, 4, 1–11. [Google Scholar]
- Fabre-Guillevin, E.; Tabrizi, R.; Coulon, V.; Monnereau, A.; Eghbali, H.; Soubeyran, I.; Soubeyran, P. Aggressive non-Hodgkin’s lymphoma: Concomitant evaluation of interleukin-2, soluble interleukin-2 receptor, interleukin-4, interleukin-6, interleukin-10 and correlation with outcome. Leuk. Lymphoma 2006, 47, 603–611. [Google Scholar] [CrossRef]
- Preti, H.A.; Cabanillas, F.; Talpaz, M.; Tucker, S.L.; Seymour, J.F.; Kurzrock, R. Prognostic value of serum interleukin-6 in diffuse large-cell lymphoma. Ann. Intern. Med. 1997, 127, 186–194. [Google Scholar] [CrossRef]
- Seymour, J.F.; Talpaz, M.; Cabanillas, F.; Wetzler, M.; Kurzrock, R. Serum interleukin-6 levels correlate with prognosis in diffuse large-cell lymphoma. J. Clin. Oncol. 1995, 13, 575–582. [Google Scholar] [CrossRef]
- Aresu, L.; Aricò, A.; Comazzi, S.; Gelain, M.E.; Riondato, F.; Mortarino, M.; Morello, E.; Stefanello, D.; Castagnaro, M. VEGF and MMP-9: Biomarkers for canine lymphoma. Vet. Comp. Oncol. 2014, 12. [Google Scholar] [CrossRef]
- Merlo, A.; Rezende, B.C.; Franchini, M.L.; Simões, D.M.; Lucas, S.R. Serum C-reactive protein concentrations in dogs with multicentric lymphoma undergoing chemotherapy. J. Am. Vet. Med. Assoc. 2007, 230, 522–526. [Google Scholar] [CrossRef]
- Mischke, R.; Waterston, M.; Eckersall, P.D. Changes in C-reactive protein and haptoglobin in dogs with lymphatic neoplasia. Vet. J. 2007, 174, 188–192. [Google Scholar] [CrossRef]
- Perry, J.A.; Thamm, D.H.; Eickhoff, J.; Avery, A.C.; Dow, S.W. Increased monocyte chemotactic protein-1 concentration and monocyte count independently associate with a poor prognosis in dogs with lymphoma. Vet. Comp. Oncol. 2011, 9. [Google Scholar] [CrossRef]
- Paoloni, M.; Davis, S.; Lana, S.; Withrow, S.; Sangiorgi, L.; Picci, P.; Hewitt, S.; Triche, T.; Meltzer, P.; Khanna, C. Canine tumor cross-species genomics uncovers targets linked to osteosarcoma progression. BMC Genomics 2009, 10. [Google Scholar] [CrossRef]
- Benoy, I.H.; Salgado, R.; Van Dam, P.; Geboers, K.; Van Marck, E.; Scharpé, S.; Vermeulen, P.B.; Dirix, L.Y. Increased serum interleukin-8 in patients with early and metastatic breast cancer correlates with early dissemination and survival. Clin. Cancer Res. 2004, 10, 7157–7162. [Google Scholar] [CrossRef]
- de Andrés, P.J.; Illera, J.C.; Cáceres, S.; Díez, L.; Pérez-Alenza, M.D.; Peña, L. Increased levels of interleukins 8 and 10 as findings of canine inflammatory mammary cancer. Vet. Immunol. Immunopathol. 2013, 152. [Google Scholar] [CrossRef]
- Lyon, D.E.; McCain, N.L.; Walter, J.; Schubert, C. Cytokine comparisons between women with breast cancer and women with a negative breast biopsy. Nurs. Res. 2008, 57, 51–58. [Google Scholar] [CrossRef]
- Klopfleisch, R.; Schütze, M.; Gruber, A.D. Downregulation of transforming growth factor beta (TGFbeta) and latent TGFbeta binding protein (LTBP)-4 expression in late stage canine mammary tumours. Vet. J. 2010, 186, 379–384. [Google Scholar] [CrossRef]
- Portela, R.F.; Fadl-Alla, B.A.; Pondenis, H.C.; Byrum, M.L.; Garrett, L.D.; Wycislo, K.L.; Borst, L.B.; Fan, T.M. Pro-tumorigenic effects of transforming growth factor beta 1 in canine osteosarcoma. J. Vet. Intern. Med. 2014, 28. [Google Scholar] [CrossRef]
- Lu, H.; Shu, X.O.; Cui, Y.; Kataoka, N.; Wen, W.; Cai, Q.; Ruan, Z.X.; Gao, Y.T.; Zheng, W. Association of genetic polymorphisms in the VEGF gene with breast cancer survival. Cancer Res. 2005, 65, 5015–5019. [Google Scholar] [CrossRef]
- Hefler, L.A.; Grimm, C.; Ackermann, S.; Malur, S.; Radjabi-Rahat, A.R.; Leodolter, S.; Beckmann, M.W.; Zeillinger, R.; Koelbl, H.; Tempfer, C.B. An interleukin-6 gene promoter polymorphism influences the biological phenotype of ovarian cancer. Cancer Res. 2003, 63, 3066–3068. [Google Scholar]
- Macarthur, M.; Hold, G.L.; El-Omar, E.M. Inflammation and Cancer II. Role of chronic inflammation and cytokine gene polymorphisms in the pathogenesis of gastrointestinal malignancy. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 286, G515–G520. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, Y.; Liu, S.; Zhu, S.; Jiang, H.; Ding, J. Association between interleukin 8−251 A/T and +781 C/T polymorphisms and osteosarcoma risk in Chinese population: a case–control study. Tumor Biol. 2016, 37. [Google Scholar] [CrossRef]
- Wang, J.; Nong, L.; Wei, Y.; Qin, S.; Zhou, Y.; Tang, Y. Association of Interleukin-12 Polymorphisms and Serum IL-12p40 Levels with Osteosarcoma Risk. DNA Cell Biol. 2013, 32. [Google Scholar] [CrossRef]
- Yoo, S.Y.; Lee, S.Y.; Yoo, N.C. Cytokine expression and cancer detection. Med. Sci. Monit. 2009, 15, RA49–RA56. [Google Scholar]
- Dranoff, G. Cytokines in cancer pathogenesis and cancer therapy. Nat. Rev. Cancer 2004, 4, 11–22. [Google Scholar] [CrossRef]
- Vail, D.; Young, K.; Pinkerton, M.E. Canine lymphoma and lymphoid leukemia. In Small Animal Clinical Oncology; Withrow, S.J., Vail, D.M., Page, R.L., Eds.; Elsevier: St. Louis, MO, USA, 2013; Volume 5, pp. 608–637. [Google Scholar]
- Marconato, L.; Gelain, M.E.; Comazzi, S. The dog as a possible animal model for human non-Hodgkin lymphoma: A review. Hematol. Oncol. 2013, 31, 1–9. [Google Scholar] [CrossRef]
- Charbonneau, B.; Maurerm, M.J.; Ansellm, S.M.; Slager, S.L.; Fredericksen, Z.S.; Ziesmer, S.C.; Macon, W.R.; Habermann, T.M.; Witzig, T.E.; Link, B.K.; et al. Pretreatment circulating serum cytokines associated with follicular and diffuse large B-cell lymphoma: A clinic-based case-control study. Cytokine 2012, 60. [Google Scholar] [CrossRef]
- Stasi, R.; Zinzani, L.; Galieni, P.; Lauta, V.M.; Damasio, E.; Dispensa, E.; Dammacco, F.; Venditti, A.; Del Poeta, G.; Cantonetti, M. Clinical implications of cytokine and soluble receptor measurements in patients with newly-diagnosed aggressive non-Hodgkin’s lymphoma. Eur. J. Haematol. 1995, 54, 9–17. [Google Scholar] [CrossRef]
- Pedersen, L.M.; Klausen, T.W.; Davidsen, U.H.; Johnsen, H.E. Early changes in serum IL-6 and VEGF levels predict clinical outcome following first-line therapy in aggressive non-Hodgkin’s lymphoma. Ann. Hematol. 2005, 84, 510–516. [Google Scholar] [CrossRef]
- Calvalido, J.; Wood, G.A.; Mutsaers, A.J.; Wood, D.; Sears, W.; Woods, J.P. Comparison of serum cytokine levels between dogs with multicentric lymphoma and healthy dogs. Vet. Immunol. Immunopathol. 2016, 182. [Google Scholar] [CrossRef]
- Hofer, J.; DeFrancesco, T.C.; Williams, L.E. Detection of tumour necrosis factor-alpha in dogs with lymphoma. Vet. Comp. Oncol. 2011, 9. [Google Scholar] [CrossRef]
- Cavalcanti, J.N.; Amstalden, E.M.I.; Guerra, J.L.; Magna, L.C. Osteosarcoma in dogs: clinical-morphological study and prognostic correlation. Brazilian J. Vet. Res. Anim. Sci. 2004, 41. [Google Scholar] [CrossRef]
- McMahon, M.; Mathie, T.; Stingle, N.; Romansik, E.; Vail, D.; London, C. Adjuvant Carboplatin and Gemcitabine Combination Chemotherapy Postamputation in Canine Appendicular Osteosarcoma. J. Vet. Intern. Med. 2011, 25, 511–517. [Google Scholar] [CrossRef]
- Mialou, V.; Philip, T.; Kalifa, C.; Perol, D.; Gentet, J.C.; Marec-Berard, P.; Pacquement, H.; Chastagner, P.; Defaschelles, A.S.; Hartmann, O. Metastatic osteosarcoma at diagnosis: Prognostic factors and long-term outcome—The French pediatric experience. Cancer 2005, 104, 1100–1109. [Google Scholar] [CrossRef]
- Mankin, H.J.; Hornicek, F.J.; Rosenberg, A.E.; Harmon, D.C.; Gebhardt, M.C. Survival data for 648 patients with osteosarcoma treated at one institution. Clin. Orthop. Relat. Res. 2004, 429, 286–291. [Google Scholar] [CrossRef]
- Kloen, P.; Gebhardt, M.C.; Perez-Atayde, A.; Rosenberg, A.E.; Springfield, D.S.; Gold, L.I.; Mankin, H.J. Expression of transforming growth factor-beta (TGF-beta) isoforms in osteosarcomas: TGF-beta3 is related to disease progression. Cancer 1997, 80, 2230–2239. [Google Scholar] [CrossRef]
- Antuofermo, E.; Miller, M.A.; Pirino, S.; Xie, J.; Badve, S.; Mohammed, S.I. Spontaneous mammary intraepithelial lesions in dogs—A model of breast cancer. Cancer Epidemiol. Biomarkers Prev. 2007, 16, 2247–2256. [Google Scholar] [CrossRef]
- Misdorp, W.; Armed Forces Institute of Pathology (U.S.); American Registry of Pathology; WHO Collaborating Center for Worldwide Reference on Comparative Oncology. Histological Classification of Mammary Tumors of the Dog and the Cat; Armed Forces Institute of Pathology in Cooperation with the American Registry of Pathology and the World Health Organization Collaborating Center for Worldwide Reference on Comparative Oncology: Washington, DC, USA, 1999; pp. 1–59. [Google Scholar]
- Gelaleti, G.B.; Jardim, B.V.; Leonel, C.; Moschetta, M.G.; Zuccari, D.A. Interleukin-8 as a prognostic serum marker in canine mammary gland neoplasias. Vet. Immunol. Immunopathol. 2012, 146. [Google Scholar] [CrossRef]
- De Larco, J.E.; Wuertz, B.R.; Yee, D.; Rickert, B.L.; Furcht, L.T. Atypical methylation of the interleukin-8 gene correlates strongly with the metastatic potential of breast carcinoma cells. PNAS 2003, 100, 13988–13993. [Google Scholar] [CrossRef]
- Schwaninger, R.; Rentsch, C.A.; Wetterwald, A.; van der Horst, G.; van Bezooijen, R.L.; van der Pluijm, G.; Löwik, C.W.; Ackermann, K.; Pyerin, W.; Hamdy, F.C.; et al. Lack of noggin expression by cancer cells is a determinant of the osteoblast response in bone metastases. Am. J. Pathol. 2007, 170, 160–175. [Google Scholar] [CrossRef]
- Peña, L.; Perez-Alenza, M.D.; Rodriguez-Bertos, A.; Nieto, A. Canine inflammatory mammary carcinoma: histopathology, immunohistochemistry and clinical implications of 21 cases. Breast Cancer Res. Treat. 2003, 78, 141–148. [Google Scholar] [CrossRef]
- Pérez Alenza, M.D.; Tabanera, E.; Peña, L. Inflammatory mammary carcinoma in dogs: 33 cases (1995–1999). J. Am. Vet. Med. Assoc. 2001, 219, 1110–1114. [Google Scholar] [CrossRef]
- Wakefield, L.M.; Yang, Y.A.; Dukhanina, O. Transforming growth factor-beta and breast cancer: Lessons learned from genetically altered mouse models. Breast Cancer Res. 2000, 2, 100–106. [Google Scholar] [CrossRef]
- Wakefield, L.M.; Piek, E.; Böttinger, E.P. TGF-beta signaling in mammary gland development and tumorigenesis. J. Mammary Gland Biol. Neoplasia 2001, 6, 67–82. [Google Scholar] [CrossRef]
- Smedley, R.C.; Spangler, W.L.; Esplin, D.G.; Kitchell, B.E.; Bergman, P.J.; Ho, H.Y.; Bergin, I.L.; Kiupel, M. Prognostic markers for canine melanocytic neoplasms: A comparative review of the literature and goals for future investigation. Vet Pathol. 2011, 48. [Google Scholar] [CrossRef]
- Simpson, R.M.; Bastian, B.C.; Michael, H.T.; Webster, J.D.; Prasad, M.L.; Conway, C.M.; Prieto, V.M.; Gary, J.M.; Goldschmidt, M.H.; Esplin, D.G.; et al. Sporadic naturally occurring melanoma in dogs as a preclinical model for human melanoma. Pigment Cell Melanoma Res. 2014, 27. [Google Scholar] [CrossRef]
- Kupcova Skalnikova, H.; Cizkova, J.; Cervenka, J.; Vodicka, P. Advances in Proteomic Techniques for Cytokine Analysis: Focus on Melanoma Research. IJMS 2017, 18, 2697. [Google Scholar] [CrossRef]
- Rosenberg, S.A. IL-2: The First Effective Immunotherapy for Human Cancer. J. Immunol. 2014, 192, 5451–5458. [Google Scholar] [CrossRef]
- Finocchiaro, L.M.; Glikin, G.C. Recent clinical trials of cancer immunogene therapy in companion animals. World J. Exp. Med. 2017, 7. [Google Scholar] [CrossRef]
- Atherton, M.J.; Morris, J.S.; McDermott, M.R.; Lichty, B.D. Cancer immunology and canine malignant melanoma: A comparative review. Vet. Immunol. Immunopathol. 2016, 169. [Google Scholar] [CrossRef]
- Finocchiaro, L.M.; Fiszman, G.L.; Karara, A.L.; Glikin, G.C. Suicide gene and cytokines combined nonviral gene therapy for spontaneous canine melanoma. Cancer Gene Ther. 2008, 15. [Google Scholar] [CrossRef]
- Thamm, D.H.; Kurzman, I.D.; Clark, M.A.; Ehrhart, E.J., 3rd; Kraft, S.L.; Gustafson, D.L.; Vail, D.M. Preclinical investigation of PEGylated tumor necrosis factor α in dogs with spontaneous tumors: Phase I evaluation. Clin. Cancer Res. 2010, 16. [Google Scholar] [CrossRef]
| Cytokine | Expression Level | Cancer Type |
|---|---|---|
| IL-1 | Increased | Mammary carcinoma (D) [9,10] |
| IL-2 | Increased | OSA (H) [11] |
| IL-6 | Increased | Lymphoma (H, D) [12,13,14,15,16,17,18], OSA (H) [11], Mammary Carcinoma (D) [9,10] |
| IL-8 | Increased | Lymphoma (H) [12,13,14], OSA (H, D) [11,19], Mammary carcinoma (H, D) [20,21] |
| IL-10 | Increased | Lymphoma (H, D) [12,13,14,15,16,17,18], Mammary carcinoma (H, D) [9,21,22] |
| IL-13 | Increased | Lymphoma (H) [12,13,14] |
| VEGF | Increased | Lymphoma (H, D) [12,13,14,15,16,17,18], OSA (H) [11] |
| CXCL9 | Increased | Lymphoma (H) [12,13,14] |
| IP-10 | Increased | Lymphoma (H) [12,13,14] |
| IFN-γ | Increased | Lymphoma (H) [12,13,14] |
| IFN-α | Increased | Mammary carcinoma (D) [9] |
| IL-12p40 | Increased | Lymphoma (H) [12,13,14] |
| MIP-1α/β | Increased | Lymphoma (H) [12,13,14] |
| HGF | Increased | Lymphoma (H) [12,13,14] |
| MCP-1 | Increased | Lymphoma (D) [15,16,17,18] |
| TGF-β | Decreased | Lymphoma (D) [15,16,17,18], Mammary carcinoma (D) [23] |
| Increased | OSA (H) [11], OSA cell lines (D) [24] | |
| TNF-α | Increased | OSA(H) [11], Mammary carcinoma (D) [9] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Irac, S.E.; Oksa, A.; Jackson, K.; Herndon, A.; Allavena, R.; Palmieri, C. Cytokine Expression in Canine Lymphoma, Osteosarcoma, Mammary Gland Tumour and Melanoma: Comparative Aspects. Vet. Sci. 2019, 6, 37. https://doi.org/10.3390/vetsci6020037
Irac SE, Oksa A, Jackson K, Herndon A, Allavena R, Palmieri C. Cytokine Expression in Canine Lymphoma, Osteosarcoma, Mammary Gland Tumour and Melanoma: Comparative Aspects. Veterinary Sciences. 2019; 6(2):37. https://doi.org/10.3390/vetsci6020037
Chicago/Turabian StyleIrac, Sergio Erdal, Annika Oksa, Karen Jackson, Aaron Herndon, Rachel Allavena, and Chiara Palmieri. 2019. "Cytokine Expression in Canine Lymphoma, Osteosarcoma, Mammary Gland Tumour and Melanoma: Comparative Aspects" Veterinary Sciences 6, no. 2: 37. https://doi.org/10.3390/vetsci6020037
APA StyleIrac, S. E., Oksa, A., Jackson, K., Herndon, A., Allavena, R., & Palmieri, C. (2019). Cytokine Expression in Canine Lymphoma, Osteosarcoma, Mammary Gland Tumour and Melanoma: Comparative Aspects. Veterinary Sciences, 6(2), 37. https://doi.org/10.3390/vetsci6020037







