Urinalysis and Urinary GGT-to-Urinary Creatinine Ratio in Dogs with Acute Pancreatitis
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Xenoulis, P.G. Diagnosis of pancreatitis in dogs and cats. J. Small Anim. Pract. 2015, 56, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Hess, R.S.; Saunders, H.M.; Van Winkle, T.J.; Shofer, F.S.; Washabau, R.J. Clinical, clinicopathologic, radiographic, and ultrasonographic abnormalities in dogs with fatal acute pancreatitis: 70 cases (1986–1995). J. Am. Vet. Med. Assoc. 1998, 213, 665–670. [Google Scholar] [PubMed]
- Pápa, K.; Máthé, A.; Abonyi-Tóth, Z.; Sterczer, A.; Psáder, R.; Hetyey, C.; Vajdovich, P.; Vörös, K. Occurrence, clinical features and outcome of canine pancreatitis (80 cases). Acta Vet. Hung. 2011, 59, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, C. Pathophysiology of acute pancreatitis: Potential application from experimental models and human medicine to dogs. J. Vet. Intern. Med. 2012, 26, 875–887. [Google Scholar] [CrossRef] [PubMed]
- Petejova, N.; Martinek, A. Acute kidney injury following acute pancreatitis: A review. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc. Czech Repub. 2013, 157, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, V.; Gori, E.; Lippi, I.; Luchetti, E.; Manca, M.L.; Pierini, A. Elevated serum creatinine and hyponatraemia as prognostic factors in canine acute pancreatitis. Aust. Vet. J. 2017, 95, 444–447. [Google Scholar] [CrossRef] [PubMed]
- Tvarijonaviciute, A.; García-Martínez, J.D.; Caldin, M.; Martínez-Subiela, S.; Tecles, F.; Pastor, J.; Ceron, J.J. Serum paraoxonase 1 (PON1) activity in acute pancreatitis of dogs. J. Small Anim. Pract. 2015, 56, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Rybak, K.; Sporek, M.; Gala-Błądzińska, A.; Mazur-Laskowska, M.; Dumnicka, P.; Walocha, J.; Drożdż, R.; Kuźniewski, M.; Ceranowicz, P.; Kuśnierz-Cabala, B. [Urinalysis in patients at the early stage of acute pancreatitis]. Prz. Lek. 2016, 73, 88–92. [Google Scholar] [PubMed]
- Clemo, F.A. Urinary enzyme evaluation of nephrotoxicity in the dog. Toxicol. Pathol. 1998, 26, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Cobrin, A.R.; Blois, S.L.; Kruth, S.A.; Abrams-Ogg, A.C.G.; Dewey, C. Biomarkers in the assessment of acute and chronic kidney diseases in the dog and cat. J. Small Anim. Pract. 2013, 54, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Lippi, I.; Perondi, F.; Meucci, V.; Bruno, B.; Gazzano, V.; Guidi, G. Clinical utility of urine kidney injury molecule-1 (KIM-1) and gamma-glutamyl transferase (GGT) in the diagnosis of canine acute kidney injury. Vet. Res. Commun. 2018, 225, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Li, Y.; Tang, Y.; Liu, F.; Yu, S.; Zhang, L.; Zeng, X.; Zhao, Y.; Fu, P. Effect of acute kidney injury on mortality and hospital stay in patient with severe acute pancreatitis. Nephrology 2015, 20, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Pahwa, N.; Jain, N. Acute kidney injury in severe acute pancreatitis: An experience from a tertiary care center. Saudi J. Kidney Dis. Transpl. 2015, 26, 56–60. [Google Scholar] [PubMed]
- Naqvi, R. Acute Kidney Injury in association with Acute Pancreatitis. Pak. J. Med. Sci. 2018, 34, 606–609. [Google Scholar] [CrossRef] [PubMed]
- Gori, E.; Lippi, I.; Guidi, G.; Perondi, F.; Pierini, A.; Marchetti, V. Acute pancreatitis and acute kidney injury in dogs. Vet. J. 2019, 245, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Finco, D.R.; Brown, S.A.; Vaden, S.L.; Ferguson, D.C. Relationship between plasma creatinine concentration and glomerular filtration rate in dogs. J. Vet. Pharmacol. Ther. 1995, 18, 418–421. [Google Scholar] [CrossRef] [PubMed]
- Osborne, C.A.; Lulich, J.P.; Albasan, H. The ins and outs of urine collection. In Nephrology and Urology of Small Animals, 1st ed.; Bartges, J., Polzin, D.J., Eds.; Blackwell Publishing Ltd.: Oxford, UK, 2014; pp. 28–42. ISBN 978-0-8138-1717-0. [Google Scholar]
- Barsanti, J.A. Urinary disorders. In Small Animal Clinical Diagnosis by Laboratory Methods, 5th ed.; Willard, M.D., Tveden, H., Eds.; Elsevier Saunders: St. Louis, MO, USA, 2012; pp. 126–155. ISBN 978-1437706574. [Google Scholar]
- Beatrice, L.; Nizi, F.; Callegari, D.; Paltrinieri, S.; Zini, E.; D’Ippolito, P.; Zatelli, A. Comparison of urine protein-to-creatinine ratio in urine samples collected by cystocentesis versus free catch in dogs. J. Am. Vet. Med. Assoc. 2010, 236, 1221–1224. [Google Scholar] [CrossRef] [PubMed]
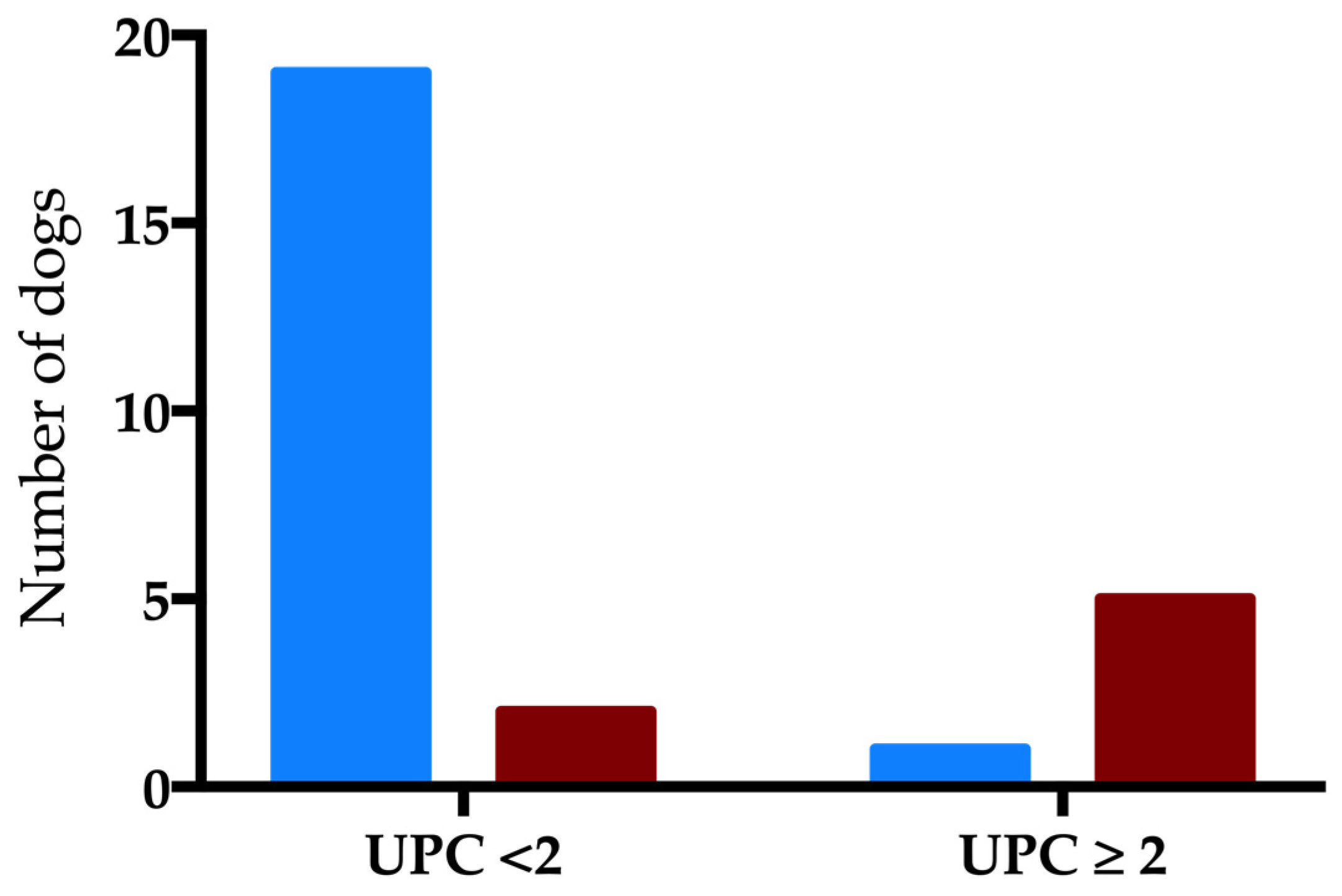
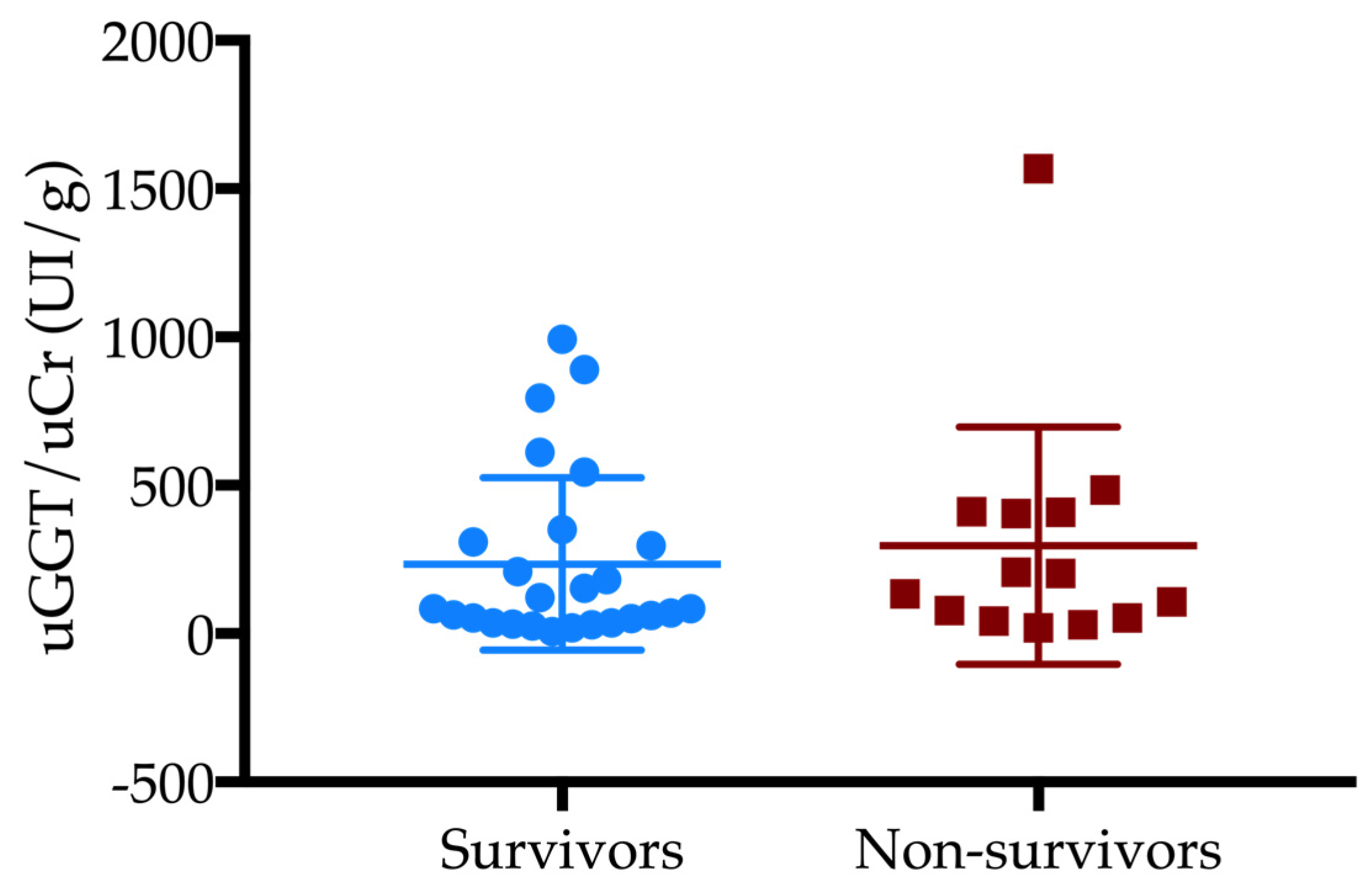
| Parameter | Survivors (n = 46) | Non-Survivors (n = 24) | p-Values |
|---|---|---|---|
| pH * | 6.5 ± 1 | 6.3 ± 0.9 | 0.35 |
| SG § | 1018 (1007–1050) | 1017 (1004–1048) | 0.42 |
| PRO | |||
| neg | 30 (43%) | 10 (14%) | 0.09 |
| TR | 0 (0%) | 0 (0%) | |
| 1+ | 8 (11%) | 6 (9%) | |
| 2+ | 5 (7%) | 2 (3%) | |
| 3+ | 3 (4%) | 6 (9%) | |
| GLU | |||
| neg | 39 (56%) | 24 (34%) | 0.24 |
| 1+ | 2 (3%) | 0 (0%) | |
| 2+ | 1 (1%) | 0 (0%) | |
| 3+ | 0 (0%) | 0 (0%) | |
| 4+ | 4 (6%) | 0 (0%) | |
| KET | |||
| neg | 38 (54%) | 20 (29%) | 0.3 |
| 1+ | 5 (7%) | 4 (6%) | |
| 2+ | 3 (4%) | 0 (0%) | |
| 3+ | 0 (0%) | 0 (0%) | |
| UBG | |||
| norm | 16 (24%) | 6 (9%) | 0.23 |
| 1+ | 24 (35%) | 11 (15%) | |
| 2+ | 5 (7%) | 3 (4%) | |
| 3+ | 1 (1%) | 3 (4%) | |
| 4+ | 0 (0%) | 1 (1%) | |
| BIL | |||
| neg | 34 (49%) | 15 (22%) | 0.022 |
| 1+ | 3 (4%) | 1 (1%) | |
| 2+ | 8 (11%) | 2 (3%) | |
| 3+ | 1 (1%) | 6 (9%) | |
| BLD/HGB | |||
| neg | 23 (33%) | 7 (10%) | 0.17 |
| 1+ | 3 (4%) | 0 (0%) | |
| 2+ | 6 (9%) | 4 (6%) | |
| 3+ | 4 (6%) | 2 (3%) | |
| 4+ | 10 (14%) | 11 (15%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gori, E.; Pierini, A.; Lippi, I.; Boffa, N.; Perondi, F.; Marchetti, V. Urinalysis and Urinary GGT-to-Urinary Creatinine Ratio in Dogs with Acute Pancreatitis. Vet. Sci. 2019, 6, 27. https://doi.org/10.3390/vetsci6010027
Gori E, Pierini A, Lippi I, Boffa N, Perondi F, Marchetti V. Urinalysis and Urinary GGT-to-Urinary Creatinine Ratio in Dogs with Acute Pancreatitis. Veterinary Sciences. 2019; 6(1):27. https://doi.org/10.3390/vetsci6010027
Chicago/Turabian StyleGori, Eleonora, Alessio Pierini, Ilaria Lippi, Noemi Boffa, Francesca Perondi, and Veronica Marchetti. 2019. "Urinalysis and Urinary GGT-to-Urinary Creatinine Ratio in Dogs with Acute Pancreatitis" Veterinary Sciences 6, no. 1: 27. https://doi.org/10.3390/vetsci6010027
APA StyleGori, E., Pierini, A., Lippi, I., Boffa, N., Perondi, F., & Marchetti, V. (2019). Urinalysis and Urinary GGT-to-Urinary Creatinine Ratio in Dogs with Acute Pancreatitis. Veterinary Sciences, 6(1), 27. https://doi.org/10.3390/vetsci6010027









