Can Chemotherapy Negatively Affect the Specific Antibody Response toward Core Vaccines in Canine Cancer Patients?
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Study Protocol
2.2. Sample Collection
2.3. Detection of Specific Antibodies by VacciCheck
2.4. Statistical Analysis
3. Results
3.1. Antibody Titers and Kinetics of Protection
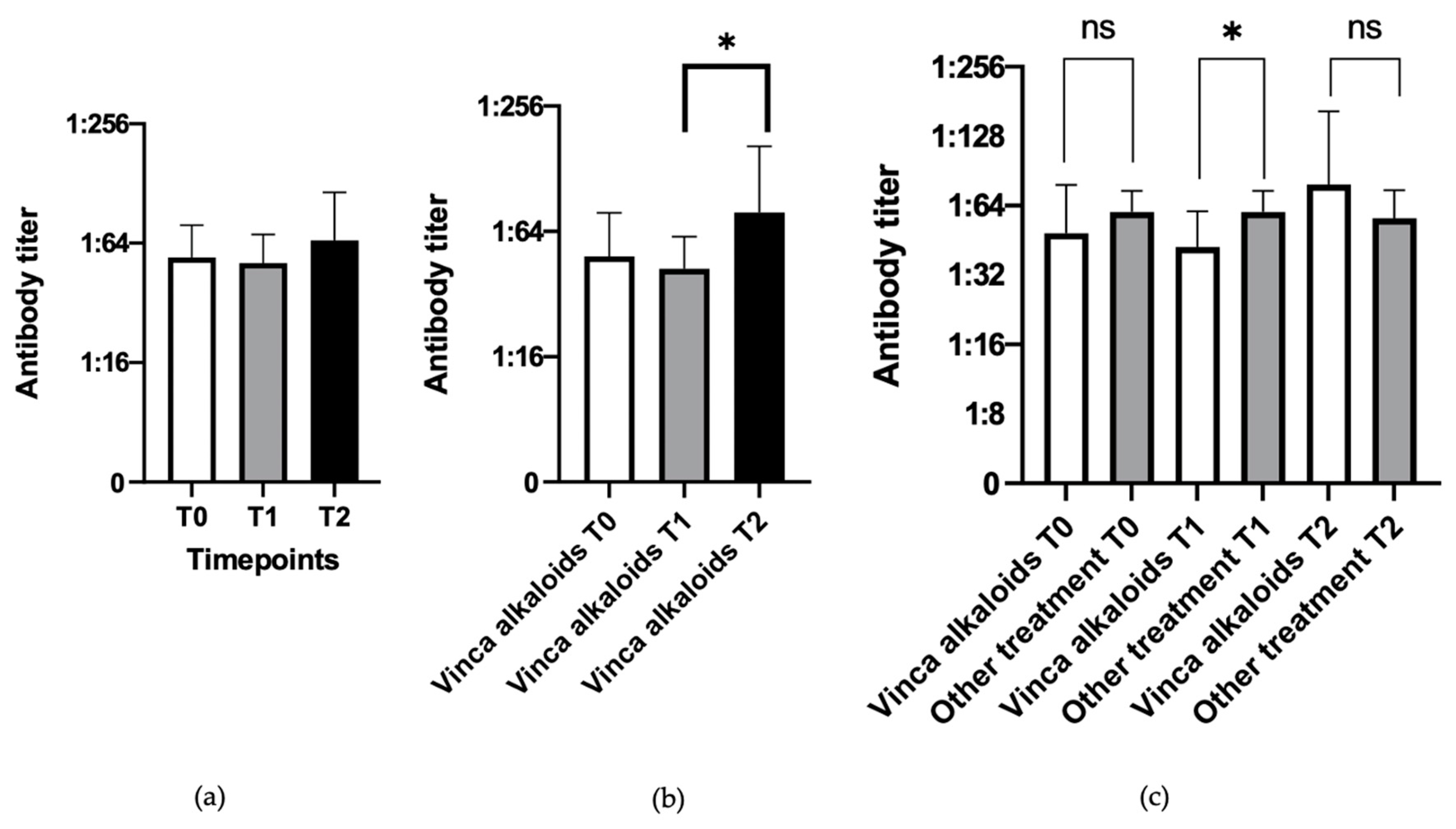
3.2. Evaluation of Influence of the Type of Chemotherapy
3.3. Evaluation of Influence of the Type of Tumor
3.4. Evaluation of Influence of the Dog Size
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paoloni, M.C.; Khanna, C. Comparative Oncology Today. Vet. Clin. N. Am. Small Anim. Pract. 2007, 37, 1023–1032. [Google Scholar] [CrossRef] [PubMed]
- Dobson, J.M. Breed-Predispositions to Cancer in Pedigree Dogs. ISRN Vet. Sci. 2013, 2013, 941275. [Google Scholar] [CrossRef]
- Gardner, H.L.; Fenger, J.M.; London, C.A. Dogs as a Model for Cancer. Annu. Rev. Anim. Biosci. 2016, 4, 199–222. [Google Scholar] [CrossRef]
- Tarone, L.; Barutello, G.; Iussich, S.; Giacobino, D.; Quaglino, E.; Buracco, P.; Cavallo, F.; Riccardo, F. Naturally Occurring Cancers in Pet Dogs as Pre-Clinical Models for Cancer Immunotherapy. Cancer Immunol. Immunother. 2019, 68, 1839–1853. [Google Scholar] [CrossRef] [PubMed]
- Dall’Ara, P. (Ed.) Vaccini e Vaccinazioni Degli Animali da Compagnia, 1st ed.; EDRA: Milano, Italy, 2020. [Google Scholar]
- Walter, C.U.; Biller, B.J.; Lana, S.E.; Bachand, A.M.; Dow, S.W. Effects of Chemotherapy on Immune Responses in Dogs with Cancer. J. Vet. Intern. Med. 2006, 20, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Elias, M.A.; Duarte, A.; Nunes, T.; Lourenço, A.M.; Braz, B.S.; Vicente, G.; Henriques, J.; Tavares, L. Influence of Chemotherapy for Lymphoma in Canine Parvovirus DNA Distribution and Specific Humoral Immunity. Comp. Immunol. Microbiol. Infect. Dis. 2014, 37, 313–320. [Google Scholar] [CrossRef]
- Henry, C.J.; McCaw, D.L.; Brock, K.V.; Stoker, A.M.; Tyler, J.W.; Tate, D.J.; Higginbotham, M.L. Association between Cancer Chemotherapy and Canine Distemper Virus, Canine Parvovirus, and Rabies Virus Antibody Titers in Tumor-Bearing Dogs. J. Am. Vet. Med. Assoc. 2001, 219, 1238–1241. [Google Scholar] [CrossRef]
- Zignol, M.; Peracchi, M.; Tridello, G.; Pillon, M.; Fregonese, F.; D’Elia, R.; Zanesco, L.; Cesaro, S. Assessment of Humoral Immunity to Poliomyelitis, Tetanus, Hepatitis B, Measles, Rubella, and Mumps in Children after Chemotherapy. Cancer 2004, 101, 635–641. [Google Scholar] [CrossRef]
- Nilsson, A.; De Milito, A.; Engstrom, P.; Nordin, M.; Narita, M.; Grillner, L.; Chiodi, F.; Bjork, O. Current Chemotherapy Protocols for Childhood Acute Lymphoblastic Leukemia Induce Loss of Humoral Immunity to Viral Vaccination Antigens. Pediatrics 2002, 109, e91. [Google Scholar] [CrossRef]
- Day, M.J.; Horzinek, M.C.; Schultz, R.D.; Squires, R.A. WSAVA Guidelines for the Vaccination of Dogs and Cats. J. Small Anim. Pract. 2016, 57, E1–E45. [Google Scholar] [CrossRef]
- Australian Veterinary Association (AVA). Vaccination of Dogs and Cats. Available online: https://www.ava.com.au/policy-advocacy/policies/companion-animals-health/vaccination-of-dogs-and-cats/ (accessed on 16 January 2023).
- British Veterinary Association (BVA). Getting Your Pet Vaccinated. Available online: https://www.bva.co.uk/media/2649/client_leaflet_9_-_getting_your_pet_vaccinated.pdf (accessed on 4 January 2023).
- Joint American Veterinary Medical Association (AVMA)-Federation of Veterinarians of Europe (FVE)-Canadian Veterinary Medical Association (CVMA) Statement on the Benefits of Animal Vaccination Programs in Advancing Animal and Human Health. Available online: https://fve.org/cms/wp-content/uploads/AVMA-CVMA-FVE_vacconation_joint-paper.docx.pdf (accessed on 18 February 2023).
- Ellis, J.; Marziani, E.; Aziz, C.; Brown, C.M.; Cohn, L.A.; Lea, C.; Moore, G.E.; Taneja, N. 2022 AAHA Canine Vaccination Guidelines. J. Am. Anim. Hosp. Assoc. 2022, 58, 213–230. [Google Scholar] [CrossRef] [PubMed]
- Hussain, K.; Khan, Y.; Ullah, Q.; Rabbani, A.H.; Naseer, O.; Raza, A.; Shahid, M.; Ali, S.; Ali, A. Canine Parvo Virus: A Review on Current Perspectives in Seroprevalence, Diagnostics and Therapeutics. Glob. Vet. 2021, 23, 113–126. [Google Scholar]
- Parrish, C.R.; Sykes, J.E. Canine Parvovirus Infections and Other Viral Enteritides. In Greene’s Infectious Diseases of the Dog and Cat; Sikes, J.E., Ed.; Elsevier: St. Louis, MO, USA, 2022; pp. 341–351. [Google Scholar]
- Decaro, N.; Buonavoglia, C. Canine Parvovirus—A Review of Epidemiological and Diagnostic Aspects, with Emphasis on Type 2c. Vet. Microbiol. 2012, 155, 1–12. [Google Scholar] [CrossRef]
- Decaro, N.; Buonavoglia, C.; Barrs, V.R. Canine Parvovirus Vaccination and Immunisation Failures: Are We Far from Disease Eradication? Vet. Microbiol. 2020, 247, 108760. [Google Scholar] [CrossRef]
- Mira, F.; Purpari, G.; Di Bella, S.; Vicari, D.; Schirò, G.; Di Marco, P.; Macaluso, G.; Battilani, M.; Guercio, A. Update on Canine Distemper Virus (CDV) Strains of Arctic-like Lineage Detected in Dogs in Italy. Vet. Ital. 2018, 54, 225–236. [Google Scholar] [PubMed]
- Sykes, J.E.; Vandevelde, M. Canine Distemper Virus Infection. In Greene’s Infectious Diseases of the Dog and Cat; Sykes, J.E., Ed.; Elsevier: St. Louis, MO, USA, 2022; pp. 271–388. [Google Scholar]
- Duque-Valencia, J.; Sarute, N.; Olarte-Castillo, X.A.; Ruíz-Sáenz, J. Evolution and Interspecies Transmission of Canine Distemper Virus—An Outlook of the Diverse Evolutionary Landscapes of a Multi-Host Virus. Viruses 2019, 11, 582. [Google Scholar] [CrossRef] [PubMed]
- Mira, F.; Puleio, R.; Schirò, G.; Condorelli, L.; Di Bella, S.; Chiaramonte, G.; Purpari, G.; Cannella, V.; Balboni, A.; Randazzo, V.; et al. Study on the Canine Adenovirus Type 1 (CAdV-1) Infection in Domestic Dogs in Southern Italy. Pathogens 2022, 11, 1254. [Google Scholar] [CrossRef]
- Decaro, N. Infectious Canine Hepatitis and Feline Adenovirus Infection. In Greene’s Infectious Diseases of the Dog and Cat; Sykes, J.E., Ed.; Elsevier: St. Louis, MO, USA, 2022; pp. 289–300. [Google Scholar]
- Eldredge, D.M.; Carlson, L.D.; Carlson, D.G.; Giffin, J.M.; Adelman, B. Comparative Age of Dogs and Humans. In Dog Owner’s Home Veterinary Handbook; Wiley Publishing, Inc.: Hoboken, NJ, USA, 2007; pp. 575–576. [Google Scholar]
- Kraus, C.; Pavard, S.; Promislow, D.E.L. The Size–Life Span Trade-Off Decomposed: Why Large Dogs Die Young. Am. Nat. 2013, 181, 492–505. [Google Scholar] [CrossRef]
- Meazzi, S.; Filipe, J.; Fiore, A.; Di Bella, S.; Mira, F.; Dall’Ara, P. Agreement between In-Clinics and Virus Neutralization Tests in Detecting Antibodies against Canine Distemper Virus (CDV). Viruses 2022, 14, 517. [Google Scholar] [CrossRef]
- Day, M.J. Small Animal Vaccination: A Practical Guide for Vets in the UK. Practice 2017, 39, 110–118. [Google Scholar] [CrossRef]
- Waner, T.; Mazar, S.; Keren-Kornblatt, E. Application of a Dot Enzyme-Linked Immunosorbent Assay for Evaluation of the Immune Status to Canine Parvovirus and Distemper Virus in Adult Dogs before Revaccination. J. Vet. Diagn. Investig. 2006, 18, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Salomon, K.; de Lange, T.; Calis, A.; Radier, O.; Krosse, J. In-Clinic Canine IgG Antinbody Titer Test Comparative Study: Result from Five Clinics. Isr. J. Vet. Med. 2022, 77, 7–14. [Google Scholar]
- Dall’Ara, P. (Ed.) Come Controllare La Protezione Verso i Vaccini Core. In Vaccini e Vaccinazioni Degli Animali da Compagnia; EDRA: Milano, Italy, 2020; pp. 189–201. [Google Scholar]
- Dall’Ara, P.; Lauzi, S.; Zambarbieri, J.; Servida, F.; Barbieri, L.; Rosenthal, R.; Turin, L.; Scarparo, E.; Filipe, J. Prevalence of Serum Antibody Titers against Core Vaccine Antigens in Italian Dogs. Life 2023, 13, 587. [Google Scholar] [CrossRef] [PubMed]
- Killey, R.; Mynors, C.; Pearce, R.; Nell, A.; Prentis, A.; Day, M.J. Long-Lived Immunity to Canine Core Vaccine Antigens in UK Dogs as Assessed by an in-Practice Test Kit. J. Small Anim. Pract. 2018, 59, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Gameiro, S.R.; Caballero, J.A.; Higgins, J.P.; Apelian, D.; Hodge, J.W. Exploitation of Differential Homeostatic Proliferation of T-Cell Subsets Following Chemotherapy to Enhance the Efficacy of Vaccine-Mediated Antitumor Responses. Cancer Immunol. Immunother. 2011, 60, 1227–1242. [Google Scholar] [CrossRef]
- Twark, L.; Dodds, W.J. Clinical Use of Serum Parvovirus and Distemper Virus Antibody Titers for Determining Revaccination Strategies in Healthy Dogs. J. Am. Vet. Med. Assoc. 2000, 217, 1021–1024. [Google Scholar] [CrossRef] [PubMed]
- Biogal Galed Labs. Acs. Ltd. VacciCheck® Titer Testing. Available online: https://www.biogal.com/products/vaccicheck/ (accessed on 4 February 2023).


| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Patient has a malignant neoplasm | Patient has a benign neoplasm or another concomitant pathology |
| Patient has to be treated with dose-intensive chemotherapy | Patient has a malignant neoplasm, but chemotherapy was not performed Chemoimmunotherapy |
| One blood sample was taken before beginning antiblastic chemotherapy | No blood sample was taken before antiblastic chemotherapy |
| Owner’s compliance | Owner’s noncompliance |
| Patient was vaccinated against CPV-2, CDV, and CAdV-1 | Patient was not vaccinated against CPV-2, CDV, and CAdV-1 |
| Patient undergoing at least two consecutive antiblastic chemotherapy treatments | Canine patient died before the end of antiblastic chemotherapy protocol |
| ID | Breed | Size | Sex | Age Category | Type of Tumor | Type of Chemotherapy | N° of Samples | Days from the Last Vaccination |
|---|---|---|---|---|---|---|---|---|
| 1 | Doberman Pinscher | Medium | NF | Adult | HS | L | 7 | 263 |
| 2 | Newfoundland Dog | Giant | IM | Senior | Lm | D | 8 | 257 |
| 3 | Crossbred | Medium | IF | Geriatric | TCUB | C | 8 | 557 |
| 4 | Pitbull | Medium | IM | Senior | UTN | C | 4 | 160 |
| 5 | Cocker Spaniel | Medium | NF | Senior | H | D | 7 | 231 |
| 6 | Border Collie | Medium | NF | Geriatric | Ly C | L | 8 | 86 |
| 7 | Crossbred | Small | NF | Adult | Ly B | CHOP | 18 | 271 |
| 8 | Beagle | Medium | NM | Senior | Ly B | CHOP | 10 | 270 |
| 9 | American Staffordshire | Medium | NF | Senior | Ly B | CHOP | 3 | 860 |
| 10 | Bull Terrier | Medium | NF | Adult | Ly B | CHOP | 10 | 120 |
| 11 | Border Collie | Large | IM | Geriatric | BC | TC | 7 | 663 |
| 12 | Bernese Mountain Dog | Giant | IM | Senior | HS | L | 13 | 257 |
| 13 | Greyhound | Medium | NF | Senior | Ly T | L | 9 | 323 |
| 14 | Bobtail | Large | NF | Geriatric | URN | TC | 6 | 434 |
| 15 | Crossbred | Large | NF | Senior | Ly T | LOPP | 14 | 109 |
| 16 | Golden Retriever | Large | NF | Geriatric | Ly B | CHOP | 9 | 209 |
| 17 | Rottweiler | Large | NF | Senior | Ly B | CHOP | 17 | 93 |
| 18 | Crossbred | Large | NF | Geriatric | M | V | 6 | 414 |
| 19 | Belgian Shepherd | Large | NF | Adult | M | V | 9 | 411 |
| 20 | Crossbred | Small | IM | Senior | Ly T | L | 6 | 341 |
| 21 | French Bouledogue | Medium | NM | Senior | M | V | 8 | 423 |
| CPV-2 | CDV | CAdV-1 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Threshold Value: 1:80 | Threshold Value: 1:32 | Threshold Value: 1:16 | |||||||
| Dog N. | T0 | T1 | T2 | T0 | T1 | T2 | T0 | T1 | T2 |
| 1 | 160 | 160 | 80 | 64 | 64 | 32 | 32 | 32 | 4 |
| 2 | 320 | 160 | 160 | 32 | 64 | 32 | 32 | 16 | 32 |
| 3 | 160 | 160 | 160 | 64 | 64 | 64 | 32 | 32 | 32 |
| 4 | 320 | 160 | 160 | 64 | 64 | 64 | 16 | 8 | 8 |
| 5 | 160 | 160 | 160 | 64 | 64 | 64 | 32 | 32 | 32 |
| 6 | 160 | 40 | 80 | 64 | 32 | 64 | 32 | 8 | 16 |
| 7 | 160 | 40 | 160 | 64 | 64 | 128 | 32 | 16 | 32 |
| 8 | 40 | 40 | 20 | 32 | 32 | 32 | 4 | 8 | 4 |
| 9 | 40 | 40 | 40 | 64 | 32 | 64 | 16 | 16 | 16 |
| 10 | 160 | 20 | 160 | 64 | 32 | 64 | 16 | 16 | 16 |
| 11 | 320 | 160 | 160 | 64 | 64 | 64 | 32 | 32 | 32 |
| 12 | 320 | 320 | 320 | 64 | 64 | 64 | 32 | 16 | 32 |
| 13 | 320 | 320 | 160 | 64 | 64 | 64 | 32 | 32 | 16 |
| 14 | 160 | 80 | 160 | 64 | 64 | 64 | 32 | 16 | 32 |
| 15 | 160 | 160 | 160 | 64 | 64 | 128 | 64 | 64 | 32 |
| 16 | 160 | 160 | 160 | 32 | 32 | 32 | 32 | 16 | 16 |
| 17 | 320 | 320 | 320 | 64 | 64 | 128 | 64 | 64 | 64 |
| 18 | 160 | 80 | 80 | 64 | 32 | 128 | 32 | 32 | 32 |
| 19 | 320 | 160 | 160 | 64 | 64 | 256 | 32 | 32 | 32 |
| 20 | 320 | 160 | 160 | 64 | 64 | 64 | 32 | 32 | 32 |
| 21 | 160 | 160 | 160 | 16 | 32 | 32 | 32 | 16 | 16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dall’Ara, P.; Filipe, J.; Pilastro, C.; Turin, L.; Lauzi, S.; Gariboldi, E.M.; Stefanello, D. Can Chemotherapy Negatively Affect the Specific Antibody Response toward Core Vaccines in Canine Cancer Patients? Vet. Sci. 2023, 10, 303. https://doi.org/10.3390/vetsci10040303
Dall’Ara P, Filipe J, Pilastro C, Turin L, Lauzi S, Gariboldi EM, Stefanello D. Can Chemotherapy Negatively Affect the Specific Antibody Response toward Core Vaccines in Canine Cancer Patients? Veterinary Sciences. 2023; 10(4):303. https://doi.org/10.3390/vetsci10040303
Chicago/Turabian StyleDall’Ara, Paola, Joel Filipe, Chiara Pilastro, Lauretta Turin, Stefania Lauzi, Elisa Maria Gariboldi, and Damiano Stefanello. 2023. "Can Chemotherapy Negatively Affect the Specific Antibody Response toward Core Vaccines in Canine Cancer Patients?" Veterinary Sciences 10, no. 4: 303. https://doi.org/10.3390/vetsci10040303
APA StyleDall’Ara, P., Filipe, J., Pilastro, C., Turin, L., Lauzi, S., Gariboldi, E. M., & Stefanello, D. (2023). Can Chemotherapy Negatively Affect the Specific Antibody Response toward Core Vaccines in Canine Cancer Patients? Veterinary Sciences, 10(4), 303. https://doi.org/10.3390/vetsci10040303








