Modulation of Immune Response and Cecal Microbiota by Dietary Fenugreek Seeds in Broilers
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Birds, Diets, and Management
2.2. RNA Isolation and RT-qPCR
2.3. Microbial DNA Extraction and 16S rRNA Sequencing
2.4. Statistical Analysis
3. Results
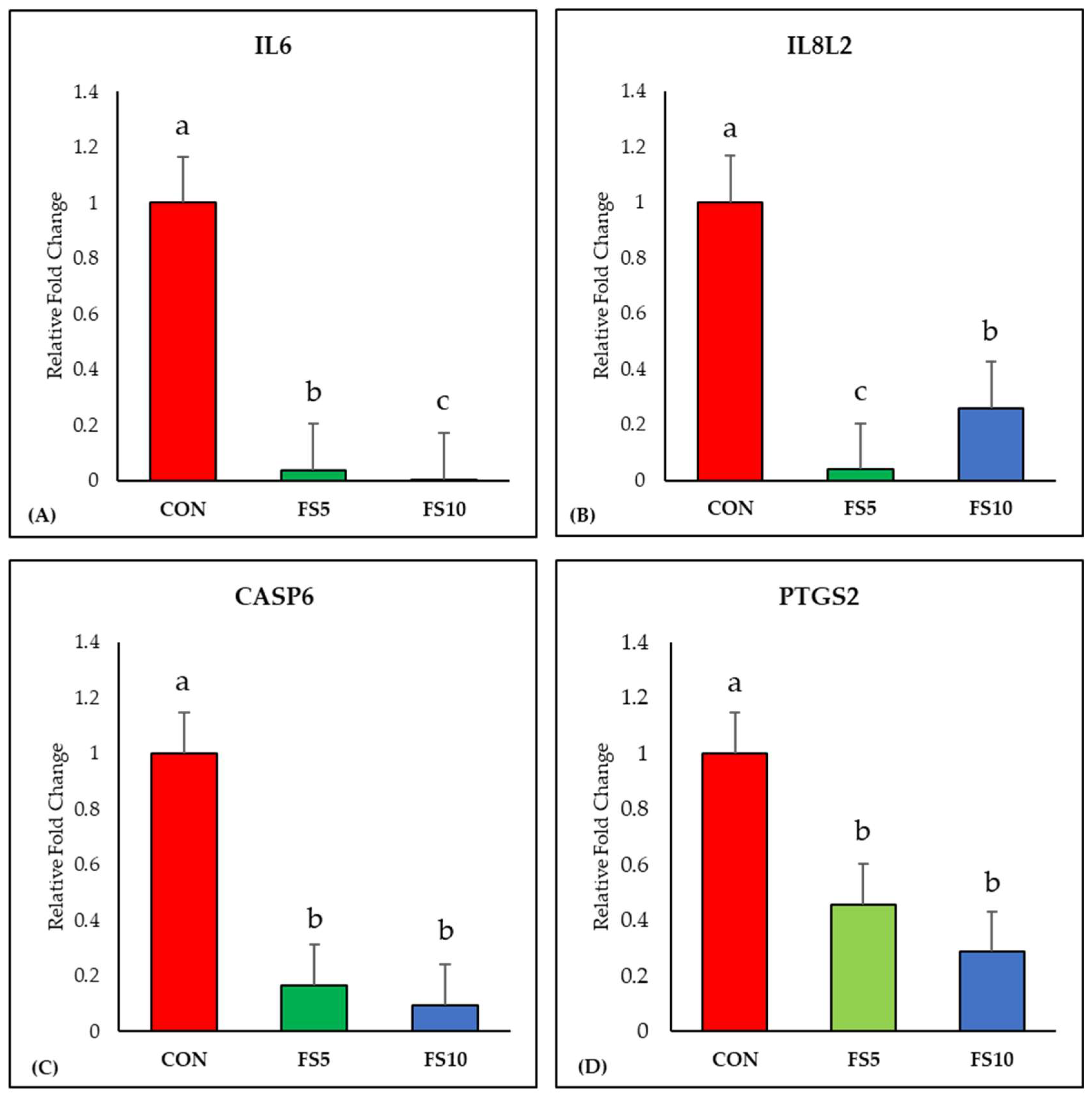
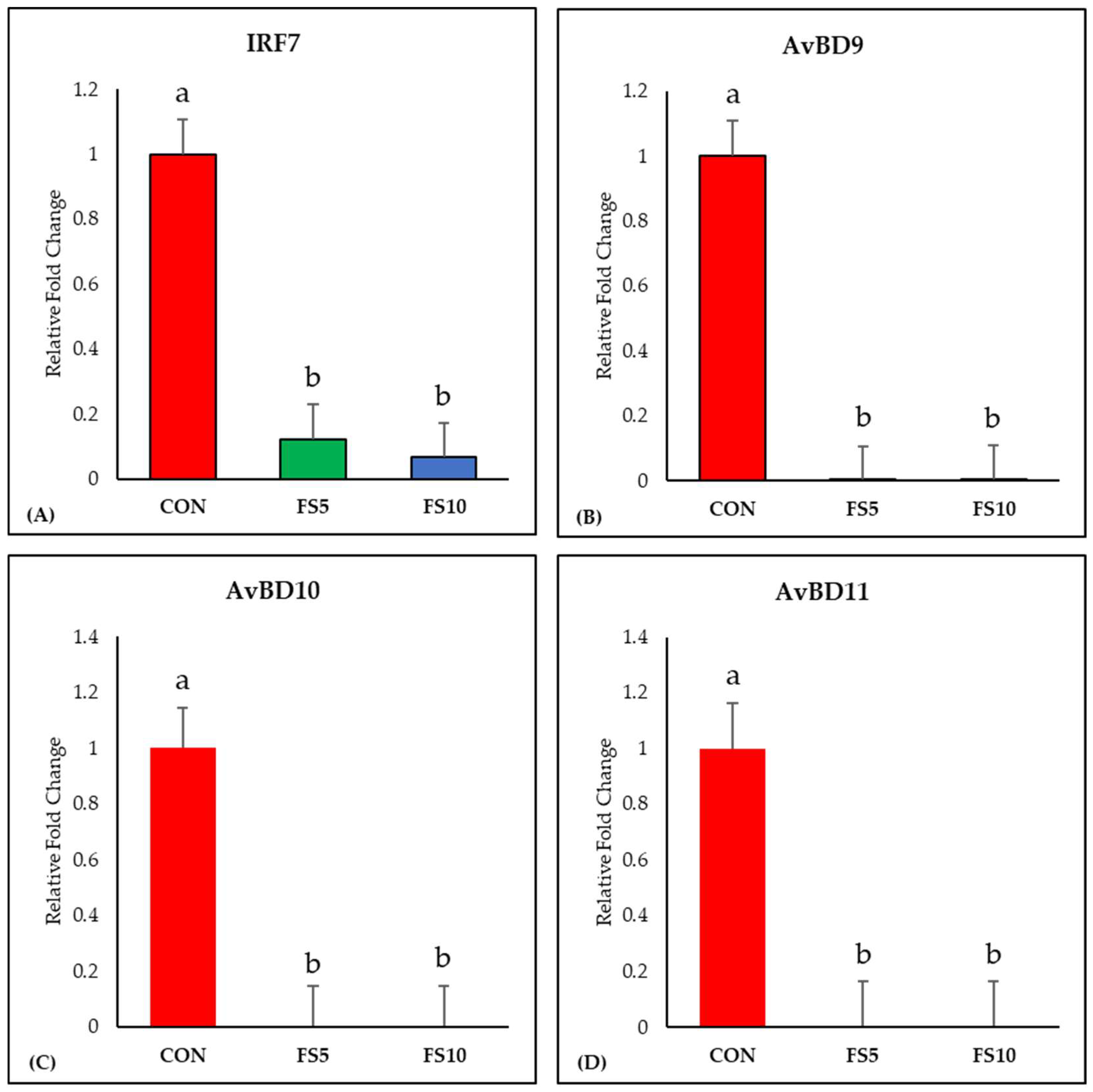
3.1. Relative Expression of Immune-Related Genes
3.1.1. Inflammation-Related Genes
3.1.2. IRF7 and Avian Defensins
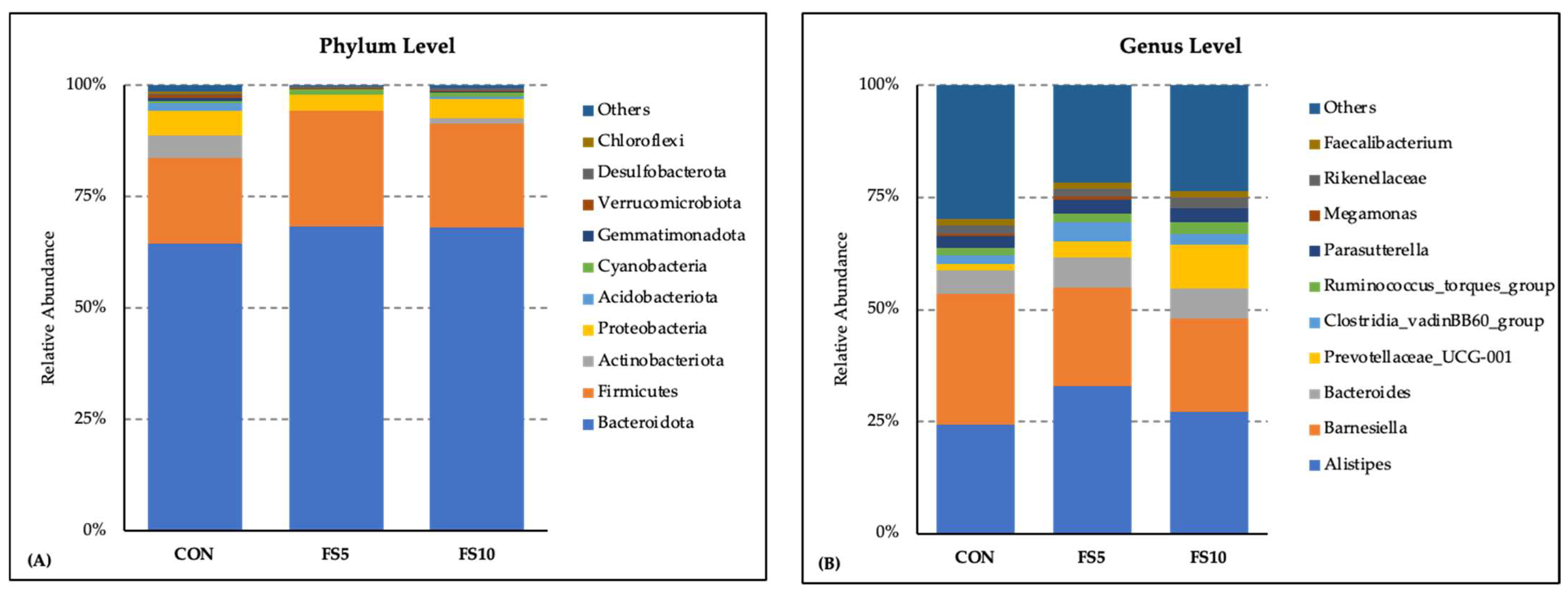
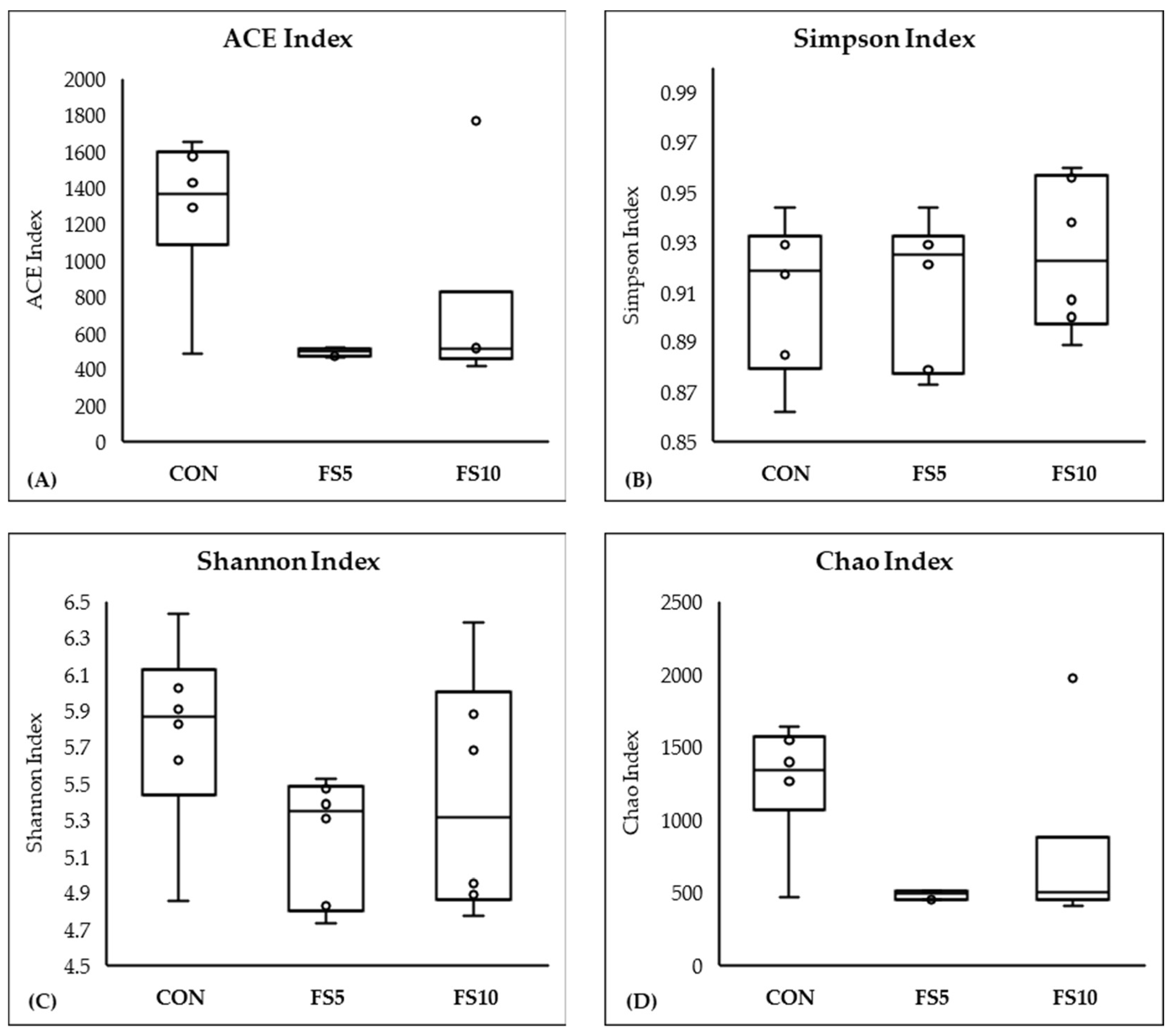
3.2. Composition and Biodiversity of the Cecal Microbiota
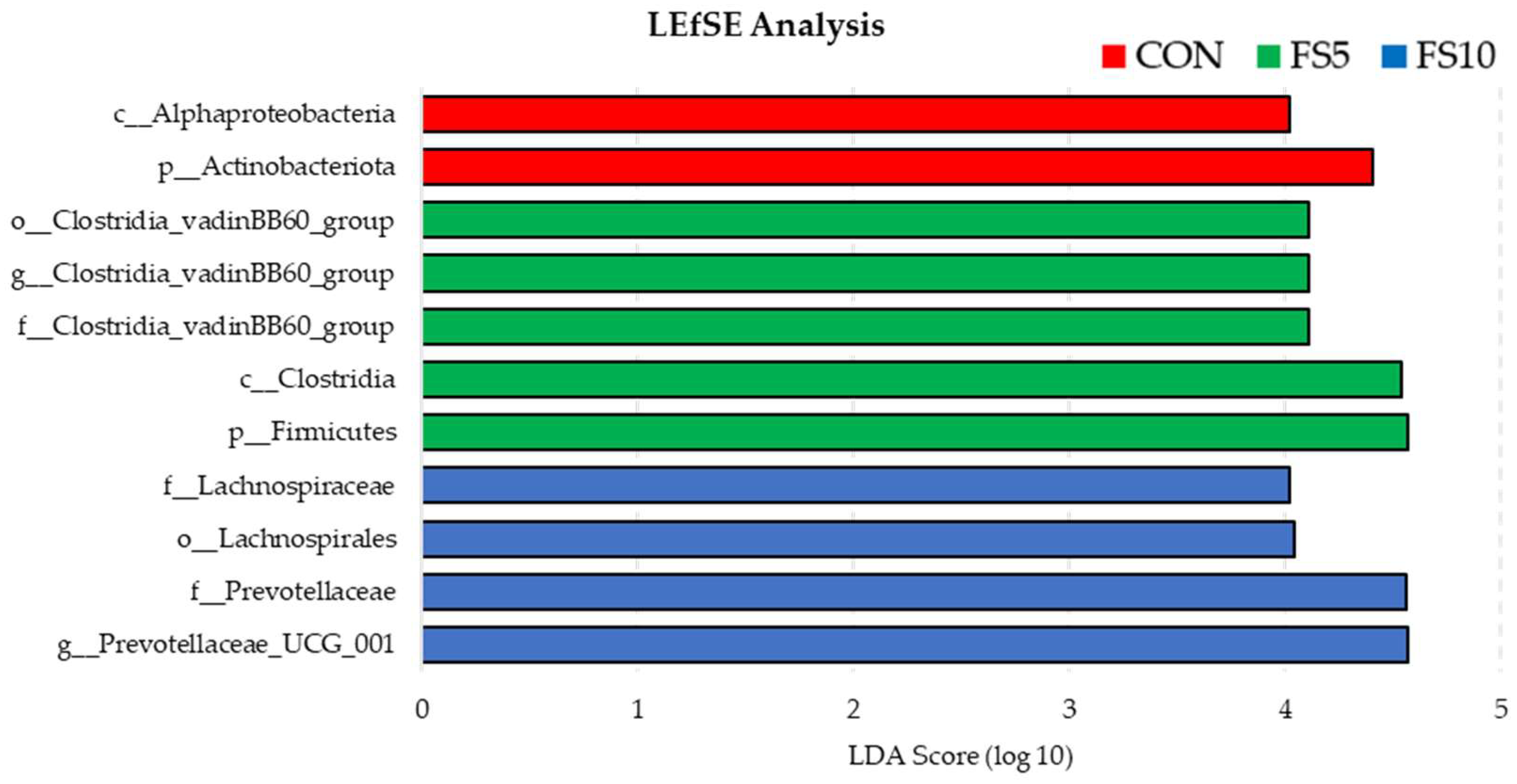
3.2.1. Microbial Enrichments
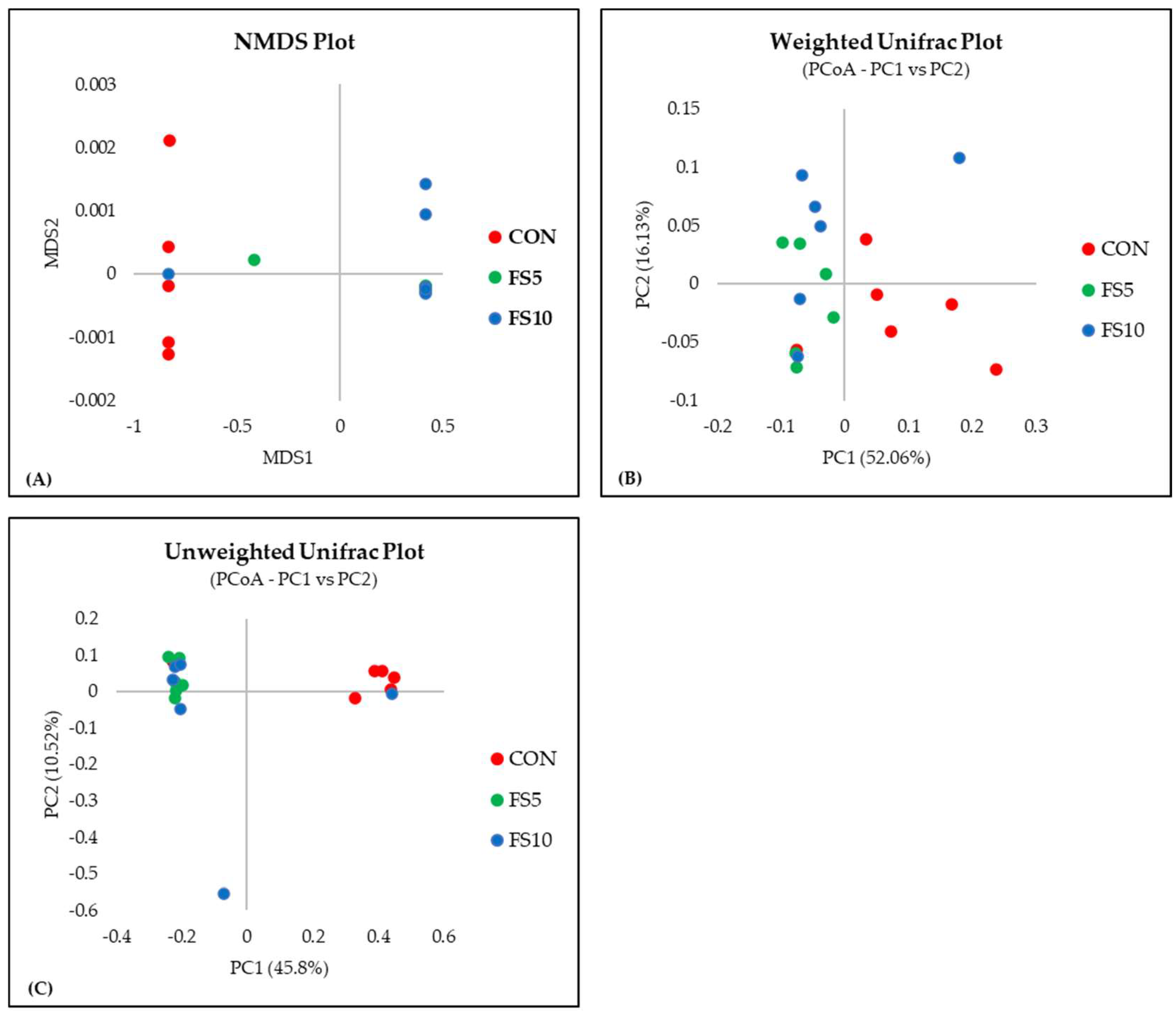
3.2.2. Alpha and Beta Diversity
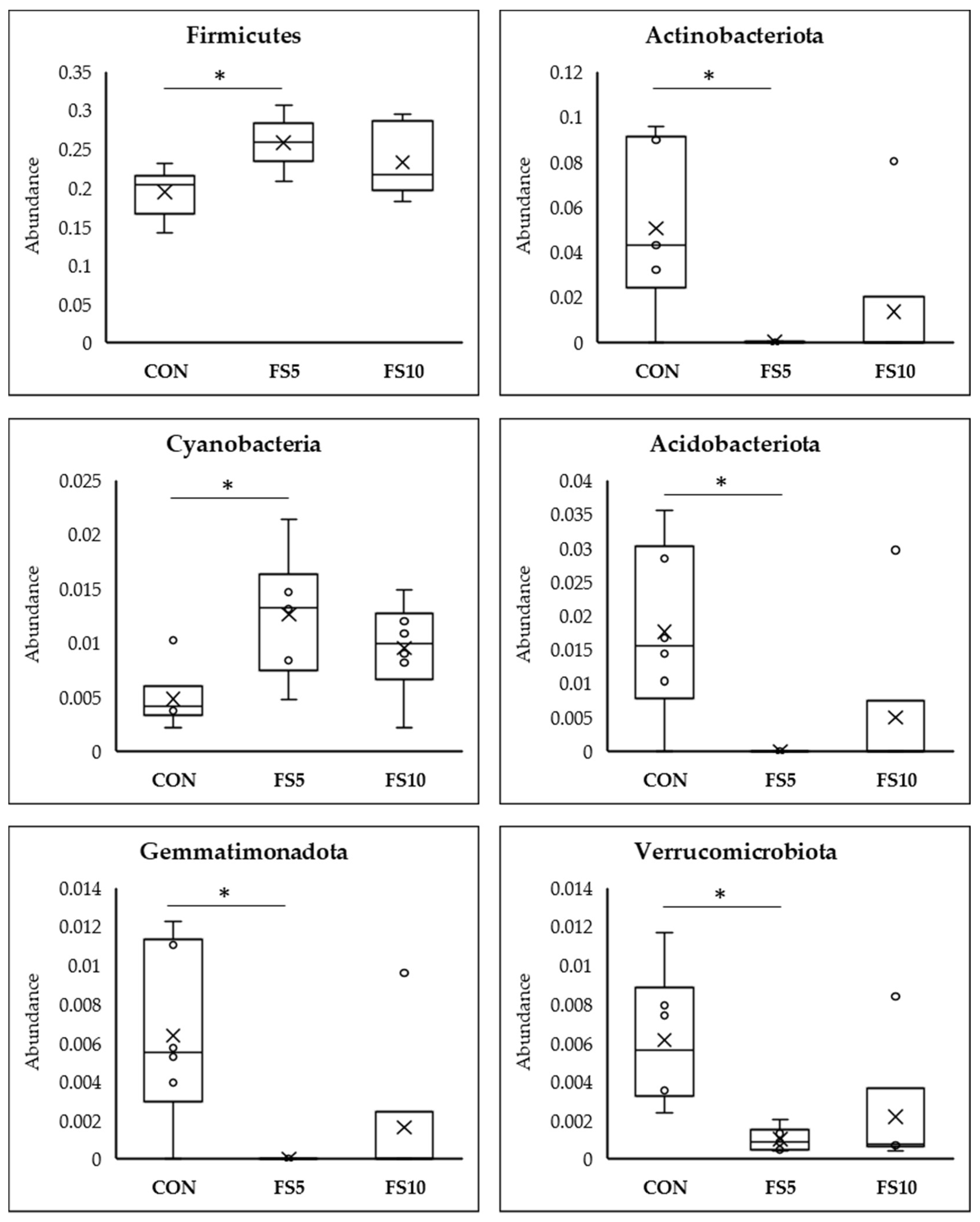
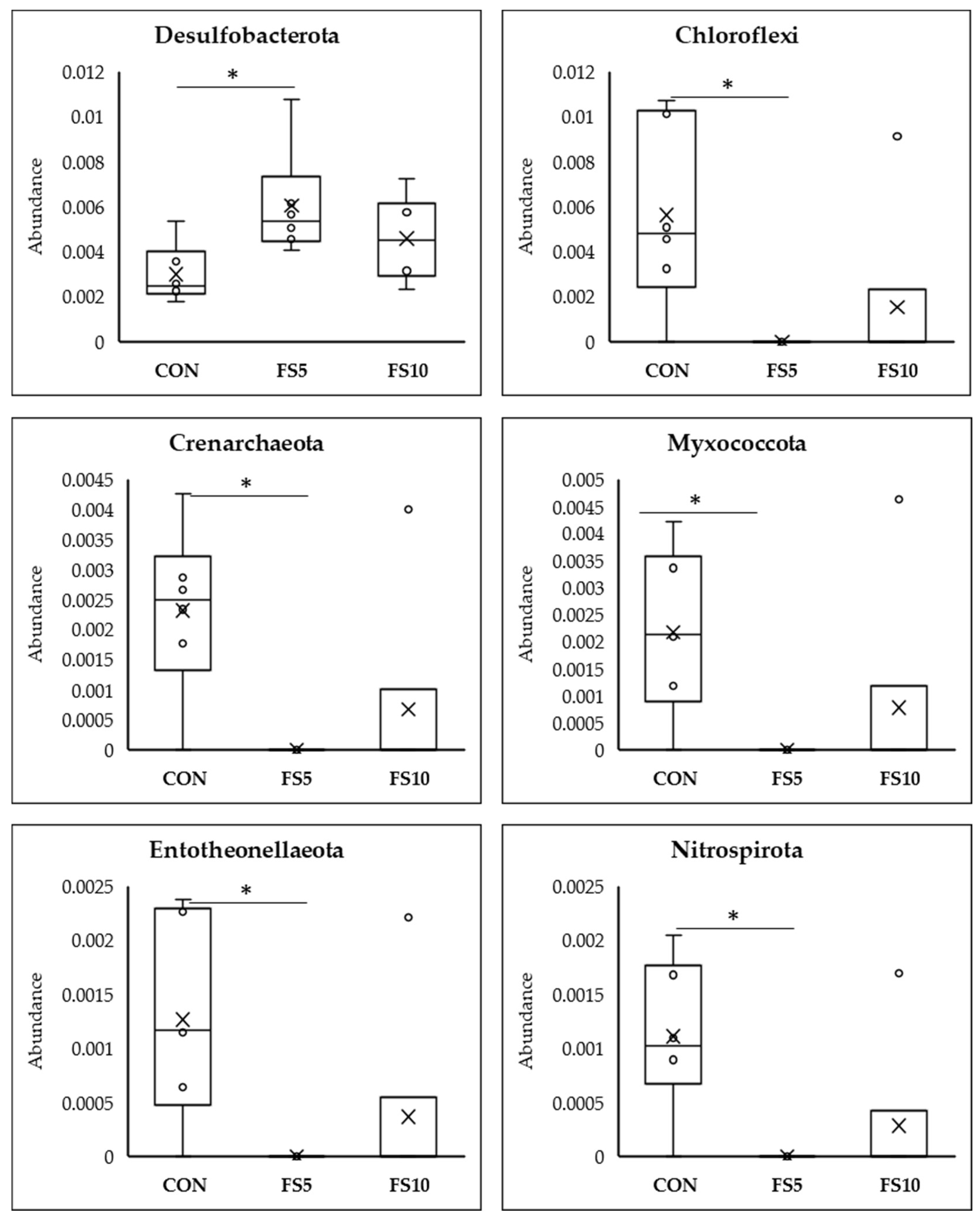
3.2.3. Differential Abundance Analysis Using Meta Stat
3.2.4. Characterization of Unique Microbiota
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mottet, A.; Tempio, G. Global Poultry Production: Current State and Future Outlook and Challenges. Worlds Poult. Sci. J. 2017, 73, 245–256. [Google Scholar] [CrossRef]
- Shuaib, M.; Paneru, D.; Hafeez, A.; Tahir, M.; Kim, W.K. The Chemical Composition of Soyhulls and Their Effect on Amino Acid and Nutrient Digestibility in Laying Hens during the Peak of Production. Animals 2023, 13, 2808. [Google Scholar] [CrossRef] [PubMed]
- Suresh, G.; Das, R.K.; Kaur Brar, S.; Rouissi, T.; Avalos Ramirez, A.; Chorfi, Y.; Godbout, S. Alternatives to Antibiotics in Poultry Feed: Molecular Perspectives. Crit. Rev. Microbiol. 2018, 44, 318–335. [Google Scholar] [CrossRef]
- Goetting, V.; Lee, K.A.; Tell, L.A. Pharmacokinetics of Veterinary Drugs in Laying Hens and Residues in Eggs: A Review of the Literature: Drug Residues in Eggs. J. Vet. Pharmacol. Ther. 2011, 34, 521–556. [Google Scholar] [CrossRef] [PubMed]
- Mund, M.D.; Khan, U.H.; Tahir, U.; Mustafa, B.-E.; Fayyaz, A. Antimicrobial Drug Residues in Poultry Products and Implications on Public Health: A Review. Int. J. Food Prop. 2017, 20, 1433–1446. [Google Scholar] [CrossRef]
- Gadde, U.; Oh, S.T.; Lee, Y.S.; Davis, E.; Zimmerman, N.; Rehberger, T.; Lillehoj, H.S. The Effects of Direct-Fed Microbial Supplementation, as an Alternative to Antibiotics, on Growth Performance, Intestinal Immune Status, and Epithelial Barrier Gene Expression in Broiler Chickens. Probiotics Antimicrob. Proteins 2017, 9, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Roberts, K.T. The Potential of Fenugreek (Trigonella foenum-graecum) as a Functional Food and Nutraceutical and Its Effects on Glycemia and Lipidemia. J. Med. Food 2011, 14, 1485–1489. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.; Rajiv, J.; Mahadevamma; Naidu, M.M.; Puranaik, J.; Srinivas, P. Effect of Fenugreek Seed Husk on the Rheology and Quality Characteristics of Muffins. Food Nutr. Sci. 2012, 03, 1473–1479. [Google Scholar] [CrossRef]
- Madhava Naidu, M.; Shyamala, B.N.; Pura Naik, J.; Sulochanamma, G.; Srinivas, P. Chemical Composition and Antioxidant Activity of the Husk and Endosperm of Fenugreek Seeds. LWT Food Sci. Technol. 2011, 44, 451–456. [Google Scholar] [CrossRef]
- Meghwal, M. A Review on the Functional Properties, Nutritional Content, Medicinal Utilization and Potential Application of Fenugreek. J. Food Process. Technol. 2012, 3, 181. [Google Scholar] [CrossRef]
- Basch, E.; Ulbricht, C.; Kuo, G.; Szapary, P.; Smith, M. Therapeutic Applications of Fenugreek. Altern. Med. Rev. 2003, 8, 20–27. [Google Scholar]
- Ahmad, A.; Alghamdi, S.S.; Mahmood, K.; Afzal, M. Fenugreek a Multipurpose Crop: Potentialities and Improvements. Saudi J. Biol. Sci. 2016, 23, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Wani, S.A.; Kumar, P. Fenugreek: A Review on Its Nutraceutical Properties and Utilization in Various Food Products. J. Saudi Soc. Agric. Sci. 2018, 17, 97–106. [Google Scholar] [CrossRef]
- Platel, K.; Srinivasan, K. Influence of Dietary Spices or Their Active Principles on Digestive Enzymes of Small Intestinal Mucosa in Rats. Int. J. Food Sci. Nutr. 1996, 47, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Al Hakeem, W.G.; Acevedo Villanueva, K.Y.; Selvaraj, R.K. The Development of Gut Microbiota and Its Changes Following C. Jejuni Infection in Broilers. Vaccines 2023, 11, 595. [Google Scholar] [CrossRef] [PubMed]
- Stadnyk, A.W. Cytokine Production by Epithelial Cells. FASEB J. 1994, 8, 1041–1047. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, T.; Akira, S.; Narazaki, M.; Taga, T. Interleukin-6 Family of Cytokines and Gp130. Blood 1995, 86, 1243–1254. [Google Scholar] [CrossRef] [PubMed]
- Gessner, D.K.; Ringseis, R.; Eder, K. Potential of Plant Polyphenols to Combat Oxidative Stress and Inflammatory Processes in Farm Animals. Anim. Physiol. Nutr. 2017, 101, 605–628. [Google Scholar] [CrossRef] [PubMed]
- Chamorro, S.; Romero, C.; Brenes, A.; Sánchez-Patán, F.; Bartolomé, B.; Viveros, A.; Arija, I. Impact of a Sustained Consumption of Grape Extract on Digestion, Gut Microbial Metabolism and Intestinal Barrier in Broiler Chickens. Food Funct. 2019, 10, 1444–1454. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Zeng, X.; Liu, J.; Yan, F.; Xiang, Z.; Wang, Y.; Tao, F.; Yang, C. Change of Serum Metabolome and Cecal Microflora in Broiler Chickens Supplemented With Grape Seed Extracts. Front. Immunol. 2020, 11, 610934. [Google Scholar] [CrossRef]
- Yang, L.; Liu, S.; Ding, J.; Dai, R.; He, C.; Xu, K.; Honaker, C.F.; Zhang, Y.; Siegel, P.; Meng, H. Gut Microbiota Co-Microevolution with Selection for Host Humoral Immunity. Front. Microbiol. 2017, 8, 1243. [Google Scholar] [CrossRef] [PubMed]
- Bruce-Keller, A.J.; Richard, A.J.; Fernandez-Kim, S.-O.; Ribnicky, D.M.; Salbaum, J.M.; Newman, S.; Carmouche, R.; Stephens, J.M. Fenugreek Counters the Effects of High Fat Diet on Gut Microbiota in Mice: Links to Metabolic Benefit. Sci. Rep. 2020, 10, 1245. [Google Scholar] [CrossRef]
- Abdel-Wareth, A.A.A.; Elkhateeb, F.S.O.; Ismail, Z.S.H.; Ghazalah, A.A.; Lohakare, J. Combined Effects of Fenugreek Seeds and Probiotics on Growth Performance, Nutrient Digestibility, Carcass Criteria, and Serum Hormones in Growing Rabbits. Livest. Sci. 2021, 251, 104616. [Google Scholar] [CrossRef]
- Paneru, D.; Tellez-Isaias, G.; Romano, N.; Lohakare, G.; Bottje, W.G.; Lohakare, J. Effect of Graded Levels of Fenugreek (Trigonella foenum-graecum L.) Seeds on the Growth Performance, Hematological Parameters, and Intestinal Histomorphology of Broiler Chickens. Vet. Sci. 2022, 9, 207. [Google Scholar] [CrossRef] [PubMed]
- Laptev, G.Y.; Filippova, V.A. Examination of the Expression of Immunity Genes and Bacterial Profiles in the Caecum of Growing Chickens Infected with Salmonella Enteritidis and Fed a Phytobiotic. Animals 2019, 9, 615. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, P.; Rothwell, L.; Galyov, E.E.; Barrow, P.A.; Burnside, J.; Wigley, P. Differential Cytokine Expression in Avian Cells in Response to Invasion by Salmonella Typhimurium, Salmonella Enteritidis and Salmonella Gallinarum The GenBank Accession Numbers for the Sequences Reported in This Paper Are AI982185 for Chicken IL-6 cDNA and AJ250838 for the Partial Chicken IL-6 Genomic Sequence, Respectively. Microbiology 2000, 146, 3217–3226. [Google Scholar] [CrossRef] [PubMed]
- Wigley, P.; Kaiser, P. Avian Cytokines in Health and Disease. Braz. J. Poult. Sci. 2003, 5, 1–14. [Google Scholar] [CrossRef]
- Zasloff, M. Antimicrobial Peptides of Multicellular Organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Steinstraesser, L.; Kraneburg, U.; Jacobsen, F.; Al-Benna, S. Host Defense Peptides and Their Antimicrobial-Immunomodulatory Duality. Immunobiology 2011, 216, 322–333. [Google Scholar] [CrossRef] [PubMed]
- van Dijk, A.; Veldhuizen, E.J.; Haagsman, H.P. Avian Defensins. Vet. Immunol. Immunopathol. 2008, 124, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Mowbray, C.A.; Niranji, S.S.; Cadwell, K.; Bailey, R.; Watson, K.A.; Hall, J. Gene Expression of AvBD6-10 in Broiler Chickens Is Independent of AvBD6, 9, and 10 Peptide Potency. Vet. Immunol. Immunopathol. 2018, 202, 31–40. [Google Scholar] [CrossRef]
- Zheng, M.; Karki, R.; Vogel, P.; Kanneganti, T.-D. Caspase-6 Is a Key Regulator of Innate Immunity, Inflammasome Activation, and Host Defense. Cell 2020, 181, 674–687.e13. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Tansey, M.G. The Role of Neuroinflammation in Parkinson’s Disease. In Neuroinflammation; Elsevier: Amsterdam, The Netherlands, 2011; pp. 403–421. ISBN 978-0-12-384913-7. [Google Scholar]
- Chen, S.; Wang, T.; Liu, P.; Yang, C.; Wang, M.; Jia, R.; Zhu, D.; Liu, M.; Yang, Q.; Wu, Y.; et al. Duck Interferon Regulatory Factor 7 (IRF7) Can Control Duck Tembusu Virus (DTMUV) Infection by Triggering Type I Interferon Production and Its Signal Transduction Pathway. Cytokine 2019, 113, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Wang, X.; Yang, L.; He, W.; Meng, T.; Zheng, K.; Xia, X.; Zhou, Y.; He, J.; Liu, C.; et al. The Effects of Fenugreek Extract on Growth Performance, Serum Biochemical Indexes, Immunity and NF-κB Signaling Pathway in Broiler. Front. Vet. Sci. 2022, 9, 882754. [Google Scholar] [CrossRef] [PubMed]
- Dibner, J.; Richards, J. Antibiotic Growth Promoters in Agriculture: History and Mode of Action. Poult. Sci. 2005, 84, 634–643. [Google Scholar] [CrossRef] [PubMed]
- Apajalahti, J.; Vienola, K. Interaction between Chicken Intestinal Microbiota and Protein Digestion. Anim. Feed Sci. Technol. 2016, 221, 323–330. [Google Scholar] [CrossRef]
- Paraskeuas, V.; Mountzouris, K.C. Broiler Gut Microbiota and Expressions of Gut Barrier Genes Affected by Cereal Type and Phytogenic Inclusion. Anim. Nutr. 2019, 5, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.; Kim, H.; Kim, I. Effects of Phytogenic Feed Additive on Growth Performance, Digestibility, Blood Metabolites, Intestinal Microbiota, Meat Color and Relative Organ Weight after Oral Challenge with Clostridium Perfringens in Broilers. Livest. Sci. 2014, 160, 82–88. [Google Scholar] [CrossRef]
- Mountzouris, K.C.; Paraskevas, V.; Tsirtsikos, P.; Palamidi, I.; Steiner, T.; Schatzmayr, G.; Fegeros, K. Assessment of a Phytogenic Feed Additive Effect on Broiler Growth Performance, Nutrient Digestibility and Caecal Microflora Composition. Anim. Feed Sci. Technol. 2011, 168, 223–231. [Google Scholar] [CrossRef]
- Pathak, M.; Mandal, G.; Patra, A.; Samanta, I.; Pradhan, S.; Haldar, S. Effects of Dietary Supplementation of Cinnamaldehyde and Formic Acid on Growth Performance, Intestinal Microbiota and Immune Response in Broiler Chickens. Anim. Prod. Sci. 2016, 57, 821–827. [Google Scholar] [CrossRef]
- Hong, J.-C.; Steiner, T.; Aufy, A.; Lien, T.-F. Effects of Supplemental Essential Oil on Growth Performance, Lipid Metabolites and Immunity, Intestinal Characteristics, Microbiota and Carcass Traits in Broilers. Livest. Sci. 2012, 144, 253–262. [Google Scholar] [CrossRef]
| Gene 1 | Accession Number | Primer Sequence (5′ → 3′) |
|---|---|---|
| AvBD9 | NM_001001611.2 | F: GTCAGGCATCTTCACAGCTG |
| R: GGCTAGGACTTCTCTGTGCA | ||
| AvBD10 | NM_001001609.2 | F: CACGTCCTGTTAGCACACTG |
| R: AGCTGCATGAACCCAAAGTG | ||
| AvBD11 | NM_001001779.1 | F: CCCTCCTTCAGTTTCCCCTT |
| R: CATCTGACTCACTGCTGCAC | ||
| IL6 | NM_204628.1 | F: TGGAAGAAGCATGGAGAGCA |
| R: GCATCCGTTCCTATGTGCTG | ||
| IL8L2 | NM_205498.1 | F: CCGGATATGCAAACACTGGC |
| R: AGAATTGAGCTGAGCCCTGT | ||
| CASP6 | NM_204726.1 | F: CGTGTTCAGTTGGACAGCAA |
| R: GGAGGGTGCAAAACTGAAGG | ||
| PTGS2 | NM_001167718.1 | F: AAAGGGGCCAGTACTGTGTT |
| R: TGCCCAGACTTGTCTTCCTT | ||
| IRF7 | NM_205372.1 | F: CACATGTTCATGCTGCTGGA |
| R: CTGGAAGGAGAGCAGGTTGA | ||
| ACTB | NM_205518.1 | F: ACTCTGTCTGGATTGGAGGC |
| R: AAAGCCATGCCAATCTCGTC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paneru, D.; Tellez-Isaias, G.; Bottje, W.G.; Asiamah, E.; Abdel-Wareth, A.A.A.; Salahuddin, M.; Lohakare, J. Modulation of Immune Response and Cecal Microbiota by Dietary Fenugreek Seeds in Broilers. Vet. Sci. 2024, 11, 57. https://doi.org/10.3390/vetsci11020057
Paneru D, Tellez-Isaias G, Bottje WG, Asiamah E, Abdel-Wareth AAA, Salahuddin M, Lohakare J. Modulation of Immune Response and Cecal Microbiota by Dietary Fenugreek Seeds in Broilers. Veterinary Sciences. 2024; 11(2):57. https://doi.org/10.3390/vetsci11020057
Chicago/Turabian StylePaneru, Deependra, Guillermo Tellez-Isaias, Walter G. Bottje, Emmanuel Asiamah, Ahmed A. A. Abdel-Wareth, Md Salahuddin, and Jayant Lohakare. 2024. "Modulation of Immune Response and Cecal Microbiota by Dietary Fenugreek Seeds in Broilers" Veterinary Sciences 11, no. 2: 57. https://doi.org/10.3390/vetsci11020057
APA StylePaneru, D., Tellez-Isaias, G., Bottje, W. G., Asiamah, E., Abdel-Wareth, A. A. A., Salahuddin, M., & Lohakare, J. (2024). Modulation of Immune Response and Cecal Microbiota by Dietary Fenugreek Seeds in Broilers. Veterinary Sciences, 11(2), 57. https://doi.org/10.3390/vetsci11020057












